A Review of the Most Recent Clinical and Neuropathological Criteria for Chronic Traumatic Encephalopathy
Abstract
1. Introduction
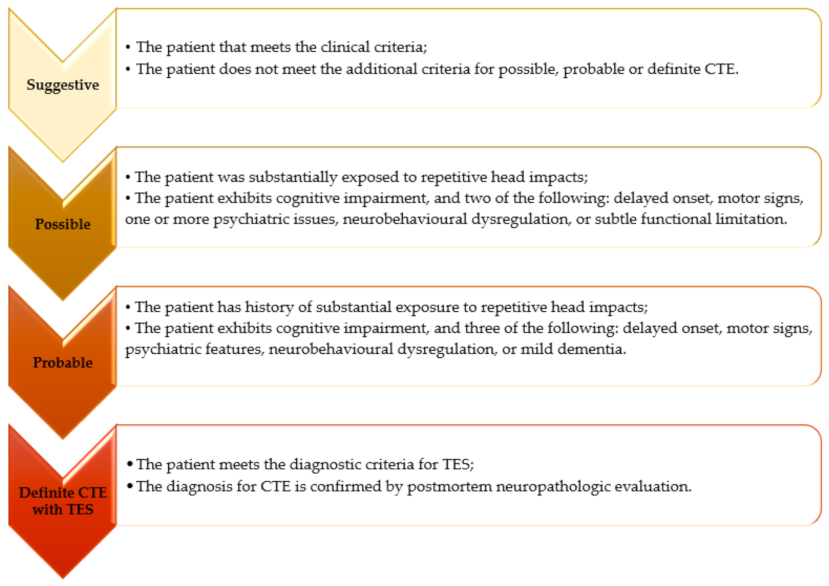
2. Clinical Diagnostic Criteria and Diagnostic Algorithm
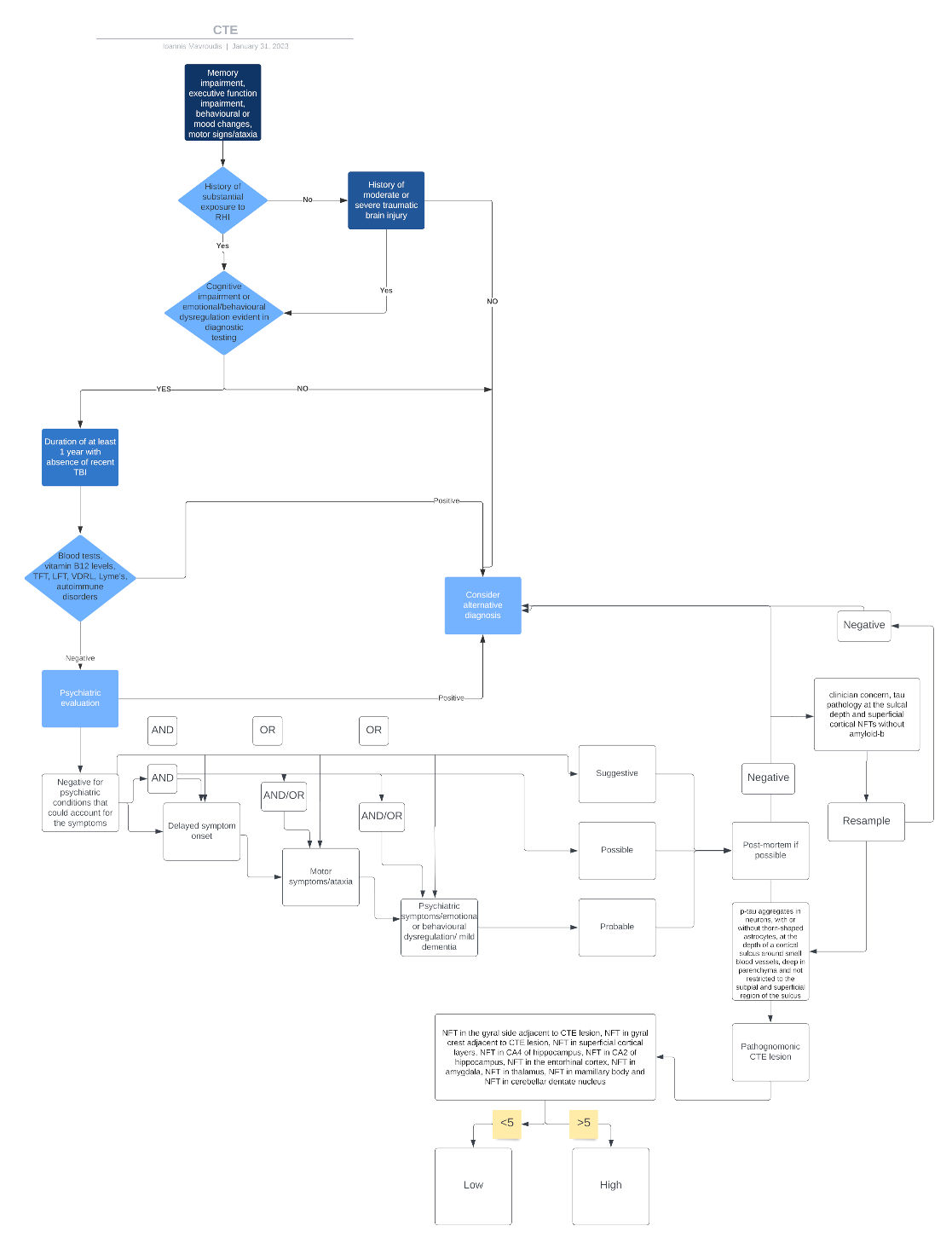
3. Diagnostic Algorithm
3.1. Symptoms Recognition and Evaluation
3.2. Exposure to Head Trauma
- 1.
- At least 5 years of active and organized participation in high-exposure contact sports, such as boxing, wrestling and martial arts, cage fighting, American football and rugby, ice hockey, or soccer.
- 2.
- Vocational activities that predispose a person to repeated head impacts: military service, leading to exposure to blasts and other explosions or multiple blows to the head; policing or other task force work, leading to exposure due to breaching locked doors and other barriers as a first responder; and other vocational activities, which may be exposed to various injuries involving multiple head impacts.
- 3.
- Domestic violence and different types of abuse survivors.
- 4.
3.3. Core Clinical Features
3.4. Symptoms Should Not Be Fully Accounted for by Other Disorders
3.5. Staging and Functionality Levels
- Stage 1, in which the patient does not have any significant changes in their daily activities, job, family, or social roles;
- Stage 2, when the patient exhibits mild functional limitation with slightly reduced performance in job, household, family, social, or community roles;
- Stage 3, which is characterized by mild dementia with definite impairment of daily activities;
- Stage 4, with moderate dementia, where the patient is not independent but can still be taken outside the home for some simple activities;
- Stage 5, with severe dementia, where the patient cannot participate in any activities outside the home.
3.6. Neuropathological Criteria
4. The Role of Biomarkers
4.1. Neuroimaging
4.1.1. Magnetic Resonance Imaging (MRI)
4.1.2. Positron Emission Tomography (PET)
4.1.3. Magnetoencephalography (MEG)
4.2. Fluid Biomarkers
4.3. Proposed Algorithm for TES/CTE Diagnosis
5. Discussion
6. Conclusions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Martland, H. Punch drunk. J. Am. Med. Assoc. 1928, 91, 1103–1107. [Google Scholar] [CrossRef]
- Brandenburg, W.; Hallervorden, J. Dementia pugilistica mit anatomischem Befund. Virchows Archiv. Pathol. Anat. 1954, 325, 680–709. [Google Scholar] [CrossRef]
- Corsellis, J.A.; Bruton, C.J.; Freeman-Browne, D. The aftermath of boxing. Psychol. Med. 1973, 3, 270–303. [Google Scholar] [CrossRef] [PubMed]
- Omalu, B.I.; DeKosky, S.T.; Minster, R.; Kamboh, M.I.; Hamilton, R.L.; Wecht, C.H. Chronic traumatic encephalopathy in a National Football League player. Neurosurgery 2005, 57, 128–134. [Google Scholar] [CrossRef] [PubMed]
