Abstract
Electrical stimulation (ES) has been shown to have beneficial effects in wound healing. It is important to assess the effects of ES on cutaneous wound healing in order to ensure optimization for clinical practice. Several different applications as well as modalities of ES have been described, including direct current (DC), alternating current (AC), high-voltage pulsed current (HVPC), low-intensity direct current (LIDC) and electrobiofeedback ES. However, no one method has been advocated as the most optimal for the treatment of cutaneous wound healing. Therefore, this review aims to examine the level of evidence (LOE) for the application of different types of ES to enhance cutaneous wound healing in the skin. An extensive search was conducted to identify relevant clinical studies utilising ES for cutaneous wound healing since 1980 using PubMed, Medline and EMBASE. A total of 48 studies were evaluated and assigned LOE. All types of ES demonstrated positive effects on cutaneous wound healing in the majority of studies. However, the reported studies demonstrate contrasting differences in the parameters and types of ES application, leading to an inability to generate sufficient evidence to support any one standard therapeutic approach. Despite variations in the type of current, duration, and dosing of ES, the majority of studies showed a significant improvement in wound area reduction or accelerated wound healing compared to the standard of care or sham therapy as well as improved local perfusion. The limited number of LOE-1 trials for investigating the effects of ES in wound healing make critical evaluation and assessment somewhat difficult. Further, better-designed clinical trials are needed to improve our understanding of the optimal dosing, timing and type of ES to be used.
1. Introduction
Acute wounds normally undergo a complex healing process, which ultimately leads to a completely healed wound [1]. The process of acute wound healing is typically divided into a series of overlapping phases, which include: haemostasis, inflammation, proliferation, wound contraction and remodeling [2]. Normal would healing in the skin should result in the restoration of skin continuity and function. Nevertheless, there are a number of responses which can occur following a cutaneous injury; normal repair in the adult human skin should typically produce a fine line permanent scar, however, abnormal healing can result in excessive healing where there is an increased deposition of connective tissue leading to the formation of hypertrophic and keloid scars or either can deficient healing where there is insufficient deposition of connective tissue and therefore, new tissue formation is incomplete and can result in the formation of chronic wounds [2].
Chronic wounds are defined as those wounds that have failed to proceed through the reparative phases of healing in less than 42 days [3,4]. There are various factors that can delay wound healing such as diabetes, vascular insufficiency, age and nutritional deficiencies [3]. Chronic wounds represent a major health burden to both the patient and the physician and impact upon global health resources. It is estimated that the total expenditure per year in the United Kingdom for managing these wounds in the National Health Service (NHS) alone is in excess of £1bn [5,6]. The actual number of patients suffering from these wounds, are on the increase, as the ageing population and the increasing incidence of risk factors such as diabetes mellitus and smoking, result in the rising incidence of chronic wound formation. Furthermore, patients have reported that these wounds can affect their quality of life due to social isolation, reduced working hours and dependency upon the healthcare system [7].
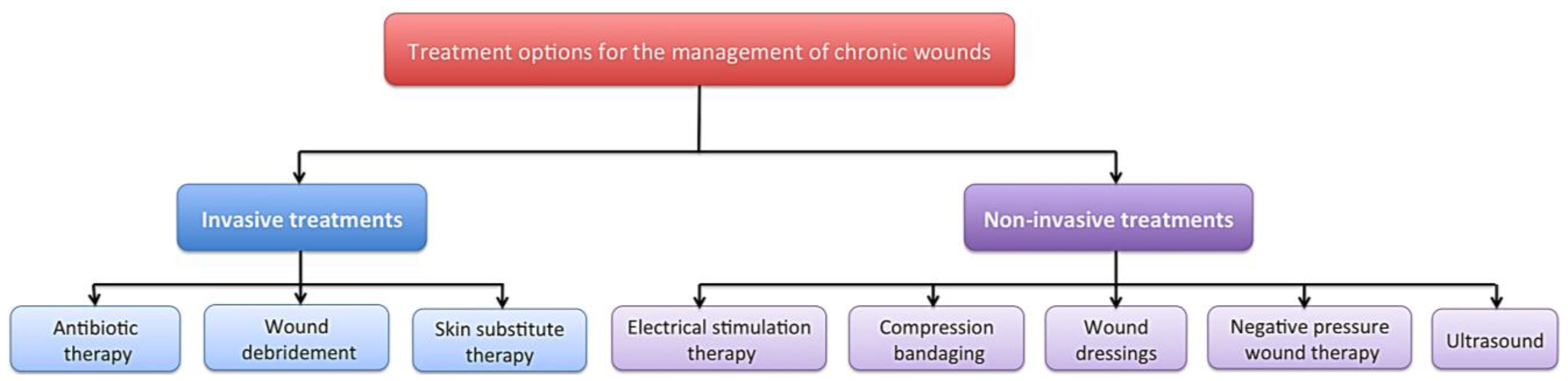
There are a range of treatment strategies available including; compression bandaging [8], wound dressings [9], negative pressure wound therapy [10], ultrasound [11], debridement [12] and skin substitutes [13], which can be expensive, time consuming and may be slow to demonstrate any positive results (Figure 1). Despite the multitude of treatment options, current regimes are not adequate, as these wounds remain a significant economic burden and a clinical problem. The use of electrical stimulation (ES) for the treatment of both acute and chronic wounds has gained prominence in the literature [14,15,16,17].
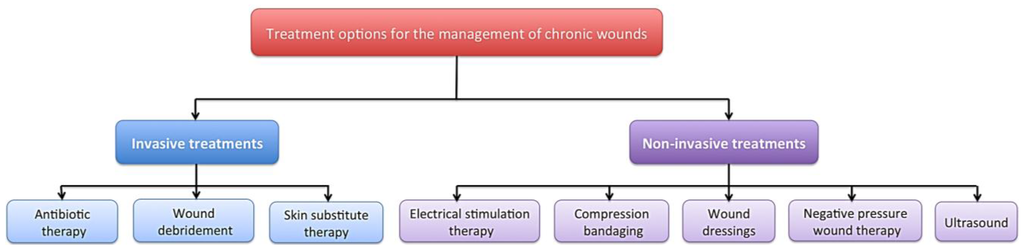
Figure 1.
A diagram to demonstrate some of the available treatment strategies for the management of chronic wounds including; compression bandaging, wound dressings, negative pressure wound therapy, ultrasound, debridement, skin substitute therapy and electrical stimulation.
Figure 1.
A diagram to demonstrate some of the available treatment strategies for the management of chronic wounds including; compression bandaging, wound dressings, negative pressure wound therapy, ultrasound, debridement, skin substitute therapy and electrical stimulation.
Many studies have advocated the use of ES therapy in conjunction with standard wound care [14,15,16,17]. ES is defined as the application of electrical current through electrodes placed on the skin either near or directly on the wound [18]. ES has been shown to have beneficial effects on the different phases of cutaneous wound healing in both chronic [19,20,21,22,23,24,25,26,27] (Figure 2) and acute wounds (Figure 3) [15,16,17,18,28,29,30,31,32,33,34]. It is suggested that ES can reduce infection, improve cellular immunity, increase perfusion, and accelerate cutaneous wound healing [35]. Undamaged human skin has an endogenous electrical potential and a transcutaneous current potential of 10–60 mV [36]. This is generated by the movement of sodium ions through Na+/K+ ATPase pumps in the epidermis [37]. Following an injury to the skin, a flow of current through the wound pathway generates a lateral electrical field and this is termed the “current of injury” or “skin battery” effect (Figure 4) [38]. Therefore, the current of injury is thought to be significant in initiating repair [38].
ES has been used for a number of clinical applications, such as pain management and wound healing including chronic and acute wounds [39]. ES devices have varying voltages, currents, modes and length of time of application. Additionally, mono- or bipolar and bi or tri-electrodes are used, as well as different types of wounds indicated for each modality. There are a number of ES devices and methods of application such as dressings, electrode placement and practitioner-assisted [40,41,42,43] (Figure 5). However, the majority of trials apply the electrodes directly on the skin, and often, directly onto the wound. Several different modalities and electrical waveforms have been described (Figure 6), including direct current (DC), alternating current (AC), high-voltage pulsed current (HVPC), and low-intensity direct current (LIDC) [44]. One of the most familiar types of ES is transcutaneous electrical nerve stimulation (TENS), which has been used frequently for pain control [44,45]. Additionally, frequency rhythmic electrical modulation systems (FREMS) is also a form of transcutaneous electrotherapy using ES that varies the pulse, frequency, duration, and voltage [46]. Recently, an electrobiofeedback device, called the Fenzian system, where its waveform was found to appear as degenerate waves (DW), which degenerate over time, has been used in the treatment of acute cutaneous wound healing and reduced the symptoms associated with abnormal skin scarring [34,47,48].
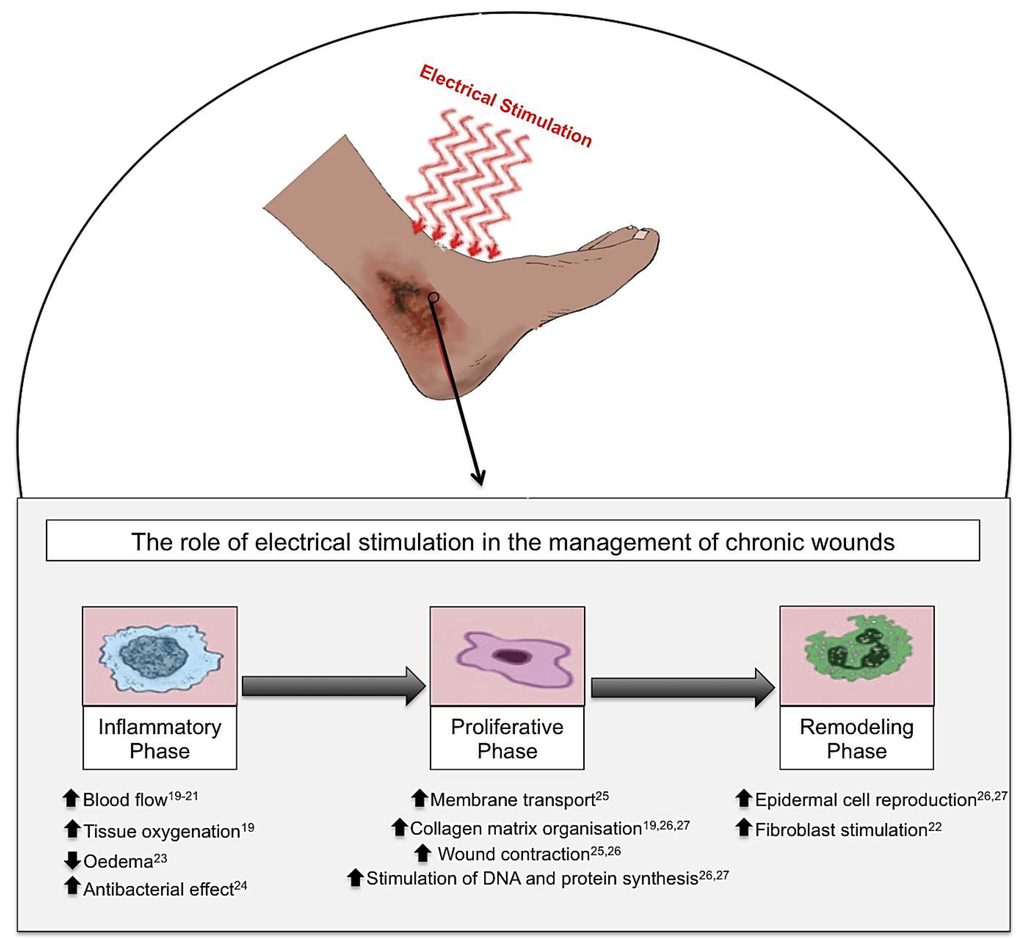
Figure 2.
