Prevalence and Risk Factors of Pregnancy-Specific Urinary Incontinence: Findings from the Diamater Cohort Study
Abstract
1. Introduction
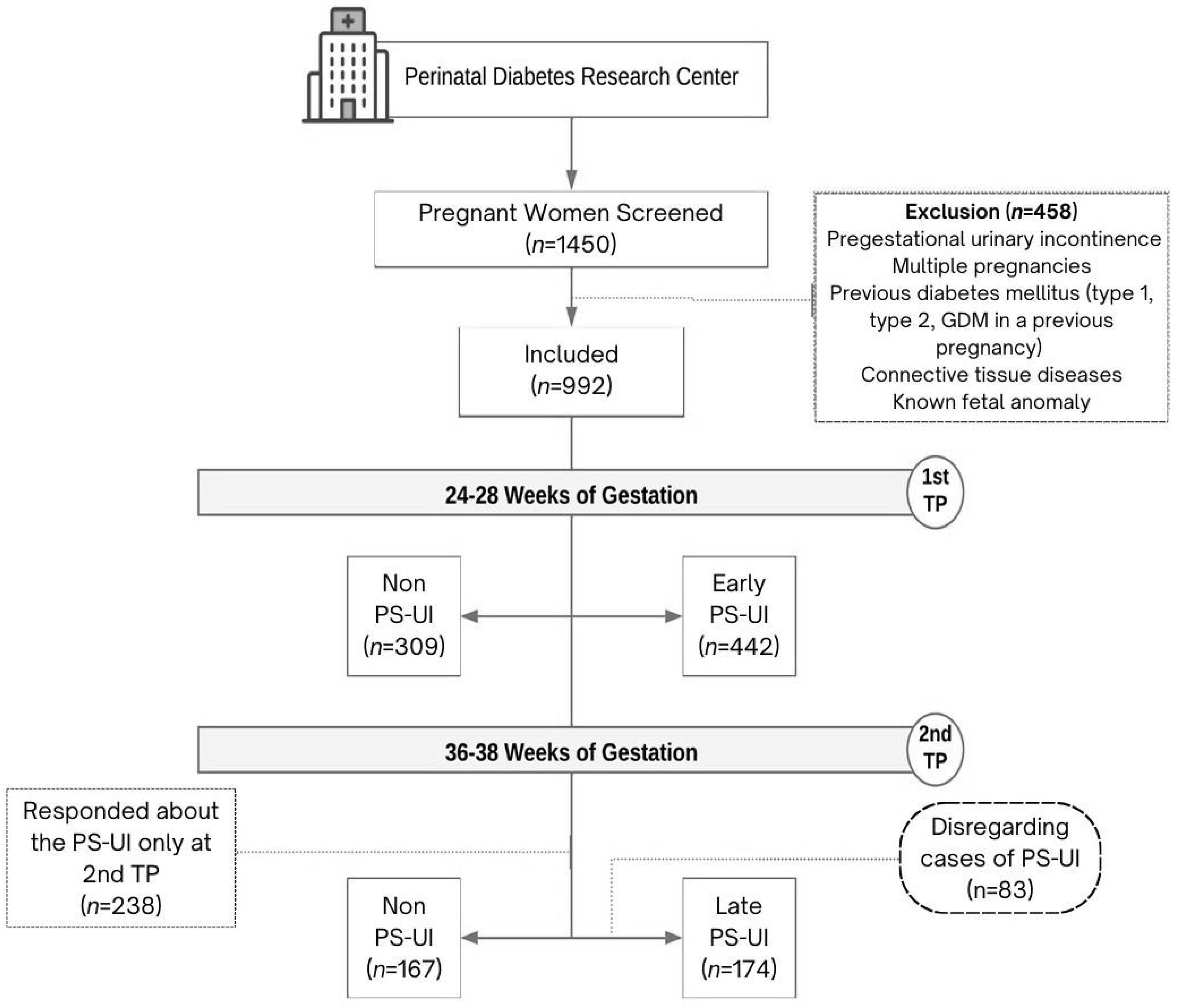
2. Method
2.1. Research Design and Subjects
2.2. Data Collection
2.3. Statistical Analysis
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| UI | urinary incontinence |
| PS-UI | pregnancy-specific urinary incontinence |
| GDM | gestational diabetes mellitus |
| OGTT | oral glucose tolerance test |
| BMI | body mass index |
References
- Ali, H.; Ahmed, A.; Olivos, C.; Khamis, K.; Liu, J. Mitigating urinary incontinence conditions using machine learning. BMC Med. Inf. Decis. Mak. 2022, 22, 243. [Google Scholar] [CrossRef]
- Irwin, D.E.; Kopp, Z.S.; Agatep, B.; Milsom, I.; Abrams, P. Worldwide prevalence estimates of lower urinary tract symptoms, overactive bladder, urinary incontinence, and bladder outlet obstruction. BJU Int. 2011, 108, 1132–1138. [Google Scholar] [CrossRef] [PubMed]
- Datar, M.; Pan, L.C.; McKinney, J.L.; Goss, T.F.; Pulliam, S.J. Healthcare resource use and cost burden of urinary incontinence to United States payers. Neurourol. Urodyn. 2022, 41, 1553–1562. [Google Scholar] [CrossRef]
- Hvidman, L.; Foldspang, A.; Mommsen, S.; Bugge Nielsen, J. Correlates of urinary incontinence in pregnancy. Int. Urogynecol. J. 2002, 13, 278–283. [Google Scholar] [CrossRef] [PubMed]
- Wesnes, S.L.; Rortveit, G.; Bø, K.; Hunskaar, S. Urinary incontinence during pregnancy. Obs. Gynecol. 2007, 109, 922–928. [Google Scholar] [CrossRef]
- Chang, S.R.; Lin, W.A.; Chang, T.C.; Lin, H.H.; Lee, C.N.; Lin, M.I. Risk factors for stress and urge urinary incontinence during pregnancy and the first year postpartum: A prospective longitudinal study. Int. Urogynecol. J. 2021, 32, 2455–2464. [Google Scholar] [CrossRef] [PubMed]
- Zhu, L.; Li, L.; Lang, J.H.; Xu, T. Prevalence and risk factors for peri- and postpartum urinary incontinence in primiparous women in China: A prospective longitudinal study. Int. Urogynecol. J. 2012, 23, 563–572. [Google Scholar] [CrossRef]
- Wang, X.; Jin, Y.; Xu, P.; Feng, S. Urinary incontinence in pregnant women and its impact on health-related quality of life. Health Qual. Life Outcomes 2022, 20, 13. [Google Scholar] [CrossRef]
- Nur Farihan, M.; Ng, B.K.; Phon, S.E.; Nor Azlin, M.I.; Nur Azurah, A.G.; Lim, P.S. Prevalence, Knowledge and Awareness of Pelvic Floor Disorder among Pregnant Women in a Tertiary Centre, Malaysia. Int. J. Environ. Res. Public Health 2022, 19, 8314. [Google Scholar] [CrossRef]
