Role of the Dietary Phytochemical Curcumin in Targeting Cancer Cell Signalling Pathways
Abstract
1. Introduction
2. Methodology
3. Curcumin and Cancer: In Vitro and In Vivo Studies
3.1. Lung Cancer
3.2. Breast Cancer
3.3. Prostate Cancer
3.4. Brain Cancer
3.5. Pancreatic Cancer
3.6. Gastric Cancer
3.7. Leukaemia
4. Clinical Trials of Curcumin in Cancer
5. Concluding Remarks and Future Perspectives
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
References
- Sohn, S.-I.; Priya, A.; Balasubramaniam, B.; Muthuramalingam, P.; Sivasankar, C.; Selvaraj, A.; Valliammai, A.; Jothi, R.; Pandian, S. Biomedical applications and bioavailability of Curcumin—An updated overview. Pharmaceutics 2021, 13, 2102. [Google Scholar] [CrossRef]
- Dei Cas, M.; Ghidoni, R. Dietary Curcumin: Correlation between bioavailability and health potential. Nutrients 2019, 11, 2147. [Google Scholar] [CrossRef] [PubMed]
- Prasad, S.; Tyagi, A.K.; Aggarwal, B.B. Recent developments in delivery, bioavailability, absorption and metabolism of Curcumin: The golden pigment from golden spice. Cancer Res. Treat. 2014, 46, 2–18. [Google Scholar] [CrossRef] [PubMed]
- Stohs, S.J.; Chen, C.Y.O.; Preuss, H.G.; Ray, S.D.; Bucci, L.R.; Ji, J.; Ruff, K.J. The fallacy of enzymatic hydrolysis for the determination of bioactive curcumin in plasma samples as an indication of bioavailability: A comparative study. BMC Complement. Altern. Med. 2019, 19, 293. [Google Scholar] [CrossRef] [PubMed]
- Pandey, A.; Chaturvedi, M.; Mishra, S.; Kumar, P.; Somvanshi, P.; Chaturvedi, R. Reductive metabolites of curcumin and their therapeutic effects. Heliyon 2020, 6, e05469. [Google Scholar] [CrossRef]
- Mahale, J.; Singh, R.; Howells, L.M.; Britton, R.G.; Khan, S.M.; Brown, K. Detection of plasma curcuminoids from dietary intake of turmeric-containing food in human volunteers. Mol. Nutr. Food Res. 2018, 62, e1800267. [Google Scholar] [CrossRef] [PubMed]
- Schiborr, C.; Kocher, A.; Behnam, D.; Jandasek, J.; Toelstede, S.; Frank, J. The oral bioavailability of curcumin from micronized powder and liquid micelles is significantly increased in healthy humans and differs between sexes. Mol. Nutr. Food Res. 2014, 58, 516–527. [Google Scholar] [CrossRef]
- Arslan, A.K.K.; Uzunhisarcikli, E.; Yerer, M.B.; Bishayee, A. The golden spice curcumin in cancer: A perspective on finalized clinical trials during the last 10 years. J. Cancer Res. Ther. 2022, 18, 19–26. [Google Scholar] [CrossRef]
- Kunnumakkara, A.B.; Harsha, C.; Banik, K.; Vikkurthi, R.; Sailo, B.L.; Bordoloi, D.; Gupta, S.C.; Aggarwal, B.B. Is curcumin bioavailability a problem in humans: Lessons from clinical trials. Expert Opin. Drug Metab. Toxicol. 2019, 15, 705–733. [Google Scholar] [CrossRef]
- Memon, H.; Patel, B.M. Immune checkpoint inhibitors in non-small cell lung cancer: A bird’s eye view. Life Sci. 2019, 233, 116713. [Google Scholar] [CrossRef]
- Wang, J.Y.; Wang, X.; Wang, X.J.; Zheng, B.Z.; Wang, Y.; Wang, X.; Liang, B. Curcumin inhibits the growth via Wnt/beta-catenin pathway in non-small-cell lung cancer cells. Eur. Rev. Med. Pharmacol. Sci. 2018, 22, 7492–7499. [Google Scholar] [CrossRef]
