Plasmolysis: Loss of Turgor and Beyond
Abstract
:1. Introduction
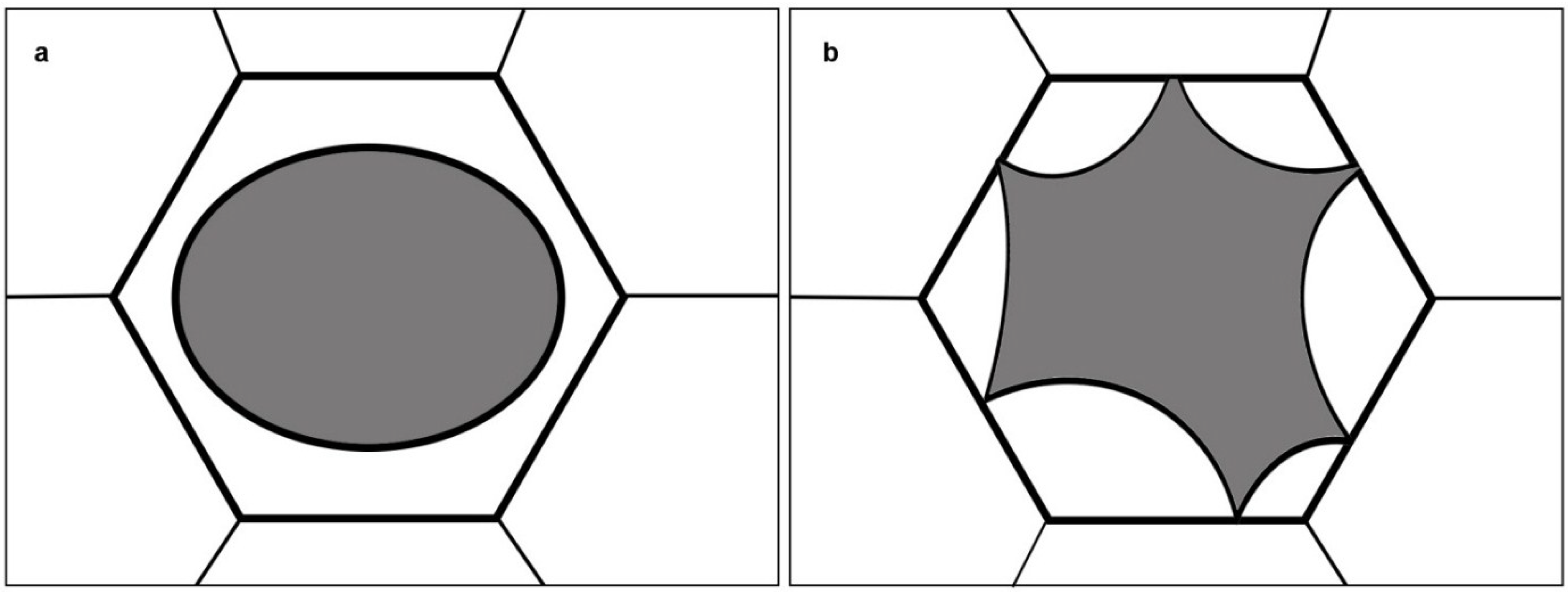
2. Results and Discussion
2.1. A Plasmolytic Cycle
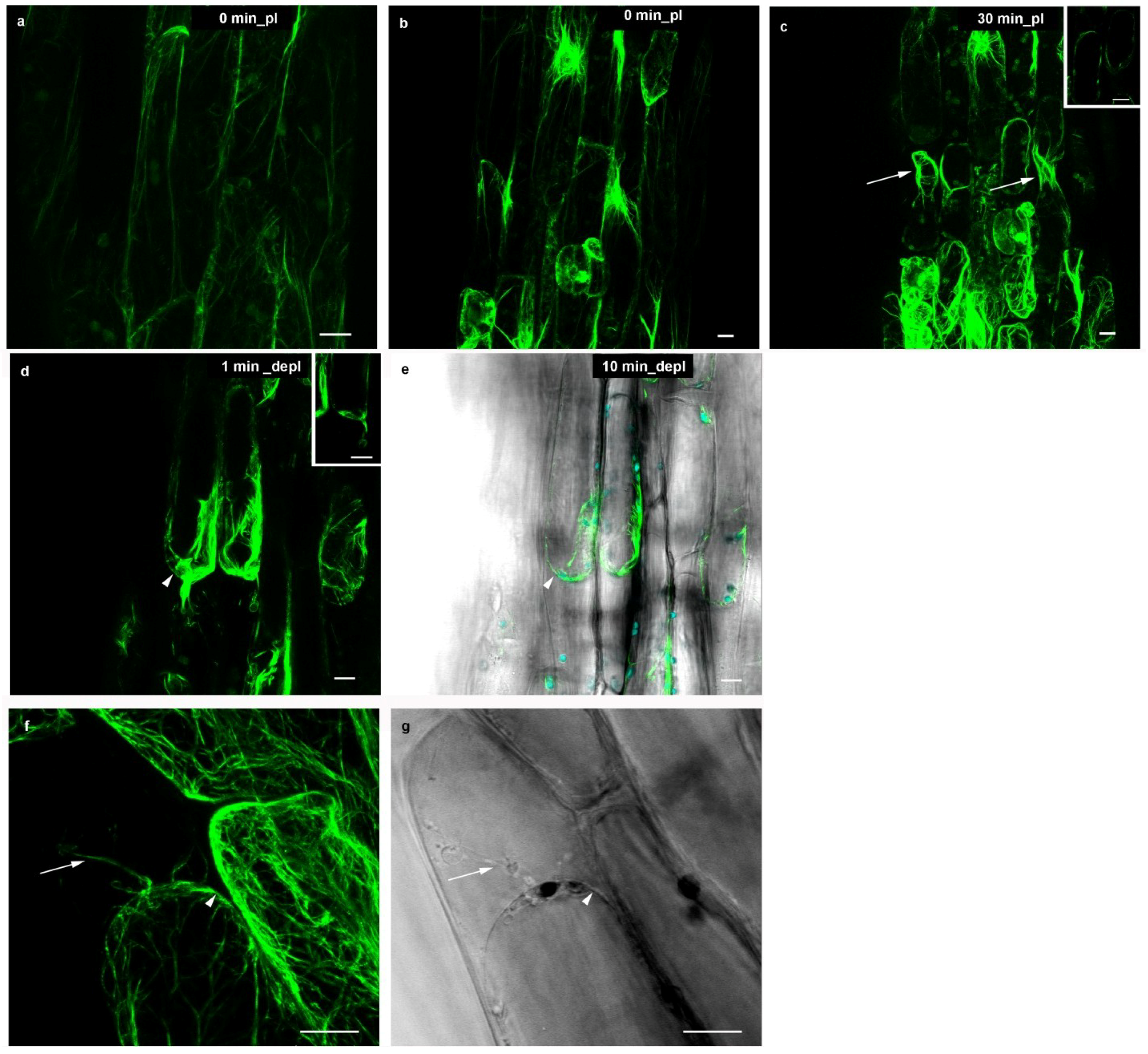
2.2. Microtubules
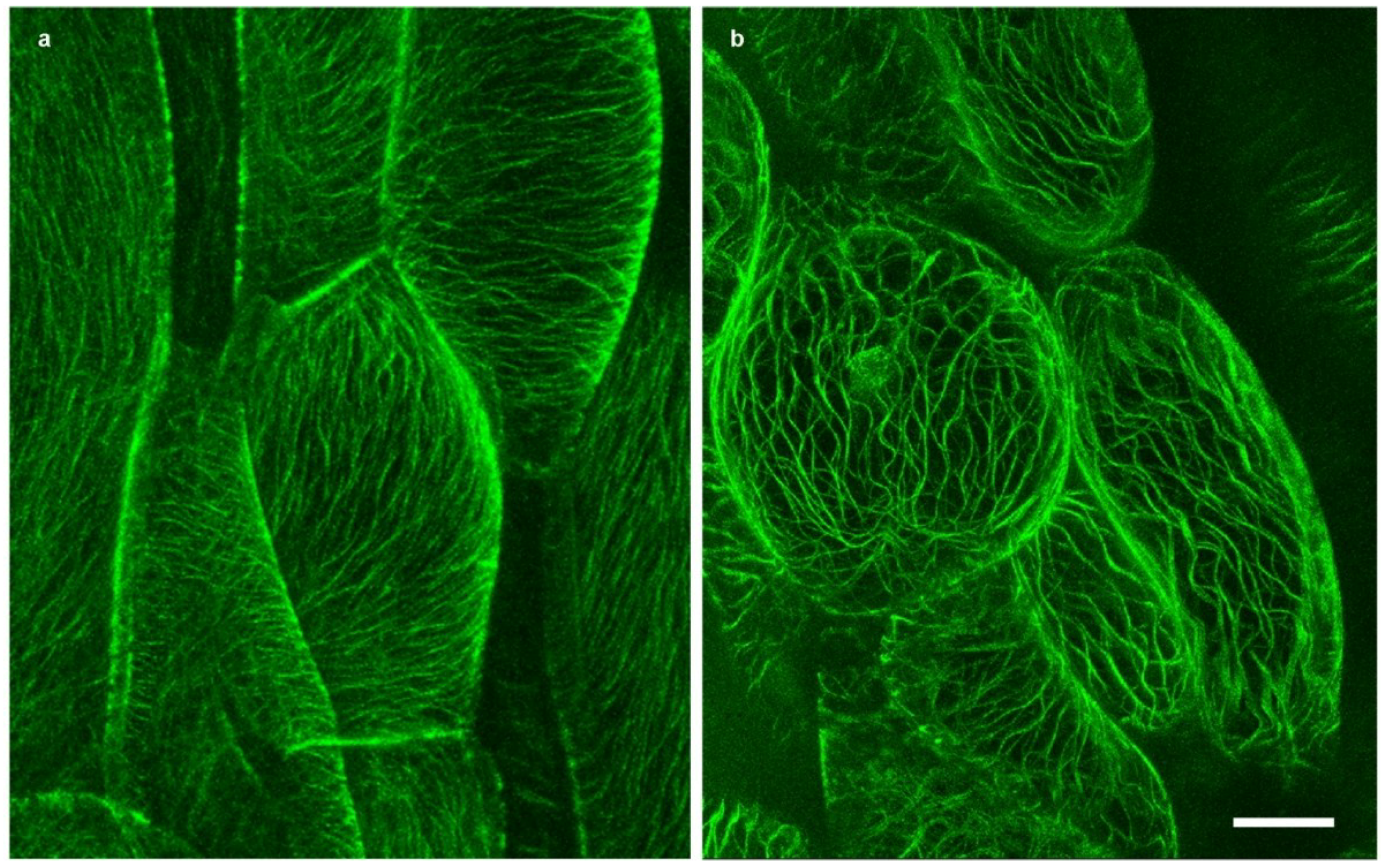

2.3. Actin Microfilaments
2.4. A Broader View
3. Experimental Section
4. Conclusions
Supplementary Files
Supplementary File 1Acknowledgments
Author Contributions
Conflicts of Interest
References
- Oparka, K.J. Plasmolysis: New insights into an old process. New Phytol. 1994, 126, 571–591. [Google Scholar] [CrossRef]
- Rojas, E.; Theriot, J.A.; Huang, K.C. Response of Escherichia coli growth rate to osmotic shock. Proc. Natl. Acad. Sci. USA 2014, 111, 7807–7812. [Google Scholar] [CrossRef] [PubMed]
- Bitsikas, V.; Karachaliou, M.; Gournas, C.; Diallinas, G. Hypertonic conditions trigger transient plasmolysis, growth arrest and blockage of transporter endocytosis in Aspergillus nidulans and Saccharomyces cerevisiae. Mol. Membr. Biol. 2011, 28, 54–68. [Google Scholar] [CrossRef] [PubMed]
- Chitcholtan, K.; Harris, E.; Yu, Y.; Harland, C.; Garrill, A. An investigation into plasmolysis in the oomycete Achlya bisexualis reveals that membrane-wall attachment points are sensitive to peptides containing the sequence RGD and that cell wall deposition can occur despite retraction of the protoplast. Can. J. Microbiol. 2012, 58, 1212–1220. [Google Scholar] [CrossRef] [PubMed]
- Stadelmann, E. Plasmolyse und Deplasmolyse. In Encyclopedia of Plant Physiology; Ruhland, W., Ed.; Springer Verlag: Berlin/Göttingen/Heidelberg, Germany, 1956; Volume 2, pp. 71–115. [Google Scholar]
- Lenné, T.; Bryant, G.; Hocart, C.H.; Huang, C.X.; Ball, M.C. Freeze avoidance: A dehydrating moss gathers no ice. Plant Cell Environ. 2010, 33, 1731–1741. [Google Scholar] [CrossRef] [PubMed]
