Positive Association between Peri-Surgical Opioid Exposure and Post-Discharge Opioid-Related Outcomes
Abstract
1. Introduction
2. Methods
2.1. Study Design and Patient Population
2.2. Outcomes
2.3. Statistical Analysis
3. Results
3.1. Baseline Patient Demographics and Clinical Characteristics
3.2. Persistent Opioid Use
3.3. ORAEs and HCRU
3.4. Multi-Test Effect on p-Values
4. Discussion
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Gerbershagen, H.J.; Aduckathil, S.; van Wijck, A.J.; Peelen, L.M.; Kalkman, C.J.; Meissner, W. Pain intensity on the first day after surgery: A prospective cohort study comparing 179 surgical procedures. Anesthesiology 2013, 118, 934–944. [Google Scholar] [CrossRef] [PubMed]
- Borges, N.C.; Pereira, L.V.; de Moura, L.A.; Silva, T.C.; Pedroso, C.F. Predictors for Moderate to Severe Acute Postoperative Pain after Cesarean Section. Pain Res. Manag. 2016, 2016, 5783817. [Google Scholar] [CrossRef] [PubMed]
- Azam, M.D.Q.; Sadat-Ali, M.; Badar, A. Pain management in knee arthroplasty: An overview. Curr. Orthop. Pract. 2016, 27, 360–370. [Google Scholar] [CrossRef]
- Choi, J.B.; Kang, K.; Song, M.K.; Seok, S.; Kim, Y.H.; Kim, J.E. Pain Characteristics after Total Laparoscopic Hysterectomy. Int. J. Med. Sci. 2016, 13, 562–568. [Google Scholar] [CrossRef] [PubMed]
- Lirk, P.; Thiry, J.; Bonnet, M.-P.; Joshi, G.P.; Bonnet, F. Pain management after laparoscopic hysterectomy: Systematic review of literature and PROSPECT recommendations. Reg. Anesth. Pain Med. 2019, 44, 425–436. [Google Scholar] [CrossRef]
- Rechberger, T.; Mack, R.J.; McCallum, S.W.; Du, W.; Freyer, A. Analgesic Efficacy and Safety of Intravenous Meloxicam in Subjects with Moderate-to-Severe Pain After Open Abdominal Hysterectomy: A Phase 2 Randomized Clinical Trial. Anesth. Analg. 2019, 128, 1309–1318. [Google Scholar] [CrossRef]
- Singh, J.A.; Lewallen, D. Predictors of pain and use of pain medications following primary Total Hip Arthroplasty (THA): 5707 THAs at 2-years and 3289 THAs at 5-years. BMC Musculoskelet. Disord. 2010, 11, 90. [Google Scholar] [CrossRef]
- Lespasio, M.J.; Guarino, A.; Sodhi, N.; Mont, A.M. Pain Management Associated with Total Joint Arthroplasty: A Primer. Perm J. 2019, 23, 18–169. [Google Scholar] [CrossRef]
- Strassels, S.A.; McNicol, E.; Suleman, R. Postoperative pain management: A practical review, part 2. Am. J. Health Syst. Pharm. 2005, 62, 2019–2025. [Google Scholar] [CrossRef]
- Brummett, C.M.; Waljee, J.F.; Goesling, J.; Moser, S.; Lin, P.; Englesbe, M.J.; Bohnert, A.S.B.; Kheterpal, S.; Nallamothu, B.K. New Persistent Opioid Use after Minor and Major Surgical Procedures in US Adults. JAMA Surg. 2017, 152, e170504. [Google Scholar] [CrossRef]
- Neuman, M.D.; Bateman, B.T.; Wunsch, H. Inappropriate opioid prescription after surgery. Lancet 2019, 393, 1547–1557. [Google Scholar] [CrossRef]
- Shafi, S.; Collinsworth, A.W.; Copeland, L.A.; Ogola, G.O.; Qiu, T.; Kouznetsova, M.; Liao, I.-C.; Mears, N.; Pham, A.T.; Wan, G.J.; et al. Association of Opioid-Related Adverse Drug Events with Clinical and Cost Outcomes Among Surgical Patients in a Large Integrated Health Care Delivery System. JAMA Surg. 2018, 153, 757–763. [Google Scholar] [CrossRef] [PubMed]
- Brat, G.A.; Agniel, D.; Beam, A.; Yorkgitis, B.; Bicket, M.; Homer, M.; Fox, K.P.; Knecht, D.B.; McMahill-Walraven, C.N.; Palmer, N.; et al. Postsurgical prescriptions for opioid naive patients and association with overdose and misuse: Retrospective cohort study. BMJ 2018, 360, j5790. [Google Scholar] [CrossRef] [PubMed]
- Chou, R.; Gordon, D.B.; de Leon-Casasola, O.A.; Rosenberg, J.M.; Bickler, S.; Brennan, T.; Carter, T.; Cassidy, C.L.; Chittenden, E.H.; Degenhardt, E.; et al. Management of Postoperative Pain: A Clinical Practice Guideline from the American Pain Society, the American Society of Regional Anesthesia and Pain Medicine, and the American Society of Anesthesiologists’ Committee on Regional Anesthesia, Executive Committee, and Administrative Council. J. Pain 2016, 17, 131–157. [Google Scholar] [PubMed]
- Walid, M.S.; Hyer, L.; Ajjan, M.; Barth, A.C.M.; Robinson, J.J.S. Prevalence of opioid dependence in spine surgery patients and correlation with length of stay. J. Opioid Manag. 2007, 3, 127–132. [Google Scholar] [CrossRef]
- Dunn, L.K.; Yerra, S.; Fang, S.; Hanak, M.F.; Leibowitz, M.K.; Tsang, S.; Durieux, M.E.; Nemergut, E.C.; Naik, B.I. Incidence and Risk Factors for Chronic Postoperative Opioid Use After Major Spine Surgery: A Cross-Sectional Study with Longitudinal Outcome. Anesth. Analg. 2018, 127, 247–254. [Google Scholar] [CrossRef]
- Young, J.C.; Wu, J.M.; Willis-Gray, M.; Pate, V.; Funk, M.J. Persistent Opioid Use after Hysterectomy in the United States, 2005–2015. Obstet. Gynecol. 2020, 135, 123–132. [Google Scholar] [CrossRef]