- McKee, A.C.; Daneshvar, D.H.; Alvarez, V.E.; Stein, T.D. The neuropathology of sport. Acta Neuropathol. 2014, 127, 29–51. [Google Scholar] [CrossRef]
- Ling, H.; Morris, H.R.; Neal, J.W.; Neal, J.W.; Lees, A.J.; Hardy, J.; Holton, J.L.; Revesz, T.; Williams, D.D. Mixed pathologies including chronic traumatic encephalopathy account for dementia in retired association football (soccer) players. Acta Neuropathol. 2017, 133, 337–352. [Google Scholar] [CrossRef]
- Lee, E.B.; Kinch, K.; Johnson, V.E.; Trojanowski, J.Q.; Smith, D.H.; Stewart, W. Chronic traumatic encephalopathy is a common co-morbidity, but less frequent primary dementia in former soccer and rugby players. Acta Neuropathol. 2019, 138, 389–399. [Google Scholar] [CrossRef]
- Omalu, B.I.; Fitzsimmons, R.P.; Hammers, J.; Bailes, J. Chronic traumatic encephalopathy in a professional American wrestler. J. Forensic Nurs. 2010, 6, 130–136. [Google Scholar] [CrossRef]
- Stewart, W.; McNamara, P.H.; Lawlor, B.; Hutchinson, S.; Farrell, M. Chronic traumatic encephalopathy: A potential late and under recognized consequence of rugby union. QJM 2016, 109, 11–15. [Google Scholar] [CrossRef]
- Buckland, M.E.; Sy, J.; Szentmariay, I.; Kullen, A.; Lee, M.; Harding, A.; Halliday, G.; Suter, C.M. Correction to: Chronic traumatic encephalopathy in two former Australian National Rugby League players. Acta Neuropathol. Commun. 2019, 7, 122. [Google Scholar] [CrossRef]
- Omalu, B.; Hammers, J.L.; Bailes, J.; Hamilton, R.L.; Kamboh, M.I.; Webster, G.; Fitzsimmons, R.P. Chronic traumatic encephalopathy in an Iraqi war veteran with posttraumatic stress disorder who committed suicide. Neurosurg. Focus 2011, 31, E3. [Google Scholar] [CrossRef]
- Priemer, D.S.; Iacono, D.; Rhodes, C.H.; Olsen, C.H.; Perl, D.P. Chronic Traumatic Encephalopathy in the Brains of Military Personnel. N. Engl. J. Med. 2022, 386, 2169–2177. [Google Scholar] [CrossRef] [PubMed]
- McKee, A.C.; Stein, T.D.; Kiernan, P.T.; Alvarez, V.E. The neuropathology of chronic traumatic encephalopathy. Brain Pathol. 2015, 25, 350–364. [Google Scholar] [CrossRef] [PubMed]
- Bieniek, K.F.; Ross, O.A.; Cormier, K.A.; Walton, R.L.; Soto-Ortolaza, A.; Johnston, A.E.; DeSaro, P.; Boylan, K.B.; Graff-Radford, N.R.; Wszolek, Z.K.; et al. Chronic traumatic encephalopathy pathology in a neurodegenerative disorders brain bank. Acta Neuropathol. 2015, 130, 877–889. [Google Scholar] [CrossRef] [PubMed]
- Ruchika, F.; Shah, S.; Neupane, D.; Vijay, R.; Mehkri, Y.; Lucke-Wold, B. Understanding the Molecular Progression of Chronic Traumatic Encephalopathy in Traumatic Brain Injury, Aging and Neurodegenerative Disease. Int. J. Mol. Sci. 2023, 24, 1847. [Google Scholar] [CrossRef]
