Exogenous Sucrose Improves the Vigor of Aged Safflower Seeds by Mediating Fatty Acid Metabolism and Glycometabolism
Abstract
1. Introduction
2. Results
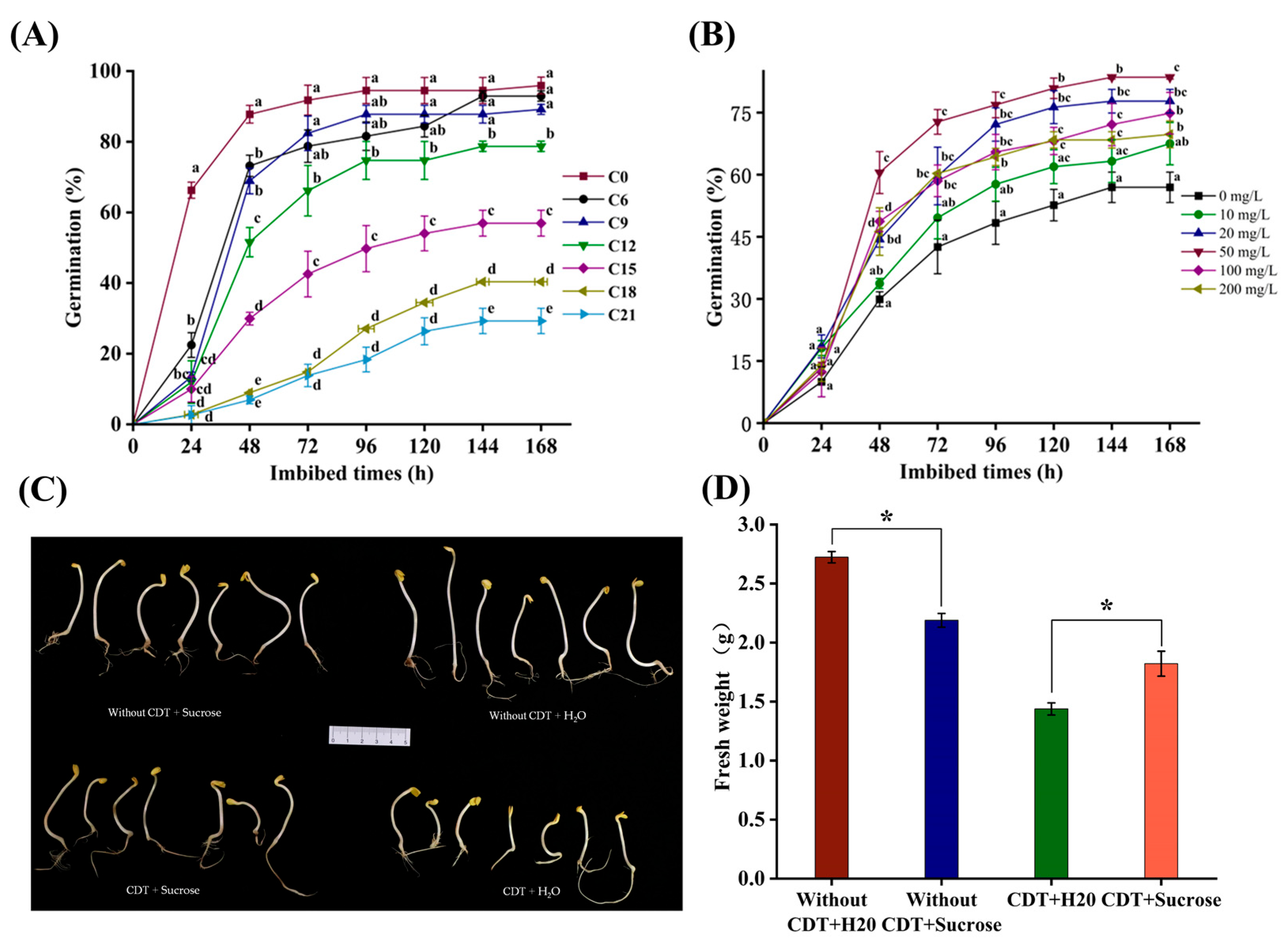
2.1. CDT Remarkably Inhibited Safflower Seed Germination
2.2. Exogenous Sucrose Treatment Promoted Germination of Aged Safflower Seeds but Not from Unaged Seeds
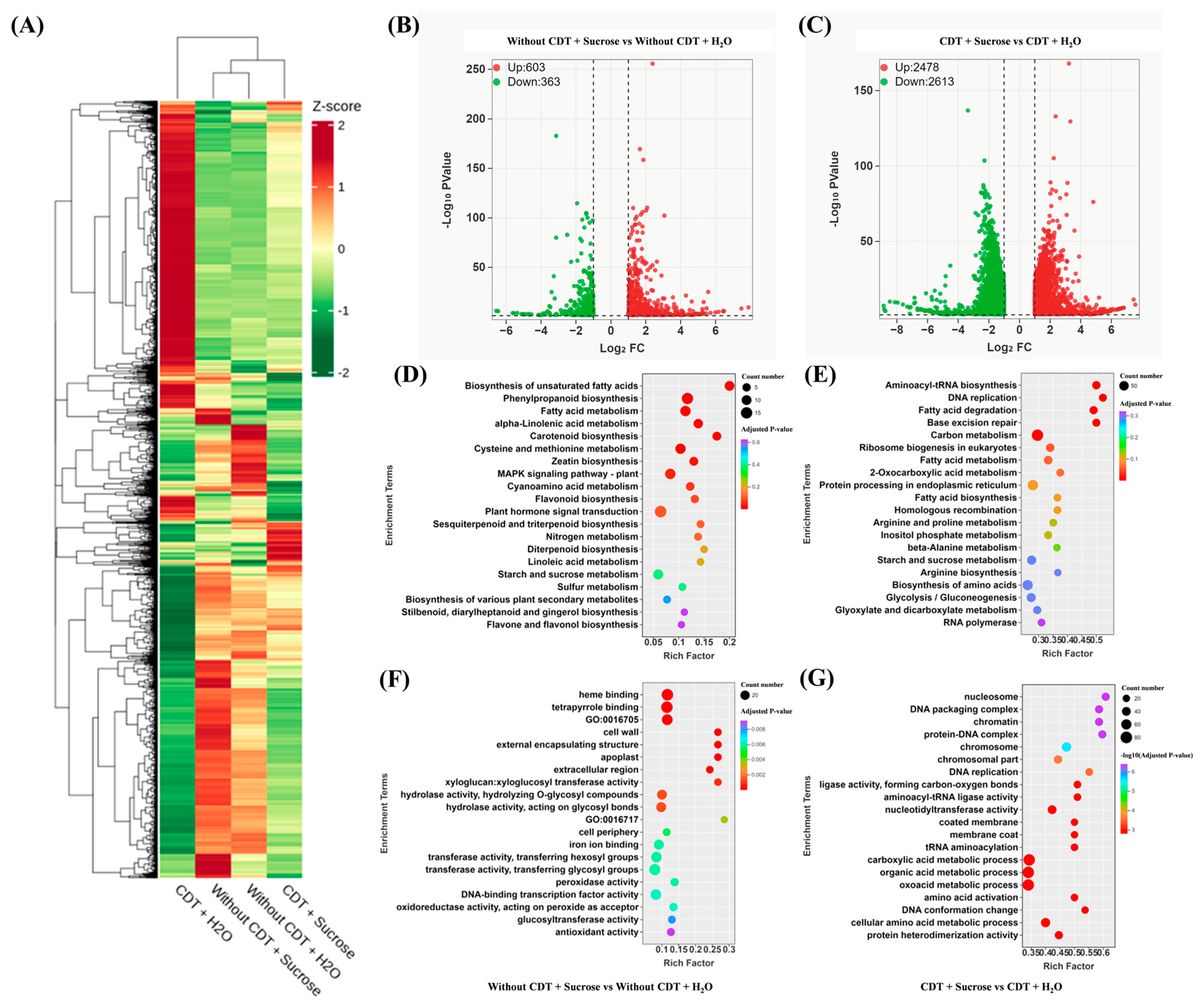
2.3. Transcriptome Analysis and Differentially Expressed Genes Between Control and Exogenous Treatments
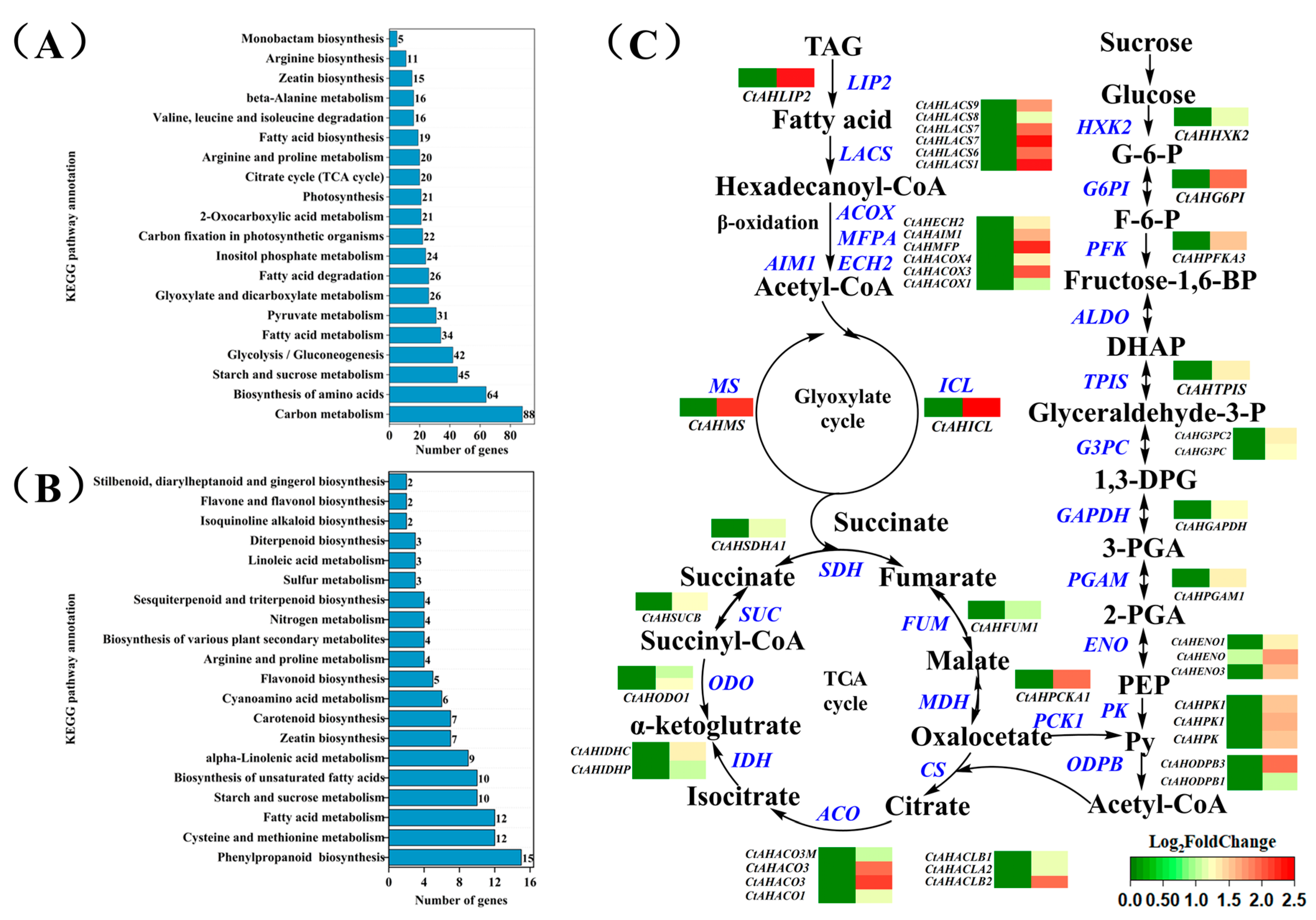
2.4. Exogenous Sucrose Increases the Transcription of Several Key Genes Involved in the Conversion of Triacylglycerols to Fatty Acids and Sugars During Imbibition in Aged Safflower Seeds
