Physiotherapy Screening for Referral of a Patient with Patent Foramen Ovale Presenting with Neck Pain as Primary Complaint: A Case Report
Abstract
1. Introduction
2. Case Presentation
2.1. Patient Information
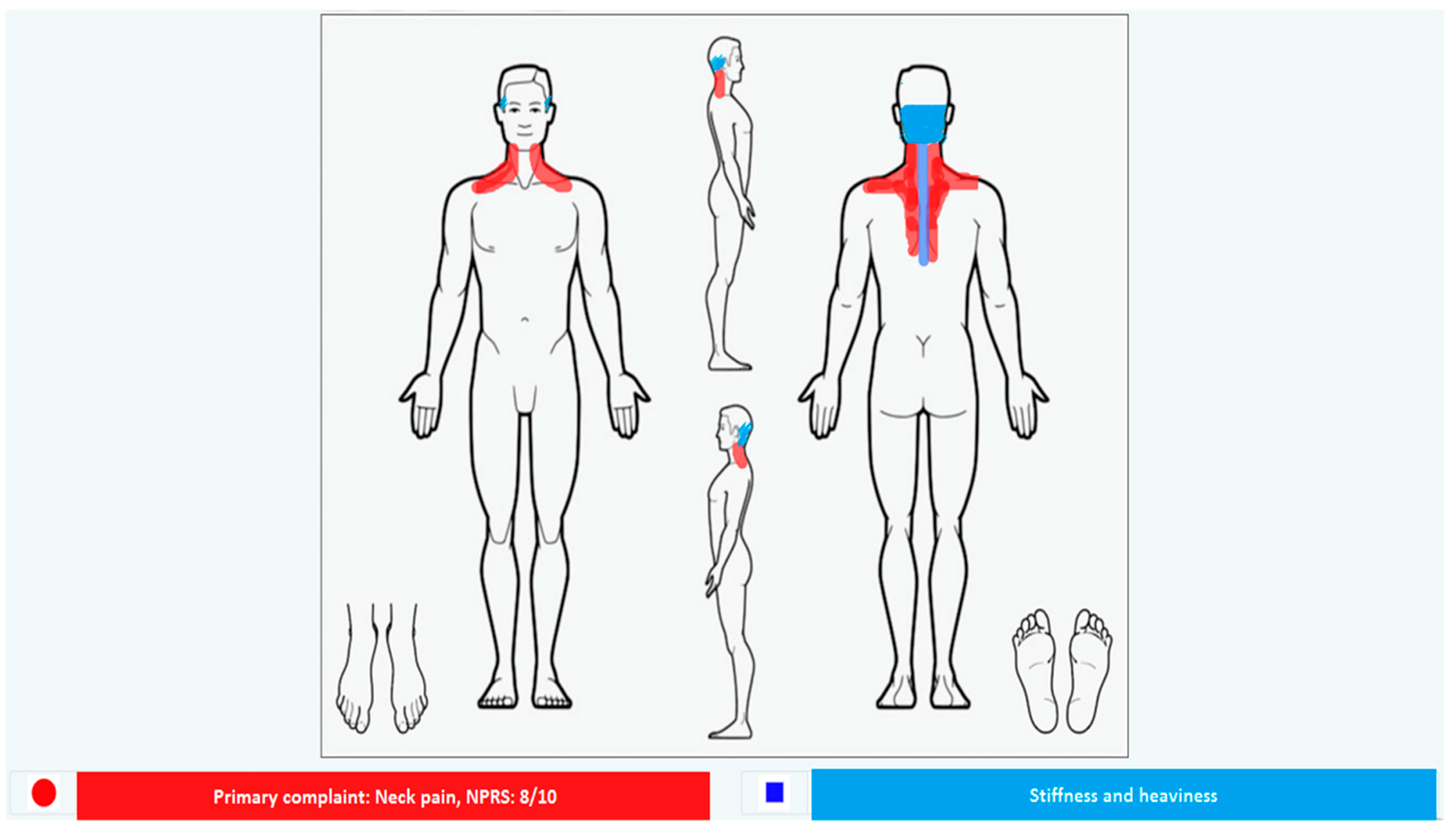
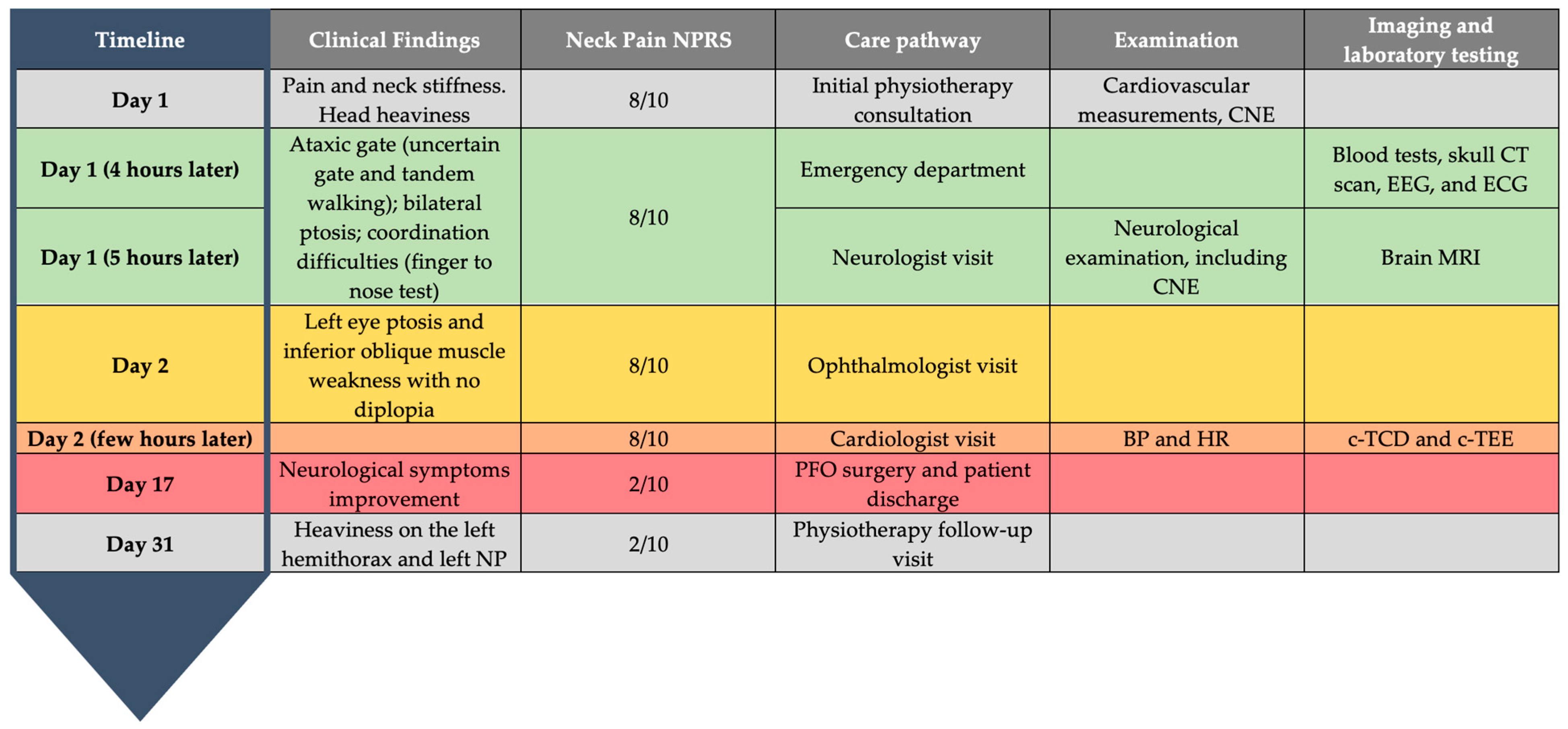
2.2. Clinical Findings
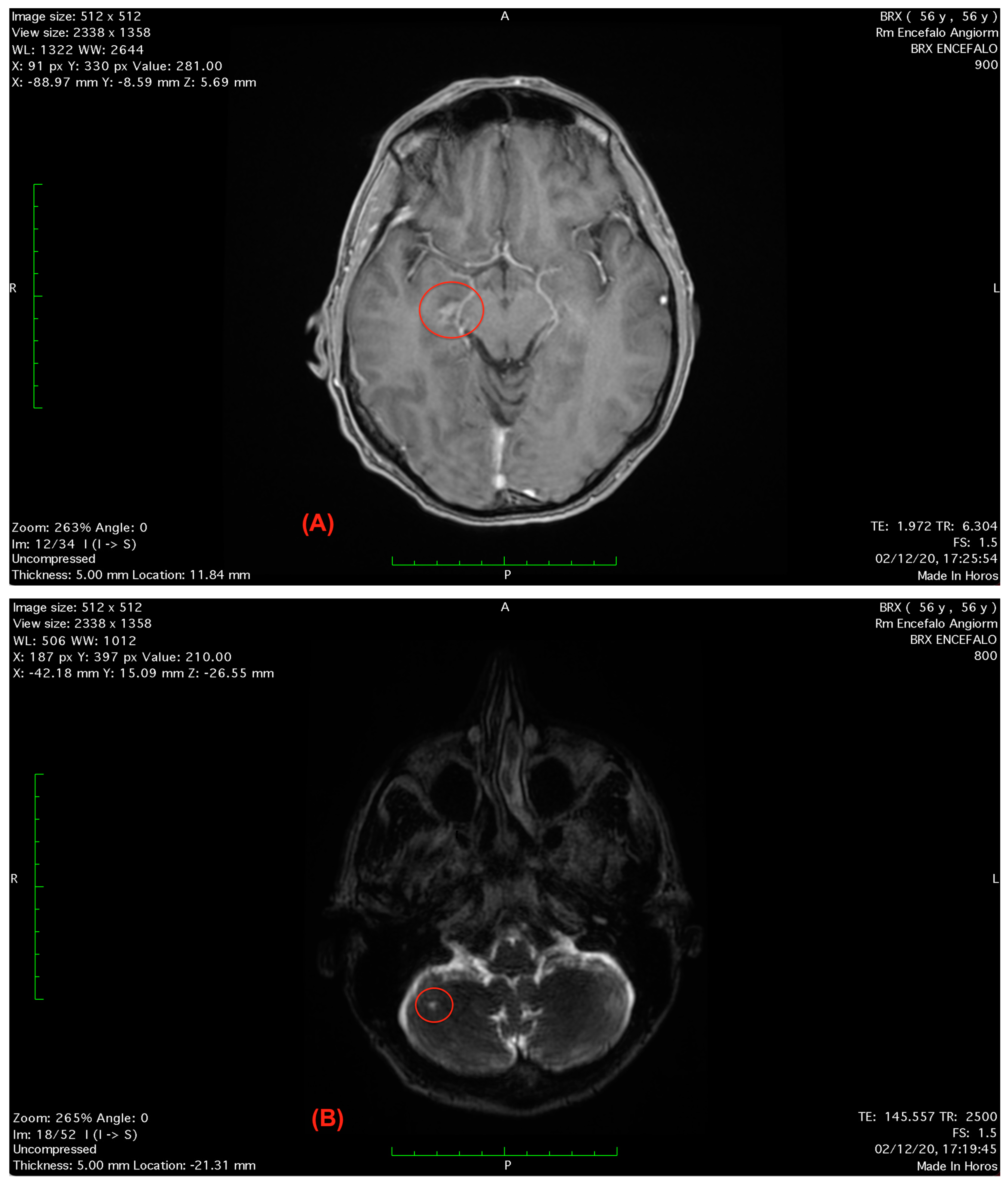
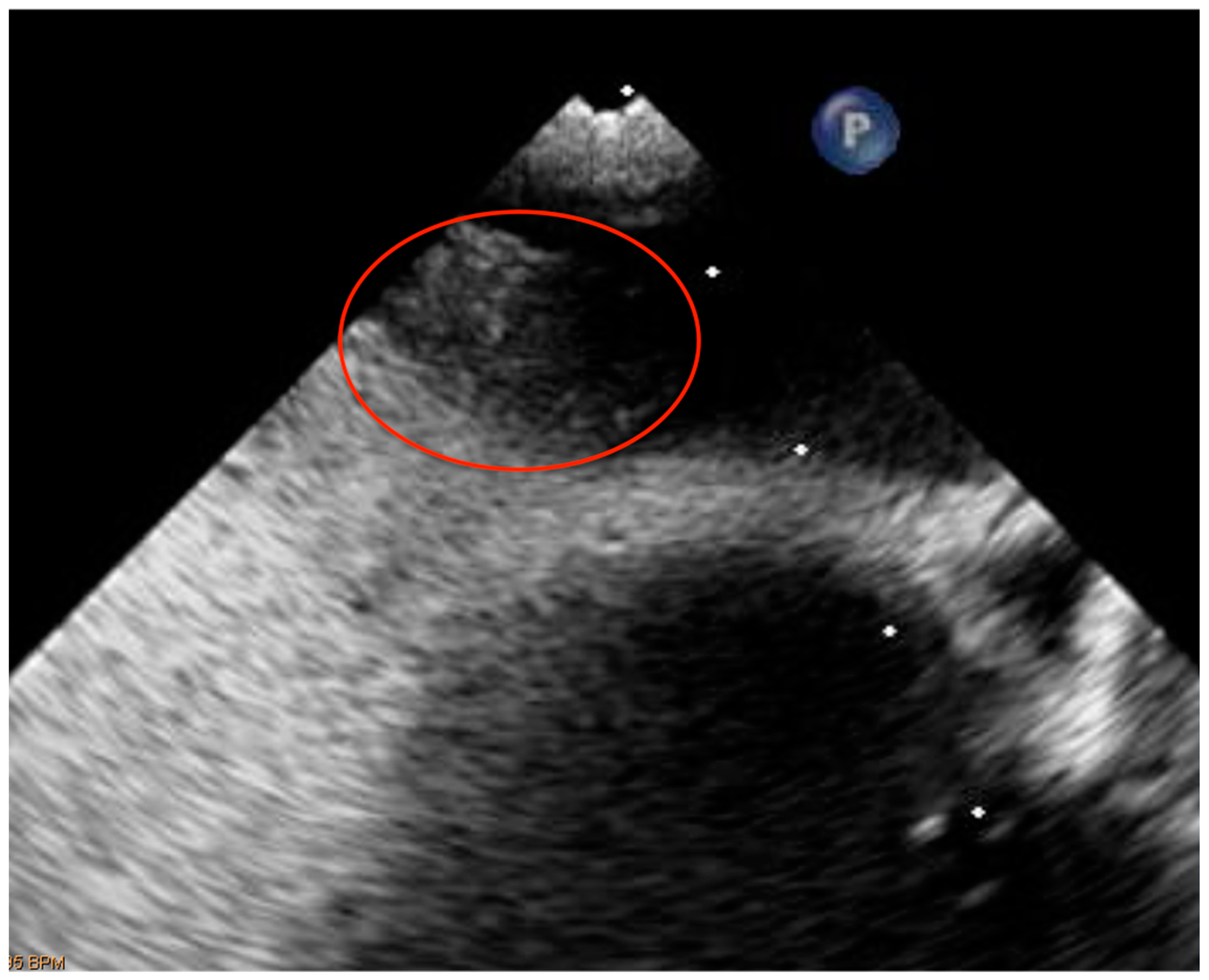
2.3. Analysis
Follow-Up and Outcomes
3. Discussion
Influence on Physiotherapy Clinical Practice
4. Conclusions
Patient Perspective
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Ethics Approval and Consent to Participate
Consent for Publication
Acknowledgments
Conflicts of Interest
Abbreviations
References
- Bovim, G.; Schrader, H.; Sand, T. Neck Pain in the General Population. Spine 1994, 19, 1307–1309. [Google Scholar] [CrossRef]
- Côté, P.; Wong, J.J.; Sutton, D.; Shearer, H.M.; Mior, S.; Randhawa, K.; Ameis, A.; Carroll, L.J.; Nordin, M.; Yu, H.; et al. Management of Neck Pain and Associated Disorders: A Clinical Practice Guideline from the Ontario Protocol for Traffic Injury Management (OPTIMa) Collaboration. Eur. Spine J. 2016, 25, 2000–2022. [Google Scholar] [CrossRef]
- Haldeman, S.; Carroll, L.; Cassidy, J.D.; Schubert, J.; Nygren, Å. The Bone and Joint Decade 2000–2010 Task Force on Neck Pain and Its Associated Disorders. Eur. Spine J. 2008, 17, 5–7. [Google Scholar] [CrossRef]
- Blanpied, P.R.; Gross, A.R.; Elliott, J.M.; Devaney, L.L.; Clewley, D.; Walton, D.M.; Sparks, C.; Robertson, E.K. Clinical Practice Guidelines Linked to the International Classification of Functioning, Disability and Health from the Orthopaedic Section of the American Physical Therapy Association. J. Orthop. Sports Phys. Ther. 2017, 47, A1–A83. [Google Scholar] [CrossRef]
- Mourad, F.; Giovannico, G.; Maselli, F.; Bonetti, F.; Fernández De Las Peñas, C.; Dunning, J. Basilar Impression Presenting as Intermittent Mechanical Neck Pain: A Rare Case Report Rehabilitation, Physical Therapy and Occupational Health. BMC Musculoskelet. Disord. 2016, 17, 4–8. [Google Scholar] [CrossRef]
- Mourad, F.P.; Maselli, F.P.; Cataldi, F.P.; Pennella, D.P.; Fernández-De-Las-Peñas, C.P.; James Dunning, D.P.T. Hip Bone Marrow Edema Presenting as Low Back Pain: A Case Report. Taylor Fr. 2018, 36, 249–257. [Google Scholar] [CrossRef]
- Mourad, F.; Patuzzo, A.; Dunning, J.R. Craniopharyngioma in a Young Woman with Symptoms Presenting as Mechanical Neck Pain Associated with Cervicogenic Headache: A Case Report Migraine and Other Primary Headaches View Project Gender Medicine in Physiotherapy View Project. Artic. Physiother. Theory Pract. 2019, 37, 549–558. [Google Scholar] [CrossRef]
- Hutting, N.; Mourad, F.; Kranenburg, R.; Wilbrink, W.; Kerry, R.; Taylor, A. What to Look Out for, What to Do, and When: 3 Key Messages for Safely Treating Neck Pain, Headache, and/or Orofacial Symptoms in Musculoskeletal Rehabilitation Settings. J. Orthop. Sports Phys. Ther. 2023, 53, 59–63. [Google Scholar] [CrossRef]
- Rushton, A.; Carlesso, L.C.; Flynn, T.; Hing, W.A.; Rubinstein, S.M.; Vogel, S.; Kerry, R. Position Statement: International Framework for Examination of the Cervical Region for Potential of Vascular Pathologies of the Neck Prior to Musculoskeletal Intervention: International IFOMPT Cervical Framework. J. Orthop. Sports Phys. Ther. 2022, 53, 7–22. [Google Scholar] [CrossRef]