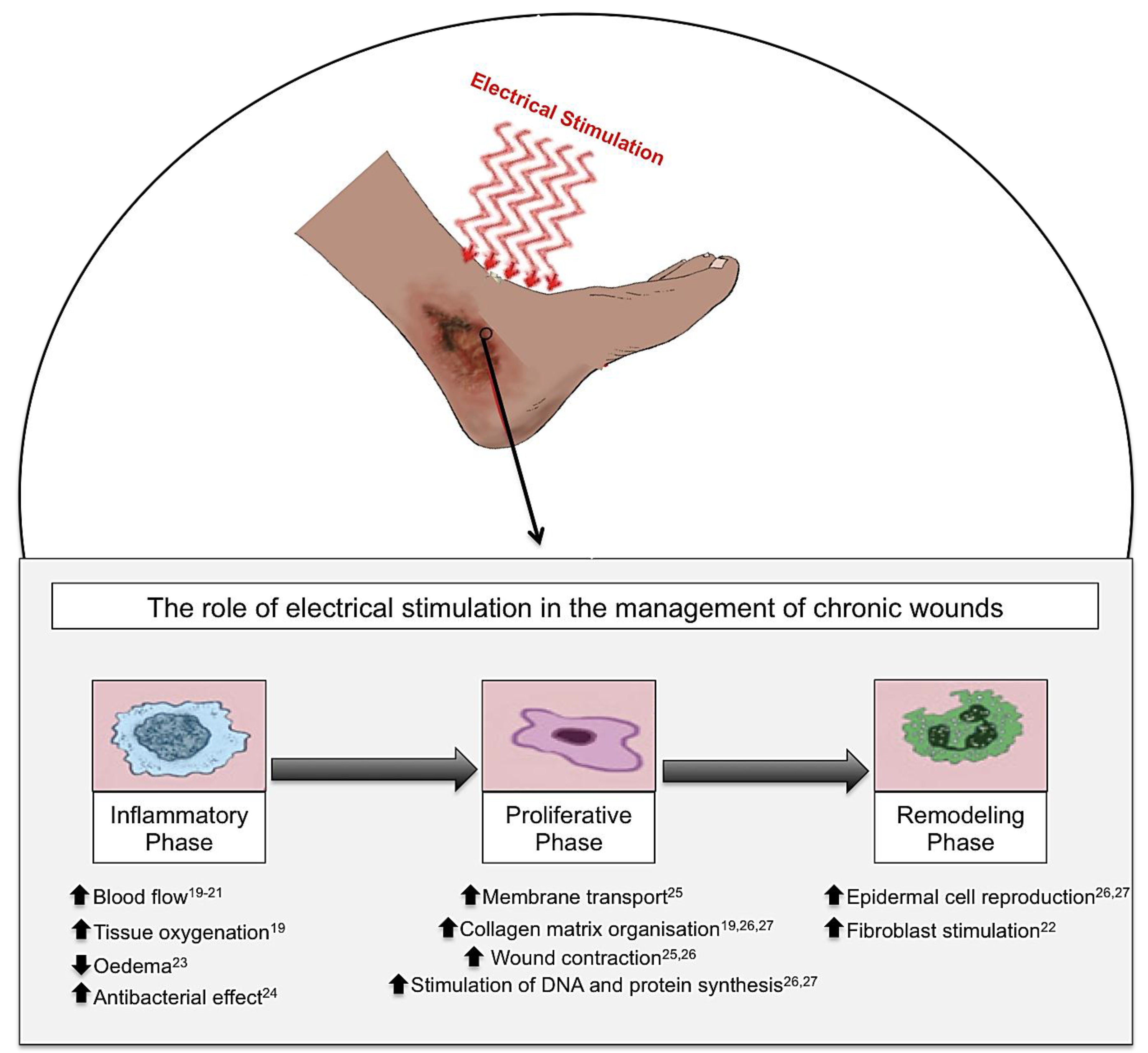
Electrical stimulation (ES), in the form of alternating current (AC), direct current (DC) and pulsed current (PC), has been shown to have beneficial effects on cutaneous wound healing in chronic wounds. When ES is applied to a chronic wound, this produces beneficial effects throughout the three phases of wound healing: inflammation, proliferation and remodelling phases. Inflammatory phase: ES increases blood flow, tissue oxygenation and stimulates fibroblasts whilst reducing oedema and providing an increased antibacterial effect. Proliferative phase: ES increases membrane transport, collagen matrix organization, wound contraction and the stimulation of DNA and protein synthesis. Remodelling phase: ES increases epidermal cell proliferation, and migration as well as stimulation of fibroblasts thus enabling enhanced wound closure [19,20,21,22,23,24,25,26,27].
Figure 2.
Electrical stimulation (ES), in the form of alternating current (AC), direct current (DC) and pulsed current (PC), has been shown to have beneficial effects on cutaneous wound healing in chronic wounds. When ES is applied to a chronic wound, this produces beneficial effects throughout the three phases of wound healing: inflammation, proliferation and remodelling phases. Inflammatory phase: ES increases blood flow, tissue oxygenation and stimulates fibroblasts whilst reducing oedema and providing an increased antibacterial effect. Proliferative phase: ES increases membrane transport, collagen matrix organization, wound contraction and the stimulation of DNA and protein synthesis. Remodelling phase: ES increases epidermal cell proliferation, and migration as well as stimulation of fibroblasts thus enabling enhanced wound closure [19,20,21,22,23,24,25,26,27].
Currently, there is a substantial body of work that supports the effectiveness of ES for cutaneous wound healing, although, there tends to be a poor understanding of the associated technology and its potential applications. Therefore, the aim of this review was to examine the results of clinical trials that use ES to accelerate cutaneous wound healing including the most common modalities and applications of ES. Additionally, we identified the level of evidence (LOE) supporting the use of ES in enhancing cutaneous wound healing.
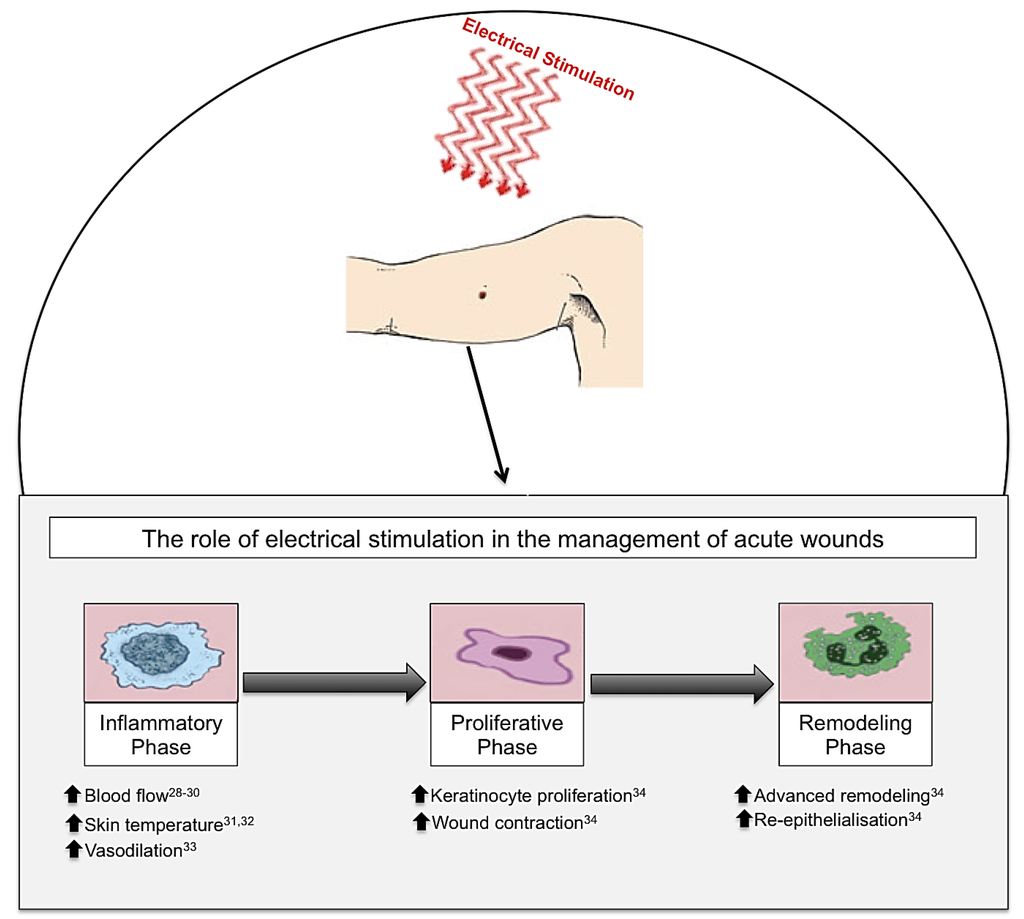
Figure 3.
Electrical stimulation (ES), in the form of biofeedback ES, direct current (DC) and pulsed current (PC), has been shown to have beneficial effects on cutaneous wound healing in acute wounds. When ES is applied to an acute wound, this produces beneficial effects throughout the three phases of wound healing: inflammation, proliferation and remodelling phases. Inflammatory phase: ES increases blood flow, skin temperature and vasodilation. Proliferative phase: ES increases keratinocyte proliferation and wound contraction. Remodelling phase: ES advances the remodelling face and increases re-epithelialisation enabling enhanced wound healing [28,29,30,31,32,33,34].
Figure 3.
Electrical stimulation (ES), in the form of biofeedback ES, direct current (DC) and pulsed current (PC), has been shown to have beneficial effects on cutaneous wound healing in acute wounds. When ES is applied to an acute wound, this produces beneficial effects throughout the three phases of wound healing: inflammation, proliferation and remodelling phases. Inflammatory phase: ES increases blood flow, skin temperature and vasodilation. Proliferative phase: ES increases keratinocyte proliferation and wound contraction. Remodelling phase: ES advances the remodelling face and increases re-epithelialisation enabling enhanced wound healing [28,29,30,31,32,33,34].
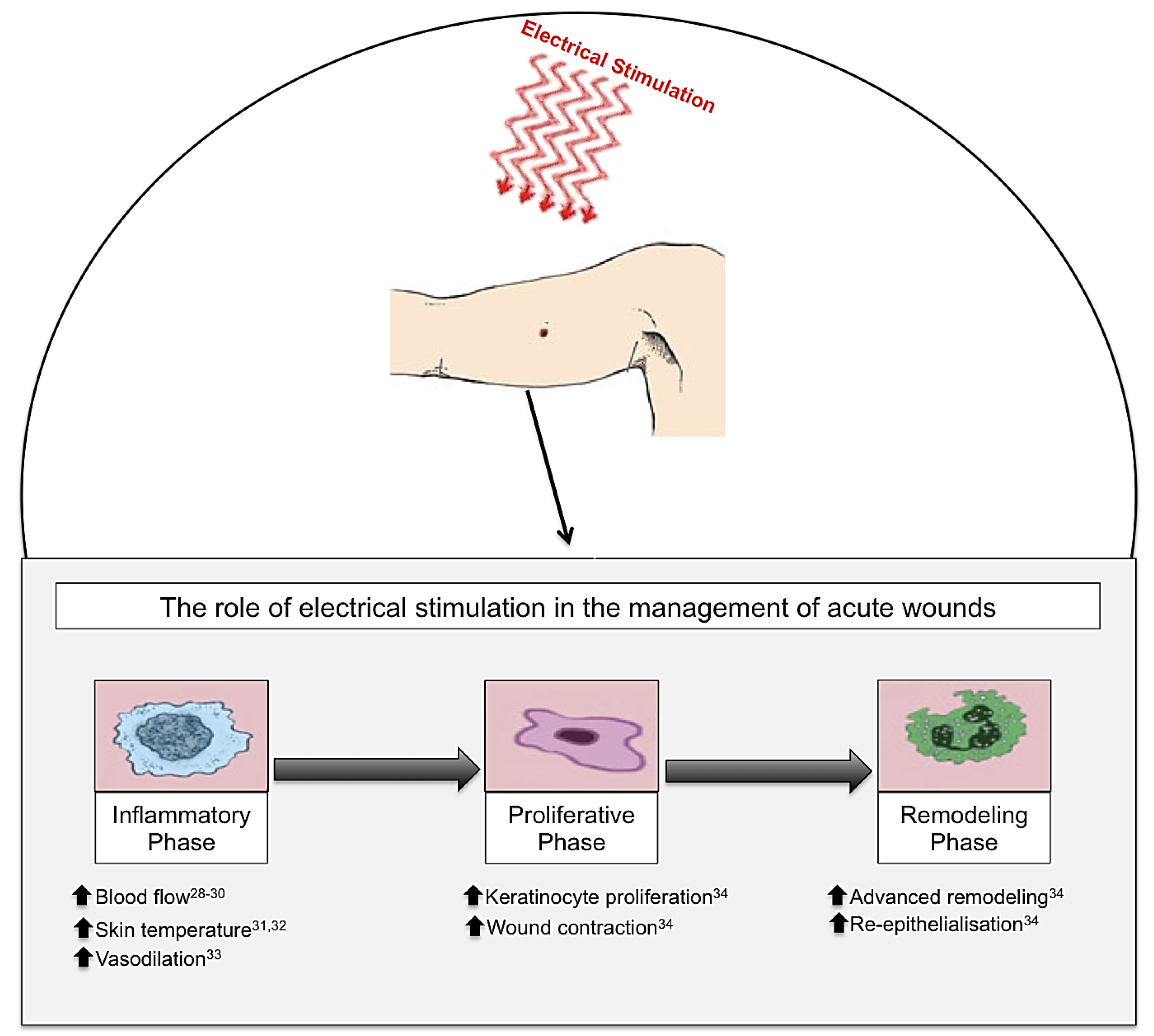
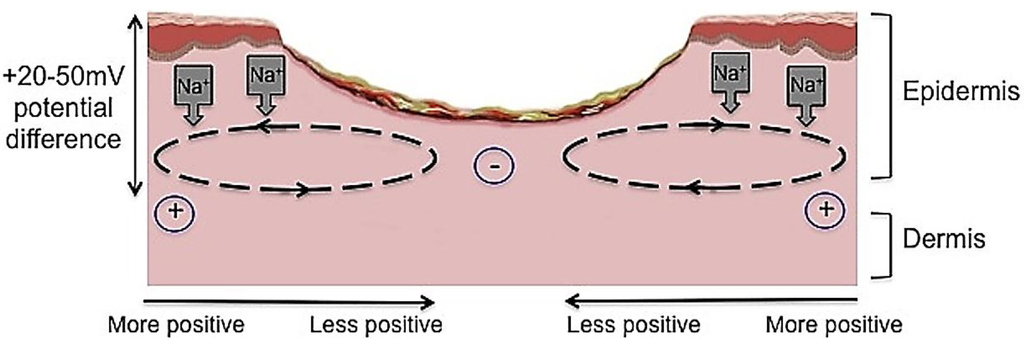
Figure 4.
