Main Poisonous and Allergenic Plant Species in Sicilian Gardens and Parks: Applications and Recommendations for Use
Abstract
1. Introduction
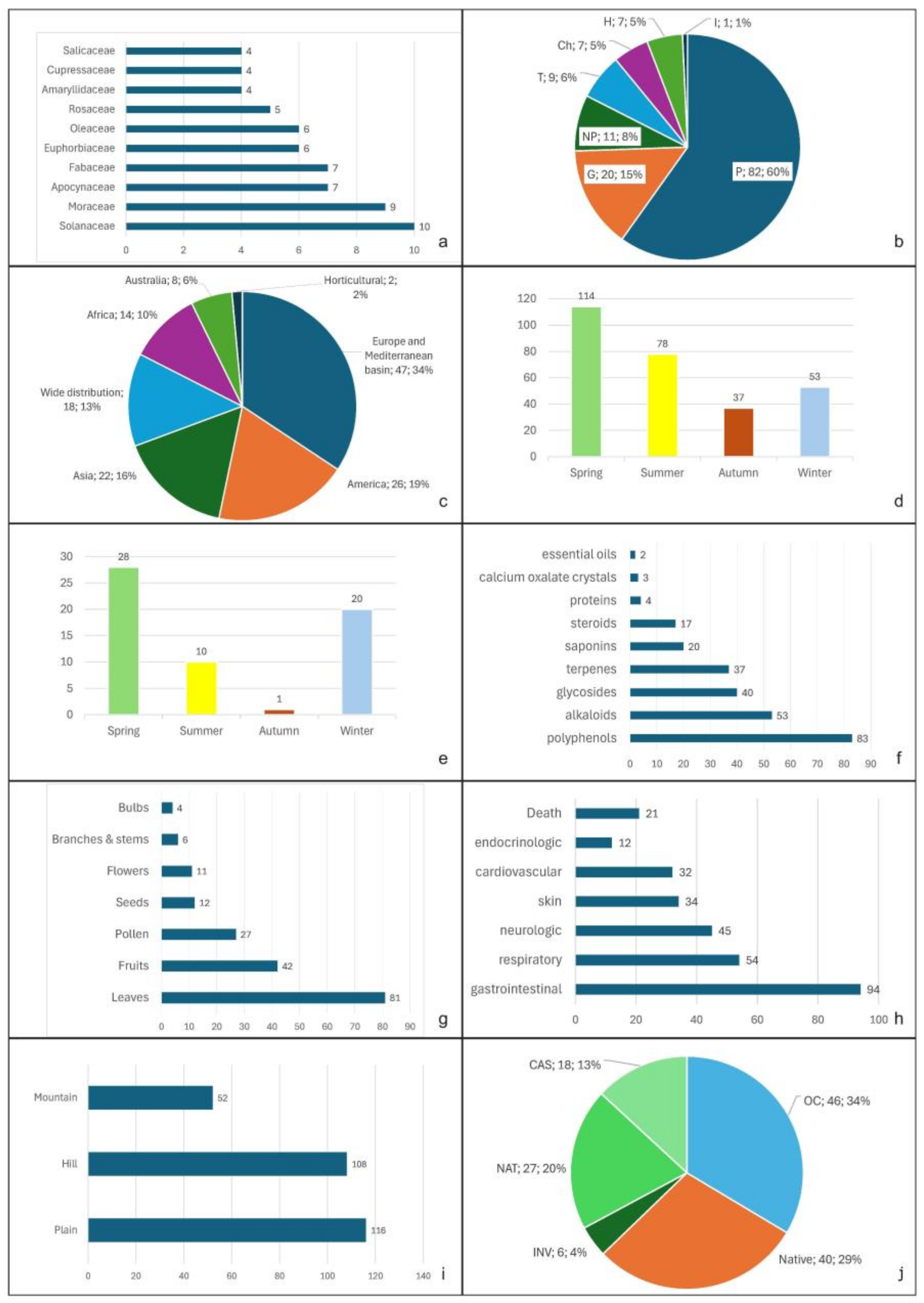
2. Results
3. Discussion and Recommendations
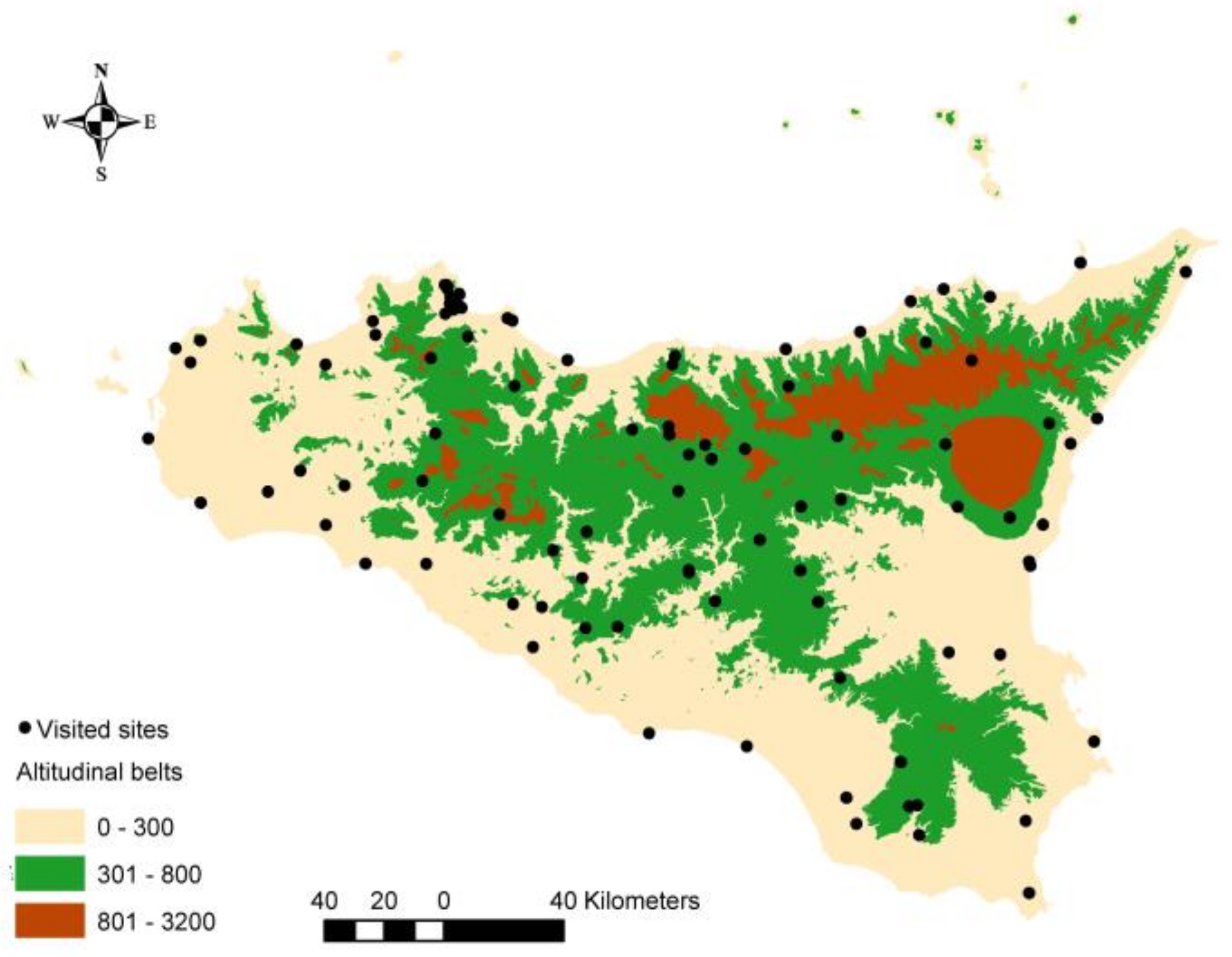
4. Materials and Methods
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Martinico, F.; La Rosa, D.; Privitera, R. Green oriented urban development for urban ecosystem services provision in a medium sized city in southern Italy. iForest 2014, 7, 385–395. [Google Scholar] [CrossRef]
- Bulgarelli, G.; Flamigni, S. Le Piante Tossiche E Velenose; Hoepli: Milano, Italy, 2010; 192p. [Google Scholar]
- Luzzi, P. Piante Ornamentali Velenose; Edagricole: Bologna, Italy, 1992; p. 148. [Google Scholar]
- Moro, P.A.; Assisi, F.; Cassetti, F.; Bissoli, M.; Borghini, R.; Davanzo, F.; Della Puppa, T.; Dimasi, V.; Ferruzzi, M.; Giarratana, T.; et al. Toxicological Hazards of Natural Environments: Clinical Reports from Poison Control Centre of Milan. Urban For. Urban Green. 2009, 8, 179–186. [Google Scholar] [CrossRef]
- Benigni, R.; Capra, C.; Cattorini, P.E. Piante Medicinali: Chimica Farmacologica e Terapia; Inverni & Della Beffa: Milano, Italy, 1962; p. 730. [Google Scholar]
- Al-Snafi, A.E. Pharmacology and Toxicology of Conium maculatum—A Review. Pharm. Chem. J. 2016, 3, 136–142. [Google Scholar]
- Mondal, J.; Panigrahi, A.K.; Khuda-Bukhsh, A.R. Anticancer Potential of Conium maculatum Extract against Cancer Cells In Vitro: Drug-DNA Interaction and Its Ability to Induce Apoptosis through ROS Generation. Pharmacogn. Mag. 2014, 10, S524–S533. [Google Scholar] [CrossRef]
- Ginsburg, H.; Deharo, E. A Call for Using Natural Compounds in the Development of New Antimalarial Treatments—An Introduction. Malar. J. 2011, 10, S1. [Google Scholar] [CrossRef]
- Vaou, N.; Stavropoulou, E.; Voidarou, C.; Tsakris, Z.; Rozos, G.; Tsigalou, C.; Bezirtzoglou, E. Interactions between Medical Plant-Derived Bioactive Compounds: Focus on Antimicrobial Combination Effects. Antibiotics 2022, 11, 1014. [Google Scholar] [CrossRef]