- Peahl, A.F.; Dalton, V.K.; Montgomery, J.R.; Lai, Y.-L.; Hu, H.M.; Waljee, J.F. Rates of New Persistent Opioid Use after Vaginal or Cesarean Birth among US Women. JAMA Netw. Open 2019, 2, e197863. [Google Scholar] [CrossRef]
- Bateman, B.T.; Cole, N.M.; Maeda, A.; Burns, S.M.; Houle, T.T.; Huybrechts, K.F.; Clancy, C.R.; Hopp, S.B.; Ecker, J.L.; Ende, H.; et al. Patterns of Opioid Prescription and Use after Cesarean Delivery. Obstet. Gynecol. 2017, 130, 29–35. [Google Scholar] [CrossRef]
- Stark, N.; Kerr, S.; Stevens, J. Prevalence and predictors of persistent post-surgical opioid use: A prospective observational cohort study. Anaesth Intensive Care 2017, 45, 700–706. [Google Scholar] [CrossRef]
- Lee, S.W.; Shen, J.; Kim, S.J.; Chun, S.-Y.; Kim, P.; Riaz, J.; Yoo, J.W.; Hwang, J. US Trends of Opioid-use Disorders and Associated Factors Among Hospitalized Patients with Spinal Conditions and Treatment from 2005 to 2014. Spine 2020, 45, 124–133. [Google Scholar] [CrossRef]
- Cook, D.J.; Kaskovich, S.W.; Pirkle, S.C.; Mica, M.A.C.; Shi, L.L.; Lee, M.J. Benchmarks of Duration and Magnitude of Opioid Consumption After Total Hip and Knee Arthroplasty: A Database Analysis of 69,368 Patients. J. Arthroplast. 2019, 34, 638–644.e1. [Google Scholar] [CrossRef] [PubMed]
- Badreldin, N.; Grobman, W.A.; Chang, K.T.; Yee, L.M. Opioid prescribing patterns among postpartum women. Am. J. Obstet. Gynecol. 2018, 219, 103.e1–103.e8. [Google Scholar] [CrossRef] [PubMed]
- Weston, E.; Raker, C.; Huang, D.; Parker, A.; Cohen, M.; Robison, K.; Mathews, C. Opioid use after minimally invasive hysterectomy in gynecologic oncology patients. Gynecol. Oncol. 2019, 155, 119–125. [Google Scholar] [CrossRef] [PubMed]
- Osmundson, S.S.; Schornack, L.A.; Grasch, J.L.; Zuckerwise, L.C.; Young, J.L.; Richardson, M. Postdischarge Opioid Use after Cesarean Delivery. Obstet. Gynecol. 2017, 130, 36–41. [Google Scholar] [CrossRef] [PubMed]
- Hah, J.M.; Bateman, B.T.; Ratliff, J.; Curtin, C.; Sun, E. Chronic Opioid Use after Surgery: Implications for Perioperative Management in the Face of the Opioid Epidemic. Anesth. Analg. 2017, 125, 1733–1740. [Google Scholar] [CrossRef] [PubMed]
- Yerneni, K.; Nichols, N.; Abecassis, Z.A.; Karras, C.L.; Tan, L.A. Preoperative Opioid Use and Clinical Outcomes in Spine Surgery: A Systematic Review. Neurosurgery 2020, 86, E490–E507. [Google Scholar] [CrossRef]
- Goesling, J.; Moser, S.E.; Zaidi, B.; Hassett, A.L.; Hilliard, P.; Hallstrom, B.; Clauw, D.J.; Brummett, C. Trends and predictors of opioid use after total knee and total hip arthroplasty. Pain 2016, 157, 1259–1265. [Google Scholar] [CrossRef]
- Quan, H.; Sundararajan, V.; Halfon, P.; Fong, A.; Burnand, B.; Luthi, J.-C.; Saunders, L.D.; Beck, C.A.; Feasby, T.E.; Ghali, W.A. Coding algorithms for defining comorbidities in ICD-9-CM and ICD-10 administrative data. Med. Care 2005, 43, 1130–1139. [Google Scholar] [CrossRef]
- Kalakoti, P.; Hendrickson, N.R.; Bedard, N.A.; Pugely, A.J. Opioid Utilization Following Lumbar Arthrodesis: Trends and Factors Associated with Long-term Use. Spine 2018, 43, 1208–1216. [Google Scholar] [CrossRef]
- Armaghani, S.J.; Lee, D.S.; Bible, J.E.; Archer, K.R.; Shau, D.N.; Kay, H.; Zhang, C.; McGirt, M.J.; Devin, C.J. Preoperative opioid use and its association with perioperative opioid demand and postoperative opioid independence in patients undergoing spine surgery. Spine 2014, 39, E1524–E1530. [Google Scholar] [CrossRef] [PubMed]
- Kim, K.Y.; Anoushiravani, A.A.; Chen, K.K.; Roof, M.; Long, W.J.; Schwarzkopf, R. Preoperative Chronic Opioid Users in Total Knee Arthroplasty-Which Patients Persistently Abuse Opiates Following Surgery? J. Arthroplast. 2018, 33, 107–112. [Google Scholar] [CrossRef] [PubMed]
- Barnabe, C.; Bessette, L.; Flanagan, C.; Leclercq, S.; Steiman, A.; Kalache, F.; Kung, T.; Pope, J.E.; Haraoui, B.; Hochman, J.; et al. Sex differences in pain scores and localization in inflammatory arthritis: A systematic review and metaanalysis. J. Rheumatol. 2012, 39, 1221–1230. [Google Scholar] [CrossRef] [PubMed]
- Keefe, F.J.; Lefebvre, J.C.; Egert, J.R.; Affleck, G.; Sullivan, M.J.; Caldwell, D.S. The relationship of gender to pain, pain behavior, and disability in osteoarthritis patients: The role of catastrophizing. Pain 2000, 87, 325–334. [Google Scholar] [CrossRef] [PubMed]
- Corney, R.H. Sex differences in general practice attendance and help seeking for minor illness. J. Psychosom. Res. 1990, 34, 525–534. [Google Scholar] [CrossRef] [PubMed]
- Key Statistics from the National Survey of Family Growth; Centers for Disease Control and Prevention: Atlanta, GA, USA, 2017.