The current of injury is thought to be significant in initiating repair. Undamaged human skin has an endogenous electrical potential and a transcutaneous current potential of 20–50 mV. This is generated by the movement of sodium ions through Na+/K+ ATPase pumps in the epidermis. The current of injury is generated through epithelial disruption. Following an injury to the skin, a flow of current through the wound pathway generates a lateral electrical field and this is termed the “current of injury” or “skin battery” effect.
Figure 4.
The current of injury is thought to be significant in initiating repair. Undamaged human skin has an endogenous electrical potential and a transcutaneous current potential of 20–50 mV. This is generated by the movement of sodium ions through Na+/K+ ATPase pumps in the epidermis. The current of injury is generated through epithelial disruption. Following an injury to the skin, a flow of current through the wound pathway generates a lateral electrical field and this is termed the “current of injury” or “skin battery” effect.
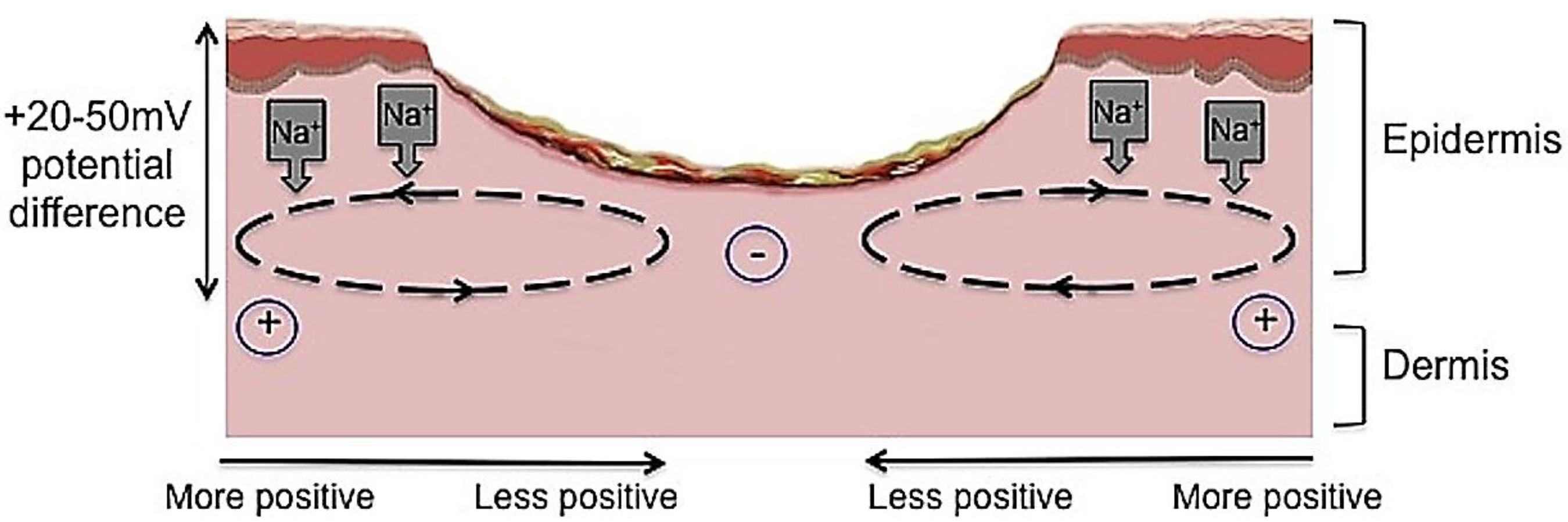
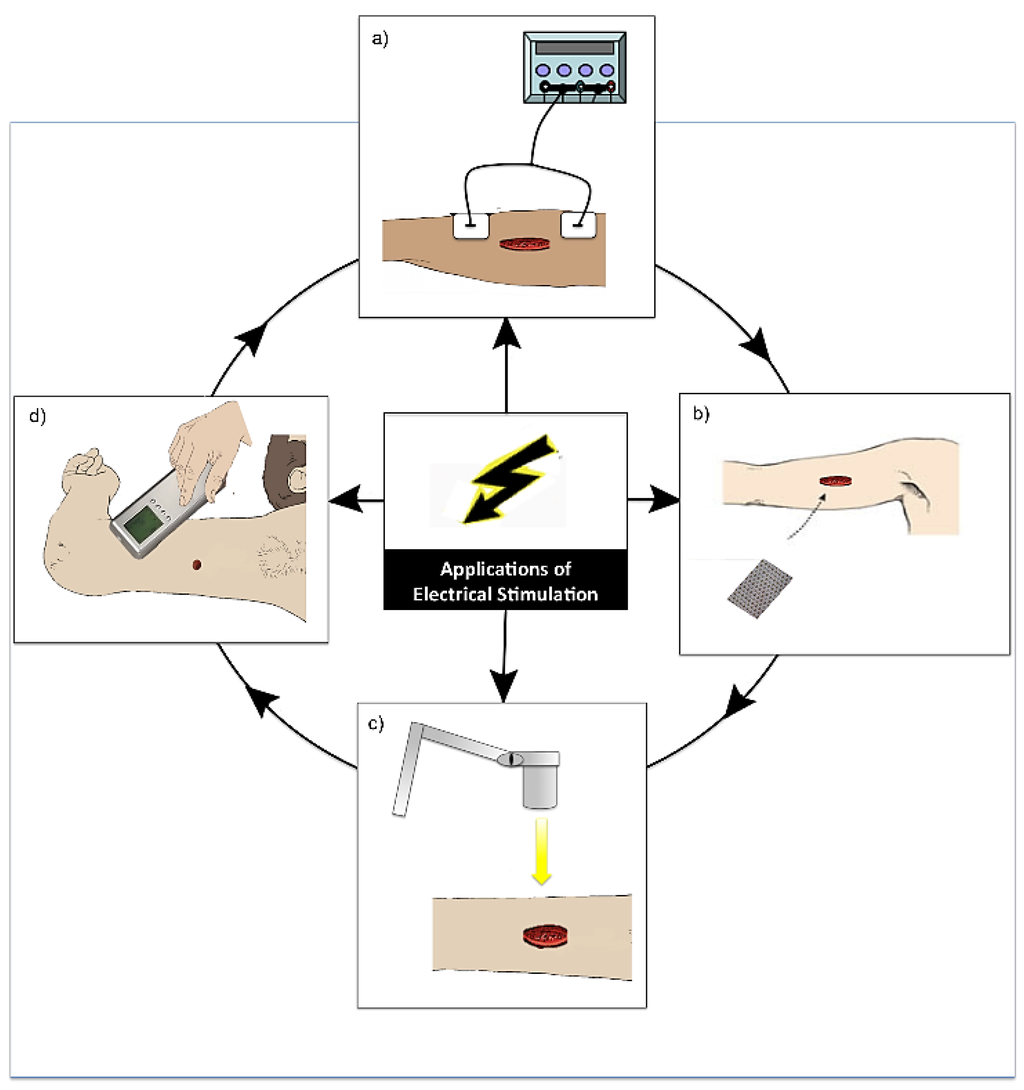
Figure 5.
Diagram demonstrating the various modes of application of electrical stimulation (ES). (a) Application of ES by electrodes placed near or on the wound site and connected to a device (this is the most common application of ES) [40]; (b) Application of a bioelectric dressing to the wound site [41]; (c) Wireless application of ES to a wound [42]; (d) Practitioner application of ES in the form electro biofeedback by the use of a device with an electrode placed in different areas around the wound site [43].
Figure 5.
Diagram demonstrating the various modes of application of electrical stimulation (ES). (a) Application of ES by electrodes placed near or on the wound site and connected to a device (this is the most common application of ES) [40]; (b) Application of a bioelectric dressing to the wound site [41]; (c) Wireless application of ES to a wound [42]; (d) Practitioner application of ES in the form electro biofeedback by the use of a device with an electrode placed in different areas around the wound site [43].
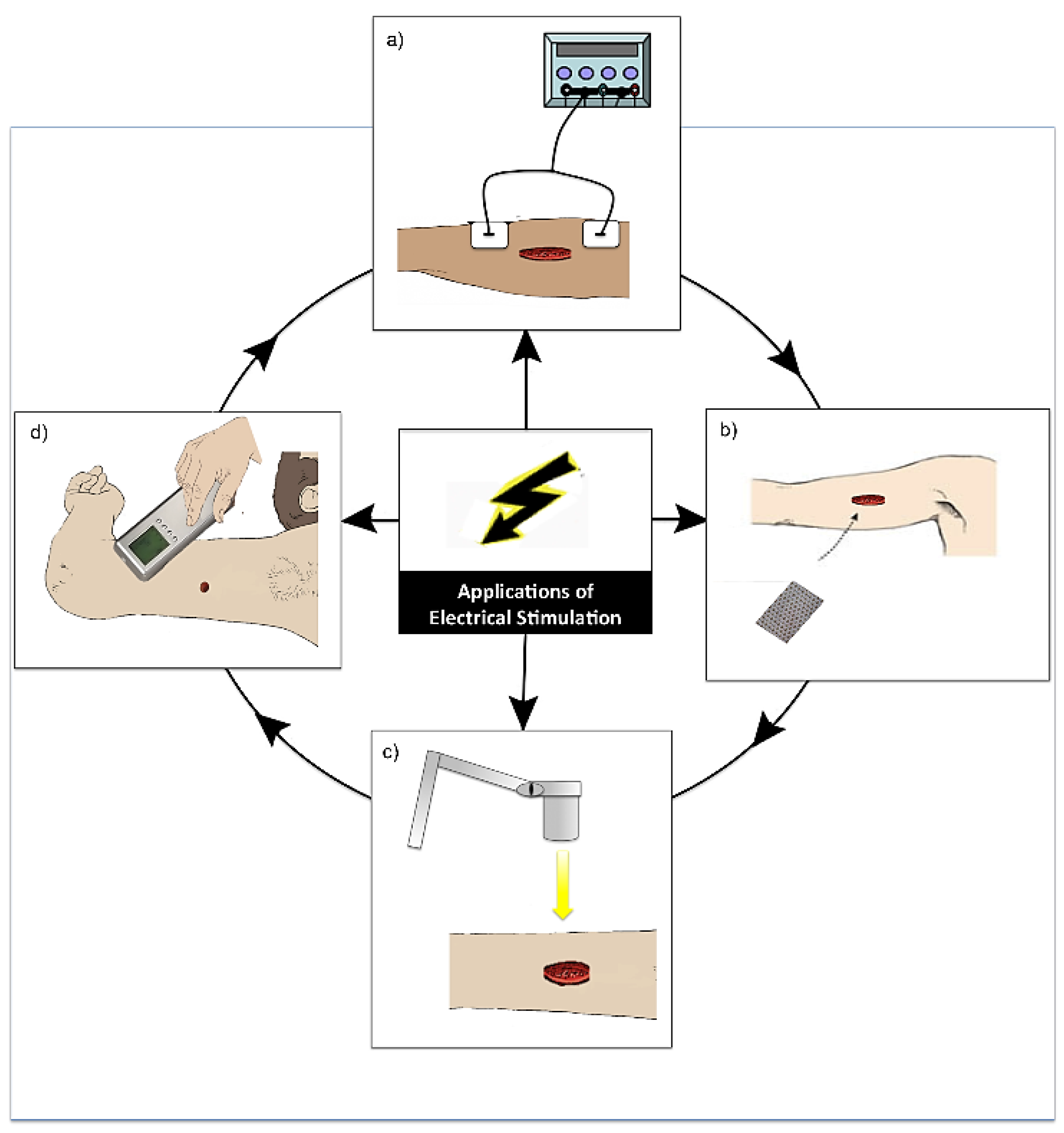
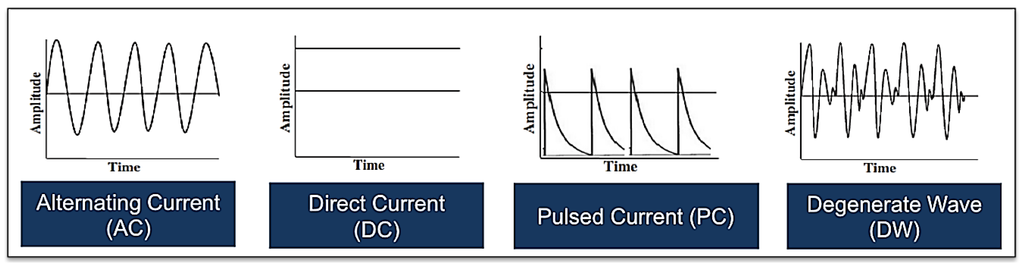
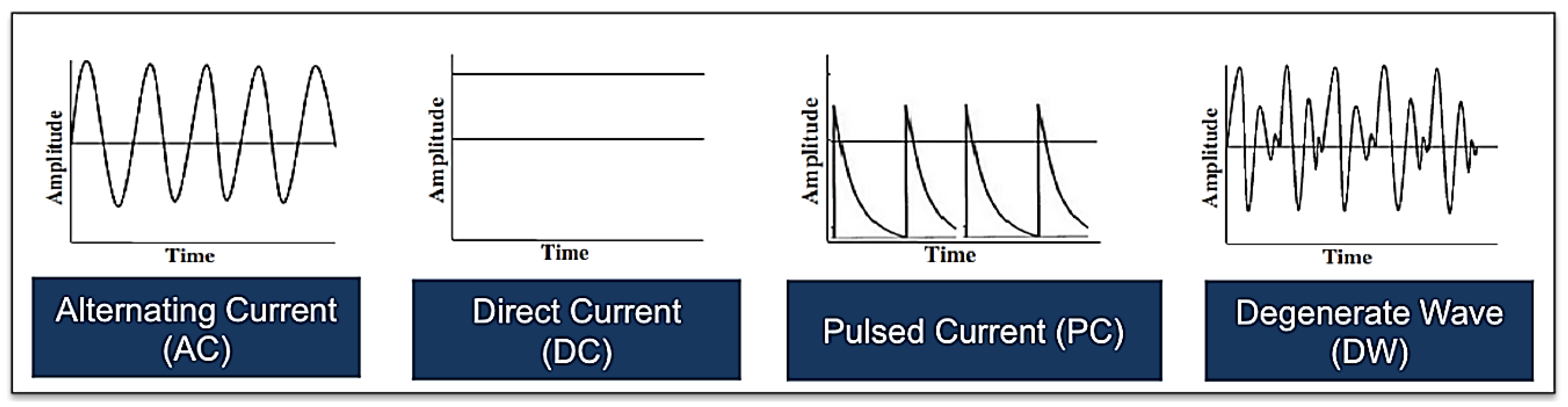
Figure 6.