- Masanotti, G.M.; Abbafati, E.; Petrella, E.; Vinciguerra, S.; Stracci, F. Intensive Tobacco Cultivations, a Possible Public Health Risk? Environ. Sci. Pollut. Res. 2019, 26, 12616–12621. [Google Scholar] [CrossRef]
- Onat, F.Y.; Yegen, B.C.; Lawrence, R.; Oktay, A.; Oklay, S. Mad Honey Poisoning in Man and Rat. Rev. Environ. Health 1991, 9, 3–10. [Google Scholar] [CrossRef]
- Mazokopakis, E.E.; Karagiannis, C.G. Coturnism as a Cause of Deadly Rhabdomyolysis in Biblical Times. Clin. Kidney J. 2022, 15, 1019–1020. [Google Scholar] [CrossRef]
- Yan, S.; Wang, K.; Al Naggar, Y.; Vander Heyden, Y.; Zhao, L.; Wu, L.; Xue, X. Natural Plant Toxins in Honey: An Ignored Threat to Human Health. J. Hazard. Mater. 2022, 424, 127682. [Google Scholar] [CrossRef]
- Banfi, E.; Colombo, M.L.; Davanzo, F.; Falciola, C.; Galasso, G.; Martino, E.; Perego, S. Piante velenose della Flora italiana nell’esperienza del Centro Antiveleni di Milano. Natura 2012, 102, 1–184. [Google Scholar]
- Botti, P.; Cipriani, F.; Dannaoui, B.; Bravi, S.; Missanelli, A. Gruppo Epintox Intossicazioni acute e avvelenamenti nei Dipartimenti di Emergenza e Urgenza in Italia. Ann. dell’Ist. Super. Sanità 2006, 42, 287–297. [Google Scholar]
- Milewski, L.M.; Khan, S.A. An Overview of Potentially Life-Threatening Poisonous Plants in Dogs and Cats. J. Vet. Emerg. Crit. Care 2006, 16, 25–33. [Google Scholar] [CrossRef]
- Stach, A.; Emberlin, J.; Smith, M.; Adams-Groom, B.; Myszkowskaet, D. Factors that determine the severity of Betula spp. pollen seasons in Poland (Poznań and Krakow) and the United Kingdom (Worcester and London). Int. J. Biometeorol. 2008, 52, 311–321. [Google Scholar] [CrossRef]
- Frenguelli, G.; Passaleva, A. La scelta delle piante destinate al verde ornamentale. Giorn It Allergol. Immunol. Clin. 2003, 13, 177–191. [Google Scholar]
- Lauriola, P.; Talluri, M. Tutti allergici? Il ruolo dei medici sentinella per l’ambiente per la prevenzione delle malattie allergiche respiratorie. Cesalpino 2022, 55, 10–14. [Google Scholar]