- Reams, N.; Eckner, J.T.; Almeida, A.A.; Aagesen, A.L.; Giordani, B.; Paulson, H.; Lorincz, M.T.; Kutcher, J.S. A Clinical Approach to the Diagnosis of Traumatic Encephalopathy Syndrome: A Review. JAMA Neurol. 2016, 73, 743–749. [Google Scholar] [CrossRef]
- Mavroudis, I.; Kazis, D.; Chowdhury, R.; Petridis, F.; Costa, V.; Balmus, I.M.; Ciobica, A.; Luca, A.C.; Radu, I.; Dobrin, R.P.; et al. Post-Concussion Syndrome and Chronic Traumatic Encephalopathy: Narrative Review on the Neuropathology, Neuroimaging and Fluid Biomarkers. Diagnostics 2022, 12, 740. [Google Scholar] [CrossRef]
- Victoroff, J. Traumatic encephalopathy: Review and provisional research diagnostic criteria. Neuro Rehabil. 2013, 32, 211–224. [Google Scholar] [CrossRef]
- Montenigro, P.H.; Bernick, C.; Cantu, R.C. Clinical features of repetitive traumatic brain injury and chronic traumatic encephalopathy. Brain Pathol. 2015, 25, 304–317. [Google Scholar] [CrossRef]
- Jordan, B.D. The clinical spectrum of sport-related traumatic brain injury. Nat. Rev. Neurol. 2013, 9, 222–230. [Google Scholar] [CrossRef]
- Katz, D.I.; Bernick, C.; Dodick, D.W.; Mez, J.; Mariani, M.L.; Adler, C.H.; Alosco, M.L.; Balcer, L.J.; Banks, S.J.; Barr, W.B.; et al. National Institute of Neurological Disorders and Stroke Consensus Diagnostic Criteria for Traumatic Encephalopathy Syndrome. Neurology 2021, 96, 848–863. [Google Scholar] [CrossRef] [PubMed]
- Ganau, M.; Lavinio, A.; Prisco, L. Delirium and agitation in traumatic brain injury patients: An update on pathological hypotheses and treatment options. Minerva Anestesiol. 2018, 84, 632–640. [Google Scholar] [CrossRef] [PubMed]
- Asken, B.M.; Sullan, M.J.; DeKosky, S.T.; Jaffee, M.S.; Bauer, R.M. Research Gaps and Controversies in Chronic Traumatic Encephalopathy. JAMA Neurol. 2017, 74, 1255. [Google Scholar] [CrossRef] [PubMed]
- Geddes, J.F.; Vowles, G.H.; Nicoll, J.A.; Révész, T. Neuronal cytoskeletal changes are an early consequence of repetitive head injury. Acta Neuropathol. 1999, 98, 171–178. [Google Scholar] [CrossRef]
- Dogan, M.; Ozdemir, O.; Sal, E.A.; Dogan, S.Z.; Ozdemir, P.; Cesur, Y.; Caksen, H. Psychotic disorder and extrapyramidal symptoms associated with vitamin B12 and folate deficiency. J. Trop. Pediatr. 2009, 55, 205–207. [Google Scholar] [CrossRef] [PubMed]
- Matsuda, H. MRI morphometry in Alzheimer’s disease. Ageing Res. Rev. 2016, 30, 17–24. [Google Scholar] [CrossRef]
- Chow, F. Neurosyphilis. Continuum (Minneap Minn). Neuroinfectious Dis. 2021, 27, 1018–1039. [Google Scholar] [CrossRef]
- Olney, N.T.; Spina, S.; Miller, B.L. Frontotemporal Dementia. Neurol. Clin. 2017, 35, 339–374. [Google Scholar] [CrossRef]
- Fanciulli, A.; Stankovic, I.; Krismer, F.; Seppi, K.; Levin, J.; Wenning, G.K. Multiple system atrophy. Int. Rev. Neurobiol. 2019, 149, 137–192. [Google Scholar] [CrossRef]
