Relationship between Depression and Cognitive Inhibition in Men with Heroin or Methamphetamine Use Disorder in First-Time Mandatory Detoxification
Abstract
1. Introduction
2. Study 1: The Relationship between Depression and Impulse Inhibition
2.1. Methods
2.1.1. Participants
2.1.2. Study Instrument
- Depression. The Beck Depression Self-Rating Inventory (BDI) was developed by Beck and is the most widely used scale in clinical psychology to assess depressive status in normal adults (18–60 years). This test uses a 13-item version of the BDI (such as “0 = I don’t feel depressed, 1 = I feel depressed or frustrated, 2 = I am depressed all day and can’t get rid of it, 3 = I am very depressed and can’t stand it anymore”), with higher total scale scores representing higher levels of depressive symptoms [29]. The total score of this scale ranges from 0 to 39, where 0−4: no clinically significant depression, 5−7: mild depressive symptoms, 8−15: moderate depressive symptoms, and >15: severe depressive symptoms [29]. In this study, Cronbach’s α coefficient for this scale was 0.818.
- Impulse inhibition. The impulse inhibition dimension of the short version of the self-control scale developed by Tangney et al. [30] was used, and this section contains six items, such as “I often act without thinking about it”. A five-point scale was used, and the higher the score, the better the impulse control. In this study, Cronbach’s α coefficient for this scale was 0.799.
2.1.3. Data Analysis and Common Method Deviation Test
2.2. Results
2.2.1. Results of Descriptive Statistics and Correlation Analysis
2.2.2. Results of Regression Analysis
2.3. Discussion of Study 1
3. Study 2: The Role of Depression in Inhibitory Control
3.1. Methods
3.1.1. Participants
3.1.2. Study Instruments
- Basic information questionnaire. A self-administered survey of demographic variables such as age, age of drug use, number of compulsory detoxifications, and type of drugs was mainly used.
- Depression. The instrument used was consistent with Study 1. The threshold of the BD score ≥ 8 (i.e., moderate to severe depression) was used as an assessment indicator in this study because of the need for treatment assessment for this level of depression [34]. In this study, Cronbach’s α coefficient for this scale was 0.819.
- Stroop color–word interference task. The Stroop color–word interference task is a test of selective attention and cognitive flexibility. It has been used in previous studies to measure individuals’ inhibitory control [35]. In the classic Stroop color–word interference task, the experimenter presents subjects with a word written in a different color and asks them to say the color of each word as quickly and as correctly as possible, regardless of the word and the meaning it represents. Three experimental conditions were included in this study: color–word agreement, color–word interference, and a control condition. In the color–word agreement condition, subjects were presented with color words written in different colors, and the name of each color word represented the same meaning as the color of the word, e.g., the Chinese word "green" was written with green paint. In the control condition, subjects were presented with neutral words in different colors. The screen background of the stimulus presentation was black. The cognitive mechanism involved in this task is called selective or directed attention because the subject must manage their attention, resist the interference of irrelevant stimuli, and inhibit or stop one response to say or do something else. The Stroop effect is usually represented by calculating the difference between the behavioral data of incongruent and congruent stimuli [36]. Color–word congruence is automatic processing, and color–word incongruence is conscious control processing.