- Sebald, V.; Schmack, J.; Egerer, M. Occurrence and Diversity of Poisonous Plants in Urban Community Gardens. Renew. Agric. Food Syst. 2023, 38, e30. [Google Scholar] [CrossRef]
- Židovec, V.; Jarić, J.; Poje, M.; Dujmović Purgar, D. Poisonous and Allergenic Plant Species in Kindergarten Gardens in Novi Zagreb City Districts. J. Cent. Eur. Agric. 2023, 24, 245–259. [Google Scholar] [CrossRef]
- Akdeniz, N.S.; Zencirkiran, M. An Evaluation of Toxic Properties of Woody Landscape Plants Used in Hospital Garden Design. HERD 2024, 17, 164–176. [Google Scholar] [CrossRef]
- Leporatti, M.L.; Guarrera, P.M.; De Giacomo, M. Wild and ornamental toxic plants in Latium region (Central Italy). Fitoterapia 1996, 67, 513–534. [Google Scholar]
- Aleo, N.; Amato, F.; Aleo, M. Le piante tossiche della flora trapanese (Sicilia). Quad. Bot. Ambient. Appl. 2011, 22, 31–49. [Google Scholar]
- Rispo, M.; De Masi, L.; Calandrelli, M.M. Assessment of Allergenic Potential in Urban Forests: A Case Study of the Royal Park of Portici in Southern Italy. iForest 2020, 13, 376–381. [Google Scholar] [CrossRef]
- Cariñanos, P.; Marinangeli, F. An Updated Proposal of the Potential Allergenicity of 150 Ornamental Trees and Shrubs in Mediterranean Cities. Urban For. Urban Green. 2021, 63, 127218. [Google Scholar] [CrossRef]
- Ciccarello, S.; Mazzola, P.; Spadaro, V. Allergens in the park and garden flora of Sicily. In Proceedings of the 112° Congresso della Società Botanica Italiana IV International Plant Science Conference (IPSC), Parma, Italy, 20–23 September 2017; p. 31. [Google Scholar]
- Istituto Nazionale di Statistica. Available online: https://www.istat.it (accessed on 12 March 2024).
- Basilone, L. Lithostratigraphy of Sicily; Springer: Berlin, Germany, 2018; p. 349. [Google Scholar] [CrossRef]
- Bazan, G.; Marino, P.; Guarino, R.; Domina, G.; Schicchi, R. Bioclimatology and vegetation series in Sicily: A geostatistical approach. Ann. Bot. Fenn. 2015, 52, 1–18. [Google Scholar] [CrossRef]