Illustrations showing one example of each of the various electrical waveforms available for the treatment of acute and chronic cutaneous wounds including alternating current, direct current, pulsed current and degenerate wave (please note that there are other subtypes of each of these waveforms).
Figure 6.
Illustrations showing one example of each of the various electrical waveforms available for the treatment of acute and chronic cutaneous wounds including alternating current, direct current, pulsed current and degenerate wave (please note that there are other subtypes of each of these waveforms).
2. Methods
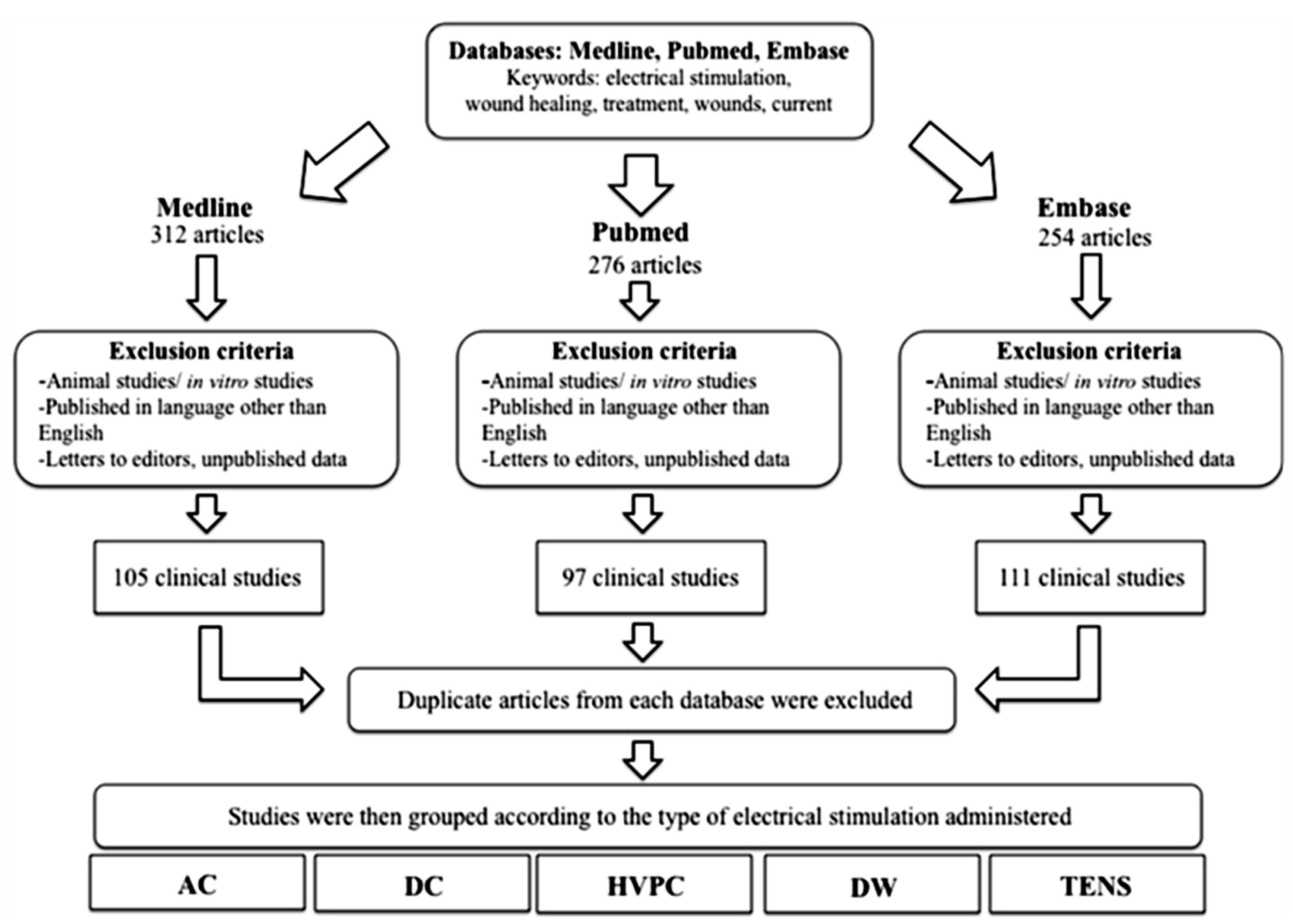
An extensive search was conducted to identify all relevant articles published in the English language, from 1980 onwards, using the following scientific and medical search engines: PubMed, Medline and EMBASE (Figure 7). Only trials involving humans were included. Keywords used in the search included a variety of combinations such as: electrical stimulation, wound healing, treatment, wounds, electric current. All retrieved articles were reviewed for their relevance on the specific topic of electrical stimulation and cutaneous wound healing and 48 were considered suitable for inclusion in this review. Clinical studies were then grouped by the primary method of ES used and then assessed and assigned an LOE adapted from the Oxford Centre for Evidence Based Medicine to establish whether valid and reliable evidence supports the use of ES for wound healing. These levels, ranging from LOE-1 to LOE-5, are based on methodology and study design. These were assigned as follows: LOE 1 = randomized control trial; LOE-2 = cohort study; LOE-3 = case-control study; LOE-4 = Case series study; LOE-5 = expert opinion or case report.
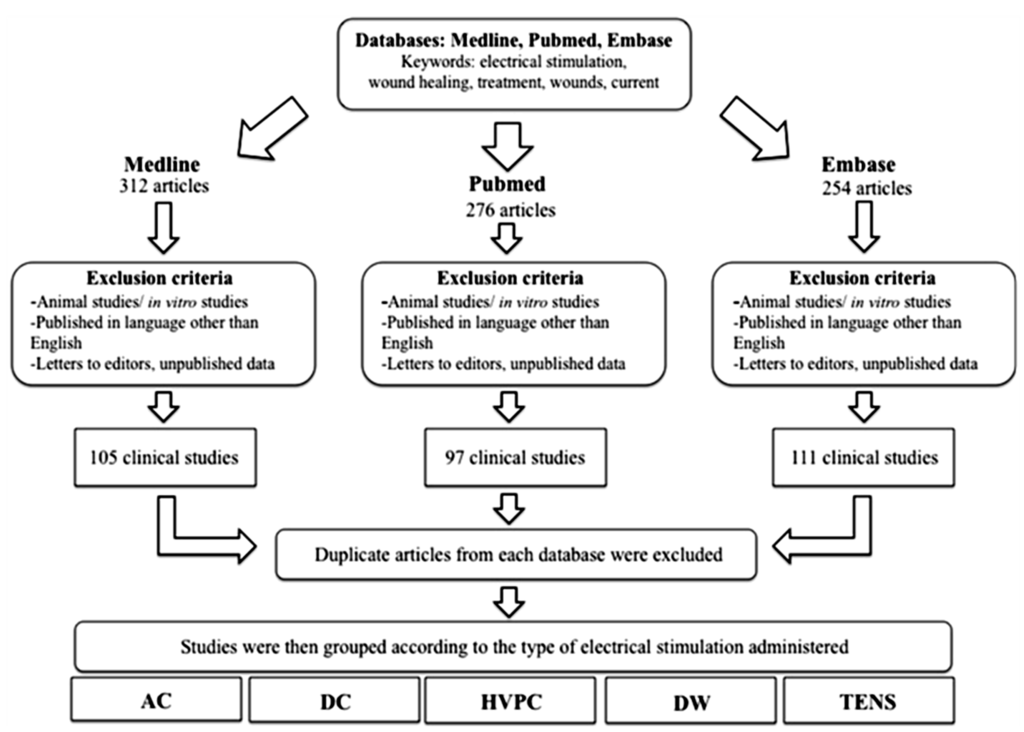
Figure 7.
A flowchart demonstrating the methodology and process of selecting relevant articles for review.
Figure 7.
A flowchart demonstrating the methodology and process of selecting relevant articles for review.
3. Results
The results will now be presented under the following headings: pulsed current, direct current, transcutaneous electrical nerve stimulation, frequency rhythmic electrical modulation system, biofeedback electrical stimulation and bioelectric dressings (Table 1). Low-frequency AC has not been used successfully in the treatment of cutaneous wound healing, due to its lack of polarity [49], therefore, this modality will not be discussed.

Table 1.
A summary table of the literature categorized under the headings; pulsed current, direct current, transcutaneous electrical nerve stimulation, frequency rhythmic electrical modulation system, biofeedback electrical stimulation and bioelectric dressings.
