Dynamic Navigation in Endodontic Surgery: A Systematic Review
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Protocol
- (P) Population: human subjects (alive/cadavers) who have undergone endodontic surgery with dynamic computer-assisted navigation systems;
- (I) Intervention: endodontic surgery performed through dynamic computer-assisted navigation systems;
- (C) Comparison: static computer-assisted, free-hand (FH), robot-assisted endodontic surgery;
- (O) Outcome(s):
- -
- Primary outcome(s): accuracy and reliability of dynamic computer-assisted navigation systems in endodontic surgery, measured as two-dimensional and three-dimensional virtual deviations (platform depth deviation, apical depth deviation, global platform deviation, global apex deviation, and angular deflection), osteotomy size (diameter, volume, depth, height, and length), and root-end resection (resected root length, residual root length, and resection angle);
- -
- Secondary outcome(s): DNS procedural duration, osteotomy duration, root-end resection duration, root-end preparation duration, root-end fill duration, total surgery duration, the dentist’s level of expertise, follow-up, outcome measures for healing following surgical endodontics (pain, swelling, and other symptoms/signs; satisfactory healing of soft tissue; sinus tract; loss of function; radiological evidence of the repair of apical periodontitis; the reformation of the periodontal ligament space; and 1-year follow-up), type and rate of complications (intra- and postoperative), and patient- and dentist-reported feedback [24].
2.2. Search Strategy
2.3. Study Selection and Eligibility Criteria
2.4. Data Extraction and Collection
- -
- Population characteristics: gender ratio, sample size, mean age/age range, comorbidities/ongoing pharmacological treatment, and dentition status;
- -
- Study characteristics: study design, first author, year, reference, journal, funding, and quality assessment;
- -
- Endodontic surgery characteristics: proximity to noble anatomical/surgically critical structures, osteotomy, apex location, root-end resection, root-end cavity preparation, root-end fill, the use of regenerative materials, and sutures;
- -
- Intervention/Comparison characteristics:
- Characteristics of dynamic computer-assisted navigation interventions: planning software, dental impression technique, radiographic imaging, navigation system, navigation software, and guidance method for navigation;
- Comparison: planning software, dental impression technique, radiographic imaging, navigation system, navigation software, and guidance method for navigation;
- -
- Primary outcome(s): two-dimensional and three-dimensional virtual deviations (platform depth deviation, apical depth deviation, angular deflection, global platform deviation, and global apex deviation), osteotomy size (diameter, volume, depth, height, and length), and root-end resection (resected root length, residual root length, and resection angle);
- -
- Secondary outcome(s): DNS procedural, osteotomy, root-end resection, root-end preparation, root-end fill, and total surgery duration; the dentist’s level of expertise; dentist-reported feedback; follow-up; outcome measures for healing following surgical endodontics (pain, swelling, and other symptoms/signs, satisfactory healing of soft tissue, sinus tract, loss of function, radiological evidence of the repair of apical periodontitis, the reformation of the periodontal ligament space, and 1-year follow-up) [24]; complications (intra- and postoperative); and patient-reported feedback.
2.5. Data Synthesis
- -
- Assess the accuracy and the reliability of dynamic computer-assisted navigation systems in endodontic surgery (platform depth deviation, apical depth deviation, global platform deviation, global apex deviation, and angular deflection);
- -
- Assess the dentist-reported and patient-reported acceptability and usability of, and satisfaction with, dynamic computer-assisted navigation systems in endodontic surgery;
- -
- Evaluate the accuracy and reliability of dynamic computer-assisted navigation systems in endodontic surgery across dentists’ levels of experience;
- -
- Compare the accuracy and reliability of dynamic computer-assisted navigation systems vs. half or full static computer-assisted, free-hand (FH), and robot-assisted endodontic surgery;
- -
- Compare the procedural time, outcome measures for healing following surgical endodontics (pain, swelling, and other symptoms/signs; satisfactory healing of soft tissue; sinus tract; loss of function; radiological evidence of the repair of apical periodontitis; the reformation of the periodontal ligament space; and 1-year follow-up), and the complications in dynamic computer-assisted navigation systems vs. half or full static computer-assisted, free-hand (FH), and robot-assisted endodontic surgery;
- -
- Compare dentist-reported and patient-reported acceptability and usability of, and satisfaction with, dynamic computer-assisted navigation systems vs. half or full static computer-assisted, free-hand (FH), and robot-assisted endodontic surgery.
2.6. Quality Assessment
3. Results
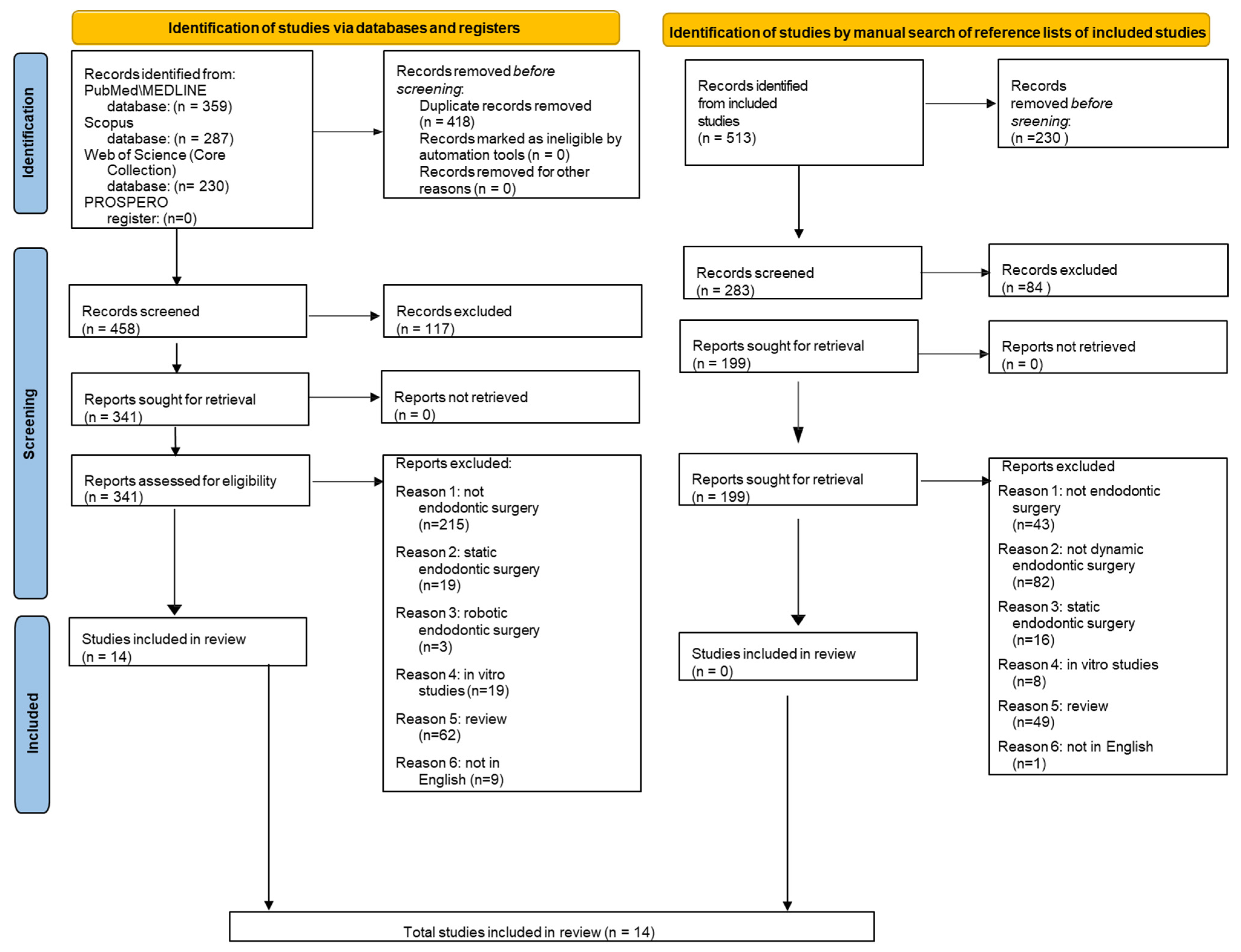
3.1. Study Selection
3.2. Study Characteristics
3.3. Sample Characteristics
3.4. Endodontic Surgery: Location and DNS Steps
3.5. Intervention Characteristics
3.6. Primary Outcome(s)
3.6.1. Virtual Deviations and Osteotomy Parameters
In Vivo Studies
Cadaver Studies
3.6.2. Virtual Deviations and Parameters of Apical Access
In Vivo Studies
Cadaver Studies
3.6.3. Virtual Deviations and Parameters of Root-End Resection
In Vivo Studies
Cadaver Studies
3.7. Secondary Outcome(s)
3.7.1. Procedural Time
In Vivo Studies
Cadaver Studies
3.7.2. Dentist Level of Expertise
3.7.3. Follow-Up
3.7.4. Outcome Measures for Healing Following Surgical Endodontics
3.7.5. Dentist- and Patient-Reported Feedback
3.8. Quality Assessment
4. Discussion
4.1. Accuracy and Reliability of Dynamic Computer-Assisted Navigation Systems in Apicectomy
4.1.1. Virtual Deviations and Osteotomy Parameters
4.1.2. Virtual Deviations and Parameters of Apical Access
4.1.3. Virtual Deviations and Parameters of Root-End Resection
4.2. Procedural Time, Dentist Level of Expertise, and Healing Outcomes
4.2.1. Procedural Time
4.2.2. Dentist Level of Expertise
4.2.3. Outcome Measures for Healing Following Surgical Endodontics
4.3. Follow-Up
4.4. Dentist- and Patient-Reported Feedback
4.5. Strengths, Limitations, and Future Perspectives
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Kim, S.; Kratchman, S. Modern Endodontic Surgery Concepts and Practice: A Review. J. Endod. 2006, 32, 601–623. [Google Scholar] [CrossRef]
- von Arx, T.; Jensen, S.S.; Hänni, S.; Friedman, S. Five-Year Longitudinal Assessment of the Prognosis of Apical Microsurgery. J. Endod. 2012, 38, 570–579. [Google Scholar] [CrossRef]
- von Arx, T. Apical Surgery: A Review of Current Techniques and Outcome. Saudi Dent. J. 2011, 23, 9–15. [Google Scholar] [CrossRef] [PubMed]
- Block, M.S.; Emery, R.W. Static or Dynamic Navigation for Implant Placement—Choosing the Method of Guidance. J. Oral Maxillofac. Surg. 2016, 74, 269–277. [Google Scholar] [CrossRef]
- Saxena, P.; Gupta, S. Static vs. Dynamic Navigation for Endodontic Microsurgery—A Comparative Review. J. Oral Biol. Craniofacial Res. 2022, 12, 410–412. [Google Scholar] [CrossRef]
- Samiei, M.; Sabanik, P.; Tavakkoli Avval, S. Guided Endodontics for Non-Surgical Root Canal Retreatment: A Systematic Review. Iran. Endod. J. 2025, 20, e3. [Google Scholar] [CrossRef]
- Hegde, V.; Mandke, L.; Memon, K.; Ansari, M.; Srilatha, S.; Mujawar, A. Dynamic Navigation in Endodontics: A Comprehensive Literature Review. J. Conserv. Dent. Endod. 2024, 27, 1202–1210. [Google Scholar] [CrossRef] [PubMed]
- Aldahmash, S.A.; Price, J.B.; Mostoufi, B.; Griffin, I.L.; Dianat, O.; Tordik, P.A.; Martinho, F.C. Real-Time 3-Dimensional Dynamic Navigation System in Endodontic Microsurgery: A Cadaver Study. J. Endod. 2022, 48, 922–929. [Google Scholar] [CrossRef] [PubMed]
- Gambarini, G.; Galli, M.; Stefanelli, L.V.; Di Nardo, D.; Morese, A.; Seracchiani, M.; De Angelis, F.; Di Carlo, S.; Testarelli, L. Endodontic Microsurgery Using Dynamic Navigation System: A Case Report. J. Endod. 2019, 45, 1397–1402.e6. [Google Scholar] [CrossRef] [PubMed]
- Cecchetti, F.; Di Girolamo, M.; Ippolito, D.G.; Baggi, L. Computer-Guided Implant Surgery: Analysis of Dynamic Navigation Systems and Digital Accuracy. J. Biol. Regul. Homeost. Agents 2020, 34, 9–17. [Google Scholar]
- Panchal, N.; Mahmood, L.; Retana, A.; Emery, R. Dynamic Navigation for Dental Implant Surgery. Oral Maxillofac. Surg. Clin. N. Am. 2019, 31, 539–547. [Google Scholar] [CrossRef]
