The Extremely High Adsorption Capacity of Fluoride by Chicken Bone Char (CBC) in Defluoridation of Drinking Water in Relation to Its Finer Particle Size for Better Human Health
Abstract
1. Introduction
2. Materials and Methods
2.1. Preparation of Chicken Bone Char (CBC)
2.2. Equilibrium Sorption Isotherms
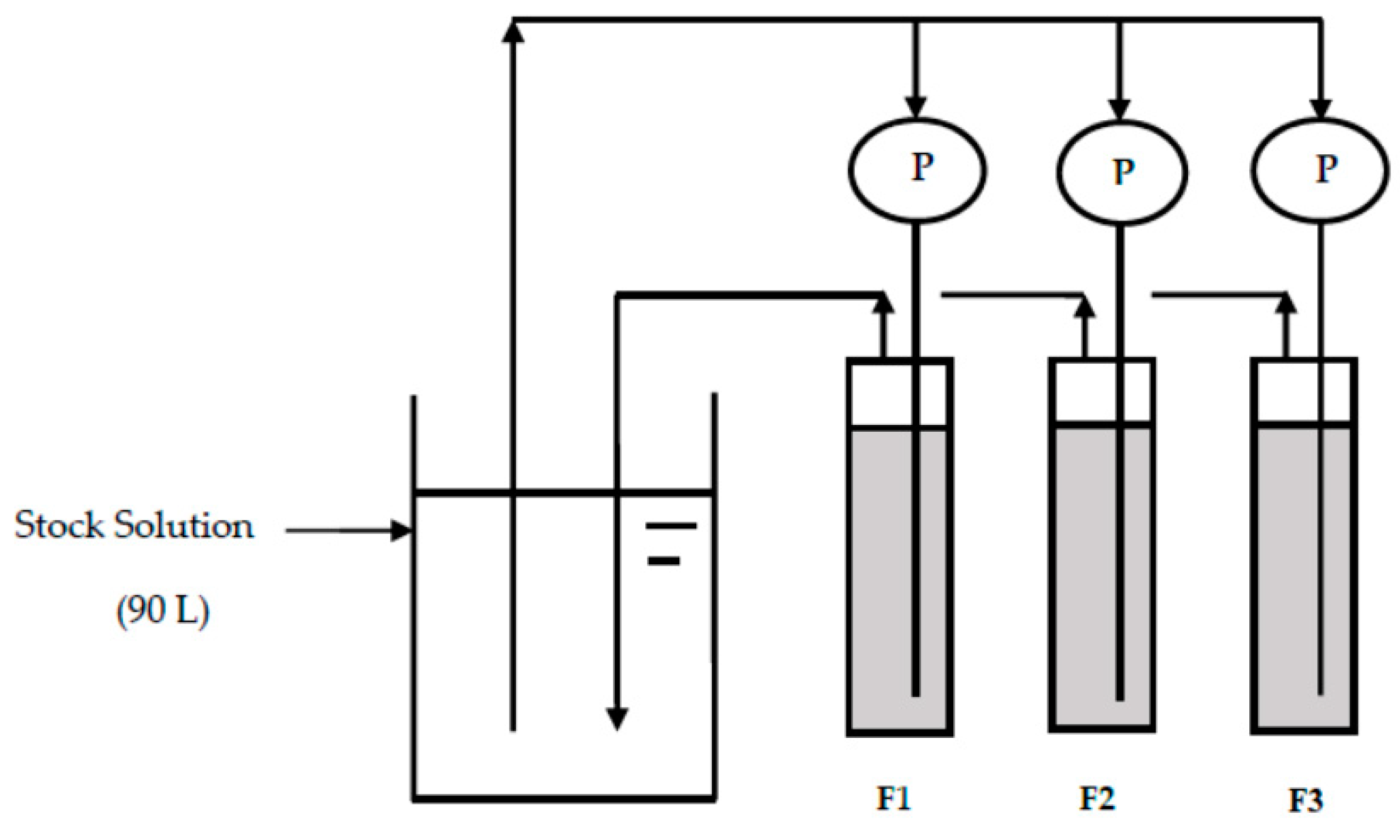
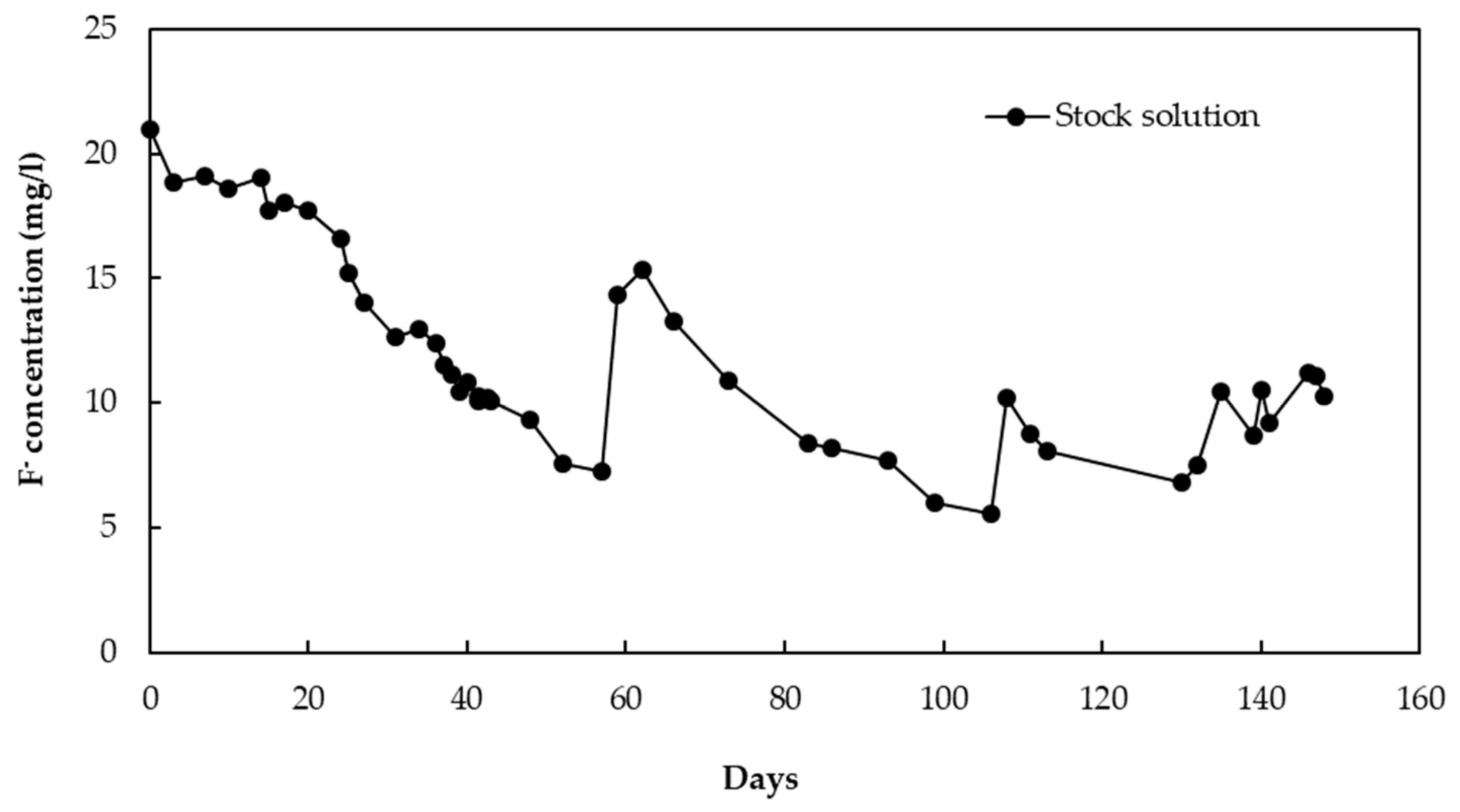
2.3. Fluoride Removal by CBC Column Experiment
3. Results and Discussion
3.1. Selecting the Best Carbonizing Temperature for the Preparation of CBC
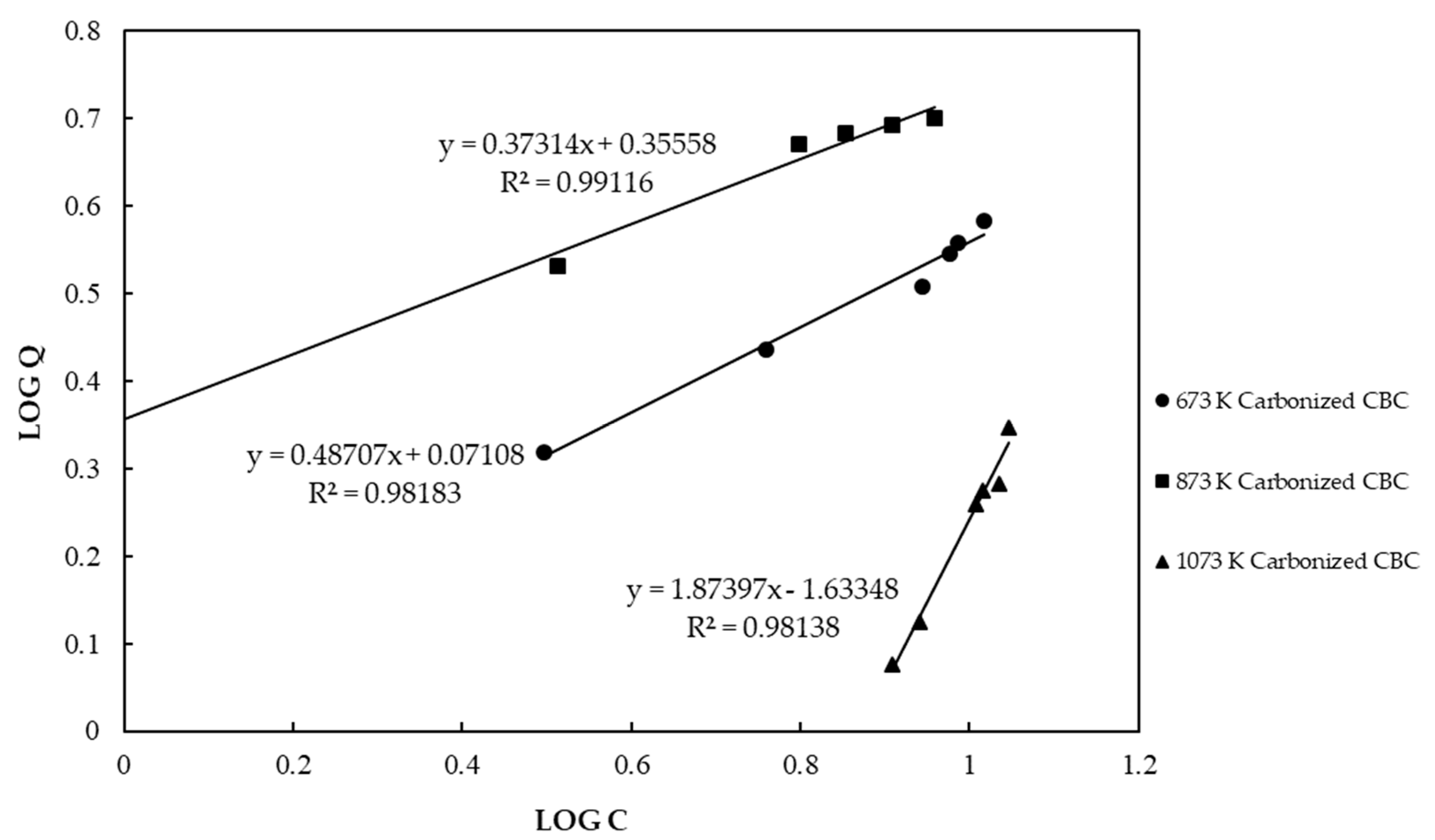
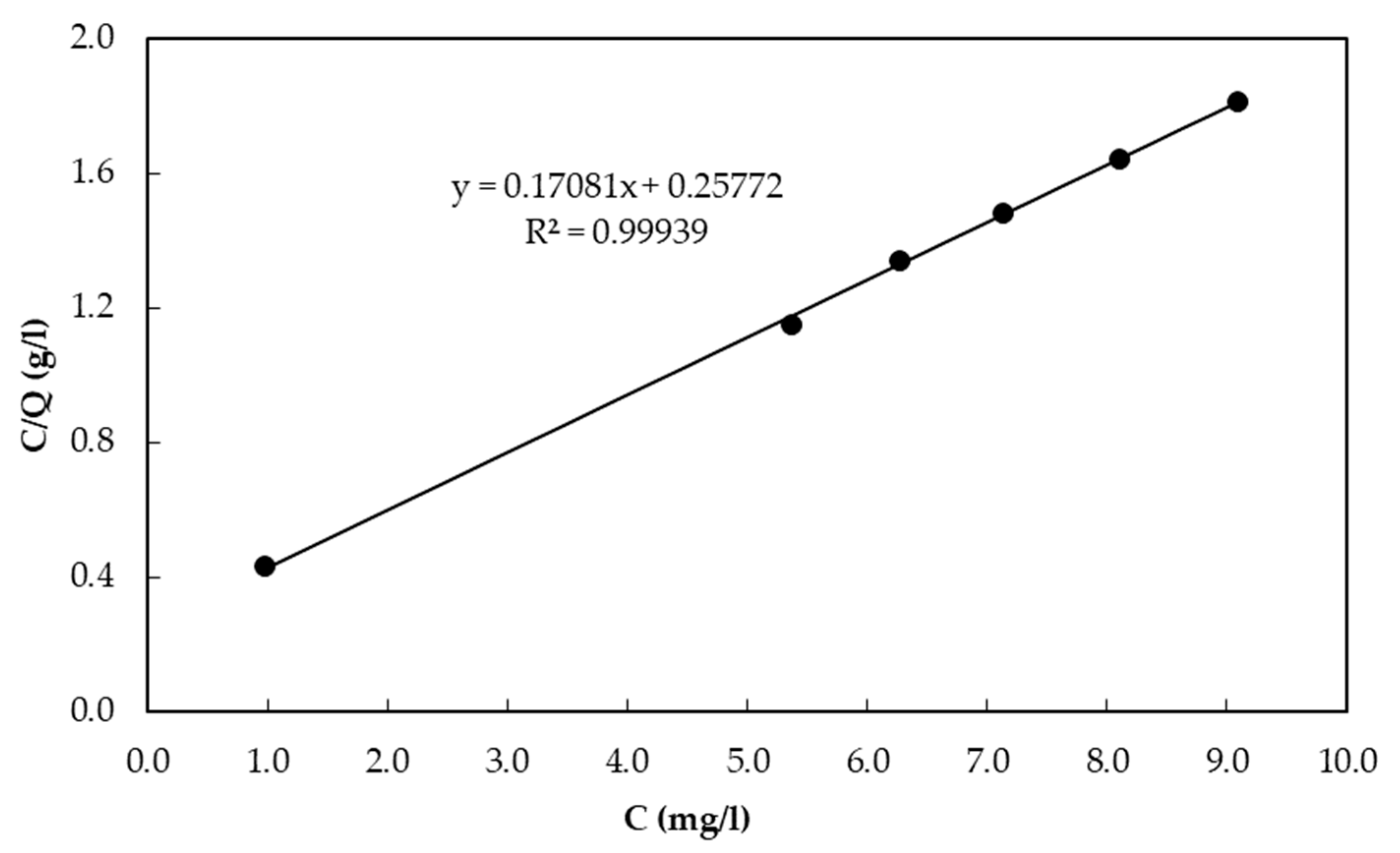
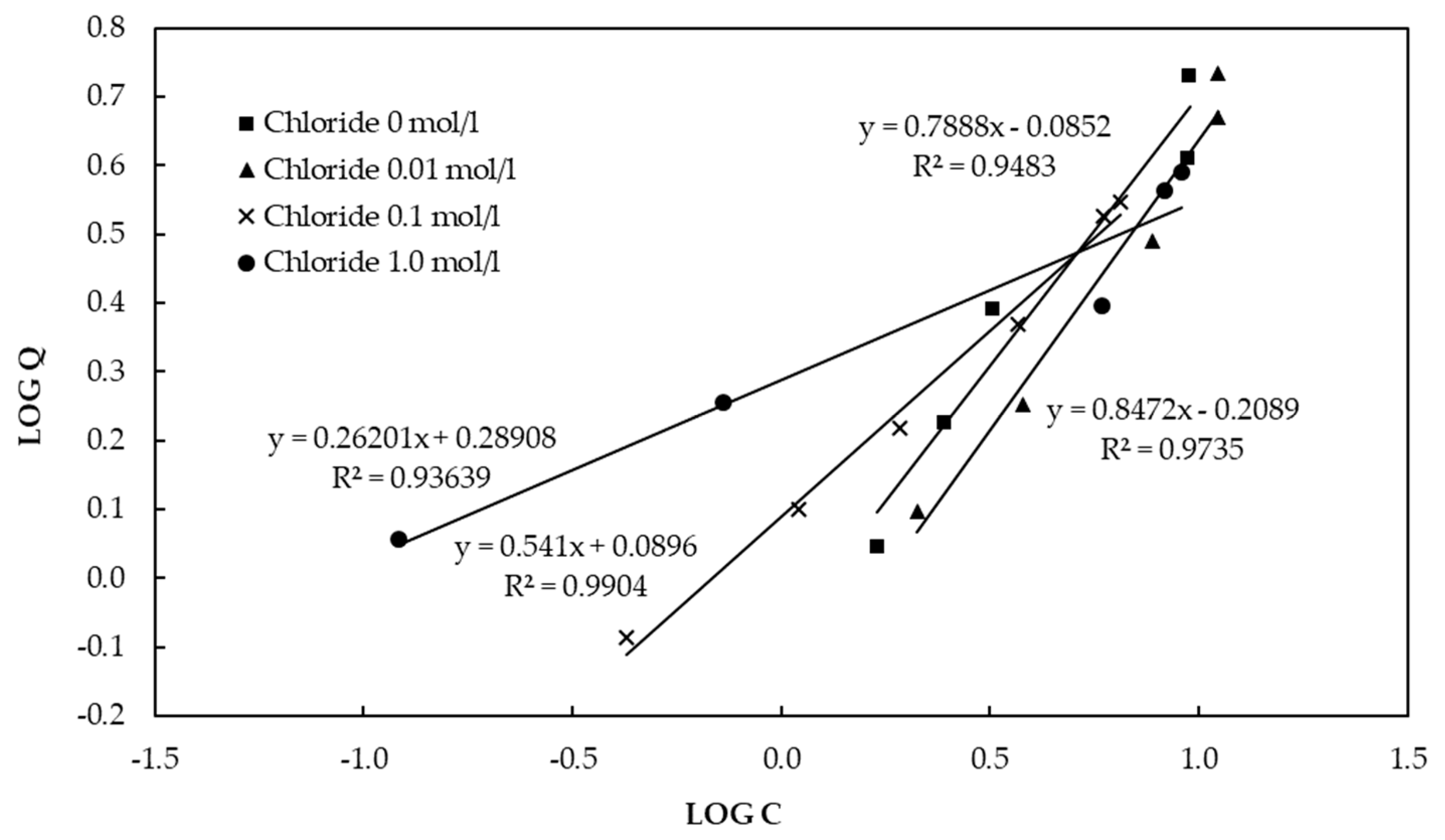
3.2. Adsorption Capacity of the CBC Based on Equilibrium Sorption Isotherms
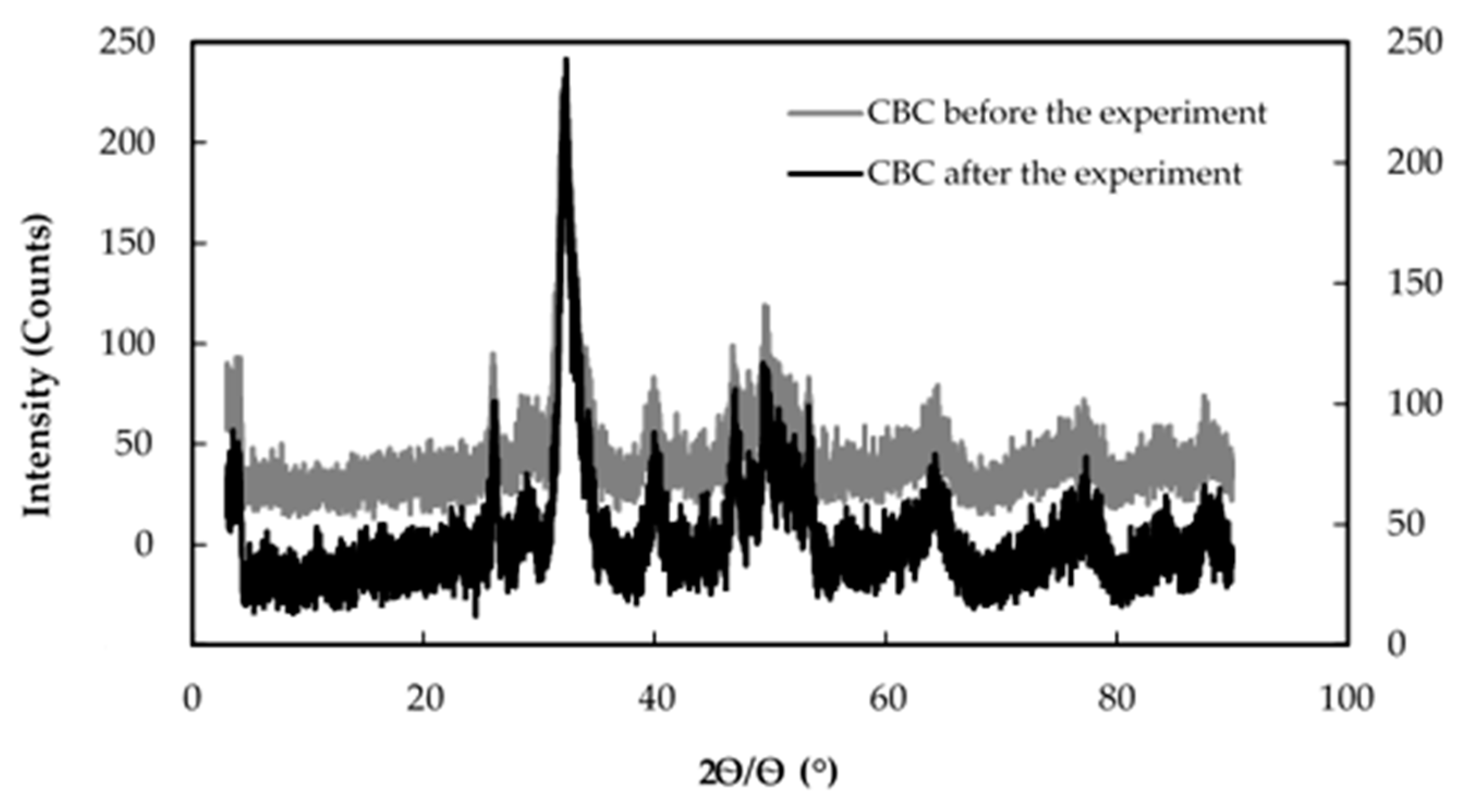
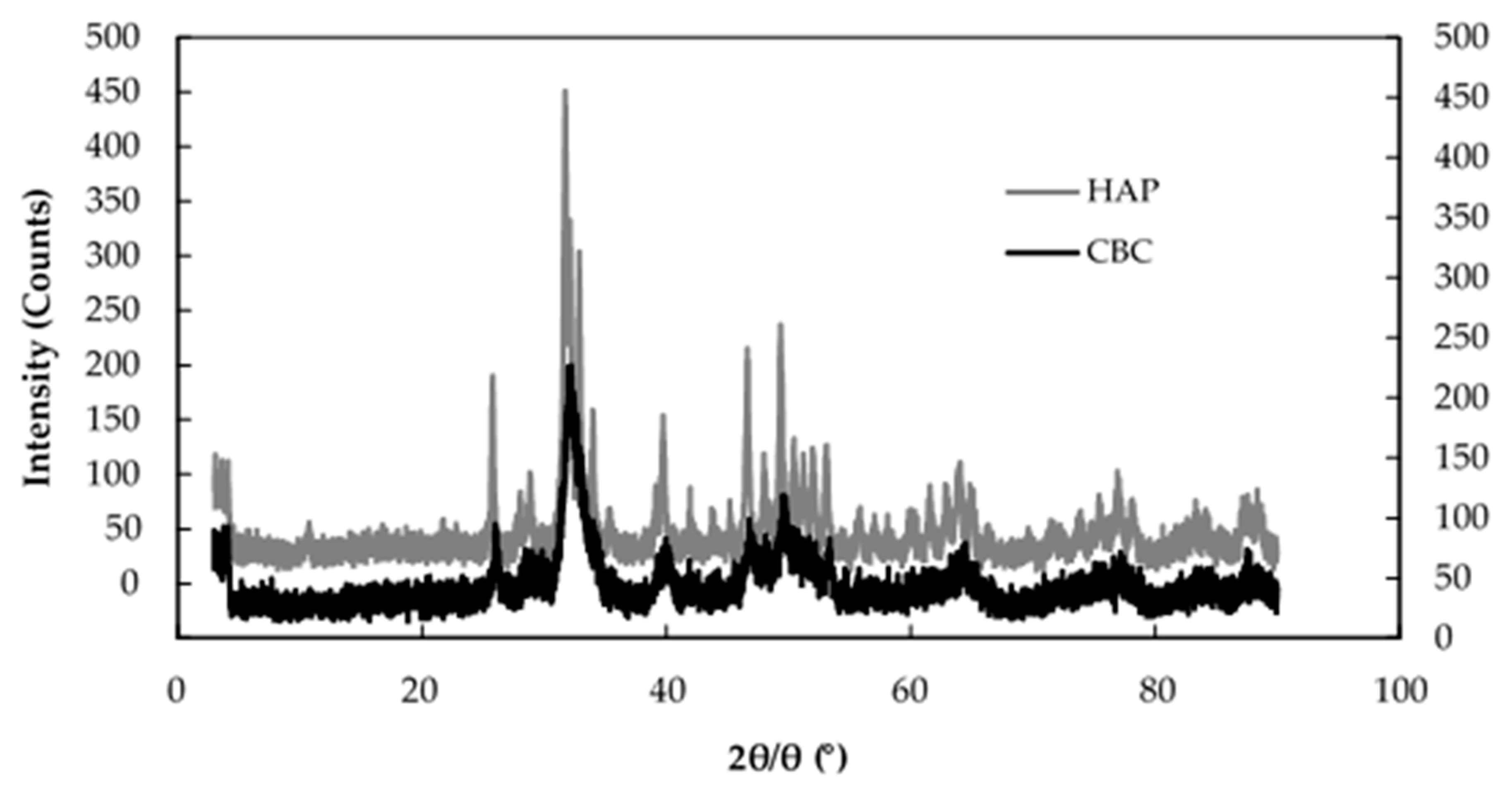
3.3. Fluoride Removal Mechanism of CBC
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Bhatnagar, A.; Kumar, E.; Sillanpaa, M. Fluoride removal from water by adsorption—A review. Chem. Eng. J. 2011, 171, 811–840. [Google Scholar] [CrossRef]
- Mohapatra, M.; Anand, S.; Mishra, B.K.; Giles, D.E.; Singh, P. Review of fluoride removal from drinking water. J. Environ. Manag. 2009, 91, 67–77. [Google Scholar] [CrossRef] [PubMed]