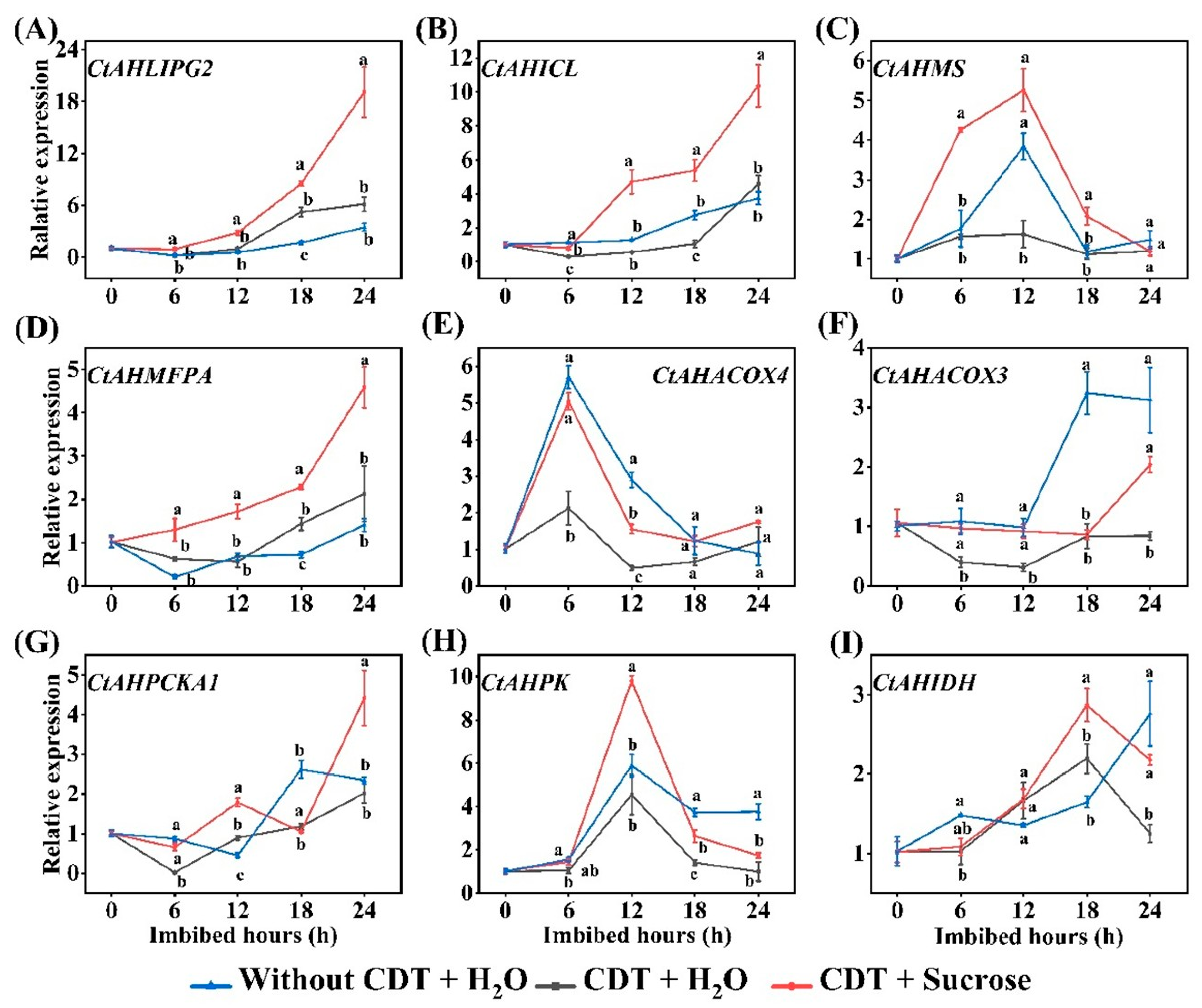
2.5. qPCR Verification for Expressions of Exogenous Sucrose Altered Genes
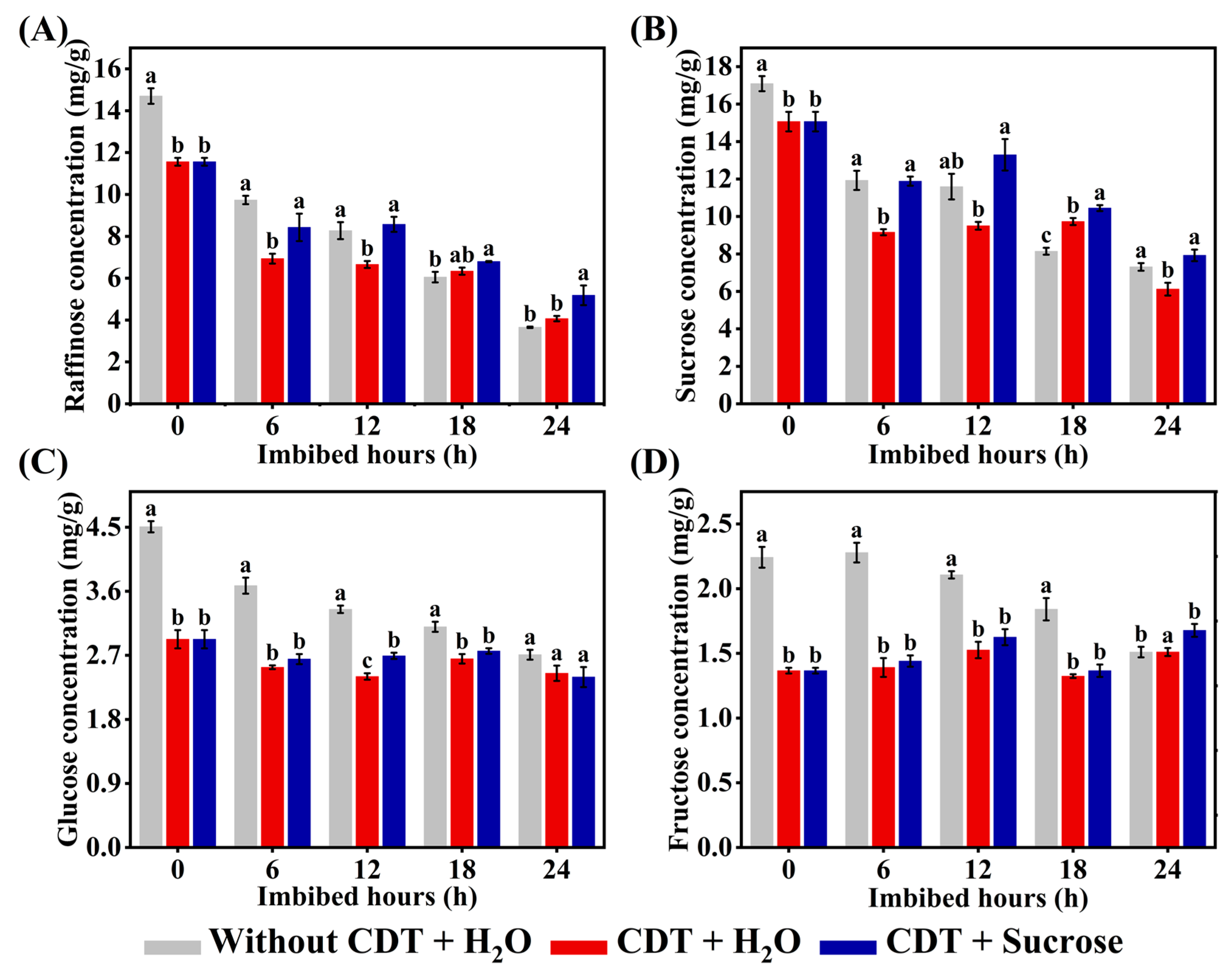
2.6. Exogenous Sucrose Treatment Increased the Concentrations of Various Sugars in Aged Safflower Seeds During Imbibition
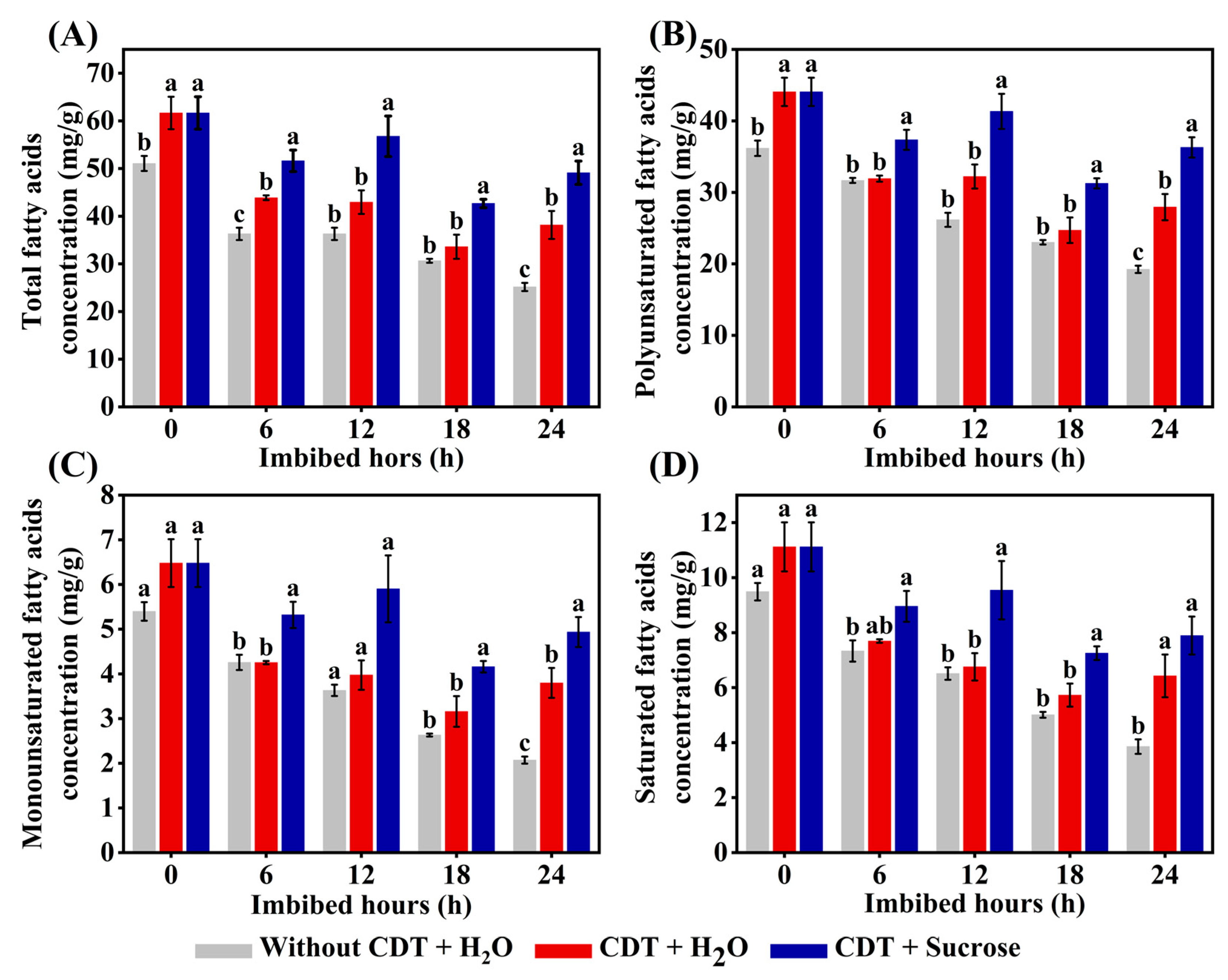
2.7. Exogenous Sucrose Treatment Increased the Concentrations of Fatty Acids in Aged Safflower Seeds During Imbibition Time
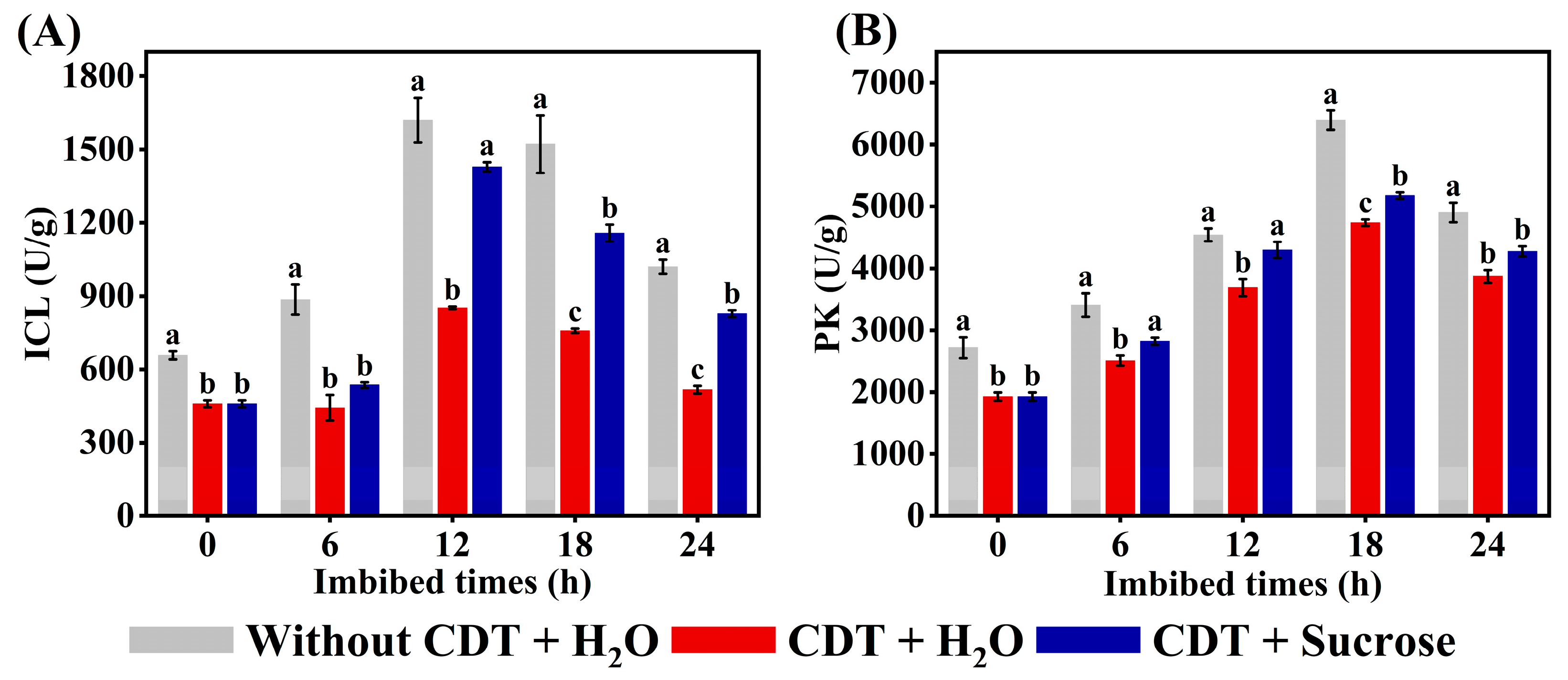
2.8. Exogenous Sucrose Increased the Activity of ICL and PK in Aged Safflower Seeds During Imbibitions