- Bussieres, A.; Stewart, G.; Al-Zoubi, F.; Decina, P.; Descarreaux, M.; Hayden, J.; Hendrickson, B.; Hincapié, C.; Pagé, I.; Passmore, S.; et al. The Treatment of Neck Pain–Associated Disorders and Whiplash-Associated Disorders: A Clinical Practice Guideline; Elsevier: Amsterdam, The Netherlands, 2016. [Google Scholar]
- Platzer, P.; Hauswirth, N.; Jaindl, M.; Chatwani, S.; Vecsei, V.; Gaebler, C. Delayed or Missed Diagnosis of Cervical Spine Injuries. J. Trauma Acute Care Surg. 2006, 61, 150–155. [Google Scholar] [CrossRef]
- Albarrati, A. Outpatient Physical Therapy Cardiovascular Assessment: Physical Therapist Perspective and Experience. Artic. Physiother. Theory Pract. 2018, 35, 843–850. [Google Scholar] [CrossRef] [PubMed]
- Finucane, L.L.M.; Downie, A.; Mercer, C.; Greenhalgh, S.M.; Boissonnault, W.G.; Pool-Goudzwaard, A.L.; Beneciuk, J.M.; Leech, R.L.; Selfe, J. International Framework for Red Flags for Potential Serious Spinal Pathologies. J. Orthop. Sports Phys. Ther. 2020, 50, 350–372. [Google Scholar] [CrossRef] [PubMed]
- Severin, R.; Wang, E.; Wielechowski, A.; Phillips, S.A. Outpatient Physical Therapist Attitudes toward and Behaviors in Cardiovascular Disease Screening: A National Survey. Phys. Ther. 2020, 99, 833–848. [Google Scholar] [CrossRef] [PubMed]
- Sizer, P.S.; Brismée, J.M.; Cook, C. Medical Screening for Red Flags in the Diagnosis and Management of Musculoskeletal Spine Pain. Pain Pract. 2007, 7, 53–71. [Google Scholar] [CrossRef]
- Mourad, F.; Lopez, G.; Cataldi, F.; Maselli, F.; Pellicciari, L.; Salomon, M.; Kranenburg, H.; Kerry, R.; Taylor, A.; Hutting, N. Assessing Cranial Nerves in Physical Therapy Practice: Findings from a Cross-Sectional Survey and Implication for Clinical Practice. Healthcare 2021, 9, 1262. [Google Scholar] [CrossRef]
- Taylor, A.; Mourad, F.; Kerry, R.; Hutting, N. A Guide to Cranial Nerve Testing for Musculoskeletal Clinicians. J. Man. Manip. Ther. 2021, 29, 376–389. [Google Scholar] [CrossRef]
- Scherer, S. Addressing Cardiovascular Risk as Part of Physical Therapist Practice—What about Practice Recommendations for Physical Therapists? Cardiopulm. Phys. Ther. J. 2009, 20, 27–29. [Google Scholar] [CrossRef]
- Faletra, A.; Bellin, G.; Dunning, J.; Fernández-de-Las-Peñas, C.; Pellicciari, L.; Brindisino, F.; Galeno, E.; Rossettini, G.; Maselli, F.; Severin, R. Assessing Cardiovascular Parameters and Risk Factors in Physical Therapy Practice: Findings from a Cross-Sectional National Survey and Implication for Clinical Practice. BMC Musculoskelet. Disord. 2022, 23, 749. [Google Scholar] [CrossRef]
- Boisonnault, W.G. Examination in Physical Therapy Practice: Screening for Medical Disease; Churchill Livingstone: London, UK, 1995. [Google Scholar]
- Feller, D.; Giudice, A.; Faletra, A.; Salomon, M.; Galeno, E.; Rossettini, G.; Brindisino, F.; Maselli, F.; Hutting, N.; Mourad, F. Identifying peripheral arterial diseases or flow limitations of the lower limb: Important aspects for cardiovascular screening for referral in physiotherapy. Musculoskelet. Sci. Pract. 2022, 61, 102611. [Google Scholar] [CrossRef]
- Maselli, F.; Palladino, M.; Barbari, V.; Storari, L.; Rossettini, G.; Testa, M. The Diagnostic Value of Red Flags in Thoracolumbar Pain: A Systematic Review. Disabil. Rehabil. 2022, 44, 1190–1206. [Google Scholar] [CrossRef]
- Debette, S.; Leys, D. Cervical-Artery Dissections: Predisposing Factors, Diagnosis, and Outcome. Lancet Neurol. 2009, 8, 668–678. [Google Scholar] [CrossRef] [PubMed]
- Kerry, R.; Taylor, A.J. Cervical Arterial Dysfunction: Knowledge and Reasoning for Manual Physical Therapists. J. Orthop. Sports Phys. Ther. 2009, 39, 378–387. [Google Scholar] [CrossRef] [PubMed]
