Dietary Intake and Genetic Background Influence Vitamin Needs during Pregnancy
Abstract
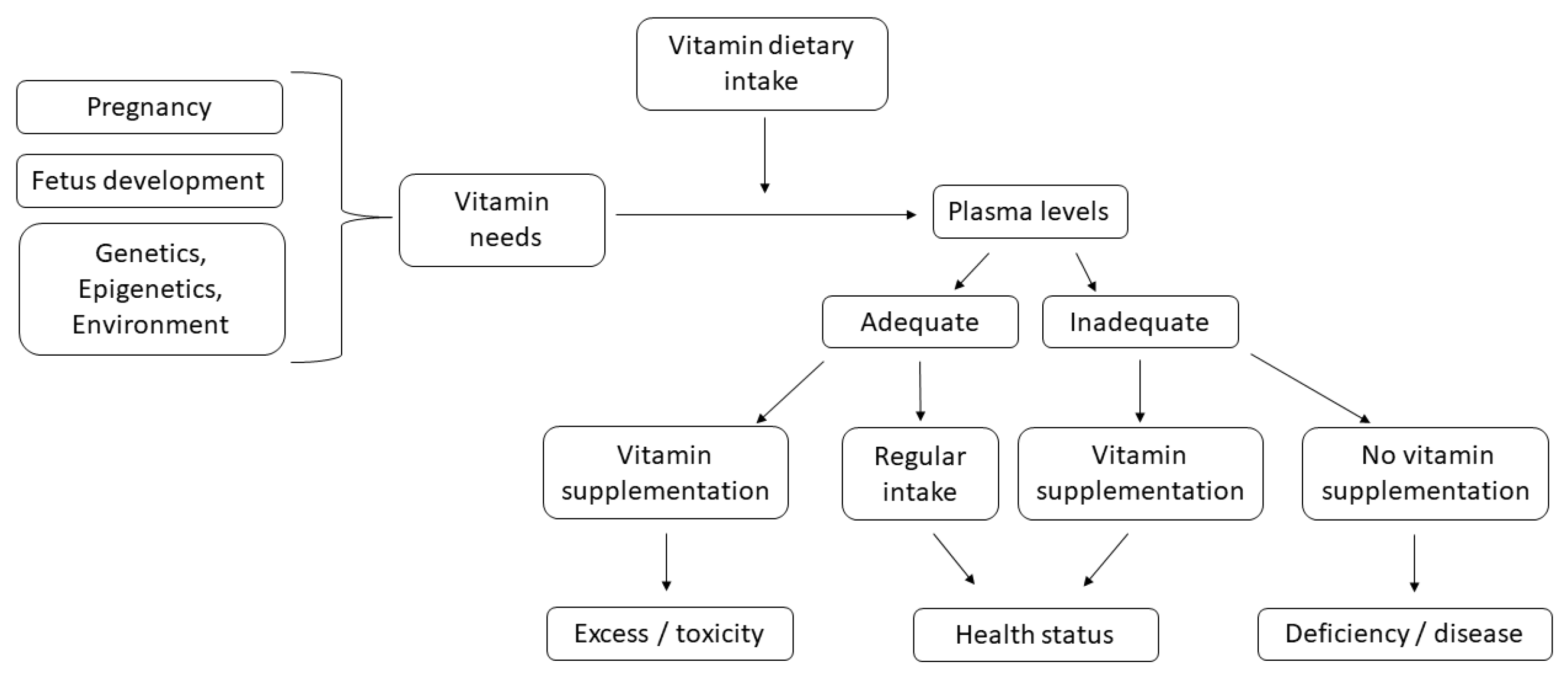
:1. Introduction
2. Relevant Findings on Fat-Soluble Vitamins
2.1. Vitamin A
2.2. Vitamin E
2.3. Vitamin D
2.4. Vitamin K
3. Relevant Findings on Water-Soluble Vitamins
3.1. Folic Acid
3.2. Vitamin B12 and Vitamin B1
3.3. Vitamin C
3.4. Other Water-Soluble Vitamins
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Shenkin, A. Micronutrients in health and disease. Postgrad. Med. J. 2006, 82, 559–567. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chauhan, P.S.; Yadav, D. Dietary Nutrients and Prevention of Alzheimer’s disease. CNS Neurol. Disord. Drug Targets 2021, 11, 217–227. [Google Scholar] [CrossRef] [PubMed]
- Amrein, K.; Scherkl, M.; Hoffmann, M.; Neuwersch-Sommeregger, S.; Kostenberger, M.; Tmava Berisha, A.; Martucci, G.; Pilz, S.; Malle, O. Vitamin D deficiency 2.0: An update on the current status worldwide. Eur. J. Clin. Nutr. 2020, 74, 1498–1513. [Google Scholar] [CrossRef]
- Ford, K.L.; Jorgenson, D.J.; Landry, E.J.L.; Whiting, S.J. Vitamin and mineral supplement use in medically complex, community-living, older adults. Appl. Physiol. Nutr. Metab. 2019, 44, 450–453. [Google Scholar] [CrossRef] [PubMed]
- Wacker, M.; Holick, M.F. Vitamin D—Effects on skeletal and extraskeletal health and the need for supplementation. Nutrients 2013, 5, 111–148. [Google Scholar] [CrossRef] [Green Version]
- Koury, M.J.; Ponka, P. New insights into erythropoiesis: The roles of folate, vitamin B12, and iron. Annu. Rev. Nutr. 2004, 24, 105–131. [Google Scholar] [CrossRef] [PubMed]
- Imam, M.U.; Zhang, S.; Ma, J.; Wang, H.; Wang, F. Antioxidants Mediate Both Iron Homeostasis and Oxidative Stress. Nutrients 2017, 9, 671. [Google Scholar] [CrossRef]
- Maggini, S.; Pierre, A.; Calder, P.C. Immune Function and Micronutrient Requirements Change over the Life Course. Nutrients 2018, 10, 1531. [Google Scholar] [CrossRef] [Green Version]
- Alpert, P.T. The Role of Vitamins and Minerals on the Immune System. Home Health Care Manag. Pract. 2017, 29, 199–202. [Google Scholar] [CrossRef]
- Han, T.; Dong, J.; Zhang, J.; Zhang, C.; Wang, Y.; Zhang, Z.; Xiang, M. Nutrient supplementation among pregnant women in China: An observational study. Public Health Nutr. 2021, 1–19. [Google Scholar] [CrossRef]
- Balogun, O.O.; da Silva Lopes, K.; Ota, E.; Takemoto, Y.; Rumbold, A.; Takegata, M.; Mori, R. Vitamin supplementation for preventing miscarriage. Cochrane Database Syst. Rev. 2016, CD004073. [Google Scholar] [CrossRef] [PubMed]
- Rose, E.G.; Murphy, M.S.Q.; Erwin, E.; Muldoon, K.A.; Harvey, A.L.J.; Rennicks White, R.; MacFarlane, A.J.; Wen, S.W.; Walker, M.C. Gestational Folate and Folic Acid Intake among Women in Canada at Higher Risk of Pre-Eclampsia. J Nutr. 2021, 151, 1976–1982. [Google Scholar] [CrossRef] [PubMed]
- Hayes, D.P. Nutritional hormesis. Eur. J. Clin. Nutr. 2007, 61, 147–159. [Google Scholar] [CrossRef] [PubMed]
- Clarke, M.W.; Burnett, J.R.; Croft, K.D. Vitamin E in human health and disease. Crit. Rev. Clin. Lab. Sci. 2008, 45, 417–450. [Google Scholar] [CrossRef] [PubMed]
- Frayne, J.; Hauck, Y.; Nguyen, T.; Liira, H.; Morgan, V.A. Reproductive planning, vitamin knowledge and use, and lifestyle risks of women attending pregnancy care with a severe mental illness. Scand. J. Prim. Health 2021, 39, 60–66. [Google Scholar] [CrossRef]
- Yamasaki, A.; Kawasaki, Y.; Takeda, K.; Harada, T.; Hasegawa, Y.; Fukushima, T.; Okazaki, R.; Makino, H.; Funaki, Y.; Sueda, Y.; et al. Relationship between Oxidative Stress, Physical Activity, and Vitamin Intake in Patients with Asthma. Yonago Acta Med. 2017, 60, 86–93. [Google Scholar] [CrossRef] [Green Version]
- Gao, F.; Guo, F.F.; Zhang, Y.D.; Yuan, Y.F.; Chen, D.J.; Bai, G.Q. Vitamin A, D, and E Levels and Reference Ranges for Pregnant Women: A Cross-Sectional Study 2017–2019. Front. Nutr. 2021, 8, 628902. [Google Scholar] [CrossRef]
- Bastos Maia, S.; Rolland Souza, A.S.; Costa Caminha, M.F.; Lins da Silva, S.; Callou Cruz, R.; Carvalho Dos Santos, C.; Batista Filho, M. Vitamin A and Pregnancy: A Narrative Review. Nutrients 2019, 11, 681. [Google Scholar] [CrossRef] [Green Version]
