Exploring the Phytochemical Composition and Biological Potential of Balkan Endemic Species Stachys scardica Griseb
Abstract
:1. Introduction
2. Results and Discussion
2.1. In Vitro Cultivation and Ex Vitro Acclimation of S. scardica
2.2. Taxonomic Position of S. scardica
2.3. The Genetic Stability of In Vitro Cultivated and Ex Vitro Adapted S. scardica Plants
2.4. NMR Fingerprinting during S. scardica Ex Situ Conservation
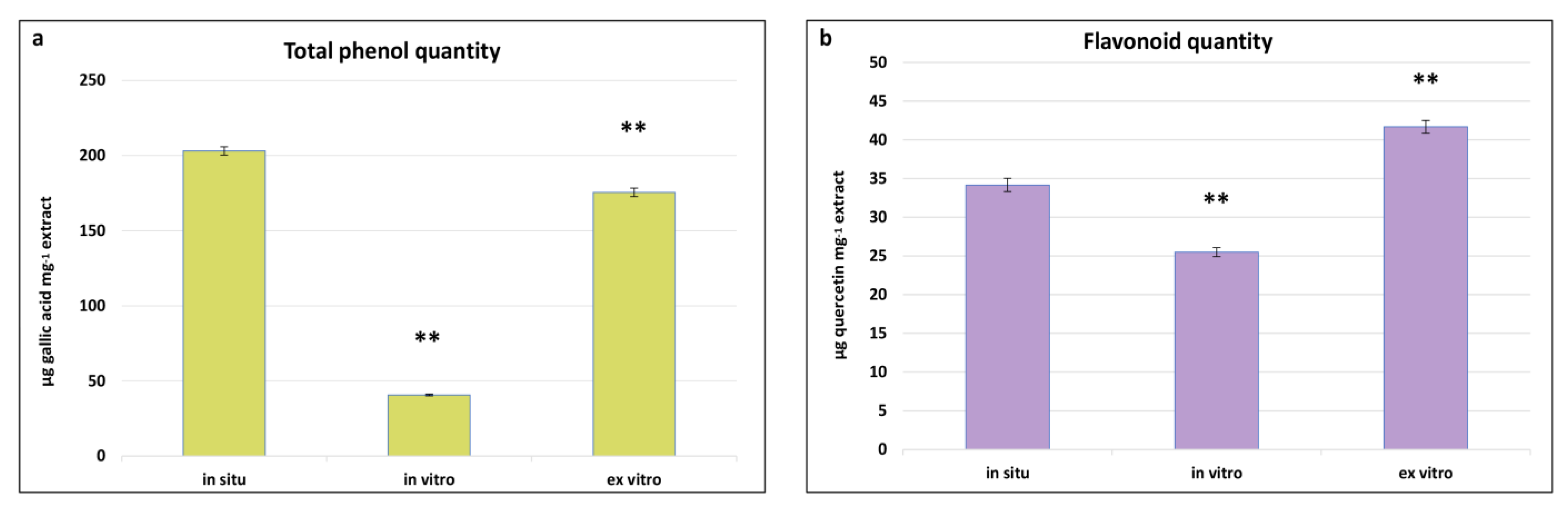
2.5. Comparative Determination of Total Phenols and Flavonoids in In Situ, In Vitro Cultivated, and Ex Vitro Adapted Plants
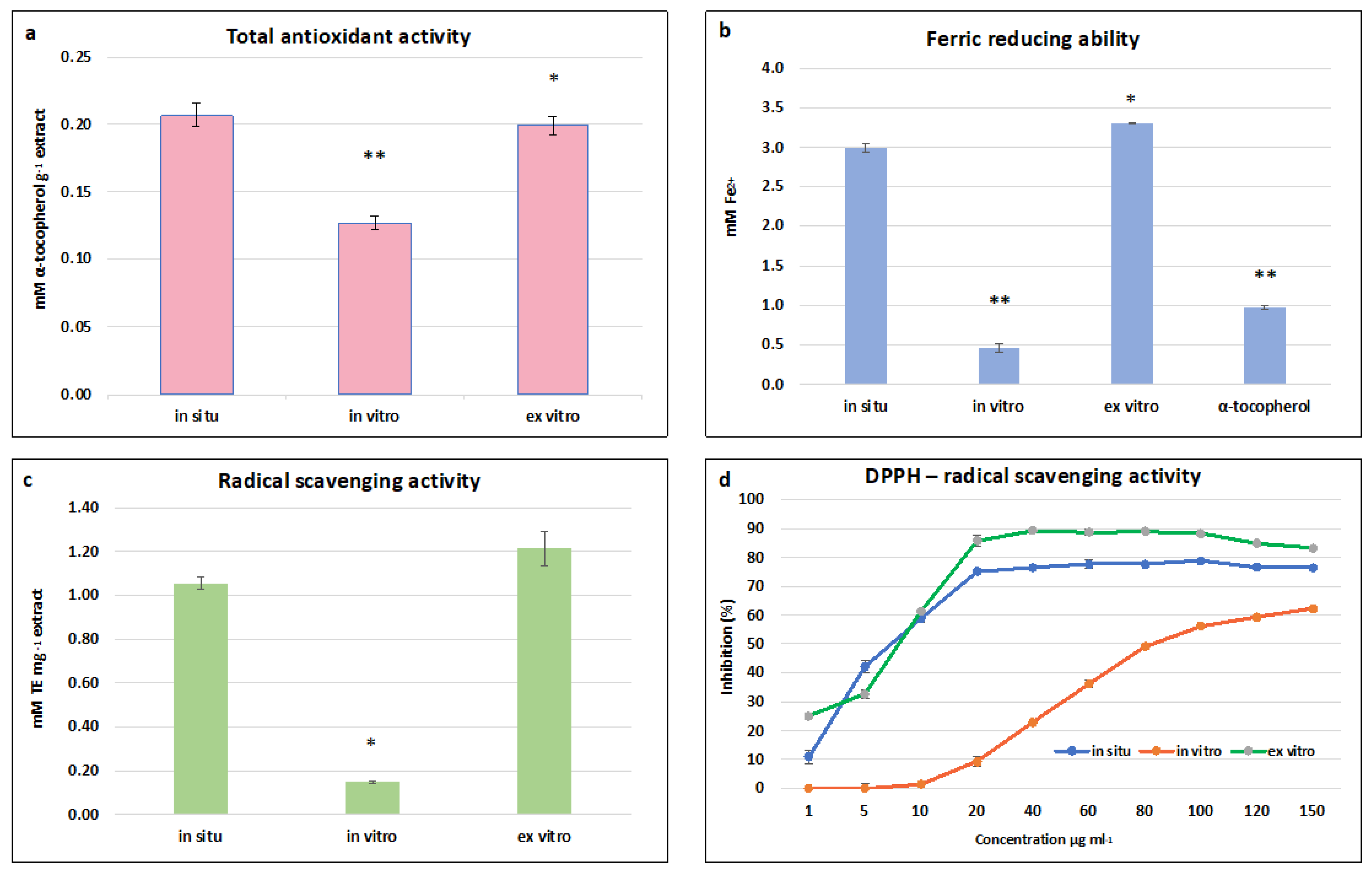
2.6. The Antioxidant and Radical Scavenging Activities of In Situ, In Vitro Cultivated, and Ex Vitro Adapted S. scardica Plants
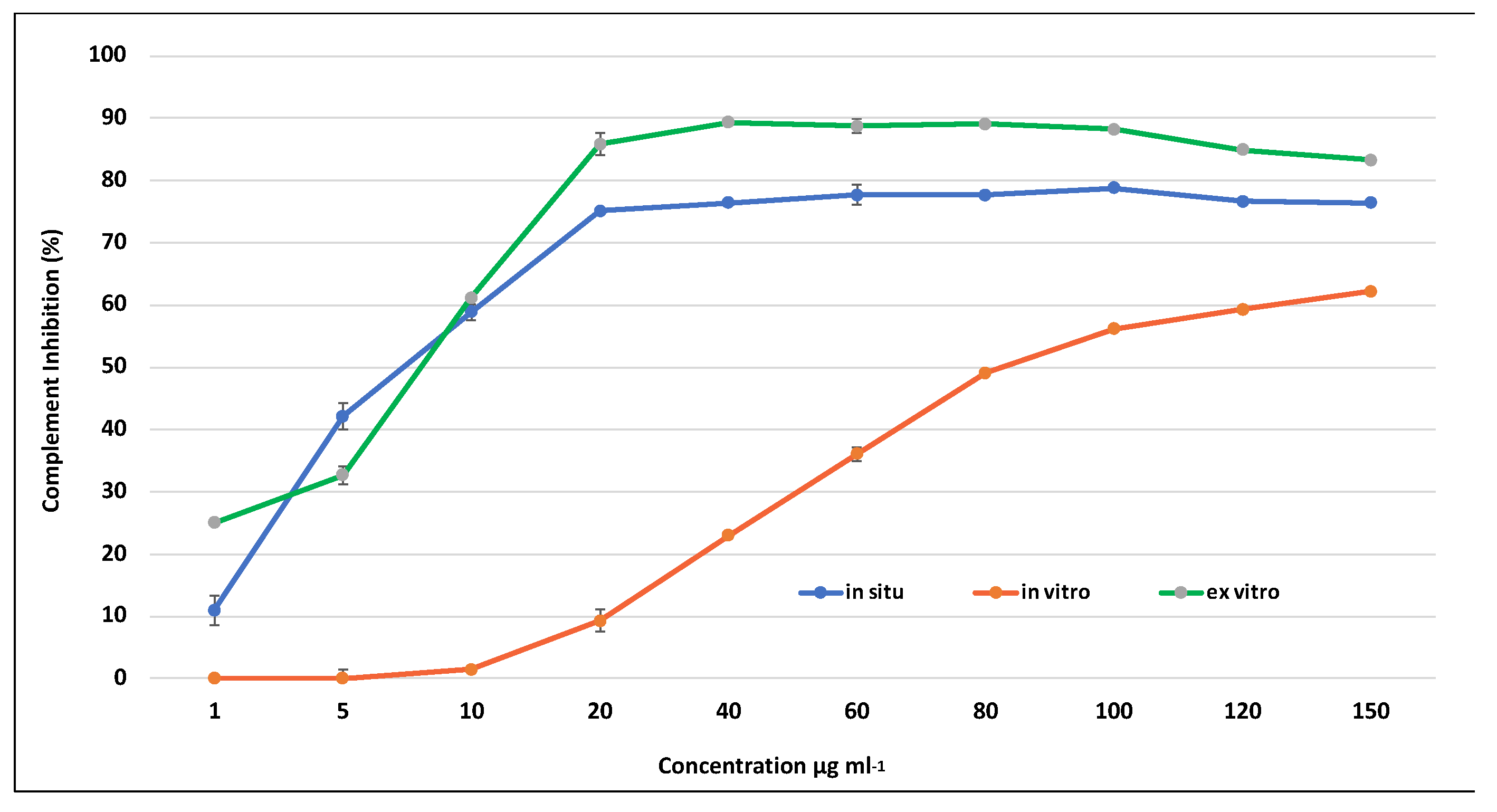
2.7. The Anti-Inflammatory Activity of S. scardica
2.8. The Anti-Microbial Activity of S. scardica
3. Materials and Methods
3.1. Chemicals
3.2. Plant Material—In Vitro Cultivation and Ex Vitro Adaptation
3.3. DNA Barcoding Analysis
3.4. Genetic Stability Assay by SRAP Markers
3.5. NMR Analyses
3.6. The Preparation of Methanolic Extracts
3.7. The Determination of Total Phenolic and Flavonoid Contents
3.8. Antioxidant and Radical Scavenging Activity Assays
3.9. Microtitre Hemolytic Complement Assay
3.10. Antimicrobial Activity
3.11. Data Analysis
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Sitarek, P.; Merecz-Sadowska, A.; Kowalczyk, T.; Wieczfinska, J.; Zajdel, R.; Śliwiński, T. Potential synergistic action of bioactive compounds from plant extracts against skin infecting microorganisms. Int. J. Mol. Sci. 2020, 21, 5105. [Google Scholar] [CrossRef] [PubMed]
- Sawada, Y.; Saito-Sasaki, N.; Mashima, E.; Nakamura, M. Daily lifestyle and inflammatory skin diseases. Int. J. Mol. Sci. 2021, 22, 5204. [Google Scholar] [CrossRef] [PubMed]
- Tampa, M.; Neagu, M.; Caruntu, C.; Georgescu, S.R. Personalized Medicine in the Field of Inflammatory Skin Disorders. J. Pers. Med. 2022, 12, 426. [Google Scholar] [CrossRef] [PubMed]
- Guo, J.W.; Jee, S.H. Strategies to develop a suitable formulation for inflammatory skin disease treatment. Int. J. Mol. Sci. 2021, 22, 6078. [Google Scholar] [CrossRef]
- Máthé, A.; Hassan, F.; Abdul Kader, A. In vitro micropropagation of medicinal and aromatic plants. In Medicinal and Aromatic Plants of the World; Springer: Berlin/Heidelberg, Germany, 2015; pp. 305–336. [Google Scholar]
- Kirkan, B. Antioxidant potential, enzyme inhibition activity, and phenolic profile of extracts from Stachys cretica subsp. vacillans. Ind. Crops Prod. 2019, 140, 11639. [Google Scholar] [CrossRef]
- Aybey, A. Antibacterial and antibiofilm properties of Phlomis and Stachys species. Bangladesh J. Bot. 2020, 49, 257–263. [Google Scholar] [CrossRef]
- Tundis, R.; Peruzzi, L.; Menichini, F. Phytochemical and biological studies of Stachys species in relation to chemotaxonomy: A review. Phytochemistry 2014, 102, 7–39. [Google Scholar] [CrossRef]
- Venditti, A.; Frezza, C.; Lorenzetti, L.M.; Maggi, F.; Serafini, M.; Bianco, A. Reassessment of the polar fraction of Stachys alopecuros (L.) Benth. subsp. divulsa (Ten.) Grande (Lamiaceae) from the Monti Sibillini National Park: A potential source of bioactive compounds. J. Intercult. Ethnopharmacol. 2017, 6, 144. [Google Scholar] [CrossRef]
- Skaltsa, H.D.; Demetzosa, C.; Lazarib, D.; Sokovicc, M. Essential oil analysis and antimicrobial activity of eight Stachys species from Greece. Phytochemistry 2003, 64, 743–752. [Google Scholar] [CrossRef]