- Boyle, A.; Reddy, U.M. Epidemiology of cesarean delivery: The scope of the problem. Semin. Perinatol. 2012, 36, 308–314. [Google Scholar] [CrossRef]
- Chan, W.V.; Le, B.; Lam, M.; Shariff, S.Z.; Gomes, T.; Lipscombe, L.; Murji, A. Opioid Prescribing Practices for Women Undergoing Elective Gynecologic Surgery. J. Minim. Invasive Gynecol. 2021, 28, 1325–1333. [Google Scholar] [CrossRef]
- Jain, N.; Phillips, F.M.; Weaver, T.; Khan, S.N. Preoperative Chronic Opioid Therapy: A Risk Factor for Complications, Readmission, Continued Opioid Use and Increased Costs After One- and Two-Level Posterior Lumbar Fusion. Spine 2018, 43, 1331–1338. [Google Scholar] [CrossRef]
- Jones, M.R.; Brovman, E.Y.; Novitch, M.B.; Rao, N.; Urman, R.D. Potential opioid-related adverse events following spine surgery in elderly patients. Clin. Neurol. Neurosurg. 2019, 186, 105550. [Google Scholar] [CrossRef]
- Vail, D.; Azad, T.; O’Connell, C.; Han, S.S.; Veeravagu, A.; Ratliff, J.K. Postoperative Opioid Use, Complications, and Costs in Surgical Management of Lumbar Spondylolisthesis. Spine 2018, 43, 1080–1088. [Google Scholar] [CrossRef]
- Manchikanti, L.; Kaye, A.M.; Knezevic, N.N.; McAnally, H.; Slavin, K.; Trescot, A.M.; Blank, S.; Pampati, V.; Abdi, S.; Grider, J.S.; et al. Responsible, Safe, and Effective Prescription of Opioids for Chronic Non-Cancer Pain: American Society of Interventional Pain Physicians (ASIPP) Guidelines. Pain Physician 2017, 20, S3–S92. [Google Scholar] [CrossRef] [PubMed]
- Overton, H.N.; Hanna, M.N.; Bruhn, W.E.; Hutfless, S.; Bicket, M.C.; Makary, M.A.; Matlaga, B.; Johnson, C.; Sheffield, J.; Shechter, R.; et al. Opioid-Prescribing Guidelines for Common Surgical Procedures: An Expert Panel Consensus. J. Am. Coll. Surg. 2018, 227, 411–418. [Google Scholar] [CrossRef] [PubMed]
- Wyles, C.C.; Hevesi, M.; Ubl, D.S.; Habermann, E.B.; Gazelka, H.M.; Trousdale, R.T.; Turner, N.S., III; Pagnano, M.W.; Mabry, T.M. Implementation of Procedure-Specific Opioid Guidelines: A Readily Employable Strategy to Improve Consistency and Decrease Excessive Prescribing Following Orthopaedic Surgery. JBJS Open Access 2020, 5, e0050. [Google Scholar] [CrossRef] [PubMed]
- Zaslansky, R.; Meissner, W.; Chapman, C.R. Pain after orthopaedic surgery: Differences in patient reported outcomes in the United States vs internationally. An observational study from the PAIN OUT dataset. Br. J. Anaesth. 2018, 120, 790–797. [Google Scholar] [CrossRef]
- Kavolus, J.J.; Ritter, M.A.; Claverie, J.G.; Salas, M.D.; Kavolus, C.H.; Trousdale, R.T. Cultural Nuance in Orthopedic Foreign Aid: Differences in Patient Concerns. J. Arthroplast. 2016, 31, 27–30. [Google Scholar] [CrossRef]
| High MME | Low MME | p-Value | |
|---|---|---|---|
| N | 3939 | 3946 | |
| MME (mg), mean (SD) | 393.49 (3466.59) | 67.38 (70.19) | <0.0001 |
| MME (mg), median | 177.02 | 41.64 | <0.0001 |
| MME (mg), IQR | 92.70–400.25 | 23.45–66.67 | |
| Index LOS (day), mean (SD) | 5.47 (6.71) | 3.96 (2.96) | <0.0001 |
| MME per day, mean (SD) | 64.86 (72.05) | 20.15 (25.70) | <0.0001 |
| MME per day, median | 40.72 | 12.26 | <0.0001 |
| MME per day, IQR | 21.47–90.49 | 7.00–21.59 | |