- Bendik, I.; Friedel, A.; Roos, F.F.; Weber, P.; Eggersdorfer, M. Vitamin D: A critical and essential micronutrient for human health. Front. Physiol. 2014, 5, 248. [Google Scholar] [CrossRef]
- Pilz, S.; Zittermann, A.; Obeid, R.; Hahn, A.; Pludowski, P.; Trummer, C.; Lerchbaum, E.; Perez-Lopez, F.R.; Karras, S.N.; Marz, W. The Role of Vitamin D in Fertility and during Pregnancy and Lactation: A Review of Clinical Data. Int. J. Environ. Res. Public Health 2018, 15, 2241. [Google Scholar] [CrossRef] [Green Version]
- Jans, G.; Guelinckx, I.; Voets, W.; Galjaard, S.; Van Haard, P.M.; Vansant, G.M.; Devlieger, R. Vitamin K1 monitoring in pregnancies after bariatric surgery: A prospective cohort study. Surg. Obes. Relat. Dis. 2014, 10, 885–890. [Google Scholar] [CrossRef] [PubMed]
- Shearer, M.J. Vitamin K in parenteral nutrition. Gastroenterology 2009, 137, S105–S118. [Google Scholar] [CrossRef] [PubMed]
- Strobel, M.; Tinz, J.; Biesalski, H.K. The importance of beta-carotene as a source of vitamin A with special regard to pregnant and breastfeeding women. Eur. J. Nutr. 2007, 46, 1–20. [Google Scholar] [CrossRef] [PubMed]
- Yee, M.M.F.; Chin, K.Y.; Ima-Nirwana, S.; Wong, S.K. Vitamin A and Bone Health: A Review on Current Evidence. Molecules 2021, 26, 1757. [Google Scholar] [CrossRef]
- Ross, A.C. Vitamin A and retinoic acid in T cell-related immunity. Am. J. Clin. Nutr. 2012, 96, 1166S–1172S. [Google Scholar] [CrossRef]
- Chen, H.; Qian, N.F.; Yan, L.Y.; Jiang, H.Q. Role of serum vitamin A and E in pregnancy. Exp. Ther. Med. 2018, 16, 5185–5189. [Google Scholar] [CrossRef] [Green Version]
- McCauley, M.E.; van den Broek, N.; Dou, L.X.; Othman, M. Vitamin A supplementation during pregnancy for maternal and newborn outcomes. Cochrane Database Syst. Rev. 2015, 10, CD008666. [Google Scholar] [CrossRef]
- Klamt, F.; Roberto de Oliveira, M.; Moreira, J.C. Retinol induces permeability transition and cytochrome c release from rat liver mitochondria. Biochim. Biophys. Acta 2005, 1726, 14–20. [Google Scholar] [CrossRef]
- Schmitt, C.; Domange, B.; Torrents, R.; de Haro, L.; Simon, N. Hypervitaminosis A Following the Ingestion of Fish Liver: Report on 3 Cases from the Poison Control Center in Marseille. Wilderness Environ. Med. 2020, 31, 454–456. [Google Scholar] [CrossRef]
- Suzuki, M.; Wang, T.; Garretto, D.; Isasi, C.R.; Cardoso, W.V.; Greally, J.M.; Quadro, L. Disproportionate Vitamin A Deficiency in Women of Specific Ethnicities Linked to Differences in Allele Frequencies of Vitamin A-Related Polymorphisms. Nutrients 2021, 13, 1743. [Google Scholar] [CrossRef]
- Pirazzi, C.; Valenti, L.; Motta, B.M.; Pingitore, P.; Hedfalk, K.; Mancina, R.M.; Burza, M.A.; Indiveri, C.; Ferro, Y.; Montalcini, T.; et al. PNPLA3 has retinyl-palmitate lipase activity in human hepatic stellate cells. Hum. Mol. Genet. 2014, 23, 4077–4085. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Mondul, A.M.; Yu, K.; Wheeler, W.; Zhang, H.; Weinstein, S.J.; Major, J.M.; Cornelis, M.C.; Mannisto, S.; Hazra, A.; Hsing, A.W.; et al. Genome-wide association study of circulating retinol levels. Hum. Mol. Genet. 2011, 20, 4724–4731. [Google Scholar] [CrossRef] [PubMed]
- Abraham, A.; Kattoor, A.J.; Saldeen, T.; Mehta, J.L. Vitamin E and its anticancer effects. Crit. Rev. Food Sci. Nutr. 2019, 59, 2831–2838. [Google Scholar] [CrossRef] [PubMed]
- Boskovic, R.; Gargaun, L.; Oren, D.; Djulus, J.; Koren, G. Pregnancy outcome following high doses of Vitamin E supplementation. Reprod. Toxicol. 2005, 20, 85–88. [Google Scholar] [CrossRef] [PubMed]
- Rumbold, A.; Ota, E.; Hori, H.; Miyazaki, C.; Crowther, C.A. Vitamin E supplementation in pregnancy. Cochrane Database. Syst. Rev. 2015, 2016, CD004069. [Google Scholar] [CrossRef]
- Janciauskiene, S. The Beneficial Effects of Antioxidants in Health and Diseases. Chronic Obstr. Pulm. Dis. J. COPD Found. 2020, 7, 182–202. [Google Scholar] [CrossRef]
- Kondo, A.; Matsuo, T.; Morota, N.; Kondo, A.S.; Okai, I.; Fukuda, H. Neural tube defects: Risk factors and preventive measures. Congenit. Anom. 2017, 57, 150–156. [Google Scholar] [CrossRef]