- Wu, Y.; Tao, B.; Lan, K.; Shen, Y.; Huang, W.; Wang, F. Reliability and Accuracy of Dynamic Navigation for Zygomatic Implant Placement. Clin. Oral Implant. Res. 2022, 33, 362–376. [Google Scholar] [CrossRef] [PubMed]
- Zhang, H.-X.; Yan, Z.-Y.; Cui, N.-H.; Sun, F.; Wu, B.-Z. Accuracy of Computer-Assisted Dynamic Navigation When Performing Coronectomy of the Mandibular Third Molar: A Pilot Study. J. Dent. 2023, 139, 104762. [Google Scholar] [CrossRef] [PubMed]
- Jain, S.D.; Carrico, C.K.; Bermanis, I.; Rehil, S. Intraosseous Anesthesia Using Dynamic Navigation Technology. J. Endod. 2020, 46, 1894–1900. [Google Scholar] [CrossRef]
- Di Spirito, F.; Gasparro, R.; Di Palo, M.P.; Sessa, A.; Giordano, F.; Rizki, I.; Allegretti, G.; Bramanti, A. Dynamic Computer-Aided Navigation System in Dentoalveolar Surgery and Maxillary Bone Augmentation in a Dental Setting: A Systematic Review. Healthcare 2025, 13, 1730. [Google Scholar] [CrossRef] [PubMed]
- Yang, X.; Zhang, Y.; Chen, X.; Huang, L.; Qiu, X. Limitations and Management of Dynamic Navigation System for Locating Calcified Canals Failure. J. Endod. 2024, 50, 96–105. [Google Scholar] [CrossRef]
- Janabi, A.; Tordik, P.A.; Griffin, I.L.; Mostoufi, B.; Price, J.B.; Chand, P.; Martinho, F.C. Accuracy and Efficiency of 3-Dimensional Dynamic Navigation System for Removal of Fiber Post from Root Canal–Treated Teeth. J. Endod. 2021, 47, 1453–1460. [Google Scholar] [CrossRef]
- Gambarini, G.; Galli, M.; Morese, A.; Stefanelli, L.V.; Abduljabbar, F.; Giovarruscio, M.; Di Nardo, D.; Seracchiani, M.; Testarelli, L. Precision of Dynamic Navigation to Perform Endodontic Ultraconservative Access Cavities: A Preliminary In Vitro Analysis. J. Endod. 2020, 46, 1286–1290. [Google Scholar] [CrossRef]
- Vasudevan, A.; Santosh, S.S.; Selvakumar, R.J.; Sampath, D.T.; Natanasabapathy, V. Dynamic Navigation in Guided Endodontics—A Systematic Review. Eur. Endod. J. 2022, 7, 81–91. [Google Scholar] [CrossRef]
- Kapoor, A.; Alagarsamy, R.; Lal, B.; Rana, A.S.; Kaur, A.; Sharma, S.; Logani, A. Dynamic Navigation in Endodontics: Scope, Benefits, and Challenges—A Systematic Review. J. Endod. 2025, 51, 879–889.e2. [Google Scholar] [CrossRef]
- Moher, D.; Shamseer, L.; Clarke, M.; Ghersi, D.; Liberati, A.; Petticrew, M.; Shekelle, P.; Stewart, L.A. Preferred Reporting Items for Systematic Review and Meta-Analysis Protocols (PRISMA-P) 2015 Statement. Syst. Rev. 2015, 4, 1. [Google Scholar] [CrossRef] [PubMed]
- Liberati, A.; Altman, D.G.; Tetzlaff, J.; Mulrow, C.; Gøtzsche, P.C.; Ioannidis, J.P.A.; Clarke, M.; Devereaux, P.J.; Kleijnen, J.; Moher, D. The PRISMA Statement for Reporting Systematic Reviews and Meta-Analyses of Studies That Evaluate Health Care Interventions: Explanation and Elaboration. PLoS Med. 2009, 6, e1000100. [Google Scholar] [CrossRef] [PubMed]
- Richardson, W.S.; Wilson, M.C.; Nishikawa, J.; Hayward, R.S. The Well-Built Clinical Question: A Key to Evidence-Based Decisions. ACP J. Club 1995, 123, A12–A13. [Google Scholar] [CrossRef] [PubMed]
- Duncan, H.F.; Nagendrababu, V.; El-Karim, I.A.; Dummer, P.M.H. Outcome Measures to Assess the Effectiveness of Endodontic Treatment for Pulpitis and Apical Periodontitis for Use in the Development of European Society of Endodontology (ESE) S3 Level Clinical Practice Guidelines: A Protocol. Int. Endod. J. 2021, 54, 646–654. [Google Scholar] [CrossRef] [PubMed]
- American Association of Endodontists. Guide to Clinical Endodontics, 6th ed.; American Association of Endodontists: Chicago, IL, USA, 2019. [Google Scholar]
- European Society of Endodontology. Quality Guidelines for Endodontic Treatment: Consensus Report of the European Society of Endodontology. Int. Endod. J. 2006, 39, 921–930. [Google Scholar] [CrossRef] [PubMed]
- Higgins, J.P.T.; Green, S. (Eds.) Cochrane Handbook for Systematic Reviews of Interventions; Wiley: Hoboken, NJ, USA, 2008. [Google Scholar]
- Gibello, U.; Cemenasco, A.; Giordano, L.; Alovisi, M.; Cortese, L.; Balocco, A.; Berutti, E.; Pasqualini, D. Combined Orthograde 3D Navigation Microsurgical Endodontic Retreatment for the Management of Persistent Apical Periodontitis in a Mandibular Molar. G. Ital. Endod. 2023, 37, 94–102. [Google Scholar] [CrossRef]
- Manishaa, B.; Sajjan, G.S.; Kinariwala, N.; Varma, K.M.; Ponnada, N.; Bagu, S.V. Endodontic Dynamic Navigation for Precise Apical Microsurgery: Case Report. Endodontology 2024, 36, 181–187. [Google Scholar] [CrossRef]
- Martinho, F.C.; Aldahmash, S.A.; Cahill, T.Y.; Gupta, S.; Dianat, O.; Mostoufi, B.; Price, J.B.; Griffin, I.; Tordik, P.A. Comparison of the Accuracy and Efficiency of a 3-Dimensional Dynamic Navigation System for Osteotomy and Root-End Resection Performed by Novice and Experienced Endodontists. J. Endod. 2022, 48, 1327–1333.e1. [Google Scholar] [CrossRef]
- Dianat, O.; Nosrat, A.; Mostoufi, B.; Price, J.B.; Gupta, S.; Martinho, F.C. Accuracy and Efficiency of Guided Root-end Resection Using a Dynamic Navigation System: A Human Cadaver Study. Int. Endod. J. 2021, 54, 793–801. [Google Scholar] [CrossRef]
- Martinho, F.C.; Rollor, C.; Westbrook, K.; Aldahmash, S.A.; Fay, G.G.; Rivera, E.; Parsa, A.; Price, J.B.; Tordik, P.A. A Cadaver-Based Comparison of Sleeve-Guided Implant-Drill and Dynamic Navigation Osteotomy and Root-End Resections. J. Endod. 2023, 49, 1004–1011. [Google Scholar] [CrossRef] [PubMed]
- Chen, C.; Zhang, R.; Qin, L.; Lin, Y.; Zhang, X.; Li, F.; Meng, L. Patient-Centered Outcome Assessment of Static and Dynamic Navigation-Aided Endodontic Microsurgery: A Randomized Controlled Trial. J. Endod. 2025, 51, 666–673. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; Huang, L.; Li, S.; Lao, S.; Yan, N.; Wu, H.; Yang, X. Endodontic Microsurgery with the Aid of Dynamic Navigation System Using Minimally Invasive Incision Approach in Anatomically Complex Scenarios: A Case Series. J. Endod. 2024, 50, 1777–1783. [Google Scholar] [CrossRef] [PubMed]
- Fu, W.; Chen, C.; Bian, Z.; Meng, L. Endodontic Microsurgery of Posterior Teeth with the Assistance of Dynamic Navigation Technology: A Report of Three Cases. J. Endod. 2022, 48, 943–950. [Google Scholar] [CrossRef]
- Villa-Machado, P.A.; Restrepo-Restrepo, F.A.; Tobón-Arroyave, S.I. Dynamically Guided Transantral Piezoelectric Endodontic Microsurgery: A Case Report with Technical Considerations. Int. Endod. J. 2024, 57, 490–500. [Google Scholar] [CrossRef] [PubMed]
- Chen, C.; Zhang, R.; Zhang, W.; Wang, F.; Wang, Z.; Qin, L.; Bian, Z.; Meng, L. Analysis of the Accuracy of a Dynamic Navigation System in Endodontic Microsurgery: A Prospective Case Series Study. J. Dent. 2023, 134, 104534. [Google Scholar] [CrossRef]
- Chen, C.; Zhang, R.; Zhang, W.; Li, F.; Wang, Z.; Qin, L.; Chen, Y.; Bian, Z.; Meng, L. Clinical and Radiological Outcomes of Dynamic Navigation in Endodontic Microsurgery: A Prospective Study. Clin. Oral Investig. 2023, 27, 5317–5329. [Google Scholar] [CrossRef]
- Lu, Y.-J.; Chiu, L.-H.; Tsai, L.-Y.; Fang, C.-Y. Dynamic Navigation Optimizes Endodontic Microsurgery in an Anatomically Challenging Area. J. Dent. Sci. 2022, 17, 580–582. [Google Scholar] [CrossRef]
- Pera, F.; Vocaturo, C.; Crupi, A.; Longhi, B.; Campagna, A.; Fiorino, A.; Gibello, U.; Roccuzzo, A. Impact of Surgeons’ Experience on Implant Placement Accuracy Using a Dynamic Navigation System: A Cadaver Pilot Study. Prosthesis 2025, 7, 20. [Google Scholar] [CrossRef]
- Iandolo, A.; Abdellatif, D.; Amato, A.; Pisano, M.; Carboncini, F.; Pontoriero, D.I.K.; Caggiano, M. Calcifications Detection and Management in the Obliterated Endodontic Space. J. Osseointegration 2023, 15, 48–52. [Google Scholar] [CrossRef]
- Di Spirito, F.; Scelza, G.; Amato, A.; Rosa, D.; Gallotti, A.; Martina, S. Prevalence of Dental Anomalies in a Sample of Growing Subjects: A Retrospective Study. Epidemiol. Prev. 2022, 46, 376–381. [Google Scholar] [CrossRef]
- Iandolo, A. The Endodontic Space. Healthcare 2023, 11, 628. [Google Scholar] [CrossRef]
- Battista, E.; Gasparro, R.; Cacciola, M.; Sammartino, G.; Marenzi, G. Dynamic Navigation System for Immediate Implant Placement in the Maxillary Aesthetic Region. Appl. Sci. 2022, 12, 5510. [Google Scholar] [CrossRef]
- Bhochhibhoya, A.; Guragain, M. Dynamic Navigation: A Novel Tool for Dental Implant Surgery. J. Nepal. Prosthodont. Soc. 2022, 5. [Google Scholar] [CrossRef]
- Marques-Guasch, J.; Bofarull-Ballús, A.; Giralt-Hernando, M.; Hernández-Alfaro, F.; Gargallo-Albiol, J. Dynamic Implant Surgery—An Accurate Alternative to Stereolithographic Guides—Systematic Review and Meta-Analysis. Dent. J. 2023, 11, 150. [Google Scholar] [CrossRef] [PubMed]
- Gulabivala, K.; Ng, Y.L. Factors That Affect the Outcomes of Root Canal Treatment and Retreatment—A Reframing of the Principles. Int. Endod. J. 2023, 56, 82–115. [Google Scholar] [CrossRef] [PubMed]