- Thomas, L.C.; Rivett, D.A.; Attia, J.R.; Levi, C. Risk Factors and Clinical Presentation of Cervical Arterial Dissection: Preliminary Results of a Prospective Case-Control Study. J. Orthop. Sports Phys. Ther. 2015, 45, 503–511. [Google Scholar] [CrossRef] [PubMed]
- Storari, L.; Barbari, V.; Brindisino, F.; Testa, M.; Filippo, M. An Unusual Presentation of Acute Myocardial Infarction in Physiotherapy Direct Access: Findings from a Case Report. Arch. Physiother. 2021, 11, 5. [Google Scholar] [CrossRef]
- Weber, M.D. Screening and Evaluation of the Cardiovascular and Pulmonary Systems in Patients Presenting with Upper Extremity Impairments; Elsevier: Amsterdam, The Netherlands, 2010. [Google Scholar]
- Cooper, R.S.; Liao, Y. Impact of Left Ventricular Hypertrophy on Ventricular Arrhythmias in the Absence of Coronary Artery Disease. J. Am. Coll. Cardiol. 1991, 17, 1277–1282. [Google Scholar] [CrossRef]
- Jouven, X.; Desnos, M.; Guerot, C.; Ducimetière, P. Predicting Sudden Death in the Population. The Paris Prospective Study I. Circulation 1999, 99, 1978–1983. [Google Scholar] [CrossRef]
- Risgaard, B.; Winkel, B.G.; Jabbari, R.; Glinge, C.; Ingemann-Hansen, O.; Thomsen, J.L.; Ottesen, G.L.; Haunsø, S.; Holst, A.G.; Tfelt-Hansen, J. Sports-related sudden cardiac death in a competitive and a noncompetitive athlete population aged 12 to 49 years: Data from an unselected nationwide study in Denmark. Heart Rhythm. 2014, 11, 1673–1681. [Google Scholar] [CrossRef]
- Savonen, K.; Lakka, T.A.; Laukkanen, J.A.; Halonen, P.M.; Rauramaa, T.H.; Salonen, J.T.; Rauramaa, R. Heart Rate Response during Exercise Test and Cardiovascular Mortality in Middle-Aged Men. Eur. Heart J. 2006, 27, 582–588. [Google Scholar] [CrossRef]
- Perry, M. Differential diagnosis: Musculoskeletal pain vs. cardiogenic pain. Research innovation scholarship entrepreneurship 18-11–2020. 2012. [Google Scholar]
- APTA. APTA Guide to Physical Therapist Practice. Phys. Ther. 2014, 81, 7–9. [Google Scholar]
- Kosuge, M.; Kimura, K.; Ishikawa, T.; Ebina, T.; Hibi, K.; Tsukahara, K.; Kanna, M.; Iwahashi, N.; Okuda, J.; Nozawa, N.; et al. Differences Between Men and Women in Terms of Clinical Features of ST-Segment Elevation Acute Myocardial Infarction. Circ. J. 2006, 70, 222–226. [Google Scholar] [CrossRef]
- Lu, L.; Liu, M.; Sun, R.; Zheng, Y.; Zhang, P. Myocardial Infarction: Symptoms and Treatments. Cell Biochem. Biophys. 2015, 72, 865–867. [Google Scholar] [CrossRef] [PubMed]
- Andreoletti, F.; Maselli, F.; Storari, L.; Vongher, A.; Erbesato, M.; Testa, M.; Turolla, A. Screening for Referral of Serious Pathology by Physical Examination Tests in Patients with Back or Chest Pain: A Systematic Review. Int. J. Environ. Res. Public Health 2022, 19, 16418. [Google Scholar] [CrossRef] [PubMed]
- Roth, G.A.; Mensah, G.A.; Johnson, C.O.; Addolorato, G.; Ammirati, E.; Baddour, L.M.; Barengo, N.C.; Beaton, A.Z.; Benjamin, E.J.; Benziger, C.P.; et al. Global Burden of Cardiovascular Diseases and Risk Factors, 1990–2019: Update From the GBD 2019 Study. J. Am. Coll. Cardiol. 2020, 76, 2982–3021. [Google Scholar] [CrossRef]
- Zuin, M.; Rigatelli, G. Thrombus Straddling a Patent Foramen Ovale: Also a Myocardial Infarction Matter? Int. J. Cardiol. 2021, 340, 24–25. [Google Scholar] [CrossRef] [PubMed]
- Alakbarzade, V.; Keteepe-Arachi, T.; Karsan, N.; Ray, R.; Pereira, A.C. Patent Foramen Ovale. Pract. Neurol. 2020, 20, 225–233. [Google Scholar] [CrossRef]