- Urena-Torres, P.; Souberbielle, J.C. Pharmacologic role of vitamin D natural products. Curr. Vasc. Pharmacol. 2014, 12, 278–285. [Google Scholar] [CrossRef]
- Caccamo, D.; Ricca, S.; Curro, M.; Ientile, R. Health Risks of Hypovitaminosis D: A Review of New Molecular Insights. Int. J. Mol. Sci. 2018, 19, 892. [Google Scholar] [CrossRef] [Green Version]
- Naidoo, Y.; Moodley, J.; Ramsuran, V.; Naicker, T. Polymorphisms within vitamin D binding protein gene within a Preeclamptic South African population. Hypertens. Pregnancy 2019, 38, 260–267. [Google Scholar] [CrossRef]
- Velaphi, S.C.; Izu, A.; Madhi, S.A.; Pettifor, J.M. Maternal and neonatal vitamin D status at birth in black South Africans. SAMJ S. Afr. Med. J. 2019, 109, 807–813. [Google Scholar] [CrossRef] [PubMed]
- van der Pligt, P.; Willcox, J.; Szymlek-Gay, E.A.; Murray, E.; Worsley, A.; Daly, R.M. Associations of Maternal Vitamin D Deficiency with Pregnancy and Neonatal Complications in Developing Countries: A Systematic Review. Nutrients 2018, 10, 640. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gernand, A.D.; Schulze, K.J.; Stewart, C.P.; West, K.P., Jr.; Christian, P. Micronutrient deficiencies in pregnancy worldwide: Health effects and prevention. Nat. Rev. Endocrinol. 2016, 12, 274–289. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Makitie, O. Causes, mechanisms and management of paediatric osteoporosis. Nat. Rev. Rheumatol. 2013, 9, 465–475. [Google Scholar] [CrossRef] [PubMed]
- Paterson, C.R.; Ayoub, D. Congenital rickets due to vitamin D deficiency in the mothers. Clin. Nutr. 2015, 34, 793–798. [Google Scholar] [CrossRef]
- Ketha, H.; Thacher, T.D.; Oberhelman, S.S.; Fischer, P.R.; Singh, R.J.; Kumar, R. Comparison of the effect of daily versus bolus dose maternal vitamin D3 supplementation on the 24,25-dihydroxyvitamin D3 to 25-hydroxyvitamin D3 ratio. Bone 2018, 110, 321–325. [Google Scholar] [CrossRef]
- Woodrow, J.P.; Sharpe, C.J.; Fudge, N.J.; Hoff, A.O.; Gagel, R.F.; Kovacs, C.S. Calcitonin plays a critical role in regulating skeletal mineral metabolism during lactation. Endocrinology 2006, 147, 4010–4021. [Google Scholar] [CrossRef] [Green Version]
- Meher, S.; Gibbons, N.; DasGupta, R. Renal stones in pregnancy. Obstet. Med. 2014, 7, 103–110. [Google Scholar] [CrossRef]
- Beer, T.M.; Lemmon, D.; Lowe, B.A.; Henner, W.D. High-dose weekly oral calcitriol in patients with a rising PSA after prostatectomy or radiation for prostate carcinoma. Cancer 2003, 97, 1217–1224. [Google Scholar] [CrossRef]
- Trump, D.L.; Potter, D.M.; Muindi, J.; Brufsky, A.; Johnson, C.S. Phase II trial of high-dose, intermittent calcitriol (1,25 dihydroxyvitamin D3) and dexamethasone in androgen-independent prostate cancer. Cancer 2006, 106, 2136–2142. [Google Scholar] [CrossRef]
- Burt, L.A.; Billington, E.O.; Rose, M.S.; Kremer, R.; Hanley, D.A.; Boyd, S.K. Reply to Burt LA; et al.: Adverse Effects of High-Dose Vitamin D Supplementation on Volumetric Bone Density Are Greater in Females Than Males. J. Bone Miner. Res. 2021, 36, 1417–1418. [Google Scholar] [CrossRef] [PubMed]
- Rajwar, E.; Parsekar, S.S.; Venkatesh, B.T.; Sharma, Z. Effect of vitamin A, calcium and vitamin D fortification and supplementation on nutritional status of women: An overview of systematic reviews. Syst. Rev. 2020, 9, 248. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Jiang, Y.F. Natural compounds as anticancer agents: Experimental evidence. World J. Exp. Med. 2012, 2, 45–57. [Google Scholar] [CrossRef] [PubMed]
- Hathcock, J.N.; Shao, A.; Vieth, R.; Heaney, R. Risk assessment for vitamin D. Am. J. Clin. Nutr. 2007, 85, 6–18. [Google Scholar] [CrossRef] [Green Version]
- Rostami, M.; Simbar, M.; Amiri, M.; Bidhendi-Yarandi, R.; Hosseinpanah, F.; Ramezani Tehrani, F. The optimal cut-off point of vitamin D for pregnancy outcomes using a generalized additive model. Clin. Nutr. 2021, 40, 2145–2153. [Google Scholar] [CrossRef]
- Zhu, J.G.; DeLuca, H.F. Vitamin D 25-hydroxylase—Four decades of searching, are we there yet? Arch. Biochem. Biophys. 2012, 523, 30–36. [Google Scholar] [CrossRef]
- Masuda, S.; Jones, G. Promise of vitamin D analogues in the treatment of hyperproliferative conditions. Mol. Cancer Ther. 2006, 5, 797–808. [Google Scholar] [CrossRef] [Green Version]