| Author | Design | Type of Wound | Type of ES | No. Patients | Parameters | Duration | LOE Outcome |
|---|---|---|---|---|---|---|---|
| Pulsed Current | |||||||
| Feedar [50] | RCT | Chronic dermal ulcers | Monophasic pulsed v sham | 47 | 29.2 V, 29.2 mA, 132 μs, polarity reversed every 3 days then daily reversal with 64 pps | 30 min twice daily for 4 weeks | 1 Reduction in wound size. Wound area reduction ES 66% vs. sham 33% (p < 0.02) |
| Gentzkow [51] | Prospective | Stage III + IV pressure ulcers | Monophasic pulsed | 61 | 128 pps, 35 mA | 30 min twice daily | 4 Complete healing achieved in 23% |
| Baker [52] | Prospective | Open diabetic ulcers | Asymmetric biphasic vs. symmetric biphasic | 80 | Not stated | Until ulcers healed | 4 60% enhanced healing with asymmetric ES |
| Franek [53] | RCT | Pressure ulcers | High-voltage pulsed v sham | 50 | 100 V, 100 μs, 100 Hz | 50 min, once daily 5 days a week for 6 weeks | 1 Improved healing rate |
| Griffin [54] | RCT | Pressure ulcers | High-voltage pulsed v sham | 17 | 200 V, 100 pps, -ve cathode applied | 1 h daily for 20 days | 1 Significant increase in healing rate |
| Houghton [55] | RCT | Pressure ulcers | High-voltage pulsed v sham | 34 | 50–100 V, 50 μs, 10–100 Hz, polarity alternated | 8 h daily for 3 months | 1 Improvement in wound appearance and stimulated healing with ES |
| Peters [56] | RCT | Diabetic foot ulcers | High-voltage pulsed v sham | 40 | 50 V, 100 μs | 8 h daily for 12 weeks | 1 Enhanced wound healing when used with standard wound care |
| Houghton [57] | RCT | Chronic leg ulcers | High-voltage pulsed v sham | 27 | 150 V, 100 μs, 100 Hz | 3 times weekly for 4 weeks | 1 Accelerated wound closure. Wound area reduction ES 44% vs. sham 16% |
| Burdge [58] | Retrospective | Chronic diabetic wounds | High-voltage pulsed | 30 | <140 V, 90–100 μs, 55.19 Hz | 45 min sessions, 3 times weekly until healed approx. 16 weeks | 4 Improved healing |
| Goldman [59] | RCT | Ischemic wounds | High-voltage pulsed v sham | 8 | 100 pps, 360 V, -ve polarity | 1 h daily for 14 weeks | 2 Increased vasodilation and dermal capillary formation |
| Ahmad [60] | RCT | Pressure ulcers | High-voltage pulsed v sham | 60 | 100–175 V, 50 μs, 120 Hz | Group 1: 45 min, Group 2: 60 min, Group 3: 120 min; daily for 5 weeks | 1 Improved healing with ES |
| Direct Current | |||||||
| Gault [61] | RCT | Ischemic ulcers | LIDC v sham | 12 | Not stated | Until healed | 1 LIDC group healed twice as fast as control |
| Adunsky [59] | RCT | Pressure ulcers | DC | 63 | Not stated | 8 weeks | 1 DC useful combined with standard wound care |
| Carley [46] | Retrospective | Sacral/below knee ulcers | LIDC | 30 | 300–500 μA for normally innervated and 500–700 μA for denervated skin | 2 h, 5 days a week for 5 weeks | 3 LIDC improved healing. Wound are reduction ES 89% vs. control 37% (p < 0.01) |
| Wirsing [42] | Controlled | Diabetic leg and foot ulcers | Wireless LIDC | 47 | 1.5 μA | 2–3 times weekly, 45–60 min sessions, for 8 weeks | 2 Significantly accelerated healing |
| Wood [62] | Placebo controlled | Chronic decubitus ulcers | Pulsed LIDC | 74 | 300–600 μA | 8 weeks | 1 Fibroblast and keratinocytes growth enhanced. Increased healing rate |
| Transcutaneous Electrical Nerve Stimulation | |||||||
| Nolan [63] | Case study | Healthy skin | TENS | 1 | Not stated | 20 minutes | 5 Does not induce increased skin temperature |
| Cramp [29] | RCT | Over median nerve | TENS | 30 | High frequency: 110 Hz, 200 μsLow frequency: 4 Hz, 200 μs | 15 minutes | 1 No difference in skin temperature and blood flow |
| Simpson [64] | RCT | Limb ischemia | TSE | 8 | Not stated | 1 hour daily for one week, then a week off and repeated for third week | 1 No improvement in pain or microcirculation |
| Cramp [28] | RCT | Health volunteers | TENS | 30 | High frequency: 110 Hz, 200 μsLow frequency: 4 Hz, 200 μs | 15 minutes | 1 Local increase in blood flow |
| Wikstrom [65] | Controlled | Blister wound | TENS | 9 | High frequency: 100 Hz. Low frequency: 2 Hz | 45 minutes | 2 Stimulated perfusion |
| Frequency Rhythmic Electrical Modulation System | |||||||
| Jankovic [66] | RCT | Leg ulcers | FREMS v control | 35 | 300 V, 1000 Hz, 10–40 μs, 100–170 μA | 40 min daily, 5 days a week for 3 weeks | 1 Accelerated ulcer healing and reduced painWound area reduction ES 82% vs. control 46% |
| Santamato [67] | RCT | Venous ulcers | FREMS v control | 20 | Not stated | 5 days a week for 3 weeks | 1 Reduced pain and area of ulcers |
| Biofeedback Electrical Stimulation | |||||||
| Ud-Din [68] | Case-series | Raised dermal scars | Biofeedback | 18 | 0.004 mA, 20–80 V, 60 Hz | Until resolved | 4 Improved scar symptoms |
| Perry [48] | Case-series | Raised dermal scars | Biofeedback | 19 | 0.004 mA, 20–80 V, 60 Hz | Until resolved | 4 Improved scar symptoms |
| Ud-Din [43] | Controlled | Acute biopsy wounds | Biofeedback | 20 | 0.004 mA, 20–80 V, 60 Hz | 2 weeks | 2 Increased blood flow and haemoglobin levels |
| Bioelectric Dressings | |||||||
| Blount [41] | Case-series | Skin graft donor sites | Bioelectric dressing | 13 | 2–10 mV, 0.6–0.7 V, 10 μA | 1 month | 4 Faster healing and improved scarring |
| Hampton [69] | Case study | Leg ulcer | Bioelectric dressing | 1 | Not stated | Until healed | 5 Improved healing |
| Hampton [70] | Case study | Pressure ulcer | Bioelectric dressing | 1 | Not stated | 12 weeks | 5 Complete healing achieved |
3.1. Pulsed Current
Pulsed current (PC) is the unidirectional or bidirectional flow of electrons or ions, and has two waveforms: monophasic or biphasic [49]. Monophasic PC can also be described as low-voltage [52] and high voltage [53,71]. Biphasic PC is bidirectional and its waveform can be asymmetric or symmetric. PC is able to mimic the physiological endogenous current [49]. PC is delivered to the wound tissues by conductive coupling with a hydrogel or moist gauze filling the defect and the electrodes of appropriate polarity placed on top [49]. The majority of studies which used pulsed current are unidirectional.
Low voltage PC (LVPC) devices deliver continuous DC and monophasic and biphasic waveforms of longer durations and lower voltages (20–35 V) [49]. A number of clinical studies used an LVPC device named woundEL® and reported beneficial outcomes when using this for the treatment of ulcers [50,51,72]. The parameters used were: a duration of 132 microseconds and 64 pulses per second.
High-voltage pulsed current (HVPC) employs a monophasic pulsed current where the pulses are delivered in doubles. Each pulse is of short duration (less that 200 micro seconds) and it has a high peak voltage (150–500 V). HVPC is typically delivered by a device with both negative and positive electrodes either placed on the wound site or proximally on the skin [49]. This application has been used in wound healing, pain relief and oedema resolution [54,55]. A randomized controlled trial (LOE-1) conducted by Peters et al. studied 40 patients with diabetic foot ulcers for 12 weeks [56]. Patients were randomized to receive HVPC or sham therapy. Patients received 20 minutes of ES every hour for 8 hours each day over the 12-week study. Most patients healed in the ES group (65% compared to the sham group 35%), but the difference was not significant (p = 0.058). However, when patient compliance was evaluated, patients that used the device at least three times a week were more likely to heal than patients that received sham therapy and patients who used ES 0, 1, or 2 times a week (p = 0.038) [56].
An RCT (LOE-1) by Houghton et al. involved 27 patients with 42 chronic leg ulcers (arterial, venous, chronic) which were assigned to either a placebo or treatment group [57]. HVPC was delivered at 150 V, 100 pps and 100 microsecond duration. Treatments lasted for 45 minutes, 3 times a week for 4 weeks. The treatment group wounds significantly reduced in size (44%) compared to the sham group (16%). However, the significant differences were not maintained at the 1-month follow-up period. A retrospective study (LOE-4) also demonstrated positive results using HVPC in 30 patients with chronic diabetic wounds [58]. Furthermore, an RCT (LOE-1) also used this modality versus sham therapy in the treatment of ischemic wounds over a 14-week period and showed that the area of the wounds decreased and microcirculation was improved [59].
An RCT (LOE-1) was conducted with 60 subjects who had chronic pressure ulcers. They were split into 4 groups; one control who received sham therapy and three groups who received HVPC for 45, 60, 120 minutes respectively daily for one week [60]. Wound surface area was measured at 0, 3 and 5 weeks and they noted a significant reduction in the groups who received HVPC for 60 min and 120 min. However, no significant differences were noted between the treatment groups.
It is evident, that it is practically impossible to standardize chronic wounds in these studies, as each wound is substantially different to the next. Additionally, the research designs and device parameters were not comparable across these studies. Therefore, further larger controlled trials are critical in order to determine the optimal dosage and mode of delivery of ES.
3.2. Direct Current
Continuous direct current is the unidirectional flow of charged particles, which flow for 1 second or longer, and is produced by batteries, thermo couplings and solar cells [73]. The length of time the current flows has been known to cause irritation and pH changes to the skin [74]. Pulsed direct current is a monophasic pulsed waveform which flows from 1 ms to 1second [49]. Direct current is able to mimic the physiological endogenous current [49]. In wound care, a low-intensity direct current (20–1000 microamps) is used to avoid damaging healthy tissue [61]. Low-intensity direct current has been shown to promote chronic wound healing by two mechanisms: galvanotaxis (by stimulating the migration of fibroblasts and keratinocytes [75] and its antimicrobial effect [61].
A study by Adunsky et al. (LOE-2), 38 patients with pressure ulcers were distributed equally between shams and treatment with DC application of electrical stimulation for 8 weeks [76]. The primary outcome was percentage change in the wound area, with the results showing that wound area reduction was 31% (ES group) vs. 4% (sham group) (p = 0.09). The relatively small sample size may have contributed to the lack of significance.
Gault et al. conducted an 8-week trial (LOE-2) using continuous LIDC to treat 76 patients with 106 ischaemic skin ulcers [61]. They applied the negative electrode directly onto the wound for three days in order to debride necrotic tissue. The current used was 200–800 microamps for two hours, three times daily. Six patients had bilateral ulcers and therefore one ulcer was treated as a control. Forty-eight of the 100 ulcers healed completely. In the patients who had controls, healing rate for the treated ulcers was 30% compared to 14.7% in the controls. Nevertheless, a larger control group would be needed for more meaningful results. In a controlled clinical trial (LOE-2) [46], 15 unspecified wounds were treated with continuous LIDC and 15 with conservative treatment for 5 weeks. The current was 300–700 microamps for two hours in two sessions per day, five days a week. The mean healing rate for the treatment group was 89%, compared to 45% for the control group. However, limitations of this study were that despite mentioning that they had conducted a follow-up, no details were reported of this. Furthermore, the randomization process was not rigorous; participants were paired according to their age, diagnosis, wound location and aetiology with each pair placed in one of two groups. Additionally, there was no blinding as this was not possible. A recent study (LOE-3) used a wireless micro current stimulation device for the treatment of 47 patients with leg and diabetic foot ulcers [42]. This was applied 2 or 3 times a week for 60 minutes per session combined with standard wound care. They demonstrated complete healing within 3 months for the majority of cases. This device is contactless and pain-free and different wounds can be treated at the same time.
Intermittent low-intensity direct current delivers a current, which goes up to 29.2 milliamps and then down to zero [73]. A double-blind multi-centred controlled trial (LOE-1) [62] evaluated the effect of this treatment on 43 patients with stage II and III pressure ulcers compared to 31 placebo (sham intermittent LIDC) patients. The current used was 300–600 milliamps. Twenty-five ulcers in the treatment group healed completely within 8 weeks (p < 0.001), compared to 4 in the control group, which had healed up to 80%. These positive results indicate a beneficial effect of intermittent LIDC, however, there was no report of randomization and no explanation for the difference in size of the two groups. Feedar et al. conducted a double-blind multi-centred RCT (LOE-1) using intermittent LIDC with 47 patients with 50 ulcers, which were split into control and treatment groups [50]. The current was applied at 35 milliamps, which was applied for 30 minutes, twice daily on a daily basis. They showed a statistically significant difference between the groups; the mean healing rate was 56% in the treatment group compared to 33% in the control group (p < 0.02).
3.3. Transcutaneous Electrical Nerve Stimulation
Transcutaneous Electrical Nerve Stimulation (TENS) is a low-frequency, pulsed electrical current transmitted by electrodes through the skin surface [77] to stimulate the peripheral nerves to produce various physiological effects [78]. The biphasic pulses are most commonly used [79,80]. TENS is considered to be one of the most common therapeutic modalities used in clinical practice for the relief of chronic and acute pain [78]. Some authors have observed that, in addition to its analgesic effects, TENS can also alter skin temperature and increase blood flow [40]. This observation has lead to various studies investigating the effect on the peripheral vascular system and how this facilitates tissue repair [81]. There are disagreements in the literature with regard to the increase in blood flow and skin temperature. Some studies have shown that TENS significantly increases skin temperature with low- (2 Hz to 4 Hz) [31] and high-frequency (75 Hz to 100 Hz) TENS [63], and in local blood flow [31]. However, some studies have not shown any significant increase of blood flow [29] and temperature [64] with the use of TENS. Interestingly, some studies suggested that when applied at the same intensity, low-frequency TENS enhanced blood flow levels more than high-frequency TENS [28,65].
3.4. Frequency Rhythmic Electrical Modulation System
Frequency rhythmic electrical modulation system (FREMS) is a form of transcutaneous electrotherapy using ES that automatically varies the pulse, frequency, duration, and voltage [46]. Two RCTs have been conducted utilising this for the treatment of chronic leg ulcers in order to improve wound healing [66,67]. The first RCT (LOE-1) recruited 35 patients and divided them into two groups [66]. One group received FREMS treatment for 2 months and the control group of no treatment. Their results showed that ulcer improvement in the treatment group was significantly higher than compared to the control group. However, a larger sample size would be needed for future studies. Another RCT (LOE-1) used FREMS treatment in 20 older patients with chronic and painful venous leg ulcers [67]. One group of 10 patients received FREMS and a topical treatment, whilst the control group received topical treatment only. 15 treatments were performed over a period of 3 weeks. They showed that there was a statistically significant decrease in ulcer area when treated with FREMS compared to the control group. Again, the small sample size means that further studies are necessary to investigate this treatment more robustly.