- Domina, G.; Di Gristina, E.; Scafidi, F.; Calvo, R.; Venturella, G.; Gargano, M. The Urban Vascular Flora of Palermo (Sicily, Italy). Plant Biosyst. 2019, 154, 627–634. [Google Scholar] [CrossRef]
- Bazan, G.; Geraci, A.; Raimondo, F.M. La componente floristica dei giardini storici siciliani. Quad. Bot. Ambient. Appl. 2005, 16, 93–126. [Google Scholar]
- Gastaldo, P. Compendio della Flora Officinale Italiana; Piccin: Padova, Italy, 1987; p. 523. [Google Scholar]
- Bruneton, J. Pharmacognosy, Phytochemistry, Medicinal Plants; Lavisior: Paris, France, 1995; p. 915. [Google Scholar]
- Galliano Raspino, M. (Ed.) Repertorio Fitoterapico, 2nd ed.; OEMF: Milano, Italy, 1996; p. 778. [Google Scholar]
- Pedretti, M. Chimica e Farmacologia delle Piante Medicinali; Studio Edizioni: Milano, Italy, 1997; p. 159. [Google Scholar]
- Poppenga, R.H. Poisonous Plants. In Molecular, Clinical and Environmental Toxicology: Volume 2: Clinical Toxicology; Luch, A., Ed.; Experientia Supplementum; Birkhäuser: Basel, Switzerland, 2010; pp. 123–175. [Google Scholar]
- Colombo, M.L.; Assisi, F.; Puppa, T.D.; Moro, P.; Bissoli, M.; Borghini, R.; Perego, S.; Galasso, G.; Davanzo, F. Most Commonly Plant Exposures and Intoxications from Outdoor Toxic Plants. J. Pharm. Sci. 2010, 2, 417–425. [Google Scholar]
- D’Amato, G.; Spieksma, F.T.M.; Bonini, S. Allergenic Pollen and Pollinosis in Europe; Blackwell Scientific Publications: Oxford, UK, 1991; 226p. [Google Scholar]
- Feliziani, V. Pollini di Interesse Allergologico: Guida al Loro Riconoscimento; Masson: Milano, Italy, 1986; 195p. [Google Scholar]
- Ciampolini, F.; Cresti, M. Atlante dei Principali Pollini Allergenici Presenti in Italia; Università di Siena: Siena, Italy, 1981; 190p. [Google Scholar]
- Raunkiaer, C. The Life Form of Plants and Statistical Geography; Claredon: Exford, UK, 1934; p. 632. [Google Scholar]
- POWO. Plants of the World Online. Facilitated by the Royal Botanic Gardens, Kew. Available online: http://www.plantsoftheworldonline.org (accessed on 2 January 2024).