| Age (year), mean (SD) | 50.18 (17.07) | 54.09 (18.56) | <0.0001 |
| Height (cm), mean (SD) | 168.22 (10.55) | 167.74 (10.66) | 0.0599 |
| Weight (kg), mean (SD) | 89.17 (22.04) | 84.69 (20.90) | <0.0001 |
| BMI (kg/m2), mean (SD) | 31.46 (7.01) | 30.07 (7.15) | <0.0001 |
| Year-2015, N (%) | 1172 (29.7%) | 878 (22.3%) | <0.0001 |
| Year-2016, N (%) | 1173 (29.7%) | 1033 (26.2%) | |
| Year-2017, N (%) | 1107 (28.1%) | 1382 (35.1%) | |
| Year-2018, N (%) | 487 (12.3%) | 653 (16.6%) | |
| Female gender, N (%) | 2733 (69.3%) | 2690 (68.3%) | 0.2452 |
| Index Surgical Procedure | |||
| Spine surgery | 1225 | 1226 | |
| Cesarean delivery | 1154 | 1155 | |
| Hysterectomy | 155 | 155 | |
| Total hip arthroplasty | 616 | 617 | |
| Total knee arthroplasty | 789 | 793 | |
| Tobacco use, N (%) | <0.0001 | ||
| Ever | 1251 (31.7%) | 961 (24.4%) | |
| Never | 2452 (62.1%) | 2749 (69.8%) | |
| Unknown | 236 (6.0%) | 236 (6.0%) | |
| Alcohol use, N (%) | 0.3599 | ||
| Yes, N (%) | 1153 (29.2%) | 1172 (29.8%) | |
| No, N (%) | 2468 (62.5%) | 2489 (63.2%) | |
| Unknown, N (%) | 318 (8.1%) | 285 (7.2%) | |
| Any opioids over 180 days before index admission, N (%) | 1014 (25.7%) | 767 (19.5%) | <0.0001 |
| Any opioids over 90 days before index admission, N (%) | 648 (16.4%) | 460 (11.7%) | <0.0001 |
| Any opioids over 30 days before index admission, N (%) | 383 (9.7%) | 262 (6.7%) | <0.0001 |
| 180-day baseline ORAE—w/ or w/o opioid exposure, N (%) | 967 (24.5%) | 781 (19.8%) | <0.0001 |
| Central nervous system disorder | 40 (1.0%) | 36 (0.9%) | 0.6392 |
| Cardiovascular | 280 (7.1%) | 252 (6.4%) | 0.2011 |
| Gastrointestinal | 400 (10.1%) | 320 (8.1%) | 0.0016 |
| Respiratory | 213 (5.4%) | 186 (4.7%) | 0.1599 |
| Skin | 133 (3.4%) | 92 (2.3%) | 0.0053 |
| Urinary | 33 (0.8%) | 14 (0.4%) | 0.0053 |
| Other | 263 (6.7%) | 135 (3.4%) | <0.0001 |
| 180-day baseline ORAE—among baseline opioid user, N (%) | 373 (9.5%) | 240 (6.1%) | <0.0001 |
| Central nervous system disorder | 21 (0.5%) | 14 (0.4%) | 0.2336 |
| Cardiovascular | 130 (3.3%) | 77 (2.0%) | 0.0002 |
| Gastrointestinal | 159 (4.0%) | 97 (2.5%) | <0.0001 |
| Respiratory | 105 (2.7%) | 68 (1.7%) | 0.0043 |
| Skin | 42 (1.1%) | 27 (0.7%) | 0.0686 |
| Urinary | 18 (0.5%) | 4 (0.1%) | 0.0028 |
| Other | 128 (3.2%) | 58 (1.5%) | <0.0001 |
| Baseline Rx pain medication, N (%) | 775 (19.6%) | 727 (18.5%) | 0.1571 |
| Gabapentin | 352 (8.9%) | 373 (9.5%) | 0.4276 |
| Pregabalin | 279 (7.1%) | 228 (5.8%) | 0.0182 |
| Celecoxib | 318 (8.1%) | 273 (6.9%) | 0.0515 |
| Meloxicam | 159 (4.0%) | 122 (3.1%) | 0.0236 |
| Charlson Comorbidity Index, N (%) | |||
| CCI 0 | 2795 (70.8%) | 2808 (71.3%) | 0.9823 |
| CCI 1 | 688 (17.4%) | 691 (17.5%) | |
| CCI 2 | 267 (6.8%) | 265 (6.7%) | |
| CCI 3+ | 189 (4.8%) | 182 (4.6%) | |
| Myocardial infarction | 56 (1.4%) | 59 (1.5%) | 0.7854 |
| Cerebrovascular disorder | 90 (2.3%) | 98 (2.5%) | 0.5631 |
| Congestive heart failure | 87 (2.2%) | 92 (2.3%) | 0.7144 |
| Diabetes, no complication | 382 (9.7%) | 401 (10.2%) | 0.4906 |
| Diabetes with complication | 152 (3.9%) | 179 (4.5%) | 0.1337 |