- Traglia, M.; Windham, G.C.; Pearl, M.; Poon, V.; Eyles, D.; Jones, K.L.; Lyall, K.; Kharrazi, M.; Croen, L.A.; Weiss, L.A. Genetic Contributions to Maternal and Neonatal Vitamin D Levels. Genetics 2020, 214, 1091–1102. [Google Scholar] [CrossRef] [Green Version]
- Wang, T.J.; Zhang, F.; Richards, J.B.; Kestenbaum, B.; van Meurs, J.B.; Berry, D.; Kiel, D.P.; Streeten, E.A.; Ohlsson, C.; Koller, D.L.; et al. Common genetic determinants of vitamin D insufficiency: A genome-wide association study. Lancet 2010, 376, 180–188. [Google Scholar] [CrossRef] [Green Version]
- Uitterlinden, A.G.; Fang, Y.; Van Meurs, J.B.; Pols, H.A.; Van Leeuwen, J.P. Genetics and biology of vitamin D receptor polymorphisms. Gene 2004, 338, 143–156. [Google Scholar] [CrossRef] [Green Version]
- Manzon, L.; Altarescu, G.; Tevet, A.; Schimmel, M.S.; Elstein, D.; Samueloff, A.; Grisaru-Granovsky, S. Vitamin D receptor polymorphism FokI is associated with spontaneous idiopathic preterm birth in an Israeli population. Eur. J. Obstet. Gynecol. Reprod. Biol. 2014, 177, 84–88. [Google Scholar] [CrossRef] [PubMed]
- Swamy, G.K.; Garrett, M.E.; Miranda, M.L.; Ashley-Koch, A.E. Maternal vitamin D receptor genetic variation contributes to infant birthweight among black mothers. Am. J. Med. Genet. Part A 2011, 155A, 1264–1271. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Rosenfeld, T.; Salem, H.; Altarescu, G.; Grisaru-Granovsky, S.; Tevet, A.; Birk, R. Maternal-fetal vitamin D receptor polymorphisms significantly associated with preterm birth. Arch. Gynecol. Obstet. 2017, 296, 215–222. [Google Scholar] [CrossRef] [PubMed]
- Baczynska-Strzecha, M.; Kalinka, J. Influence of Apa1 (rs7975232), Taq1 (rs731236) and Bsm1 (rs154410) polymorphisms of vitamin D receptor on preterm birth risk in the Polish population. Ginekol. Pol. 2016, 87, 763–768. [Google Scholar] [CrossRef] [Green Version]
- Motohashi, Y.; Yamada, S.; Yanagawa, T.; Maruyama, T.; Suzuki, R.; Niino, M.; Fukazawa, T.; Kasuga, A.; Hirose, H.; Matsubara, K.; et al. Vitamin D receptor gene polymorphism affects onset pattern of type 1 diabetes. J. Clin. Endocrinol. Metab. 2003, 88, 3137–3140. [Google Scholar] [CrossRef] [Green Version]
- Tizaoui, K.; Kaabachi, W.; Hamzaoui, A.; Hamzaoui, K. Contribution of VDR polymorphisms to type 1 diabetes susceptibility: Systematic review of case-control studies and meta-analysis. J. Steroid Biochem. Mol. Biol. 2014, 143, 240–249. [Google Scholar] [CrossRef]
- Oh, J.Y.; Barrett-Connor, E. Association between vitamin D receptor polymorphism and type 2 diabetes or metabolic syndrome in community-dwelling older adults: The Rancho Bernardo Study. Metabolism 2002, 51, 356–359. [Google Scholar] [CrossRef]
- Zhou, Q.; Wen, S.; Liu, M.; Zhang, S.; Jin, X.; Liu, A. Association between Gene Polymorphisms of Vitamin D Receptor and Gestational Diabetes Mellitus: A Systematic Review and Meta-Analysis. Int. J. Environ. Res. Public Health 2020, 18, 205. [Google Scholar] [CrossRef]
- Knabl, J.; Vattai, A.; Ye, Y.; Jueckstock, J.; Hutter, S.; Kainer, F.; Mahner, S.; Jeschke, U. Role of Placental VDR Expression and Function in Common Late Pregnancy Disorders. Int. J. Mol. Sci. 2017, 18, 2340. [Google Scholar] [CrossRef] [Green Version]
- Yu, F.; Cui, L.L.; Li, X.; Wang, C.J.; Ba, Y.; Wang, L.; Li, J.; Li, C.; Dai, L.P.; Li, W.J. The genetic polymorphisms in vitamin D receptor and the risk of type 2 diabetes mellitus: An updated meta-analysis. Asia Pac. J. Clin. Nutr. 2016, 25, 614–624. [Google Scholar] [CrossRef]
- Shi, A.; Wen, J.; Liu, G.; Liu, H.; Fu, Z.; Zhou, J.; Zhu, Y.; Liu, Y.; Guo, X.; Xu, J. Genetic variants in vitamin D signaling pathways and risk of gestational diabetes mellitus. Oncotarget 2016, 7, 67788–67795. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sandrim, V.C.; Palei, A.C.; Luizon, M.R.; Izidoro-Toledo, T.C.; Cavalli, R.C.; Tanus-Santos, J.E. eNOS haplotypes affect the responsiveness to antihypertensive therapy in preeclampsia but not in gestational hypertension. Pharm. J. 2010, 10, 40–45. [Google Scholar] [CrossRef] [PubMed]