- Hage-Fransen, M.A.; Wiezer, M.; Otto, A.; Wieffer-Platvoet, M.S.; Slotman, M.H.; Nijhuis-van der Sanden, M.W.; Pool-Goudzwaard, A.L. Pregnancy- and obstetric-related risk factors for urinary incontinence, fecal incontinence, or pelvic organ prolapse later in life: A systematic review and meta-analysis. Acta Obstet. Gynecol. Scand. 2021, 100, 373–382. [Google Scholar] [CrossRef]
- Daly, D.; Clarke, M.; Begley, C. Urinary incontinence in nulliparous women before and during pregnancy: Prevalence, incidence, type, and risk factors. Int. Urogynecol. J. 2018, 29, 353–362. [Google Scholar] [CrossRef] [PubMed]
- Piculo, F.; Marini, G.; Vesentini, G.; Morceli, G.; Damasceno, D.C.; Sobrevia, L.; Barbosa, A.M.; Rudge, M.V. Pregnancy-specific urinary incontinence in women with gestational hyperglycemia worsens the occurrence and severity of urinary incontinence and quality of life over the first year postpartum. Eur. J. Obstet. Gynecol. Reprod. Biol. 2020, 252, 336–343. [Google Scholar] [CrossRef]
- Mason, L.; Glenn, S.; Walton, I.; Hughes, C. Women’s reluctance to seek help for stress incontinence during pregnancy and following childbirth. Midwifery 2001, 17, 212–221. [Google Scholar] [CrossRef]
- Anyanwu, L.-J.J.C.; Anyanwu, O.M.; Yakubu, A.A. Missed opportunities for breast awareness information among women attending the maternal and child health services of an urban tertiary hospital in Northern Nigeria. J. Cancer Res. Ther. 2016, 12, 765–769. [Google Scholar] [CrossRef]
- Nagraj, S.; Kennedy, S.H.; Norton, R.; Jha, V.; Praveen, D.; Hinton, L.; Hirst, J.E. Cardiometabolic Risk Factors in Pregnancy and Implications for Long-Term Health: Identifying the Research Priorities for Low-Resource Settings. Front. Cardiovasc. Med. 2020, 7, 40. [Google Scholar] [CrossRef] [PubMed]
- Tudor Car, L.; Van Velthoven, M.H.; Brusamento, S.; Elmoniry, H.; Car, J.; Majeed, A.; Tugwell, P.; Welch, V.; Marusic, A.; Atun, R. Integrating prevention of mother-to-child HIV transmission programs to improve uptake: A systematic review. PLoS ONE 2012, 7, e35268. [Google Scholar] [CrossRef]
- Rudge, M.V.; Souza, F.P.; Abbade, J.F.; Hallur, R.L.; Marcondes, J.P.C.; Piculo, F.; Marini, G.; Vesentini, G.; Thabane, L.; Witkin, S.S.; et al. Study protocol to investigate biomolecular muscle profile as predictors of long-term urinary incontinence in women with gestational diabetes mellitus. BMC Pregnancy Childbirth 2020, 20, 117. [Google Scholar] [CrossRef]
- Rudge, M.V.C.; Calderon, I.D.M.P.; Ramos, M.D.; Brasil, M.A.M.; Rugolo, L.M.S.S.; Bossolan, G.; Odland, J.Ø. Hiperglicemia materna diária diagnosticada pelo perfil glicêmico: Um problema de saúde pública materno e perinatal. Rev. Bras. Ginecol. Obstet. 2005, 27, 691–697. [Google Scholar] [CrossRef]
