Effects of Aviation Mutagenesis on Soil Chemical Indexes, Enzyme Activities, and Metabolites of Dahongpao (Camellia sinensis) Tea Trees
Abstract
1. Introduction
2. Results
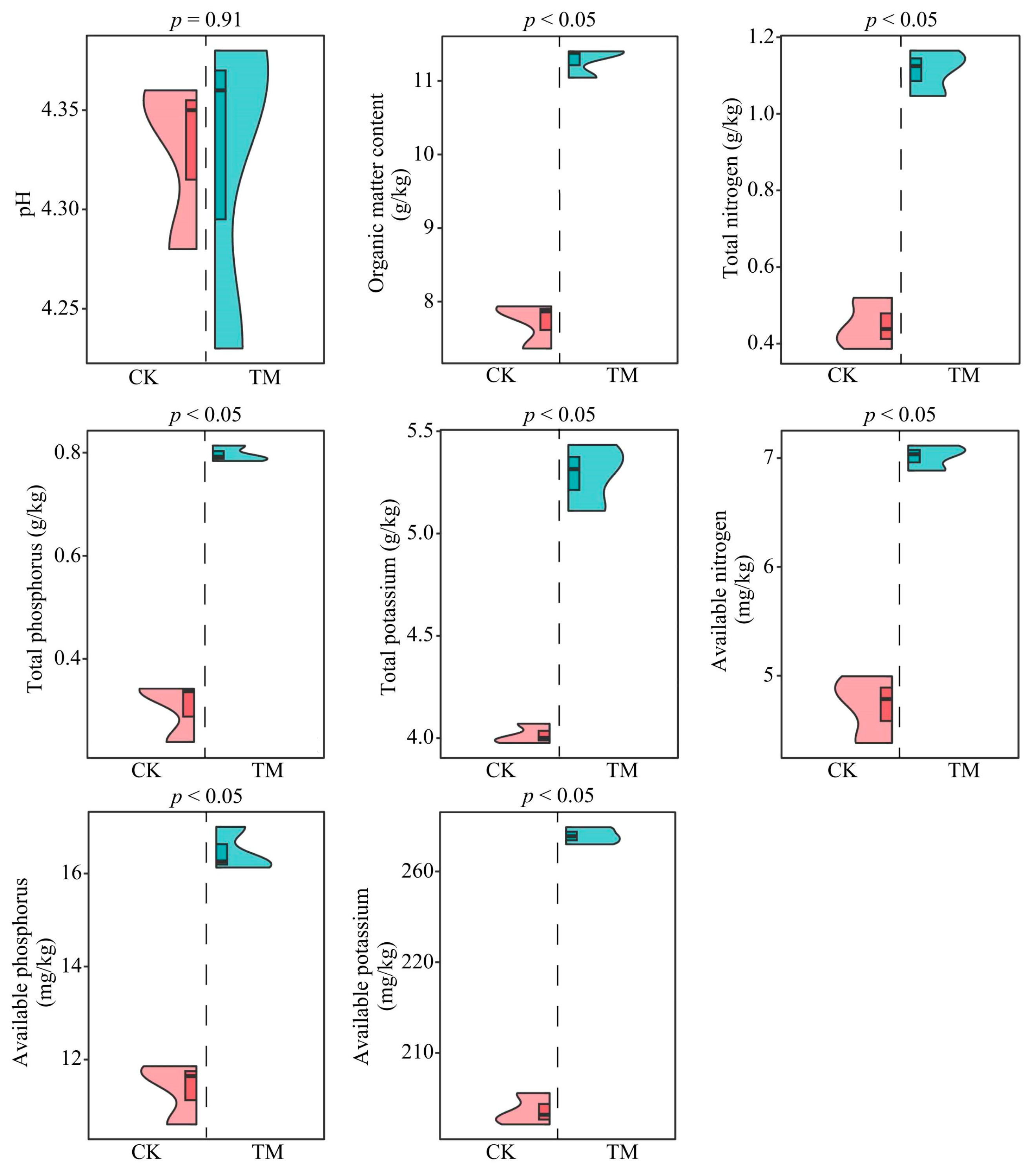
2.1. Analysis of Chemical Indexes of Tea Tree Rhizosphere Soil
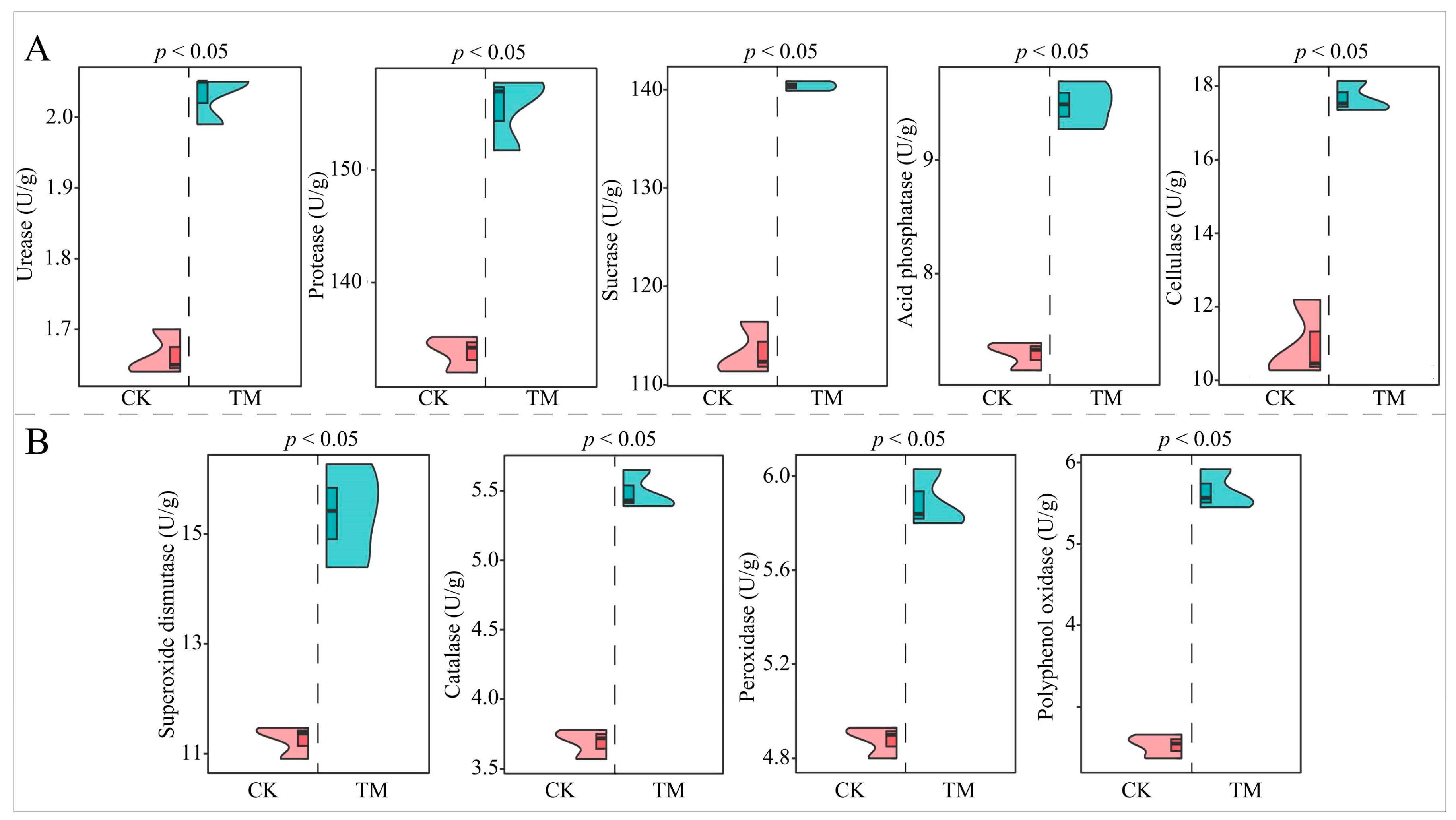
2.2. Analysis of Rhizosphere Soil Enzyme Activities of Tea Tree
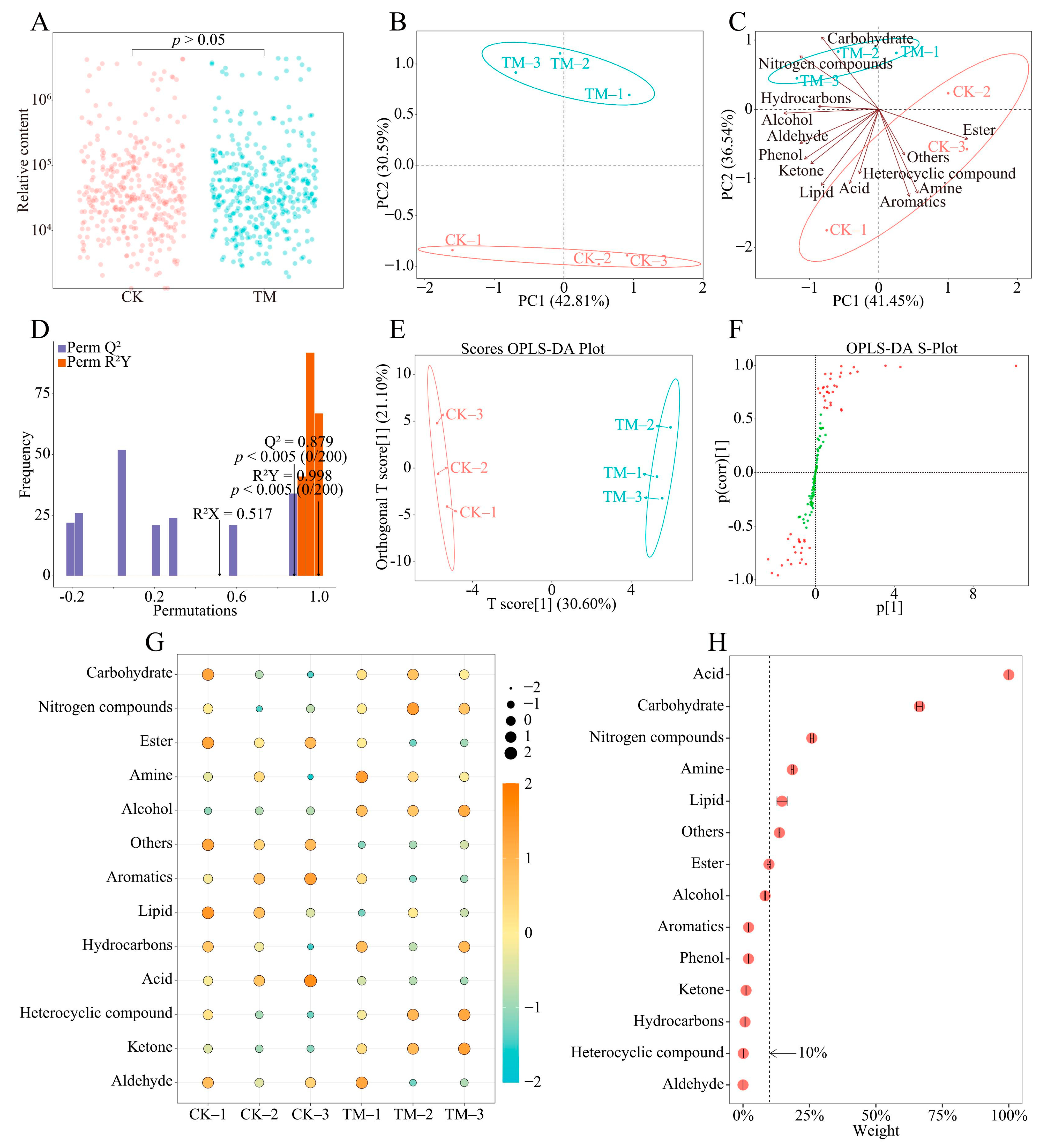
2.3. Analysis of Rhizosphere Soil Metabolites of Tea Tree
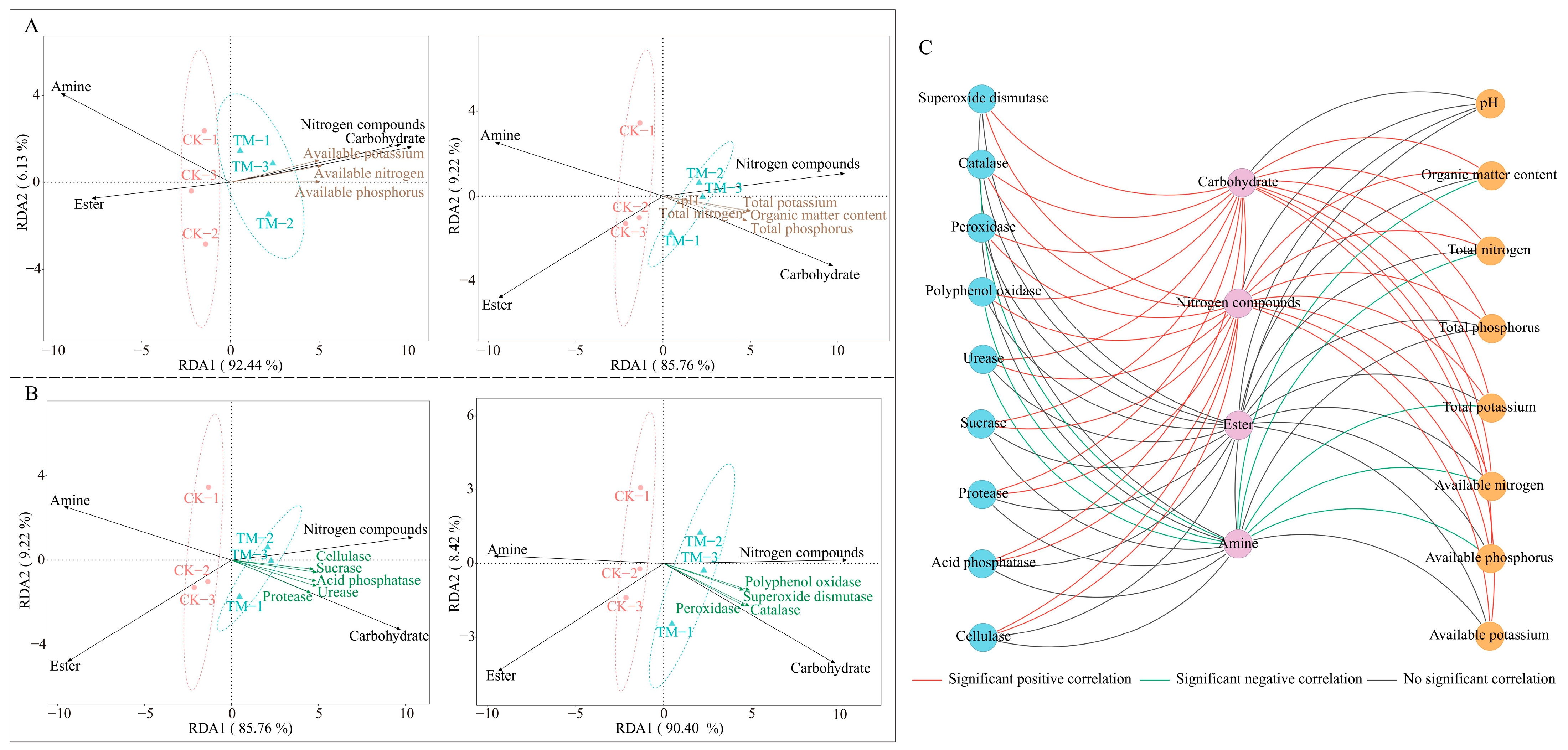
2.4. Interaction Analysis
3. Discussion
4. Materials and Methods
4.1. Samples and Sampling
4.2. Determination of Soil Chemical Indexes
4.3. Determination of Soil Enzyme Activity
4.4. Extraction and Determination of Soil Metabolites
4.5. Statistical Analysis
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Ferl, R.; Wheeler, R.; Levine, H.; Paul, A. Plants in space. Curr. Opin. Plant Biol. 2002, 5, 258–263. [Google Scholar] [CrossRef] [PubMed]
- Furukawa, S.; Nagamatsu, A.; Nenoi, M.; Fujimori, A.; Kakinuma, S.; Katsube, T.; Wang, B.; Tsuruoka, C.; Shirai, T.; Nakamura, A.; et al. Space radiation biology for “Living in Space”. BioMed Res. Int. 2020, 2020, 25. [Google Scholar] [CrossRef] [PubMed]
- Xia, P.; Li, Q.; Liang, Z.; Zhang, X.; Yan, K. Spaceflight breeding could improve the volatile constituents of Andrographis paniculata. Ind. Crops Prod. 2021, 171, 113967. [Google Scholar] [CrossRef]