- Goldstein, L.E.; Fisher, A.M.; Tagge, C.A.; Zhang, X.-L.; Velisek, L.; Sullivan, J.A.; Upreti, C.; Kracht, J.M.; Ericsson, M.; Wojnarowicz, M.W.; et al. Chronic traumatic encephalopathy in blast-exposed military veterans and a blast neurotrauma mouse model. Sci. Transl. Med. 2012, 4, 134ra60. [Google Scholar]
- Roberts, G.W.; Whitwell, H.L.; Acland, P.R.; Bruton, C.J. Dementia in a punch-drunk wife. Lancet Lond. Engl. 1990, 335, 918–919. [Google Scholar] [CrossRef] [PubMed]
- Bieniek, K.F.; Cairns, N.J.; Crary, J.F.; Dickson, D.W.; Folkerth, R.D.; Keene, C.D.; Litvan, I.; Perl, D.P.; Stein, T.D.; Vonsattel, J.P.; et al. The Second NINDS/NIBIB Consensus Meeting to Define Neuropathological Criteria for the Diagnosis of Chronic Traumatic Encephalopathy. J. Neuropathol. Exp. Neurol. 2021, 80, 210–219. [Google Scholar] [CrossRef] [PubMed]
- Johnson, V.E.; Stewart, W.; Smith, D.H. Widespread tau and amyloid-beta pathology many years after a single traumatic brain injury in humans. Brain Pathol. 2012, 22, 142–149. [Google Scholar] [CrossRef] [PubMed]
- Klein, A.P.; Tetzlaff, J.E.; Bonis, J.M.; Nelson, L.D.; Mayer, A.; Huber, D.L.; Harezlak, J.; Mathews, V.P.; Ulmer, J.L.; Sinson, G.P.; et al. Prevalence of potentially clinically significant MRI findings in athletes with and without sport-related concussion. J. Neurotrauma 2019, 36, 1776–1785. [Google Scholar] [CrossRef] [PubMed]
- Asken, B.M.; DeKosky, S.T.; Clugston, J.R.; Jaffee, M.S.; Bauer, R.M. Diffusion tensor imaging (DTI) findings in adult civilian, military, and sport-related mild traumatic brain injury (mTBI): A systematic critical review. Brain Imaging Behav. 2011, 12, 585–612. [Google Scholar] [CrossRef] [PubMed]
- Lepage, C.; Muehlmann, M.; Tripodis, Y.; Hufschmidt, J.; Stamm, J.; Green, K.; Wrobel, P.; Schultz, V.; Weir, I.; Alosco, M.L.; et al. Limbic system structure volumes and associated neurocognitive functioning in former NFL players. Brain Imaging Behav. 2019, 13, 725–734. [Google Scholar] [CrossRef]
- Champagne, A.A.; Peponoulas, E.; Terem, I.; Ross, A.; Tayebi, M.; Chen, Y.; Coverdale, N.S.; Nielsen, P.; Wang, A.; Shim, V.; et al. Novel strain analysis informs about injury susceptibility of the corpus callosum to repeated impacts. Brain Commun. 2019, 1, fcz021. [Google Scholar] [CrossRef]
- Schultz, V.; Stern, R.A.; Tripodis, Y.; Stamm, J.; Wrobel, P.; Lepage, C.; Weir, I.; Guenette, J.P.; Chua, A.; Alosco, M.L.; et al. Age at first exposure to repetitive head impacts is associated with smaller thalamic volumes in former professional american football players. J. Neurotrauma 2018, 35, 278–285. [Google Scholar] [CrossRef]