- Mjengera, H.; Mkongo, G. Appropriate deflouridation technology for use in flourotic areas in Tanzania. Phys. Chem. Earth. 2003, 28, 1097–1104. [Google Scholar] [CrossRef]
- World Health Organization. Guidelines for Drinking—Water Quality, 4th ed.; WHO: Geneva, Switzerland, 2011. [Google Scholar]
- Meenakshi; Maheshwari, R.C. Fluoride in drinking water and its removal. J. Hazard. Mater. 2006, B137, 456–463. [Google Scholar]
- Rojas-Mayorga, C.K.R.; Petriciolet, A.B.; Villarreal, I.A.A.; Montoya, V.H.; Virgen, M.R.M.; Gomez, R.T.; Moran, M.A.M. Optimization of pyrolysis conditions and adsorption properties of bone char for fluoride removal from water. J. Anal. Appl. Pyrolysis 2013, 104, 10–18. [Google Scholar] [CrossRef]
- Medellin-Castillo, N.A.; Leyva-Ramos, R.; Ocampo-Perez, R.; Cruz, R.F.G.D.L.; Argon-Pina, A.; Martinez-Rosales, J.M.; Guerrero-Coronado, R.M.; Fuentes-Rubio, L. Adsorption of fluoride from water solution on bone char. Ind. Eng. Chem. Res. 2007, 46, 9205–9212. [Google Scholar] [CrossRef]
- Feng, L.; Xu, W.; Liu, T.; Liu, J. Heat regeneration of hydroxyapatite/attapulgite composite beads for defluoridation of drinking water. J. Hazard. Mater. 2012, 221–222, 228–235. [Google Scholar] [CrossRef] [PubMed]