- Mantovska, D.I.; Zhiponova, M.K.; Georgiev, M.I.; Alipieva, K.; Tsacheva, I.; Simova, S.; Yordanova, Z.P. Biological activity and NMR-fingerprinting of balkan endemic species Stachys thracica Davidov. Metabolites 2022, 12, 251. [Google Scholar] [CrossRef]
- Khanavi, M.; Hajimahmoodi, M.; Cheraghi-Niroomand, M.; Kargar, Z.; Ajani, Y.; Hadjiakhoondi, A.; Oveisi, M.R. Comparison of the antioxidant activity and total phenolic contents in some Stachys species. AJB 2009, 8, 1143–1147. [Google Scholar]
- Šliumpaitė, I.; Venskutonis, P.R.; Murkovic, M.; Ragažinskienė, O. Antioxidant properties and phenolic composition of wood betony (Betonica officinalis L., syn. Stachys officinalis L.). Ind. Crops. Prod. 2013, 50, 715–722. [Google Scholar] [CrossRef]
- Shakeri, A.; D’Urso, G.; Taghizadeh, S.F.; Piacente, S.; Norouzi, S.; Soheili, V.; Asili, J.; Salarbashi, D. LC-ESI/LTQOrbitrap/MS/MS and GC–MS profiling of Stachys parviflora L. and evaluation of its biological activities. J. Pharm. Biomed. Anal. 2019, 168, 209–216. [Google Scholar] [CrossRef] [PubMed]
- Paun, G.; Neagu, E.; Moroeanu, V.; Albu, C.; Ursu, T.M.; Zanfirescu, A.; Negres, S.; Chirita, C.; Radu, G.L. Anti-inflammatory and antioxidant activities of the Impatiens noli-tangere and Stachys officinalis polyphenolic-rich extracts. Rev. Bras. Farmacogn. 2018, 28, 57–64. [Google Scholar] [CrossRef]
- Háznagy-Radnai, E.; Balogh, A.; Czigle, S.; Máthé, I.; Hohmann, J.; Blazsó, G. Antiinflammatory Activities of Hungarian Stachys species and Their Iridoids. Phytother. Res. 2011, 26, 505–509. [Google Scholar] [CrossRef] [PubMed]
- Golemanski, V.; Peev, D.R. (Eds.) Red Data Book of the Republic of Bulgaria; Bulgarian Academy of Sciences: Sofia, Bulgaria, 2015. [Google Scholar]
- Bankova, V.; Koeva-Todorovska, J.; Stambolijskab, T.; Ignatova-Groceva, M.; Todorova, D.; Popov, S. Polyphenols in Stachys and Betonica Species (Lamiaceae). Zeitschrift für Naturforschung C 1999, 54, 876–880. [Google Scholar] [CrossRef]
- Murashige, T.; Skoog, F. A revised medium for rapid growth and bio assays with tobacco tissue cultures. Physiol. Plant. 1962, 15, 473–497. [Google Scholar] [CrossRef]
- Güleryüz, G.; Kırmızı, S.; Arslan, H.; Sakar, F.S. Dormancy and germination in Stachys germanica L. subsp. bithynica (Boiss.) Bhattacharjee seeds: Effects of short-time moist chilling and plant growth regulators. Flora Morphol. Distrib. Funct. Ecol. 2011, 206, 943–948. [Google Scholar] [CrossRef]
- Kozhuharova, E. New Ex Situ Collection of Rare and Threatened Medicinal Plants in the Pirin Mts. (Bulgaria). Ekoloji 2009, 18, 32–44. [Google Scholar] [CrossRef]
- Cüce, M.; Bekircan, T.; Laghari, A.H.; Sökmen, M.; Sökmen, A.; Uçar, E.Ö.; Kılıç, A.O. Antioxidant phenolic constituents, antimicrobial and cytotoxic properties of Stachys annua L. from both natural resources and micropropagated plantlets. IJTK 2017, 16, 407–416. [Google Scholar]
- Yordanova, Z.P.; Rogova, M.A.; Kapchina-Toteva, V.M. In vitro propagation of the Balkan endemic species Verbascum eriophorum Godr. Bulg. J. Agric. Sci. 2016, 22, 767–771. [Google Scholar]
- Yordanova, Z.P.; Rogova, M.A.; Zhiponova, M.K.; Georgiev, M.I.; Kapchina-Toteva, V.M. Comparative determination of the essential oil composition in Bulgarian endemic plant Achillea thracica Velen. during the process of ex situ conservation. Phytochem. Lett. 2017, 20, 456–461. [Google Scholar] [CrossRef]
