From MINI to Meaningful Change—A German Pilot Study to Improve Patient Outcomes in End-of-Life Care
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Design
2.2. Intervention and Study Procedure
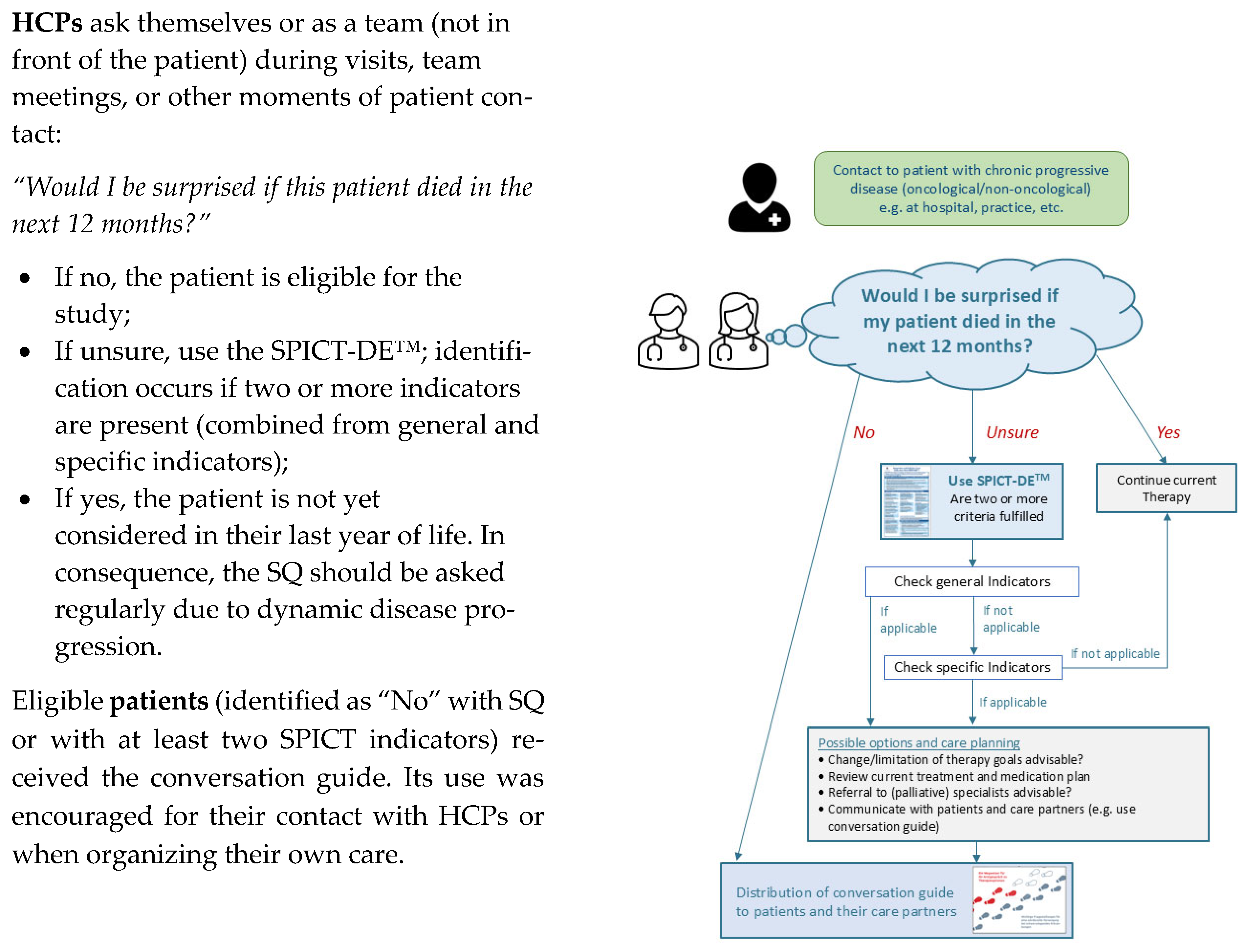
2.3. Recruitment Procedure
- Initial Screening: HCPs (medical and nursing staff) on participating wards (Ward 1, primarily treating non-cancer patients; Ward 2, cancer inpatients/outpatients) identified eligible patients. This occurred during regular contact (visits and team meetings) or with study personnel (researchers (JG, BW) or study nurse (LW)) present at least twice weekly. Inclusion criteria were (a) patients who were identified via the Surprise Question (“Would you be surprised if this patient died within 12 months?” to which the answer was recorded as “no”) and the SPICT-DE™ tool; (b) aware of their incurable disease and limited prognosis; (c) aged ≥ 18 years; (d) capable of providing informed consent; (e) cognitively able to participate; and (f) fluent in German;
- Recruitment Log Completion: For each flagged patient, HCPs completed a standardized recruitment log and confirmed all inclusion criteria, especially the patient’s awareness of their condition’s incurability;
- Patient Contact Form: HCPs provided identified patients with a contact form to indicate their willingness to be contacted by the study team. These forms were securely collected by study personnel from the physician’s office. Study personnel regularly contacted wards to ensure ongoing screening;
- Informed Consent and Baseline: Study personnel retrieved forms, contacted interested patients, and provided detailed study information. If patients were interested, the conversation guide (QPS) was distributed and their written informed consent was obtained. Care partner contact details were also collected for burden assessment, with this data to be published separately (Grimm et al., in preparation [21]). A baseline assessment (T0) was scheduled only after consent was secured.
2.4. Data Collection
2.5. Measurements
2.6. Statistical Analysis
3. Results
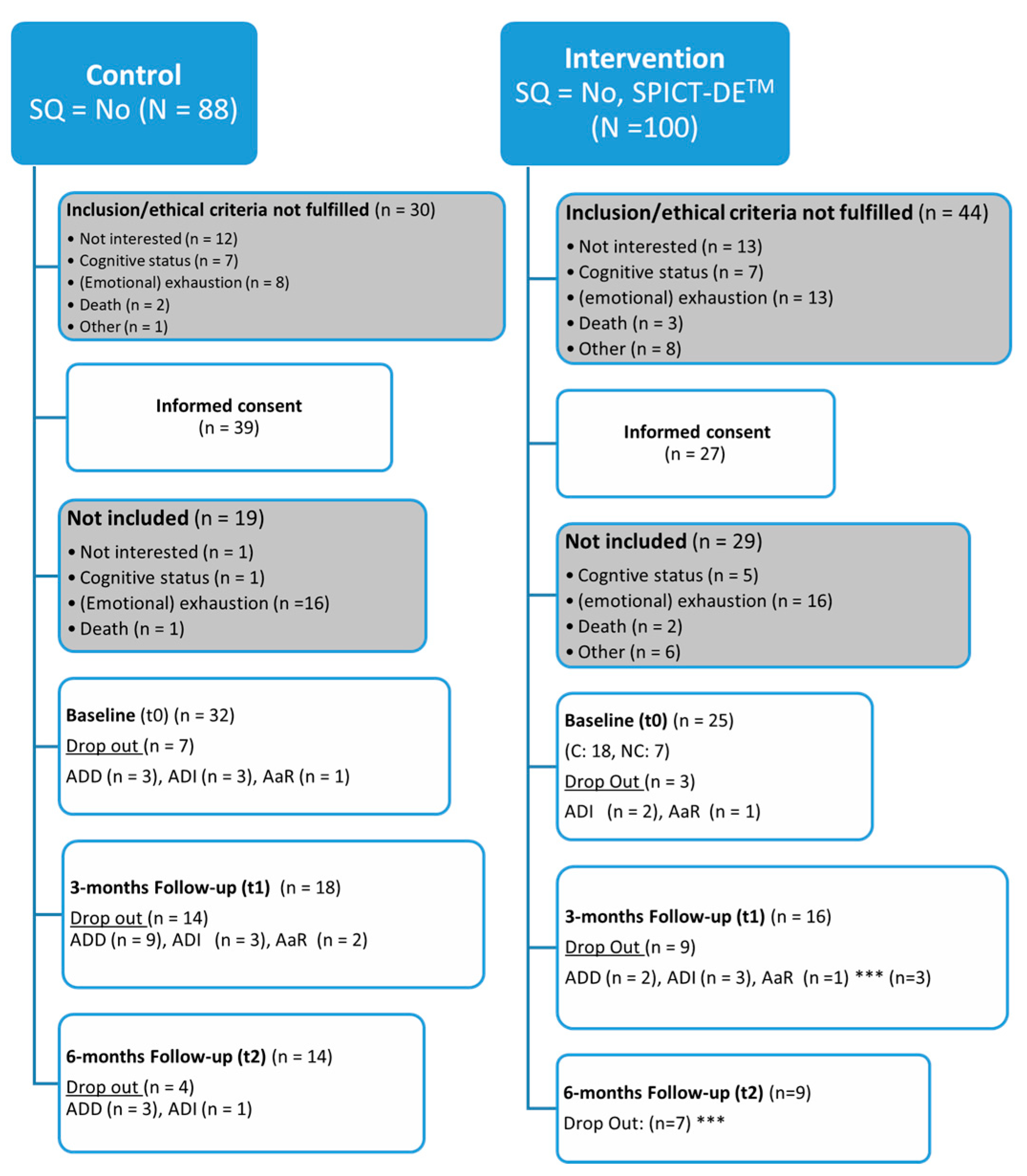
3.1. Allocation and Dropout
3.2. General Sample Characteristics at Baseline
3.3. Self-Reported Health-Related Quality of Life with the Short-Form Health Service (SF-12)
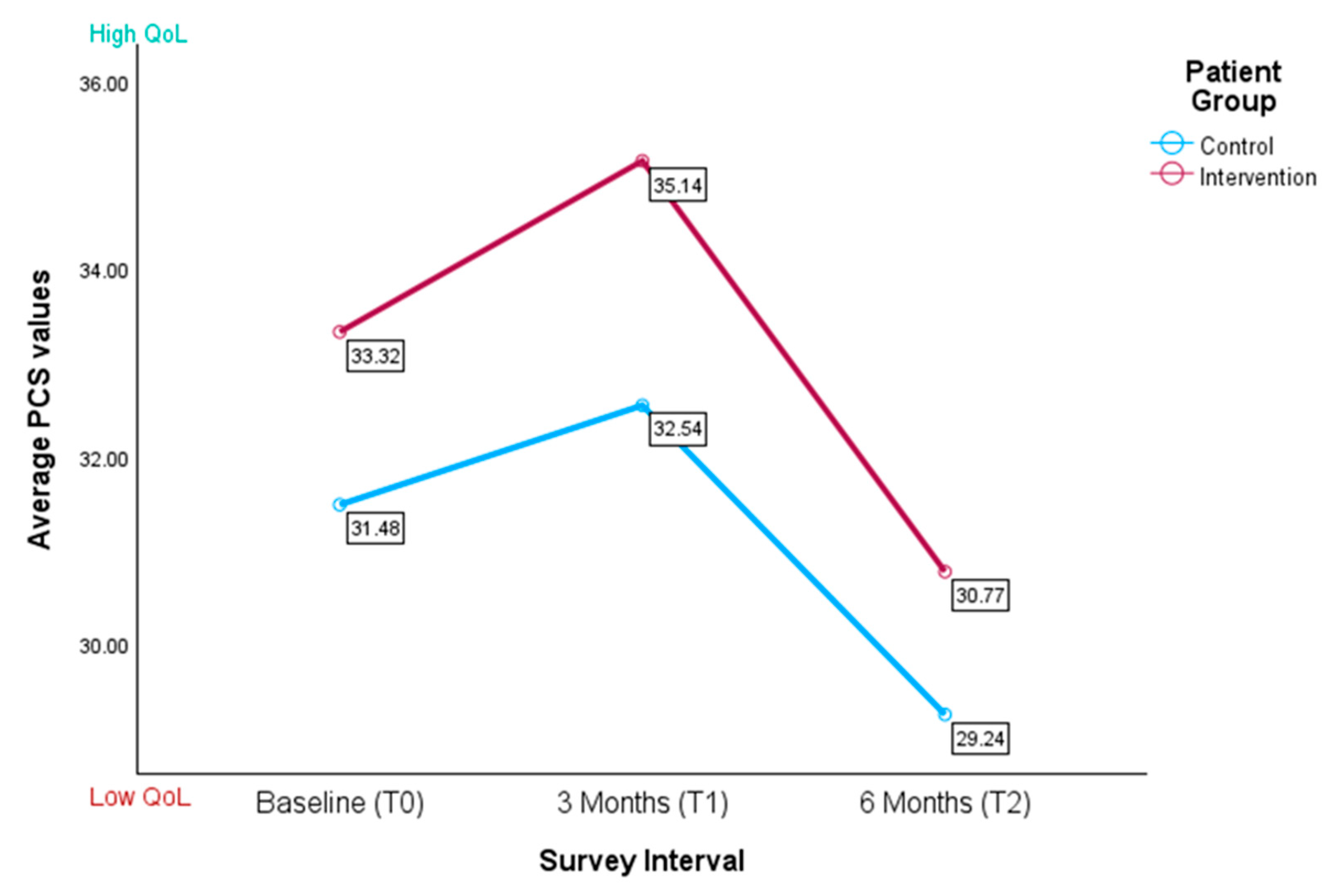
3.3.1. Physical Component Score
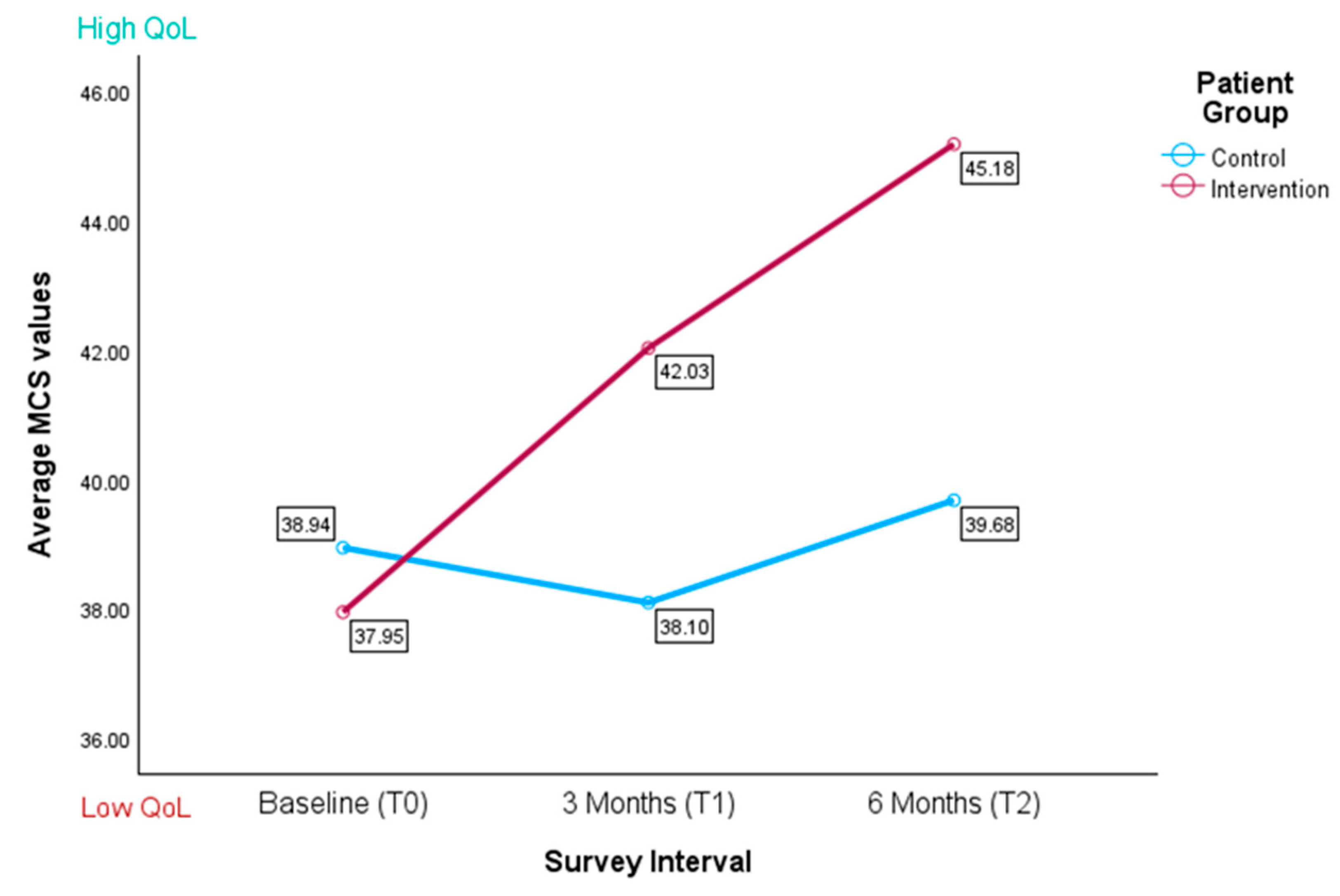
3.3.2. Mental Component Score (MCS)
3.4. Palliative Care Needs Using the Integrated Palliative Care Outcome Scale (IPOS)
3.4.1. IPOS Number of Physical, Emotional, and Communication Problems
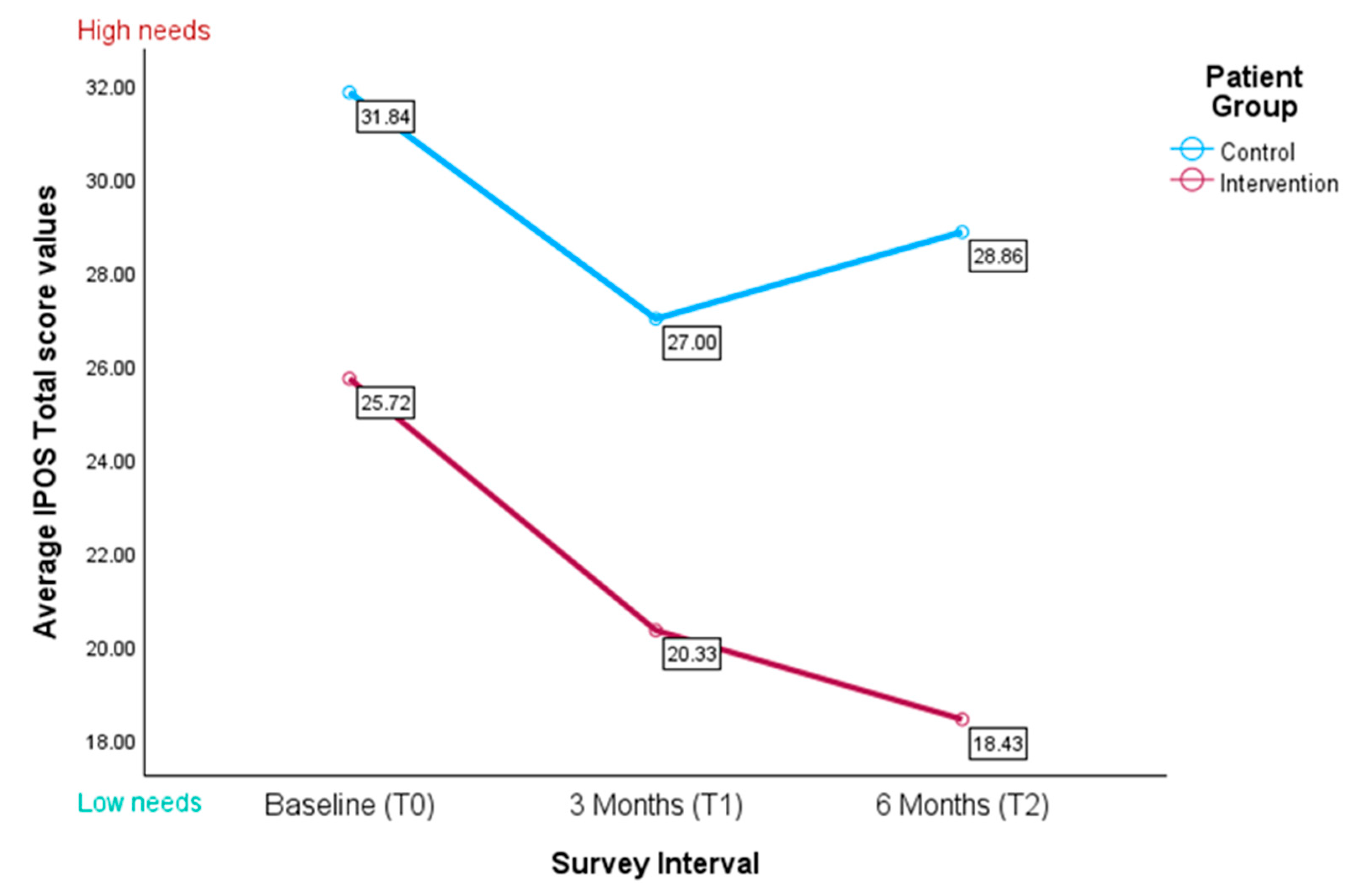
3.4.2. IPOS Total Scores
3.4.3. IPOS Subscale for Physical Symptoms
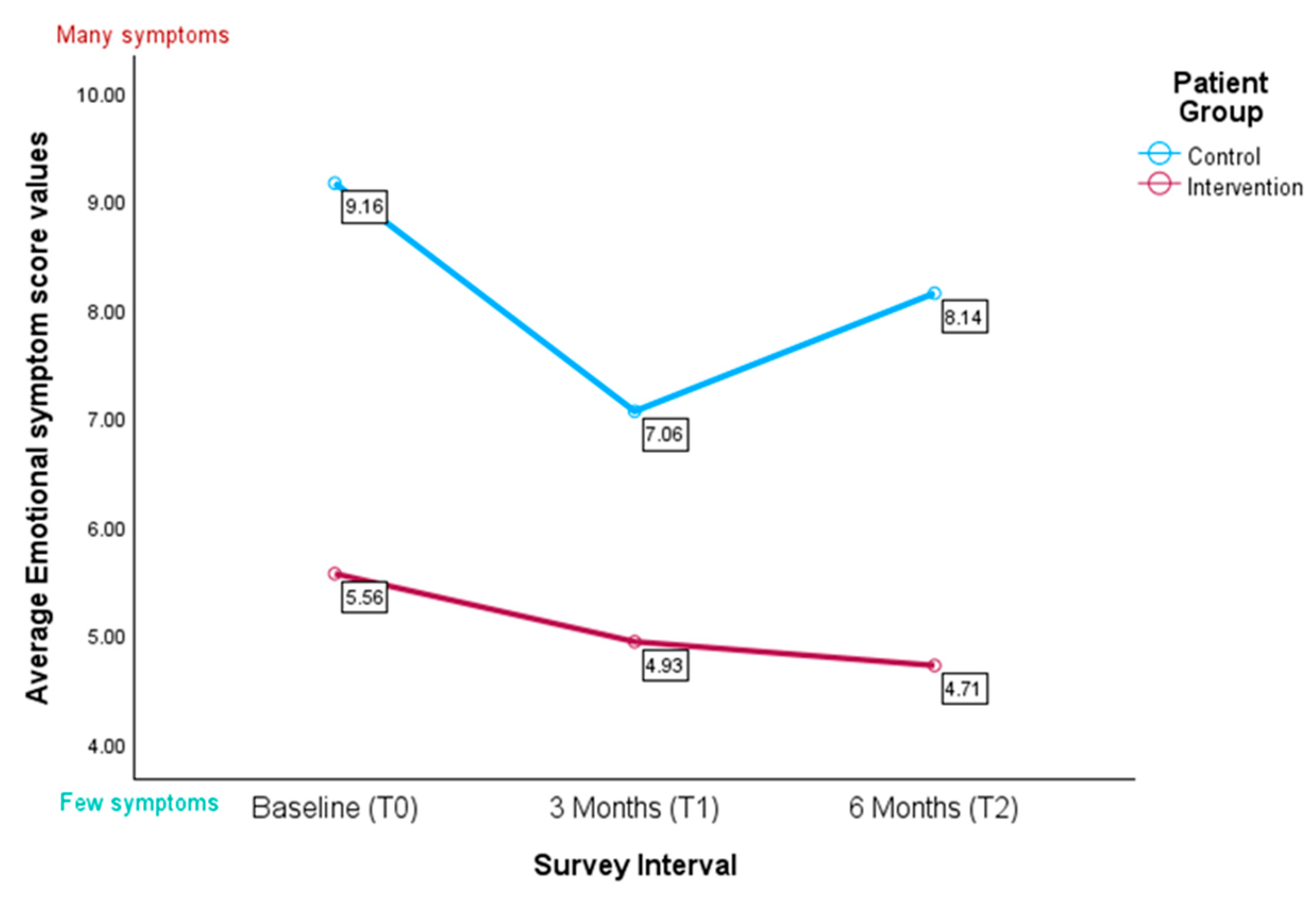
3.4.4. IPOS Subscale for Emotional Symptoms
“I don’t want to have anything else besides therapy. Otherwise, it’s too much. I’d like to be able to stop thinking about my illness.” (Pat_KG_FC4).(extracted as free text comments)
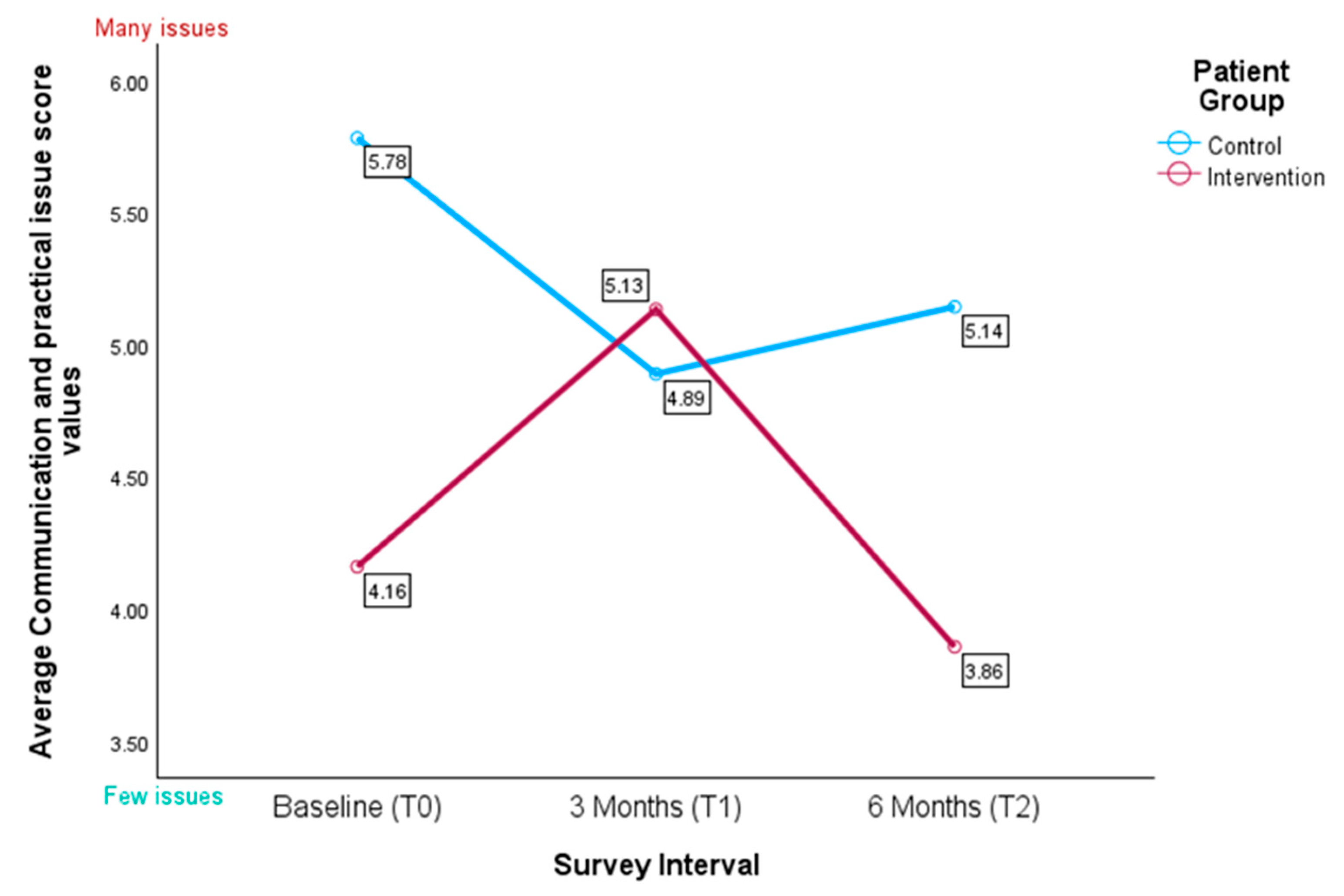
3.4.5. IPOS Subscale for Communication and Practical Issues
“Can I do that to my wife, having her care for me at home until the end? I would rather die at home than in a nursing home or something like that, but I don’t even dare to think about it or talk to her about it just yet” (PAT_KG_CP15).(extracted as free text comments)