- Yadav, A.K.; Kaushik, C.P.; Haritash, A.K.; Kansal, A.; Rani, N. Defluoridation of ground water using brick powder as an adsorbent. J. Hazard. Mater. 2006, 128, 289–293. [Google Scholar] [CrossRef] [PubMed]
- Herath, H.M.A.S.; Kubota, K.; Kawakami, T.; Nagasawa, S.; Motoyama, A.; Weragoda, S.K.; Chaminda, G.G.T.; Yatigammana, S. Potential risk of drinking water to human health in Sri Lanka. J. Environ. Forensics 2017, 18, 241–250. [Google Scholar] [CrossRef]
- Ma, W.; Ya, F.; Wang, R.; Zhao, Y. Fluoride removal from drinking water by adsorption using bone char as a biosorbent. Int. J. Environ. Technol. Manag. 2008, 9, 59–69. [Google Scholar] [CrossRef]
- Sakhare, N.; Lunge, S.; Rayalu, S.; Bakardjiva, S.; Subrt, J.; Devotta, S.; Labhsetwar, N. Defluoridation of water using calcium aluminate materials. Chem. Eng. J. 2012, 203, 406–414. [Google Scholar] [CrossRef]
- Essadki, A.H.; Gourich, B.; Vial, C.; Delmas, H.; Bennajah, M. Defluoridation of drinking water by electrocoagulation/electroflotation in a stirred tank reactor with a comparative performance to an external-loop airlift reactor. J. Hazard. Mater. 2009, 168, 1325–1333. [Google Scholar] [CrossRef] [PubMed]