- Mantovska, D.I.; Zhiponova, M.K.; Georgiev, M.I.; Grozdanova, T.; Gerginova, D.; Alipieva, K.; Simova, S.; Popova, M.; Kapchina-Toteva, V.M.; Yordanova, Z.P. In vitro multiplication and NMR fingerprinting of rare Veronica caucasica M. Bieb. Molecules 2021, 26, 5888. [Google Scholar] [CrossRef] [PubMed]
- Panayotova, L.G.; Ivanova, T.A.; Bogdanova, Y.Y.; Gussev, C.V.; Stanilova, M.I.; Bosseva, Y.Z.; Stoeva, T.D. In vitro cultivation of plant species from sandy dunes along the Bulgarian Black Sea Coast. Phytol. Balc. 2008, 14, 119–123. [Google Scholar]
- Salmaki, Y.; Zarre, S.; Ryding, O.; Lindqvist, C.; Bräuchler, C.; Heubl, G.; Barber, J.; Bendiksby, M. Molecular phylogeny of tribe Stachydeae (Lamiaceae subfamily Lamioideae). Mol. Phylogenet. Evol. 2013, 69, 535–551. [Google Scholar] [CrossRef] [PubMed]
- Bhattacharjee, R. Taxonomic studies in Stachys: II. A new infrageneric classification of Stachys L. Notes R. Bot. Gard. Edinburgh 1980, 38, 65–96. [Google Scholar]
- Kang, Y.; Deng, Z.; Zang, R.; Long, W. DNA barcoding analysis and phylogenetic relationships of tree species in tropical cloud forests. Sci. Rep. 2017, 7, 12564. [Google Scholar] [CrossRef] [PubMed]
- Aneva, I.; Zhelev, P.; Bonchev, G.; Boycheva, I.; Simeonova, S.; Kancheva, D. DNA barcoding study of representative Thymus species in Bulgaria. Plants 2022, 11, 270. [Google Scholar] [CrossRef]
- Petrova, D.; Gašić, U.; Yocheva, L.; Hinkov, A.; Yordanova, Z.; Chaneva, G.; Mantovska, D.; Paunov, M.; Ivanova, L.; Rogova, M.; et al. Catmint (Nepeta nuda L.) phylogenetics and metabolic responses in variable growth conditions. Front. Plant Sci. 2022, 13, 866777. [Google Scholar] [CrossRef]
- Tobyn, G.; Denham, A.; Whitelegg, M. The Western Herbal Tradition: 2000 Years of Medicinal Plant Knowledge; Singing Dragon: London, UK, 2016. [Google Scholar]
- Venditti, A.; Serrilli, A.M.; Di Cecco, M.; Ciaschetti, G.; Andrisano, T.; Bianco, A. Phytochemical composition of polar fraction of Stachys germanica L. subsp. salviifolia (Ten.) Gams, a typical plant of Majella National Park. Nat. Prod. Res. 2013, 27, 190–193. [Google Scholar] [CrossRef]
- Duta-Cornescu, G.; Constantin, N.; Pojoga, D.M.; Nicuta, D.; Simon-Gruita, A. Somaclonal variation—Advantage or disadvantage in micropropagation of the medicinal plants. Int. J. Mol. Sci. 2023, 24, 838. [Google Scholar] [CrossRef] [PubMed]
- Li, G.; Quiros, C.F. Sequence-related amplified polymorphism (SRAP), a new marker system based on a simple PCR reaction: Its application to mapping and gene tagging in Brassica. Theor. Appl. Genet. 2001, 103, 455–461. [Google Scholar] [CrossRef]
- Zagorcheva, T.; Stanev, S.; Rusanov, K.; Atanassov, I. SRAP markers for genetic diversity assessment of lavender (Lavandula angustifolia mill.) varieties and breeding lines. Biotechnol. Biotechnol. Equip. 2020, 34, 303–308. [Google Scholar] [CrossRef]
- Sarfaraz, D.; Rahimmalek, M.; Saeidi, G. Polyphenolic and molecular variation in Thymus species using HPLC and SRAP analyses. Sci. Rep. 2021, 11, 5019. [Google Scholar] [CrossRef] [PubMed]
- Alekseeva, M.; Zagorcheva, T.; Rusanova, M.; Rusanov, K.; Atanassov, I. Genetic and Flower Volatile Diversity in Natural Populations of Origanum vulgare subsp. hirtum (Link) Ietsw. in Bulgaria: Toward the Development of a Core Collection. Front. Plant Sci. 2021, 12, 679063. [Google Scholar] [CrossRef] [PubMed]
- Asande, L.K.; Ombori, O.; Nyaboga, E.N.; Oduor, R.O. Efficient shoot organogenesis using leaf disc and nodal explants of passion fruit (Passiflora edulis Sims) and genetic fidelity assessment using sequence-related amplified polymorphism (SRAP) markers. Int. J. Agron. 2020, 2020, 3205710. [Google Scholar] [CrossRef]