- Ndiaye, N.C.; Said, E.S.; Stathopoulou, M.G.; Siest, G.; Tsai, M.Y.; Visvikis-Siest, S. Epistatic study reveals two genetic interactions in blood pressure regulation. BMC Med. Genet. 2013, 14, 2. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wang, L.; Ma, J.; Manson, J.E.; Buring, J.E.; Gaziano, J.M.; Sesso, H.D. A prospective study of plasma vitamin D metabolites, vitamin D receptor gene polymorphisms, and risk of hypertension in men. Eur. J. Nutr. 2013, 52, 1771–1779. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Swapna, N.; Vamsi, U.M.; Usha, G.; Padma, T. Risk conferred by FokI polymorphism of vitamin D receptor (VDR) gene for essential hypertension. Indian J. Hum. Genet. 2011, 17, 201–206. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Caccamo, D.; Cannata, A.; Ricca, S.; Catalano, L.M.; Montalto, A.F.; Alibrandi, A.; Ercoli, A.; Granese, R. Role of Vitamin-D Receptor (VDR) single nucleotide polymorphisms in gestational hypertension development: A case-control study. PLoS ONE 2020, 15, e0239407. [Google Scholar] [CrossRef] [PubMed]
- Karras, S.N.; Dursun, E.; Alaylioglu, M.; Gezen-Ak, D.; Annweiler, C.; Al Anouti, F.; Fakhoury, H.M.A.; Bais, A.; Kiortsis, D. Investigating the Role of Functional Polymorphism of Maternal and Neonatal Vitamin D Binding Protein in the Context of 25-Hydroxyvitamin D Cutoffs as Determinants of Maternal-Neonatal Vitamin D Status Profiles in a Sunny Mediterranean Region. Nutrients 2021, 13, 3082. [Google Scholar] [CrossRef]
- Shahrook, S.; Ota, E.; Hanada, N.; Sawada, K.; Mori, R. Vitamin K supplementation during pregnancy for improving outcomes: A systematic review and meta-analysis. Sci. Rep. 2018, 8, 11459. [Google Scholar] [CrossRef] [Green Version]
- Booth, S.L. Vitamin K: Food composition and dietary intakes. Food Nutr. Res. 2012, 56, 5505. [Google Scholar] [CrossRef] [Green Version]
- Imbrescia, K.; Moszczynski, Z. Vitamin K. In StatPearls; StatPearls Publishing: Treasure Island, FL, USA, 2021. [Google Scholar]
- Weston, B.W.; Monahan, P.E. Familial deficiency of vitamin K-dependent clotting factors. Haemophilia 2008, 14, 1209–1213. [Google Scholar] [CrossRef] [Green Version]
- Zhang, B.; Ginsburg, D. Familial multiple coagulation factor deficiencies: New biologic insight from rare genetic bleeding disorders. J. Thromb. Haemost. 2004, 2, 1564–1572. [Google Scholar] [CrossRef] [PubMed]
- Hafizi, S.; Dahlback, B. Gas6 and protein S. Vitamin K-dependent ligands for the Axl receptor tyrosine kinase subfamily. FEBS J. 2006, 273, 5231–5244. [Google Scholar] [CrossRef] [PubMed]
- Ozakpinar, O.B.; Sahin, S.; Verimli, N.; Simsek, G.G.; Maurer, A.M.; Eroglu, M.; Tetik, S.; Uras, F. Association between the growth arrest-specific 6 (Gas6) gene polymorphism c.834 + 7G>A and preeclampsia. J. Matern. Fetal Neonatal. Med. 2016, 29, 1149–1153. [Google Scholar] [CrossRef] [PubMed]
- Devalia, V.; Hamilton, M.S.; Molloy, A.M.; British Committee for Standards in Haematology. Guidelines for the diagnosis and treatment of cobalamin and folate disorders. Br. J. Haematol 2014, 166, 496–513. [Google Scholar] [CrossRef] [PubMed]
- Stabler, S.P. Vitamin B12 deficiency. N. Engl. J. Med. 2013, 368, 2041–2042. [Google Scholar] [CrossRef] [PubMed]
- Frank, L.L. Thiamin in Clinical Practice. JPEN J. Parenter. Enteral. Nutr. 2015, 39, 503–520. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Pincemail, J.; Vanbelle, S.; Degrune, F.; Cheramy-Bien, J.P.; Charlier, C.; Chapelle, J.P.; Giet, D.; Collette, G.; Albert, A.; Defraigne, J.O. Lifestyle Behaviours and Plasma Vitamin C and beta-Carotene Levels from the ELAN Population (Liege, Belgium). J. Nutr. Metab. 2011, 2011, 494370. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Refsum, H. Folate, vitamin B12 and homocysteine in relation to birth defects and pregnancy outcome. Br. J. Nutr. 2001, 85, S109–S113. [Google Scholar] [CrossRef]
- Vanhees, K.; Vonhogen, I.G.C.; van Schooten, F.J.; Godschalk, R.W.L. You are what you eat, and so are your children: The impact of micronutrients on the epigenetic programming of offspring. Cell Mol. Life Sci. 2014, 71, 271–285. [Google Scholar] [CrossRef]