| Para/hemiplegia | 34 (0.9%) | 36 (0.9%) | 0.816 |
| Peptic ulcer | 11 (0.3%) | 14 (0.4%) | 0.5508 |
| Pulmonary disorder | 491 (12.4%) | 436 (11.1%) | 0.051 |
| Peripheral vascular disorder | 101 (2.6%) | 128 (3.2%) | 0.0723 |
| Renal disorder | 111 (2.8%) | 100 (2.5%) | 0.435 |
| Dementia | 8 (0.2%) | 12 (0.3%) | 0.3726 |
| Rheumatoid Arthritis | 105 (2.7%) | 95 (2.4%) | 0.466 |
| Liver, mild | 85 (2.2%) | 54 (1.4%) | 0.0077 |
| Liver, moderate to severe | 15 (0.4%) | 2 (0.1%) | 0.0016 |
| AIDS/HIV | 13 (0.3%) | 13 (0.3%) | 0.9964 |
| Cancer | 0 (0.0%) | 0 (0.0%) | n/a |
| Cancer, metastasis | 0 (0.0%) | 0 (0.0%) | n/a |
| Race/Ethnicity, N (%) | 0.0100 | ||
| Non-hispanic white | 3184 (80.7%) | 3071 (78.0%) | |
| Hispanic/Latino | 389 (9.9%) | 461 (11.7%) | |
| Other | 335 (8.5%) | 383 (9.7%) | |
| Unknown | 31 (0.8%) | 31 (0.8%) | |
| Index opioids, any, N (%) | 3939 (99.8%) | 3889 (98.7%) | <0.0001 |
| Hydrocodone | 415 (10.5%) | 463 (11.8%) | 0.0909 |
| Buprenorphine | 16 (0.4%) | 0 (0.0%) | <0.0001 |
| Butorphanol | 0 (0.0%) | 1 (0.0%) | 0.3177 |
| Codeine | 5 (0.1%) | 7 (0.2%) | 0.5655 |
| Fentanyl | 1429 (36.2%) | 1165 (29.6%) | <0.0001 |
| Hydromorphone | 2654 (67.3%) | 1794 (45.5%) | <0.0001 |
| Meperidine | 404 (10.2%) | 247 (6.3%) | <0.0001 |
| Methadone | 90 (2.3%) | 31 (0.8%) | <0.0001 |
| Morphine | 1268 (32.1%) | 1090 (27.7%) | <0.0001 |
| Nalbuphine | 249 (6.3%) | 214 (5.4%) | 0.0898 |
| Oxycodone | 3588 (90.9%) | 3064 (77.8%) | <0.0001 |
| Oxymorphone | 2 (0.1%) | 0 (0.0%) | 0.2495 |
| Pentazocine | 0 (0.0%) | 0 (0.0%) | n/a |
| Remifentanil | 1733 (43.9%) | 505 (12.8%) | <0.0001 |
| Sufentanil | 167 (4.2%) | 179 (4.5%) | 0.5203 |
| Tapentadol | 114 (2.9%) | 111 (2.8%) | 0.8287 |
| Tramadol | 1666 (42.2%) | 1738 (44.1%) | 0.1168 |
| Anesthetic technique, N (%) | <0.0001 | ||
| General | 2147 (54.4%) | 1578 (40.1%) | |
| Other | 48 (1.2%) | 39 (1.0%) | |
| Regional | 1703 (43.2%) | 2254 (57.2%) | |
| None | 0 (0.0%) | 1 (0.0%) | |
| Unknown | 41 (1.0%) | 74 (1.9%) | |
| Health plan, N (%) | <0.0001 | ||
| Commercial | 2096 (53.1%) | 2041 (51.8%) | |
| Medicare | 1069 (27.1%) | 1326 (33.7%) | |
| Medicaid | 574 (14.5%) | 432 (11.0%) | |
| None | 45 (1.1%) | 32 (0.8%) | |
| Other | 155 (3.9%) | 115 (2.9%) | |
| Outcomes and Subgroup | High MME | Low MME | p-Value | |
|---|---|---|---|---|
| Unadjusted | Holm | |||
| 1+ Rx Opioid 31–180 days | ||||
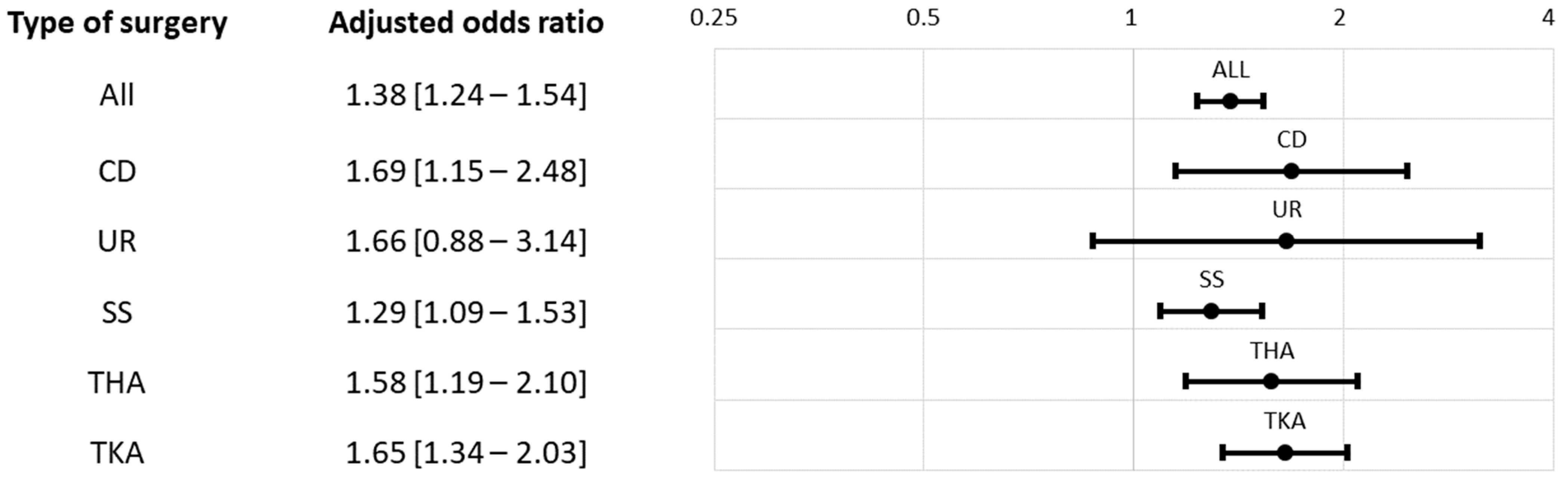
| All, N (%) | 1318 (33.5%) | 964 (24.4%) | <0.0001 | <0.0001 |
| CD | 91 (7.9%) | 44 (3.8%) | <0.0001 | <0.0001 |
| UR | 31 (20.0%) | 21 (13.5%) | 0.1285 | 0.1285 |
| SS | 520 (42.4%) | 451 (36.8%) | 0.0042 | 0.0042 |
| THA | 245 (39.8%) | 141 (22.9%) | <0.0001 | <0.0001 |
| TKA | 431 (54.6%) | 307 (38.7%) | <0.0001 | <0.0001 |
| 1+ Rx Opioid 91–180 days | ||||
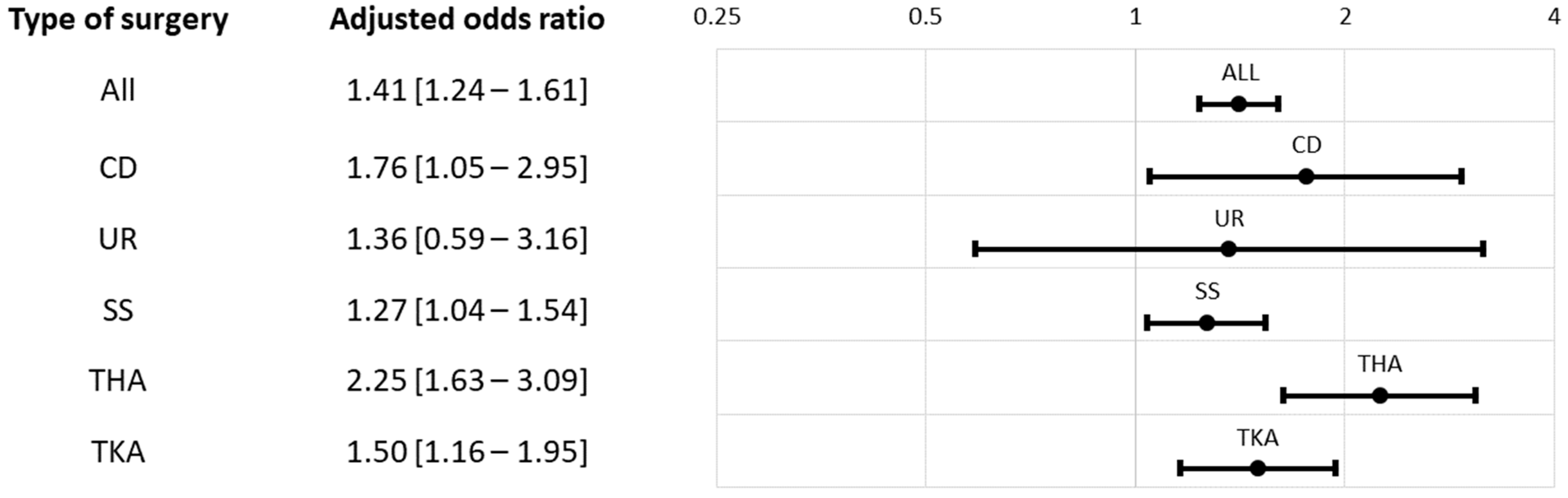
| All, N (%) | 771 (19.6%) | 513 (13.0%) | <0.0001 | <0.0001 |
| CD | 52 (4.5%) | 23 (2.0%) | 0.0007 | 0.0013 |
| UR | 14 (9.0%) | 12 (7.7%) | 0.6820 | 0.6820 |
| SS | 304 (24.8%) | 262 (21.4%) | 0.0430 | 0.0859 |
| THA | 153 (24.8%) | 69 (11.2%) | <0.0001 | <0.0001 |
| TKA | 248 (31.4%) | 147 (18.5%) | <0.0001 | <0.0001 |