- Jonaityte, E.M.; Bilvinaite, G.; Drukteinis, S.; Torres, A. Accuracy of Dynamic Navigation for Non-Surgical Endodontic Treatment: A Systematic Review. J. Clin. Med. 2022, 11, 3441. [Google Scholar] [CrossRef]
- Monsarrat, P.; Arcaute, B.; Peters, O.A.; Maury, E.; Telmon, N.; Georgelin-Gurgel, M.; Maret, D. Interrelationships in the Variability of Root Canal Anatomy among the Permanent Teeth: A Full-Mouth Approach by Cone-Beam CT. PLoS ONE 2016, 11, e0165329. [Google Scholar] [CrossRef]
- Wang, H.; Xu, X.; Bian, Z.; Liang, J.; Chen, Z.; Hou, B.; Qiu, L.; Chen, W.; Wei, X.; Hu, K.; et al. Expert Consensus on Apical Microsurgery. Int. J. Oral Sci. 2025, 17, 2. [Google Scholar] [CrossRef]
- Thakur, V.; Kaul, R. Apicoectomy: A Review of Clinical Concepts and Techniques. J. Oral Res. Rev. 2025, 17, 77–82. [Google Scholar] [CrossRef]
- Agarwal, T.; Govind, S. Comprehensive Overview of the Apicoectomy Procedure: Indications, Technique, and Postoperative Care. J. Biomed. Pharm. Res. 2024, 13, 80–85. [Google Scholar] [CrossRef]
- Fu, M.; Zhao, S.; Zhou, X.; Hou, B.; Zhang, C. Removal of a Fractured File beyond the Apical Foramen Using Robot-Assisted Endodontic Microsurgery: A Clinical Report. BMC Oral Health 2025, 25, 8. [Google Scholar] [CrossRef] [PubMed]
- Ma, L.; Ye, M.; Wu, M.; Chen, X.; Shen, S. A Retrospective Study of Dynamic Navigation System-Assisted Implant Placement. BMC Oral Health 2023, 23, 759. [Google Scholar] [CrossRef] [PubMed]
- Kim, Y.; Roh, B.-D.; Shin, Y.; Kim, B.S.; Choi, Y.; Ha, A. Morphological Characteristics and Classification of Mandibular First Molars Having 2 Distal Roots or Canals: 3-Dimensional Biometric Analysis Using Cone-Beam Computed Tomography in a Korean Population. J. Endod. 2018, 44, 46–50. [Google Scholar] [CrossRef]
- Kim, S.-Y.; Yang, S.-E. Cone-Beam Computed Tomography Study of Incidence of Distolingual Root and Distance from Distolingual Canal to Buccal Cortical Bone of Mandibular First Molars in a Korean Population. J. Endod. 2012, 38, 301–304. [Google Scholar] [CrossRef] [PubMed]
- Jin, G.; Kim, K.; Roh, B.; Lee, C.; Lee, S. Buccal Bone Plate Thickness of the Asian People. J. Endod. 2005, 31, 430–434. [Google Scholar] [CrossRef]
- Zahedi, S.; Mostafavi, M.; Lotfirikan, N. Anatomic Study of Mandibular Posterior Teeth Using Cone-Beam Computed Tomography for Endodontic Surgery. J. Endod. 2018, 44, 738–743. [Google Scholar] [CrossRef]
- Huumonen, S.; Kvist, T.; Gröndahl, K.; Molander, A. Diagnostic Value of Computed Tomography in Re-treatment of Root Fillings in Maxillary Molars. Int. Endod. J. 2006, 39, 827–833. [Google Scholar] [CrossRef] [PubMed]
- Mekhdieva, E.; Del Fabbro, M.; Alovisi, M.; Scotti, N.; Comba, A.; Berutti, E.; Pasqualini, D. Dynamic Navigation System vs. Free-Hand Approach in Microsurgical and Non-Surgical Endodontics: A Systematic Review and Meta-Analysis of Experimental Studies. J. Clin. Med. 2023, 12, 5845. [Google Scholar] [CrossRef]
- Ribeiro, D.; Reis, E.; Marques, J.A.; Falacho, R.I.; Palma, P.J. Guided Endodontics: Static vs. Dynamic Computer-Aided Techniques—A Literature Review. J. Pers. Med. 2022, 12, 1516. [Google Scholar] [CrossRef]
- Wang, F.; Cai, X.; Sun, W.; Chen, C.; Meng, L. Application of Dynamic Navigation Technology in Oral and Maxillofacial Surgery. Clin. Oral Investig. 2024, 29, 13. [Google Scholar] [CrossRef]
- Isufi, A.; Hsu, T.-Y.; Chogle, S. Robot-Assisted and Haptic-Guided Endodontic Surgery: A Case Report. J. Endod. 2024, 50, 533–539.e1. [Google Scholar] [CrossRef]
- Ambu, E.; Gori, B.; Marruganti, C.; Malvicini, G.; Bordone, A.; Giberti, L.; Grandini, S.; Gaeta, C. Influence of Calcified Canals Localization on the Accuracy of Guided Endodontic Therapy: A Case Series Study. Dent. J. 2023, 11, 183. [Google Scholar] [CrossRef]
- Fornara, R.; Pisano, M.; Salvati, G.; Malvicini, G.; Iandolo, A.; Gaeta, C. Management of Calcified Canals with a New Type of Endodontic Static Guide: A Case Report. Dent. J. 2024, 12, 166. [Google Scholar] [CrossRef]
- Liu, S.-M.; Peng, L.; Zhao, Y.-J.; Han, B.; Wang, X.-Y.; Wang, Z.-H. The Learning Curve of a Dynamic Navigation System Used in Endodontic Apical Surgery. J. Dent. Sci. 2024, 19, 2247–2255. [Google Scholar] [CrossRef]
- Pisano, M.; Sangiovanni, G.; Frucci, E.; Scorziello, M.; De Benedetto, G.; Iandolo, A. Evaluation of the Accuracy of Electronic Apex Locators in Modern Endodontics: An Umbrella Review. Medicina 2024, 60, 1709. [Google Scholar] [CrossRef]
- Orosco, F.A.; Bernardineli, N.; Garcia, R.B.; Bramante, C.M.; Duarte, M.A.H.; Moraes, I.G. de In Vivo Accuracy of Conventional and Digital Radiographic Methods in Confirming Root Canal Working Length Determination by Root ZX. J. Appl. Oral Sci. 2012, 20, 522–525. [Google Scholar] [CrossRef]
- Tang, W.L.; Chao, X.Y.; Ye, Z.; Liu, M.W.; Jiang, H. The Use of Dynamic Navigation Systems as a Component of Digital Dentistry. J. Dent. Res. 2024, 103, 119–128. [Google Scholar] [CrossRef]
- Amador-Cabezalí, A.; Pardal-Peláez, B.; Quispe-López, N.; Lobato-Carreño, M.; Sanz-Sánchez, Á.; Montero, J. Influence of the Retrograde Filling Material on the Success of Periapical Surgery. Systematic Review and Meta-Analysis by Groups. Coatings 2022, 12, 1140. [Google Scholar] [CrossRef]
- Remschmidt, B.; Rieder, M.; Gsaxner, C.; Gaessler, J.; Payer, M.; Wallner, J. Augmented Reality-Guided Apicoectomy Based on Maxillofacial CBCT Scans. Diagnostics 2023, 13, 3037. [Google Scholar] [CrossRef]
- von Arx, T.; Janner, S.F.M.; Jensen, S.S.; Bornstein, M.M. The Resection Angle in Apical Surgery: A CBCT Assessment. Clin. Oral Investig. 2016, 20, 2075–2082. [Google Scholar] [CrossRef]
- Jang, Y.; Hong, H.-T.; Roh, B.-D.; Chun, H.-J. Influence of Apical Root Resection on the Biomechanical Response of a Single-Rooted Tooth: A 3-Dimensional Finite Element Analysis. J. Endod. 2014, 40, 1489–1493. [Google Scholar] [CrossRef]
- Sauveur, G.; Boccara, E.; Colon, P.; Sobel, M.; Boucher, Y. A Photoelastimetric Analysis of Stress Induced by Root-End Resection. J. Endod. 1998, 24, 740–743. [Google Scholar] [CrossRef]
- Gagliani, M.; Taschieri, S.; Molinari, R. Ultrasonic Root-End Preparation: Influence of Cutting Angle on the Apical Seal. J. Endod. 1998, 24, 726–730. [Google Scholar] [CrossRef]
- de, O. Dal Piva, A.M.; Tribst, J.P.M.; de A. e Souza, R.O.; Borges, A.L.S. Influence of Alveolar Bone Loss and Cement Layer Thickness on the Biomechanical Behavior of Endodontically Treated Maxillary Incisors: A 3-Dimensional Finite Element Analysis. J. Endod. 2017, 43, 791–795. [Google Scholar] [CrossRef]
- Pisano, M.; Di Spirito, F.; Martina, S.; Sangiovanni, G.; D’Ambrosio, F.; Iandolo, A. Intentional Replantation of Single-Rooted and Multi-Rooted Teeth: A Systematic Review. Healthcare 2022, 11, 11. [Google Scholar] [CrossRef]
- Pereira, T.C.; Boutsioukis, C.; Dijkstra, R.J.B.; Petridis, X.; Versluis, M.; de Andrade, F.B.; van de Meer, W.J.; Sharma, P.K.; van der Sluis, L.W.M.; So, M.V.R. Biofilm Removal from a Simulated Isthmus and Lateral Canal during Syringe Irrigation at Various Flow Rates: A Combined Experimental and Computational Fluid Dynamics Approach. Int. Endod. J. 2021, 54, 427–438. [Google Scholar] [CrossRef] [PubMed]
- Nakakura-Ohshima, K.; Quispe-Salcedo, A.; Sano, H.; Hayasaki, H.; Ohshima, H. The Effects of Reducing the Root Length by Apicoectomy on Dental Pulp Revascularization Following Tooth Replantation in Mice. Dent. Traumatol. 2021, 37, 677–690. [Google Scholar] [CrossRef] [PubMed]
- Morgan, L.A.; Marshall, J.G. The Topography of Root Ends Resected with Fissure Burs and Refined with Two Types of Finishing Burs. Oral Surg. Oral Med. Oral Pathol. Oral Radiol. Endodontol. 1998, 85, 585–591. [Google Scholar] [CrossRef]
- Kontakiotis, E.G.; Lagoudakos, T.A.; Georgopoulou, M.K. The Influence of Root-end Resection and Root-end Cavity Preparation on Microleakage of Root Filled Teeth in Vitro. Int. Endod. J. 2004, 37, 403–407. [Google Scholar] [CrossRef]
- Xu, F.; Gong, Y.; Sina, A.; Wang, C.; Pouria, A.; Yu, M.; Tu, J.; Na, S. A Prospective Randomized Study on the Efficacy of Real-Time Dynamic Navigation in Deep Horizontal Mandibular Third Molar Extractions. BMC Oral Health 2024, 24, 1234. [Google Scholar] [CrossRef]
- Jorba-Garcia, A.; Figueiredo, R.; Gonzalez-Barnadas, A.; Camps-Font, O.; Valmaseda-Castellon, E. Accuracy and the Role of Experience in Dynamic Computer Guided Dental Implant Surgery: An in-Vitro Study. Med. Oral Patol. Oral Cir. Bucal 2018, 24, e76–e83. [Google Scholar] [CrossRef] [PubMed]
- Graf, T.; Keul, C.; Wismeijer, D.; Güth, J.F. Time and Costs Related to Computer-assisted versus Non-computer-assisted Implant Planning and Surgery. A Systematic Review. Clin. Oral Implant. Res. 2021, 32, 303–317. [Google Scholar] [CrossRef] [PubMed]
