Adverse Childhood Events, Post-Traumatic Stress Disorder, Infectious Encephalopathies and Immune-Mediated Disease
Abstract
1. Introduction
2. Materials and Methods
3. Results
3.1. Relevant Contributors
3.1.1. Trauma
3.1.2. Post-Traumatic Stress Disorder
3.1.3. Intrusive Symptoms
3.1.4. Adverse Childhood Events
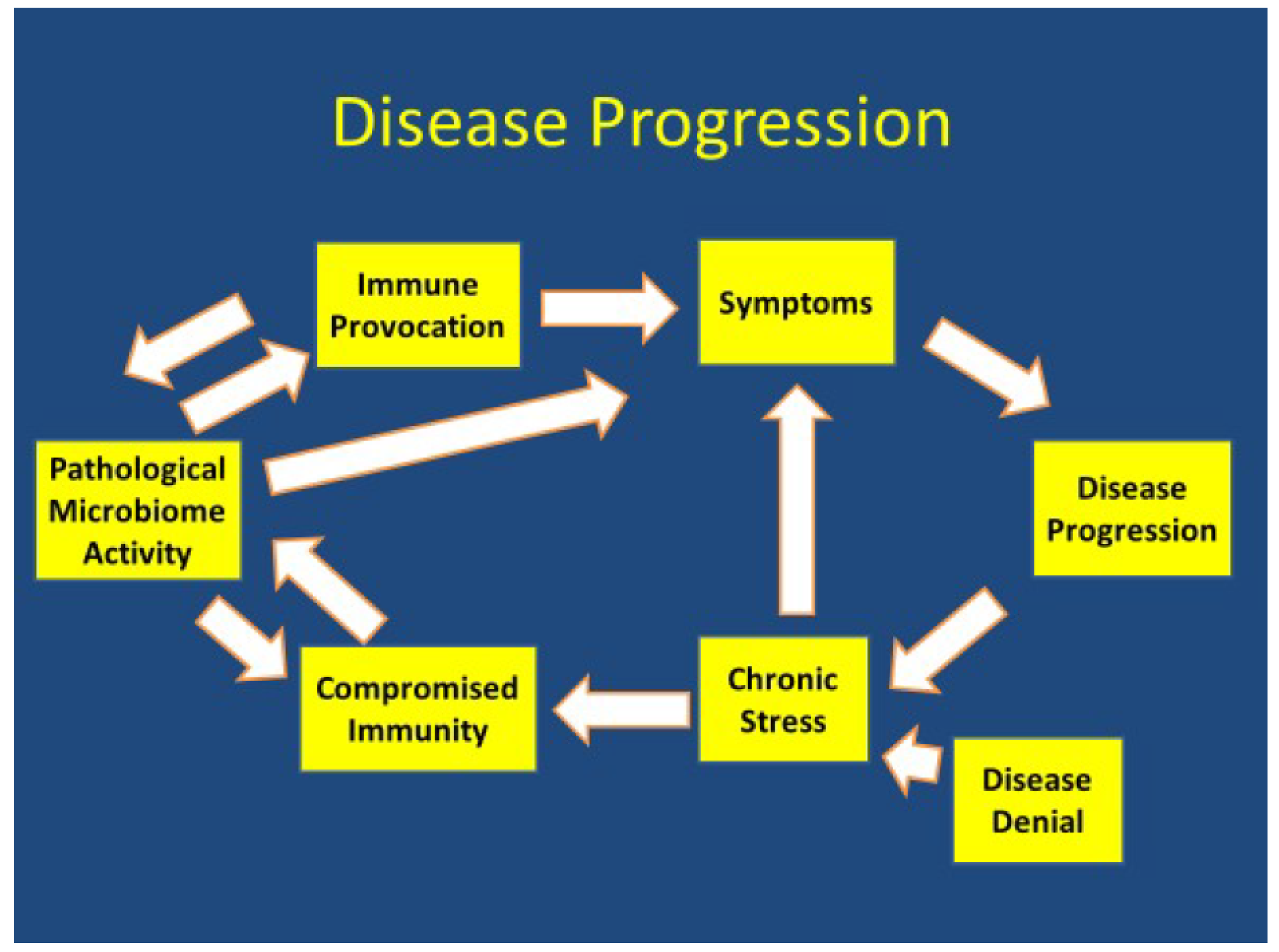
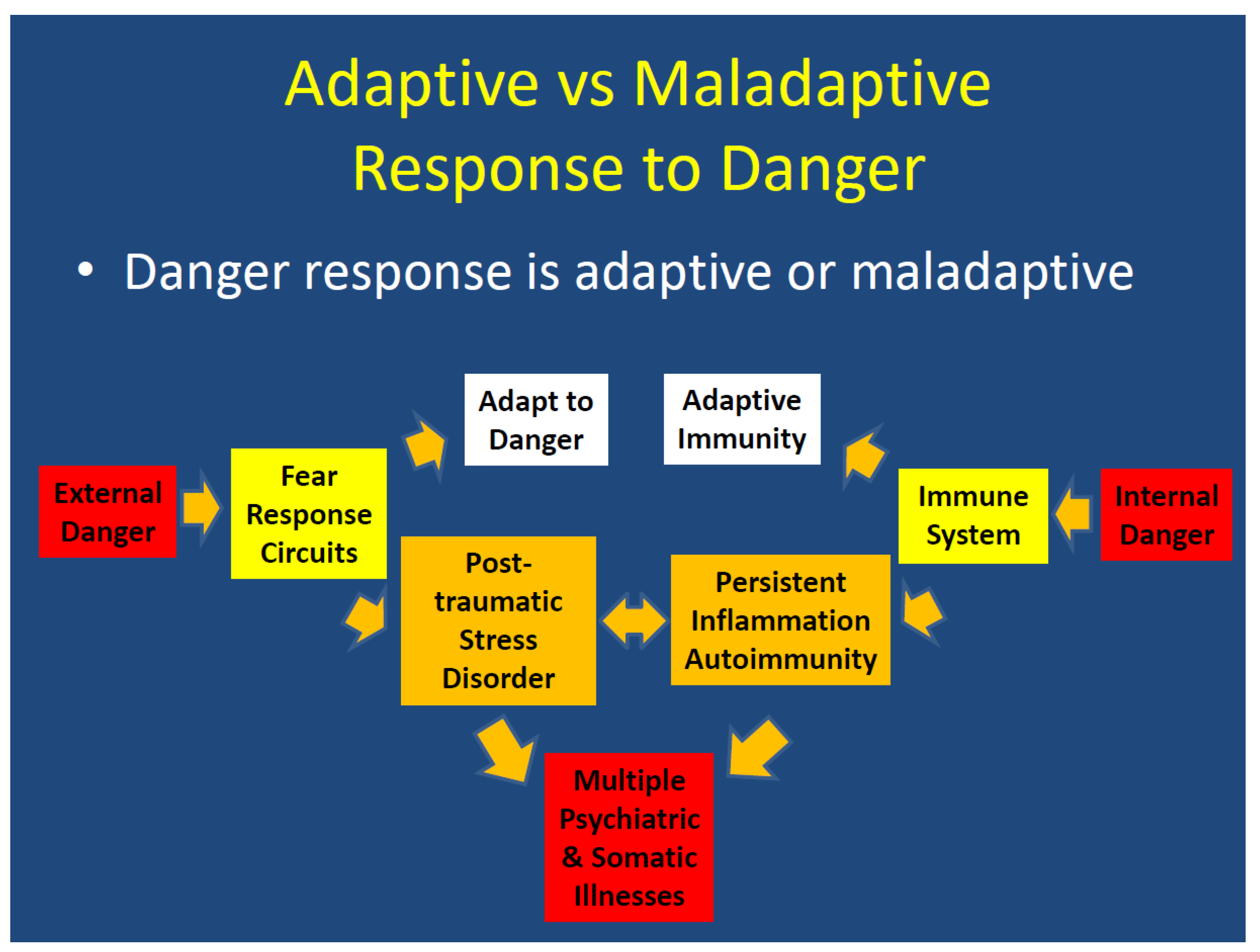
3.2. Pathophysiology
4. Discussion
4.1. Overview
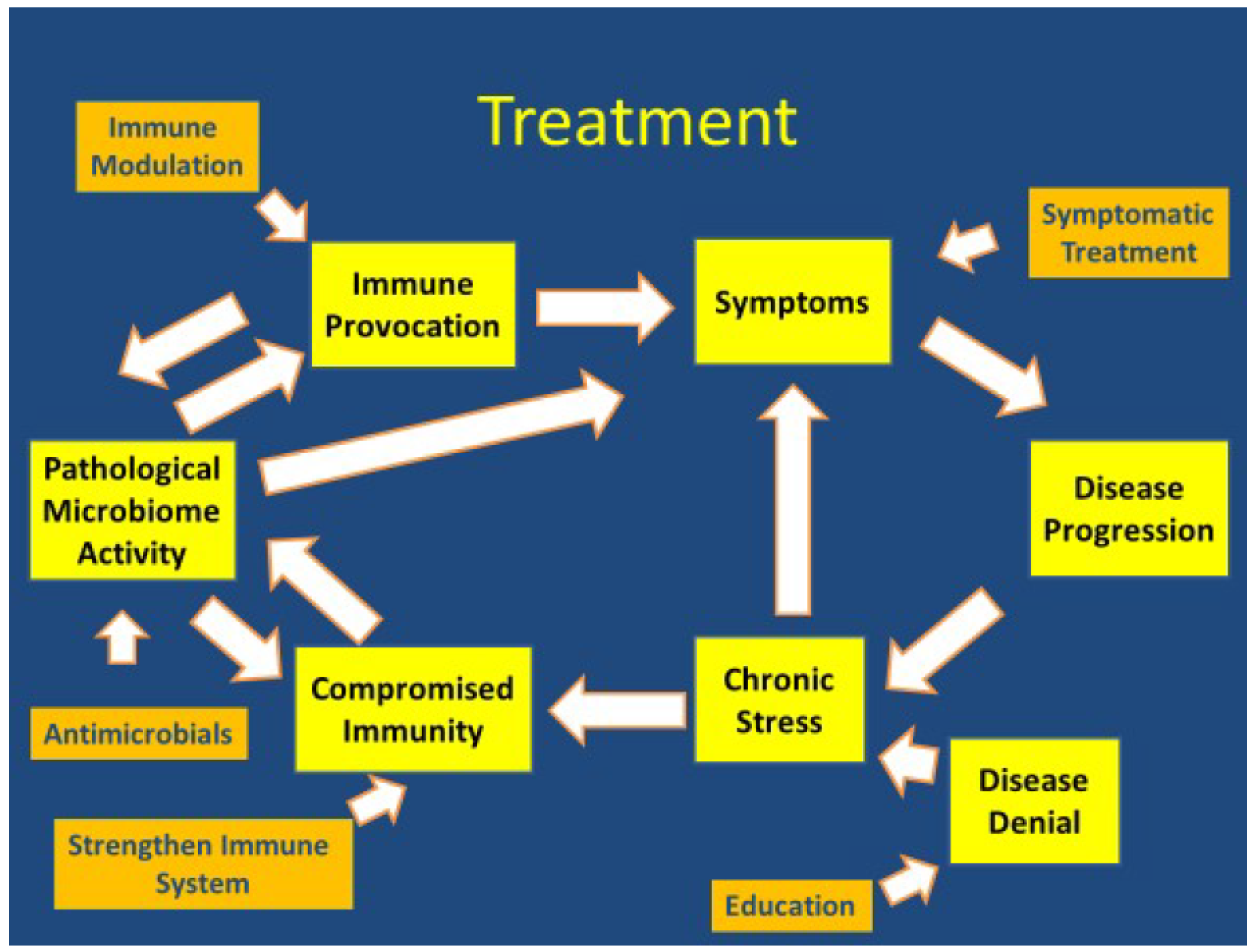
4.2. Treatment Implications
Funding
Acknowledgments
Conflicts of Interest
References
- Van der Kolk, B. The Body Keeps the Score: Brain, Mind, and Body in the Healing of Trauma; Penguin: New York, NY, USA, 2014. [Google Scholar]
- Dube, S.R.; Fairweather, D.; Pearson, W.S.; Felitti, V.J.; Anda, R.F.; Croft, J.B. Cumulative Childhood Stress and Autoimmune Diseases in Adults. Psychosom. Med. 2009, 71, 243–250. [Google Scholar] [CrossRef] [PubMed]
- Lin, J.E.; Neylan, T.C.; Epel, E.; O’Donovan, A. Associations of childhood adversity and adulthood trauma with C-reactive protein: A cross-sectional population-based study. Brain Behav. Immun. 2015, 53, 105–112. [Google Scholar] [CrossRef] [PubMed]
- Copeland, W.E.; Wolke, D.; Lereya, S.T.; Shanahan, L.; Worthman, C.; Costello, E.J. Childhood bullying involvement predicts low-grade systemic inflammation into adulthood. Proc. Natl. Acad. Sci. USA 2014, 111, 7570–7575. [Google Scholar] [CrossRef] [PubMed]
- Felitti, V.J.; Anda, R.F.; Nordenberg, D.; Williamson, D.F.; Spitz, A.M.; Edwards, V.; Koss, M.P.; Marks, J.S. Relationship of Childhood Abuse and Household Dysfunction to Many of the Leading Causes of Death in Adults: The Adverse Childhood Experiences (ACE) Study. Am. J. Prev. Med. 1998, 14, 245–258. [Google Scholar] [CrossRef]
- Fallon, B.A.; Levin, E.S.; Schweitzer, P.J.; Hardesty, D. Inflammation and central nervous system Lyme disease. Neurobiol. Dis. 2010, 37, 534–541. [Google Scholar] [CrossRef]