- Overell, J.R.; Bone, I.; Lees, K.R. Interatrial Septal Abnormalities and Stroke: A Meta-Analysis of Case-Control Studies. Neurology 2000, 55, 1172–1179. [Google Scholar] [CrossRef]
- Valensi, P.; Lorgis, L.; Cottin, Y. Prevalence, Incidence, Predictive Factors and Prognosis of Silent Myocardial Infarction: A Review of the Literature. Arch. Cardiovasc. Dis. 2011, 104, 178–188. [Google Scholar] [CrossRef]
- Riley, D.S.; Barber, M.S.; Kienle, G.S.; Aronson, J.K.; von Schoen-Angerer, T.; Tugwell, P.; Kiene, H.; Helfand, M.; Altman, D.G.; Sox, H.; et al. CARE guidelines for case reports: Explanation and elaboration document. J. Clin. Epidemiol. 2017, 89, 218–235. [Google Scholar] [CrossRef]
- Young, I.A.; Dunning, J.; Butts, R.; Mourad, F.; Cleland, J.A. Reliability, Construct Validity, and Responsiveness of the Neck Disability Index and Numeric Pain Rating Scale in Patients with Mechanical Neck Pain without Upper Extremity Symptoms. Physiother. Theory Pract. 2019, 35, 1328–1335. [Google Scholar] [CrossRef]
- Ten Cate, H. Trombocytopenia: One of the markers of disseminated intravascular coagulation. Pathophysiol. Haemost. Thromb. 2003, 33, 413–416. [Google Scholar] [CrossRef]
- Sielski, J.; Wałek, P.; Siuda, M. Acute myocardial infarction and chronic leukaemia with significant thrombocytopenia. Kardiol. Pol. 2011, 69, 1072–1074. [Google Scholar] [PubMed]
- Matthai, W.H., Jr. Thrombocytopenia in cardiovascular patients: Diagnosis and management. Chest 2005, 127 (Suppl. S2), 46S–52S. [Google Scholar] [CrossRef] [PubMed]
- Unger, T.; Borghi, C.; Charchar, F.; Khan, N.A.; Poulter, N.R.; Prabhakaran, D.; Ramirez, A.; Schlaich, M.; Stergiou, G.S.; Tomaszewski, M.; et al. 2020 International Society of Hypertension global hypertension practice guidelines. Am. Heart Assoc. 2020, 75, 1334–1357. [Google Scholar]
- Wang, C.C.; Chao, J.K.; Chang, Y.H.; Chou, C.L.; Kao, C.L. Care for patients with musculoskeletal pain during the COVID-19 pandemic: Physical therapy and rehabilitation suggestions for pain management. J. Chin. Med. Assoc. 2020, 83, 822–824. [Google Scholar] [CrossRef]
- Rondoni, A.; Rossettini, G.; Ristori, D. Intrarater and Inter-Rater Reliability of Active Cervical Range of Motion in Patients with Nonspecific Neck Pain Measured with Technological and Common Use Devices: A Systematic Review With Meta-regression. J. Manip. Physiol. Ther. 2017, 40, 597–608. [Google Scholar] [CrossRef]
- Hatzaras, I.; Tranquilli, M.; Coady, M.; Barrett, P.M.; Bible, J.; Elefteriades, J.A. Weight lifting and aortic dissection: More evidence for a connection. Cardiology 2007, 107, 103–106. [Google Scholar] [CrossRef]
- Gambazza, S.; Bargeri, S.; Campanini, I.; Meroni, R.; Turolla, A.; Castellini, G.; Gianola, S. Prevalence and Associated Factors of COVID-19 across Italian Regions: A Secondary Analysis from a National Survey on Physiotherapists. Arch. Physiother. 2021, 11, 30. [Google Scholar] [CrossRef]
- Severin, R.; Sabbahi, A.; Albarrati, A.; Phillips, S.A.; Arena, S. Blood Pressure Screening by Outpatient Physical Therapists: A Call to Action and Clinical Recommendations. Phys. Ther. 2020, 100, 1008–1019. [Google Scholar] [CrossRef]
- Kayrak, M.; Bacaksiz, A.; Vatankulu, M.A.; Ayhan, S.S.; Kaya, Z.; Ari, H.; Sonmez, O.; Gok, H. Exaggerated Blood Pressure Response to Exercise—A New Portent of Masked Hypertension. Clin. Exp. Hypertens. 2010, 32, 560–568. [Google Scholar] [CrossRef]
- Currie, K.D.; Floras, J.S.; La Gerche, A.; Goodman, J.M. Exercise Blood Pressure Guidelines: Time to Re-evaluate What is Normal and Exaggerated? Sports Med. 2018, 48, 1763–1771. [Google Scholar] [CrossRef]