- Devnath, G.P.; Kumaran, S.; Rajiv, R.; Shaha, K.K.; Nagaraj, A. Fatal Folic Acid Toxicity in Humans. J. Forensic Sci. 2017, 62, 1668–1670. [Google Scholar] [CrossRef]
- De Martinis, M.; Sirufo, M.M.; Nocelli, C.; Fontanella, L.; Ginaldi, L. Hyperhomocysteinemia is Associated with Inflammation, Bone Resorption, Vitamin B12 and Folate Deficiency and MTHFR C677T Polymorphism in Postmenopausal Women with Decreased Bone Mineral Density. Int. J. Environ. Res. Public Health 2020, 17, 4260. [Google Scholar] [CrossRef] [PubMed]
- Chen, H.; Yang, X.; Lu, M. Methylenetetrahydrofolate reductase gene polymorphisms and recurrent pregnancy loss in China: A systematic review and meta-analysis. Arch. Gynecol. Obstet. 2016, 293, 283–290. [Google Scholar] [CrossRef] [PubMed]
- Furness, D.L.; Fenech, M.F.; Khong, Y.T.; Romero, R.; Dekker, G.A. One-carbon metabolism enzyme polymorphisms and uteroplacental insufficiency. Am. J. Obstet. Gynecol. 2008, 199, 276.e1–276.e8. [Google Scholar] [CrossRef] [PubMed]
- Laanpere, M.; Altmae, S.; Stavreus-Evers, A.; Nilsson, T.K.; Yngve, A.; Salumets, A. Folate-mediated one-carbon metabolism and its effect on female fertility and pregnancy viability. Nutr. Rev. 2010, 68, 99–113. [Google Scholar] [CrossRef] [PubMed]
- Nair, R.R.; Khanna, A.; Singh, R.; Singh, K. Association of maternal and fetal MTHFR A1298C polymorphism with the risk of pregnancy loss: A study of an Indian population and a meta-analysis. Fertil. Steril. 2013, 99, 1311–1318.e4. [Google Scholar] [CrossRef] [PubMed]
- Tiwari, D.; Bose, P.D.; Das, S.; Das, C.R.; Datta, R.; Bose, S. MTHFR (C677T) polymorphism and PR (PROGINS) mutation as genetic factors for preterm delivery, fetal death and low birth weight: A Northeast Indian population based study. Meta Gene 2015, 3, 31–42. [Google Scholar] [CrossRef] [PubMed]
- Behere, R.V.; Deshmukh, A.S.; Otiv, S.; Gupte, M.D.; Yajnik, C.S. Maternal Vitamin B12 Status During Pregnancy and Its Association With Outcomes of Pregnancy and Health of the Offspring: A Systematic Review and Implications for Policy in India. Front. Endocrinol. 2021, 12, 288. [Google Scholar] [CrossRef]
- Gong, M.; Dong, W.; He, T.; Shi, Z.; Huang, G.; Ren, R.; Huang, S.; Qiu, S.; Yuan, R. MTHFR 677C>T polymorphism increases the male infertility risk: A meta-analysis involving 26 studies. PLoS ONE 2015, 10, e0121147. [Google Scholar] [CrossRef]
- Facco, F.; You, W.; Grobman, W. Genetic thrombophilias and intrauterine growth restriction: A meta-analysis. Obstet. Gynecol. 2009, 113, 1206–1216. [Google Scholar] [CrossRef]
- Chedraui, P.; Andrade, M.E.; Salazar-Pousada, D.; Escobar, G.S.; Hidalgo, L.; Ramirez, C.; Spaanderman, M.E.; Kramer, B.W.; Gavilanes, A.W. Polymorphisms of the methylenetetrahydrofolate reductase gene (C677T and A1298C) in the placenta of pregnancies complicated with preeclampsia. Gynecol. Endocrinol. 2015, 31, 569–572. [Google Scholar] [CrossRef]
- Liew, S.C.; Gupta, E.D. Methylenetetrahydrofolate reductase (MTHFR) C677T polymorphism: Epidemiology, metabolism and the associated diseases. Eur. J. Med. Genet. 2015, 58, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Wu, H.; Zhu, P.; Geng, X.; Liu, Z.; Cui, L.; Gao, Z.; Jiang, B.; Yang, L. Genetic polymorphism of MTHFR C677T with preterm birth and low birth weight susceptibility: A meta-analysis. Arch. Gynecol. Obstet. 2017, 295, 1105–1118. [Google Scholar] [CrossRef] [PubMed]
- Blancquaert, D.; Storozhenko, S.; Loizeau, K.; De Steur, H.; De Brouwer, V.; Viaene, J.; Ravanel, S.; Rebeille, F.; Lambert, W.; Van Der Straeten, D. Folates and Folic Acid: From Fundamental Research Toward Sustainable Health. Crit. Rev. Plant Sci. 2010, 29, 14–35. [Google Scholar] [CrossRef]
- Shelnutt, K.P.; Kauwell, G.P.; Gregory, J.F., 3rd; Maneval, D.R.; Quinlivan, E.P.; Theriaque, D.W.; Henderson, G.N.; Bailey, L.B. Methylenetetrahydrofolate reductase 677C→T polymorphism affects DNA methylation in response to controlled folate intake in young women. J. Nutr. Biochem. 2004, 15, 554–560. [Google Scholar] [CrossRef] [PubMed]
- Jankovic-Karasoulos, T.; Furness, D.L.; Leemaqz, S.Y.; Dekker, G.A.; Grzeskowiak, L.E.; Grieger, J.A.; Andraweera, P.H.; McCullough, D.; McAninch, D.; McCowan, L.M.; et al. Maternal folate, one-carbon metabolism and pregnancy outcomes. Matern. Child. Nutr. 2021, 17, e13064. [Google Scholar] [CrossRef] [PubMed]