- Herath, H.M.A.S. Public Health and Groundwater Quality in Sri Lanka and Defluoridation of Drinking Water in Relation to Chronic Kidney Disease of Unknown Etiology. Ph.D. Thesis, Toyama Prefectural University, Imizu, Toyama Prefecture, Japan, 2018. [Google Scholar]
- Nicolas, S.; Guihard, L.; Marchand, A.; Bariou, B.; Amrane, A.; Mazighi, A.; Mameri, N.; Midaoui, A.E. Defluoridation of brackish northern Sahara groundwater—Activity product calculations in order to optimize pretreatment before reverse osmosis. Desalination 2010, 256, 9–15. [Google Scholar] [CrossRef]
- Cui, H.; Li, Q.; Qian, Y.; Tang, R.; An, H.; Zhai, J. Defluoridation of water via electrically controlled anion exchange by polyaniline modified electrode reactor. Water Res. 2011, 45, 5736–5744. [Google Scholar] [CrossRef] [PubMed]
- Sivasankar, V.; Rajkumar, S.; Murugesh, S.; Darchen, A. Influence of shaking or stirring dynamic methods in the defluoridation behavior of activated tamarind fruit shell carbon. Chem. Eng. J. 2012, 197, 162–172. [Google Scholar] [CrossRef]
- Loganathan, P.; Vigneswaran, S.; Kandasamy, J.; Naidu, R. Defluoridation of drinking water using adsorption processes. J. Hazard. Mater. 2013, 248–249, 1–19. [Google Scholar] [CrossRef] [PubMed]