3.1.3. Experimental Design and Procedure
3.1.4. Data Analysis
3.2. Results
3.2.1. Demographic information of participants
3.2.2. Stroop Effect Validation
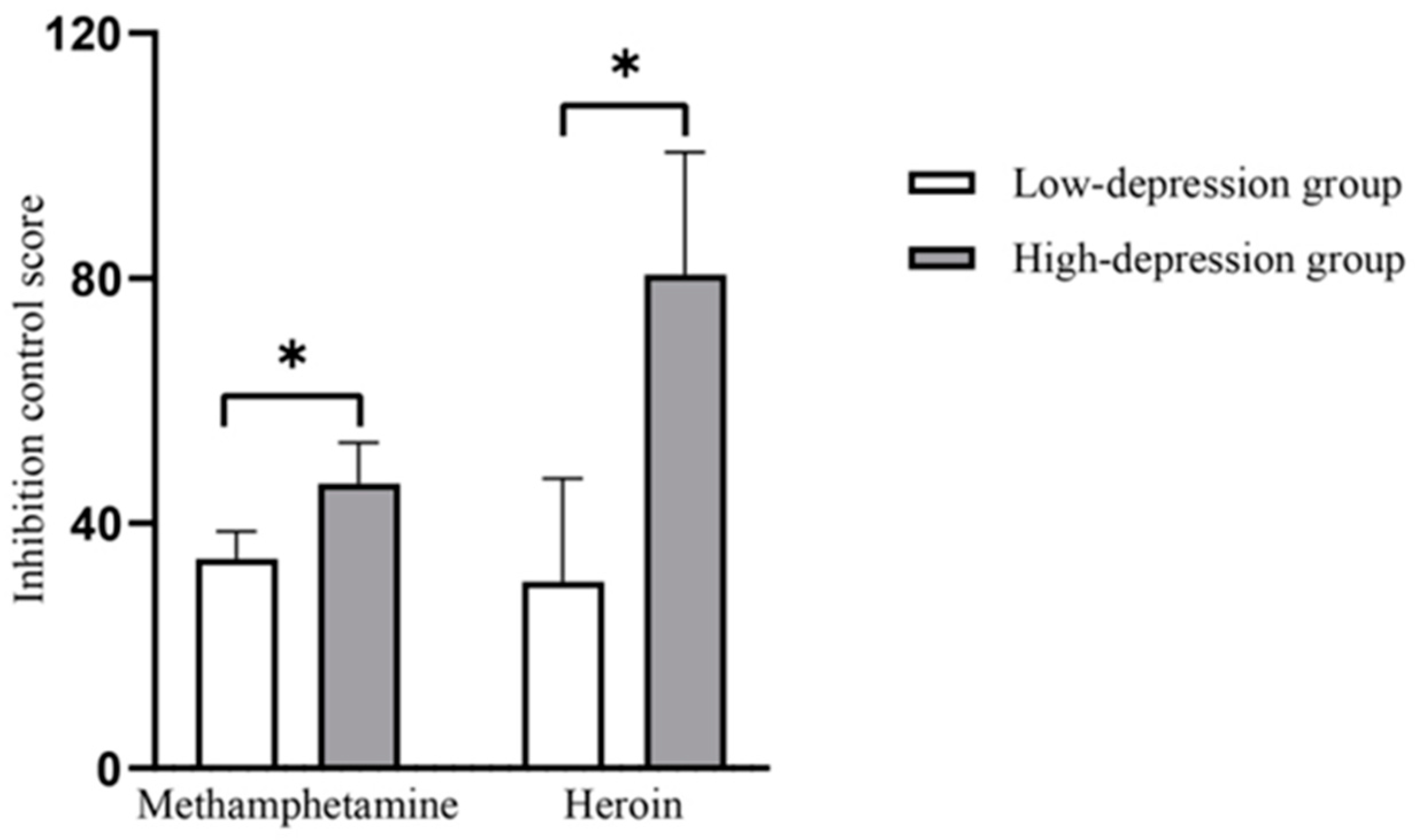
3.2.3. ANOVA Results for Inhibitory Control Scores
3.3. Discussion of Study 2
4. General Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- World Drug Report 2022. Available online: https://www.unodc.org/unodc/data-and-analysis/world-drug-report-2022.html (accessed on 15 December 2022).