- Lee, J.K.; Wu, J.; Banks, S.; Bernick, C.; Massand, M.G.; Modic, M.T.; Ruggieri, P.; Jones, S.E. Prevalence of traumatic findings on routine MRI in a large cohort of professional fighters. AJNR Am. J. Neuroradiol. 2017, 38, 1303–1310. [Google Scholar] [CrossRef]
- Zhang, H.; Schneider, T.; Wheeler-Kingshott, C.A.; Alexander, D.C. NODDI: Practical in vivo neurite orientation dispersion and density imaging of the human brain. Neuroimage 2012, 61, 1000–1016. [Google Scholar] [CrossRef]
- Thompson, P.W.; Ye, L.; Morgenstern, J.L.; Sue, L.; Beach, T.G.; Judd, D.J.; Shipley, N.J.; Libri, V.; Lockhart, A. Interaction of the amyloid imaging tracer FDDNP with hallmark Alzheimer’s disease pathologies. J. Neurochem. 2009, 109, 623–630. [Google Scholar] [CrossRef] [PubMed]
- Leung, K. 2-(4-(2-[(18)F]Fluoroethyl)piperidin-1-yl)benzo[4;5]imidazo[1,2-a]pyrimidine. In Molecular Imaging and Contrast Agent Database (MICAD); National Center for Biotechnology Information: Bethesda, MD, USA, 2004. [Google Scholar]
- Agdeppa, E.D.; Kepe, V.; Liu, J.; Flores-Torres, S.; Satyamurthy, N.; Petric, A.; Cole, G.M.; Small, G.W.; Huang, S.C.; Barrio, J.R. Binding characteristics of radiofluorinated 6-dialkylamino-2-naphthylethylidene derivatives as positron emission tomography imaging probes for beta-amyloid plaques in Alzheimer’s disease. J. Neurosci. 2001, 21, Rc189. [Google Scholar] [CrossRef] [PubMed]
- Harada, R.; Okamura, N.; Furumoto, S.; Tago, T.; Yanai, K.; Arai, H.; Kudo, Y. Characteristics of tau and its ligands in PET Imaging. Biomolecules 2016, 6, 7. [Google Scholar] [CrossRef] [PubMed]
- Peitz, G.W.; Wilde, E.A.; Grandhi, R. Magnetoencephalography in the Detection and Characterization of Brain Abnormalities Associated with Traumatic Brain Injury: A Comprehensive Review. Med. Sci. 2021, 9, 7. [Google Scholar] [CrossRef]
- Stern, R.A.; Tripodis, Y.; Baugh, C.M.; Fritts, N.G.; Martin, B.M.; Chaisson, C.; Cantu, R.C.; Joyce, J.A.; Shah, S.; Ikezu, T.; et al. Preliminary study of plasma exosomal tau as a potential biomarker for chronic traumatic encephalopathy. J. Alzheimers Dis. 2016, 51, 1099–1109. [Google Scholar] [CrossRef]
- Oliver, J.M.; Jones, M.T.; Kirk, K.M.; Gable, D.A.; Repshas, J.T.; Johnson, T.A.; Andréasson, U.; Norgren, N.; Blennow, K.; Zetterberg, H. Serum Neurofilament Light in American Football Athletes over the Course of a Season. J. Neurotrauma 2016, 33, 1784–1789. [Google Scholar] [CrossRef]
- Siman, R.; Toraskar, N.; Dang, A.; McNeil, E.; McGarvey, M.; Plaum, J.; Maloney, E.; Grady, M.S. A panel of neuronenriched proteins as markers for traumatic brain injury in humans. J. Neurotrauma 2009, 26, 1867–1877. [Google Scholar] [CrossRef]