- Küster, E. Über Inhaltsverlagerungen in plasmolysierten Zellen. Flora 1910, 100, 267–287. [Google Scholar]
- Stadelmann, E. Zu Plasmolyse und Deplasmolyse von Allium-Epidermen. Protoplasma 1964, 59, 14–68. [Google Scholar] [CrossRef]
- Fischer, J.M.C.; Peterson, C.A.; Bols, N.C. A new fluorescent test for cell vitality using calcofuor white M2R. Stain Technol. 1985, 60, 69–79. [Google Scholar] [PubMed]
- Luyet, B.J.; Gehenio, P.M. The survival of moss vitrified in liquid air and its relation to water conten. Biodynamica 1938, 42, 1–7. [Google Scholar]
- Strugger, S. Praktikum der Zell- und Gewebephysiologie der Pflanze, 2nd ed.; Springer Verlag: Berlin/Göttingen/Heidelberg, Germany, 1949; Volume 1, pp. 89–93. [Google Scholar]
- Ruoslahti, E.; Pierschbacher, M.D. New perspectives in cell adhesion: RGD and integrins. Science 1987, 238, 491–497. [Google Scholar] [CrossRef] [PubMed]
- Hostetter, M.K. RGD-mediated adhesion in fungal pathogens of humans, plants and insects. Curr. Opin. Microbiol. 2000, 3, 344–348. [Google Scholar] [CrossRef] [PubMed]
- Lu, B.; Chen, F.; Gong, Z.H.; Xie, H.; Zhang, J.H.; Liang, J.S. Intracellular localization of integrin-like protein and its roles in osmotic stress-induced abscisic acid biosynthesis in Zea mays. Protoplasma 2007, 232, 35–43. [Google Scholar] [CrossRef] [PubMed]
- Ambrose, C.; Allard, J.F.; Cytrynbaum, E.N.; Wasteneys, G.O. A CLASP-modulated cell edge barrier mechanism drives cell-wide cortical microtubule organization in Arabidopsis. Nat. Commun. 2011, 2, e430. [Google Scholar] [CrossRef]
- Lei, L.; Li, S.; Bashline, L.; Gu, Y. Dissecting the molecular mechanism underlying the intimate relationship between cellulose microfibrils and cortical microtubules. Front. Plant Sci. 2014, 5, e90. [Google Scholar] [CrossRef]