- Koob, G.F.; Volkow, N.D. Neurocircuitry of Addiction. Neuropsychopharmacology 2010, 35, 217–238. [Google Scholar] [CrossRef] [PubMed]
- Miyake, A.; Friedman, N.P.; Emerson, M.J.; Witzki, A.H.; Howerter, A.; Wager, T.D. The Unity and Diversity of Executive Functions and Their Contributions to Complex “Frontal Lobe” Tasks: A Latent Variable Analysis. Cogn. Psychol. 2000, 41, 49–100. [Google Scholar] [CrossRef] [PubMed]
- Getzmann, S.; Wascher, E.; Schneider, D. The role of inhibition for working memory processes: ERP evidence from a short-term storage task. Psychophysiology 2018, 55, e13026. [Google Scholar] [CrossRef] [PubMed]
- Wang, D.; Zhu, T.; Zhou, C.; Chang, Y.-K. Aerobic exercise training ameliorates craving and inhibitory control in methamphetamine dependencies: A randomized controlled trial and event-related potential study. Psychol. Sport Exerc. 2017, 30, 82–90. [Google Scholar] [CrossRef]
- Lindgren, K.P.; Hendershot, C.S.; Ramirez, J.J.; Bernat, E.; Rangel-Gomez, M.; Peterson, K.P.; Murphy, J.G. A dual process perspective on advances in cognitive science and alcohol use disorder. Clin. Psychol. Rev. 2019, 69, 83–96. [Google Scholar] [CrossRef] [PubMed]
- Failing, M.; Theeuwes, J. Selection history: How reward modulates selectivity of visual attention. Psychon. Bull. Rev. 2018, 25, 514–538. [Google Scholar] [CrossRef]
- Hitch, G.J.; Allen, R.J.; Baddeley, A.D. Attention and binding in visual working memory: Two forms of attention and two kinds of buffer storage. Atten. Percept. Psychophys. 2020, 82, 280–293. [Google Scholar] [CrossRef]
- Stacy, A.W.; Wiers, R.W. Implicit cognition and addiction: A tool for explaining paradoxical behavior. Annu. Rev. Clin. Psychol. 2010, 6, 551–575. [Google Scholar] [CrossRef]
- Rømer Thomsen, K.; Blom Osterland, T.; Hesse, M.; Feldstein Ewing, S.W. The intersection between response inhibition and substance use among adolescents. Addict. Behav. 2018, 78, 228–230. [Google Scholar] [CrossRef]
- Brooks, S.J.; Wiemerslage, L.; Burch, K.H.; Maiorana, S.A.; Cocolas, E.; Schiöth, H.B.; Kamaloodien, K.; Stein, D.J. The impact of cognitive training in substance use disorder: The effect of working memory training on impulse control in methamphetamine users. Psychopharmacology 2017, 234, 1911–1921. [Google Scholar] [CrossRef]
- Ersche, K.D.; Williams, G.B.; Robbins, T.W.; Bullmore, E.T. Meta-analysis of structural brain abnormalities associated with stimulant drug dependence and neuroimaging of addiction vulnerability and resilience. Curr. Opin. Neurobiol. 2013, 23, 615–624. [Google Scholar] [CrossRef] [PubMed]
- Feil, J.; Sheppard, D.; Fitzgerald, P.B.; Yücel, M.; Lubman, D.I.; Bradshaw, J.L. Addiction, compulsive drug seeking, and the role of frontostriatal mechanisms in regulating inhibitory control. Neurosci Biobehav Rev 2010, 35, 248–275. [Google Scholar] [CrossRef] [PubMed]
- Mizoguchi, H.; Yamada, K. Methamphetamine use causes cognitive impairment and altered decision-making. Neurochem. Int. 2019, 124, 106–113. [Google Scholar] [CrossRef] [PubMed]
- Farhadian, M.; Akbarfahimi, M.; Hassani Abharian, P.; Hosseini, S.G.; Shokri, S. Assessment of Executive Functions in Methamphetamine-addicted Individuals: Emphasis on Duration of Addiction and Abstinence. Basic Clin. Neurosci. 2017, 8, 147–153. [Google Scholar] [CrossRef]
- Volkow, N.D.; Koob, G.F.; McLellan, A.T. Neurobiologic Advances from the Brain Disease Model of Addiction. N. Engl. J. Med. 2016, 374, 363–371. [Google Scholar] [CrossRef]