- Herath, H.M.A.S.; Kawakami, T.; Tafu, M. Regeneration of exhausted chicken bone char (CBC) to optimize its usage in the defluoridation of drinking water. J. Ecotechnol. Res. 2018, 18, 39–46. [Google Scholar]
- Larsen, M.J.; Pearce, E.I.F.; Ravnholt, G. The effectiveness of bone char in the defluoridation of water in relation to its crystallinity, carbon content and dissolution pattern. Arch. Oral Biol. 1994, 39, 807–816. [Google Scholar] [CrossRef]
- Leyva-Ramos, R.; Rivera-Utrilla, J.; Medellin-Castillo, N.A.; Sanchez-Polo, M. Kinetic modeling of fluoride adsorption from aqueous solution onto bone char. Chem. Eng. J. 2010, 158, 458–467. [Google Scholar] [CrossRef]
- Brunson, L.R.; Sabatini, D.A. An evaluation of fish bone char as an appropriate arsenic and fluoride removal technology for emerging regions. Environ. Eng. Sci. 2009, 26, 1777–1784. [Google Scholar] [CrossRef]
- Kaseva, M.E. Optimization of regenerated bone char for fluoride removal in drinking water: A case study in Tanzania. J. Water Health 2006, 4, 139–147. [Google Scholar] [CrossRef] [PubMed]
- Kawasaki, N.; Ogata, F.; Tominaga, H.; Yamaguchi, I. Removal of fluoride ion by bone char produced from animal biomass. J. Oleo Sci. 2009, 58, 529–535. [Google Scholar] [CrossRef] [PubMed]