- Shahim, P.; Tegner, Y.; Wilson, D.H.; Randall, J.; Skillbäck, T.; Pazooki, D.; Kallberg, B.; Blennow, K.; Zetterberg, H. Blood biomarkers for brain injury in concussed professional ice hockey players. JAMA Neurol. 2014, 71, 684–692. [Google Scholar] [CrossRef]
- Zetterberg, H.; Tanriverdi, F.; Unluhizarci, K.; Selcuklu, A.; Kelestimur, F.; Blennow, K. Sustained release of neuron-specific enolase to serum in amateur boxers. Brain Inj. 2009, 23, 723–726. [Google Scholar] [CrossRef]
- Stein, D.M.; Lindell, A.; Murdock, K.R.; Kufera, J.A.; Menaker, J.; Keledjian, K.; Bochicchio, G.V.; Aarabi, B.; Scalea, T.M. Relationship of serum and cerebrospinal fluid biomarkers with intracranial hypertension and cerebral hypoperfusion after severe traumatic brain injury. J. Trauma 2011, 70, 1096–1103. [Google Scholar] [CrossRef]
- McMahon, P.J.; Panczykowski, D.M.; Yue, J.K.; Puccio, A.M.; Inoue, T.; Sorani, M.D.; Lingsma, H.F.; Maas, A.I.; Valadka, A.B.; Yuh, E.L.; et al. Measurement of the glial fibrillary acidic protein and its breakdown products GFAP-BDP biomarker for the detection of traumatic brain injury compared to computed tomography and magnetic resonance imaging. J. Neurotrauma 2015, 32, 527–533. [Google Scholar] [CrossRef] [PubMed]
- Edwards, K.A.; Greer, K.; Leete, J.; Lai, C.; Devoto, C.; Qu, B.X.; Yarnell, A.M.; Polejaeva, E.; Dell, K.C.; LoPresti, M.L.; et al. Neuronally-derived tau is increased in experienced breachers and is associated with neurobehavioral symptoms. Sci. Rep. 2021, 11, 19527. [Google Scholar] [CrossRef] [PubMed]
- Gill, J.; Mustapic, M.; Diaz-Arrastia, R.; Lange, R.; Gulyani, S.; Diehl, T.; Motamedi, V.; Osier, N.; Stern, R.A.; Kapogiannis, D. Higher exosomal tau, amyloid-beta 42, and IL-10 are associated with mild TBIs andchronic symptoms in military personnel. Brain Inj. 2018, 32, 1277–1284. [Google Scholar] [CrossRef] [PubMed]
- Olivera, A.; Lejbman, N.; Jeromin, A.; French, L.M.; Kim, H.S.; Cashion, A.; Mysliwiec, V.; Diaz-Arrastia, R.; Gill, J. Peripheral total tau in military personnel who sustain traumatic brain injuries during deployment. JAMA Neurol. 2015, 72, 1109–1116. [Google Scholar] [CrossRef]
- Lewis, J.M.; Dhawan, S.; Obirieze, A.C.; Sarno, B.; Akers, J.; Heller, M.J.; Chen, C.C. Plasma Biomarker for Post-concussive Syndrome: A Pilot Study Using an Alternating Current Electro-Kinetic Platform. Front. Neurol. 2020, 11, 685. [Google Scholar] [CrossRef]
- Di Battista, A.P.; Churchill, N.; Rhind, S.G.; Richards, D.; Hutchison, M.G. Evidence of a distinct peripheral inflammatory profile in sport-related concussion. J. Neuroinflamm. 2019, 16, 17. [Google Scholar] [CrossRef]
- Shahim, P.; Tegner, Y.; Marklund, N.; Höglund, K.; Portelius, E.; Brody, D.L.; Blennow, K.; Zetterberg, H. Astroglial activation and altered amyloid metabolism in human repetitive concussion. Neurology 2017, 88, 1400–1407. [Google Scholar] [CrossRef]