- Koob, G.F.; Le Moal, M. Drug Addiction, Dysregulation of Reward, and Allostasis. Neuropsychopharmacology 2001, 24, 97–129. [Google Scholar] [CrossRef]
- Courtney, K.E.; Ray, L.A. Methamphetamine: An update on epidemiology, pharmacology, clinical phenomenology, and treatment literature. Drug Alcohol. Depend 2014, 143, 11–21. [Google Scholar] [CrossRef]
- Potvin, S.; Pelletier, J.; Grot, S.; Hébert, C.; Barr, A.M.; Lecomte, T. Cognitive deficits in individuals with methamphetamine use disorder: A meta-analysis. Addict. Behav. 2018, 80, 154–160. [Google Scholar] [CrossRef]
- Al-Zahrani, M.A.; Elsayed, Y.A. The impacts of substance abuse and dependence on neuropsychological functions in a sample of patients from Saudi Arabia. Behav. Brain Funct. 2009, 5, 48. [Google Scholar] [CrossRef]
- Ouzir, M.; Errami, M. Etiological theories of addiction: A comprehensive update on neurobiological, genetic and behavioural vulnerability. Pharmacol. Biochem. Behav. 2016, 148, 59–68. [Google Scholar] [CrossRef]
- Dierker, L.; Selya, A.; Lanza, S.; Li, R.; Rose, J. Depression and marijuana use disorder symptoms among current marijuana users. Addict. Behav. 2018, 76, 161–168. [Google Scholar] [CrossRef] [PubMed]
- Giannouli, V.; Stamovlasis, D.; Tsolaki, M. Longitudinal Study of Depression on Amnestic Mild Cognitive Impairment and Financial Capacity. Clin. Gerontol. 2022, 45, 708–714. [Google Scholar] [CrossRef] [PubMed]
- Torres, I.J.; Qian, H.; Basivireddy, J.; Chakrabarty, T.; Wong, H.; Lam, R.W.; Yatham, L.N. Three-year longitudinal cognitive functioning in patients recently diagnosed with bipolar disorder. Acta Psychiatr. Scand. 2020, 141, 98–109. [Google Scholar] [CrossRef] [PubMed]
- LeMoult, J.; Gotlib, I.H. Depression: A cognitive perspective. Clin. Psychol. Rev. 2019, 69, 51–66. [Google Scholar] [CrossRef] [PubMed]
- Beevers, C.G. Cognitive vulnerability to depression: A dual process model. Clin. Psychol. Rev. 2005, 25, 975–1002. [Google Scholar] [CrossRef]
- Zhao, C.; Liu, Z.; Zhao, D.; Liu, Y.; Liang, J.; Tang, Y.; Liu, Z.; Zheng, J. Drug abuse in China. Ann. N. Y. Acad. Sci. 2004, 1025, 439–445. [Google Scholar] [CrossRef]
- Liu, Y.; Li, M.; Fan, M.; Song, Y.; Yu, H.; Zhi, X.; Xiao, K.; Lai, S.; Zhang, J.; Jin, X.; et al. Chromodomain Y-like Protein–Mediated Histone Crotonylation Regulates Stress-Induced Depressive Behaviors. Biol. Psychiatry 2019, 85, 635–649. [Google Scholar] [CrossRef]
- Beck, A.T.; Steer, R.A. Internal consistencies of the original and revised beck depression inventory. J. Clin. Psychol. 1984, 40, 1365–1367. [Google Scholar] [CrossRef]
- Tangney, J.P.; Baumeister, R.F.; Boone, A.L. High Self-Control Predicts Good Adjustment, Less Pathology, Better Grades, and Interpersonal Success. J. Pers. 2004, 72, 271–324. [Google Scholar] [CrossRef]
- Malhotra, N.K.; Kim, S.S.; Patil, A. Common Method Variance in IS Research: A Comparison of Alternative Approaches and a Reanalysis of Past Research. Manag. Sci. 2006, 52, 1865–1883. [Google Scholar] [CrossRef]
- Sun, Q.; Li, G.; Li, N.; Wang, C.; Wang, X. Analysis of the health status of drug detainees in compulsory isolation and rehabilitation. China Drug Dependy. 2009, 18, 318–321. [Google Scholar] [CrossRef]
- Zhang, X.; Xu, H.; Gu, J.; Lau, J.T.F.; Hao, C.; Zhao, Y.; Davis, A.; Hao, Y. Depression, suicidal ideation, and related factors of methadone maintenance treatment users in Guangzhou, China. AIDS Care 2016, 28, 851–856. [Google Scholar] [CrossRef] [PubMed]