- Li, X.; Ma, S.; Yang, P.; Sun, B.; Zhang, Y.; Sun, Y.; Hao, M.; Mou, R.; Jia, Y. Anticancer effects of curcumin on nude mice bearing lung cancer A549 cell subsets SP and NSP cells. Oncol. Lett. 2018, 16, 6756–6762. [Google Scholar] [CrossRef] [PubMed]
- Wu, G.Q.; Chai, K.Q.; Zhu, X.M.; Jiang, H.; Wang, X.; Xue, Q.; Zheng, A.H.; Zhou, H.Y.; Chen, Y.; Chen, X.C.; et al. Anti-cancer effects of curcumin on lung cancer through the inhibition of EZH2 and NOTCH1. Oncotarget 2016, 7, 26535–26550. [Google Scholar] [CrossRef] [PubMed]
- Wang, C.; Song, X.; Shang, M.; Zou, W.; Zhang, M.; Wei, H.; Shao, H. Curcumin exerts cytotoxicity dependent on reactive oxygen species accumulation in non-small-cell lung cancer cells. Future Oncol. 2019, 15, 1243–1253. [Google Scholar] [CrossRef] [PubMed]
- Zhou, G.Z.; Li, A.F.; Sun, Y.H.; Sun, G.C. A novel synthetic curcumin derivative MHMM-41 induces ROS-mediated apoptosis and migration blocking of human lung cancer cells A549. Biomed. Pharmacother. 2018, 103, 391–398. [Google Scholar] [CrossRef] [PubMed]
- Jin, H.; Qiao, F.; Wang, Y.; Xu, Y.; Shang, Y. Curcumin inhibits cell proliferation and induces apoptosis of human non-small cell lung cancer cells through the upregulation of miR-192-5p and suppression of PI3K/Akt signaling pathway. Oncol. Rep. 2015, 34, 2782–2789. [Google Scholar] [CrossRef] [PubMed]
- Man, S.; Zhang, L.; Cui, J.; Yang, L.; Ma, L.; Gao, W. Curcumin enhances the anti-cancer effects of Paris Saponin II in lung cancer cells. Cell Prolif. 2018, 51, e12458. [Google Scholar] [CrossRef]
- Jin, X.; Wang, J.; Shen, H.; Ran, R.; Xu, K.; Zhang, W.; Tong, X.; Feng, L. Curcumin co-treatment ameliorates resistance to gefitinib in drug- resistant NCI-H1975 lung cancer cells. J. Tradit. Chin. Med. 2017, 37, 355–360. [Google Scholar]
- Baharuddin, P.; Satar, N.; Fakiruddin, K.S.; Zakaria, N.; Lim, M.N.; Yusoff, N.M.; Zakaria, Z.; Yahaya, B.H. Curcumin improves the efficacy of cisplatin by targeting cancer stem-like cells through p21 and cyclin D1-mediated tumour cell inhibition in non-small cell lung cancer cell lines. Oncol. Rep. 2016, 35, 13–25. [Google Scholar] [CrossRef]
- Lheureux, S.; Denoyelle, C.; Ohashi, P.S.; De Bono, J.S.; Mottaghy, F.M. Molecularly targeted therapies in cancer: A guide for the nuclear medicine physician. Eur. J. Nucl. Med. Mol. Imaging 2017, 44, 41–54. [Google Scholar] [CrossRef]
- Yang, S.X.; Polley, E.; Lipkowitz, S. New insights on PI3K/AKT pathway alterations and clinical outcomes in breast cancer. Cancer Treat. Rev. 2016, 45, 87–96. [Google Scholar] [CrossRef]
- Januskeviciene, I.; Petrikaite, V. Heterogeneity of breast cancer: The importance of interaction between different tumor cell populations. Life Sci. 2019, 239, 117009. [Google Scholar] [CrossRef]
- Steelman, L.S.; Martelli, A.M.; Cocco, L.; Libra, M.; Nicoletti, F.; Abrams, S.L.; McCubrey, J.A. The therapeutic potential of mTOR inhibitors in breast cancer. Br. J. Clin. Pharmacol. 2016, 82, 1189–1212. [Google Scholar] [CrossRef]
- Hu, S.; Xu, Y.; Meng, L.; Huang, L.; Sun, H. Curcumin inhibits proliferation and promotes apoptosis of breast cancer cells. Exp. Ther. Med. 2018, 16, 1266–1272. [Google Scholar] [CrossRef] [PubMed]