- Mohanta, T.K.; Mishra, A.K.; Mohanta, Y.K.; Al-Harrasi, A. Space breeding: The next-generation crops. Front. Plant Sci. 2021, 12, 2407. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.; Chen, Y.; Wang, W.; Wang, H.; Cai, J.; Wang, M.; Li, D.; Zhang, B.; Huang, K. Effects of space treatment on biological and growth characteristics of Camellia sinensis. Chin. Bull. Bot. 2020, 55, 564–572. [Google Scholar] [CrossRef]
- Pii, Y.; Mimmo, T.; Tomasi, N.; Terzano, R.; Cesco, S.; Crecchio, C. Microbial interactions in the rhizosphere: Beneficial influences of plant growth-promoting rhizobacteria on nutrient acquisition process. A review. Biol. Fert. Soils 2015, 51, 403–415. [Google Scholar] [CrossRef]
- Bowles, T.M.; Acosta-Martínez, V.; Calderón, F.; Jackson, L.E. Soil enzyme activities, microbial communities, and carbon and nitrogen availability in organic agroecosystems across an intensively-managed agricultural landscape. Soil Biol. Biochem. 2014, 68, 252–262. [Google Scholar] [CrossRef]
- Lin, S.; Liu, Z.; Wang, Y.; Li, J.; Wang, G.; Ye, J.; Wang, H.; He, H. Soil metagenomic analysis on changes of functional genes and microorganisms involved in nitrogen-cycle processes of acidified tea soils. Front. Plant Sci. 2022, 13, 998178. [Google Scholar] [CrossRef] [PubMed]