- Afman, L.A.; Lievers, K.J.A.; van der Put, N.M.J.; Trijbels, F.J.M.; Blom, H.J. Single nucleotide polymorphisms in the transcobalamin gene: Relationship with transcobalamin concentrations and risk for neural tube defects. Eur. J. Hum. Genet. 2002, 10, 433–438. [Google Scholar] [CrossRef] [PubMed]
- Christensen, K.E.; Rohlicek, C.V.; Andelfinger, G.U.; Michaud, J.; Bigras, J.L.; Richter, A.; MacKenzie, R.E.; Rozen, R. The MTHFD1 p.Arg653Gln Variant Alters Enzyme Function and Increases Risk for Congenital Heart Defects. Hum. Mutat. 2009, 30, 212–220. [Google Scholar] [CrossRef]
- Parle-McDermott, A.; Kirke, P.N.; Mills, J.L.; Molloy, A.M.; Cox, C.; O’Leary, V.B.; Pangilinan, F.; Conley, M.; Cleary, L.; Brody, L.C.; et al. Confirmation of the R653Q polymorphism of the trifunctional C1-synthase enzyme as a maternal risk for neural tube defects in the Irish population. Eur. J. Hum. Genet. 2006, 14, 768–772. [Google Scholar] [CrossRef] [Green Version]
- Wilson, D. Pre-conceptional Vitamin/Folic Acid Supplementation 2007: The Use of Folic Acid in Combination with a Multivitamin Supplement for the Prevention of Neural Tube Defects and Other Congenital Anomalies. J. Obstet. Gynaecol. Can. 2007, 29, 1003–1013. [Google Scholar] [CrossRef]
- Wilson, R.D.; Genetics, C.; Wilson, R.D.; Audibert, F.; Brock, J.A.; Carroll, J.; Cartier, L.; Gagnon, A.; Johnson, J.A.; Langlois, S.; et al. Pre-conception Folic Acid and Multivitamin Supplementation for the Primary and Secondary Prevention of Neural Tube Defects and Other Folic Acid-Sensitive Congenital Anomalies. J. Obstet. Gynaecol. Can. 2015, 37, 534–552. [Google Scholar] [CrossRef] [Green Version]
- Molloy, A.M.; Kirke, P.N.; Brody, L.C.; Scott, J.M.; Mills, J.L. Effects of folate and vitamin B12 deficiencies during pregnancy on fetal, infant, and child development. Food Nutr. Bull. 2008, 29, S101–S111; discussion S112–105. [Google Scholar] [CrossRef] [PubMed]
- Baker, H.; DeAngelis, B.; Holland, B.; Gittens-Williams, L.; Barrett, T., Jr. Vitamin profile of 563 gravidas during trimesters of pregnancy. J. Am. Coll. Nutr. 2002, 21, 33–37. [Google Scholar] [CrossRef]
- Kloss, O.; Eskin, N.A.M.; Suh, M. Thiamin deficiency on fetal brain development with and without prenatal alcohol exposure. Biochem. Cell. Biol. 2018, 96, 169–177. [Google Scholar] [CrossRef] [Green Version]
- Butterworth, R.F. Maternal thiamine deficiency: Still a problem in some world communities. Am. J. Clin. Nutr. 2001, 74, 712–713. [Google Scholar] [CrossRef] [PubMed]
- Ortega, R.M.; Martinez, R.M.; Andres, P.; Marin-Arias, L.; Lopez-Sobaler, A.M. Thiamin status during the third trimester of pregnancy and its influence on thiamin concentrations in transition and mature breast milk. Br. J. Nutr. 2004, 92, 129–135. [Google Scholar] [CrossRef] [PubMed]
- Bartakova, V.; Pleskacova, A.; Kuricova, K.; Pacal, L.; Dvorakova, V.; Belobradkova, J.; Tomandlova, M.; Tomandl, J.; Kankova, K. Dysfunctional protection against advanced glycation due to thiamine metabolism abnormalities in gestational diabetes. Glycoconj. J. 2016, 33, 591–598. [Google Scholar] [CrossRef] [PubMed]
- Zeng, W.Q.; Al-Yamani, E.; Acierno, J.S., Jr.; Slaugenhaupt, S.; Gillis, T.; MacDonald, M.E.; Ozand, P.T.; Gusella, J.F. Biotin-responsive basal ganglia disease maps to 2q36.3 and is due to mutations in SLC19A3. Am. J. Hum. Genet. 2005, 77, 16–26. [Google Scholar] [CrossRef] [Green Version]
- Fradin, D.; Bougneres, P. Three common intronic variants in the maternal and fetal thiamine pyrophosphokinase gene (TPK1) are associated with birth weight. Ann. Hum. Genet. 2007, 71, 578–585. [Google Scholar] [CrossRef]
- Nwogu, C.M.; Okunade, K.S.; Adenekan, M.A.; Sekumade, A.I.; John-olabode, S.; Oluwole, A.A. Association between maternal serum homocysteine concentrations in early pregnancy and adverse pregnancy outcomes. Ann. Afr. Med. 2020, 19, 113–118. [Google Scholar] [CrossRef]
- Rumbold, A.; Ota, E.; Nagata, C.; Shahrook, S.; Crowther, C.A. Vitamin C supplementation in pregnancy. Cochrane Database Syst. Rev. 2015, 2015, CD004072. [Google Scholar] [CrossRef]