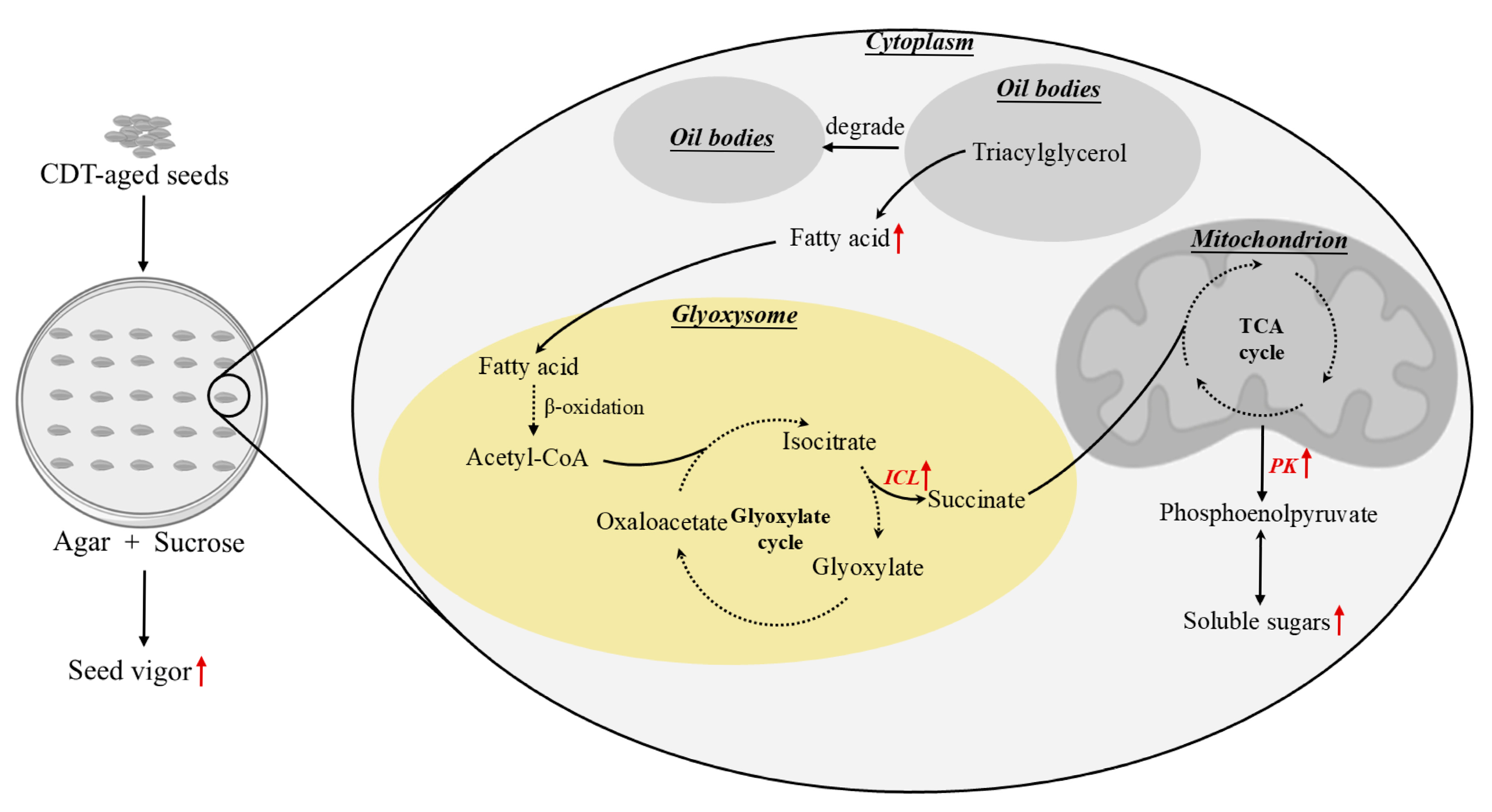
2.9. Changes in the Interaction of Glyoxysome with Oil Bodies Under Sucrose Treatment
3. Discussion
3.1. Exogenous Sucrose Has a Positive Effect on Germination in Aged Safflower Seeds, but Not in Unaged Ones
3.2. Exogenous Sucrose Promoted the Hydrolysis of Triacylglycerol to Sugars in Aged Safflower Seeds
3.3. The Glyoxysomes Plays an Important Role in the Conversion of Fatty Acids to Sugars
3.4. Perspectives Research on Seed Vigor Repair Mechanisms
4. Materials and Methods
4.1. Plant Materials
4.2. Controlled Deterioration Treatment
4.3. Germination Test and Seeding Establishment
4.4. Determination of Various Sugar Concentration
4.5. Fatty Acid Extraction and Measurements
4.6. Measurement of Enzyme Activity
4.7. RNA Extraction, Sequencing, and Data Analysis
4.8. Real-Time Quantitative Polymerase Chain Reaction
4.9. Transmission Electron Microscopy
4.10. Statistical Analysis
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Zohary, D. Monophyletic vs. polyphyletic origin of the crops on which agriculture was founded in the Near East. Genet. Resour. Crop Evol. 2019, 46, 133–142. [Google Scholar] [CrossRef]
- FAO. Faostat/en. 2020. Available online: https://www.fao.org/faostat/en/#data/QCL (accessed on 26 September 2024).