- Sharman, J.E.; LaGerche, A. Exercise blood pressure: Clinical relevance and correct measurement. J. Hum. Hypertens. 2015, 29, 351–358. [Google Scholar] [CrossRef] [PubMed]
- Wielemborek-Musial, K.; Szmigielska, K.; Leszczynska, J.; Jegier, A. Blood Pressure Response to Submaximal Exercise Test in Adults. Biomed. Res. Int. 2016, 2016, 5607507. [Google Scholar] [CrossRef] [PubMed]
- Schultz, M.G.; Otahal, P.; Cleland, V.J.; Blizzard, L.; Marwick, T.H.; Sharman, J.E. Exercise-induced hypertension, cardiovascular events, and mortality in patients undergoing exercise stress testing: A systematic review and meta-analysis. Am. J. Hypertens. 2013, 26, 357–366. [Google Scholar] [CrossRef] [PubMed]
- Schultz, M.G.; Hare, J.L.; Marwick, T.H.; Stowasser, M.; Sharman, J.E. Masked Hypertension Is “Unmasked” by Low-Intensity Exercise Blood Pressure. Blood Press. 2011, 20, 284–289. [Google Scholar] [CrossRef] [PubMed]
- Schultz, M.G.; Otahal, P.; Picone, D.S.; Sharman, J.E. Clinical Relevance of Exaggerated Exercise Blood Pressure. J. Am. Coll. Cardiol. 2015, 66, 1843–1845. [Google Scholar] [CrossRef]
- Cook, C.E.; Hegedus, E.; Pietrobon, R.; Goode, A. A pragmatic neurological screen for patients with suspected cord compressive myelopathy. Phys. Ther. 2007, 87, 1233–1242. [Google Scholar] [CrossRef] [PubMed]
- Matuszak, J.M.; McVige, J.; McPherson, J.; Willer, B.; Leddy, J. A Practical Concussion Physical Examination Toolbox: Evidence-Based Physical Examination for Concussion. Sports Health 2016, 8, 260–269. [Google Scholar] [CrossRef]
- Ioannidis, S.G.; Mitsias, P.D. Patent Foramen Ovale in Cryptogenic Ischemic Stroke: Direct Cause, Risk Factor, or Incidental Finding? Front. Neurol. 2020, 11, 567. [Google Scholar] [CrossRef]
- Miranda, B.; Fonseca, A.C.; Ferro, J.M. Patent Foramen Ovale and Stroke. J. Neurol. 2018, 265, 1943–1949. [Google Scholar] [CrossRef]
- Drighil, A.; El Mosalami, H.; Elbadaoui, N.; Chraibi, S.; Bennis, A. Patent foramen ovale: A new disease? Int. J. Cardiol. 2007, 122, 1–9. [Google Scholar] [CrossRef]
- Desai, A.J.; Fuller, C.J.; Jesurum, J.T.; Reisman, M. Patent foramen ovale and cerebrovascular diseases. Nat. Clin. Pract. Cardiovasc. Med. 2006, 3, 446–455. [Google Scholar] [CrossRef] [PubMed]
- Tullio, M.D.; Sacco, R.L.; Venketasubramanian, N.; Sherman, D.; Mohr, J.P.; Homma, S. Comparison of Diagnostic Techniques for the Detection of a Patent Foramen Ovale in Stroke Patients. Stroke 1993, 24, 1020–1024. [Google Scholar] [CrossRef] [PubMed]
- Alsheikh-Ali, A.A.; Thaler, D.E.; Kent, D.M. Patent Foramen Ovale in Cryptogenic Stroke: Incidental or Pathogenic? Stroke 2009, 40, 2349–2355. [Google Scholar] [CrossRef] [PubMed]
- Homma, S.; Di Tullio, M.R.; Sacco, R.L.; Sciacca, R.R.; Smith, C.; Mohr, J.P. Surgical Closure of Patent Foramen Ovale in Cryptogenic Stroke Patients. Stroke 1997, 28, 2376–2381. [Google Scholar] [CrossRef]
- Mi, M.; Block, P.; Boderick, J. PFO Closure for Cryptogenic Stroke. Cardiol. Rev. 2017, 25, 147–157. [Google Scholar] [CrossRef]
- Meier, B.; Lock, J.E. Contemporary Management of Patent Foramen Ovale. Circulation 2003, 107, 5–9. [Google Scholar] [CrossRef]
- Onorato, E.; Melzi, G.; Casilli, F.; Pedon, L.; Rigatelli, G.; Carrozza, A.; Maiolino, P.; Zanchetta, M.; Morandi, E.; Angeli, S.; et al. Patent Foramen Ovale with Paradoxical Embolism: Mid-Term Results of Transcatheter Closure in 256 Patients. J. Intervent. Cardiol. 2003, 16, 43–50. [Google Scholar] [CrossRef]