- Forde, B.G.; Lea, P.J. Glutamate in plants: Metabolism, regulation, and signalling. J. Exp. Bot. 2007, 58, 2339–2358. [Google Scholar] [CrossRef] [PubMed]
- Parthasarathy, A.; Savka, M.A.; Hudson, A.O. The synthesis and role of β-alanine in plants. Front. Plant Sci. 2019, 10, 921. [Google Scholar] [CrossRef]
- Panchal, P.; Miller, A.J.; Giri, J. Organic acids: Versatile stress-response roles in plants. J. Exp. Bot. 2021, 72, 4038–4052. [Google Scholar] [CrossRef]
- Lopez-Bucio, J.; De la Vega, O.M.; Guevara-Garcia, A.; Herrera-Estrella, L. Enhanced phosphorus uptake in transgenic tobacco plants that overproduce citrate. Nat. Biotechnol 2000, 18, 450–453. [Google Scholar] [CrossRef]
- Xiang, Y.; Kang, F.; Xiang, Y.; Jiao, Y. Effects of humic acid-modified magnetic Fe3O4/MgAl-layered double hydroxide on the plant growth, soil enzyme activity, and metal availability. Ecotoxicol. Environ. Saf. 2019, 182, 109424. [Google Scholar] [CrossRef] [PubMed]
- Shahbaz, A.M.; Oki, Y.; Adachi, T.; Murata, Y.; Khan, M.H.R. Phosphorus starvation induced root-mediated pH changes in solublization and acquisition of sparingly soluble P sources and organic acids exudation by Brassica cultivars. J. Soil Sci. Plant Nutr 2006, 52, 623–633. [Google Scholar] [CrossRef]
- Mora-Macías, J.; Ojeda-Rivera, J.O.; Gutiérrez-Alanís, D.; Yong-Villalobos, L.; Oropeza-Aburto, A.; Raya-González, J.; Jiménez-Domínguez, G.; Chávez-Calvillo, G.; Rellán-Álvarez, R.; Herrera-Estrella, L. Malate-dependent Fe accumulation is a critical checkpoint in the root developmental response to low phosphate. Proc. Natl. Am. Sci. USA 2017, 114, E3563–E3572. [Google Scholar] [CrossRef] [PubMed]
- Lehner, A.; Chopera, D.R.; Peters, S.W.; Keller, F.; Mundree, S.G.; Thomson, J.A.; Farrant, J.M. Protection mechanisms in the resurrection plant Xerophyta viscosa: Cloning, expression, characterisation and role of XvINO1, a gene coding for a myo-inositol 1-phosphate synthase. Funct. Plant Biol. 2008, 35, 26–39. [Google Scholar] [CrossRef] [PubMed]
- Kim, Y.H.; Khan, A.L.; Waqas, M.; Lee, I.J. Silicon regulates antioxidant activities of crop plants under abiotic-induced oxidative stress: A review. Front. Plant Sci. 2017, 8, 510. [Google Scholar] [CrossRef] [PubMed]
- Brownstein, K.J.; Thomas, A.L.; Nguyen, H.T.; Gang, D.R.; Folk, W.R. Changes in the Harpagide, Harpagoside, and Verbascoside Content of Field Grown Scrophularia lanceolata and Scrophularia marilandica in Response to Season and Shade. Metabolites 2021, 11, 464. [Google Scholar] [CrossRef] [PubMed]
- Georgiev, M.I.; Ivanovska, N.; Alipieva, K.; Dimitrova, P.; Verpoorte, R. Harpagoside: From Kalahari Desert to pharmacy shelf. Phytochem. 2013, 92, 8–15. [Google Scholar] [CrossRef] [PubMed]
- Tomou, E.M.; Barda, C.; Skaltsa, H. Genus Stachys: A review of traditional uses, phytochemistry and bioactivity. Medicines 2020, 7, 63. [Google Scholar] [CrossRef]
- Alipieva, K.; Korkina, L.; Orhan, I.E.; Georgiev, M.I. Verbascoside—A review of its occurrence,(bio) synthesis and pharmacological significance. Biotechnol. Adv. 2014, 32, 1065–1076. [Google Scholar] [CrossRef]
- Naveed, M.; Hejazi, V.; Abbas, M.; Kamboh, A.A.; Khan, G.J.; Shumzaid, M.; Ahmad, F.; Babazadeh, D.; FangFang, X.; Modarresi-Ghazani, F.; et al. Chlorogenic acid (CGA): A pharmacological review and call for further research. Biomed. Pharmacother. 2018, 97, 67–74. [Google Scholar] [CrossRef]