- Sharifi, R.S.; Namvar, A.; Sharifi, R.S. Grain filling and fatty acid composition of safflower fertilized with integrated nitrogen fertilizer and biofertilizers. Pesqui. Agropecuária Bras. 2017, 52, 236–243. [Google Scholar] [CrossRef]
- Matthaus, B.; Özcan, M.M.; Al Juhaimi, F.Y. Fatty acid composition and tocopherol profiles of safflower (Carthamus tinctorius L.) seed oils. Nat. Prod. Res. 2015, 29, 193–196. [Google Scholar] [CrossRef]
- Amini, H.; Arzani, A.; Karami, M. Effect of water deficiency on seed quality and physiological traits of different safflower genotypes. Turk. J. Biol. 2014, 38, 271–282. [Google Scholar] [CrossRef]
- Goel, A.; Goel, A.K.; Sheoran, I.S. Changes in oxidative stress enzymes during artificial ageing in cotton (Gossypium hirsutum L.) seeds. J. Plant Physiol. 2003, 160, 1093–1100. [Google Scholar] [CrossRef]
- Koornneef, M.; Bentsink, L.; Hilhorst, H. Seed dormancy and germination. Curr. Opin. Plant Biol. 2022, 5, 33–36. [Google Scholar] [CrossRef]
- Bewley, J.D.; Bradford, K.J.; Hilhorst, H.W.M.; Nonogaki, H. Germination. In Seeds; Springer: New York, NY, USA, 2013; pp. 133–135. [Google Scholar]
- Gran, P.; Visscher, T.W.; Bai, B.; Nijveen, H.; Mahboubi, A.; Bakermans, L.L.; Willems, L.A.J.; Bentsink, L. Unravelling the dynamics of seed-stored mRNAs during seed priming. New Phytol. 2025. [Google Scholar] [CrossRef]
- Gu, J.; Hou, D.; Li, Y.; Chao, H.; Zhang, K.; Wang, H.; Xiang, J.; Raboanatahiry, N.; Wang, B.; Li, M. Integration of proteomic and genomic approaches to dissect seed germination vigor in Brassica napus seeds differing in oil content. BMC Plant Biol. 2019, 19, 21. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.; Zuo, Q.; Zheng, J.; You, J.; Yang, G.; Leng, S. Salt stress decreases seed yield and postpones growth process of canola (Brassica napus L.) by changing nitrogen and carbon characters. Sci. Rep. 2022, 12, 17884. [Google Scholar] [CrossRef]
- Wei, J.; Xu, L.; Shi, Y.; Cheng, T.; Tan, W.; Zhao, Y.; Li, C.; Yang, X.; Ouyang, L.; Wei, M.; et al. Transcriptome profile analysis of Indian mustard (Brassica juncea L.) during seed germination reveals the drought stress-induced genes associated with energy, hormone, and phenylpropanoid pathways. Plant Physiol. Biochem. 2023, 200, 107750. [Google Scholar] [CrossRef] [PubMed]
- Ghobadi, M.E.; Ghobadi, M.; Zebarjadi, A. Effect of waterlogging at different growth stages on some morphological traits of wheat varieties. Int. J. Biometeorol. 2017, 61, 635–645. [Google Scholar] [CrossRef]
- Seneviratne, M.; Rajakaruna, N.; Rizwan, M.; Madawala, H.M.S.P.; Ok, Y.S.; Vithanage, M. Heavy metal-induced oxidative stress on seed germination and seedling development: A critical review. Environ. Geochem. Health 2019, 41, 1813–1831. [Google Scholar] [CrossRef]
- Hura, T. Wheat and barley: Acclimatization to abiotic and biotic stress. Int. J. Mol. Sci. 2020, 21, 7423. [Google Scholar] [CrossRef]
- Rajjou, L.; Duval, M.; Gallardo, K.; Catusse, J.; Bally, J.; Job, C.; Job, D. Seed germination and vigor. Annu. Rev. Plant Biol. 2012, 63, 507–533. [Google Scholar] [CrossRef]
- Zhou, W.; Chen, F.; Zhao, S.; Yang, C.; Meng, Y.; Shuai, H.; Luo, X.; Dai, Y.; Yin, H.; Du, J.; et al. DA-6 promotes germination and seedling establishment from aged soybean seeds by mediating fatty acid metabolism and glycometabolism. J. Exp. Bot. 2019, 70, 101–114. [Google Scholar] [CrossRef]
- Zhang, M.; Li, B.; Wan, Z.; Chen, X.; Liu, C.; Liu, C.; Zhou, Y. Exogenous Spermidine Promotes Germination of Aged Sorghum Seeds by Mediating Sugar Metabolism. Plants 2022, 11, 2853. [Google Scholar] [CrossRef] [PubMed]
- Theodoulou, F.L.; Eastmond, P.J. Seed storage oil catabolism: A story of give and take. Curr. Opin. Plant Biol. 2012, 15, 322–328. [Google Scholar] [CrossRef] [PubMed]
- Quettier, A.L.; Shaw, E.; Eastmond, P.J. SUGAR-DEPENDENT6 encodes a mitochondrial flavin adenine dinucleotide-dependent glycerol-3-p dehydrogenase, which is required for glycerol catabolism and post germinative seedling growth in Arabidopsis. Plant Physiol. 2008, 148, 519–528. [Google Scholar] [CrossRef]
- Hurlock, A.K.; Roston, R.L.; Wang, K.; Benning, C. Lipid trafficking in plant cells. Traffic 2014, 15, 915–932. [Google Scholar] [CrossRef] [PubMed]
- Huang, Y.; Cai, S.; Ruan, X.; Xu, J.; Cao, D. CSN improves seed vigor of aged sunflower seeds by regulating the fatty acid, glycometabolism, and abscisic acid metabolism. J. Adv. Res. 2021, 33, 1–13. [Google Scholar] [CrossRef]
- Zhou, L.; Lu, L.; Chen, C.; Zhou, T.; Wu, Q.; Wen, F.; Chen, J.; Pritchard, H.W.; Peng, C.; Pei, J.; et al. Comparative changes in sugars and lipids show evidence of a critical node for regeneration in safflower seeds during aging. Front. Plant Sci. 2022, 13, 1020478. [Google Scholar] [CrossRef] [PubMed]
- Lv, T.; Li, J.; Zhou, L.; Zhou, T.; Pritchard, H.W.; Ren, C.; Chen, J.; Yan, J.; Pei, J. Aging-Induced Reduction in Safflower Seed Germination via Impaired Energy Metabolism and Genetic Integrity Is Partially Restored by Sucrose and DA-6 Treatment. Plants 2024, 13, 659. [Google Scholar] [CrossRef]
- Chen, C.; Wang, R.; Dong, S.; Wang, J.; Ren, C.X.; Chen, C.P.; Yan, J.; Zhou, T.; Wu, Q.H.; Pei, J.; et al. Integrated proteome and lipidome analysis of naturally aged safflower seeds varying in vitality. Plant Biol. 2022, 24, 266–277. [Google Scholar] [CrossRef]
- Li, L.; Sheen, J. Dynamic and diverse sugar signaling. Curr. Opin. Plant Biol. 2016, 33, 116–125. [Google Scholar] [CrossRef]