“Although I’ve tried to discuss my worries and fears, it hasn’t worked because I always see someone else. The main contact person is good, but you hardly see them as a patient because they have so little time. The constant change of doctors makes you feel like a number, so you can’t really talk about your fears.” [...] (PAT_IG_KR4).(extracted as free text comments)
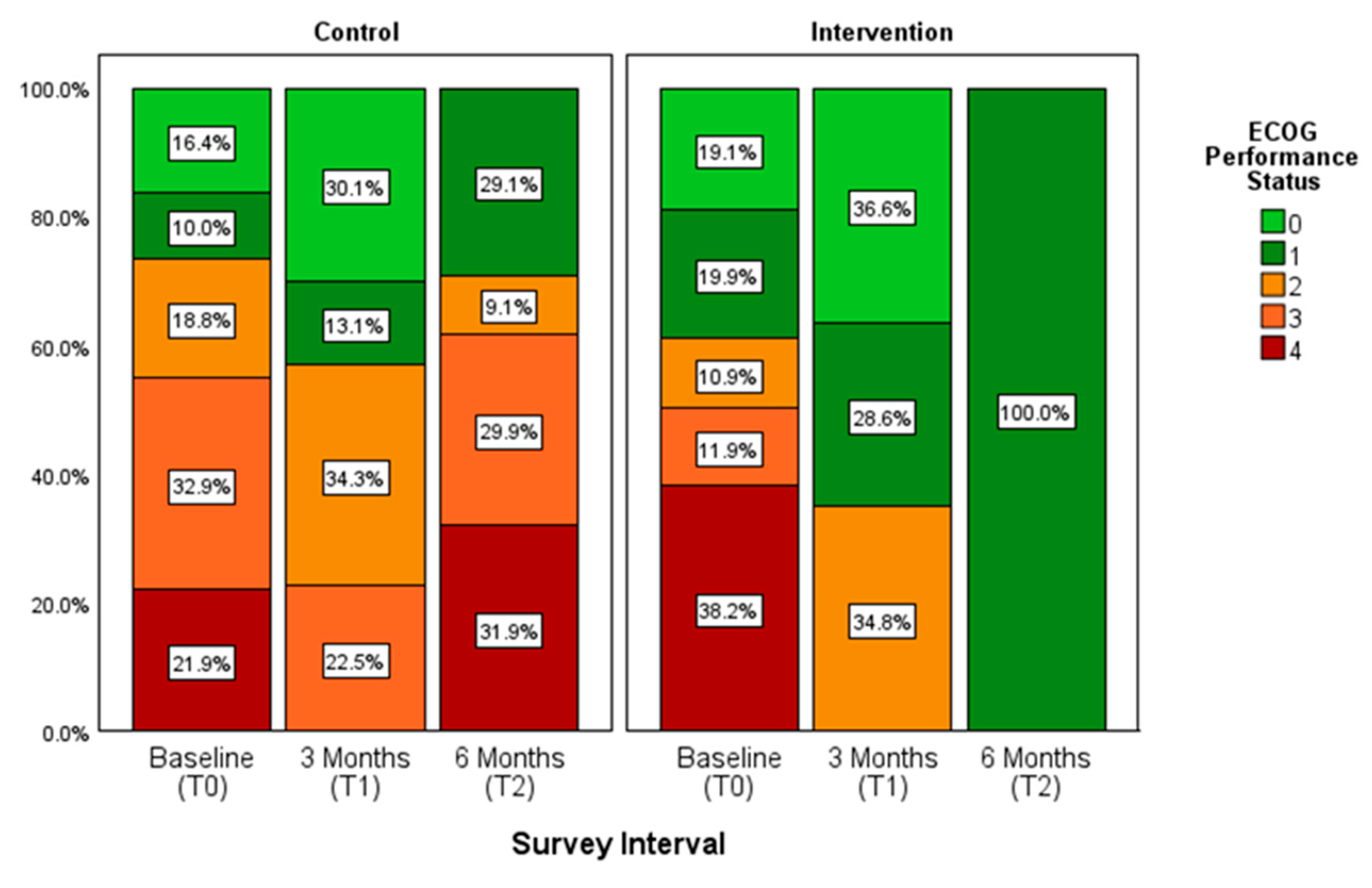
3.5. Cooperative Oncology Group (ECOG) Performance Status
4. Discussion
4.1. Allocation and General Sample Characteristics
4.2. Outcome Measures
4.2.1. SF-12
4.2.2. IPOS
4.2.3. ECOG
4.3. Strengths, Limitations, and Lessons Learned
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| AaR | Attrition at random |
| ADD | Attrition due to death |
| ADI | Attrition due to illness |
| CG | Control group |
| ECOG | Eastern Cooperative Oncology Group Performance Status |
| HCPs | Healthcare professionals |
| IG | Intervention group |
| ICC | Intraclass Correlation Coefficient |
| IPOS | Integrated Palliative Care Outcome Scale |
| LYOL-C-II | Last Year of Life Study Cologne—Part 2 |
| M | Mean |
| MCS-12 | Mental Component Score |
| MINI | Minimally Invasive Intervention |
| PCS-12 | Physical Component Score |
| PROMS | Patient-reported outcome measures |
| QoL | Quality of life |
| QPS | Question prompt sheets |
| SF12 | Short-Form Health Service |
| SD | Standard deviation |
| SPICT-DETM | Supportive and Palliative Care Indicators Tool |
| SQ | Surprise Question |
| T (0-4) | Timepoints |
Appendix A
| PCS | MCS | |||
|---|---|---|---|---|
| Control M (SD) | Intervention M (SD) | Control M (SD) | Intervention M (SD) | |
| T0 | 31.34 (5.53) | 33.40 (5.50) | 38.63 (7.53) | 38.09 (7.25) |
| T1 | 32.54 (6.51) | 35.14 (7.20) | 38.10 (7.06) | 42.03 (4.82) |
| T2 | 29.24 (5.55) | 29.74 (4.93) | 39.68 (7.27) | 44.67 (6.24) |
| A. Fixed-Effect Estimates | ||||||
| Parameter | β | Std. Error | df | t-Value | p-Value | 95% CI |
| Intercept | 20.30 | 5.01 | 71.41 | 4.05 | <0.001 | [10.31, 30.29] |
| Group (control) | 8.77 | 3.99 | 95.46 | 2.20 | 0.030 * | [0.854, 16.69] |
| Time (T0 vs. T2) | 4.53 | 3.23 | 42.63 | 1.40 | 0.168 | [−1.98, 11.04] |
| Time (T1 vs. T2) | 2.22 | 2.89 | 56.43 | 0.77 | 0.445 | [−3.56, 8.00] |
| Group × Time (control × T0) | −2.97 | 4.05 | 39.41 | −0.74 | 0.476 | [−11.15, 5.21] |
| Group × Time (control × T1) | −3.39 | 3.56 | 54.27 | −0.95 | 0.346 | [−10.53, 3.75] |
| Gender (female) | 1.59 | 3.92 | 51.78 | 0.41 | 0.687 | [−6.29, 9.46] |
| Ward (internal medicine) | −0.31 | 3.59 | 51.63 | −0.09 | 0.932 | [−7.51, 6.89] |
| Age | 0.21 | 0.11 | 44.75 | 1.87 | 0.068 | [−0.016, 0.43] |
| B. Type III Tests of Fixed Effects | ||||||
| Parameters | Num df | Den df | F | p-value | ||
| Group | 1 | 56.79 | 6.44 | 0.014 * | ||
| Time | 2 | 46.74 | 1.91 | 0.159 | ||
| Group × Time | 2 | 47.68 | 0.45 | 0.638 | ||
| Gender | 1 | 51.78 | 0.16 | 0.687 | ||
| Ward | 1 | 51.63 | 0.01 | 0.932 | ||
| Age | 1 | 44.75 | 3.50 | 0.068 | ||
| C. Variance Components and AR (1) Parameter | ||||||
| Component | Estimate | Std. Error | 95% CI | |||
| Random-intercept variance | 8.87 | 90.02 | [0, 3.87× 109] ** | |||
| Residual variance | 85.90 | 89.54 | [11.14, 666.55] | |||
| AR(1) | 0.62 | 0.39 | [−0.48, 0.96] | |||
| A. Fixed-Effect Estimates | ||||||
| Parameter | β | Std. Error | df | t-Value | p-Value | 95% CI |
| Intercept | 4.05 | 1.14 | 77.12 | 3.55 | 0.001 | [1.78, 6.31] |
| Group (control) | 1.09 | 1.02 | 95.46 | 1.07 | 0.288 | [−0.94, 3.12] |
| Time (T0 vs. T2) | 0.22 | 0.91 | 40.63 | 0.24 | 0.812 | [−1.62, 2.06] |