- Metzger, B.E.; Gabbe, S.G.; Persson, B.; Lowe, L.P.; Dyer, A.R.; Oats, J.J.; Buchanan, T.A. International association of diabetes and pregnancy study groups recommendations on the diagnosis and classification of hyperglycemia in pregnancy. Diabetes Care 2010, 33, 676–682. [Google Scholar] [CrossRef] [PubMed]
- Abrams, P.; Cardozo, L.; Fall, M.; Griffiths, D.; Rosier, P.; Ulmsten, U.; Van Kerrebroeck, P.; Victor, A.; Wein, A. The standardization of terminology in lower urinary tract function: Report from the standardization sub-committee of the International Continence Society. Urology 2003, 61, 37–49. [Google Scholar] [CrossRef] [PubMed]
- Pizzoferrato, A.C.; Briant, A.R.; Le Grand, C.; Gaichies, L.; Fauvet, R.; Fauconnier, A.; Fritel, X. Influence of prenatal urinary incontinence and mode of delivery in postnatal urinary incontinence: A systematic review and meta-analysis. J. Gynecol. Obstet. Hum. Reprod. 2023, 52, 102536. [Google Scholar] [CrossRef]
- Tahra, A.; Bayrak, O.; Dmochowski, R. The Epidemiology and Population-Based Studies of Women with Lower Urinary Tract Symptoms: A Systematic Review. Türk Urol. Derg./Turk. J. Urol. 2022, 48, 155–165. [Google Scholar] [CrossRef]
- Fitzgerald, M.P.; Brensinger, C.M. What We Don’t Know About Pelvic Floor Disorders in Women. Obstet. Gynecol. Clin. N. Am. 2021, 48, 665–678. [Google Scholar] [CrossRef]
- Lamerton, T.J.; Mielke, G.I.; Brown, W.J. Urinary incontinence in young women: Risk factors, management strategies, help-seeking behavior, and perceptions about bladder control. Neurourol. Urodyn. 2020, 39, 2284–2292. [Google Scholar] [CrossRef] [PubMed]
- Baruch, Y.; Manodoro, S.; Barba, M.; Cola, A.; Re, I.; Frigerio, M. Prevalence and Severity of Pelvic Floor Disorders during Pregnancy: Does the Trimester Make a Difference? Healthcare 2023, 11, 1096. [Google Scholar] [CrossRef] [PubMed]
- Ferreira, B.L.; Souza, R.T.; Dutra, L.O.; Moura, C.S. Urinary incontinence in obese women: A systematic review and meta-analysis. Int. Urogynecol. J. 2020, 31, 2215–2224. [Google Scholar] [CrossRef] [PubMed]
- Subak, L.L.; Wing, R.; West, D.S.; Franklin, F.; Vittinghoff, E.; Creasman, J.M.; Richter, H.E.; Myers, D.; Burgio, K.L.; Gorin, A.A.; et al. Weight Loss to Treat Urinary Incontinence in Overweight and Obese Women. N. Engl. J. Med. 2009, 360, 481–490. [Google Scholar] [CrossRef] [PubMed]
- Yazdany, T.; Jakus-Waldman, S.; Jeppson, P.C.; Schimpf, M.O.; Yurteri-Kaplan, L.A.; Ferzandi, T.R.; Weber-LeBrun, E.; Knoepp, L.; Mamik, M.; Viswanathan, M.; et al. American Urogynecologic Society Systematic Review: The Impact of Weight Loss Intervention on Lower Urinary Tract Symptoms and Urinary Incontinence in Overweight and Obese Women. Female Pelvic Med. Reconstr. Surg. 2020, 26, 16–29. [Google Scholar] [CrossRef]