- Frezza, C.; de Vita, D.; Toniolo, C.; Ventrone, A.; Tomassini, L.; Foddai, S.; Nicoletti, M.; Guiso, M.; Bianco, A.; Serafini, M. Harpagide: Occurrence in plants and biological activities-A review. Fitoterapia 2020, 147, 104764. [Google Scholar] [CrossRef] [PubMed]
- Venditti, A.; Bianco, A.; Nicoletti, M.; Quassinti, L.; Bramucci, M.; Lupidi, G.; Vitali, L.A.; Papa, F.; Vittori, S.; Petrelli, D.; et al. Characterization of secondary metabolites, biological activity and glandular trichomes of Stachys tymphaea Hausskn. from the Monti Sibillini National Park (Central Apennines, Italy). Chem. Biodivers. 2014, 11, 245–261. [Google Scholar] [CrossRef] [PubMed]
- Tungmunnithum, D.; Thongboonyou, A.; Pholboon, A.; Yangsabai, A. Flavonoids and other phenolic compounds from medicinal plants for pharmaceutical and medical aspects: An overview. Medicines 2018, 5, 93. [Google Scholar] [CrossRef] [PubMed]
- Mntovska, D.; Chaneva, G.; Yordanova, Z. Comparative determination of phenolic content and radical scavenging activity of the Bulgarian endemic species Stachys bulgarica Degen & Neic. Oxid. Commun. 2022, 45, 40–51. [Google Scholar]
- Trouba, K.J.; Hamadeh, H.K.; Amin, R.P.; Germolec, D.R. Oxidative stress and its role in skin disease. ARS 2002, 4, 665–673. [Google Scholar] [CrossRef] [PubMed]
- Kruk, J.; Duchnik, E. Oxidative stress and skin diseases: Possible role of physical activity. Asian Pac. J. Cancer Prev. 2014, 15, 561–568. [Google Scholar] [CrossRef] [PubMed]
- Barygina, V.; Becatti, M.; Lotti, T. Low dose cytokines reduce oxidative stress in primarylesional fibroblasts obtained from psoriatic patients. J. Dermatol. Sci. 2016, 83, 242–244. [Google Scholar] [CrossRef] [PubMed]
- Khan, A.Q.; Agha, M.V.; Sheikhan, K.S.A.; Younis, S.M.; Al Tamimi, M.; Alam, M.; Ahmad, A.; Uddin, S.; Buddenkotte, J.; Steinhoff, M. Targeting deregulated oxidative stress in skin inflammatory diseases: An update on clinical importance. Biomed. Pharmacother. 2022, 154, 113601. [Google Scholar] [CrossRef]
- Jose, A.; Labala, S.; Venuganti, V.V.K. Co-delivery of curcumin and STAT3 siRNA using deformable cationic liposomes to treat skin cancer. J. Drug Target. 2017, 25, 330–341. [Google Scholar] [CrossRef]
- Vundać, V.B.; Brantner, A.H.; Plazibat, M. Content of polyphenolic constituents and antioxidant activity of some Stachys taxa. Food Chem. 2007, 104, 1277–1281. [Google Scholar] [CrossRef]
- Lu, H.; Tian, Z.; Cui, Y.; Liu, Z.; Ma, X. Chlorogenic acid: A comprehensive review of the dietary sources, processing effects, bioavailability, beneficial properties, mechanisms of action, and future directions. CRFSFS 2020, 19, 3130–3158. [Google Scholar] [CrossRef] [PubMed]
- Luhata, L.P.; Usuki, T. Free radical scavenging activities of verbascoside and isoverbascoside from the leaves of Odontonema strictum (Acanthaceae). Bioorganic Med. Chem. Lett. 2022, 59, 128528. [Google Scholar]
- Dunkelberger, J.R.; Song, W.C. Complement and its role in innate and adaptive immune responses. Cell Res. 2010, 20, 34–50. [Google Scholar] [CrossRef] [PubMed]
- Giang, J.; Seelen, M.A.; Van Doorn, M.B.; Rissmann, R.; Prens, E.P.; Damman, J. Complement activation in inflammatory skin diseases. Front. Immunol. 2018, 9, 639. [Google Scholar] [CrossRef] [PubMed]
- Ghias, M.H.; Hyde, M.J.; Tomalin, L.E.; Morgan, B.P.; Alavi, A.; Lowes, M.A.; Piguet, V. Role of the complement pathway in inflammatory skin diseases: A focus on hidradenitis suppurativa. JID 2020, 140, 531–536. [Google Scholar] [CrossRef] [PubMed]