- Hernaez, R.; Kramer, J.R.; Khan, A.; Phillips, J.; McCallister, K.; Chaffin, K.; Hernandez, A.P.; Fullington, H.; Ortiz, C.; Blackwell, J.-M.; et al. Depression and Anxiety Are Common Among Patients With Cirrhosis. Clin. Gastroenterol. Hepatol. 2022, 20, 194–203.e191. [Google Scholar] [CrossRef] [PubMed]
- Diamond, A. Executive functions. Annu. Rev. Psychol. 2013, 64, 135–168. [Google Scholar] [CrossRef] [PubMed]
- Stroop, J.R. Studies of interference in serial verbal reactions. J. Exp. Psychol. Anim. Learn Cogn. 1935, 18, 643–662. [Google Scholar] [CrossRef]
- Colich, N.L.; Foland-Ross, L.C.; Eggleston, C.; Singh, M.K.; Gotlib, I.H. Neural Aspects of Inhibition Following Emotional Primes in Depressed Adolescents. J. Clin. Child Adolesc. Psychol. 2016, 45, 21–30. [Google Scholar] [CrossRef]
- Beck, A.T.; Bredemeier, K. A Unified Model of Depression: Integrating Clinical, Cognitive, Biological, and Evolutionary Perspectives. Clin. Psychol. Sci. 2016, 4, 596–619. [Google Scholar] [CrossRef]
- Humphrey, C.; Bucci, S.; Varese, F.; Degnan, A.; Berry, K. Paranoia and negative schema about the self and others: A systematic review and meta-analysis. Clin. Psychol. Rev. 2021, 90, 102081. [Google Scholar] [CrossRef]
- Epp, A.M.; Dobson, K.S.; Dozois, D.J.A.; Frewen, P.A. A systematic meta-analysis of the Stroop task in depression. Clin. Psychol. Rev. 2012, 32, 316–328. [Google Scholar] [CrossRef]
- Joyal, M.; Wensing, T.; Levasseur-Moreau, J.; Leblond, J.T.; Sack, A.; Fecteau, S. Characterizing emotional Stroop interference in posttraumatic stress disorder, major depression and anxiety disorders: A systematic review and meta-analysis. PLoS ONE 2019, 14, e0214998. [Google Scholar] [CrossRef]
- Hofmann, W.; Friese, M.; Strack, F. Impulse and Self-Control From a Dual-Systems Perspective. Perspect. Psychol. Sci. 2009, 4, 162–176. [Google Scholar] [CrossRef] [PubMed]
- Wolff, W.; Martarelli, C.S. Bored Into Depletion? Toward a Tentative Integration of Perceived Self-Control Exertion and Boredom as Guiding Signals for Goal-Directed Behavior. Perspect. Psychol. Sci. 2020, 15, 1272–1283. [Google Scholar] [CrossRef] [PubMed]
- Inzlicht, M.; Schmeichel, B.J.; Macrae, C.N. Why self-control seems (but may not be) limited. Trends Cogn. Sci. 2014, 18, 127–133. [Google Scholar] [CrossRef] [PubMed]
- Evans, J.S.B.T. Dual-processing accounts of reasoning, judgment, and social cognition. Annu. Rev. Psychol. 2008, 59, 255–278. [Google Scholar] [CrossRef] [PubMed]
- Sun, Y.; Zhang, Y. A review of theories and models applied in studies of social media addiction and implications for future research. Addict. Behav. 2021, 114, 106699. [Google Scholar] [CrossRef]
- May, A.C.; Aupperle, R.L.; Stewart, J.L. Dark Times: The Role of Negative Reinforcement in Methamphetamine Addiction. Front. Psychiatry 2020, 11, 114. [Google Scholar] [CrossRef]
- Morgado, P.; Sousa, N.; Cerqueira, J.J. The impact of stress in decision making in the context of uncertainty. J. Neurosci. Res. 2015, 93, 839–847. [Google Scholar] [CrossRef]
- Ramey, T.; Regier, P.S. Cognitive impairment in substance use disorders. CNS Spectr. 2019, 24, 102–113. [Google Scholar] [CrossRef]
- Dawe, S.; Gullo, M.J.; Loxton, N.J. Reward drive and rash impulsiveness as dimensions of impulsivity: Implications for substance misuse. Addict. Behav. 2004, 29, 1389–1405. [Google Scholar] [CrossRef]