- Ye, J.; Wang, Y.; Lin, S.; Wang, Y.; Chen, P.; Hong, L.; Jia, X.; Kang, J.; Wu, Z.; Wang, H. Metabolomics analysis of the effect of acidification on rhizosphere soil microecosystem of tea tree. Front. Plant Sci. 2023, 14, 1137465. [Google Scholar] [CrossRef]
- Shen, F.T.; Lin, S.H. Priming effects of cover cropping on bacterial community in a tea plantation. Sustainability 2021, 13, 4345. [Google Scholar] [CrossRef]
- Tan, X.; Xie, H.; Yu, J.; Wang, Y.; Xu, J.; Xu, P.; Ma, B. Host genetic determinants drive compartment-specific assembly of tea plant microbiomes. Plant Biotechnol. J. 2022, 20, 2174–2186. [Google Scholar] [CrossRef] [PubMed]
- Sun, Y.; Duan, C.; Cao, N.; Ding, C.; Huang, Y.; Wang, J. Biodegradable and conventional microplastics exhibit distinct microbiome, functionality, and metabolome changes in soil. J. Hazard. Mater. 2022, 424, 127282. [Google Scholar] [CrossRef]
- Wu, C.; Ma, Y.; Wang, D.; Shan, Y.; Song, X.; Hu, H.; Ren, X.; Ma, X.; Cui, J.; Ma, Y. Integrated microbiology and metabolomics analysis reveal plastic mulch film residue affects soil microorganisms and their metabolic functions. J. Hazard. Mater. 2022, 423, 127258. [Google Scholar] [CrossRef] [PubMed]
- Liang, L.L.; Song, Y.K.; Qian, W.J.; Ruan, J.Y.; Ding, Z.T.; Zhang, Q.F.; Hu, J.H. Metabolomics analysis reveals the responses of tea plants to excessive calcium. J. Sci. Food Agric. 2021, 101, 5678–5687. [Google Scholar] [CrossRef] [PubMed]