- Bhalerao, A.; Ayoub, A.; Marimuthu, M.; Wahab, A.; Kumar, V. Cost-Effectiveness of the Flapless Insertion of Zygomatic Implants Using Dynamic Navigation—A Retrospective Study. Ann. Maxillofac. Surg. 2023, 13, 211–215. [Google Scholar] [CrossRef]
- Sharma, S.; Haldar, P.; Kumar, V.; Chawla, A.; Logani, A. Learning Curve for Dynamic Navigation Procedure during Endodontic Management of Permanent Maxillary Anterior Teeth with Pulp Canal Calcification: A Risk-Adjusted Cumulative Summation Analysis of a Single Operator’s Experience. J. Endod. 2025, 51, 295–302. [Google Scholar] [CrossRef]
- Sun, T.-M.; Lee, H.-E.; Lan, T.-H. The Influence of Dental Experience on a Dental Implant Navigation System. BMC Oral Health 2019, 19, 222. [Google Scholar] [CrossRef]
- Selvaganesh, S.; Nesappan, T. Comparative Evaluation of the Accuracy, Operator Comfort and Time Taken for Implant Placement among Different Practitioners under Dynamic Navigation. J. Clin. Diagn. Res. 2023, 17, 10–14. [Google Scholar] [CrossRef]
- Tsesis, I.; Fuss, Z.; Lin, S.; Tilinger, G.; Peled, M. Analysis of Postoperative Symptoms Following Surgical Endodontic Treatment. Quintessence Int. 2003, 34, 756–760. [Google Scholar] [PubMed]
- Von Arx, T.; Vinzens-Majaniemi, T.; Bürgin, W.; Jensen, S.S. Changes of Periodontal Parameters Following Apical Surgery: A Prospective Clinical Study of Three Incision Techniques. Int. Endod. J. 2007, 40, 959–969. [Google Scholar] [CrossRef]
- Vonarx, T.; Jensen, S.; Hanni, S. Clinical and Radiographic Assessment of Various Predictors for Healing Outcome 1 Year After Periapical Surgery. J. Endod. 2007, 33, 123–128. [Google Scholar] [CrossRef] [PubMed]
- di Lauro, A.E.; Valletta, A.; Aliberti, A.; Cangiano, M.; Dolce, P.; Sammartino, G.; Gasparro, R. The Effectiveness of Autologous Platelet Concentrates in the Clinical and Radiographic Healing after Endodontic Surgery: A Systematic Review. Materials 2023, 16, 7187. [Google Scholar] [CrossRef]
- Zuolo, M.L.; Ferreira, M.O.F.; Gutmann, J.L. Prognosis in Periradicular Surgery: A Clinical Prospective Study. Int. Endod. J. 2000, 33, 91–98. [Google Scholar] [CrossRef] [PubMed]
- Ng, Y.; Gulabivala, K. Factors That Influence the Outcomes of Surgical Endodontic Treatment. Int. Endod. J. 2023, 56, 116–139. [Google Scholar] [CrossRef] [PubMed]
- Liu, C.; Liu, X.; Wang, X.; Liu, Y.; Bai, Y.; Bai, S.; Zhao, Y. Endodontic Microsurgery With an Autonomous Robotic System: A Clinical Report. J. Endod. 2024, 50, 859–864. [Google Scholar] [CrossRef] [PubMed]

| Study | Sample Characteristics | Endodontic Surgery | Intervention Characteristics | Primary Outcome(s) | Secondary Outcome(s) |
|---|---|---|---|---|---|
| Aldahmash 2022 J Endod [8] Randomized controlled trial American Association of Endodontists, Foundation for Endodontics | Population Sample size: n = 2 (cadavers) Gender ratio: MD Mean/Range age: MD Comorbidities/ongoing pharmacological treatment: NA Tooth/Teeth Treated: MD Type: MD Position: MD Previous endodontic treatment: MD Pulp tests (CPT, HPT, EPT): NA Percussion examination: NA Palpation examination: NA Diagnosis: NA Treated roots Number: n = 24 | DNS step(s) (n = roots): Proximity to noble anatomical/surgically critical structures: MD Osteotomy: n = 24 Apex location: n = 24 Root-end resection: n = 24 FH step(s): Root-end cavity preparation: Endodontic microscope + ProUltra ultrasonic surgical tips (n = 24) Root-end fill: Endodontic microscope + EndoSequence® (n = 24) Use of regenerative materials: NA Sutures: NA | Dental impression technique: MD Radiographic imaging: CBCT (n = 24) Planning software: X-Guide® (n = 24) Navigation software: X-Guide® (n = 24) Navigation system: DNS (n = 24) Guidance method for navigation: Radiopaque fiducial markers (X-clip) embedded in thermoplastic stent + real-time tracking via optical motion-tracking cameras (n = 24) | Virtual accuracy metrics in 2D/3D deviations (mm/°) (n = roots): Platform depth deviation: 1.09 ± 1.40 mm (n = 24) Apical depth deviation: 1.26 ± 1.39 mm (n = 24) Angular deflection: 1.10 ± 0.78° (n = 24) Global platform deviation: 0.60 ± 0.18 mm (n = 24) Global apex deviation:1.07 ± 1.55 mm (n = 24) Osteotomy size (mm/mm3) (n = roots): Diameter: MD Volume: 82.37 ± 61.40 mm3 (n = 24) Depth: 6.69 ± 2.38 mm (n = 24) Height: 3.72 ± 0.67 mm (n = 24) Length: 4.05 ± 0.13 mm (n = 24) Root-end resection (mm/°): Resected root length: 2.98 ± 0.27 mm (n = 24) Residual root length: 10.19 ± 2.36 mm (n = 24) Resection angle: 9.05 ± 7.78° (n = 24) | DNS procedural duration (s/min): 550 ± 264 s/~9.2 ± 4.4 min (average per treated root, n = 24) Osteotomy duration: MD Root-end resection duration: MD Root-end preparation duration: MD/+ filling: 250 ± 176 s/~4.2 ± 2.9 min (n = 24) Root-end fill duration: MD Total surgery duration (DNS + FH) (s/min): 800 ± 271 s/~13.3 ± 4.5 min (n = 24) Dentist level of expertise: EE (n = 1) Dentist-reported feedback: MD Complications (intra- and postoperative), type and rate (n = roots): Incomplete root-end resection, 4.16% (n = 1) |
| Chen 2025 J Endod [33] Randomized controlled trial General Program of National Natural Science Foundation of China; Fundamental Research Funds for the Central Universities; Wuhan Special Project on Knowledge Innovation; Key R&D projects of Hubei Provincial Science and Technology Plan; Research Project of School and Hospital of Stomatology Wuhan University | Population Sample size: n = 30 (alive) Gender ratio: 10 M/20 F Mean/Range age: 30.13 ± 10.19 yo/MD Comorbidities/Ongoing pharmacological treatment: MD Tooth/Teeth Treated: n = 34 Type: Anterior (5), posterior (29) Position: 25 maxilla/9 mandible Previous endodontic treatment: Yes, MD (timing), n = 34 Pulp tests (CPT, HPT, EPT): MD Percussion examination: MD Palpation examination: MD Diagnosis: Apical periodontitis in previously root canal-treated teeth (n = 34) Treated roots Number: n = 50 | DNS step(s) (n = roots): Proximity to noble anatomical/surgically critical structures: MD Osteotomy: n = 50 Apex location: n = 50 Root-end resection: n = 50 FH step(s): Root-end cavity preparation: Endodontic microscope + diamond-coated ultrasonic microtips (n = 50) Root-end fill: Endodontic microscope + iRoot BP Plus® (n = 50) Use of regenerative materials: NO Sutures: Monofilament sutures (n = 50) | Dental impression technique: MD Radiographic imaging: CBCT (n = 50) Planning software: DHC-ENDO1® (n = 50) Navigation software: DHC-ENDO1® (n = 50) Navigation system: DHC-ENDO1® (n = 50) Guidance method for navigation: Marker-based registration via radiopaque fiducial markers embedded in a registration device filled with silicone impression material + real-time infrared optical tracking (n = 50) | Virtual accuracy metrics in 2D/3D deviations (mm/°) (n = roots): Platform depth deviation: MD Apical depth deviation: MD Angular deflection: MD Global platform deviation: MD Global apex deviation: MD Osteotomy size (mm/mm3) (n = roots): Diameter: 4 mm (n = 50) Volume: MD Depth: MD Height: MD Length: MD Root-end resection (mm/°): Resected root length: 3 mm (n = 50) Residual root length: MD Resection angle: MD | DNS procedural duration (s/min): 292.48 ± 180.05 s/~4.9 ± 3.0 min (average per treated root, n = 50) The following was reported: Operation duration median: 234.50 s/~3.9 min (average per treated root, n = 50); operation duration min, max: ranged from 60.0 s (~1 min) to 894.0 s (~14.9 min). Osteotomy duration: MD Root-end resection duration: MD Root-end preparation duration: MD Root-end fill duration: MD Total surgery duration (DNS + FH) (s/min): MD Dentist level of expertise: EE (n = MD) Dentist-reported feedback: MD Follow-up: 1 week (n = 30) Outcome measures for healing following surgical endodontics (No. teeth/%): Pain, swelling, and other symptoms/signs: Yes, postoperative pain was short-lived, peaking early and progressively decreasing over time. Swelling peaked on day 2 and gradually decreased thereafter. Symptoms were assessed daily using patient questionnaires during the first postoperative week (n = 30 patients). Number of teeth not defined. Satisfactory healing of soft tissue: MD Sinus tract: No, presence of sinus tract not described preoperatively or postoperatively (n = 34/100%) Loss of function: MD Radiological evidence of repair of apical periodontitis: Yes (n = 34/100%) Reformation of the periodontal ligament space: MD 1-year follow-up (yes/no): No (n = 34/100%) Complications (intra- and postoperative), type and rate (n = roots): No (n = 50/100%) Patient-reported feedback: MD |