| Time (T1 vs. T2) | 1.29 | 0.94 | 60.59 | 1.36 | 0.177 | [−0.59, 3.17] |
| Group × Time (control × T0) | 0.51 | 1.13 | 37.23 | 0.45 | 0.657 | [−1.78, 2.80] |
| Group × Time (control × T1) | −1.46 | 1.18 | 55.73 | −1.24 | 0.222 | [−3.83, 0.91] |
| Gender (female) | 0.07 | 0.81 | 46.96 | 0.08 | 0.936 | [−1.57, 1.70] |
| Ward (internal medicine) | −0.33 | 0.74 | 48.15 | −0.45 | 0.656 | [−1.82, 1.16] |
| Age | 0.03 | 0.02 | 35.29 | 1.54 | 0.133 | [−0.01, 0.08] |
| B. Type III Tests of Fixed Effects | ||||||
| Parameters | Num df | Den df | F | p-value | ||
| Group | 1 | 42.60 | 2.08 | 0.157 | ||
| Time | 2 | 56.31 | 0.47 | 0.626 | ||
| Group × Time | 2 | 57.67 | 2.44 | 0.096 | ||
| Gender | 1 | 46.96 | 0.01 | 0.936 | ||
| Ward | 1 | 48.15 | 0.20 | 0.656 | ||
| Age | 1 | 35.29 | 2.36 | 0.133 | ||
| C. Variance Components and AR (1) Parameter | ||||||
| Component | Estimate | Std. Error | 95% CI | |||
| Random-intercept variance | 0.75 | 1.61 | [0.01, 50.49] | |||
| Residual variance | 4.20 | 1.61 | [1.98, 8.92] | |||
| AR(1) | 0.06 | 0.37 | [–0.58, 0.65] | |||
References
- Voltz, R. Palliativ … und Jetzt? 10 Erkenntnisse zum Umgang mit schwerer Krankheit und für die letzte Lebensphase, 1st ed.; W. Kohlhammer: Stuttgart, Germany, 2022; ISBN 978-3-17-041178-4. [Google Scholar]
- Kremeike, K.; Bausewein, C.; Freytag, A.; Junghanss, C.; Marx, G.; Schnakenberg, R.; Schneider, N.; Schulz, H.; Wedding, U.; Voltz, R. DNVF-Memorandum Versorgungsforschung im letzten Lebensjahr. Gesundheitswesen 2022. [Google Scholar] [CrossRef] [PubMed]
- Fliedner, M.C.; Zambrano, S.C.; Eychmueller, S. Public perception of palliative care: A survey of the general population. Palliat. Care Soc. Pract. 2021, 15, 26323524211017546. [Google Scholar] [CrossRef]
- Knaul, F.M.; Arreola-Ornelas, H.; Kwete, X.J.; Bhadelia, A.; Rosa, W.E.; Touchton, M.; Méndez-Carniado, O.; Vargas Enciso, V.; Pastrana, T.; Friedman, J.R.; et al. The evolution of serious health-related suffering from 1990 to 2021: An update to The Lancet Commission on global access to palliative care and pain relief. Lancet Glob. Health 2025, 13, e422–e436. [Google Scholar] [CrossRef] [PubMed]
- Voltz, R.; Dust, G.; Schippel, N.; Hamacher, S.; Payne, S.; Scholten, N.; Pfaff, H.; Rietz, C.; Strupp, J. Improving regional care in the last year of life by setting up a pragmatic evidence-based Plan-Do-Study-Act cycle: Results from a cross-sectional survey. BMJ Open 2020, 10, e035988. [Google Scholar] [CrossRef] [PubMed]
- Strupp, J.; Kasdorf, A.; Dust, G.; Hower, K.I.; Seibert, M.; Werner, B.; Kuntz, L.; Schulz-Nieswandt, F.; Meyer, I.; Pfaff, H.; et al. Last Year of Life Study-Cologne (LYOL-C) (Part II): Study protocol of a prospective interventional mixed-methods study in acute hospitals to analyse the implementation of a trigger question and patient question prompt sheets to optimise patient-centred care. BMJ Open 2021, 11, e048681. [Google Scholar] [CrossRef]
- Strupp, J.; Hanke, G.; Schippel, N.; Pfaff, H.; Karbach, U.; Rietz, C.; Voltz, R. Last Year of Life Study Cologne (LYOL-C): Protocol for a cross-sectional mixed methods study to examine care trajectories and transitions in the last year of life until death. BMJ Open 2018, 8, e021211. [Google Scholar] [CrossRef]
- Kasdorf, A.; Dust, G.; Hamacher, S.; Schippel, N.; Rietz, C.; Voltz, R.; Strupp, J. The last year of life for patients dying from cancer vs. non-cancer causes: A retrospective cross-sectional survey of bereaved relatives. Support. Care Cancer 2022, 30, 4971–4979. [Google Scholar] [CrossRef]