- Brown, R.W.; Chadwick, D.R.; Zang, H.; Jones, D.L. Use of metabolomics to quantify changes in soil microbial function in response to fertiliser nitrogen supply and extreme drought. Soil Biol. Biochem. 2021, 160, 108351. [Google Scholar] [CrossRef]
- Soares, J.C.; Santos, C.S.; Carvalho, S.M.; Pintado, M.M.; Vasconcelos, M.W. Preserving the nutritional quality of crop plants under a changing climate: Importance and strategies. Plant Soil 2019, 443, 1–26. [Google Scholar] [CrossRef]
- Briat, J.F.; Gojon, A.; Plassard, C.; Rouached, H.; Lemaire, G. Reappraisal of the central role of soil nutrient availability in nutrient management in light of recent advances in plant nutrition at crop and molecular levels. Eur. J. Agron. 2020, 116, 126069. [Google Scholar] [CrossRef]
- Das, P.P.; Singh, K.R.; Nagpure, G.; Mansoori, A.; Singh, R.P.; Ghazi, I.A.; Kumar, A.; Singh, J. Plant-soil-microbes: A tripartite interaction for nutrient acquisition and better plant growth for sustainable agricultural practices. Environ. Res. 2022, 214, 113821. [Google Scholar] [CrossRef]
- Zielewicz, W.; Swędrzyński, A.; Dobrzyński, J.; Swędrzyńska, D.; Kulkova, I.; Wierzchowski, P.S.; Wróbel, B. Effect of forage plant mixture and biostimulants application on the yield, changes of botanical composition, and microbiological soil activity. Agronomy 2021, 11, 1786. [Google Scholar] [CrossRef]