| Chen 2023 Clin Oral Investig [38] Prospective study General Program of National Natural Scientific Foundation of China; Wuhan Special Project on Knowledge Innovation | Population Sample size: n = 32 (alive) Gender ratio: 14 M/18 F Mean/Range age: 31.4 yo/18 yo-59 yo Comorbidities/Ongoing pharmacological treatment: MD Tooth/Teeth Treated: n = 46 Type: 29 anterior, 11 posterior + n = 6 MD (treated but not followed up and not reported in the article) Position: 32 maxilla/8 mandible + n = 6 MD (treated but not followed up and not reported in the article) Previous endodontic treatment: Yes, MD (timing) (n = 46) Pulp tests (CPT, HPT, EPT): NA Percussion examination: MD Palpation examination: MD Diagnosis: Apical periodontitis in previously root canal-treated teeth (n = 46) Treated roots Number: n = 51 | DNS step(s) (n = roots): Proximity to noble anatomical/surgically critical structures: Yes—The study included roots in proximity to the maxillary sinus, mandibular canal, and mental nerve (number of roots: MD) Osteotomy: n = 51 Apex location: n = 51 Root-end resection: n = 51 FH step(s): Root-end cavity preparation: Endodontic microscope + high-speed handpiece with a diamond bur or ET18D ultrasonic tip (n = 51) Root-end fill: Endodontic microscope + iRoot BP Plus® (n = 51) Use of regenerative materials: No Sutures: MD | Dental impression technique: MD Radiographic imaging: CBCT (n = 51) Planning software: DHC-ENDO1® (n = 51) Navigation software: DHC-ENDO1® (n = 51) Navigation system: DHC-ENDO1® (n = 51) Guidance method for navigation: Marker-based registration via radiopaque fiducial markers embedded in a registration device filled with silicone impression material + real-time infrared optical tracking (n = 51) | Virtual accuracy metrics in 2D/3D deviations (mm/°) (n = roots): Platform depth deviation: MD Apical depth deviation: MD Angular deflection: MD Global platform deviation: MD Global apex deviation: MD Osteotomy size (mm/mm3) (n = roots): Diameter: MD Volume: MD Depth: MD Height: MD Length: MD Root-end resection (mm/°): Resected root length: 3 mm (n = 51) Residual root length: MD Resection angle: MD | DNS procedural duration (s/min): MD Osteotomy duration: MD Root-end resection duration: MD Root-end preparation duration: MD Root-end fill duration: MD Total surgery duration (DNS + FH) (s/min): MD Dentist level of expertise: EE (n = 1) Dentist-reported feedback: MD Follow-up: Between 12 and 20 months; mean 13 months (n = 28/32 patients and 40/46 teeth) Outcome measures for healing following surgical endodontics (No. teeth/%): Pain, swelling, and other symptoms/signs: Yes (n = 2/40, 5%) Satisfactory healing of soft tissue: MD Sinus tract: Yes (n = 2/40, 5%) Loss of function: MD Radiological evidence of repair of apical periodontitis: On periapical radiography, 38/40 teeth showed complete or incomplete healing; 2/40 showed unsatisfactory healing. CBCT was performed in 35/40 teeth. Five teeth were excluded. On CBCT, 33/35 teeth showed complete healing; 2/35 showed unsatisfactory healing. Reformation of the periodontal ligament space: MD 1-year follow-up (yes/no): Yes (n = 40/46 86, 95%) Complications (intra- and postoperative), type and rate (n = roots): Postoperative sinus tract, 3.9% (n = 2/51) Patient-reported feedback: MD |
| Chen 2023 J Dent [37] Case series General Program of National Natural Scientific Foundation of China; Wuhan Special Project on Knowledge Innovation | Population Sample size: n = 9 (alive) Gender ratio: 2 M/7 F Mean/Range age: 29.6 yo/18 yo-48 yo Comorbidities/Ongoing pharmacological treatment: MD Tooth/Teeth Treated: n = 11 Type: 1.1 (×2), 1.2, 1.3, 1.6, 2.1 (×2), 2.2, 3.6 (×2), 4.1 Position: 8 maxilla/3 mandible Previous endodontic treatment: Yes (n = 11); timing reported for 2 teeth (2 months, 4 years) Pulp tests (CPT, HPT, EPT): NA Percussion examination: MD Palpation examination: MD Diagnosis: Apical periodontitis in previously root canal-treated teeth (n = 11) Treated roots Number: n = 12 | DNS step(s) (n = roots): Proximity to noble anatomical/surgically critical structures: MD Osteotomy: n = 12 Apex location: n = 12 Root-end resection: n = 12 FH step(s): Root-end cavity preparation: Endodontic microscope + diamond-coated ultrasonic microtips (n = 12) Root-end fill: Endodontic microscope + iRoot BP Plus® (n = 12) Use of regenerative materials: NO Sutures: Monofilament sutures (n = 12) | Dental impression technique: MD Radiographic imaging: CBCT (n = 12) Planning software: DHC-ENDO1® (n = 12) Navigation software: DHC-ENDO1® (n = 12) Navigation system: DHC-ENDO1® (n = 12) Guidance method for navigation: Marker-based registration via radiopaque fiducial markers embedded in a registration device filled with silicone impression material + real-time infrared optical tracking (n = 12) | Virtual accuracy metrics in 2D/3D deviations (mm/°) (n = roots): Platform depth deviation: MD Apical depth deviation: MD Angular deflection: 6.24 ± 3.69° (n = 12) Global platform deviation: 1.05 ± 0.74 mm (n = 12) Global apex deviation: 1.20 ± 0.67 mm (n = 12) Osteotomy size (mm/mm3) (n = roots): Diameter: MD Volume: MD Depth: MD Height: MD Length: MD Root-end resection (mm/°): Resected root length: MD Residual root length: MD Resection angle: MD Length deviation of root-end resection: 0.30 mm (IQR: 0.26) (n = 12) Angle deviation of root-end resection: 3.49° (IQR: 4.41) (n = 12) Depth of surgical path: ≤5 mm: Platform deviation: 1.31 ± 0.86 mm (n = 12) Apex deviation: 1.44 ± 0.75 mm (n = 12) Angular deviation: 5.80 ± 2.93° (n = 12) Length deviation of root-end resection: 0.31 (0.28) mm (n = 12) Angle deviation of root-end resection: 3.45 ± 2.05° (n = 12) >5 mm: Platform deviation: 0.70 ± 0.37 mm (n = 12) Apex deviation: 0.88 ± 0.41 mm (n = 12) Angular deviation: 6.86 ± 4.88° (n = 12) Length deviation of root-end resection: 0.26 ± 0.12 mm (n = 12) Angle deviation of root-end resection: 6.92 ± 7.05° (n = 12) | DNS procedural duration (s/min): MD Osteotomy duration: MD Root-end resection duration: MD Root-end preparation duration: MD Root-end fill duration: MD Total surgery duration (DNS + FH) (s/min): MD Dentist level of expertise: EE (n = 1) Dentist-reported feedback: MD Follow-up: Between 12 and 20 months; mean: 13.1 months (n = 8) Outcome measures for healing following surgical endodontics (No. teeth/%): Pain, swelling, and other symptoms/signs: MD Satisfactory healing of soft tissue: MD Sinus tract: Yes (n = 1/10, 10%); one tooth lost to follow-up (10/11 teeth evaluated) Loss of function: MD Radiological evidence of repair of apical periodontitis: Complete healing (n = 6), incomplete healing (n = 2), unsatisfactory healing (n = 1) (10 out of 11 teeth were evaluated at follow-up; however, radiological evidence of apical periodontitis repair was not available for 1 of these 10 teeth) Reformation of the periodontal ligament space: MD 1-year follow-up (yes/no): Yes (n = 10, 100%) Complications (intra- and post-operative), type and rate (n = roots): fistula present at follow-up, 10% (n = 1) Patient-reported feedback: MD |
| Dianat 2021 Int Endod J [31] Randomized controlled trial None | Population Sample size: n = 2 (cadavers) Gender ratio: MD Mean/Range age: MD Comorbidities/Ongoing pharmacological treatment: NA Tooth/Teeth Treated: n = 20 Type: Anteriors and canines (n = 10),premolars (n = 4), and molars (n = 6) Position: 10 maxilla/10 mandible Previous endodontic treatment: MD Pulp tests (CPT, HPT, EPT): NA Percussion examination: NA Palpation examination: NA Diagnosis: NA Treated roots Number: n = 20 | DNS step(s) (n = roots): Proximity to noble anatomical/surgically critical structures: MD Osteotomy: n = 20 Apex location: n = 20 Root-end resection: n = 20 FH step(s): Root-end cavity preparation: NO Root-end fill: NO Use of regenerative materials: NA Sutures: NA | Dental impression technique: MD Radiographic imaging: CBCT (n = 20) Planning software: X-Guide® (n = 20) Navigation software: X-Guide® (n = 20) Navigation system: DNS (n = 20) Guidance method for navigation: Radiopaque fiducial markers (X-clip) embedded in thermoplastic stent + real-time tracking via optical motion-tracking cameras (n = 20) | Virtual accuracy metrics in 2D/3D deviations (mm/°) (n = roots): Platform depth deviation: MD Apical depth deviation: MD Angular deflection: 2.54 ± 2.62°; ≤5 mm: 2.7 ±2.1°; >5 mm: 2.44 ± 0.97° (n = 20) Global platform deviation: 0.70 ± 0.19 mm; ≤5 mm: 0.73 ±0.38 mm; >5 mm: 0.68 ± 0.49 mm (n = 20) Global apex deviation: 0.65 ± 0.09 mm; ≤5 mm: 0.63 ± 0.33 mm >5 mm: 0.65 ± 0.27 mm (n = 20) Osteotomy size (mm/mm3) (n = roots): Diameter: MD Volume: MD Depth: 5.31 ± 1.82 mm (n = 20) Height: MD Length: MD Root-end resection (mm/°): Resected root length: 3 mm (n = 20) Residual root length: MD Resection angle: MD | DNS procedural duration (s/min): 212 ± 49 s/~3.5 ± 0.8 min (average per treated root, n = 20) Osteotomy duration: MD Root-end resection duration: MD Root-end preparation duration: MD Root-end fill duration: MD Total surgery duration (DNS + FH) (s/min): MD Dentist level of expertise: EE (n = 1) Dentist-reported feedback: MD Complications (intra- and postoperative), type and rate (n = roots): incomplete root-end resection, 10% (n = 2 roots), all occurred in roots ≤5 mm from buccal cortical plate (2/12, 16.7%) |