- De Vries, H. Eine Methode zur Analyse der Turgorkraft. Jahrb. Wiss. Bot. 1877, 14, 427–601. [Google Scholar]
- Hecht, K. Studien über den Vorgang der Plasmolyse. Beitr. Biol. Pflanz. 1912, 11, 133–189. [Google Scholar]
- Sitte, P. Zellfeinbau bei plasmolyse. Protoplasma 1963, 57, 304–333. [Google Scholar] [CrossRef]
- Tilney, L.G.; Cooke, T.J.; Connelly, P.S.; Tilney, M.S. The structure of plasmodesmata as revealed by plasmolysis, detergent extraction, and protease digestion. J. Cell Biol. 1991, 112, 739–747. [Google Scholar] [CrossRef] [PubMed]
- Pont-Lezica, R.F.; McNally, J.G.; Pickard, B.G. Wall-to-membrane linkers in onion epidermis: Some hypotheses. Plant Cell Environ. 1993, 16, 111–123. [Google Scholar] [CrossRef]
- Lang, I.; Barton, D.A.; Overall, R.L. Membrane-wall attachements in plasmolysed plant cells. Protoplasma 2004, 224, 231–243. [Google Scholar] [CrossRef] [PubMed]
- Cleary, A.L. Plasma membrane-cell wall connections: Roles in mitosis and cytokinesis revealed by plasmolysis of Tradescantia virginiana leaf epidermal cells. Protoplasma 2001, 215, 21–34. [Google Scholar] [CrossRef] [PubMed]
- Lang-Pauluzzi, I.; Gunning, B.E.S. A plasmolytic cycle: The fate of cytoskeletal elements. Protoplasma 2000, 212, 174–185. [Google Scholar] [CrossRef]
- Komis, G.; Quader, H.; Galatis, B.; Apostolakos, P. Macrotubule-dependent protoplast volume regulation in plasmolysed root-tip cells of Triticum turgidum: Involvement of phospholipase D. New Phytol. 2006, 171, 737–750. [Google Scholar] [CrossRef] [PubMed]
- Komis, G.; Apostolakos, P.; Galatis, B. Hyperosmotic stress-induced actin filament reorganization in leaf cells of Chlorophyton comosum. J. Exp. Bot. 2002, 53, 1699–1710. [Google Scholar] [CrossRef] [PubMed]