- Bransfield, R.C. The Psychoimmunology of Lyme/Tick-Borne Diseases and its Association with Neuropsychiatric Symptoms. Open Neurol. J. 2012, 6, 88–93. [Google Scholar] [CrossRef]
- Bransfield, R. Relationship of Inflammation and Autoimmunity to Psychiatric Sequelae in Lyme Disease. Psychiatr. Ann. 2012, 42, 337–341. [Google Scholar] [CrossRef]
- Ramesh, G.; Didier, P.J.; England, J.D.; Santana-Gould, L.; Doyle-Meyers, L.A.; Martin, D.S.; Jacobs, M.B.; Philipp, M.T. Inflammation in the Pathogenesis of Lyme Neuroborreliosis. Am. J. Pathol. 2015, 185, 1344–1360. [Google Scholar] [CrossRef]
- Bransfield, R.C.; Aidlen, D.M.; Cook, M.J.; Javia, S. A Clinical Diagnostic System for Late-Stage Neuropsychiatric Lyme Borreliosis Based upon an Analysis of 100 Patients. Healthcare 2020, 8, 13. [Google Scholar] [CrossRef]
- Bransfield, R.C. Intrusive symptoms and infectious encephalopathies. Neurol. Psychiatry Brain Res. 2016, 22, 3–4. [Google Scholar] [CrossRef]
- Swedo, S.E.; Leonard, H.L.; Garvey, M.; Mittleman, B.; Allen, A.J.; Perlmutter, S.; Lougee, L.; Dow, S.; Zamkoff, J.; Dubbert, B.K. Pediatric autoimmune neuropsychiatric disorders associated with streptococcal infections: Clinical description of the first 50 cases. Am. J. Psychiatry 1998, 155, 264–271. [Google Scholar] [CrossRef] [PubMed]
- Grant, J.E.; Drummond, L.; Nicholson, T.R.; Fagan, H.; Baldwin, D.S.; Fineberg, N.A.; Chamberlain, S.R. Obsessive-compulsive symptoms and the Covid-19 pandemic: A rapid scoping review. Neurosci. Biobehav. Rev. 2021, 132, 1086–1098. [Google Scholar] [CrossRef] [PubMed]
- Nezgovorova, V.; Ferretti, C.J.; Pallanti, S.; Hollander, E. Modulating neuroinflammation in COVID-19 patients with obsessive-compulsive disorder. J. Psychiatry Res. 2022, 149, 367–373. [Google Scholar] [CrossRef]
- Breitschwerdt, E.B.; Greenberg, R.; Maggi, R.G.; Mozayeni, B.R.; Lewis, A.; Bradley, J.M. Bartonella henselae Bloodstream Infection in a Boy With Pediatric Acute-Onset Neuropsychiatric Syndrome. J. Central Nerv. Syst. Dis. 2019, 11, 1179573519832014. [Google Scholar] [CrossRef]