| Fu 2022 J Endod [35] Case series General Program of National Natural Science Foundation of China | Population Sample size: n = 3 (alive) Gender ratio: 2 M/1 F Mean/Range age: 26.67 yo/26-27 yo Comorbidities/Ongoing pharmacological treatment: MD/No (n = 1)- MD/MD (n = 2) Tooth/Teeth Treated: n = 3 Type: 1.6 (n = 1), 3.6 (n = 2) Position: 1 maxilla/2 mandible Previous endodontic treatment: Yes; 2 years (n = 1), 4 years (n = 2) Pulp tests (CPT, HPT, EPT): NA Percussion examination: Yes (n = 3) Palpation examination: No (n = 2), MD (n = 1) Diagnosis: apical periodontitis in previously root canal-treated teeth (n = 3) Treated roots Number: n = 7 | DNS step(s) (n = roots): Proximity to noble anatomical/surgically critical structures: Yes, maxillary sinus (n = 3) Osteotomy: n = 7 Apex location: n = 7 Root-end resection: n = 7 FH step(s): Root-end cavity preparation: Endodontic microscope + ultrasonic tips (n = 7) Root-end fill: Endodontic microscope + iRoot BP Plus® (n = 7) Use of regenerative materials: Bio-Oss + Bio-Gide (n = 3) Sutures: Monofilament sutures (n = 7) | Dental impression technique: MD Radiographic imaging: CBCT (n = 7) Planning software: DHC-ENDO1® (n = 7) Navigation software: DHC-ENDO1® (n = 7) Navigation system: DHC-ENDO1® (n = 7) Guidance method for navigation: Marker-based registration via radiopaque fiducial markers embedded in a registration device filled with silicone impression material + real-time infrared optical tracking (n = 7) | Virtual accuracy metrics in 2D/3D deviations (mm/°) (n = roots): Platform depth deviation: MD Apical depth deviation: MD Angular deflection: MD Global platform deviation: MD Global apex deviation: MD Osteotomy size (mm/mm3) (n = roots): Diameter: 4 mm (n = 7) Volume: MD Depth: MD Height: MD Length: MD Root-end resection (mm/°): Resected root length: 3 mm (n = 7) Residual root length: MD Resection angle: MD | DNS procedural duration (s/min): MD Osteotomy duration: MD Root-end resection duration: MD Root-end preparation duration: MD Root-end fill duration: MD Total surgery duration (DNS + FH) (s/min): MD Dentist level of expertise: MD Dentist-reported feedback: MD Follow-up: 3 months (n = 1), 6 months (n = 1), 9 months (n = 1) Outcome measures for healing following surgical endodontics (No. teeth/%): Pain, swelling, and other symptoms/signs: No (n = 3/100%) Satisfactory healing of soft tissue: MD Sinus tract: No, presence of sinus tract described preoperatively and healed postoperatively in n = 2/3 teeth (66.7%); one tooth did not present sinus tract preoperatively Loss of function: MD Radiological evidence of repair of apical periodontitis: Yes (n = 3/100%) Reformation of the periodontal ligament space: MD 1-year follow-up (yes/no): No (n = 3/100%) Complications (intra- and postoperative), type and rate (n = roots): No (n = 7/100%) Patient-reported feedback: MD |
| Gambarini 2019 J Endod [9] Case report None | Population Sample size: n = 1 (alive) Gender ratio: 1 M Mean/Range age: 34 yo Comorbidities/Ongoing pharmacological treatment: MD Tooth/Teeth Treated: n = 1 Type: 1.2 Position: 1 maxilla Previous endodontic treatment: Yes, 3 years (n = 1) Pulp tests (CPT, HPT, EPT): MD Percussion examination: Yes (n = 1) Palpation examination: MD Diagnosis: apical periodontitis in previously root canal-treated teeth (n = 1) Treated roots Number: n = 1 | DNS step(s) (n = roots): Proximity to noble anatomical/surgically critical structures: MD Osteotomy: n = 1 Apex location: n = 1 Root-end resection: n = 1 FH step(s): Root-end cavity preparation: Endodontic microscope + ultrasonic BK3-R tip (n = 1) Root-end fill: Endodontic microscope + EndoSequence® (n = 1) Use of regenerative materials: No Sutures: Resorbable Vicryl Plus 4-0 (n = 1) | Dental impression technique: MD Radiographic imaging: CBCT (n = 1) Planning software: Navident® (n = 1) Navigation software: Navident® (n = 1) Navigation system: Navident® (n = 1) Guidance method for navigation: Intra-oral trace registration with optical stereoscopic tracking (n = 1) | Virtual accuracy metrics in 2D/3D deviations (mm/°) (n = roots): Platform depth deviation: MD Apical depth deviation: MD Angular deflection: MD Global platform deviation: MD Global apex deviation: MD Osteotomy size (mm/mm3) (n = roots): Diameter: ~ 3 mm (n = 1) Volume: MD Depth: MD Height: MD Length: MD Root-end resection (mm/°): Resected root length: 3 mm (n = 1) Residual root length: MD Resection angle: 10° (n = 1) | DNS procedural duration (s/min): MD Osteotomy duration: MD Root-end resection duration: MD Root-end preparation duration: MD Root-end fill duration: MD Total surgery duration (DNS + FH) (s/min): Less than 2700 s/Less than 45 min (n = 1 root) Dentist level of expertise: NE (n = 1, supervised by experienced endodontists) Dentist-reported feedback: The system was described as easy to use, accurate, and supportive for inexperienced users. The learning curve was reported as rapid, and the trace registration was considered easy to learn and perform (n = 1 NE supervised by experienced endodontists) [Excluding cadavers] Follow-up: 1, 3, 6 months (n = 1) Outcome measures for healing following surgical endodontics (No. teeth/%): Pain, swelling, and other symptoms/signs: No (n = 1/100%) Satisfactory healing of soft tissue: MD Sinus tract: No, presence of sinus tract not described preoperatively or postoperatively (n = 1/100%) Loss of function: MD Radiological evidence of repair of apical periodontitis: Yes (n = 1/100%) Reformation of the periodontal ligament space: MD 1-year follow-up (yes/no): No (n = 1/100%) Complications (intra- and postoperative), type and rate (n = roots): No (n = 1) Patient-reported feedback: MD |
| Gibello 2023 Giornale Italiano di Endodonzia [28] Case report None | Population Sample size: n = 1 (alive) Gender ratio: 1 F Mean/Range age: 32 yo Comorbidities/Ongoing pharmacological treatment: No/No (n = 1) Tooth/Teeth Treated: n = 1 Type: 3.6 Position: 1 mandible Previous endodontic treatment: Yes (inadequate), MD (n = 1) Pulp tests (CPT, HPT, EPT): NA Percussion examination: Yes (tenderness to percussion) (n = 1) Palpation examination: MD Diagnosis: Apical periodontitis, due to an inadequate endodontic treatment and a separated instrument (n = 1) Treated roots Number: n = 1 | DNS step(s) (n = roots): Proximity to noble anatomical/surgically critical structures: Yes, inferior alveolar nerve (n = 1) Osteotomy: n = 1 Apex location: n = 1 Root-end resection: n = 1 FH FH step(s): Root-end cavity preparation: Endodontic microscope + ultrasonic tips (n = 1) Root-end fill: Endodontic microscope + EndoSequence® (n = 1) Use of regenerative materials: Collagen sponge placed to support the cortical bone block (n = 1) Sutures: Resorbable Vicryl Plus 6-0 (n = 1) | Dental impression technique: Intra-oral digital impression (.STL) (n = 1) Radiographic imaging: CBCT (n = 1) Planning software: Navident® (n = 1) Navigation software: Navident® (n = 1) Navigation system: Navident® (n = 1) Guidance method for navigation: CBCT (DICOM) and STL matched in planning software + intra-oral trace registration with optical stereoscopic tracking (n = 1) | Virtual accuracy metrics in 2D/3D deviations (mm/°) (n = roots): Platform depth deviation: MD Apical depth deviation: MD Angular deflection: MD Global platform deviation: MD Global apex deviation: MD Osteotomy size (mm/mm3) (n = roots): Diameter: MD Volume: MD Depth: MD Height: MD Length: MD Root-end resection (mm/°): Resected root length: MD Residual root length: MD Resection angle: MD | DNS procedural duration (s/min): MD Osteotomy duration: MD Root-end resection duration: MD Root-end preparation duration: MD Root-end fill duration: MD Total surgery duration (DNS + FH) (s/min): MD Dentist level of expertise: MD Dentist-reported feedback: MD [Excluding cadavers] Follow-up: 4 months and 12 months (n = 1) Outcome measures for healing following surgical endodontics (No. teeth/%): Pain, swelling, and other symptoms/signs: No (n = 1/100%) Satisfactory healing of soft tissue: Yes (n = 1/100%) Sinus tract: No, presence of sinus tract not described preoperatively or postoperatively (n = 1/100%) Loss of function: MD Radiological evidence of repair of apical periodontitis: Yes (n = 1/100%) Reformation of the periodontal ligament space: MD 1-year follow-up (yes/no): Yes (n = 1/100%) Complications (intra- and postoperative), type and rate (n = roots): No (n = 1/100%) Patient-reported feedback: MD |