- Schippel, N.; Dust, G.; von Reeken, C.; Voltz, R.; Strupp, J.; Rietz, C. Can we determine burdensome transitions in the last year of life based on time of occurrence and frequency? An explanatory mixed-methods study. Palliat. Support. Care 2022, 20, 637–645. [Google Scholar] [CrossRef]
- Scholten, N.; Günther, A.L.; Pfaff, H.; Karbach, U. The size of the population potentially in need of palliative care in Germany--an estimation based on death registration data. BMC Palliat. Care 2016, 15, 29. [Google Scholar] [CrossRef]
- Goswami, P. Advance Care Planning and End-Of-Life Communications: Practical Tips for Oncology Advanced Practitioners. J. Adv. Pract. Oncol. 2021, 12, 89–95. [Google Scholar] [CrossRef]
- Epstein, R.M.; Duberstein, P.R.; Fenton, J.J.; Fiscella, K.; Hoerger, M.; Tancredi, D.J.; Xing, G.; Gramling, R.; Mohile, S.; Franks, P.; et al. Effect of a Patient-Centered Communication Intervention on Oncologist-Patient Communication, Quality of Life, and Health Care Utilization in Advanced Cancer: The VOICE Randomized Clinical Trial. JAMA Oncol. 2017, 3, 92–100. [Google Scholar] [CrossRef]
- Kasdorf, A.; Dust, G.; Vennedey, V.; Rietz, C.; Polidori, M.C.; Voltz, R.; Strupp, J. What are the risk factors for avoidable transitions in the last year of life? A qualitative exploration of professionals’ perspectives for improving care in Germany. BMC Health Serv. Res. 2021, 21, 147. [Google Scholar] [CrossRef]
- Debenedetti, S.; Cosmai, S.; Cattani, D.; Mancin, S.; Cangelosi, G.; Petrelli, F.; Mazzoleni, B. Language Validation and Cultural Adaptation of the Italian Version of the Family Caregiver Quality of Life Scale. Nurs. Rep. 2024, 14, 2302–2312. [Google Scholar] [CrossRef] [PubMed]
- Braun, M.; Scholz, U.; Hornung, R.; Martin, M. Die subjektive Belastung pflegender Ehepartner von Demenzkranken. Hinweise zur Validität der deutschen Version des Zarit Burden Interviews. Z. Gerontol. Geriatr. 2010, 43, 111–119. [Google Scholar] [CrossRef]
- Wagle, K.C.; Skopelja, E.N.; Campbell, N.L. Caregiver-Based Interventions to Optimize Medication Safety in Vulnerable Elderly Adults: A Systematic Evidence-Based Review. J. Am. Geriatr. Soc. 2018, 66, 2128–2135. [Google Scholar] [CrossRef]
- Locatelli, G.; Zeffiro, V.; Occhino, G.; Rebora, P.; Caggianelli, G.; Ausili, D.; Alvaro, R.; Riegel, B.; Vellone, E. Effectiveness of Motivational Interviewing on contribution to self-care, self-efficacy, and preparedness in caregivers of patients with heart failure: A secondary outcome analysis of the MOTIVATE-HF randomized controlled trial. Eur. J. Cardiovasc. Nurs. 2022, 21, 801–811. [Google Scholar] [CrossRef]
- Haroen, H.; Maulana, S.; Harlasgunawan, A.R.; Rahmawati, S.; Riansyah, A.; Musthofa, F.; Pardosi, J.F. Barriers and Facilitators of Early Palliative Care in the Trajectory of People Living with Chronic Condition: A Mini Review Using Socio-ecological Framework to Inform Public Health Strategy. J. Multidiscip. Healthc. 2024, 17, 4189–4197. [Google Scholar] [CrossRef] [PubMed]
- Harnischfeger, N.; Rath, H.M.; Oechsle, K.; Bergelt, C. Addressing palliative care and end-of-life issues in patients with advanced cancer: A systematic review of communication interventions for physicians not specialised in palliative care. BMJ Open 2022, 12, e059652. [Google Scholar] [CrossRef] [PubMed]