- Bransfield, R.C. Lyme disease, comorbid tick-borne diseases, and neuropsychiatric disorders. Psychiatry Times 2007, 24, 59–61. [Google Scholar]
- Qiu, D.; Li, Y.; Li, L.; He, J.; Ouyang, F.; Xiao, S. Infectious Disease Outbreak and Post-Traumatic Stress Symptoms: A Systematic Review and Meta-Analysis. Front. Psychol. 2021, 12, 668784. [Google Scholar] [CrossRef]
- Kessler, R.C. National Comorbidity Replication Study. Arch. Gen. Psychiatry 1995, 53, 1048. [Google Scholar] [CrossRef]
- Fallon, B.A.; Schwartzberg, M.; Bransfield, R.; Zimmerman, B.; Scotti, A.; Weber, C.A.; Liebowitz, M.R. Late-Stage Neuropsychiatric Lyme Borreliosis: Differential Diagnosis and Treatment. J. Psychosom. Res. 1995, 36, 295–300. [Google Scholar] [CrossRef]
- Stein, S.L.; Solvason, H.B.; Biggart, E.; Spiegel, D. A 25-year-old woman with hallucinations, hypersexuality, nightmares, and a rash [clinical conference]. Am. J. Psychiatry 1996, 153, 545–551. [Google Scholar]
- Bar, K.-J.; Jochum, T.; Hager, F.; Meissner, W.; Sauer, H. Painful Hallucinations and Somatic Delusions in a Patient With the Possible Diagnosis of Neuroborreliosis. Clin. J. Pain 2005, 21, 362–363. [Google Scholar] [CrossRef]
- Bransfield, R.C. Can infections and immune reactions to them cause violent behavior? Neurol. Psychiatry Brain Res. 2012, 18, 42. [Google Scholar] [CrossRef]
- Bransfield, R.C. Suicide and Lyme and associated diseases. Neuropsychiatr. Dis. Treat. 2017, 13, 1575–1587. [Google Scholar] [CrossRef] [PubMed]
- Bransfield, R.C. Aggressiveness, violence, homicidality, homicide, and Lyme disease. Neuropsychiatr. Dis. Treat. 2018, 14, 693–713. [Google Scholar] [CrossRef] [PubMed]
- Bransfield, R.C. Neuropsychiatric Lyme Borreliosis: An Overview with a Focus on a Specialty Psychiatrist’s Clinical Practice. Healthcare 2018, 6, 104. [Google Scholar] [CrossRef] [PubMed]