- Komis, G.; Apostolakos, P.; Galatis, B. Altered patterns of tubulin polymerization in dividing leaf cells of Chlorophyton comosum after a hyperosmotic treatment. New Phytol. 2001, 149, 193–207. [Google Scholar] [CrossRef]
- Ambrose, C.; Wasteneys, G.O. Nanoscale and geometric influences on the microtubule cytoskeleton in plants: Thinking inside and outside the box. Protoplasma 2012, 249, S69–S76. [Google Scholar] [CrossRef] [PubMed]
- Volgger, M.; Lang, I.; Ovečka, M.; Lichtscheidl, I. Plasmolysis and cell wall deposition in wheat root hairs under osmotic stress. Protoplasma 2010, 243, 51–62. [Google Scholar] [CrossRef] [PubMed]
- Oparka, K.J.; Prior, D.A.M.; Crawford, J.W. Behaviour of plasma membrane, cortical ER and plasmoldesmata during plasmolysis of onion epidermal cells. Plant Cell Environ. 1994, 17, 163–171. [Google Scholar] [CrossRef]
- Oparka, K.J.; Prior, D.A.M.; Crawford, J.W. Membrane conservation during plasmolysis. In Membranes: Specialized Functions in Plants; Smallwood, M., Knox, J.P., Bowles, D.J., Eds.; BIOS Scientific Publishers Ltd.: Milton, UK, 1996; pp. 39–56. [Google Scholar]
- Marc, J.; Granger, C.L.; Brincat, J.; Fisher, D.D.; Kao, T.H.; McCubbin, A.G.; Cyr, R.J. A GFP-MAP4 reporter gene for visualizing cortical microtubule rearrangements in living epidermal cells. Plant Cell 1998, 10, 1927–1939. [Google Scholar] [PubMed]
- Shaw, S.L.; Kamyar, R.; Ehrhardt, D.W. Sustained microtubule treadmilling in Arabidopsis cortical arrays. Science 2003, 300, 1715–1718. [Google Scholar] [CrossRef] [PubMed]