- Hung, J.; Landzberg, M.J.; Jenkins, K.J.; King, M.E.E.; Lock, J.E.; Palacios, I.F.; Lang, P. Closure of Patent Foramen Ovale for Paradoxical Emboli: Intermediate-Term Risk of Recurrent Neurological Events Following Transcatheter Device Placement. J. Am. Coll. Cardiol. 2000, 35, 1311–1316. [Google Scholar] [CrossRef]
- Wahl, A.; Meier, B.; Haxel, B.; Nedeltchev, K.; Arnold, M.; Eicher, E.; Sturzenegger, M.; Seiler, C.; Mattle, H.P.; Windecker, S. Prognosis after percutaneous closure of patent foramen ovale for paradoxical embolism. Neurology 2001, 57, 1330–1332. [Google Scholar] [CrossRef]
- Schuchlenz, H.W.; Weihs, W.; Berghold, A.; Lechner, A.; Schmidt, R. Secondary prevention after cryptogenic cerebrovascular events in patients with patent foramen ovale. Int. J. Cardiol. 2005, 101, 77–82. [Google Scholar] [CrossRef]
- Piscitelli, D.; Furmanek, M.P.; Meroni, R.; De Caro, W.; Pellicciari, L. Direct Access in Physical Therapy: A Systematic Review. Clin. Ter. 2018, 169, e249–e260. [Google Scholar] [CrossRef] [PubMed]
- Maselli, F.; Piano, L.; Cecchetto, S.; Storari, L.; Rossettini, G.; Mourad, F. Direct Access to Physical Therapy: Should Italy Move Forward? Int. J. Environ. Res. Public. Health 2022, 19, 555. [Google Scholar] [CrossRef] [PubMed]
- Maselli, F.; Piano, L.; Cecchetto, S.; Storari, L.; Rossettini, G.; Mourad, F. Would Moving Forward Mean Going Back? Comment on “Maselli et al. Direct Access to Physical Therapy: Should Italy Move Forward? Int. J. Environ. Res. Public Health 2022, 19, 555.”. Int. J. Environ. Res. Public Health 2022, 19, 4620. [Google Scholar] [CrossRef] [PubMed]
- Hutting, N.; Kerry, R.; Kranenburg, R.; Mourad, F.; Taylor, A. Assessing Vascular Function in Patients with Neck Pain, Headache, and/or Orofacial Pain: Part of the Job Description of All Physical Therapists. J. Orthop. Sports Phys. Ther. 2021, 51, 418–421. [Google Scholar] [CrossRef] [PubMed]
- Lip, P.Z.Y.; Lip, G.Y.H. Patent Foramen Ovale and Migraine Attacks: A Systematic Review. Am. J. Med. 2014, 127, 411–420. [Google Scholar] [CrossRef]
- Lechat, P.; Mas, J.L.; Lascault, G.; Loron, P.; Theard, M.; Klimczac, M.; Drobinski, G.; Thomas, D.; Grosgogeat, Y. Prevalence of patent foramen ovale in patients with stroke. N. Engl. J. Med. 1988, 318, 1148–1152. [Google Scholar] [CrossRef]
- Homma, S.; Sacco, R.L. Patent Foramen Ovale and Stroke. Circulation 2005, 112, 1063–1072. [Google Scholar] [CrossRef]
- Katsanos, A.H.; Spence, J.D.; Bogiatzi, C.; Parissis, J.; Giannopoulos, S.; Frogoudaki, A.; Safouris, A.; Voumvourakis, K.; Tsivgoulis, G. Recurrent Stroke and Patent Foramen Ovale: A Systematic Review and Meta-Analysis. Stroke 2014, 45, 3352–3359. [Google Scholar] [CrossRef]
- Klötzsch, C.; Sliwka, U.; Berlit, P.; Noth, J. An increased frequency of patent foramen ovale in patients with transient globalamnesia. Analysis of 53 consecutive patients. Arch. Neurol. 1996, 53, 504–508. [Google Scholar] [CrossRef]
- Bury, T.J.; Stokes, E.K. Direct access and patient/client self-referral to physiotherapy: A review of contemporary practice within the European Union. Physiotherapy 2013, 99, 285–291. [Google Scholar] [CrossRef]
- Thomas, L.C. Cervical arterial dissection: An overview and implications for manipulative therapy practice. Man. Ther. 2016, 21, 2–9. [Google Scholar] [CrossRef]
- Hypertension in Adults: Diagnosis and Management. National Institute for Health and Care Excellence (NICE): London, UK, (NICE Guideline, No. 136). Available online: https://www.ncbi.nlm.nih.gov/books/NBK547161/ (accessed on 18 March 2022).