- Eckman, E.A.; Pacheco-Quinto, J.; Herdt, A.R.; Halperin, J. Neuroimmunomodulators in Neuroborreliosis and Lyme Encephalopathy. Clin. Infect. Dis. 2018, 67, 80–88. [Google Scholar] [CrossRef]
- De Bellis, M.D. The Psychobiology of Neglect. Child Maltreatment 2005, 10, 150–172. [Google Scholar] [CrossRef]
- Felitti, V.J. The Relation Between Adverse Childhood Experiences and Adult Health: Turning Gold into Lead. Perm. J. 2002, 6, 44–47. [Google Scholar]
- O’Donovan, A.; Neylan, T.C.; Metzler, T.; Cohen, B.E. Lifetime exposure to traumatic psychological stress is associated with elevated inflammation in the Heart and Soul Study. Brain Behav. Immun. 2012, 26, 642–649. [Google Scholar] [CrossRef]
- Dantzer, R.; O’Connor, J.C.; Freund, G.G.; Johnson, R.W.; Kelley, K.W. From inflammation to sickness and depression: When the immune system subjugates the brain. Nat. Rev. Neurosci. 2008, 9, 46–56. [Google Scholar] [CrossRef]
- Eisenberger, N.I.; Berkman, E.T.; Inagaki, T.; Rameson, L.T.; Mashal, N.M.; Irwin, M. Inflammation-Induced Anhedonia: Endotoxin Reduces Ventral Striatum Responses to Reward. Biol. Psychiatry 2010, 68, 748–754. [Google Scholar] [CrossRef]
- Raison, C.L.; Miller, A.H. Role of Inflammation in Depression: Implications for Phenomenology, Pathophysiology and Treatment. Mod. Trends Pharm. 2013, 28, 33–48. [Google Scholar] [CrossRef]
- Harris, T.B.; Ferrucci, L.; Tracy, R.P.; Corti, M.C.; Wacholder, S.; Ettinger, W.H., Jr.; Heimovitz, H.; Cohen, H.J.; Wallace, R. Associations of elevated Interleukin-6 and C-Reactive protein levels with mortality in the elderly. Am. J. Med. 1999, 106, 506–512. [Google Scholar] [CrossRef]
- Libby, P. Inflammation in atherosclerosis. Nature 2002, 420, 868–874. [Google Scholar] [CrossRef] [PubMed]
- O’Donovan, A.; Cohen, B.E.; Seal, K.H.; Bertenthal, D.; Margaretten, M.; Nishimi, K.; Neylan, T.C. Elevated Risk for Autoimmune Disorders in Iraq and Afghanistan Veterans with Posttraumatic Stress Disorder. Biol. Psychiatry 2014, 77, 365–374. [Google Scholar] [CrossRef] [PubMed]