- Kasdorf, A.; Werner, B.; Grimm, J.S.; Dust, G.; Voltz, R.; Strupp, J.; on behalf of the Cologne Research and Development Network (CoRe-Net). Promising Concept, Challenging Implementation: Evaluating the Implementation of a Minimally Invasive Intervention (MINI) to Identify Patients in Their Last Year of Life: A Multi-Methods Feasibility Study with University Hospital Staff in Germany. Correspondence: Department of Palliative Medicine, University Hospital Cologne, Cologne, North Rhine-Westphalia, Germany. 2025; (manuscript in preparation). [Google Scholar]
- Grimm, J.S.; Kasdorf, A.; Voltz, R.; Strupp, J.; on behalf of the Cologne Research and Development Network (CoRe-Net). “Alone it would be simply impossible to cope with the disease and the burdensome organization around it” Perceived care partner burden in end-of-life care.; Correspondence Department of Palliative Medicine, University Hospital Cologne, Cologne, North Rhine-Westphalia, Germany. 2025; (manuscript in preparation). [Google Scholar]
- Ware, J.; Kosinski, M.; Keller, S.D. A 12-Item Short-Form Health Survey: Construction of scales and preliminary tests of reliability and validity. Med. Care 1996, 34, 220–233. [Google Scholar] [CrossRef]
- Murtagh, F.E.; Ramsenthaler, C.; Firth, A.; Groeneveld, E.I.; Lovell, N.; Simon, S.T.; Denzel, J.; Guo, P.; Bernhardt, F.; Schildmann, E.; et al. A brief, patient- and proxy-reported outcome measure in advanced illness: Validity, reliability and responsiveness of the Integrated Palliative care Outcome Scale (IPOS). Palliat. Med. 2019, 33, 1045–1057. [Google Scholar] [CrossRef]
- Oken, M.M.; Creech, R.H.; Tormey, D.C.; Horton, J.; Davis, T.E.; McFadden, E.T.; Carbone, P.P. Toxicity and response criteria of the Eastern Cooperative Oncology Group. Am. J. Clin. Oncol. 1982, 5, 649–655. [Google Scholar] [CrossRef]
- van der Zee, E.N.; Noordhuis, L.M.; Epker, J.L.; van Leeuwen, N.; Wijnhoven, B.P.L.; Benoit, D.D.; Bakker, J.; Kompanje, E.J.O. Assessment of mortality and performance status in critically ill cancer patients: A retrospective cohort study. PLoS ONE 2021, 16, e0252771. [Google Scholar] [CrossRef]
- von Elm, E.; Altman, D.G.; Egger, M.; Pocock, S.J.; Gøtzsche, P.C.; Vandenbroucke, J.P. The Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) statement: Guidelines for reporting observational studies. J. Clin. Epidemiol. 2008, 61, 344–349. [Google Scholar] [CrossRef]
- Aaronson, E.L.; George, N.; Ouchi, K.; Zheng, H.; Bowman, J.; Monette, D.; Jacobsen, J.; Jackson, V. The Surprise Question Can Be Used to Identify Heart Failure Patients in the Emergency Department Who Would Benefit from Palliative Care. J. Pain Symptom Manag. 2019, 57, 944–951. [Google Scholar] [CrossRef]
- Afshar, K.; Feichtner, A.; Boyd, K.; Murray, S.; Jünger, S.; Wiese, B.; Schneider, N.; Müller-Mundt, G. Systematic development and asjustment of the German version of the Supportive and Palliative Care Indicators Tool (SPICT-DE). BMC Palliat. Care 2018, 17, 27. [Google Scholar] [CrossRef] [PubMed]
- Afshar, K.; van Baal, K.; Schleef, T.; Frerichs, H.; Schneider, N. Handlungsempfehlungen für die Versorgung von Menschen am Lebensende und ihren Angehörigen: Ergebnisse aus dem Projekt OPAL Optimale Versorgung am Lebensende, 2nd ed.; Medizinische Hoschule Hannover, Intitute für Allgemeinmedizin und Palliativmedizin: Hanover, Germany, 2023; Available online: https://www.mhh.de/fileadmin/mhh/allgemeinmedizin/downloads/weitere_Downloadelemente/AllPallMed_Broschuere_OPAL.pdf (accessed on 5 March 2025).