- Voigt, B.; Timmers, A.C.J.; Samaj, J.; Muller, J.; Baluska, F.; Menzel, D. GFP-FABD2 fusion construct allows in vivo visualization of the dynamic actin cytoskeleton in all cells of Arabidopsis seedlings. Eur. J. Cell Biol. 2005, 84, 595–608. [Google Scholar] [CrossRef] [PubMed]
- Baskin, T.I. On the alignment of cellulose microfibrils by cortical microtubules: A review and a model. Protoplasma 2001, 215, 150–171. [Google Scholar] [CrossRef] [PubMed]
- Gutierrez, R.; Lindeboom, J.J.; Paredez, A.R.; Emons, A.M.C.; Ehrhardt, D.W. Arabidopsis cortical microtubules position cellulose synthase delivery to the plasma membrane and interact with cellulose synthase trafficking compartments. Nat. Cell Biol. 2009, 11, 797–806. [Google Scholar] [CrossRef] [PubMed]
- Somerville, C. Cellulose synthesis in higher plants. Annu. Rev. Cell Dev. Biol. 2006, 22, 53–78. [Google Scholar] [CrossRef] [PubMed]
- Vain, T.; Crowell, E.F.; Timpano, H.; Biot, E.; Desprez, T.; Mansoori, N.; Trindade, L.M.; Pagant, S.; Robert, S.; Hoefte, H.; et al. The cellulase KORRIGAN is part of the cellulose synthase complex. Plant Physiol. 2014, 165, 1521–1532. [Google Scholar] [CrossRef] [PubMed]
- Bashline, L.; Du, J.; Gu, Y. The trafficking and behavior of cellulose synthase and a glimpse of potential cellulose synthesis regulators. Front. Biol. 2011, 6, 377–383. [Google Scholar] [CrossRef]
- Strzelecka-Golaszewska, H. Divalent cations, nucleotides, and actin structure. Results Probl. Cell Differ. 2001, 32, 23–41. [Google Scholar] [PubMed]
- Wen, Q.; Janmey, P.A. Polymer physics of the cytoskeleton. Curr. Opin. Solid State Mater. Sci. 2011, 15, 177–182. [Google Scholar] [CrossRef] [PubMed]
- Liu, Q.; Qiao, F.; Ismail, A.; Chang, X.; Nick, P. The plant cytoskeleton controls regulatory volume increase. BBA Biomembr. 2013, 1828, 2111–2120. [Google Scholar] [CrossRef]