- Ismail, Z.Z.; Abdelkareem, H.N. Sustainable approach for recycling waste lamb and chicken bones for fluoride removal from water followed by reusing fluoride-bearing waste in concrete. Waste Manag. 2015, 45, 66–75. [Google Scholar] [CrossRef] [PubMed]
- Fawell, J.; Bailey, K.; Chilton, J.; Dahi, E.; Fewtrell, L.; Magara, Y. Fluoride in Drinking Water; World Health Organization: Geneva, Switzerland; IWA Publishing: London, UK, 2006. [Google Scholar]
- Abe, I.; Iwasaki, S.; Tokimoto, T.; Kawasaki, N.; Nakamura, T.; Tanada, S. Adsorption of fluoride ions onto carbonaceous materials. J. Colloid Interface Sci. 2004, 275, 35–39. [Google Scholar] [CrossRef] [PubMed]
- Ooi, C.Y.; Hamdi, M.; Ramesh, S. Properties of hydroxyapatite produced by annealing of bovine bone. Ceram. Int. 2007, 33, 1171–1177. [Google Scholar] [CrossRef]
- Mwaniki, D.L. Fluoride sorption characteristics of different grades of bone charcoal, based on batch tests. J. Dent. Res. 1992, 71, 1310–1315. [Google Scholar] [CrossRef] [PubMed]


| F− in the Solution | mg |
|---|---|
| F− added to the solution | 2831 |
| F− lost from the solution by leakage | 492 |