- Eraly, S.A.; Nievergelt, C.; Maihofer, A.X.; Barkauskas, D.A.; Biswas, N.; Agorastos, A.; O’Connor, D.T.; Baker, D.G. Assessment of Plasma C-Reactive Protein as a Biomarker of Posttraumatic Stress Disorder Risk. JAMA Psychiatry 2014, 71, 423–431. [Google Scholar] [CrossRef]
- Heath, N.M.; Chesney, S.A.; Gerhart, J.I.; Goldsmith, R.E.; Luborsky, J.L.; Stevens, N.R.; Hobfoll, S.E. Interpersonal violence, PTSD, and inflammation: Potential psychogenic pathways to higher C-reactive protein levels. Cytokine 2013, 63, 172–178. [Google Scholar] [CrossRef]
- Bob, P.; Raboch, J.; Maes, M.; Susta, M.; Pavlat, J.; Jasova, D.; Vevera, J.; Uhrova, J.; Benáková, H.; Zima, T. Depression, traumatic stress and interleukin-6. J. Affect. Disord. 2010, 120, 231–234. [Google Scholar] [CrossRef]
- Gill, J.; Vythilingam, M.; Page, G.G. Low cortisol, high DHEA, and high levels of stimulated TNF-α, and IL-6 in women with PTSD. J. Traum. Stress 2008, 21, 530–539. [Google Scholar] [CrossRef]
- Ritov, G.; Ardi, Z.; Richter-Levin, G. Differential activation of amygdala, dorsal and ventral hippocampus following an exposure to a reminder of underwater trauma. Front. Behav. Neurosci. 2014, 8, 18. [Google Scholar] [CrossRef]
- Zimmerman, G.; Shaltiel, G.; Barbash, S.; Cohen, J.; Gasho, C.J.; Shenhar-Tsarfaty, S.; Shalev, H.; Berliner, S.A.; Shelef, I.; Shoham, S.; et al. Post-traumatic anxiety associates with failure of the innate immune receptor TLR9 to evade the pro-inflammatory NFκB pathway. Transl. Psychiatry 2012, 2, e78. [Google Scholar] [CrossRef]
- Lim, L.; Hart, H.; Howells, H.; Mehta, M.A.; Simmons, A.; Mirza, K.; Rubia, K. Altered white matter connectivity in young people exposed to childhood abuse: A tract-based spatial statistics (TBSS) and tractography study. J. Psychiatry Neurosci. 2019, 44, E11–E20. [Google Scholar] [CrossRef] [PubMed]