- Gandek, B.; Ware, J.E.; Aaronson, N.K.; Apolone, G.; Bjorner, J.B.; Brazier, J.E.; Bullinger, M.; Kaasa, S.; Leplege, A.; Prieto, L.; et al. Cross-validation of item selection and scoring for the SF-12 Health Survey in nine countries: Results from the IQOLA Project. International Quality of Life Assessment. J. Clin. Epidemiol. 1998, 51, 1171–1178. [Google Scholar] [CrossRef] [PubMed]
- Drixler, K.; Morfeld, M.; Glaesmer, H.; Brähler, E.; Wirtz, M.A. Validierung der Messung gesundheitsbezogener Lebensqualität mittels des Short-Form-Health-Survey-12 (SF-12 Version 2.0) in einer deutschen Normstichprobe. Z. Psychosom. Med. Psychother. 2020, 66, 272–286. [Google Scholar] [CrossRef] [PubMed]
- Wirtz, M.A.; Morfeld, M.; Glaesmer, H.; Brähler, E. Normierung des SF-12 Version 2.0 zur Messung der gesundheitsbezogenen Lebensqualität in einer deutschen bevölkerungsrepräsentativen Stichprobe. Diagnostica 2018, 64, 215–226. [Google Scholar] [CrossRef]
- Miles, M.B.; Huberman, A.M.; Saldaña, J. Qualitative Data Analysis: A Methods Sourcebook, 3rd ed.; SAGE: Thousand Oaks, CA, USA, 2014; ISBN 978-1-4522-5787-7. [Google Scholar]
- Higginson, I.J.; Evans, C.J.; Grande, G.; Preston, N.; Morgan, M.; McCrone, P.; Lewis, P.; Fayers, P.; Harding, R.; Hotopf, M.; et al. Evaluating complex interventions in end of life care: The MORECare statement on good practice generated by a synthesis of transparent expert consultations and systematic reviews. BMC Med. 2013, 11, 111. [Google Scholar] [CrossRef]
- Oriani, A.; Dunleavy, L.; Sharples, P.; Perez Algorta, G.; Preston, N.J. Are the MORECare guidelines on reporting of attrition in palliative care research populations appropriate? A systematic review and meta-analysis of randomised controlled trials. BMC Palliat. Care 2020, 19, 6. [Google Scholar] [CrossRef]
- Bandura, A. Social cognitive theory of self-regulation. Organ. Behav. Hum. Decis. Process. 1991, 50, 248–287. [Google Scholar] [CrossRef]
- Adolfsson, K.; Kreicbergs, U.; Bratthäll, C.; Holmberg, E.; Björk-Eriksson, T.; Stenmarker, M. Referral of patients with cancer to palliative care: Attitudes, practices and work-related experiences among Swedish physicians. Eur. J. Cancer Care (Engl.) 2022, 31, e13680. [Google Scholar] [CrossRef]
- Salins, N.; Ghoshal, A.; Hughes, S.; Preston, N. How views of oncologists and haematologists impacts palliative care referral: A systematic review. BMC Palliat. Care 2020, 19, 175. [Google Scholar] [CrossRef] [PubMed]
- Francis, L.; Robertson, N. Healthcare practitioners’ experiences of breaking bad news: A critical interpretative meta synthesis. Patient Educ. Couns. 2023, 107, 107574. [Google Scholar] [CrossRef] [PubMed]
- Ratna, H. The Importance of Effective Communication in Healthcare Practice. Harv. Public Health Rev. 2019, 23, 1–6. [Google Scholar] [CrossRef]
- Dale MacLaine, T.; Lowe, N.; Dale, J. The use of simulation in medical student education on the topic of breaking bad news: A systematic review. Patient Educ. Couns. 2021, 104, 2670–2681. [Google Scholar] [CrossRef]
- Wang, X.; Liu, M.; Li, J.; Wang, Z.; Liang, Q.; Yan, Z.; Wang, J.; Luan, X. Relationship between quality of life, fear of disease progression, and coping styles in patients with pulmonary hypertension: A network analysis. Res. Nurs. Health 2023, 46, 546–557. [Google Scholar] [CrossRef]
- Wang, C.; Bi, S.; Lu, Y.; Li, Y.; Han, B.; Xu, M.; Meng, G.; Zhou, Q. Availability and stability of palliative care for family members of terminally ill patients in an integrated model of health and social care. BMC Palliat. Care 2024, 23, 140. [Google Scholar] [CrossRef]
- Marschner, N.; Zacharias, S.; Lordick, F.; Hegewisch-Becker, S.; Martens, U.; Welt, A.; Hagen, V.; Gleiber, W.; Bohnet, S.; Kruggel, L.; et al. Association of Disease Progression with Health-Related Quality of Life Among Adults with Breast, Lung, Pancreatic, and Colorectal Cancer. JAMA Netw. Open 2020, 3, e200643. [Google Scholar] [CrossRef]
- Salamanca-Balen, N.; Merluzzi, T.V.; Chen, M. The effectiveness of hope-fostering interventions in palliative care: A systematic review and meta-analysis. Palliat. Med. 2021, 35, 710–728. [Google Scholar] [CrossRef]
- Merluzzi, T.V.; Salamanca-Balen, N.; Philip, E.J.; Salsman, J.M.; Chirico, A. Integration of Psychosocial Theory into Palliative Care: Implications for Care Planning and Early Palliative Care. Cancers 2024, 16, 342. [Google Scholar] [CrossRef]
- Tolstrup, L.K.; Ditzel, H.M.; Giger, A.-K.W.; Jørgensen, T.L.; Ryg, J.; Dieperink, K.B. Determinants of poor quality of life in older patients with cancer: A longitudinal mixed methods study (PROGNOSIS-Q). J. Geriatr. Oncol. 2023, 14, 101500. [Google Scholar] [CrossRef]
- Käsmann, L.; Taugner, J.; Eze, C.; Roengvoraphoj, O.; Dantes, M.; Gennen, K.; Karin, M.; Petrukhnov, O.; Tufman, A.; Belka, C.; et al. Performance Status and Its Changes Predict Outcome for Patients with Inoperable Stage III NSCLC Undergoing Multimodal Treatment. Anticancer Res. 2019, 39, 5077–5081. [Google Scholar] [CrossRef] [PubMed]
- Azam, F.; Latif, M.F.; Farooq, A.; Tirmazy, S.H.; AlShahrani, S.; Bashir, S.; Bukhari, N. Performance Status Assessment by Using ECOG (Eastern Cooperative Oncology Group) Score for Cancer Patients by Oncology Healthcare Professionals. Case Rep. Oncol. 2019, 12, 728–736. [Google Scholar] [CrossRef] [PubMed]
- Kumar, D.; Neeman, E.; Zhu, S.; Sun, H.; Kotak, D.; Liu, R. Revisiting the Association of ECOG Performance Status with Clinical Outcomes in Diverse Patients with Cancer. J. Natl. Compr. Canc. Netw. 2024, 22, e237111. [Google Scholar] [CrossRef]
- Dunleavy, L.; Preston, N.; Walshe, C. Health care professional recruitment of patients and family carers to palliative care randomised controlled trials: A qualitative multiple case study. Palliat. Med. 2023, 37, 1540–1553. [Google Scholar] [CrossRef]
- LeBlanc, T.W.; Lodato, J.E.; Currow, D.C.; Abernethy, A.P. Overcoming recruitment challenges in palliative care clinical trials. J. Oncol. Pract. 2013, 9, 277–282. [Google Scholar] [CrossRef] [PubMed]
- Walshe, C.; Dunleavy, L.; Preston, N.; Payne, S.; Ellershaw, J.; Taylor, V.; Mason, S.; Nwosu, A.C.; Gadoud, A.; Board, R.; et al. Understanding barriers and facilitators to palliative and end-of-life care research: A mixed method study of generalist and specialist health, social care, and research professionals. BMC Palliat. Care 2024, 23, 159. [Google Scholar] [CrossRef] [PubMed]