| Li 2024 J Endod [34] Case series Natural Science Foundation of Guangdong; the Science and Technology Planning Project of Guangzhou, Featured Clinical Technique of Guangzhou and the Plan for enhancing scientific research in GMU | Population Sample size: n = 4 (alive) Gender ratio: 4 F Mean/Range age: 29 yo/24-34 yo Comorbidities/Ongoing pharmacological treatment: MD Tooth/Teeth Treated: n = 4 Type: 2.5, 3.3, 3.4, 3.5 Position: 1 maxilla/3 mandible Previous endodontic treatment: Yes; 2 months (n = 1), 3 years (n = 1), 4 years (n = 1), 5 years (n = 1) Pulp tests (CPT, HPT, EPT): NA Percussion examination: YES (n = 4) Palpation examination: YES (n = 2) Diagnosis: Apical periodontitis in previously root canal-treated teeth (n = 3); apical periodontitis in previously root canal-treated teeth and odontogenic maxillary sinusitis (n = 1) Treated roots Number: n = 4 | DNS step(s) (n = roots): Proximity to noble anatomical/surgically critical structures: mental foramen (n = 1), inferior alveolar nerve (n = 1), maxillary sinus (n = 1), implant and bone graft (n = 1) Osteotomy: n = 4 Apex location: n = 4 Root-end resection: n = 4 FH step(s): Root-end cavity preparation: Endodontic microscope + ultrasonic tips (n = 4) Root-end fill: Endodontic microscope + iRoot BP Plus® (n = 4) Use of regenerative materials: CGF membranes/fragments (n = 3); collagen membrane (Bio-Gide) (n = 1) Sutures: Monofilament sutures 6-0 (n = 4) | Dental impression technique: MD Radiographic imaging: CBCT (n = 4) Planning software: Integrated IRIS-100® (n = 4) Navigation software: Integrated IRIS-100® (n = 4) Navigation system: Integrated IRIS-100® (n = 4) Guidance method for navigation: Radiopaque fiducial markers with silicone-based registration device during CBCT + infrared optical tracking (n = 4) | Virtual accuracy metrics in 2D/3D deviations (mm/°) (n = roots): Platform depth deviation: MD Apical depth deviation: MD Angular deflection: MD Global platform deviation: MD Global apex deviation: MD Osteotomy size (mm/mm3) (n = roots): Diameter: MD Volume: MD Depth: MD Height: MD Length: MD Root-end resection (mm/°): Resected root length: 3 mm (n = 4) Residual root length: MD Resection angle: MD | DNS procedural duration (s/min): MD Osteotomy duration: MD Root-end resection duration: MD Root-end preparation duration: MD Root-end fill duration: MD Total surgery duration (DNS + FH) (s/min): MD Dentist level of expertise: MD Dentist-reported feedback: MD [Excluding cadavers] Follow-up: 3 months (n = 1), 5 months (n = 1), 12 months (n = 2) Outcome measures for healing following surgical endodontics (No. teeth/%): Pain, swelling, and other symptoms/signs: No (n = 4/100%) Satisfactory healing of soft tissue: Yes (n = 4/100%) Sinus tract: No, presence of sinus tract not described preoperatively or postoperatively (n = 4/100%) Loss of function: MD Radiological evidence of repair of apical periodontitis: Yes (n = 4/100%), density increase (n = 1), lesion reduction (n = 1), radiographic healing (n = 2) Reformation of the periodontal ligament space: MD 1-year follow-up (yes/no): Yes (n = 2/50%) Complications (intra- and postoperative), type and rate (n = roots): No (n = 4/100%) Patient-reported feedback: MD |
| Lu 2022 J Dent Sci [39] Case report None | Population Sample size: n = 1 (alive) Gender ratio: 1 F Mean/Range age: 41 yo Comorbidities/Ongoing pharmacological treatment: MD Tooth/Teeth Treated: n = 1 Type: 3.6 Position: 1 mandible Previous endodontic treatment: Yes; months ago (n = 1) Pulp tests (CPT, HPT, EPT): NA Percussion examination: Yes (n = 1) Palpation examination: Yes (n = 1) Diagnosis: Periapical lesion with sinus tract tracing to the distobuccal root after root canal retreatment (n = 1) Treated roots Number: n = 2 | DNS step(s) (n = roots): Proximity to noble anatomical/surgically critical structures: Yes, inferior alveolar nerve (n = 2) Osteotomy: n = 2 Apex location: n = 2 Root-end resection: n = 2 FH step(s): Root-end cavity preparation: Endodontic microscope (n = 2) Root-end fill: Endodontic microscope (n = 2) Use of regenerative materials: The buccal bone plate was intactly removed and repositioned to enhance healing (n = 2) Sutures: MD | Dental impression technique: MD Radiographic imaging: CBCT (n = 2) Planning software: X-Guide® (n = 2) Navigation software: X-Guide® (n = 2) Navigation system: DNS (n = 2) Guidance method for navigation: Radiopaque fiducial markers (X-clip) embedded in thermoplastic stent + real-time tracking via optical motion-tracking cameras (n = 2) | Virtual accuracy metrics in 2D/3D deviations (mm/°) (n = roots): Platform depth deviation: MD Apical depth deviation: MD Angular deflection: MD Global platform deviation: MD Global apex deviation: MD Osteotomy size (mm/mm3) (n = roots): Diameter: MD Volume: MD Depth: MD Height: MD Length: MD Root-end resection (mm/°): Resected root length: MD Residual root length: MD Resection angle: MD | DNS procedural duration (s/min): MD Osteotomy duration: MD Root-end resection duration: MD Root-end preparation duration: MD Root-end fill duration: MD Total surgery duration (DNS + FH) (s/min): MD Dentist level of expertise: MD Dentist-reported feedback: MD [Excluding cadavers] Follow-up: 5 months (n = 1) Outcome measures for healing following surgical endodontics (No. teeth/%): Pain, swelling, and other symptoms/signs: MD Satisfactory healing of soft tissue: MD Sinus tract: No, presence of sinus tract described preoperatively and healed postoperatively (n = 1/100%) Loss of function: MD Radiological evidence of repair of apical periodontitis: Yes (n = 1/100%), bone healing at the periapical area Reformation of the periodontal ligament space: MD 1-year follow-up (yes/no): NO (n = 1/100%) Complications (intra- and postoperative), type and rate (n = roots): No (n = 2/100%) Patient-reported feedback: MD |
| Manishaa 2024 Endodontology [29] Case series None | Population Sample size: n = 2 (alive) Gender ratio: 2 F Mean/Range age: 18 yo Comorbidities/Ongoing pharmacological treatment: MD Tooth/Teeth Treated: n = 2 Type: 1.2, 1.4 Position: 2 maxilla Previous endodontic treatment: Yes, 2 weeks prior (n = 1); no (n = 1) Pulp tests (CPT, HPT, EPT): NA (n = 1); yes (CPT, EPT) (n = 1) Percussion examination: Yes (n = 2) Palpation examination: MD Diagnosis: Apical periodontitis in an endodontically treated tooth with separated instruments (n = 1); apical periodontitis (n = 1) Treated roots Number: n = 2 | DNS step(s) (n = roots): Proximity to noble anatomical/surgically critical structures: MD Osteotomy: n = 2 Apex location: n = 2 Root-end resection: n = 2 FH step(s): Root-end cavity preparation: Endodontic microscope + S12-70D ultrasonic instrument (n = 2) Root-end fill: Endodontic microscope + MTA (n = 2) Use of regenerative materials: i-PRF + Osseograft used for defect filling and bone regeneration (n = 1) Sutures: Resorbable Vicryl Plus 4-0 (n = 2) | Dental impression technique: Digital model scanning (STL files from scanned physical impressions) (n = 2) Radiographic imaging: CBCT (n = 2) Planning software: Navident® (n = 2) Navigation software: Navident® (n = 2) Navigation system: Navident® (n = 2) Guidance method for navigation: CBCT (DICOM) and STL matched in planning software + Intra-oral trace registration with optical stereoscopic tracking (n = 2) | Virtual accuracy metrics in 2D/3D deviations (mm/°) (n = roots): Platform depth deviation: MD Apical depth deviation: MD Angular deflection: MD Global platform deviation: MD Global apex deviation: MD Osteotomy size (mm/mm3) (n = roots): Diameter: MD Volume: MD Depth: MD Height: MD Length: MD Root-end resection (mm/°): Resected root length: MD Residual root length: MD Resection angle: MD | DNS procedural duration (s/min): MD Osteotomy duration: MD Root-end resection duration: MD Root-end preparation duration: MD Root-end fill duration: MD Total surgery duration (DNS + FH) (s/min): MD Dentist level of expertise: MD Dentist-reported feedback: MD Follow-up: 1 month, 12 months (n = 2) Outcome measures for healing following surgical endodontics (No. teeth/%): Pain, swelling, and other symptoms/signs: No (n = 2/100%) Satisfactory healing of soft tissue: Yes (n = 1/50%) Sinus tract: No, presence of sinus tract not described preoperatively or postoperatively (n = 2/100%) Loss of function: No (n = 2/100%) Radiological evidence of repair of apical periodontitis: Yes (n = 2/100%) Reformation of the periodontal ligament space: MD 1-year follow-up (yes/no): Yes (n = 2/100%) Complications (intra- and postoperative), type and rate (n = roots): No (n = 2/100%) Patient-reported feedback: High satisfaction |