3.5. Biofeedback ES
An electro biofeedback device termed the Fenzian system (Fenzian Ltd, Hungerford, UK), where its waveform was deciphered and shown to resemble degenerate waves, has been used successfully in the treatment of symptoms in keloid and hypertrophic scarring and in accelerating the process of acute wound healing in the skin [34,47,48,82]. It is a transcutaneous low intensity device, which detects changes in skin impedance. This device forms part of an electrobiofeedback link with the individual’s normal physiological repair. This modality follows the theory that the normal electrical potential of skin forms a global electrical network reflecting the underlying neurological activity through changes in skin impedance [82]. Using a concentric electrode the device detects the skin’s electrical impedance and adjusts the outgoing microcurrent electrical biofeedback impulses [82]. The device delivers 0.004 milliamps, 20–80 V, has a frequency default of 60 Hz and impulses which last approximately six-hundredth of a second.
It has been used successfully to alleviate the symptoms for pain, pruritus and inflammation in two case series (LOE-4) on raised dermal scars [48,68]. It is postulated that this treatment can be beneficial in the treatment of abnormal skin scarring as it may negate the need for long-term pain medications. Furthermore, a clinical trial (LOE-2) was conducted involving multiple temporal punch biopsies treated with biofeedback ES and demonstrated increased blood flow and haemoglobin levels in acute cutaneous wounds (on day 14 post-wounding) created in 20 human volunteers compared to controls which had not received ES [43]. This treatment modality accelerated the rate of cutaneous wound healing in all cases as evidenced by gene and protein studies showing up-regulated angiogenesis and down-regulated inflammation [34]. Additional larger randomised controlled trials are required to investigate this treatment further to identify if this could be beneficial in patients with chronic wounds.
3.6. Bioelectric Dressings
Bioelectric dressings are emerging as a useful method of delivering ES to the wound site. However, studies of these specific modalities are lacking. Procellera® is a woven metallic bandage with embedded microbatteries, which is used as a dressing for partial or full thickness wounds. The mechanism of action is delivery of ES to the wound site. It produces a low voltage of 2–10 mV by microbatteries of Ag and Zn metals which are inside a woven material and are activated by the moisture in the wound delivers 0.6–0.7 V at 10 microamps. In a study by Blount et al. [41], 13 patients had skin grafting and the Procellera® dressing was applied to half of the donor site area. They noted improved healing, scarring and patient subjective outcomes. However, a larger trial is required to substantiate these results further. Another bioelectric wound dressing, named the PosiFect RD® DC device, has been used in treating pressure and venous ulcers [69,70]. This dressing contains a miniature electrical circuit delivering a microcurrent to the wound bed for a minimum of 48 hours and has shown promise in treating these chronic wounds.
4. Discussion
The reported studies demonstrate considerable variability in the parameters of ES application, leading to difficulty in generating sufficient evidence to support any one standard therapeutic approach. Most studies reported successful positive outcomes using ES to accelerate wound healing. Nonetheless, the differences in types of ulcerations or wounds, ES parameter settings and limited power of study design make synthesis of the results difficult and to draw conclusions as to an optimal mode of ES or type of ES device.
The level of evidence assigned for each study showed variations amongst the types of ES. The majority of LOE-1 were for HVPC, TENS, FREMS and DC. The available evidence (n = 4) suggests that HVPC is most beneficial for pressure ulcer treatment, with these LOE-1 studies demonstrating improved healing rates with the application of this modality. Despite a limited number of studies, HVPC has also shown positive results when used in diabetic (n = 2), ischaemic (n = 1) and chronic leg ulcers (n = 1). However, it is not apparent if other types of wounds such as acute wounds or venous ulcers would respond differently to this therapy. FREMS has been used in the treatment of leg ulcers and have shown promising results by accelerating ulcer healing and the area of the ulcer. Nevertheless, there were only two RCTs (LOE-1); thus, it is difficult to identify whether this treatment is effective in other wound types. Additionally, HVPC and DC stimulation demonstrate higher levels of evidence when compared to biofeedback ES and bioelectric dressings, which are based on case series and case study reports. There is limited clinical evidence regarding ES application for acute wounds in comparison to chronic wounds.
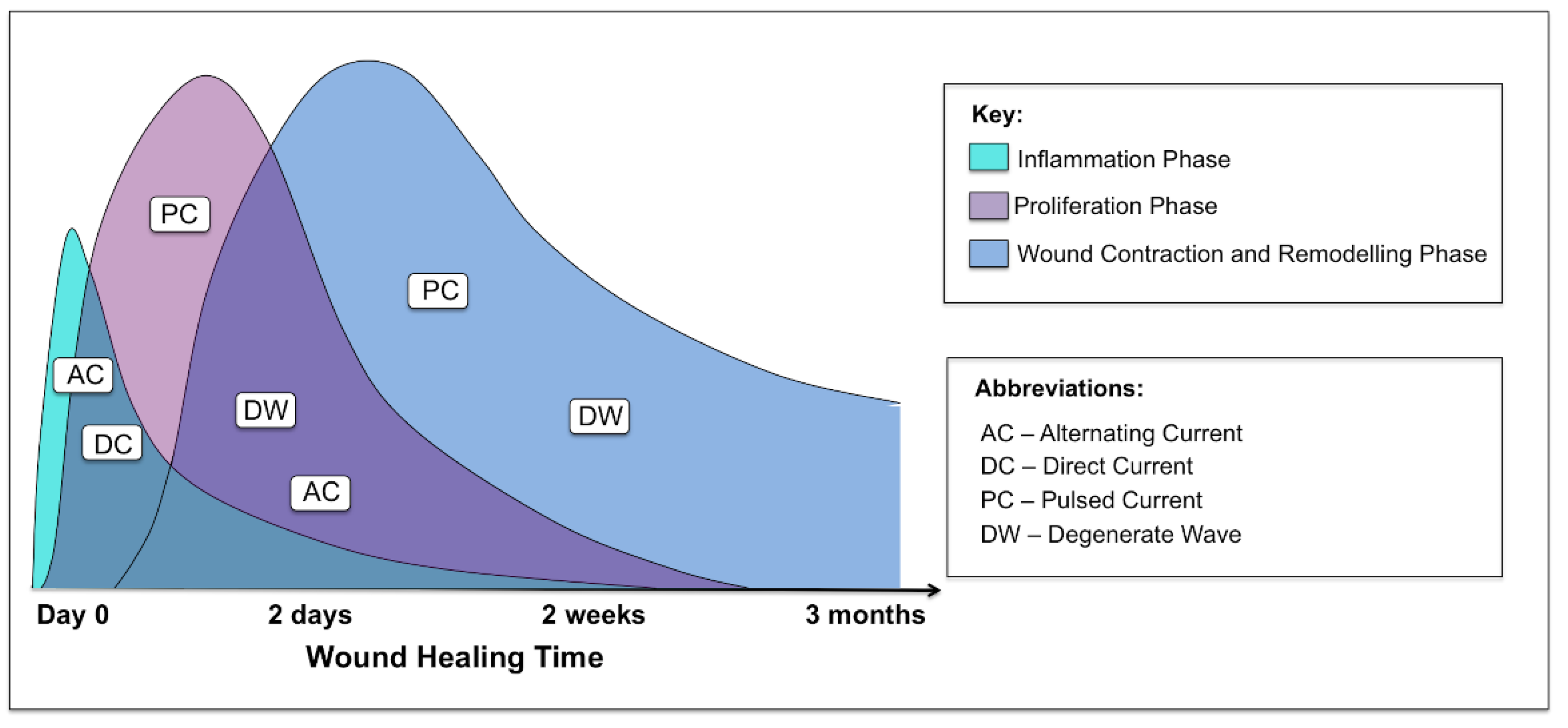
ES has mainly been evaluated in pressure ulcers, venous ulcers, vascular ulcers and diabetic foot wounds. One of the challenges in interpreting these data is the variation in outcome measurements, type of ES, and how the therapy was dosed in the trials. Most of the studies were small and many had a short treatment period and limited follow-up. In addition, many of the studies did not use complete wound healing (i.e., complete wound closure) as the primary outcome. Due to the short duration of the studies, change in wound area was often used instead of wound closure. As it is difficult to standardize chronic wounds, it is important to look at acute wound studies for the effects ES has on these wounds. There was a lack of human controlled trials investigating the role of ES in acute cutaneous wounds. Biofeedback ES has been shown to be an effective method for enhancing cutaneous wound healing. A significantly increased blood flow was noted on day 14 in a controlled study [43]. Nevertheless, based on the findings to date, it remains difficult to ascertain which phases of wound healing this particular device would be optimal for. Importantly, it is of note that not all applications and modalities of ES have an effect on all phases of wound healing (Figure 8).
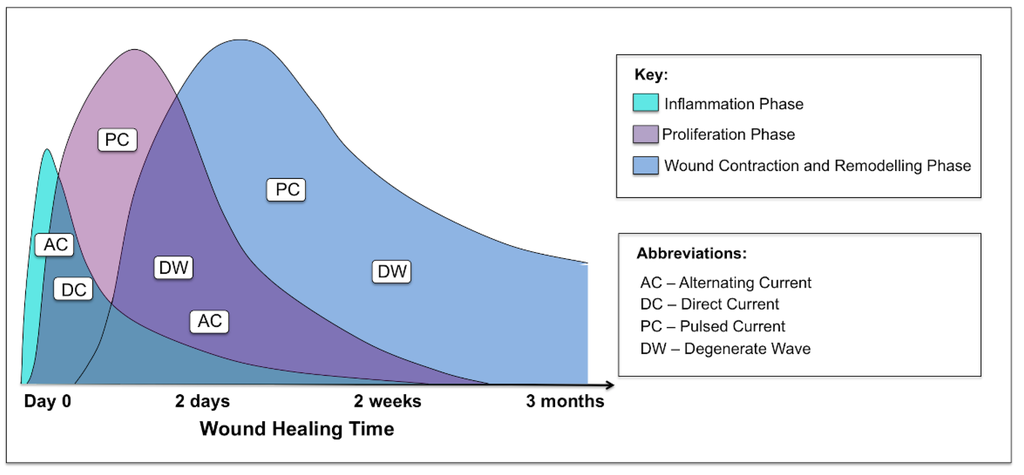
Figure 8.
Graphical representation of the three phases of acute cutaneous wound healing and where the different waveforms of electrical stimulation are effective in each phase: inflammatory, proliferative and remodelling.
Figure 8.
Graphical representation of the three phases of acute cutaneous wound healing and where the different waveforms of electrical stimulation are effective in each phase: inflammatory, proliferative and remodelling.
The majority of studies used unidirectional ES with the electrodes placed in or around the wound site. Additionally, some studies have suggested that the application of certain polarities at specific stages of wound healing may accelerate wound closure [60]. Moreover, it has been shown that electrical stimulation induces the migration of keratinocytes, which contribute to the skin’s first line of defence against pathogens, a key process in wound healing [69]. In a study by Guo et al, they showed that after a one-hour period the physiological electrical field enabled human dermal fibroblasts to begin migrating toward the anode, in a direction opposite to that of keratinocytes, which migrate toward the cathode [83]. This is also suggested in a study by Ahmad et al. [60], where they identified that applying the anode in the wound could enhance wound healing. A further study was conducted to see which was more appropriate for wound repair: anodal or cathodal microamperage direct current electrical stimulation. Application of continuous microamperage direct current is a plausible method of treatment due to the inherent potential difference between a wound and its surrounding intact skin. The study concluded that anodal microamperage direct current is more effective than cathodal microamperage direct current in healing skin wounds as it decreases the wound surface area faster, allowing for faster wound healing than cathodal electrical stimulation [84].
The majority of studies evaluated the effects of ES in patients with wounds of various aetiologies, with many having their chronic wound for a variable number of years. It is pertinent to understand when is the best time to apply ES. It may be necessary to commence treatment as soon as the wound occurs, and the exact frequency to treat with. Additionally, it may be necessary to change a chronic wound into an acute wound and then commence ES therapy [29]. When comparing the device parameters for similar wound types, it is noted that there are some variations. Pressure ulcers are a common wound type, which is used in a number of studies in particular with HVPC. Franek et al. [53] used parameters set at 100 V, 100 microseconds, 100 Hz for 50 minutes once daily. Griffin et al. [54] used a voltage of 200 V, Houghton et al. [55] used between 50 and 100 V and Ahmad et al. [60] applied 100–175 V. Therefore, the voltages applied in these studies tend to vary. Additionally, the length of time ES is to be performed is approximately similar across some studies; 50 minutes [53] and 60 minutes [54], whilst Griffin et al. [54] applied HVPC over a period of 8 hours per day. Ahmad et al. [60] compared different durations of treatment over three groups; 45 minutes, 60 minutes and 120 minutes. They noted that 60 and 120 minute groups when applied for 7 days a week for 5 weeks demonstrated optimal healing compared to the 45 minute group. In diabetic wounds, similar parameters were used for HVPC across some studies [51,71]. These studies used an interphase interval of 100 microseconds and a voltage between 50 and 140 V; however, duration of treatment times varied. Further studies comparing the parameters for different wound types and types of ES would be useful to identify the optimal settings for each device in specific wounds.