| Type of Surgery | ORAE Type | High MME n (%) | Low MME n (%) | p-Value | |
|---|---|---|---|---|---|
| Unadjusted | Holm | ||||
| All | Any | 1073 (27.2%) | 837 (21.2%) | <0.01 | <0.01 |
| Central nervous system | 111 (2.8%) | 94 (2.4%) | 0.22 | 0.45 | |
| Cardiovascular | 326 (8.3%) | 219 (5.5%) | <0.01 | <0.01 | |
| Gastrointestinal | 433 (11.0%) | 332 (8.4%) | <0.01 | <0.01 | |
| Respiratory | 276 (7.0%) | 215 (5.4%) | <0.01 | 0.02 | |
| Genitourinary | 98 (2.5%) | 63 (1.6%) | <0.01 | 0.02 | |
| Skin | 101 (2.6%) | 85 (2.2%) | 0.23 | 0.45 | |
| Others | 459 (11.7%) | 310 (7.9%) | <0.01 | <0.01 | |
| CD | Any | 220 (19.1%) | 138 (11.9%) | <0.01 | <0.01 |
| Central nervous system | 10 (0.9%) | 6 (0.5%) | 0.31 | 0.31 | |
| Cardiovascular | 70 (6.1%) | 37 (3.2%) | <0.01 | <0.01 | |
| Gastrointestinal | 97 (8.4%) | 68 (5.9%) | 0.02 | 0.06 | |
| Respiratory | 26 (2.3%) | 8 (0.7%) | <0.01 | 0.01 | |
| Genitourinary | 8 (0.7%) | 2 (0.2%) | 0.06 | 0.11 | |
| Skin | 34 (2.9%) | 13 (1.1%) | <0.01 | 0.01 | |
| Others | 67 (5.8%) | 39 (3.4%) | <0.01 | 0.02 | |
| UR | Any | 59 (38.1%) | 47 (30.3%) | 0.15 | 0.60 |
| Central nervous system | 8 (5.2%) | 1 (0.6%) | 0.02 | 0.13 | |
| Cardiovascular | 22 (14.2%) | 12 (7.7%) | 0.07 | 0.41 | |
| Gastrointestinal | 37 (23.9%) | 20 (12.9%) | 0.01 | 0.10 | |
| Respiratory | 10 (6.5%) | 11 (7.1%) | 0.82 | 1.00 | |
| Genitourinary | 9 (5.8%) | 3 (1.9%) | 0.08 | 0.41 | |
| Skin | 4 (2.6%) | 7 (4.5%) | 0.36 | 1.00 | |
| Others | 23 (14.8%) | 25 (16.1%) | 0.75 | 1.00 | |
| SS | Any | 390 (31.8%) | 328 (26.8%) | <0.01 | 0.04 |
| Central nervous system | 69 (5.6%) | 62 (5.1%) | 0.53 | 0.85 | |
| Cardiovascular | 126 (10.3%) | 91 (7.4%) | 0.01 | 0.08 | |
| Gastrointestinal | 167 (13.6%) | 142 (11.6%) | 0.13 | 0.51 | |
| Respiratory | 108 (8.8%) | 92 (7.5%) | 0.24 | 0.71 | |
| Genitourinary | 55 (4.5%) | 38 (3.1%) | 0.07 | 0.36 | |
| Skin | 26 (2.1%) | 32 (2.6%) | 0.43 | 0.85 | |
| Others | 188 (15.3%) | 130 (10.6%) | <0.01 | <0.01 | |
| THA | Any | 156 (25.3%) | 128 (20.7%) | 0.06 | 0.38 |
| Central nervous system | 9 (1.5%) | 7 (1.1%) | 0.61 | 1.00 | |
| Cardiovascular | 32 (5.2%) | 31 (5.0%) | 0.89 | 1.00 | |
| Gastrointestinal | 49 (8.0%) | 41 (6.6%) | 0.38 | 1.00 | |
| Respiratory | 45 (7.3%) | 29 (4.7%) | 0.05 | 0.38 | |
| Genitourinary | 14 (2.3%) | 8 (1.3%) | 0.20 | 0.98 | |
| Skin | 17 (2.8%) | 12 (1.9%) | 0.35 | 1.00 | |
| Others | 73 (11.9%) | 52 (8.4%) | 0.05 | 0.37 | |
| TKA | Any | 248 (31.4%) | 196 (24.7%) | <0.01 | 0.02 |
| Central nervous system | 15 (1.9%) | 18 (2.3%) | 0.61 | 1.00 | |
| Cardiovascular | 76 (9.6%) | 48 (6.1%) | <0.01 | 0.05 | |
| Gastrointestinal | 83 (10.5%) | 61 (7.7%) | 0.05 | 0.25 | |
| Respiratory | 87 (11.0%) | 75 (9.5%) | 0.30 | 1.00 | |
| Genitourinary | 12 (1.5%) | 12 (1.5%) | 0.99 | 1.00 | |
| Skin | 20 (2.5%) | 21 (2.6%) | 0.89 | 1.00 | |
| Others | 108 (13.7%) | 64 (8.1%) | <0.01 | <0.01 | |
| Type of Surgery (n, High MME vs. Low MME) | Mean (SD) | Median [IQR]; Min–Max | ||||||
|---|---|---|---|---|---|---|---|---|
| High MME | Low MME | p-Value * | High MME | Low MME | p-Value † | |||