| F− remaining in the solution | 483 |
| F− adsorbed by CBC (55 g*3) | 1855 |
| F− Adsorption Capacity (mg/g) | ||||
|---|---|---|---|---|
| F− Content in CBC before Adsorption | F− Content in CBC after Adsorption | Net Adsorption | Averaged Value | |
| 0.46 | F1 | 12.13 | 11.67 | 11.1 |
| F2 | 12.05 | 11.59 | ||
| F3 | 10.43 | 9.97 | ||
| Particle Size | State of CBC | BET Surface Area (m2/g) |
|---|---|---|
| 106–212 μm | CBC before the fluoride adsorption | 126 |
| CBC after the fluoride adsorption | 136 |
| Solution | Concentration (mg/L) | |||||||
|---|---|---|---|---|---|---|---|---|
| F− | Cl− | PO43− | Na+ | NH4+ | K+ | Mg2+ | Ca2+ | |
| Initial solution | 20 | 0 | 0 | 19 | 0 | 0 | 0 | 0 |
| Final solution | 10 | 87 | 0 | 61 | 0 | 3 | 5 | 10 |
| State of CBC | No. of Moles in 100 g of CBC | |||
|---|---|---|---|---|
| PO43− | Ca2+ | F− | OH− | |
| CBC before the fluoride adsorption | 0.371 | 0.691 | 0.002 | 0.138 |
| CBC after the fluoride adsorption | 0.351 | 0.684 | 0.061 | 0.075 |
| Solution | pH | No. of OH− Moles (µ moles/L) | Alkalinity (μeq/L) |
|---|---|---|---|
| Initial solution | 5.21 | 0.002 | 0 |