- Zhu, M.; Zang, Y.; Zhang, X.; Shang, S.; Xue, S.; Chen, J.; Tang, X. Insights into the regulation of energy metabolism during the seed-to-seedling transition in marine angiosperm Zostera marina L.: Integrated metabolomic and transcriptomic analysis. Front. Plant Sci. 2023, 14, 1130292. [Google Scholar] [CrossRef] [PubMed]
- Zhou, W.; Yang, Y.; Zheng, C.; Luo, X.; Chandrasekaran, U.; Yin, H.; Chen, F.; Meng, Y.; Chen, L.; Shu, K. Flooding represses soybean seed germination by mediating anaerobic respiration, glycometabolism and phytohormones biosynthesis. Environ. Exp. Bot. 2021, 188, 104491. [Google Scholar] [CrossRef]
- Bhattacharyya, S.; Sen-Mandi, S. Studies into causes of non-germination of aged wheat seeds. Ann. Bot. 1985, 56, 475–479. [Google Scholar] [CrossRef]
- Eastmond, P.J.; Germain, V.; Lange, P.R.; Bryce, J.H.; Smith, S.M.; Graham, I.A. Postgerminative growth and lipid catabolism in oilseeds lacking the glyoxylate cycle. Proc. Natl. Acad. Sci. USA 2000, 97, 5669–5674. [Google Scholar] [CrossRef] [PubMed]
- Borek, S.; Nuc, K. Sucrose controls storage lipid breakdown on gene expression level in germinating yellow lupine (Lupinus luteus L.) seeds. J. Plant Physiol. 2011, 168, 1795–1803. [Google Scholar] [CrossRef]
- Wang, B.; Zhang, Y.; Haque, M.E.; Xu, W.; Li, F.; Liu, A. Transcriptomic analyses reveal complex and interconnected sucrose signaling cascades in developing seeds of castor bean. J. Plant Physiol. 2018, 221, 1–10. [Google Scholar] [CrossRef]
- Yu, J.; Lee, H.; Heo, H.; Jeong, H.S.; Sung, J.; Lee, J. Sucrose-induced abiotic stress improves the phytochemical profiles and bioactivities of mung bean sprouts. Food Chem. 2023, 400, 134069. [Google Scholar] [CrossRef]
- Lara-Núñez, A.; García-Ayala, B.B.; Garza-Aguilar, S.M.; Flores-Sánchez, J.; Sánchez-Camargo, V.A.; Bravo-Alberto, C.E.; Vázquez-Santana, S.; Vázquez-Ramos, J.M. Glucose and sucrose differentially modify cell proliferation in maize during germination. Plant Physiol. Biochem. 2017, 113, 20–31. [Google Scholar] [CrossRef]
- Cui, S.; Hayashi, Y.; Otomo, M.; Mano, S.; Oikawa, K.; Hayashi, M.; Nishimura, M. Sucrose Production Mediated by Lipid Metabolism Suppresses the Physical Interaction of Peroxisomes and Oil Bodies during Germination of Arabidopsis thaliana. J. Biol. Chem. 2016, 291, 19734–19745. [Google Scholar] [CrossRef]
- Galland, M.; Boutet-Mercey, S.; Lounifi, I.; Godin, B.; Balzergue, S.; Grandjean, O.; Morin, H.; Perreau, F.; Debeaujon, I.; Rajjou, L. Compartmentation and dynamics of flavone metabolism in dry and germinated rice seeds. Plant Cell Physiol. 2014, 55, 1646–1659. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Jiang, W.; Li, C.; Wang, Z.; Lu, C.; Cheng, J.; Wei, S.; Yang, J.; Yang, Q. Integrated transcriptomic and metabolomic analyses elucidate the mechanism of flavonoid biosynthesis in the regulation of mulberry seed germination under salt stress. BMC Plant Biol. 2024, 24, 132. [Google Scholar] [CrossRef] [PubMed]
- Kalaivani, V.; Nikarika, R.; Shoma, N.; Arunraj, R. Delayed hydrolysis of Raffinose Family Oligosaccharides (RFO) affects critical germination of chickpeas. 3 Biotech 2021, 11, 298. [Google Scholar] [CrossRef] [PubMed]
- Formela-Luboińska, M.; Chadzinikolau, T.; Drzewiecka, K.; Jeleń, H.; Bocianowski, J.; Kęsy, J.; Labudda, M.; Jeandet, P.; Morkunas, I. The Role of Sugars in the Regulation of the Level of Endogenous Signaling Molecules during Defense Response of Yellow Lupine to Fusarium oxysporum. Int. J. Mol. Sci. 2020, 21, 4133. [Google Scholar] [CrossRef]
- Salam, B.B.; Barbier, F.; Danieli, R.; Teper-Bamnolker, P.; Ziv, C.; Spíchal, L.; Aruchamy, K.; Shnaider, Y.; Leibman, D.; Shaya, F.; et al. Sucrose promotes stem branching through cytokinin. Plant Physiol. 2021, 185, 1708–1721. [Google Scholar] [CrossRef]
- Oracz, K.; Stawska, M. Cellular Recycling of Proteins in Seed Dormancy Alleviation and Germination. Front. Plant Sci. 2016, 7, 1128. [Google Scholar] [CrossRef]
- Galland, M.; Huguet, R.; Arc, E.; Cueff, G.; Job, D.; Rajjou, L. Dynamic proteomics emphasizes the importance of selective mRNA translation and protein turnover during Arabidopsis seed germination. Mol. Cell. Proteom. 2014, 13, 252–268. [Google Scholar] [CrossRef]
- Huang, Y.; Mei, G.; Fu, X.; Wang, Y.; Ruan, X.; Cao, D. Ultrasonic Waves Regulate Antioxidant Defense and Gluconeogenesis to Improve Germination From Naturally Aged Soybean Seeds. Front. Plant Sci. 2022, 13, 833858. [Google Scholar] [CrossRef]
- Hu, X.; Zhou, Q. Novel hydrated graphene ribbon unexpectedly promotes aged seed germination and root differentiation. Sci. Rep. 2014, 4, 3782. [Google Scholar] [CrossRef]
- Nile, S.H.; Thiruvengadam, M.; Wang, Y.; Samynathan, R.; Shariati, M.A.; Rebezov, M.; Nile, A.; Sun, M.; Venkidasamy, B.; Xiao, J.; et al. Nano-priming as emerging seed priming technology for sustainable agriculture-recent developments and future perspectives. J. Nanobiotechnol. 2022, 20, 254. [Google Scholar] [CrossRef]
- Li, Y.; Liang, L.; Li, W.; Ashraf, U.; Ma, L.; Tang, X.; Pan, S.; Tian, H.; Mo, Z. ZnO nanoparticle-based seed priming modulates early growth and enhances physio-biochemical and metabolic profiles of fragrant rice against cadmium toxicity. J. Nanobiotechnol. 2021, 19, 75. [Google Scholar] [CrossRef]
- Gupta, N.; Singh, P.M.; Sagar, V.; Pandya, A.; Chinnappa, M.; Kumar, R.; Bahadur, A. Seed Priming with ZnO and Fe3O4 Nanoparticles Alleviate the Lead Toxicity in Basella alba L. through Reduced Lead Uptake and Regulation of ROS. Plants 2022, 11, 2227. [Google Scholar] [CrossRef]
- Basnet, R.K.; Del Carpio, D.P.; Xiao, D.; Bucher, J.; Jin, M.; Boyle, K.; Fobert, P.; Visser, R.G.; Maliepaard, C.; Bonnema, G. A Systems Genetics Approach Identifies Gene Regulatory Networks Associated with Fatty Acid Composition in Brassica rapa Seed. Plant Physiol. 2016, 170, 568–585. [Google Scholar] [CrossRef] [PubMed]