Koel et al. [85] summarized the results of effect studies with ES as an additional treatment to standard wound care. They used forest plots and identified the healing rate, which was expressed as the percentage area reduction within 4 weeks of treatment. Their results showed that unidirectional ES and standard wound care increases the reduction in wound surface area by 30.8%. In pressure ulcers, the results increased to 42.7% by 4 weeks. Additionally, they noted that unidirectional ES is most beneficial for pressure ulcers, whereas venous leg ulcers and diabetic foot ulcers have had positive results with bidirectional ES.
ES therapy is considered safe and easy to use, as no device-related complications or adverse effects have been reported to date. ES application is relatively cost effective compared to other comparative treatments. In those ES modalities which are administered by a practitioner, this can be performed by a single experienced practitioner and there is often no pain associated with the treatments.
Some authors suggested that compliance might be a factor that affects cutaneous wound healing in ES studies [56,69]. However, in most studies, therapy was provided in a hospital or clinic setting, therefore, patients attending clinic appointments determined the main measure of compliance. The study by Peters et al. [56] was the only study that provided an ES device for patients to use at home and they recorded the number of hours the device was used. There was no significant difference in the compliance rates between the two treatment groups. There was a trend demonstrating a dose response with ES. A higher proportion of wounds healed in compliant patients in the ES treatment group (71%), non-compliant patients in the ES treatment group (50%), compliant patients in the sham group (39%), and non-compliant patients in the sham group (29%) [56].
In summary, despite variations in the type of current, duration, and dosing of ES, the majority of studies showed a significant improvement in wound area reduction or wound healing compared to the standard of care or sham therapy as well as improved local perfusion. Furthermore, no device-related complications or adverse effects have been reported in the existing literature, therefore, indicating that the therapy is safe and easy to use. Additionally, as ES decreases bacterial infection, increases local perfusion and accelerates wound healing, it targets these main factors of significance in wound management. There are several questions which remain unanswered, including, the optimal method of delivering ES, identifying which wound types respond better to treatment and the ideal anatomical location, frequency, duration and time to commence the application of ES for each wound type. Overall, the evidence to date infers that further clinical trials are much needed to aid in better understanding the optimal dosing, timing and type of ES to be used and to optimize the effectiveness and appropriate clinical application. No doubt, this is likely to be achieved in the future by comparing the effects of different ES modalities, treatment durations and frequencies on the rate and quality of healing in similar cutaneous wounds.
Author Contributions
Sara Ud-Din wrote the paper. Ardeshir Bayat wrote and edited the paper and is the senior author.
Conflict of Interest
The authors declare no conflict of interest.
References
- Monaco, J.L.; Lawrence, W.T. Acute wound healing: An overview. Clin. Plast. Surg. 2003, 30, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Greaves, N.S.; Iqbal, S.A.; Baguneid, M.; Bayat, A. The role of skin substitutes in the management of chronic cutaneous wounds. Wound Repair Regen. 2013, 21, 194–210. [Google Scholar] [CrossRef] [PubMed]
- Fonder, M.A.; Lazarus, G.S.; Cowan, D.A.; Aronson-Cook, B.; Kohli, A.R.; Mamelak, A.J. Treating the chronic wound: A practical approach to the care of nonhealing wounds and wound care dressings. J. Am. Acad. Dermatol. 2008, 58, 185–206. [Google Scholar] [CrossRef] [PubMed]
- Singh, A.; Halder, S.; Chumber, S.; Misra, M.C.; Sharma, L.K.; Srivastava, A. Meta-analysis of randomized controlled trials on hydrocolloid occlusive dressing versus conventional gauze dressing in the healing of chronic wounds. Asian J. Surg. 2004, 27, 326–332. [Google Scholar] [CrossRef] [PubMed]
- Posnett, J.; Franks, P.J. The burden of chronic wounds in the UK. Nurs. Times 2008, 104, 44–45. [Google Scholar] [PubMed]
- Mustoe, T.A.; O’Shaughnessy, K.; Kloeters, O. Chronic wound pathogenesis and current treatment strategies: A unifying hypothesis. Plast. Reconstr. Surg. 2006, 117, 35S–41S. [Google Scholar] [CrossRef] [PubMed]
- Vileikyte, L. Diabetic foot ulcers: A quality of life issue. Diabetes Metab. Res. Rev. 2001, 17, 246–249. [Google Scholar] [CrossRef] [PubMed]
- Amsler, F.; Willenberg, T.; Blättler, W. In search of optimal compression therapy for venous leg ulcers: A meta-anlysis of studies comparing divers bandages with specially designed stockings. J. Vasc. Surg. 2009, 50, 668–674. [Google Scholar] [CrossRef] [PubMed]
- Carter, J.M.; Tingley-Kelley, K.; Warriner, I.R.A. Silver treatments and silver-impregnated dressings for the healing of leg wounds and ulcers: A systematic review and meta-analysis. J. Am. Acad. Dermatol. 2010, 63, 668–679. [Google Scholar] [CrossRef] [PubMed]
- Armstrong, D.G.; Marston, W.A.; Reyzelman, A.M.; Kirsner, R.S. Comparison of negative pressure wound therapy with an ultraportable mechanically powered device vs. traditional electrically powered device for the treatment of chronic lower extremity ulcers: A multicenter randomized- controlled trial. Wound Repair Regen. 2011, 19, 173–180. [Google Scholar] [CrossRef] [PubMed]
- Cullum, N.A.; Al-Kurdi, D.; Bell-Syer, S.E. Therapeutic ultrasound for venous leg ulcers. Cochrane Database Syst. Rev. 2010. [Google Scholar] [CrossRef]
- Dumville, J.C.; Worthy, G.; Soares, M.O.; Bland, J.M.; Cullum, N.; Dowson, C.; Iglesias, C.; McCaughan, D.; Mitchell, J.L.; Nelson, E.A.; et al. VenUS II: A randomised controlled trial of larval therapy in the management of leg ulcers. Health Technol. Assess. 2009, 13, 1–206. [Google Scholar] [CrossRef] [PubMed]
- Greaves, N.S.; Benatar, B.; Baguneid, M.; Bayat, A. Single-stage application of a novel decellularized dermis for treatment-resistant lower limb ulcers: Positive outcomes assessed by SIAscopy, laser perfusion, and 3D imaging, with sequential timed histological analysis. Wound Repair Regen. 2013, 21, 813–822. [Google Scholar] [CrossRef] [PubMed]
- Snyder, R.J.; Kirsner, R.S.; Warriner, R.A.; Lavery, L.A.; Hanft, J.R.; Sheehan, P. Consensus recommendations on advancing the standard of care for treating neuropathic foot ulcers in patients with diabetes. Ostomy Wound Manag. 2010, 56, S1–S24. [Google Scholar]
- Suh, H.; Petrofsky, J.; Fish, A.; Hernandez, V.; Mendoza, E.; Collins, K.; Yang, T.; Abdul, A.; Batt, J.; Lawson, D. A new electrode design to improve outcomes in the treatment of chronic non-healing wounds in diabetes. Diabetes Technol. Ther. 2009, 11, 315–322. [Google Scholar] [CrossRef] [PubMed]
- Petrofsky, J.; Lawson, D.; Prowse, M.; Suh, H.J. Effects of a 2-, 3- and 4-electrode stimulator design on current dispersion on the surface and into the limb during electrical stimulation in controls and patients with wounds. J. Med. Eng. Technol. 2008, 32, 485–497. [Google Scholar] [CrossRef] [PubMed]
- Jünger, M.; Arnold, A.; Zuder, D.; Stahl, H.W.; Heising, S. Local therapy and treatment costs of chronic, venous leg ulcers with electrical stimulation (Dermapulse): A prospective, placebo controlled, double blind trial. Wound Repair Regen. 2008, 16, 480–487. [Google Scholar] [CrossRef]
- Isseroff, R.R.; Dahle, S.E. Electrical stimulation therapy and wound healing: Where are we now? Adv. Wound Care 2012, 1, 238–243. [Google Scholar] [CrossRef]
- Reger, S.I.; Hyodo, A.; Negami, S.; Kambic, H.E.; Sahgal, V. Experimental wound healing with electrical stimulation. Artif. Organs 1999, 23, 460–462. [Google Scholar] [CrossRef] [PubMed]
- Gagnier, K.A.; Manix, N.; Baker, L. The effects of electrical stimulation on cutaneous oxygen supply in paraplegics. Phys. Ther. 1988, 68, 835–839. [Google Scholar]
- Peters, E.J.; Armstrong, D.G.; Wunderlich, R.P.; Bosma, J.; Stacpoole-Shea, S.; Lavery, L.A. The benefit of electrical stimulation to enhance perfusion in persons with diabetes mellitus. J. Foot Ankle Surg. 1998, 37, 396–400. [Google Scholar] [CrossRef] [PubMed]
- Rouabhia, M.; Park, H.; Meng, S.; Derbali, H.; Zhang, Z. Electrical stimulation promotes wound healing by enhancing dermal fibroblast activity and promoting myofibroblast transdifferentiation. PLoS One 2013, 8, e71660. [Google Scholar] [CrossRef] [PubMed]
- Young, S.; Hampton, S.; Tadej, M. Study to evaluate the effect of low-intensity pulsed electrical currents on levels of oedema in chronic non-healing wounds. J. Wound Care 2011, 20, 370–373. [Google Scholar] [CrossRef]
- Daeschlein, G.; Assadian, O.; Kloth, L.C.; Meinl, C.; Ney, F.; Kramer, A. Antibacterial activity of positive and negative polarity low-voltage pulsed current (LVPC) on six typical gram-positive and gram-negative bacterial pathogens of chronic wounds. Wound Repair Regen. 2007, 15, 399–403. [Google Scholar] [CrossRef] [PubMed]
- Alvarez, O.M.; Mertz, P.M.; Smerbeck, R.V.; Eaglstein, W.H. The healing of superficial skin wounds is stimulated by external electrical current. J. Invest. Dermatol. 1983, 81, 144–148. [Google Scholar] [CrossRef] [PubMed]
- Bourguignon, G.J.; Bourguignon, L.Y. Electric stimulation of protein and DNA synthesis in human fibroblasts. FASEB J. 1987, 1, 398–402. [Google Scholar] [PubMed]
- Bassett, C.A.L.; Herrmann, I. The effect of electrostatic fields on macromolecular synthesis by fibroblasts in vitro. J. Cell Biol. 1968, 9, 39. [Google Scholar]
- Cramp, A.F.L.; Gilsenan, C.; Lowe, A.S.; Walsh, D.M. The effect of high- and low-frequency transcutaneous electrical nerve stimulation upon cutaneous blood flow and skin temperature in healthy subjects. Clin. Physiol. 2000, 20, 150–157. [Google Scholar] [CrossRef] [PubMed]
- Cramp, A.F.; Noble, J.G.; Lowe, A.S.; Walsh, D.M. Transcutaneous electrical nerve stimulation (TENS): The effect of electrode placement upon cutaneous blood flow and skin temperature. Acupunct. Electrother. Res. 2001, 26, 25–37. [Google Scholar] [PubMed]
- Cramp, F.L.; McCullough, G.R.; Lowe, A.S.; Walsh, D.M. Transcutaneous electric nerve stimulation: The effect of intensity on local and distal cutaneous blood flow and skin temperature in healthy subjects. Arch. Phys. Med. Rehabil. 2002, 83, 5–9. [Google Scholar] [CrossRef] [PubMed]
- Kaada, B.; Olsen, E.; Eielsen, O. In search of mediators of skin vasodilation induced by transcutaneous nerve stimulation: III. Increase in plasma VIP in normal subjects and in Raynaud’s disease. Gen. Pharmacol. 1984, 15, 107–113. [Google Scholar] [CrossRef] [PubMed]
- Kaada, B.; Helle, K.B. In search of mediators of skin vasodilation induced by transcutaneous nerve stimulation: IV. In vitro bioassay of the vasoinhibitory activity of sera from patients suffering from peripheral ischemia. Gen. Pharmacol. 1984, 15, 115–122. [Google Scholar]
- Abram, S.E.; Asiddao, C.B.; Reynolds, A.C. Increased skin temperature during transcutaneous electrical stimulation. Anesth. Analg. 1980, 59, 22–25. [Google Scholar] [PubMed]