- Loughead, J.; Falcone, M.; Wileyto, E.P.; Albelda, B.; Audrain-McGovern, J.; Cao, W.; Kurtz, M.M.; Gur, R.C.; Lerman, C. Can brain games help smokers quit?: Results of a randomized clinical trial. Drug Alcohol. Depend 2016, 168, 112–118. [Google Scholar] [CrossRef]
- Sweeney, M.M.; Rass, O.; DiClemente, C.; Schacht, R.L.; Vo, H.T.; Fishman, M.J.; Leoutsakos, J.-M.S.; Mintzer, M.Z.; Johnson, M.W. Working Memory Training for Adolescents with Cannabis Use Disorders: A Randomized Controlled Trial. J. Child Adolesc. Subst. Abuse 2018, 27, 211–226. [Google Scholar] [CrossRef] [PubMed]
- Sanchis-Segura, C.; Becker, J.B. Why we should consider sex (and study sex differences) in addiction research. Addict. Biol. 2016, 21, 995–1006. [Google Scholar] [CrossRef] [PubMed]
- Pettinati, H.M.; O’Brien, C.P.; Dundon, W.D. Current Status of Co-Occurring Mood and Substance Use Disorders: A New Therapeutic Target. Am. J. Psychiatry 2013, 170, 23–30. [Google Scholar] [CrossRef] [PubMed]
| Variable | 1 | 2 | 3 | 4 |
|---|---|---|---|---|
| 1. Age | 1 | |||
| 2. FDG | 0.687 *** | 1 | ||
| 3. Depression | −0.005 | −0.023 | 1 | |
| 4. SII | 0.124 *** | 0.117 *** | −0.218 *** | 1 |
| M | 34.47 | 24.33 | 7.05 | 3.204 |
| SD | 7.360 | 6.946 | 5.240 | 0.750 |
| Predictor Variable | Outcome Variable: SII | ||||
|---|---|---|---|---|---|
| b | SE | t | p | 95% CI | |
| Constant | 3.008 | 0.151 | 19.975 | <0.001 | (2.712, 3.303) |
| Age | 0.008 | 0.006 | 1.434 | 0.152 | (−0.003, 0.019) |
| FDG | 0.006 | 0.006 | 0.942 | 0.346 | (−0.006, 0.017) |
| Depression | −0.031 | 0.006 | −5.281 | <0.001 | (−0.043, −0.020) |
| Joint explanatory power | R2 = 0.062*** | ||||
| Overall Significance | F(3,567) = 12.462*** | ||||
| Type of First-Time Drug Use | Type of Depression | n (%) | Age | FDG | ||
|---|---|---|---|---|---|---|
| M | SD | M | SD | |||
| Methamphetamine | Low depression | 223 (59.63) | 33.18 | 6.897 | 24.03 | 7.092 |
| High depression | 121 (32.35) | 34.27 | 6.613 | 24.74 | 6.621 | |
| Heroin | Low depression | 17 (4.54) | 42.94 | 7.949 | 23.35 | 5.314 |
| High depression | 13 (3.48) | 45.69 | 8.087 | 23.23 | 5.732 | |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Xin, Y.; Zhong, X.; Zhang, X.; Chen, Y.; Xin, W.; Liu, C.; Fu, H.; Feng, C.; Zuo, G. Relationship between Depression and Cognitive Inhibition in Men with Heroin or Methamphetamine Use Disorder in First-Time Mandatory Detoxification. Healthcare 2023, 11, 70. https://doi.org/10.3390/healthcare11010070
Xin Y, Zhong X, Zhang X, Chen Y, Xin W, Liu C, Fu H, Feng C, Zuo G. Relationship between Depression and Cognitive Inhibition in Men with Heroin or Methamphetamine Use Disorder in First-Time Mandatory Detoxification. Healthcare. 2023; 11(1):70. https://doi.org/10.3390/healthcare11010070
Chicago/Turabian StyleXin, Yong, Xiao Zhong, Xiaoqian Zhang, Youping Chen, Wei Xin, Chuanjun Liu, Haojie Fu, Chun Feng, and Guoguo Zuo. 2023. "Relationship between Depression and Cognitive Inhibition in Men with Heroin or Methamphetamine Use Disorder in First-Time Mandatory Detoxification" Healthcare 11, no. 1: 70. https://doi.org/10.3390/healthcare11010070
APA StyleXin, Y., Zhong, X., Zhang, X., Chen, Y., Xin, W., Liu, C., Fu, H., Feng, C., & Zuo, G. (2023). Relationship between Depression and Cognitive Inhibition in Men with Heroin or Methamphetamine Use Disorder in First-Time Mandatory Detoxification. Healthcare, 11(1), 70. https://doi.org/10.3390/healthcare11010070