- Pieroni, A.; Pachaly, P.; Huang, Y.; Van Poel, B.; Vlietinck, A.J. Studies on anti-complementary activity of extracts and isolated flavones from Ligustrum vulgare and Phillyrea latifolia leaves (Oleaceae). J. Ethnopharmacol. 2000, 70, 213–217. [Google Scholar] [CrossRef]
- Maleki, N.; Garjani, A.; Nazemiyeh, H.; Nilfouroushan, N.; Sadat, A.E.; Allameh, Z.; Hasannia, N. Potent anti-inflammatory activities of hydroalcoholic extract from aerial parts of Stachys inflata on rats. J. Ethnopharmacol. 2001, 75, 213–218. [Google Scholar] [CrossRef]
- Laggoune, S.; Zeghib, A.; Kabouche, A.; Kabouche, Z.; Maklad, Y.A.; Leon, F.; Brouard, I.; Bermejo, J.; Calliste, C.A.; Duroux, J.L. Components and antioxidant, anti-inflammatory, anti-ulcer and antinociceptive activities of the endemic species Stachys mialhesi de Noe. Arab. J. Chem. 2016, 9, S191–S197. [Google Scholar] [CrossRef]
- Ventola, C.L. The antibiotic resistance crisis: Part 1: Causes and threats. PT 2015, 40, 277. [Google Scholar]
- Ozdemir, F.A.; Kocak, G.; Kursat, M. Efficient callus formation and these callus antibacterial activities of a valuable medicinal plant Stachys cretica L. subsp. garana (Boiss) Rech. Prog. Nutr. 2017, 19, 92–96. [Google Scholar]
- Didry, N.; Seidel, V.; Dubreuil, L.; Tillequin, F.; Bailleul, F. Isolation and antibacterial activity of phenylpropanoid derivatives from Ballota nigra. J. Ethnopharmacol. 1999, 67, 197–202. [Google Scholar] [CrossRef] [PubMed]
- Souza, P.A.D.; Silva, C.G.; Machado, B.R.P.; Lucas, N.C.D.; Leitão, G.G.; Eleutherio, E.C.A.; Ortiz, G.M.D.; Benchetrit, L.C. Evaluation of antimicrobial, antioxidant and phototoxic activities of extracts and isolated compounds from Stachytarpheta cayennensis (Rich.) Vahl, Verbenaceae. Rev. Bras. Farmacogn. 2010, 20, 922–928. [Google Scholar] [CrossRef]
- Agampodi, V.A.; Katavic, P.; Collet, C.; Collet, T. Antibacterial and anti-inflammatory activity of extracts and major constituents derived from Stachytarpheta indica Linn. leaves and their potential implications for wound healing. Appl. Biochem. Biotechnol. 2022, 194, 6213–6254. [Google Scholar] [CrossRef] [PubMed]
- Kumar, S.; Stecher, G.; Li, M.; Knyaz, C.; Tamura, K. MEGA X: Molecular Evolutionary Genetics Analysis across computing platforms. Mol. Biol. Evol. 2018, 35, 1547–1549. [Google Scholar] [CrossRef] [PubMed]
- Ratnasingham, S.; Hebert, P.D. BOLD: The Barcode of Life Data System (http://www.barcodinglife.org). Mol. Ecol. Notes 2007, 7, 355–364. [Google Scholar] [CrossRef] [PubMed]
- Zahmanov, G.; Alipieva, K.; Simova, S.; Georgiev, M.I. Metabolic differentiations of dwarf elder by NMR-based metabolomics. Phytochem. Lett. 2015, 11, 404–409. [Google Scholar] [CrossRef]
- Marchev, A.S.; Dinkova-Kostova, A.T.; György, Z.; Mirmazloum, I.; Aneva, I.Y.; Georgiev, M.I. Rhodiola rosea L.: From golden root to green cell factories. Phytochem. Rev. 2016, 15, 515–536. [Google Scholar] [CrossRef]
- Singleton, V.L.; Orthofer, R.; Lamuela-Raventós, R.M. Analysis of total phenols and other oxidation substrates and antioxidants by means of folin-ciocalteu reagent. Meth. Enzymol. 1999, 299, 152–178. [Google Scholar]
- Chang, C.C.; Yang, M.H.; Wen, H.M.; Chern, J.C. Estimation of total flavonoid content in propolis by two complementary colorimetric methods. JFDA 2002, 10, 178–182. [Google Scholar]
- Prieto, P.; Pineda, M.; Aguilar, M. Spectrophotometric quantitation of antioxidant capacity through the formation of a phosphomolybdenum complex: Specific application to the determination of vitamin E. Anal. Biochem. 1999, 269, 337–341. [Google Scholar] [CrossRef]