- Lim, L.; Howells, H.; Radua, J.; Rubia, K. Aberrant structural connectivity in childhood maltreatment: A meta-analysis. Neurosci. Biobehav. Rev. 2020, 116, 406–414. [Google Scholar] [CrossRef] [PubMed]
- Tendolkar, I.; Mårtensson, J.; Kühn, S.; Klumpers, F.; Fernández, G. Physical neglect during childhood alters white matter connectivity in healthy young males. Hum. Brain Mapp. 2017, 39, 1283–1290. [Google Scholar] [CrossRef] [PubMed]
- Tanti, A.; Kim, J.J.; Wakid, M.; Davoli, M.-A.; Turecki, G.; Mechawar, N. Child abuse associates with an imbalance of oligodendrocyte-lineage cells in ventromedial prefrontal white matter. Mol. Psychiatry 2017, 23, 2018–2028. [Google Scholar] [CrossRef] [PubMed]
- Lutz, P.-E.; Tanti, A.; Gasecka, A.; Barnett-Burns, S.; Kim, J.J.; Zhou, Y.; Chen, G.G.; Wakid, M.; Shaw, M.; Almeida, D.; et al. Association of a History of Child Abuse With Impaired Myelination in the Anterior Cingulate Cortex: Convergent Epigenetic, Transcriptional, and Morphological Evidence. Am. J. Psychiatry 2017, 174, 1185–1194. [Google Scholar] [CrossRef]
- Kato, T.A.; Kanba, S. Are microglia minding us? Digging up the unconscious mind-brain relationship from a neuropsychoanalytic approach. Front. Psychol. 2013, 7, 13. [Google Scholar]
- Chen, M.A.; LeRoy, A.S.; Majd, M.; Chen, J.Y.; Brown, R.L.; Christian, L.M.; Fagundes, C.P. Immune and Epigenetic Pathways Linking Childhood Adversity and Health Across the Lifespan. Front. Psychol. 2021, 12, 788351. [Google Scholar] [CrossRef]
- Jiang, S.; Postovit, L.; Cattaneo, A.; Binder, E.B.; Aitchison, K.J. Epigenetic Modifications in Stress Response Genes Associated With Childhood Trauma. Front. Psychiatry 2019, 10, 808. [Google Scholar] [CrossRef]
- Danese, A.; Lewis, S. Psychoneuroimmunology of Early-Life Stress: The Hidden Wounds of Childhood Trauma? Neuropsychopharmacology 2016, 42, 99–114. [Google Scholar] [CrossRef]
- Bransfield, R. The psychoimmunology of Lyme and associated diseases. Neurol. Psychiatry Brain Res. 2014, 20, 8. [Google Scholar] [CrossRef]