- Wojtaszek, P.; Baluska, F.; Kasprowicz, A.; Luczak, M.; Volkmann, D. Domain-specific mechanosensory transmission of osmotic and enzymatic cell wall disturbances to the actin cytoskeleton. Protoplasma 2007, 230, 217–230. [Google Scholar] [CrossRef] [PubMed]
- Singh, J. Ultrastructural alterations in cells of hardened and non-hardened winter rye during hyperosmotic and extracellular freezing stresses. Protoplasma 1979, 98, 329–341. [Google Scholar] [CrossRef]
- Lichtscheidl, I.K.; Url, W.G. Organization and dynamics of cortical endoplasmic reticulum in inner epidermal cells of onion bulb scales. Protoplasma 1990, 157, 203–215. [Google Scholar] [CrossRef]
- Pickard, B.G.; Ding, J.P. The mechanosensory calcium-selective ion-channel: Key component of a plasmalemmal control center. Aust. J. Plant Physiol. 1993, 20, 439–459. [Google Scholar] [CrossRef] [PubMed]
- Murashige, T.; Skoog, F. A revised medium for rapid growth and bio assays with tobacce tissue cultures. Physiol. Plant. 1962, 15, 473–497. [Google Scholar] [CrossRef]
© 2014 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lang, I.; Sassmann, S.; Schmidt, B.; Komis, G. Plasmolysis: Loss of Turgor and Beyond. Plants 2014, 3, 583-593. https://doi.org/10.3390/plants3040583
Lang I, Sassmann S, Schmidt B, Komis G. Plasmolysis: Loss of Turgor and Beyond. Plants. 2014; 3(4):583-593. https://doi.org/10.3390/plants3040583
Chicago/Turabian StyleLang, Ingeborg, Stefan Sassmann, Brigitte Schmidt, and George Komis. 2014. "Plasmolysis: Loss of Turgor and Beyond" Plants 3, no. 4: 583-593. https://doi.org/10.3390/plants3040583
APA StyleLang, I., Sassmann, S., Schmidt, B., & Komis, G. (2014). Plasmolysis: Loss of Turgor and Beyond. Plants, 3(4), 583-593. https://doi.org/10.3390/plants3040583