- Benzie, I.F.; Strain, J.J. Ferric reducing/antioxidant power assay: Direct measure of total antioxidant activity of biological fluids and modified version for simultaneous measurement of total antioxidant power and ascorbic acid concentration. Meth. Enzymol. 1999, 299, 15–27. [Google Scholar]
- White, T.J.; Bruns, T.; Lee, S.; Taylor, J. Amplifications and direct sequencing of fungal ribosomal RNA genes for phylogenetics. In PCR Protocols: A Guide to Methods and Applications; Innis, M., Gelfand, D., Sninsky, J., White, T., Eds.; Academic Press: San Diego, CA, USA, 1990; pp. 315–322. [Google Scholar]
- Levin, R.A.; Wagner, W.L.; Hoch, P.C.; Nepokroeff, M.; Pires, J.C.; Zimmer, E.A.; Sytsma, K.J. Family-level relationships of Onagraceae based on chloroplast rbcL and ndhF data. Am. J. Bot. 2003, 90, 107–115. [Google Scholar] [CrossRef] [PubMed]
- Fazekas, A.J.; Burgess, K.S.; Kesanakurti, P.R.; Graham, S.W.; Newmaster, S.G.; Husband, B.C.; Percy, D.M.; Hajibabaei, M.; Barrett, S.C. Multiple multilocus DNA barcodes from the plastid genome discriminate plant species equally well. PLoS ONE 2008, 3, e2802. [Google Scholar] [CrossRef] [PubMed]
- Kuzmina, M.L.; Johnson, K.L.; Barron, H.R.; Hebert, P.D. Identification of the vascular plants of Churchill, Manitoba, using a DNA barcode library. BMC Ecol. 2012, 12, 25. [Google Scholar] [CrossRef]
- Tate, J.A.; Simpson, B.B. Paraphyly of Tarasa (Malvaceae) and diverse origins of the polyploid species. Syst. Bot. 2003, 28, 723–737. [Google Scholar]
- Sang, T.; Crawford, D.J.; Stuessy, T.F. Chloroplast DNA phylogeny, reticulate evolution, and biogeography of Paeonia (Paeoniaceae). Am. J. Bot. 1997, 84, 1120–1136. [Google Scholar] [CrossRef]
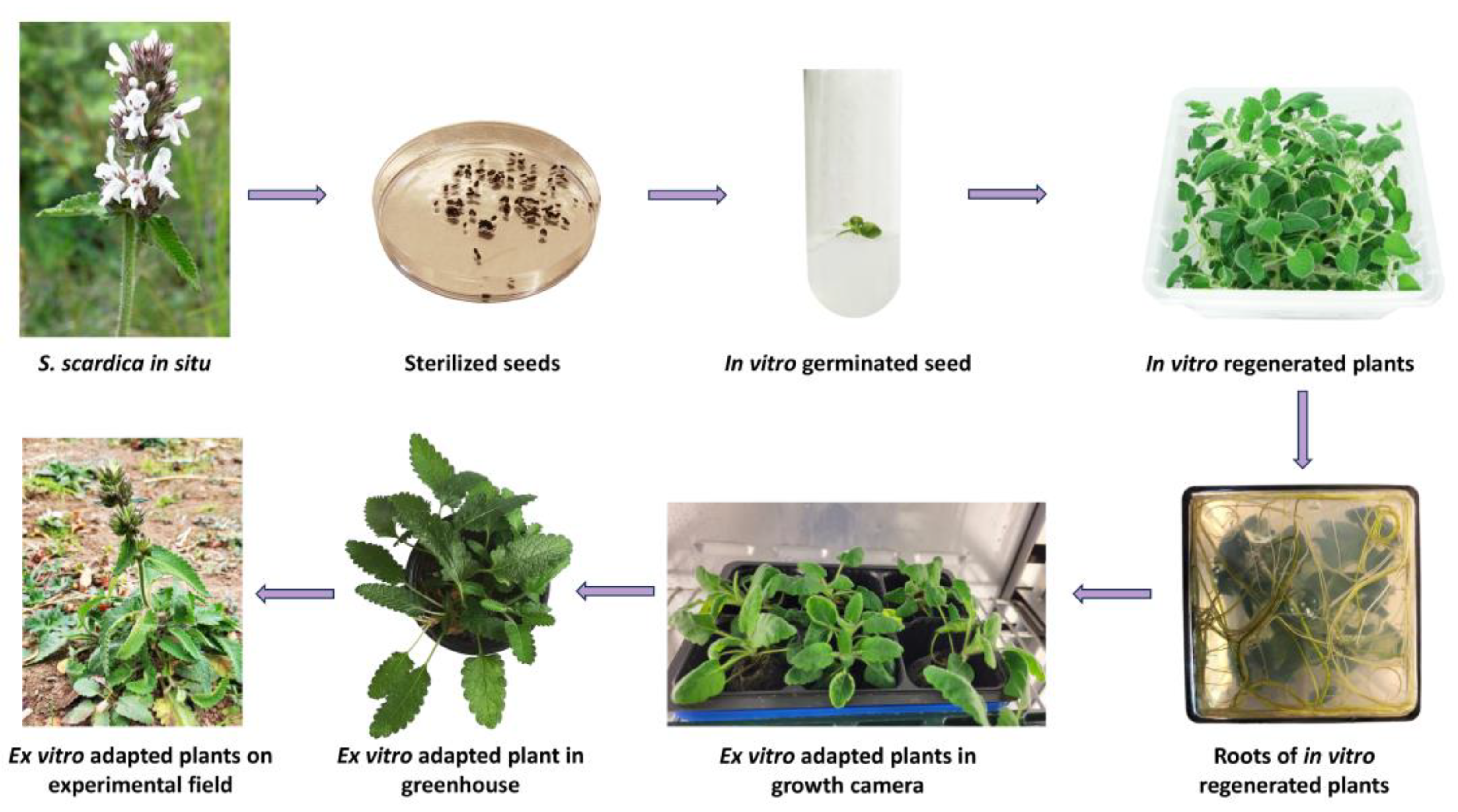


| Variants | Fresh Weight [g] | Dry Weight [g] | Number of Shoots | Number of Internodes | Growth Index | Root Length [cm] | Callus Formation |
|---|---|---|---|---|---|---|---|
| Control | 0.34 ± 0.2 | 0.05 ± 0.0 | 2.2 ± 0.40 | 1.54 ± 0.47 | 3.3 ± 1.1 | 5.73 ± 1.6 | - |
| 0.1 mg/L BA | 0.18 ± 0.04 | 0.04 ± 0.02 | 2 ± 0.00 | 1.5 ± 0.5 | 3.0 ± 1.0 | - | - |
| 0.5 mg/L BA | 0.53 ± 0.10 | 0.075 * ± 0.01 | 3.2 * ± 0.4 | 1.56 ± 0.25 | 5.0 ± 1.0 | - | - |
| 1 mg/L BA | 0.76 * ± 0.28 | 0.085 ± 0.02 | 6.4 * ± 1.2 | 1.67 ± 0.16 | 10.8 ± 2.8 | - | + 1 |
| 1.5 mg/L BA | 0.98 * ± 0.16 | 0.083 * ± 0.01 | 16.2 * ± 2.0 | 1.3 * ± 0.25 | 22.6 ± 2.4 | - | + 1 |
| 2 mg/L BA | 0.9 * ± 0.07 | 0.08 * ± 0.01 | 17.3 * ± 3.1 | 1.5 ± 0.29 | 24.0 ± 8.6 | - | + 2 |
| 2.5 mg/L BA | 1.38 * ± 0.22 | 0.12 * ± 0.01 | 15.2 * ± 2.7 | 1.6 * ± 0.35 | 23.8 ± 3.2 | - | + 2 |
| rbcL | matK | trnH-psbA | ITS | |
|---|---|---|---|---|
| S. scardica | S. officinalis | S. officinalis | B. officinalis | B. officinalis |
| (100%) | (100%) | (99%) | (84%) | |
| S. macrantha | S. macrantha | B. macrantha | ||
| (99%) | (99%) | (87%) | ||
| B. alopecuros | S. alopecuros | |||
| (96%) | (86%) |
| Metabolite | S. scardica In Situ | S. scardica In Vitro | S. scardica Ex Vitro | Selected Signals, Multiplicity and Coupling Constant a |
|---|---|---|---|---|
| Alanine | + | + | + | δ 1.47 (d, J = 7.2) |
| Glutamine | + | δ 2.13 (m)/δ 2.45 (m)/δ 3.74 (t) | ||
| α-Glucose | + | + | + | δ 5.17 (d, J = 3.8) |
| β-Glucose | + | + | + | δ 4.56 (d, J = 7.9)/3.18 (dd, J = 7.9, 9.2) |
| Sucrose | + | + | + | δ 5.37 (d, J = 3.8) |
| Acetic acid | + | + | δ 1.92 (s) | |
| Lactic acid | + | δ 1.31 (d, J = 6.9)/δ 4.08 m | ||