| Final solution | 7.82 | 0.661 | 762 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Herath, H.M.A.S.; Kawakami, T.; Tafu, M. The Extremely High Adsorption Capacity of Fluoride by Chicken Bone Char (CBC) in Defluoridation of Drinking Water in Relation to Its Finer Particle Size for Better Human Health. Healthcare 2018, 6, 123. https://doi.org/10.3390/healthcare6040123
Herath HMAS, Kawakami T, Tafu M. The Extremely High Adsorption Capacity of Fluoride by Chicken Bone Char (CBC) in Defluoridation of Drinking Water in Relation to Its Finer Particle Size for Better Human Health. Healthcare. 2018; 6(4):123. https://doi.org/10.3390/healthcare6040123
Chicago/Turabian StyleHerath, H. M. Ayala S., Tomonori Kawakami, and Masamoto Tafu. 2018. "The Extremely High Adsorption Capacity of Fluoride by Chicken Bone Char (CBC) in Defluoridation of Drinking Water in Relation to Its Finer Particle Size for Better Human Health" Healthcare 6, no. 4: 123. https://doi.org/10.3390/healthcare6040123
APA StyleHerath, H. M. A. S., Kawakami, T., & Tafu, M. (2018). The Extremely High Adsorption Capacity of Fluoride by Chicken Bone Char (CBC) in Defluoridation of Drinking Water in Relation to Its Finer Particle Size for Better Human Health. Healthcare, 6(4), 123. https://doi.org/10.3390/healthcare6040123