- Kerr, D.; Milnes, S.; Ammentorp, J.; McKie, C.; Dunning, T.; Ostaszkiewicz, J.; Wolderslund, M.; Martin, P. Challenges for nurses when communicating with people who have life-limiting illness and their families: A focus group study. J. Clin. Nurs. 2020, 29, 416–428. [Google Scholar] [CrossRef]
- Bruun, A.; Oostendorp, L.; Bloch, S.; White, N.; Mitchinson, L.; Sisk, A.-R.; Stone, P. Prognostic decision-making about imminent death within multidisciplinary teams: A scoping review. BMJ Open 2022, 12, e057194. [Google Scholar] [CrossRef]
- Ramos, K.; Kaufman, B.G.; Winger, J.G.; Boggins, A.; van Houtven, C.H.; Porter, L.S.; Hastings, S.N. Knowledge, goals, and misperceptions about palliative care in adults with chronic disease or cancer. Palliat. Support. Care 2023, 22, 1707–1713. [Google Scholar] [CrossRef]
- Kitta, A.; Hagin, A.; Unseld, M.; Adamidis, F.; Diendorfer, T.; Masel, E.K.; Kirchheiner, K. The silent transition from curative to palliative treatment: A qualitative study about cancer patients’ perceptions of end-of-life discussions with oncologists. Support. Care Cancer 2021, 29, 2405–2413. [Google Scholar] [CrossRef] [PubMed]
- Lelorain, S. Discussing Prognosis with Empathy to Cancer Patients. Curr. Oncol. Rep. 2021, 23, 42. [Google Scholar] [CrossRef]
- Amroud, M.S.; Raeissi, P.; Hashemi, S.-M.; Reisi, N.; Ahmadi, S.-A. Investigating the challenges and barriers of palliative care delivery in Iran and the World: A systematic review study. J. Educ. Health Promot. 2021, 10, 246. [Google Scholar] [CrossRef]
- Nipp, R.D.; El-Jawahri, A.; Moran, S.M.; D’Arpino, S.M.; Johnson, P.C.; Lage, D.E.; Wong, R.L.; Pirl, W.F.; Traeger, L.; Lennes, I.T.; et al. The relationship between physical and psychological symptoms and health care utilization in hospitalized patients with advanced cancer. Cancer 2017, 123, 4720–4727. [Google Scholar] [CrossRef]
- Colosimo, K.; Nissim, R.; Pos, A.E.; Hales, S.; Zimmermann, C.; Rodin, G. “Double awareness” in psychotherapy for patients living with advanced cancer. J. Psychother. Integr. 2018, 28, 125–140. [Google Scholar] [CrossRef]
- Petrilli, C.M.; Saint, S.; Jennings, J.J.; Caruso, A.; Kuhn, L.; Snyder, A.; Chopra, V. Understanding patient preference for physician attire: A cross-sectional observational study of 10 academic medical centres in the USA. BMJ Open 2018, 8, e021239. [Google Scholar] [CrossRef] [PubMed]

| Characteristics | Control n (%) M (SD) | Intervention n (%) M (SD) | Total n (%) M (SD) |
|---|---|---|---|
| Care partner enrolled in study | |||
| No | 18 (52.9) | 19 (73.1) | 37 (61.7) |
| Yes | 16 (47.1) | 7 (26.9) | 23 (38.3) |
| Care setting of recruitment | |||
| Internal medicine unit | 16 (48.5) | 7 (28.0) | 23 (39.7) |
| Gynecological inpatient unit | 4 (12.1) | 2 (8.0) | 6 (10.3) |
| Gynecological outpatient unit | 13 (39.4) | 16 (64.0) | 30 (50.0) |
| Highest school qualification * | |||
| High school or lower | 12 (36.4) | 10 (43.5) | 22 (39.3) |
| Vocational degree | 14 (42.4) | 7 (30.4) | 21 (37.5) |
| University degree | 7 (21.2) | 6 (26.1) | 13 (23.2) |
| Age | 64.76 ± 13.63 | 61.32 ± 13.17 | 63.28 ± 13.43 |
| Sex | |||
| Female | 25 (75.8) | 21 (84.0) | 46 (79.3) |
| Male | 8 (24.2) | 4 (16.0) | 12 (20.7) |
| Relationship status | |||
| Single | 2 (6.1) | 7 (29.2) | 9 (15.8) |
| Married/civil partnership | 19 (57.6) | 12 (50.0) | 31 (53.6) |
| Divorced | 7 (21.2) | 2 (8.3) | 9 (15.8) |
| Widowed | 5 (15.2) | 3 (12.5) | 8 (14.0) |
| Religion (NA 2) | |||
| None | 6 (18.8) | 6 (25.0) | 12 (21.4) |
| Christianity | 24 (75.0) | 18 (75.0) | 42 (75.0) |
| Islam | 2 (6.3) | 0 (0.0) | 2 (3.6) |
| Employment | |||
| No | 25 (78.1) | 16 (80.7) | 40 (71.4) |
| Currently on sick leave | 3 (9.4) | 4 (16.7) | 7 (12.5) |
| Yes | 4 (12.5) | 4 (16.7) | 8 (14.3) |
| Children (NA 1) | |||
| No | 4 (12.1) | 8 (33.3) | 12 (21.1) |
| Yes | 29 (87.9) | 16 (66.7) | 45 (78.9) |
| 1 | 10 (31.3) | 4 (16.7) | 14 (25.0) |
| 2 | 10 (31.1) | 9 (37.5) | 19 (33.9) |
| 3 or more | 8 (25.0) | 3 (12.5) | 11 (19.6) |
| Living situation (NA 1) | |||
| Living alone | 6 (18.2) | 9 (37.5) | 15 (26.3) |
| Living with another person | 27 (81.8.4) | 15 (62.5) | 42 (73.7) |
| Care Grade ** (NA 1) | |||
| No | 10 (30.3) | 11 (45.8) | 21 (36.8) |
| Yes | 23 (69.7) | 13 (54.2) | 36 (63.2) |
| Care grade 1 | 1 (3.0) | 1 (4.2) | 2 (3.5) |
| Care grade 2 | 8 (24.2) | 2 (8.3) | 10 (17.5) |
| Care grade 3 | 11 (33.3) | 5 (20.8) | 16 (28.1) |
| Care grade 4 | 3 (9.1) | 4 (16.7) | 7 (12.3) |
| Unsure | 0 (0.0) | 1 (4.2) | 1 (1.8) |
| Planned/applied for | 2 (6.1) | 0 (0.0) | 2 (3.5) |
| Knowledge about incurability (NA 3) | |||
| <24 h | 0 (0.0) | 2 (8.7) | 2 (3.6) |
| 1 week to <1 month | 0 (0.0) | 1 (4.3) | 1 (1.8) |
| 1 to <6 months | 4 (12.5) | 1 (4.3) | 5 (9.1) |
| 6 months to <1 year | 1 (3.1) | 0 (0.0) | 1 (1.8) |
| ≥1 year | 27 (84.4) | 19 (82.6) | 47 (86.6) |
| Financial problems | |||
| No | 16 (50.0) | 14 (58.3) | 30 (51.7) |
| Rather not | 2 (6.3) | 4 (16.7) | 6 (10.3) |
| Rather yes | 4 (12.5) | 3 (12.5) | 7 (12.1) |
| Yes | 10 (31.3) | 2 (8.3) | 12 (21.4) |
| Not sure | 0 (0.0) | 1 (4.2) | 1 (1.8) |
| A. Fixed-Effect Estimates | ||||||
| Parameter | β | Std. Error | df | t-Value | p-Value | 95% CI |
| Intercept | 35.53 | 2.98 | 86.82 | 11.26 | <0.001 | [27.61, 39.44] |
| Group (control) | –1.16 | 2.55 | 79.50 | –0.46 | 0.650 | [–6.23, 3.91] |
| Time (T0 vs. T2) | 2.64 | 2.17 | 30.96 | 1.22 | 0.233 | [–1.79, 7.08] |
| Time (T1 vs. T2) | 3.48 | 2.15 | 60.86 | 1.62 | 0.111 | [–1.82, 7.79] |
| Group × Time (control × T0) | 0.09 | 2.64 | 28.10 | 0.03 | 0.974 | [–5.32, 5.49] |
| Group × Time (control × T1) | 0.45 | 2.62 | 59.06 | 0.17 | 0.865 | [–4.80, 5.69] |
| Gender (female) | –1.88 | 2.16 | 55.28 | –0.87 | 0.387 | [–6.20, 2.44] |
| Ward (internal medicine) | –4.45 | 1.98 | 54.69 | –2.24 | 0.029 * | [–8.43, –0.48] |
| Age | −0.03 | 0.06 | 45.33 | −0.42 | 0.675 | [–0.15, 0.10] |
| B. Type III Tests of Fixed Effects | ||||||
| Parameters | Num df | Den df | F | p-value | ||
| Group | 1 | 60.78 | 0.44 | 0.511 | ||
| Time | 2 | 47.70 | 4.00 | 0.025 * | ||
| Group × Time | 2 | 49.06 | 0.02 | 0.977 | ||
| Gender | 1 | 55.28 | 0.76 | 0.387 | ||
| Ward | 1 | 54.69 | 5.04 | 0.029 * | ||
| Age | 1 | 45.33 | 0.18 | 0.675 | ||
| C. Variance Components and AR (1) Parameter | ||||||
| Component | Estimate | Std. Error | 95% CI | |||
| Random-intercept variance | 11.20 | 11.70 | [1.44, 86.85] | |||
| Residual variance | 20.44 | 10.77 | [7.28, 57.38] | |||
| AR(1) | 0.17 | 0.44 | [–0.61, 0.79] | |||
| A. Fixed-Effect Estimates | ||||||
| Parameter | β | Std. Error | df | t-Value | Sig. | 95% CI |