- Mao, L.; He, X.; Ye, S.; Wang, S. Soil aggregate-associated carbon-cycle and nitrogen-cycle enzyme activities as affected by stand age in Chinese fir plantations. J. Soil Sci. Plant Nut. 2023, 23, 4361–4372. [Google Scholar] [CrossRef]
- Tian, J.; Ge, F.; Zhang, D.; Deng, S.; Liu, X. Roles of phosphate solubilizing microorganisms from managing soil phosphorus deficiency to mediating biogeochemical P cycle. Biology 2021, 10, 158. [Google Scholar] [CrossRef] [PubMed]
- Zheng, W.; Dong, H.; Wang, Z.; Tao, Y. Effect of straw returning and nitrogen application rate on soil enzymatic activities. Agric. Res. 2023, 12, 163–171. [Google Scholar] [CrossRef]
- Withers, E.; Hill, P.W.; Chadwick, D.R.; Jones, D.L. Use of untargeted metabolomics for assessing soil quality and microbial function. Soil Biol. Biochem. 2020, 143, 107758. [Google Scholar] [CrossRef]
- Wang, H.; Liu, S.; Schindlbacher, A.; Wang, J.; Yang, Y.; Song, Z.; You, Y.; Shi, Z.; Li, Z.; Chen, L.; et al. Experimental warming reduced topsoil carbon content and increased soil bacterial diversity in a subtropical planted forest. Soil Biol. Biochem. 2019, 133, 155–164. [Google Scholar] [CrossRef]