| Variable | Non-PS-UI | PS-UI | p-Value | ||
|---|---|---|---|---|---|
| Partnership status | Married | 311 (83.4%) | 515 (83.6%) | 0.926 | |
| Not married | 62 (16.6%) | 101 (16.4%) | |||
| Education level | basic level | 27 (7.2%) | 42 (6.9%) | 0.014 | |
| high school | 229 (61.1%) | 427 (69.7%) | |||
| college/university | 119 (31.7%) | 144 (23.5%) | |||
| Ethnicity | Caucasian | 299 (81%) | 495 (80.5%) | 0.835 | |
| Non-Caucasian | 70 (19%) | 120 (19.5%) | |||
| Pregestational BMI (kg/m2) | 26.9 ± 6.25 | 28.48 ± 7.28 | 0.002 | ||
| BMI–1st TP (kg/m2) | 29.4 ± 6.04 | 30.8 ± 6.85 | 0.004 | ||
| BMI–2nd TP (kg/m2) | 31.6 ± 6.20 | 33.75 ± 6.51 | 0.002 | ||
| Gestational weight gain–1st TP (kg) | 6.39 ± 5.04 | 6.08 ± 6.19 | 0.477 | ||
| Gestational weight gain–2nd TP (kg) | 11.76 ± 7.34 | 11.36 ± 7.63 | 0.613 | ||
| Fasting blood glucose (mg/dL) | 79.48 ± 12.71 | 80.11 ± 14.44 | 0.583 | ||
| OGTT–fasting (mg/dL) | 74.82 ± 12.66 | 76.11 ± 11.06 | 0.161 | ||
| OGTT–1 h (mg/dL) | 116.7 ± 31.27 | 119.17 ± 32.7 | 0.321 | ||
| OGTT–2 h (mg/dL) | 105.08 ± 28.1 | 107.01 ± 8.88 | 0.378 | ||
| Chronic coughing | 0 | 9 (1.8%) | 0.025 | ||
| Constipation | 91 (31.5%) | 152 (28.9%) | 0.439 | ||
| Fecal incontinence | 3 (1.1%) | 6 (1.2%) | 0.887 | ||
| Previous arterial hypertension | 24 (8.3%) | 53 (10.2%) | 0.376 | ||
| Alcohol consumption | 1 (0.4%) | 9 (1.8%) | 0.091 | ||
| Smoking in pregnancy | 12 (4.3%) | 26 (5%) | 0.615 | ||
| Physical activity | 66 (23.1%) | 76 (14.7%) | 0.003 | ||
| Gestational diabetes mellitus | 81 (22%) | 154 (25.2%) | 0.242 | ||
| Pregnancy-induced hypertension | 2 (3%) | 8 (4.4%) | 0.611 | ||
| Urinary tract infection | 7 (10.8%) | 22 (12.4%) | 0.735 | ||
| Weeks of Gestation | |||
|---|---|---|---|
| n | % | ||
| 24–28 weeks | |||
| 1st TP | (1) non-PS-UI | 309 | 41.15 |
| (2) Early PS-UI | 442 | 58.85 | |
| 36–38 weeks | |||
| 2nd TP | (1) non-PS-UI | 167 | 49.0 |
| (2) Late PS-UI | 174 | 51.0 | |
| 1st and 2nd TP | |||
| (1) non-PS-UI | 376 | 37.9 | |
| (2) PS-UI | 616 | 62.1 |
| Variable | 1st TP | 2nd TP | |||||
|---|---|---|---|---|---|---|---|
| Non-PS-UI (n = 309) | Early PS-UI (n = 442) | p-Value | Non-PS-UI (n = 167) | Late PS-UI (n = 174) | p-Value | ||
| Partnership status | Married | 261 (84.5%) | 367 (83%) | 0.601 | 136 (82.4%) | 148 (85.1%) | 0.511 |
| Not married | 48 (15.5%) | 75 (17%) | 29 (17.6%) | 26 (14.9%) | |||
| Education level | basic level | 22 (7.1%) | 27 (6.1%) | 0.007 | 11 (6.6%) | 15 (8.7%) | 0.210 |