- Siega-Riz, A.M.; Promislow, J.H.; Savitz, D.A.; Thorp, J.M., Jr.; McDonald, T. Vitamin C intake and the risk of preterm delivery. Am. J. Obstet. Gynecol. 2003, 189, 519–525. [Google Scholar] [CrossRef]
- Stratakis, C.A.; Taymans, S.E.; Daruwala, R.; Song, J.; Levine, M. Mapping of the human genes (SLC23A2 and SLC23A1) coding for vitamin C transporters 1 and 2 (SVCT1 and SVCT2) to 5q23 and 20p12, respectively. J. Med. Genet. 2000, 37, E20. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Erichsen, H.C.; Engel, S.A.; Eck, P.K.; Welch, R.; Yeager, M.; Levine, M.; Siega-Riz, A.M.; Olshan, A.F.; Chanock, S.J. Genetic variation in the sodium-dependent vitamin C transporters, SLC23A1, and SLC23A2 and risk for preterm delivery. Am. J. Epidemiol. 2006, 163, 245–254. [Google Scholar] [CrossRef] [PubMed]
- Duarte-Salles, T.; Mendez, M.A.; Morales, E.; Bustamante, M.; Rodriguez-Vicente, A.; Kogevinas, M.; Sunyer, J. Dietary benzo(a)pyrene and fetal growth: Effect modification by vitamin C intake and glutathione S-transferase P1 polymorphism. Environ. Int. 2012, 45, 1–8. [Google Scholar] [CrossRef] [Green Version]
- Mielgo-Ayuso, J.; Aparicio-Ugarriza, R.; Olza, J.; Aranceta-Bartrina, J.; Gil, A.; Ortega, R.M.; Serra-Majem, L.; Varela-Moreiras, G.; Gonzalez-Gross, M. Dietary Intake and Food Sources of Niacin, Riboflavin, Thiamin and Vitamin B(6) in a Representative Sample of the Spanish Population. The Anthropometry, Intake, and Energy Balance in Spain (ANIBES) Study dagger. Nutrients 2018, 10, 846. [Google Scholar] [CrossRef] [Green Version]
- Yoshii, K.; Hosomi, K.; Sawane, K.; Kunisawa, J. Metabolism of Dietary and Microbial Vitamin B Family in the Regulation of Host Immunity. Front. Nutr. 2019, 6, 48. [Google Scholar] [CrossRef] [Green Version]
- Dror, D.K.; Allen, L.H. Interventions with vitamins B6, B12 and C in pregnancy. Paediatr. Perinat. Epidemiol. 2012, 26 (Suppl. S1), 55–74. [Google Scholar] [CrossRef]
- Simpson, J.L.; Bailey, L.B.; Pietrzik, K.; Shane, B.; Holzgreve, W. Micronutrients and women of reproductive potential: Required dietary intake and consequences of dietary deficiency or excess. Part I--Folate, Vitamin B12, Vitamin B6. J. Matern. Fetal Neonatal. Med. 2010, 23, 1323–1343. [Google Scholar] [CrossRef]
| Fat-Soluble Vitamins | Reference Levels | Recommended Intake | Recommended Intake in Early Pregnancy | Recommended Intake in Late Pregnancy | References |
|---|---|---|---|---|---|
| Vitamin A | 0.2–0.6 mg/L | 700 µg/day | 770 µg/day | 1300 µg/day | [17,18] |
| Vitamin E | 7.4–23.0 mg/L | 15 mg/day | 15 mg/day | 19 mg/day | [17] |
| Vitamin D | 75–200 nmol/L | 15 µg/day | 25 µg/day | 35 µg/day | [19,20] |
| Vitamin K | 0.09–1.99 nmol/l | 90 µg/day | 90 µg/day | - | [21,22] |
| Water-Soluble Vitamins | Plasma Levels | Nutrient Intake under Physiological Conditions | Recommended Intake in Early Pregnancy | Recommended Intake in Late Pregnancy | References |
|---|---|---|---|---|---|
| Vitamin B9 | 2.2–17 ng/ml | 400 µg/day | 600 µg/day | 500 µg/day | [85] |
| Vitamin B12 | 2–9 µg/ml | 2.4 µg/day | 2.6 µg/day | 2.8 µg/day | [86] |
| Vitamin B1 | 5–12 μg/dL | 1.2 mg/day | 1.4 mg/day | - | [87] |
| Vitamin C | 6–14 mg/L | 75 mg/day | 85 mg/day | 120 mg/day | [88] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bertuccio, M.P.; Currò, M.; Caccamo, D.; Ientile, R. Dietary Intake and Genetic Background Influence Vitamin Needs during Pregnancy. Healthcare 2022, 10, 768. https://doi.org/10.3390/healthcare10050768
Bertuccio MP, Currò M, Caccamo D, Ientile R. Dietary Intake and Genetic Background Influence Vitamin Needs during Pregnancy. Healthcare. 2022; 10(5):768. https://doi.org/10.3390/healthcare10050768
Chicago/Turabian StyleBertuccio, Maria Paola, Monica Currò, Daniela Caccamo, and Riccardo Ientile. 2022. "Dietary Intake and Genetic Background Influence Vitamin Needs during Pregnancy" Healthcare 10, no. 5: 768. https://doi.org/10.3390/healthcare10050768
APA StyleBertuccio, M. P., Currò, M., Caccamo, D., & Ientile, R. (2022). Dietary Intake and Genetic Background Influence Vitamin Needs during Pregnancy. Healthcare, 10(5), 768. https://doi.org/10.3390/healthcare10050768