- Sebastian, A.; Syed, F.; Perry, D.; Balamurugan, V.; Colthurst, J.; Chaudhry, I.H.; Bayat, A. Acceleration of cutaneous healing by electrical stimulation: Degenerate electrical waveform down-regulates inflammation, up-regulates angiogenesis and advances remodelling in temporal punch biopsies in a human volunteer study. Wound Repair Regen. 2011, 19, 693–708. [Google Scholar] [CrossRef] [PubMed]
- Szuminky, N.J.; Albers, A.C.; Unger, P.; Eddy, J.G. Effect of narrow, pulsed high voltages on bacterial viability. Phys. Ther. 1994, 74, 660–667. [Google Scholar] [PubMed]
- Foulds, I.S.; Barker, A.T. Human skin battery potentials and their possible role in wound healing. Br. J. Dermatol. 1983, 109, 515–522. [Google Scholar] [CrossRef] [PubMed]
- McGinnis, M.E.; Vanable, J.W., Jr. Voltage gradients in newt limb stumps. Prog. Clin. Biol. Res. 1986, 210, 213–238. [Google Scholar] [PubMed]
- Barker, A.T.; Jaffe, L.F.; Vanable, J.W., Jr. The glabrous epidermis of cavies contains a powerful battery. Am. J. Physiol. 1982, 242, R358–R366. [Google Scholar] [PubMed]
- Thakral, G.; Lafontaine, J.; Najafi, B.; Talal, T.K.; Kim, P.; Lavery, L.A. Electrical stimulation to accelerate wound healing. Diabet. Foot Ankle 2013. [Google Scholar] [CrossRef]
- Atalay, C.; Yilmaz, K.B. The effect of transcutaneous electrical nerve stimulation on postmastectomy skin flap necrosis. Breast Cancer Res. Treat. 2009, 117, 611–614. [Google Scholar] [CrossRef] [PubMed]
- Blount, A.L.; Foster, S.; Rapp, D.A.; Wilcox, R. The use of bioelectric dressings in skin graft harvest sites: A prospective case series. J. Burn Care Res. 2012, 33, 354–357. [Google Scholar] [CrossRef] [PubMed]
- Wirsing, P.G.; Habrom, A.D.; Zehnder, T.M.; Friedli, S.; Blatti, M. Wireless micro current stimulation—An innovative electrical stimulation method for the treatment of patients with leg and diabetic foot ulcers. Int. Wound J. 2013. [Google Scholar] [CrossRef]
- Ud-Din, S.; Perry, D.; Giddings, P.; Colthurst, J.; Zaman, K.; Cotton, S.; Whiteside, S.; Morris, J.; Bayat, A. Electrical stimulation increases blood flow and haemoglobin levels in acute cutaneous wounds without affecting wound closure time: Evidenced by non-invasive assessment of temporal biopsy wounds in human volunteers. Exp. Dermatol. 2012, 21, 758–764. [Google Scholar] [CrossRef]
- Stillings, D. A survey of the history of electrical stimulation for pain to 1900. Med. Instrum. 1975, 9, 255–259. [Google Scholar] [PubMed]
- Kloth, L.C.; Feedar, J.A. Acceleration of wound healing with high voltage, monophasic, pulsed current. Phys. Ther. 1988, 68, 503–508. [Google Scholar]
- Carley, P.J.; Wainapel, S.F. Electrotherapy for acceleration of wound healing: Low intensity direct current. Arch. Phys. Med. Rehabil. 1985, 66, 443–446. [Google Scholar] [PubMed]
- Sebastian, A.; Syed, F.; McGrouther, D.A.; Colthurst, J.; Paus, R.; Bayat, A. A novel in vitro assay for electrophysiological research on human skin fibroblasts: Degenerate electrical waves downregulate collagen I expression in keloid fibroblasts. Exp. Dermatol. 2011, 20, 64–68. [Google Scholar] [CrossRef]
- Perry, D.; Colthurst, J.; Giddings, P.; McGrouther, D.A.; Morris, J.; Bayat, A. Treatment of symptomatic abnormal skin scars with electrical stimulation. J. Wound Care 2010, 19, 447–453. [Google Scholar]
- Kloth, L.C. Electrical stimulation technologies for wound healing. Adv. Wound Care 2014, 3, 81–90. [Google Scholar] [CrossRef]
- Feedar, J.A.; Kloth, L.C.; Gentzkow, G.D. Chronic dermal ulcer healing enhanced with monophasic pulsed electrical stimulation. Phys. Ther. 1991, 71, 639–649. [Google Scholar] [PubMed]
- Gentzkow, G.D.; Alon, G.; Taler, G.A.; Eltorai, I.M.; Montroy, R.E. Healing of refactory stage III and IV pressure ulcers by a new electrical stimulation device. Wounds 1993, 5, 160–172. [Google Scholar]
- Baker, L.L.; Chanbers, R.; Demuth, S.K.; Villar, F. Effects of electrical stimulation on wound healing in patients with diabetic ulcers. Diabetes Care 1997, 20, 405–412. [Google Scholar] [CrossRef] [PubMed]
- Franek, A.; Kostur, R.; Polak, A.; Taradaj, J.; Szlachta, Z.; Blaszczak, E.; Dolibog, P.; Dolibog, P.; Koczy, B.; Kucio, C. Using high voltage electrical stimulation in the treatment of recalcitrant pressure ulcers: Results of a randomised, controlled clinical study. Ostomy Wound Manag. 2012, 58, 30–44. [Google Scholar]
- Griffin, J.W.; Tooms, R.E.; Mendius, R.A.; Clifft, J.K.; Vander Zwaag, R.; el-Zeky, F. Efficacy of high voltage pulsed current for healing of pressure ulcers in patients with spinal cord injury. Phys. Ther. 1991, 71, 433–442. [Google Scholar] [PubMed]
- Houghton, P.E.; Campbell, K.E.; Fraser, C.H.; Harris, C.; Keast, D.H.; Potter, P.J.; Hayes, K.C.; Woodbury, M.G. Electrical stimulation therapy increases rate of healing of pressure ulcers in community-dwelling people with spinal cord injury. Arch. Phys. Med. Rehabil. 2010, 91, 669–678. [Google Scholar] [CrossRef] [PubMed]
- Peters, E.J.; Lavery, L.A.; Armstrong, D.G.; Fleischli, J.G. Electric stimulation as an adjunct to heal diabetic foot ulcers: A randomized clinical trial. Arch. Phys. Med. Rehabil. 2001, 82, 721–725. [Google Scholar] [CrossRef] [PubMed]
- Houghton, P.E.; Kincaid, C.B.; Lovell, M.; Campbell, K.E.; Keast, D.H.; Woodbury, M.G.; Harris, K.A. Effect of electrical stimulation on chronic leg ulcer size and appearance. Phys. Ther. 2003, 83, 17–28. [Google Scholar] [PubMed]
- Burdge, J.J.; Hartman, J.F.; Wright, M.L. A study of HVPC as an adjunctive therapy in limb salvage for chronic diabetic wounds of the lower extremity. Ostomy Wound Manag. 2009, 55, 30–38. [Google Scholar]
- Goldman, R.; Rosen, M.; Brewley, B.; Golden, M. Electrotherapy promotes healing and microcirculation of infrapopliteal ischemic wounds: A prospective pilot study. Adv. Skin Wound Care 2004, 17, 284–294. [Google Scholar] [CrossRef] [PubMed]
- Ahmad, E.T. High-voltage pulsed galvanic stimulation: Effect of treatment duration on healing of chronic pressure ulcers. Mediterr. Counc. Burn. Fire Disasters 2008, 21, 124–128. [Google Scholar]
- Gault, W.R.; Gatens, P.F., Jr. Use of low intensity direct current in management of ischemic skin ulcers. Phys. Ther. 1976, 56, 265–269. [Google Scholar] [PubMed]
- Wood, J.M.; Evans, P.E., 3rd; Schallreuter, K.U.; Jacobson, W.E.; Sufit, R.; Newman, J.; White, C.; Jacobson, M. A multicenter study on the use of pulsed low-intensity direct current for healing chronic stage II and stage III decubitus ulcers. Arch. Dermatol. 1993, 129, 999–1009. [Google Scholar] [CrossRef] [PubMed]
- Nolan, M.F.; Hartsfield, J.K.; Witters, D.M.; Wason, P.J. Failure of transcutaneous electrical nerve stimulation in the conventional and burst modes to alter digital skin temperature. Arch. Phys. Med. Rehabil. 1993, 74, 182–187. [Google Scholar] [PubMed]
- Simpson, K.H.; Ward, J. A Randomized, double-blind, crossover study of the use of transcutaneous spinal electroanalgesia in patients with pain from chronic critical limb ischemia. J. Pain Symptom Manag. 2004, 28, 511–516. [Google Scholar]
- Wikstrom, S.O.; Svedman, P.; Svensson, H.; Tanweer, A.S. Effect of transcutaneous nerve stimulation on microcirculation in intact skin and blister wounds in healthy volunteers. Scand. J. Plast. Reconstr. Surg. Hand Surg. 1999, 33, 195–201. [Google Scholar] [CrossRef] [PubMed]
- Janković, A.; Binić, I. Frequency rhythmic electrical modulation system in the treatment of chronic painful leg ulcers. Arch. Dermatol. Res. 2008, 300, 377–383. [Google Scholar] [CrossRef] [PubMed]
- Santamato, A.; Panza, F.; Fortunato, F.; Portincasa, A.; Frisardi, V.; Cassatella, G.; Valente, M.; Seripa, D.; Ranieri, M.; Fiore, P. Effectiveness of the frequency rhythmic electrical modulation system for the treatment of chronic and painful venous leg ulcers in older adults. Rejuvenation Res. 2012, 15, 281–287. [Google Scholar] [CrossRef] [PubMed]
- Ud-Din, S.; Giddings, P.; Colthurst, J.; Whiteside, S.; Morris, J.; Bayat, A. Significant reduction of symptoms of scarring with electrical stimulation: Evaluated with subjective and objective assessment tools. Wounds 2013, 25, 212–224. [Google Scholar]
- Hampton, S.; King, L. Healing an intractable wound using bio-electric stimulation therapy. Br. J. Nurs. 2005, 14, S30–S32. [Google Scholar] [CrossRef] [PubMed]
- Hampton, S.; Collins, F. Treating a pressure ulcer with bio-electric stimulation therapy. Br. J. Nurs. 2006, 15, S14–S18. [Google Scholar] [CrossRef] [PubMed]
- Franek, A.; Taradaj, J.; Polak, A.; Cierpka, L.; Blaszczak, E. Efficacy of high voltage stimulation for healing of venous leg ulcers in surgically and conservatively treated patients. Phlebologie 2006, 35, 127–133. [Google Scholar]
- Kloth, L.C. Wound healing with conductive electrical stimulation—It’s the dosage that counts. J. Wound Technol. 2009, 6, 30. [Google Scholar]
- Ramadan, A.; Elsaidy, M.; Zyada, R. Effect of low-intensity direct current on the healing of chronic wounds: A literature review. J. Wound Care 2008, 17, 292–296. [Google Scholar] [CrossRef] [PubMed]
- Newton, R.A.; Karselis, T.C. Skin pH following high voltage pulsed galvanic stimulation. Phys. Ther. 1983, 63, 1593–1596. [Google Scholar] [PubMed]
- Ojingwa, J.C.; Isseroff, R.R. Electrical stimulation of wound healing. J. Investig. Dermatol. 2003, 121, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Adunsky, A.; Ohry, A. Decubitus direct current treatment (DDCT) of pressure ulcers: Results of a randomized double-blinded placebo controlled study. Arch. Gerontol. Geriatr. 2005, 41, 261–269. [Google Scholar] [CrossRef] [PubMed]
- Chen, C.; Johnson, M.I. A comparison of transcutaneous electrical nerve stimulation (TENS) at 3 and 80 pulses per second on cold-pressor pain in healthy human participants. Clin. Physiol. Funct. Imaging 2010, 30, 260–268. [Google Scholar] [CrossRef] [PubMed]
- Liebano, R.E.; Rakel, B.; Vance, C.G.; Walsh, D.M.; Sluka, K.A. An investigation of the development of analgesic tolerance to TENS in humans. Pain 2011, 152, 335–342. [Google Scholar] [CrossRef] [PubMed]
- Kjartansson, J.; Lundeberg, T.; Körlof, B. Transcutaneous electrical nerve stimulation (TENS) in ischemic tissue. Plast. Reconstr. Surg. 1988, 81, 813–815. [Google Scholar] [PubMed]
- Johnson, M.I. Transcutaneous Electrical Nerve Stimulation (TENS). In Electrotherapy: Evidence-Based Practice, 12th ed.; Watson, T., Ed.; Elsevier: Toronto, ON, Canada, 2008. [Google Scholar]
- Sherry, J.E.; Oehrlein, K.M.; Hegge, K.S.; Morgan, B.J. Effect of burst-mode transcutaneous electrical nerve stimulation on peripheral vascular resistance. Phys. Ther. 2001, 81, 1183–1191. [Google Scholar] [PubMed]
- Colthurst, J.; Giddings, P. A retrospective case note review of the Fenzian electrostimulation system: A novel non-invasive, non-pharmacological treatment. Pain Clin. 2007, 19, 7–14. [Google Scholar] [CrossRef]
- Guo, A.; Song, B.; Reid, B.; Gu, Y.; Forrester, J.V.; Jahoda, C.A.; Zhao, M. Effects of physiological electric fields on migration of human dermal fibroblasts. J. Investig. Dermatol. 2010, 130, 2320–2327. [Google Scholar] [CrossRef] [PubMed]
- Talebi, G.; Torkaman, G.; Firoozabadi, M.; Shariat, S. Effect of anodal and cathoda microamperage direct current electrical stimulation on injury potential and wound size in guinea pigs. J. Rehabil. Res. Dev. 2008, 45, 153–159. [Google Scholar] [CrossRef] [PubMed]
- Koel, G.; Houghton, P.E. Electrostimulation: Current status, strength of evidence guidelines, and meta-analysis. Adv. Wound Care (New Rochelle) 2014, 3, 118–126. [Google Scholar] [CrossRef]
© 2014 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