| high school | 191 (61.8%) | 319 (72.5%) | 93 (55.7%) | 108 (62.4%) | |||
| college/university | 96 (31.1%) | 94 (21.4%) | 63 (5.2%) | 50 (28.9%) | |||
| Ethnicity | Caucasian | 253 (82.4%) | 365 (82.6%) | 0.952 | 124 (76.1%) | 130 (75.1%) | 0.843 |
| Non-Caucasian | 54 (17.6%) | 77 (17.4%) | 39 (23.9%) | 43 (24.9%) | |||
| Pregestational BMI (kg/m2) | 27.04 ± 6.28 | 28.5 ± 7.33 | 0.003 | 27.31 ± 6.42 | 29.47 ± 6.69 | 0.003 | |
| BMI–1st TP (kg/m2) | 29.49 ± 6.1 | 30.83 ± 6.87 | 0.005 | 29.58 ± 6.8 | 30.24 ± 6.64 | 0.637 | |
| BMI–2nd TP (kg/m2) | 31.23 ± 6.76 | 33.49 ± 6.33 | 0.213 | 31.67 ± 6.2 | 33.92 ± 6.64 | 0.002 | |
| Gestational weight gain–1st TP (kg) | 6.31 ± 5.01 | 6.09 ± 6.26 | 0.614 | 6.18 ± 7.73 | 5.85 ± 4.97 | 0.833 | |
| Gestational weight gain–2nd TP (kg) | 10.81 ± 5.55 | 10.62 ± 8.76 | 0.863 | 11.6 ± 8.18 | 11.81 ± 6.83 | 0.802 | |
| Fasting blood glucose (mg/dL) | 79.86 ± 12.4 | 79.42 ± 12.23 | 0.691 | 81.63 ± 13.26 | 82.12 ± 19.37 | 0.826 | |
| OGTT–fasting (mg/dL) | 74.58 ± 12.83 | 75.77 ± 10.58 | 0.206 | 78.46 ± 11.73 | 77.84 ± 13.2 | 0.755 | |
| OGTT–1 h (mg/dL) | 115.65 ± 29.98 | 118.24 ± 32.9 | 0.311 | 126.02 ± 35.13 | 123.81 ± 31.5 | 0.755 | |
| OGTT–2 h (mg/dL) | 105.08 ± 26.77 | 106.19 ± 28.86 | 0.615 | 112.05 ± 33.39 | 111.15 ± 28.87 | 0.862 | |
| Chronic coughing | 0 | 1 (0.3%) | 0.426 | 0 | 8 (6.7%) | 0.012 | |
| Constipation | 79 (31.6%) | 113 (28.7%) | 0.430 | 27 (27.8%) | 39 (29.5%) | 0.778 | |
| Fecal incontinence | 1 (0.4%) | 3 (0.8%) | 0.568 | 2 (2.2%) | 3 (2.5%) | 0.879 | |
| Previous arterial hypertension | 17 (6.8%) | 29 (7.3%) | 0.795 | 12 (12.4%) | 24 (19.4%) | 0.163 | |
| Alcohol | 1 (0.4%) | 4 (1%) | 0.391 | 0 | 5 (4.2%) | 0.047 | |
| Smoking | 8 (3.2%) | 19 (4.8%) | 0.328 | 6 (6.6%) | 7 (5.9%) | 0.832 | |
| Physical activity | 53 (21.4%) | 51 (12.8%) | 0.004 | 22 (22.9%) | 25 (20.7%) | 0.689 | |
| Gestational diabetes mellitus | 70 (23.0%) | 103 (23.5%) | 0.877 | 116 (70.7%) | 121 (70.3%) | 0.939 | |
| Pregnancy-induced hypertension | 0 | 3 (4.2%) | 0.185 | 3 (3.8%) | 5 (4.6%) | 0.778 | |
| Urinary infection | 1 (2.6%) | 7 (9.5%) | 0.184 | 9 (11.3%) | 15 (14.4%) | 0.526 | |
| Variable | Early PS-UI (n = 442) | Late PS-UI (n = 442) | PS-UI (1st plus 2nd TP) | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| OR * | 95% CI ** | p-Value | OR * | 95% CI ** | p-Value | OR * | 95% CI ** | p-Value | |||||
| Education level | basic level | ||||||||||||
| high school | 1.28 | 0.62–2.63 | 0.172 | 0.62 | 0.04 | 9.28 | 0.491 | 1.21 | 0.58–2.51 | 0.304 | |||
| college/university | 0.89 | 0.40–1–96 | 0.359 | 1.20 | 0.08–19.02 | 0.629 | 0.91 | 0.41–2.04 | 0.493 | ||||