| Unadjusted | Holm | Unadjusted | Holm | |||||
| Number of readmissions | ||||||||
| All (3939 vs. 3946) | 0.18 (0.51) | 0.13 (0.44) | <0.01 | <0.01 | 0 [0–0]; 0–5 | 0 [0–0]; 0–7 | <0.01 | <0.01 |
| CD (1154 vs. 1155) | 0.05 (0.25) | 0.02 (0.15) | <0.01 | <0.01 | 0 [0–0]; 0–3 | 0 [0–0]; 0–2 | <0.01 | <0.01 |
| UR (155 vs. 155) | 0.12 (0.45) | 0.11 (0.43) | 0.80 | 1.00 | 0 [0–0]; 0–4 | 0 [0–0]; 0–3 | 0.57 | 1.00 |
| SS (1225 vs. 1226) | 0.22 (0.59) | 0.18 (0.57) | 0.05 | 0.36 | 0 [0–0]; 0–5 | 0 [0–0]; 0–7 | <0.01 | 0.04 |
| THA (616 vs. 617) | 0.29 (0.66) | 0.15 (0.40) | <0.01 | <0.01 | 0 [0–0]; 0–5 | 0 [0–0]; 0–3 | <0.01 | <0.01 |
| TKA (789 vs. 793) | 0.25 (0.52) | 0.20 (0.50) | 0.04 | 0.36 | 0 [0–0]; 0–3 | 0 [0–0]; 0–5 | 0.02 | 0.12 |
| Cumulative length of stay | ||||||||
| All (3939 vs. 3946) | 1.09 (4.75) | 0.64 (3.56) | <0.01 | <0.01 | 0 [0–0]; 0–109 | 0 [0–0]; 0–109 | <0.01 | <0.01 |
| CD (1154 vs. 1155) | 0.20 (1.30) | 0.07 (0.73) | <0.01 | 0.03 | 0 [0–0]; 0–26 | 0 [0–0]; 0–16 | <0.01 | <0.01 |
| UR (155 vs. 155) | 0.94 (6.86) | 0.70 (2.98) | 0.70 | 1.00 | 0 [0–0]; 0–83 | 0 [0–0]; 0–21 | 0.62 | 1.00 |
| SS (1225 vs. 1226) | 1.87 (7.21) | 1.17 (5.53) | <0.01 | 0.06 | 0 [0–0]; 0–109 | 0 [0–0]; 0–109 | <0.01 | 0.02 |
| THA (616 vs. 617) | 1.29 (3.64) | 0.55 (2.30) | <0.01 | <0.01 | 0 [0–0]; 0–41 | 0 [0–0]; 0–42 | <0.01 | <0.01 |
| TKA (789 vs. 793) | 1.08 (2.84) | 0.70 (2.89) | <0.01 | 0.07 | 0 [0–0]; 0–32 | 0 [0–0]; 0–45 | <0.01 | 0.02 |
| Cumulative length of stay, among ever admitted | ||||||||
| All (561 vs. 399) | 7.6 (10.41) | 6.3 (9.5) | 0.54 | 1.00 | 4 [3–8]; 1–109 | 4 [2–6]; 1–109 | 0.21 | 1.00 |
| CD (45 vs. 19) | 5.1 (4.38) | 4.3 (3.9) | 0.6 | 1.00 | 4 [2–6]; 1–26 | 3 [2–4]; 2–16 | 0.68 | 1.00 |
| UR (15 vs. 12) | 9.7 (20.67) | 9.1 (6.4) | 0.63 | 1.00 | 4 [2–5]; 1–83 | 7.5 [4.5–13]; 2–21 | 0.50 | 1.00 |
| SS (203 vs. 153) | 11.3 (14.4) | 9.4 (13.0) | 0.48 | 1.00 | 6 [3–11]; 1–109 | 5 [4–10]; 1–109 | 0.50 | 1.00 |
| THA (130 vs. 84) | 6.1 (5.8) | 4.1 (5.0) | <0.01 | <0.01 | 4 [3–8]; 1–41 | 3 [2–4]; 1–42 | <0.01 | <0.01 |
| TKA (168 vs. 131) | 5.1 (4.2) | 4.2 (6.0) | 0.72 | 1.00 | 4 [3–6]; 2–32 | 2 [2–4]; 1–45 | 0.93 | 1.00 |
| Average length of inpatient stay per admission, among ever admitted | ||||||||
| All (561 vs. 399) | 5.4 (5.6) | 4.4 (3.7) | <0.01 | <0.01 | 4 [3–6]; 1–55 | 3 [2–5]; 1–31 | <0.01 | <0.01 |
| CD (45 vs. 19) | 4.0 (2.7) | 3.6 (3.2) | 0.61 | 1.00 | 3 [2–5]; 1–14 | 3 [2–3.5]; 2–16 | 0.27 | 1.00 |
| UR (15 vs. 12) | 5.1 (5.3) | 6.5 (3.9) | 0.44 | 1.00 | 4 [2–5]; 1–20.75 | 5.7 [4.25–7.5]; 2–17 | 0.08 | 0.65 |
| SS (203 vs. 153) | 7.7 (8.2) | 6.0 (4.8) | 0.02 | 0.12 | 5.5 [3–8]; 1–54.5 | 5 [3–7]; 1–31 | 0.06 | 0.45 |
| THA (130 vs. 84) | 4.1 (2.3) | 3.4 (2.3) | 0.03 | 0.23 | 3.25 [3–5]; 1–14 | 3 [2–4]; 1–14 | <0.01 | <0.01 |
| TKA (168 vs. 131) | 4.2 (2.1) | 3.1 (1.7) | <0.01 | <0.01 | 4 [3–5]; 2–14 | 2 [2–4]; 1–10.75 | <0.01 | <0.01 |
| Type of Surgery (n, High MME vs. Low MME) | Mean (SD) | Median [IQR]; Min–Max | ||||||
|---|---|---|---|---|---|---|---|---|
| High MME | Low MME | p-Value * | High MME | Low MME | p-Value † | |||
| Unadjusted | Holm | Unadjusted | Holm | |||||