- Xie, X.G.; Zhang, F.M.; Wang, X.X.; Li, X.G.; Dai, C.C. Phomopsis liquidambari colonization promotes continuous cropping peanut growth by improving the rhizosphere microenvironment, nutrient uptake and disease incidence. J. Sci. Food Agric. 2019, 99, 1898–1907. [Google Scholar] [CrossRef] [PubMed]
- Hartmann, M.; Six, J. Soil structure and microbiome functions in agroecosystems. Nat. Rev. Earth Environ. 2023, 4, 4–18. [Google Scholar] [CrossRef]
- Chen, Y.; Yang, L.; Zhang, L.; Li, J.; Zheng, Y.; Yang, W.; Deng, L.; Gao, Q.; Mi, Q.; Li, X.; et al. Autotoxins in continuous tobacco cropping soils and their management. Front. Plant Sci. 2023, 14, 1106033. [Google Scholar] [CrossRef]
- Wang, Y.; Zhang, Q.; Li, J.; Lin, S.; Jia, X.; Zhang, Q.; Ye, J.; Wang, H.; Wu, Z. Study on the effect of pH on rhizosphere soil fertility and the aroma quality of tea trees and their interactions. Agriculture 2023, 13, 1739. [Google Scholar] [CrossRef]
- Yuan, H.; Cao, G.; Hou, X.; Huang, M.; Du, P.; Tan, T.; Zhang, Y.; Zhou, H.; Liu, X.; Liu, L.; et al. Development of a widely targeted volatilomics method for profiling volatilomes in plants. Mol. Plant 2022, 15, 189–202. [Google Scholar] [CrossRef]
- Li, Z.; Sun, X.; Xu, T.; Dai, W.; Yan, Q.; Li, P.; Fang, Y.; Ding, J. Insight into the dynamic variation and retention of major aroma volatile compounds during the milling of Suxiang japonica rice. Food Chem. 2023, 405, 134468. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Jia, M.; Cheng, P.; Wang, Y.; Pang, X.; Li, M.; Hong, L.; Zhang, Q.; Chen, Y.; Jia, X.; Ye, J.; et al. Effects of Aviation Mutagenesis on Soil Chemical Indexes, Enzyme Activities, and Metabolites of Dahongpao (Camellia sinensis) Tea Trees. Plants 2024, 13, 1291. https://doi.org/10.3390/plants13101291
Jia M, Cheng P, Wang Y, Pang X, Li M, Hong L, Zhang Q, Chen Y, Jia X, Ye J, et al. Effects of Aviation Mutagenesis on Soil Chemical Indexes, Enzyme Activities, and Metabolites of Dahongpao (Camellia sinensis) Tea Trees. Plants. 2024; 13(10):1291. https://doi.org/10.3390/plants13101291
Chicago/Turabian StyleJia, Miao, Pengyuan Cheng, Yuhua Wang, Xiaomin Pang, Mingzhe Li, Lei Hong, Qi Zhang, Yiling Chen, Xiaoli Jia, Jianghua Ye, and et al. 2024. "Effects of Aviation Mutagenesis on Soil Chemical Indexes, Enzyme Activities, and Metabolites of Dahongpao (Camellia sinensis) Tea Trees" Plants 13, no. 10: 1291. https://doi.org/10.3390/plants13101291
APA StyleJia, M., Cheng, P., Wang, Y., Pang, X., Li, M., Hong, L., Zhang, Q., Chen, Y., Jia, X., Ye, J., & Wang, H. (2024). Effects of Aviation Mutagenesis on Soil Chemical Indexes, Enzyme Activities, and Metabolites of Dahongpao (Camellia sinensis) Tea Trees. Plants, 13(10), 1291. https://doi.org/10.3390/plants13101291