| Succinic acid | + | + | δ 2.48 (s) | |
| Formic acid | + | + | + | δ 8.45 (s) |
| Malic acid | + | δ 2.80 (dd, J = 16.9, 8.2)/δ 2.93 (dd, J= 16.9, 3.9) | ||
| Choline | + | δ 3.19 (s) | ||
| Allobetonicoside | + | δ 6.46 (d, J = 6.4)/δ 6.10 (bs)/δ 5.92 (d, J = 1.3)/ δ 5.30 (d, J = 8.3)/δ 5.02 (dd, J = 6.4, 1.2)/δ 4.71 (d, J = 7.8)/δ 2.33 (s) | ||
| 8-OAc-harpagide | + | δ 6.42 (d, J = 6.5)/δ 6.04 (d, J = 1.3)/δ 4.96 (dd, J = 6.5, 1.7)/ δ 4.67 (d, J = 7.9)/δ 2.04 (s)/δ 1.43 (s) | ||
| Verbascoside | + | + | + | δ 7.63 (d, J = 15.9)/δ 7.14 (d, J = 2.0)/7.05 (dd, J = 8.3, 2.0)/ δ 6.67 (dd, J = 8.3, 2.0)/δ 6.34 (d, J = 15.9)/4.93 (t, J = 9.6)/4.47 (d, J = 7.9)/ δ 2.81 (t, J = 7.2) 1.04 (d, J = 6.4) |
| Leucosepthoside A | + | - | + | δ 7.70 (d, J = 15.8)/δ 7.23 (d, J = 1.9)/7.16 (dd, J = 8.3, 2.0)/ δ 6.89 (dd, J = 8.3, 2.0)/δ 6.41 (d, J = 16.0)/4.93 (t, J = 9.6)/4.47 (d, J = 7.9)/ δ 3.88 (s)/δ 2.81 (t, J = 7.1) 1.04 (d, J = 6.4) |
| Chlorogenic acid | + | + | + | δ 7.60 (d, J = 15.7)/δ 7.13 (d, J = 2.2)/δ 7.06 (dd, J = 8.2, 2.2)/ δ 6.86 (d, J = 8.3)/δ 6.33 (d, J = 15.9)/δ 5.30 (td, J = 4.9, 10.9)/δ 4.18 (br q, J = 3.1) |
| Trigonelline | + | - | + | δ 9.12 (s)/δ 8.83 (m)/δ 8.07 (m)/δ 4.43 (s) |
| Test Strain | Inhibiton Zone (mm) | ||||
|---|---|---|---|---|---|
| (+) Control | 5% DMSO | Stachys scardica In Vitro | Stachys scardica Ex Vitro | ||
| Bacillus cereus ATCC 11778 | Gentamicin | 19.17 ± 0.45 | NZ | 8.09 ± 0.11 c | 12.43 ± 0.04 c |
| 10 µg/disk | |||||
| Staphylococcus epidermidis ATCC 12228 | Gentamicin | 24.51 ± 0.42 | NZ | 8.74 ± 0.21 c | 24.93 ± 0.78 a |
| 10 µg/disk | |||||
| Propionibacterium acnes (an isolate) | Clindamycin | 17.65 ± 0.50 | NZ | 7.78 ± 0.16 c | 26.58 ± 0.55 c |
| 2 µg/disk; | |||||
| Escherichia coli ATCC 25922 | Gentamicin | 18.74 ± 0.59 | NZ | NZ | NZ |
| 10 µg/disk | |||||
| Malassezia furfur ATCC14521 | Nystatin | 17.38 ± 0.77 | NZ | NZ | NZ |
| 100 units/disk | |||||
| Candida albicans ATCC10231 | Nystatin | 22.0 ± 0.09 | NZ | NZ | NZ |
| 100 units/disk | |||||
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mantovska, D.I.; Zhiponova, M.K.; Petrova, D.; Alipieva, K.; Bonchev, G.; Boycheva, I.; Evstatieva, Y.; Nikolova, D.; Tsacheva, I.; Simova, S.; et al. Exploring the Phytochemical Composition and Biological Potential of Balkan Endemic Species Stachys scardica Griseb. Plants 2024, 13, 30. https://doi.org/10.3390/plants13010030
Mantovska DI, Zhiponova MK, Petrova D, Alipieva K, Bonchev G, Boycheva I, Evstatieva Y, Nikolova D, Tsacheva I, Simova S, et al. Exploring the Phytochemical Composition and Biological Potential of Balkan Endemic Species Stachys scardica Griseb. Plants. 2024; 13(1):30. https://doi.org/10.3390/plants13010030
Chicago/Turabian StyleMantovska, Desislava I., Miroslava K. Zhiponova, Detelina Petrova, Kalina Alipieva, Georgi Bonchev, Irina Boycheva, Yana Evstatieva, Dilyana Nikolova, Ivanka Tsacheva, Svetlana Simova, and et al. 2024. "Exploring the Phytochemical Composition and Biological Potential of Balkan Endemic Species Stachys scardica Griseb" Plants 13, no. 1: 30. https://doi.org/10.3390/plants13010030
APA StyleMantovska, D. I., Zhiponova, M. K., Petrova, D., Alipieva, K., Bonchev, G., Boycheva, I., Evstatieva, Y., Nikolova, D., Tsacheva, I., Simova, S., & Yordanova, Z. P. (2024). Exploring the Phytochemical Composition and Biological Potential of Balkan Endemic Species Stachys scardica Griseb. Plants, 13(1), 30. https://doi.org/10.3390/plants13010030