- Faraoni, P.; Sereni, E.; Gnerucci, A.; Cialdai, F.; Monici, M.; Ranaldi, F. Glyoxylate cycle activity in Pinus pinea seeds during germination in altered gravity conditions. Plant Physiol. Biochem. 2019, 139, 389–394. [Google Scholar] [CrossRef] [PubMed]
- Adham, A.R.; Zolman, B.K.; Millius, A.; Bartel, B. Mutations in Arabidopsis acyl-CoA oxidase genes reveal distinct and overlapping roles in beta-oxidation. Plant J. 2005, 41, 859–874. [Google Scholar] [CrossRef]
- Eastmond, P.J.; Graham, I.A. The multifunctional protein AtMFP2 is co-ordinately expressed with other genes of fatty acid beta-oxidation during seed germination in Arabidopsis thaliana (L.). Heynh. Biochem. Soc. Trans. 2000, 28, 95–99. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Liu, Y.; Zolman, B.K. Metabolic Alterations in the Enoyl-CoA Hydratase 2 Mutant Disrupt Peroxisomal Pathways in Seedlings. Plant Physiol. 2019, 180, 1860–1876. [Google Scholar] [CrossRef] [PubMed]
- Delker, C.; Zolman, B.K.; Miersch, O.; Wasternack, C. Jasmonate biosynthesis in Arabidopsis thaliana requires peroxisomal beta-oxidation enzymes--additional proof by properties of pex6 and aim1. Phytochemistry 2007, 68, 1642–1650. [Google Scholar] [CrossRef] [PubMed]
- Pinfield-Wells, H.; Rylott, E.L.; Gilday, A.D.; Graham, S.; Job, K.; Larson, T.R.; Graham, I.A. Sucrose rescues seedling establishment but not germination of Arabidopsis mutants disrupted in peroxisomal fatty acid catabolism. Plant J. 2005, 43, 861–872. [Google Scholar] [CrossRef]
- Rylott, E.L.; Eastmond, P.J.; Gilday, A.D.; Slocombe, S.P.; Larson, T.R.; Baker, A.; Graham, I.A. The Arabidopsis thaliana multifunctional protein gene (MFP2) of peroxisomal beta-oxidation is essential for seedling establishment. Plant J. 2006, 45, 930–941. [Google Scholar] [CrossRef]
- Domergue, J.B.; Abadie, C.; Limami, A.; Way, D.; Tcherkez, G. Seed quality and carbon primary metabolism. Plant Cell Environ. 2019, 42, 2776–2788. [Google Scholar] [CrossRef]
- Graham, I.A. Seed storage oil mobilization. Annu. Rev. Plant Biol. 2008, 59, 115–142. [Google Scholar] [CrossRef]
- Nigam, M.; Mishra, A.P.; Salehi, B.; Kumar, M.; Sahrifi-Rad, M.; Coviello, E.; Iriti, M.; Sharifi-Rad, J. Accelerated ageing induces physiological and biochemical changes in tomato seeds involving MAPK pathways. Sci. Hortic. 2019, 248, 20–28. [Google Scholar] [CrossRef]
- Zhang, W.; Mace, W.J.; Matthew, C.; Card, S.D. The Impact of Endophyte Infection, Seed Aging, and Imbibition on Selected Sugar Metabolite Concentrations in Seed. J. Agric. Food Chem. 2019, 67, 6921–6929. [Google Scholar] [CrossRef]
- Bernal-Lugo, I.; Leopold, A.C. Changes in Soluble Carbohydrates during Seed Storage. Plant Physiol. 1992, 98, 1207–1210. [Google Scholar] [CrossRef]
- Pereira Lima, J.J.; Buitink, J.; Lalanne, D.; Rossi, R.F.; Pelletier, S.; da Silva, E.A.A.; Leprince, O. Molecular characterization of the acquisition of longevity during seed maturation in soybean. PLoS ONE 2017, 12, e0180282. [Google Scholar] [CrossRef]
- Li, W.; Huang, L.; Liu, N.; Pandey, M.K.; Chen, Y.; Cheng, L.; Guo, J.; Yu, B.; Luo, H.; Zhou, X.; et al. Key Regulators of Sucrose Metabolism Identified through Comprehensive Comparative Transcriptome Analysis in Peanuts. Int. J. Mol. Sci. 2021, 22, 7266. [Google Scholar] [CrossRef] [PubMed]
- Eastmond, P.J.; Astley, H.M.; Parsley, K.; Aubry, S.; Williams, B.P.; Menard, G.N.; Craddock, C.P.; Nunes-Nesi, A.; Fernie, A.R.; Hibberd, J.M. Arabidopsis uses two gluconeogenic gateways for organic acids to fuel seedling establishment. Nat. Commun. 2015, 6, 6659. [Google Scholar] [CrossRef]
- Silva, M.F.D.; Soares, J.M.; Xavier, W.A.; Silva, F.C.D.S.; Silva, F.L.D.; Silva, L.J.D. The role of the biochemical composition of soybean seeds in the tolerance to deterioration under natural and artificial aging. Heliyon 2023, 9, e21628. [Google Scholar] [CrossRef]
- Diantina, S.; McGill, C.; Millner, J.; Nadarajan, J.; Pritchard, H.W.; Colville, L.; Clavijo McCormick, A. Seed viability and fatty acid profiles of five orchid species before and after ageing. Plant Biol. 2022, 24, 168–175. [Google Scholar] [CrossRef]
- Kozuka, T.; Sawada, Y.; Imai, H.; Kanai, M.; Hirai, M.Y.; Mano, S.; Uemura, M.; Nishimura, M.; Kusaba, M.; Nagatani, A. Regulation of Sugar and Storage Oil Metabolism by Phytochrome during De-etiolation. Plant Physiol. 2020, 182, 1114–1129. [Google Scholar] [CrossRef]
- Hayashi, Y.; Sato, N.; Shinozaki, A.; Watanabe, M. Increase in peroxisome number and the gene expression of putative glyoxysomal enzymes in Chlamydomonas cells supplemented with acetate. J. Plant Res. 2015, 128, 177–185. [Google Scholar] [CrossRef] [PubMed]
- Zhao, S.; Garcia, D.; Zhao, Y.; Huang, D. Hydro-Electro Hybrid Priming Promotes Carrot (Daucus carota L.) Seed Germination by Activating Lipid Utilization and Respiratory Metabolism. Int. J. Mol. Sci. 2021, 22, 11090. [Google Scholar] [CrossRef] [PubMed]
- Wu, W.L.; Hsiao, Y.Y.; Lu, H.C.; Liang, C.K.; Fu, C.H.; Huang, T.H.; Chuang, M.H.; Chen, L.J.; Liu, Z.J.; Tsai, W.C. Expression regulation of MALATE SYNTHASE involved in glyoxylate cycle during protocorm development in Phalaenopsis aphrodite (Orchidaceae). Sci. Rep. 2020, 10, 10123. [Google Scholar] [CrossRef]
- Ma, Z.; Marsolais, F.; Bernards, M.A.; Sumarah, M.W.; Bykova, N.V.; Igamberdiev, A.U. Glyoxylate cycle and metabolism of organic acids in the scutellum of barley seeds during germination. Plant Sci. 2016, 248, 37–44. [Google Scholar] [CrossRef]