- Dumitra¸s, D.-A.; Bunea, A.; Vodnar, D.C.; Hanganu, D.; Pall, E.; Cenariu, M.; Gal, A.F.; Andrei, S. Phytochemical Characterizationof Taxus baccata L. Aril with Emphasis on Evaluation of the Antiproliferative and Pro-Apoptotic Activity of Rhodoxanthin. Antioxidants 2022, 11, 1039. [Google Scholar] [CrossRef]
- Di Tizio, A.; Łuczaj, T.J.; Quave, C.L.; Redži´c, S.; Pieroni, A. Traditional Food and Herbal Uses of Wild Plants in the Ancient South-Slavic Diaspora of Mundimitar/Montemitro (Southern Italy). J. Ethnobiol. Ethnomed. 2012, 8, 21. [Google Scholar] [CrossRef]
- Southcott, R.V.; Haegi, L.A.R. Plant hair dermatitis. Med. J. Aust. 1992, 156, 623–624+627–629+631–632. [Google Scholar] [CrossRef]
- Rajhans, S.; Pandya, H.; Kumar, S.P.; Bhadresha, K.; Yadav, D.K.; Rawal, R.; Ansari, H.; Dave, R.; Sindhav, G. Assessment of Cytotoxic Effects of Latex from Cascabela thevetia (L.) Lippold and Plumeria alba L. via In vitro and In silico Approaches. J. Nat. Remedies 2023, 23, 89–104. [Google Scholar] [CrossRef]
- Mohammad, H.; Alzweiri, M. Phytochemistry and pharmacological activities of Ficus carica latex: A systematic review. J. Chin. Pharm. Sci. 2022, 31, 81–96. [Google Scholar] [CrossRef]
- Galasso, G.; Conti, F.; Peruzzi, L.; Alessandrini, A.; Ardenghi, N.M.G.; Bacchetta, G.; Banfi, E.; Barberis, L.; Bernardo, L.; Bouvet, D.; et al. A second update to the checklist of the vascular flora alien to Italy. Plant Biosyst. 2024, 1–44. [Google Scholar] [CrossRef]
- Bartolucci, F.; Peruzzi, L.; Galasso, G.; Alessandrini, A.; Ardenghi, N.M.G.; Bacchetta, G.; Banfi, E.; Barberis, G.; Bernardo, L.; Bouvet, D.; et al. A second update to the checklist of the vascular flora native to Italy. Plant Biosyst. 2024, 1–78. [Google Scholar] [CrossRef]
- Ghisleni, G.; Caretto, F. Alcune piante d’appartamento tossiche per il cane e il gatto. Veterinaria 1999, 13, 65–73. [Google Scholar]
- Botha, C.J.; Penrith, M.-L. Potential Plant Poisonings in Dogs and Cats in Southern Africa: Review Article. J. S. Afr. Vet. Assoc. 2009, 80, 63–74. [Google Scholar] [CrossRef] [PubMed]
- Berny, P.; Caloni, F.; Croubels, S.; Sachana, M.; Vandenbroucke, V.; Davanzo, F.; Guitart, R. Animal Poisoning in Europe. Part 2: Companion Animals. Vet. J. 2010, 183, 255–259. [Google Scholar] [CrossRef] [PubMed]
- Caloni, F.; Cortinovis, C.; Rivolta, M.; Alonge, S.; Davanzo, F. Plant Poisoning in Domestic Animals: Epidemiological Data from an Italian Survey (2000–2011). Vet. Rec. 2013, 172, 580. [Google Scholar] [CrossRef]
- Anadón, A.; Martínez-Larrañaga, M.R.; Ares, I.; Martínez, M.A. Chapter 62—Poisonous Plants of the Europe. In Veterinary Toxicology, 3rd ed.; Gupta, R.C., Ed.; Academic Press: Cambridge, MA, USA, 2018; pp. 891–909. [Google Scholar]
- Devarinti, S.R. Pollen Allergy: Common Weeds in Telangana and Their Management Measures. JBFBP 2015, 6, 2. [Google Scholar] [CrossRef]
- Leveau, P. Intoxications des enfants par les plantes. J. Pédiatr. Puéric. 2021, 34, 237–243. [Google Scholar] [CrossRef]