| Ethnicity | Caucasian | 1.22 | 0.76–1.96 | 0.416 | 2.65 | 0.46–15.15 | 0.274 | 1.28 | 0.88–0.78 | 0.519 | |||
| Non-Caucasian | |||||||||||||
| Age (years) | 0.99 | 0.96–1.02 | 0.375 | 1.01 | 0.92–1.12 | 0.805 | 0.99 | 0.96–1.03 | 0.706 | ||||
| Pregestational BMI | 1.03 | 1.01–1.06 | 0.014 | 1.02 | 0.93–1.11 | 0.716 | 1.04 | 1.01–1.07 | 0.006 | ||||
| Constipation | 1.21 | 0.84–0.83 | 0.312 | 1.02 | 0.26–4.03 | 0.974 | 0.78 | 0.54–1.13 | 0.193 | ||||
| Fecal incontinence | 3.02 | 0.29–31.82 | 0.358 | 2.81 | 0.26–29.92 | 0.392 | |||||||
| Previous arterial hypertension | 0.83 | 0.41–1.70 | 0.616 | 1.4 | 0.16–12.32 | 0.761 | 0.84 | 0.41–1.76 | 0.652 | ||||
| Smoking | 2.08 | 0.78–5.53 | 0.141 | 1.85 | 0.69–4.91 | 0.220 | |||||||
| Physical activity | 0.51 | 0.32–0.80 | 0.003 | 0.72 | 0.13–3.88 | 0.700 | 0.5 | 0.32–0.79 | 0.003 | ||||
| Gestational diabetes mellitus | 1.10 | 1.01–1.06 | 0.649 | 0.94 | 0.25–3.58 | 0.928 | 1.16 | 0.75–1.81 | 0.500 | ||||
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bassin, H.C.M.; Barbosa, A.M.P.; Prudencio, C.B.; Sobrevia, L.; Barbosa, V.P.; Nunes, S.K.; Rossignoli, P.d.S.; Pedroni, C.R.; França, D.C.H.; Catinelli, B.B.; et al. Prevalence and Risk Factors of Pregnancy-Specific Urinary Incontinence: Findings from the Diamater Cohort Study. Healthcare 2025, 13, 2141. https://doi.org/10.3390/healthcare13172141
Bassin HCM, Barbosa AMP, Prudencio CB, Sobrevia L, Barbosa VP, Nunes SK, Rossignoli PdS, Pedroni CR, França DCH, Catinelli BB, et al. Prevalence and Risk Factors of Pregnancy-Specific Urinary Incontinence: Findings from the Diamater Cohort Study. Healthcare. 2025; 13(17):2141. https://doi.org/10.3390/healthcare13172141
Chicago/Turabian StyleBassin, Henrique Caetano Mingoranci, Angélica Mércia Pascon Barbosa, Caroline Baldini Prudencio, Luis Sobrevia, Vitoria Pascon Barbosa, Sthefanie Kenickel Nunes, Patrícia de Souza Rossignoli, Cristiane Rodrigues Pedroni, Danielle Cristina Honório França, Bruna Bologna Catinelli, and et al. 2025. "Prevalence and Risk Factors of Pregnancy-Specific Urinary Incontinence: Findings from the Diamater Cohort Study" Healthcare 13, no. 17: 2141. https://doi.org/10.3390/healthcare13172141
APA StyleBassin, H. C. M., Barbosa, A. M. P., Prudencio, C. B., Sobrevia, L., Barbosa, V. P., Nunes, S. K., Rossignoli, P. d. S., Pedroni, C. R., França, D. C. H., Catinelli, B. B., Carvalho, C. N. F. D., Avramidis, R. E., Magyori, A. B. M., Sartorao-Filho, C. I., Rudge, M. V. C., & Diamater Study Group. (2025). Prevalence and Risk Factors of Pregnancy-Specific Urinary Incontinence: Findings from the Diamater Cohort Study. Healthcare, 13(17), 2141. https://doi.org/10.3390/healthcare13172141