- Eastmond, P.J. SUGAR-DEPENDENT1 encodes a patatin domain triacylglycerol lipase that initiates storage oil breakdown in germinating Arabidopsis seeds. Plant Cell 2006, 18, 665–675. [Google Scholar] [CrossRef] [PubMed]
- Campobenedetto, C.; Grange, E.; Mannino, G.; van Arkel, J.; Beekwilder, J.; Karlova, R.; Garabello, C.; Contartese, V.; Bertea, C.M. A Biostimulant Seed Treatment Improved Heat Stress Tolerance During Cucumber Seed Germination by Acting on the Antioxidant System and Glyoxylate Cycle. Front. Plant Sci. 2020, 11, 836. [Google Scholar] [CrossRef]
- Song, Y.; Gao, X.; Wu, Y. Key Metabolite Differences Between Korean Pine (Pinus koraiensis) Seeds With Primary Physiological Dormancy and No-Dormancy. Front. Plant Sci. 2021, 12, 767108. [Google Scholar] [CrossRef]
- He, Y.; Cheng, J.; He, Y.; Yang, B.; Cheng, Y.; Yang, C.; Zhang, H.; Wang, Z. Influence of isopropylmalate synthase OsIPMS1 on seed vigour associated with amino acid and energy metabolism in rice. Plant Biotechnol. J. 2019, 17, 322–337. [Google Scholar] [CrossRef] [PubMed]
- Yang, B.; Chen, M.; Zhan, C.; Liu, K.; Cheng, Y.; Xie, T.; Zhu, P.; He, Y.; Zeng, P.; Tang, H.; et al. Identification of OsPK5 involved in rice glycolytic metabolism and GA/ABA balance for improving seed germination via genome-wide association study. J. Exp. Bot. 2022, 73, 3446–3461. [Google Scholar] [CrossRef]
- Guo, Z.; Zhao, J.; Wang, M.; Song, S.; Xia, Z. Sulfur dioxide promotes seed germination by modulating reactive oxygen species production in maize. Plant Sci. 2021, 312, 111027. [Google Scholar] [CrossRef] [PubMed]
- Lei, K.; Sun, S.; Zhong, K.; Li, S.; Hu, H.; Sun, C.; Zheng, Q.; Tian, Z.; Dai, T.; Sun, J. Seed soaking with melatonin promotes seed germination under chromium stress via enhancing reserve mobilization and antioxidant metabolism in wheat. Ecotoxicol. Environ. Saf. 2021, 220, 112241. [Google Scholar] [CrossRef] [PubMed]
- Chen, L.; Lu, B.; Liu, L.; Duan, W.; Jiang, D.; Li, J.; Zhang, K.; Sun, H.; Zhang, Y.; Li, C.; et al. Melatonin promotes seed germination under salt stress by regulating ABA and GA3 in cotton (Gossypium hirsutum L.). Plant Physiol. Biochem. 2021, 162, 506–516. [Google Scholar] [CrossRef] [PubMed]
- Ventura, L.; Donà, M.; Macovei, A.; Carbonera, D.; Buttafava, A.; Mondoni, A.; Rossi, G.; Balestrazzi, A. Understanding the molecular pathways associated with seed vigor. Plant Physiol. Biochem. 2012, 60, 196–206. [Google Scholar] [CrossRef]
- Kiran, K.R.; Deepika, V.B.; Swathy, P.S.; Prasad, K.; Kabekkodu, S.P.; Murali, T.S.; Satyamoorthy, K.; Muthusamy, A. ROS-dependent DNA damage and repair during germination of NaCl primed seeds. J. Photochem. Photobiol. B 2020, 213, 112050. [Google Scholar] [CrossRef]
- Forti, C.; Shankar, A.; Singh, A.; Balestrazzi, A.; Prasad, V.; Macovei, A. Hydropriming and Biopriming Improve Medicago truncatula Seed Germination and Upregulate DNA Repair and Antioxidant Genes. Genes 2020, 11, 242. [Google Scholar] [CrossRef]
- Millennium Seed Bank Partnership. Germination Testing: Procedures and Evaluation. 2022. Available online: https://brahmsonline.kew.org/Content/Projects/msbp/resources/Training/13a-Germination-testing-procedures.pdf (accessed on 11 July 2025).
- Zhou, T.; Qiu, X.; Zhao, L.; Yang, W.; Wen, F.; Wu, Q.; Yan, J.; Xu, B.; Chen, J.; Ma, Y.; et al. Optimal light intensity and quality increased the saffron daughter corm yield by inhibiting the degradation of reserves in mother corms during the reproductive stage. Ind. Crops Prod. 2022, 176, 114396. [Google Scholar] [CrossRef]
- Yang, C.; Iqbal, N.; Hu, B.; Zhang, Q.; Wu, H.; Liu, X.; Zhang, J.; Liu, W.; Yang, W.; Liu, J. Targeted metabonomics analysis of fatty acid in soybean seed by GC-MS reveal the metabolic manipulation of shading in intercropping system. Anal. Methods 2017, 9, 2144–2152. [Google Scholar] [CrossRef]
- Wu, Z.; Liu, H.; Zhan, W.; Yu, Z.; Qin, E.; Liu, S.; Yang, T.; Xiang, N.; Kudrna, D.; Chen, Y.; et al. The chromosome-scale reference genome of safflower (Carthamus tinctorius L.) provides insights into linoleic acid and flavonoid biosynthesis. Plant Biotechnol. J. 2021, 19, 1725–1742. [Google Scholar] [CrossRef] [PubMed]
- Livak, K.J.; Schmittgen, T.D. Analysis of relative gene expression data using real-time quantitative PCR and the 2−ΔΔCT method. Methods 2001, 25, 402–408. [Google Scholar] [CrossRef] [PubMed]

Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lv, T.; Zhong, L.; Li, J.; Chen, C.; Xian, B.; Zhou, T.; Ren, C.; Chen, J.; Pei, J.; Yan, J. Exogenous Sucrose Improves the Vigor of Aged Safflower Seeds by Mediating Fatty Acid Metabolism and Glycometabolism. Plants 2025, 14, 2301. https://doi.org/10.3390/plants14152301
Lv T, Zhong L, Li J, Chen C, Xian B, Zhou T, Ren C, Chen J, Pei J, Yan J. Exogenous Sucrose Improves the Vigor of Aged Safflower Seeds by Mediating Fatty Acid Metabolism and Glycometabolism. Plants. 2025; 14(15):2301. https://doi.org/10.3390/plants14152301
Chicago/Turabian StyleLv, Tang, Lin Zhong, Juan Li, Cuiping Chen, Bin Xian, Tao Zhou, Chaoxiang Ren, Jiang Chen, Jin Pei, and Jie Yan. 2025. "Exogenous Sucrose Improves the Vigor of Aged Safflower Seeds by Mediating Fatty Acid Metabolism and Glycometabolism" Plants 14, no. 15: 2301. https://doi.org/10.3390/plants14152301
APA StyleLv, T., Zhong, L., Li, J., Chen, C., Xian, B., Zhou, T., Ren, C., Chen, J., Pei, J., & Yan, J. (2025). Exogenous Sucrose Improves the Vigor of Aged Safflower Seeds by Mediating Fatty Acid Metabolism and Glycometabolism. Plants, 14(15), 2301. https://doi.org/10.3390/plants14152301