| Martinho 2022 J Endod [30] Randomized controlled trial AAE Foundation for Endodontics | Population Sample size: n = 4 (cadavers) Gender ratio: MD Mean/Range age: MD Comorbidities/Ongoing pharmacological treatment: NA Tooth/Teeth Treated: n = 38 Type: Anterior and canine n = 16, premolars n = 12, molars n = 10 Position: 18 maxilla/20 mandible Previous endodontic treatment: MD Pulp tests (CPT, HPT, EPT): NA Percussion examination: NA Palpation examination: NA Diagnosis: MD Treated roots Number: n = 38 | DNS step(s) (n = roots): Proximity to noble anatomical/surgically critical structures: MD Osteotomy: n = 38 Apex location: n = 38 Root-end resection: n = 38 FH step(s): Root-end cavity preparation: No Root-end fill: No Use of regenerative materials: NA Sutures: No | Dental impression technique: MD Radiographic imaging: CBCT (n = 38) Planning software: X-Guide® (n = 38) Navigation software: X-Guide® (n = 38) Navigation system: DNS (n = 38) Guidance method for navigation: Radiopaque fiducial markers (X-clip) embedded in thermoplastic stent + real-time tracking via optical motion-tracking cameras (n = 38) | Virtual accuracy metrics in 2D/3D deviations (mm/°) (n = roots): Platform depth deviation: EE: 0.8 ± 0.3 mm (n = 19), NE: 1.7 ± 0.6 mm (n = 19) Apical depth deviation: EE: 0.78 ± 0.5 mm (n = 19), NE: 1.5 ± 1.1 mm (n = 19) Angular deflection: EE: 1.3 ± 0.9° (n = 19), NE: 2.5 ± 0.8° (n = 19) Global platform deviation: EE: 0.70 ± 0.2 mm (n = 19), NE: 1.0 ± 0.4 mm (n = 19) Global apex deviation: EE: 0.66 ± 0.5 mm (n = 19), NE: 1.2 ± 0.5 mm (n = 19) Osteotomy size (mm/mm3) (n = roots): Diameter: MD Volume: MD Depth: MD Height: MD Length: MD Root-end resection (mm/°): Resected root length: 3 mm (n = 38) Residual root length: MD Resection angle: MD | DNS procedural duration (s/min): EE: 257 ± 90 s/~4.3 ± 1.5 min (average per treated root, n = 19 roots), NE: 460 ± 50 s/~7.7 ± 0.8 min (average per treated root, n = 19 roots) Osteotomy duration: MD Root-end resection duration: MD Root-end preparation duration: NO Root-end fill duration: No Total surgery duration (DNS + FH) (s/min): NA (no root-end cavity preparation and root-end fill) Dentist level of expertise: EE (n = 1), NE (n = 1) Dentist-reported feedback: MD Complications (intra- and postoperative), type and rate (n = roots): Type of complication not specified, 5.26% (1/19 roots) in EE and 5.26% (1/19 roots) in NE Patient-reported feedback: NA |
| Martinho 2023 J Endod [32] Randomized controlled trial American Association of Endodontists Foundation | Population Sample size: n = MD (cadavers) Gender ratio: MD Mean/Range age: MD Comorbidities/Ongoing pharmacological treatment: NA Tooth/Teeth Treated: MD Type: MD Position: MD Previous endodontic treatment: MD Pulp tests (CPT, HPT, EPT): NA Percussion examination: NA Palpation examination: MD Diagnosis: MD Treated roots Number: n = 25 | DNS step(s) (n = roots): Proximity to noble anatomical/surgically critical structures: MD Osteotomy: n = 25 Apex location: n = 25 Root-end resection: n = 25 FH step(s): Root-end cavity preparation: Endodontic microscope + ProUltra ultrasonic surgical tips (n = 25) Root-end fill: Endodontic microscope + EndoSequence® (n = 25) Use of regenerative materials: NA Sutures: No | Dental impression technique: MD Radiographic imaging: CBCT (n = 25) Planning software: X-Guide® (n = 25) Navigation software: X-Guide® (n = 25) Navigation system: DNS (n = 25) Guidance method for navigation: Radiopaque fiducial markers (X-clip) embedded in thermoplastic stent + real-time tracking via optical motion-tracking cameras (n = 25) | Virtual accuracy metrics in 2D/3D deviations (mm/°) (n = roots): Platform depth deviation: 1.13 ± 0.47 mm (n = 25) Apical depth deviation: 1.28 ± 0.64 mm (n = 25) Angular deflection: 1.94 ± 0.22 ° (n = 25) Global platform deviation: 1.00 ± 0.28 mm (n = 25) Global apex deviation: 1.14 ± 0.25 mm (n = 25) Osteotomy size (mm/mm3) (n = roots): Diameter: MD Volume: 82.27 ± 29.33 mm3 (n = 25) Depth: MD Height: MD Length: MD Root-end resection (mm/°): Resected root length: 3.17 ± 0.59 mm (n = 25) Residual root length: MD Resection angle: 5.66 ± 2.1° (n = 25) | DNS procedural duration (s/min): 280 ± 71 s/~4.7 ± 1.2 min (average per treated root, n = 25 roots) Osteotomy duration: MD Root-end resection duration: MD Root-end preparation duration: MD Root-end fill duration: MD Total surgery duration (DNS + FH) (s/min): MD Dentist level of expertise: EE (n = MD) Dentist-reported feedback: MD Complications (intra- and postoperative), type and rate (n = roots): Incomplete root-end resection, 16% (n = 1) Patient-reported feedback: NA |
| Villa-Machado 2024 Int Endod J [36] Case report None | Population Sample size: n = 1 (alive) Gender ratio: 1 M Mean/Range age: 58 yo Comorbidities/Ongoing pharmacological treatment: No/No (n = 1) Tooth/Teeth Treated: n = 1 Type: 2.6 Position: 1 maxilla Previous endodontic treatment: Yes, 2 years (n = 1) Pulp tests (CPT, HPT, EPT): NA Percussion examination: Yes (n = 1) Palpation examination: Yes (n = 1) Diagnosis: Apical periodontitis in a previously root canal-treated tooth (n = 1) Treated roots Number: n = 3 | DNS step(s) (n = roots): Proximity to noble anatomical/surgically critical structures: Maxillary sinus (n = 3) Osteotomy: n = 3 Apex location: n = 3 Root-end resection: n = 3 FH step(s): Root-end cavity preparation: Endodontic microscope + diamond-coated ultrasonic microtips (n = 3) Root-end fill: Endodontic microscope + EndoSequence® (n = 3) Use of regenerative materials: Autologous PRF for bone regeneration and sinus protection (n = 3) Sutures: Monofilament sutures 6-0 (n = 3) | Dental impression technique: MD Radiographic imaging: CBCT (n = 3) Planning software: Navident® (n = 3) Navigation software: Navident® (n = 3) Navigation system: Navident® (n = 3) Guidance method for navigation: Intra-oral trace registration with optical stereoscopic tracking (n = 3) | Virtual accuracy metrics in 2D/3D deviations (mm/°) (n = roots): Platform depth deviation: MD Apical depth deviation: MD Angular deflection: MD Global platform deviation: MD Global apex deviation: MD Osteotomy size (mm/mm3) (n = roots): Diameter: MD Volume: MD Depth: MD Height: MD Length: MD Root-end resection (mm/°): Resected root length: 3 mm (n = 3) Residual root length: MD Resection angle: <10° (n = 3) | DNS procedural duration (s/min): MD Osteotomy duration: MD Root-end resection duration: MD Root-end preparation duration: MD Root-end fill duration: MD Total surgery duration (DNS + FH) (s/min): MD Dentist level of expertise: EE (n = 2) Dentist-reported feedback: MD Follow-up: every 3 months; clinical assessments reported at 3, 6, and 24 months (n = 1) Outcome measures for healing following surgical endodontics (No. teeth/%): Pain, swelling, and other symptoms/signs: No (n = 1/100%) Satisfactory healing of soft tissue: MD Sinus tract: No, presence of sinus tract not described preoperatively or postoperatively (n = 1/100%) Loss of function: MD Radiological evidence of repair of apical periodontitis: Yes (n = 1/100%) Reformation of the periodontal ligament space: Yes (n = 1/100%), CBCT at 24 months showed normal PDL space around all roots 1-year follow-up (yes/no): No (n = 1/100%) Complications (intra- and postoperative), type and rate (n = roots): No (n = 3) Patient-reported feedback: MD |
| Parameter | Mean Value | Standard Deviation | |
|---|---|---|---|
| Platform depth deviation | 1.17 | 0.84 | mm |
| Global platform deviation | 0.83 | 0.34 | mm |
| Angular deflection | 2.29 | 1.69 | ° |
| Osteotomy diameter | 3.98 | mm | |
| Osteotomy depth | 6.06 | 2.14 | mm |
| Osteotomy height | 3.72 | 0.67 | mm |
| Osteotomy length | 4.05 | 0.13 | mm |
| Osteotomy volume | 82.32 | 47.83 | mm3 |
| Apical depth deviation | 1.21 | 0.99 | mm |
| Global apex deviation | 0.98 | 0.79 | mm |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Di Spirito, F.; Gasparro, R.; Di Palo, M.P.; De Benedetto, G.; Giordano, F.; Amato, M.; Bramanti, A. Dynamic Navigation in Endodontic Surgery: A Systematic Review. Healthcare 2025, 13, 2151. https://doi.org/10.3390/healthcare13172151
Di Spirito F, Gasparro R, Di Palo MP, De Benedetto G, Giordano F, Amato M, Bramanti A. Dynamic Navigation in Endodontic Surgery: A Systematic Review. Healthcare. 2025; 13(17):2151. https://doi.org/10.3390/healthcare13172151
Chicago/Turabian StyleDi Spirito, Federica, Roberta Gasparro, Maria Pia Di Palo, Giuseppina De Benedetto, Francesco Giordano, Massimo Amato, and Alessia Bramanti. 2025. "Dynamic Navigation in Endodontic Surgery: A Systematic Review" Healthcare 13, no. 17: 2151. https://doi.org/10.3390/healthcare13172151
APA StyleDi Spirito, F., Gasparro, R., Di Palo, M. P., De Benedetto, G., Giordano, F., Amato, M., & Bramanti, A. (2025). Dynamic Navigation in Endodontic Surgery: A Systematic Review. Healthcare, 13(17), 2151. https://doi.org/10.3390/healthcare13172151