| Intercept | 43.97 | 3.75 | 86.39 | 11.72 | <0.001 | [36.51, 51.42] |
| Group (control) | −4.49 | 3.28 | 79.41 | −1.37 | 0.175 | [−11.01, 2.04] |
| Time (T0 vs. T2) | −6.21 | 2.79 | 39.95 | −2.23 | 0.032 * | [−11.85, 0.57] |
| Time (T1 vs. T2) | −2.45 | 3.04 | 56.84 | −0.81 | 0.424 | [−8.55, 3.64] |
| Group × Time (control × T0) | 5.52 | 3.71 | 38.02 | 1.64 | 0.110 | [−1.30, 12.34] |
| Group × Time (control × T1) | 1.18 | 3.76 | 53.67 | 0.32 | 0.754 | [−6.36, 8.72] |
| Gender (female) | 0.47 | 2.64 | 53.38 | 0.18 | 0.859 | [−4.82, 5.76] |
| Ward (internal medicine) | −0.52 | 2.41 | 51.50 | −0.22 | 0.830 | [−5.36, 4.32] |
| Age | 0.037 | 0.07 | 39.47 | 0.51 | 0.611 | [−0.12–0.18] |
| B. Type III Tests of Fixed Effects | ||||||
| Parameters | Num df | Den df | F | p-value | ||
| Group | 1 | 51.64 | 1.58 | 0.215 | ||
| Time | 2 | 54.25 | 2.24 | 0.116 | ||
| Group × Time | 2 | 55.34 | 2.00 | 0.146 | ||
| Gender | 1 | 53.38 | 0.03 | 0.859 | ||
| Ward | 1 | 51.50 | 0.05 | 0.830 | ||
| Age | 1 | 39.47 | 0.26 | 0.611 | ||
| C. Variance Components and AR (1) Parameter | ||||||
| Component | Estimate | Std. Error | 95% CI | |||
| Random-intercept variance | 19.04 | 7.41 | [7.71, 47.03] | |||
| Residual variance | 31.88 | 7.41 | [20.21, 50.27] | |||
| AR(1) | −0.20 | 0.26 | [−0.62, 0.31] | |||
| Pannel A. Fixed-Effect Estimates | ||||||
| Parameter | β | Std. Error | df | t-Value | Sig. | 95% CI |
| Intercept | 9.06 | 3.65 | 67.61 | 2.48 | 0.016 * | [−1.77, 16.34] |
| Group (control) | 5.71 | 2.72 | 85.11 | 2.10 | 0.039 * | [0.31, 11.12] |
| Time (T0 vs. T2) | 3.75 | 2.00 | 29.96 | 2.87 | 0.071 | [−0.34, 7.84] |
| Time (T1 vs. T2) | 0.60 | 1.86 | 50.69 | 0.32 | 0.747 | [−3.14, 4.35] |
| Group × Time (control × T0) | −5.13 | 2.50 | 27.76 | −2.05 | 0.050 | [−10.25, −0.01] |
| Group × Time (control × T1) | −1.24 | 2.30 | 49.61 | −0.54 | 0.594 | [−5.86, 3.39] |
| Gender (female) | 1.95 | 2.94 | 50.28 | 0.67 | 0.509 | [−3.95, 7.85] |
| Ward (internal medicine) | 0.05 | 2.69 | 49.74 | 0.02 | 0.984 | [−5.34, 5.45] |
| Age | 0.15 | 0.08 | 45.00 | 1.76 | 0.085 | [−0.02, 0.31] |
| Panel B Type III Tests of Fixed Effects | ||||||
| Parameters | Num df | Den df | F | p-value | ||
| Group | 1 | 55.48 | 3.43 | 0.069 | ||
| Time | 2 | 39.19 | 0.98 | 0.384 | ||
| Group × Time | 2 | 39.82 | 3.13 | 0.055 | ||
| Gender | 1 | 50.28 | 0.44 | 0.509 | ||
| Ward | 1 | 49.74 | 0.00 | 0.984 | ||
| Age | 1 | 45.00 | 3.11 | 0.085 | ||
| C. Variance Components and AR (1) Parameter | ||||||
| Component | Estimate | Std. Error | 95% CI | |||
| Random-intercept variance | 29.31 | 17.46 | [9.12, 94.21] | |||
| Residual variance | 21.62 | 14.83 | [5.63, 82.95] | |||
| AR(1) | 0.38 | 0.43 | [−0.53, 0.88] | |||
| A. Fixed-Effect Estimates | ||||||
| Parameter | β | Std. Error | df | t-Value | Sig. | 95% CI |
| Intercept | 5.90 | 1.76 | 71.98 | 3.35 | 0.001 | [2.39, 9.41] |
| Group (control) | 2.76 | 1.54 | 99.25 | 1.80 | 0.075 | [−0.29, 5.81] |
| Time (T0 vs. T2) | 0.30 | 1.36 | 40.59 | 0.22 | 0.825 | [−2.45, 3.05] |
| Time (T1 vs. T2) | 0.06 | 1.30 | 64.24 | 0.05 | 0.963 | [−2.53, 2.65] |
| Group × Time (control × T0) | 0.84 | 1.70 | 36.50 | 0.49 | 0.625 | [−2.60, 4.28] |
| Group × Time (control × T1) | −0.70 | 1.62 | 60.09 | −0.43 | 0.668 | [−3.93, 2.54] |
| Gender (female) | −0.51 | 1.30 | 49.04 | −0.39 | 0.699 | [−3.11, 2.10] |
| Ward (internal medicine) | −0.58 | 1.18 | 50.54 | −0.49 | 0.625 | [−3.00, 1.80] |
| Age | 0.02 | 0.04 | 38.81 | 0.45 | 0.653 | [−0.06, 0.09] |
| B. Type III Tests of Fixed Effects | ||||||
| Parameters | Num df | Den df | F | p-value | ||
| Group | 1 | 49.20 | 10.44 | 0.002 * | ||
| Time | 2 | 54.31 | 1.33 | 0.272 | ||
| Group × Time | 2 | 55.93 | 0.78 | 0.466 | ||
| Gender | 1 | 49.04 | 0.15 | 0.699 | ||
| Ward | 1 | 50.54 | 0.24 | 0.625 | ||
| Age | 1 | 38.81 | 0.21 | 0.653 | ||
| C. Variance Components and AR (1) Parameter | ||||||
| Component | Estimate | Std. Error | 95% CI | |||
| Random-intercept variance | 0.37 | 0.43 | [−0.53, 0.88] | |||
| Residual variance | 0.37 | 8.00 | [0, 5.73 × 1015] ** | |||
| AR(1) | 11.29 | 8.00 | [2.83, 45.05] | |||
| Pannel A. Fixed-Effect Estimates | ||||||
| Parameter | β | Std. Error | df | t-Value | p-Value | 95% CI |
| Intercept | 1.19 | 0.50 | 69.92 | 2.39 | 0.020 * | [0.20, 2.18] |
| Group (control) | 0.78 | 0.37 | 85.60 | 2.09 | 0.040 * | [0.04, 1.51] |
| Time (T0 vs. T2) | 0.45 | 0.28 | 36.51 | 1.61 | 0.115 | [−0.12, 1.02] |
| Time (T1 vs. T2) | 0.24 | 0.27 | 51.66 | 0.87 | 0.386 | [−0.31, 0.79] |
| Group × Time (control × T0) | −0.55 | 0.34 | 34.35 | −1.62 | 0.115 | [−1.25, 0.14] |
| Group × Time (control × T1) | –0.43 | 0.33 | 50.51 | –1.30 | 0.200 | [−1.10, 0.24] |
| Gender (female) | −0.02 | 0.39 | 51.18 | −0.04 | 0.970 | [−0.80, 0.77] |
| Ward (internal medicine) | 1.05 | 0.35 | 51.01 | 3.00 | 0.004 * | [0.35, 1.74] |
| Age | 0.00 | 0.01 | 45.79 | 0.37 | 0.712 | [−0.02, 0.03] |
| Panel B. Type III Tests of Fixed Effects | ||||||
| Parameters | Num df | Den df | F | p-value | ||
| Group | 1 | 56.03 | 3.04 | 0.087 | ||
| Time | 2 | 44.00 | 0.95 | 0.394 | ||
| Group × Time | 2 | 44.48 | 1.32 | 0.278 | ||
| Gender | 1 | 51.18 | 0.00 | 0.970 | ||
| Ward | 1 | 51.01 | 9.02 | 0.004 * | ||
| Age | 1 | 54.79 | 0.14 | 0.712 | ||
| C. Variance Components and AR (1) Parameter | ||||||
| Component | Estimate | Std. Error | 95% CI | |||
| Random-intercept variance | 0.54 | 0.19 | [0.27, 1.09] | |||
| Residual variance | 0.32 | 0.13 | [0.15, 0.71] | |||
| AR(1) | 0.19 | 0.34 | [−0.45, 0.71] | |||
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Grimm, J.S.; Kasdorf, A.; Voltz, R.; Strupp, J. From MINI to Meaningful Change—A German Pilot Study to Improve Patient Outcomes in End-of-Life Care. Healthcare 2025, 13, 2024. https://doi.org/10.3390/healthcare13162024
Grimm JS, Kasdorf A, Voltz R, Strupp J. From MINI to Meaningful Change—A German Pilot Study to Improve Patient Outcomes in End-of-Life Care. Healthcare. 2025; 13(16):2024. https://doi.org/10.3390/healthcare13162024
Chicago/Turabian StyleGrimm, Jana Sophie, Alina Kasdorf, Raymond Voltz, and Julia Strupp. 2025. "From MINI to Meaningful Change—A German Pilot Study to Improve Patient Outcomes in End-of-Life Care" Healthcare 13, no. 16: 2024. https://doi.org/10.3390/healthcare13162024
APA StyleGrimm, J. S., Kasdorf, A., Voltz, R., & Strupp, J. (2025). From MINI to Meaningful Change—A German Pilot Study to Improve Patient Outcomes in End-of-Life Care. Healthcare, 13(16), 2024. https://doi.org/10.3390/healthcare13162024