| Number of office visits | ||||||||
| All (3939 vs. 3946) | 5.36 (6.91) | 5.05 (6.23) | 0.04 | 0.29 | 3 [1–7]; 0–109 | 3 [1–6]; 0–67 | 0.03 | 0.21 |
| CD (1154 vs. 1155) | 1.89 (2.70) | 1.49 (2.13) | <0.01 | <0.01 | 1 [0–3]; 0–34 | 1 [0–2]; 0–21 | <0.01 | 0.01 |
| UR (155 vs. 155) | 3.26 (4.53) | 2.23 (3.08) | 0.02 | 0.16 | 2 [0–5]; 0–32 | 1 [0–3]; 0–18 | 0.10 | 0.83 |
| SS (1225 vs. 1226) | 6.15 (8.00) | 5.64 (6.30) | 0.08 | 0.64 | 4 [2–7]; 0–109 | 4 [2–7]; 0–67 | 0.59 | 1.00 |
| THA (616 vs. 617) | 6.29 (5.74) | 6.21 (5.85) | 0.82 | 1.00 | 4 [2–8]; 0–49 | 4 [2–8]; 0–40 | 0.40 | 1.00 |
| TKA (789 vs. 793) | 8.88 (8.13) | 8.94 (7.76) | 0.88 | 1.00 | 6 [3–12]; 0–51 | 6 [3–13]; 0–47 | 0.63 | 1.00 |
| Number of ER visits | ||||||||
| All (3939 vs. 3946) | 0.24 (0.86) | 0.18 (0.91) | <0.01 | 0.08 | 0 [0–0]; 0–16 | 0 [0–0]; 0–32 | <0.01 | <0.01 |
| CD (1154 vs. 1155) | 0.24 (0.80) | 0.16 (0.53) | <0.01 | 0.03 | 0 [0–0]; 0–12 | 0 [0–0]; 0–6 | 0.03 | 0.26 |
| UR (155 vs. 155) | 0.35 (1.09) | 0.05 (0.26) | <0.01 | <0.01 | 0 [0–0]; 0–10 | 0 [0–0]; 0–2 | <0.01 | <0.01 |
| SS (1225 vs. 1226) | 0.24 (0.92) | 0.30 (1.44) | 0.18 | 1.00 | 0 [0–0]; 0–16 | 0 [0–0]; 0–32 | 0.40 | 1.00 |
| THA (616 vs. 617) | 0.22 (0.85) | 0.10 (0.48) | <0.01 | 0.03 | 0 [0–0]; 0–10 | 0 [0–0]; 0–5 | <0.01 | <0.01 |
| TKA (789 vs. 793) | 0.22 (0.78) | 0.14 (0.53) | <0.01 | 0.08 | 0 [0–0]; 0–8 | 0 [0–0]; 0–7 | 0.05 | 0.41 |
| Number of telephone encounters | ||||||||
| All (3939 vs. 3946) | 1.28 (1.63) | 1.09 (1.42) | <0.01 | <0.01 | 1 [0–2]; 0–11 | 1 [0–2]; 0–11 | <0.01 | <0.01 |
| CD (1154 vs. 1155) | 0.46 (0.78) | 0.33 (0.68) | <0.01 | <0.01 | 0 [0–1]; 0–5 | 0 [0–0]; 0–4 | <0.01 | <0.01 |
| UR (155 vs. 155) | 1.17 (1.32) | 1.52 (1.27) | 0.02 | 0.15 | 1 [0–2]; 0–6 | 1 [1–2]; 0–5 | <0.01 | 0.04 |
| SS (1225 vs. 1226) | 1.14 (1.19) | 1.16 (1.12) | 0.72 | 1.00 | 1 [0–2]; 0–9 | 1 [0–2]; 0–8 | 0.24 | 1.00 |
| THA (616 vs. 617) | 2.00 (2.09) | 1.40 (1.71) | <0.01 | <0.01 | 1 [0–3]; 0–10 | 1 [0–2]; 0–9 | <0.01 | <0.01 |
| TKA (789 vs. 793) | 2.13 (2.08) | 1.76 (1.86) | <0.01 | <0.01 | 2 [1–3]; 0–11 | 1 [0–2]; 0–11 | <0.01 | <0.01 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kim, K.; Biskupiak, J.E.; Babin, J.L.; Ilham, S. Positive Association between Peri-Surgical Opioid Exposure and Post-Discharge Opioid-Related Outcomes. Healthcare 2023, 11, 115. https://doi.org/10.3390/healthcare11010115
Kim K, Biskupiak JE, Babin JL, Ilham S. Positive Association between Peri-Surgical Opioid Exposure and Post-Discharge Opioid-Related Outcomes. Healthcare. 2023; 11(1):115. https://doi.org/10.3390/healthcare11010115
Chicago/Turabian StyleKim, Kibum, Joseph E. Biskupiak, Jennifer L. Babin, and Sabrina Ilham. 2023. "Positive Association between Peri-Surgical Opioid Exposure and Post-Discharge Opioid-Related Outcomes" Healthcare 11, no. 1: 115. https://doi.org/10.3390/healthcare11010115
APA StyleKim, K., Biskupiak, J. E., Babin, J. L., & Ilham, S. (2023). Positive Association between Peri-Surgical Opioid Exposure and Post-Discharge Opioid-Related Outcomes. Healthcare, 11(1), 115. https://doi.org/10.3390/healthcare11010115