- Moldofsky, H. Nonrestorative Sleep, Musculoskeletal Pain, Fatigue in Rheumatic Disorders, and Allied Syndromes: A Historical Perspective. In Sleep Medicine; Springer: New York, NY, USA, 2015; pp. 423–431. [Google Scholar] [CrossRef]
- Brady, K.; Pearlstein, T.; Asnis, G.M.; Baker, D.; Rothbaum, B.; Sikes, C.R.; Farfel, G.M. Efficacy and safety of sertraline treatment of posttraumatic stress disorder: A randomized controlled trial. JAMA 2020, 283, 1837–1844. [Google Scholar] [CrossRef] [PubMed]
- Marshall, R.D.; Beebe, K.L.; Oldham, M.; Zaninelli, R. Efficacy and Safety of Paroxetine Treatment for Chronic PTSD: A Fixed-Dose, Placebo-Controlled Study. Am. J. Psychiatry 2001, 158, 1982–1988. [Google Scholar] [CrossRef] [PubMed]
- Hudson, S.M.; Whiteside, T.E.; Lorenz, R.A.; Wargo, K.A. Prazosin for the Treatment of Nightmares Related to Posttraumatic Stress Disorder. Prim. Care Companion CNS Disord. 2012, 14, PCC.11r01222. [Google Scholar] [CrossRef]
- Berlant, J. Topiramate as a therapy for chronic posttraumatic stress disorder. Psychiatry Edgmont 2006, 3, 40–45. [Google Scholar]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bransfield, R.C. Adverse Childhood Events, Post-Traumatic Stress Disorder, Infectious Encephalopathies and Immune-Mediated Disease. Healthcare 2022, 10, 1127. https://doi.org/10.3390/healthcare10061127
Bransfield RC. Adverse Childhood Events, Post-Traumatic Stress Disorder, Infectious Encephalopathies and Immune-Mediated Disease. Healthcare. 2022; 10(6):1127. https://doi.org/10.3390/healthcare10061127
Chicago/Turabian StyleBransfield, Robert C. 2022. "Adverse Childhood Events, Post-Traumatic Stress Disorder, Infectious Encephalopathies and Immune-Mediated Disease" Healthcare 10, no. 6: 1127. https://doi.org/10.3390/healthcare10061127
APA StyleBransfield, R. C. (2022). Adverse Childhood Events, Post-Traumatic Stress Disorder, Infectious Encephalopathies and Immune-Mediated Disease. Healthcare, 10(6), 1127. https://doi.org/10.3390/healthcare10061127