- Blaylock, R.L.; Maroon, J. Immunoexcitotoxicity as a central mechanism in chronic traumatic encephalopathy-A unifying hypothesis. Surg. Neurol. Int. 2011, 2, 107. [Google Scholar] [CrossRef]
- Javidi, E.; Magnus, T. Autoimmunity After Ischemic Stroke and Brain Injury. Front. Immunol. 2019, 10, 686. [Google Scholar] [CrossRef]
| Study | Mandatory Condition | Symptoms | Symptom Duration/ Progression | Comments |
|---|---|---|---|---|
| Jordan [20] | Exposure to repetitive head impact events | Two or more symptoms:
| At least one year, in the absence of new head trauma | Diagnosis can be supported by neuroimaging |
| Montenigro et al. [19] | Exposure to head trauma events | At least one core symptom:
| At least one year; Progressive course, or delayed onset. | Diagnostic criteria should be applied after the exclusion of other conditions that could account for the symptoms. |
Two supportive features:
| ||||
| Reams et al. [16] | Exposure to repetitive head impact events | Cognitive decline, confirmed by neuropsychological testing | Longer than two years; Progressive course, late symptom onset. | Provides classification for TES/CTE according to the predominant symptomatology and based on symptoms progression. |
| NINDS Consensus [21] | Exposure to repetitive head impact events originating from contact sports, military service, or other causes |
| Progressive course | Progressive course of the symptoms not fully accounted for by any other neurologic, psychiatric, or medical conditions. |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mavroudis, I.; Balmus, I.-M.; Ciobica, A.; Luca, A.-C.; Gorgan, D.L.; Dobrin, I.; Gurzu, I.L. A Review of the Most Recent Clinical and Neuropathological Criteria for Chronic Traumatic Encephalopathy. Healthcare 2023, 11, 1689. https://doi.org/10.3390/healthcare11121689
Mavroudis I, Balmus I-M, Ciobica A, Luca A-C, Gorgan DL, Dobrin I, Gurzu IL. A Review of the Most Recent Clinical and Neuropathological Criteria for Chronic Traumatic Encephalopathy. Healthcare. 2023; 11(12):1689. https://doi.org/10.3390/healthcare11121689
Chicago/Turabian StyleMavroudis, Ioannis, Ioana-Miruna Balmus, Alin Ciobica, Alina-Costina Luca, Dragos Lucian Gorgan, Irina Dobrin, and Irina Luciana Gurzu. 2023. "A Review of the Most Recent Clinical and Neuropathological Criteria for Chronic Traumatic Encephalopathy" Healthcare 11, no. 12: 1689. https://doi.org/10.3390/healthcare11121689
APA StyleMavroudis, I., Balmus, I.-M., Ciobica, A., Luca, A.-C., Gorgan, D. L., Dobrin, I., & Gurzu, I. L. (2023). A Review of the Most Recent Clinical and Neuropathological Criteria for Chronic Traumatic Encephalopathy. Healthcare, 11(12), 1689. https://doi.org/10.3390/healthcare11121689