- Bromfield, S.; Muntner, P. High Blood Pressure: The Leading Global Burden of Disease Risk Factor and the Need for Worldwide Prevention Programs; Springer: Berlin/Heidelberg, Germany, 2013. [Google Scholar] [CrossRef]
- Kessler, C.S.; Joudeh, Y. Evaluation and treatment of severe asymptomatic hypertension. Am. Fam. Physician. 2010, 81, 470–476. [Google Scholar]
- Banik, S. Blood Pressure and Incidence of Twelve Cardiovascular Diseases: Lifetime Risks, Healthy Life-Years Lost and Age-Specific Associations in 1.25 Million People. J. Neuroanaesth. Crit. Care 2014, 1, 224–225. [Google Scholar] [CrossRef]
- Luft, F.C. Twins in Cardiovascular Genetic Research. Hypertension 2001, 37, 350–356. [Google Scholar] [CrossRef] [PubMed]
- Fagard, R.; Brguljan, J.; Staessen, J.; Thijs, L.; Derom, C.; Thomis, M.; Vlietinck, R. Heritability of Conventional an Ambulatory Blood Pressures: A Study in Twins. Hypertension 1995, 26, 919–924. [Google Scholar] [CrossRef] [PubMed]
- Gangwisch, J.E. A review of evidence for the link between sleep duration and hypertension. Am. J. Hypertens. 2014, 27, 1235–1242. [Google Scholar] [CrossRef] [PubMed]
- Palagini, L.; Bruno, R.M.; Gemignani, A.; Baglioni, C.; Ghiadoni, L.; Riemann, D. Sleep Loss and Hypertension: A Systematic Review Send Orders of Reprints at Bspsaif@emirates.Net.Ae Sleep Loss and Hypertension: A Systematic Review. Curr. Pharm. Des. 2013, 19, 2409–2419. [Google Scholar] [CrossRef] [PubMed]
- Lin, I.; Wiles, L.; Waller, R.; Goucke, R.; Nagree, Y.; Gibberd, M.; Straker, L.; Maher, C.G.; O’sullivan, P.P.B. What Does Best Practice Care for Musculoskeletal Pain Look like? Eleven Consistent Recommendations from High-Quality Clinical Practice Guidelines: Systematic Review. Br. J. Sport. Med. 2020, 54, 79–86. [Google Scholar] [CrossRef]
- Arnold, M.; Cumurciuc, R.; Stapf, C.; Favrole, P.; Berthet, K.; Bousser, M.G. Pain as the only symptom of cervical artery dissection. J. Neurol. Neurosurg. Psychiatry 2006, 77, 1021–1024. [Google Scholar] [CrossRef]
- Patel, R.R.; Adam, R.; Maldjian, C.; Lincoln, C.M.; Yuen, A.; Arneja, A. Cervical carotid artery dissection: Current review of diagnosis and treatment. Cardiol. Rev. 2012, 20, 145–152. [Google Scholar] [CrossRef]
- Redekop, G. Extracranial Carotid and Vertebral Artery Dissection: A Review. Can. J. Neurol. Sci. 2008, 35, 146–152. [Google Scholar] [CrossRef] [PubMed]
- Koletsos, N.; Dipla, K.; Triantafyllou, A.; Gkaliagkousi, E.; Sachpekidis, V.; Zafeiridis, A.; Douma, S. A brief submaximal isometric exercise test ‘unmasks’ systolic and diastolic masked hypertension. J. Hypertens. 2019, 37, 710–719. [Google Scholar] [CrossRef] [PubMed]
- Gorostidi, M.; Vinyoles, E.; Banegas, J.R.; de la Sierra, A. Prevalence of white-coat and masked hypertension in national and international registries. Hypertens. Res. 2015, 38, 1–7. [Google Scholar] [CrossRef]
- Peacock, J.; Diaz, K.M.; Viera, A.J.; Schwartz, J.E.; Shimbo, D. Unmasking masked hypertension: Prevalence, clinical implications, diagnosis, correlates and future directions. J. Hum. Hypertens. 2014, 28, 521–528. [Google Scholar] [CrossRef]
- Zhang, D.; Shen, X.; Qi, X. Resting Heart Rate and All-Cause and Cardiovascular Mortality in the General Population: A Meta-Analysis. Can. Med. Assoc. 2016, 188, E53–E63. [Google Scholar] [CrossRef]
- Fletcher, G.F.; Ades, P.A.; Kligfield, P.; Arena, R.; Balady, G.J.; Bittner, V.A.; Coke, L.A.; Fleg, J.L.; Forman, D.E.; Gerber, T.C.; et al. Exercise standards for testing and training: A scientific statement from the American Heart Association. Circulation 2013, 128, 873–934. [Google Scholar] [CrossRef] [PubMed]
- Maselli, F.; Testa, M. Superficial peroneal nerve schwannoma presenting as lumbar radicular syndrome in a non-competitive runner. J. Back Musculoskelet. Rehabil. 2019, 32, 361–365. [Google Scholar] [CrossRef]
- Bayles, M.P. ACSM’s Guidelines for Exercise Testing and Prescription; Lippincott Williams & Wilkins: Philadelphia, PA, USA, 2013. [Google Scholar]
- Boissonnault, W.G.; Ross, M.D. Physical therapists referring patients to physicians: A review of case reports and series. J. Orthop. Sports Phys. Ther. 2012, 42, 446–454. [Google Scholar] [CrossRef]
- Ross, M.D.; Boissonnault, W.G. Red Flags: To Screen or Not to Screen? J. Orthop. Sports Phys. Ther. 2010, 40, 682–684. [Google Scholar] [CrossRef]
- Taylor, A.; Kerry, R.; Mourad, F.; Hutting, N. Vascular flow limitations affecting the cervico-cranial region: Understanding ischaemia. Brazilian journal of physical therapy. 2023, 27, 100493. [Google Scholar] [CrossRef]
- Mourad, F.; Giudice, A.; Maritati, G.; Maselli, F.; Kranenburg, R.; Taylor, A.; Kerry, R.; Hutting, N. A guide to identify cervical autonomic dysfunctions (and associated conditions) in patients with musculoskeletal disorders in physical therapy practice. Braz. J. Phys. Ther. 2023, 27, 100495. [Google Scholar] [CrossRef]
| Time (minutes) | Heart Rate (bpm) | Blood Pressure (mmHg) | RPE | Symptoms |
|---|---|---|---|---|
| 0 (RHR and RBP were used as baseline pre-exercise measurement) | 68 | 130/80 | 0 | Neck pain and head heaviness |
| 3 | 91 | 145/85 | 0 | Neck pain and stiffness, head heaviness |
| 6 | 96 | 160/95 | 5 | Familiar NP onset |
| 9 | 101 | 190/100 | 8 | Sharp BP and NP increase, progressive unsteady gait |
| Immediately after exercise | 97 | 180/95 | 7 | Unsteady gait and NP |
| 2 min after exercise | 84 | 155/85 | 4 | Unsteady gait and NP |
| 4 min after exercise | 72 | 130/80 | 2 | Unsteady gait and NP |
| PFO: what is it? | The patent foramen ovale is a congenital heart defect, consisting of an opening in the inter-atrial septum resulting from incomplete coverage of the ostium secundum, by the septum secundum [65]. |
| Major risk factors linked with PFO-referable strokes | PFO morphology and its size, young age, right-to-left shunt degree, atrial septal aneurysm, intrinsic coagulation–anticoagulation systems imbalance, other atrial abnormalities co-existence (as right atrial septal pouch, Eustachian valve and Chiari’s network) [62]. |