- Davanzo, F.; Miaglia, S.; Perego, S.; Assisi, F.; Bissoli, M.; Borghini, R.; Cassetti, F.; Puppa, T.D.; Dimasi, V.; Falciola, C.; et al. Plant Poisoning: Increasing Relevance, a Problem of Public Health and Education. North-Western Italy, Piedmont Region. J. Pharm. Sci. Res. 2011, 3, 1338–1343. [Google Scholar]
- Di Gristina, E.; Domina, G.; Barone, G. The Alien Vascular Flora of Stromboli and Vulcano (Aeolian Islands, Italy). Ital. Bot. 2021, 12, 63–75. [Google Scholar] [CrossRef]
- Bolarinwa, I.F.; Orfila, C.; Morgan, M.R. Determination of amygdalin in apple seeds, fresh apples and processed apple juices. Food Chem. 2015, 170, 437–442. [Google Scholar] [CrossRef] [PubMed]
- Mazzola, P.; Di Martino, C. La florula decorativa del promontorio di Monte Pellegrino (Palermo). Quad. Bot. Ambient. Appl. 1996, 4, 35–48. [Google Scholar]
- Rossini Oliva, S.; Raimondo, F.M.; Valdés, B. The ornamental flora of Western Sicily. Bocconea 2003, 16, 1171–1176. [Google Scholar]
- Domina, G.; Mazzola, P. Flora ornamentale delle isole circumsiciliane. Quad. Bot. Ambient. Appl. 2008, 19, 55–67. [Google Scholar]
- Schicchi, R.; Speciale, M. Alberi di Palermo, Guida al Riconoscimento; Palermo University Press: Palermo, Italy, 2020; p. 335. [Google Scholar]
- Traverso, O. Botanica Orticola; M. Ponzio: Pavia, Italy, 1926; 1367p. [Google Scholar]
- Bailey, L.H. The Standard Cyclopedia of Horticolture, 1–3; Mcmillian & Co.: New York, NY, USA, 1963. [Google Scholar]
- Cullen, J.; Knees, S.G.; Cubey, H.S. The European Garden Flora, 2nd ed., 1–5; Cambridge University Press: Cambridge, UK, 2011. [Google Scholar]
- Pignatti, S. Flora d’Italia, 2nd ed., 1; New Business Media: Milano, Italy, 2017. [Google Scholar]
- Pignatti, S. Flora d’Italia, 2nd ed., 2; New Business Media: Milano, Italy, 2017. [Google Scholar]
- Pignatti, S. Flora d’Italia, 2nd ed., 3; New Business Media: Milano, Italy, 2018. [Google Scholar]
- Pignatti, S.; Guarino, R.; La Rosa, M. Flora d’Italia, 2nd ed., 4 & Flora Digitale; New Business Media: Milano, Italy, 2019. [Google Scholar]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Domina, G.; Di Gristina, E.; Barone, G. Main Poisonous and Allergenic Plant Species in Sicilian Gardens and Parks: Applications and Recommendations for Use. Plants 2024, 13, 1031. https://doi.org/10.3390/plants13071031
Domina G, Di Gristina E, Barone G. Main Poisonous and Allergenic Plant Species in Sicilian Gardens and Parks: Applications and Recommendations for Use. Plants. 2024; 13(7):1031. https://doi.org/10.3390/plants13071031
Chicago/Turabian StyleDomina, Gianniantonio, Emilio Di Gristina, and Giulio Barone. 2024. "Main Poisonous and Allergenic Plant Species in Sicilian Gardens and Parks: Applications and Recommendations for Use" Plants 13, no. 7: 1031. https://doi.org/10.3390/plants13071031
APA StyleDomina, G., Di Gristina, E., & Barone, G. (2024). Main Poisonous and Allergenic Plant Species in Sicilian Gardens and Parks: Applications and Recommendations for Use. Plants, 13(7), 1031. https://doi.org/10.3390/plants13071031