| Clinical conditions frequently related with PFO | Decompression illness in scuba divers, platypnea–orthodeoxia syndrome, cryptogenic stroke, cerebral fat embolism syndrome, [70] obstructive sleep apnea (OSAS), migraine [64] with aura [65,79] and transient global amnesia [83]. |
| PFO diagnostic procedures | The gold standard technique is represented by the contrast transesophageal echocardiography (c-TEE) but contrast transcranial doppler (c-TCD) and contrast-transthoracic echocardiography (c-TTE) are also utilized to establish the hemodynamic relevance and seriousness of the right-to-left shunting through a PFO [64,65,66]. |
| PFO medical management | Trans-catheter closure consists of a percutaneous insertion of a device to close the interatrial septum and has been shown the less invasive surgical technique which allows patients to be discharged from hospital in 24 h [65]. |
| Upper motor neuron examination | The upper motor neuron examination includes assessment of reflexes, strength, coordination, muscle tone, muscle bulk and abnormal movements. Some of these may be detected through careful observation. However, specific maneuvers/tests (i.e., hyperreflexia, ankle clonus, finger rolling, pronator drift, Babinski sign and Hoffman’s reflex) help in the detection of abnormality. Abnormal findings prompt the suspicion of a central motor dysfunction in need urgent referral [15,61]. |
| Cranial nerve examination | Cranial nerve examination is an integral part of a complete neurological examination and consists of assessing a subset of the twelve cranial nerves in order to identify any abnormalities in their function. Subtle or frank cranial nerve palsy may precede more serious pathologies mimicking neck pain or headache in early stages [24,96,97]. |
| Peripheral neurological examination | The peripheral neurological examination consists of the assessment of the sensibility (including pain, temperature and light touch testing), the muscle strengths and deep tendon reflexes. Careful observation and specific tests are used to detect any abnormalities. The main purpose is to localize where in the peripheral nervous system the lesion is [15]. |
| Blood pressure measurement | High systolic and diastolic pressure are the most common risk factors for cardiovascular pathologies and stroke [86]. These parameters are important to evaluate, in both during resting or physical activity, for the patient’ safety—preventing potentially fatal events—and the decision to refer and its urgency [12,14,47,52,58,98,99,100]. |
| Heart rate measurement | Heart rate is an indirect parameter of coronary blood flow, myocardial performance and oxygen demand. It is a vital sign which can be easily monitored [47,101]. |
| Blood pressure measurement during exercise | Together with electrocardiographic and heart rate monitoring, BP measurement during clinical exercise testing (GXT) must be considered in those patients at risk of masked hypertension. Exaggerated increases in systolic BP (SBP) and/or diastolic BP (DBP) during GXT are related with a 36% increased risk of cardiovascular events and mortality, and extended risk of hidden hypertension in normotensive subjects. Thus, exaggerated blood pressure response to exercise (EBPR) is a useful clinical test for diagnostic and prognostic purposes [54]. Both the American College of Sports Medicine (ACSM) and the American Heart Association (AHA) identify an increase of 8–12 mmHg of the SBP or 10 mmHg of the metabolic equivalent (MET; 3.5 mL∙kg−1∙min−1) as ‘‘normal’’ measurement. Additionally, both recommend GXT cessation when an increase of SBP > 210–250 mmHg and DBP > 115 mmHg is observed [102,103,104]. EBPR may help to unmask unmanifested cardiovascular diseases (e.g., coarctation of aorta), enhancing risk stratification and the sensitivity of stress studies (e.g., electrocardiogram), ensuring the early diagnosis and appropriate medical management [54]. The subjects had to continue the test until 90% of their adjusted-age maximal heart rate (HR) was obtained. SBP and DBP has to be recorded just before the exercise and during the last 30–45 s of every 3 min during the exercise [53]. At the end of the test, BP has to be recorded after three minutes of recovery with the patient in a sitting position. That is, based on the BP measurements, the test is evaluated as normal or exaggerated. The test must be stopped only at the occurrence of one of the following: achievement of more than 90% of the age-predicted maximal heart rate, severe fatigue or chest discomfort [54]. |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lopez, G.; Cataldi, F.; Bellin, G.; Dunning, J.; Fernández-de-las-Peñas, C.; Galeno, E.; Meroni, R.; Maselli, F.; Mourad, F. Physiotherapy Screening for Referral of a Patient with Patent Foramen Ovale Presenting with Neck Pain as Primary Complaint: A Case Report. Healthcare 2023, 11, 1165. https://doi.org/10.3390/healthcare11081165
Lopez G, Cataldi F, Bellin G, Dunning J, Fernández-de-las-Peñas C, Galeno E, Meroni R, Maselli F, Mourad F. Physiotherapy Screening for Referral of a Patient with Patent Foramen Ovale Presenting with Neck Pain as Primary Complaint: A Case Report. Healthcare. 2023; 11(8):1165. https://doi.org/10.3390/healthcare11081165
Chicago/Turabian StyleLopez, Giovanni, Fabio Cataldi, Giuseppe Bellin, James Dunning, César Fernández-de-las-Peñas, Erasmo Galeno, Roberto Meroni, Filippo Maselli, and Firas Mourad. 2023. "Physiotherapy Screening for Referral of a Patient with Patent Foramen Ovale Presenting with Neck Pain as Primary Complaint: A Case Report" Healthcare 11, no. 8: 1165. https://doi.org/10.3390/healthcare11081165
APA StyleLopez, G., Cataldi, F., Bellin, G., Dunning, J., Fernández-de-las-Peñas, C., Galeno, E., Meroni, R., Maselli, F., & Mourad, F. (2023). Physiotherapy Screening for Referral of a Patient with Patent Foramen Ovale Presenting with Neck Pain as Primary Complaint: A Case Report. Healthcare, 11(8), 1165. https://doi.org/10.3390/healthcare11081165