- Coker-Gurkan, A.; Celik, M.; Ugur, M.; Arisan, E.D.; Obakan-Yerlikaya, P.; Durdu, Z.B.; Palavan-Unsal, N. Curcumin inhibits autocrine growth hormone-mediated invasion and metastasis by targeting NF-kappaB signaling and polyamine metabolism in breast cancer cells. Amino Acids 2018, 50, 1045–1069. [Google Scholar] [CrossRef] [PubMed]
- Coker-Gurkan, A.; Bulut, D.; Genc, R.; Arisan, E.D.; Obakan-Yerlikaya, P.; Palavan-Unsal, N. Curcumin prevented human autocrine growth hormone (GH) signaling mediated NF-kappaB activation and miR-183-96-182 cluster stimulated epithelial mesenchymal transition in T47D breast cancer cells. Mol. Biol. Rep. 2019, 46, 355–369. [Google Scholar] [CrossRef]
- Zou, J.; Zhu, L.; Jiang, X.; Wang, Y.; Wang, Y.; Wang, X.; Chen, B. Curcumin increases breast cancer cell sensitivity to cisplatin by decreasing FEN1 expression. Oncotarget 2018, 9, 11268–11278. [Google Scholar] [CrossRef]
- Attia, Y.M.; El-Kersh, D.M.; Ammar, R.A.; Adel, A.; Khalil, A.; Walid, H.; Eskander, K.; Hamdy, M.; Reda, N.; Mohsen, N.E.; et al. Inhibition of aldehyde dehydrogenase-1 and p-glycoprotein-mediated multidrug resistance by curcumin and vitamin D3 increases sensitivity to paclitaxel in breast cancer. Chem. Biol. Interact. 2020, 315, 108865. [Google Scholar] [CrossRef] [PubMed]
- Siegel, R.L.; Miller, K.D.; Jemal, A. Cancer statistics, 2020. CA Cancer J. Clin. 2020, 70, 7–30. [Google Scholar] [CrossRef]
- Lonergan, P.E.; Tindall, D.J. Androgen receptor signaling in prostate cancer development and progression. J. Carcinog. 2011, 10, 20. [Google Scholar] [CrossRef]
- Watson, P.A.; Arora, V.K.; Sawyers, C.L. Emerging mechanisms of resistance to androgen receptor inhibitors in prostate cancer. Nat. Rev. Cancer 2015, 15, 701–711. [Google Scholar] [CrossRef] [PubMed]
- Tomeh, M.A.; Hadianamrei, R.; Zhao, X. A Review of Curcumin and Its Derivatives as Anticancer Agents. Int. J. Mol. Sci. 2019, 20, 1033. [Google Scholar] [CrossRef] [PubMed]
- Thapa, D.; Ghosh, R. Chronic inflammatory mediators enhance prostate cancer development and progression. Biochem. Pharmacol. 2015, 94, 53–62. [Google Scholar] [CrossRef] [PubMed]
- Hayden, M.S.; Ghosh, S. NF-kappaB, the first quarter-century: Remarkable progress and outstanding questions. Genes Dev. 2012, 26, 203–234. [Google Scholar] [CrossRef]
- Kavya, K.; Kumar, M.N.; Patil, R.H.; Hegde, S.M.; Kiran Kumar, K.M.; Nagesh, R.; Babu, R.L.; Ramesh, G.T.; Chidananda Sharma, S. Differential expression of AP-1 transcription factors in human prostate LNCaP and PC-3 cells: Role of Fra-1 in transition to CRPC status. Mol. Cell Biochem. 2017, 433, 13–26. [Google Scholar] [CrossRef]
- Killian, P.H.; Kronski, E.; Michalik, K.M.; Barbieri, O.; Astigiano, S.; Sommerhoff, C.P.; Pfeffer, U.; Nerlich, A.G.; Bachmeier, B.E. Curcumin inhibits prostate cancer metastasis in vivo by targeting the inflammatory cytokines CXCL1 and -2. Carcinogenesis 2012, 33, 2507–2519. [Google Scholar] [CrossRef]
- Zhao, W.; Zhou, X.; Qi, G.; Guo, Y. Curcumin suppressed the prostate cancer by inhibiting JNK pathways via epigenetic regulation. J. Biochem. Mol. Toxicol. 2018, 32, e22049. [Google Scholar] [CrossRef]
- Dorai, T.; Gehani, N.; Katz, A. Therapeutic potential of curcumin in human prostate cancer-I. curcumin induces apoptosis in both androgen-dependent and androgen-independent prostate cancer cells. Prostate Cancer Prostatic. Dis. 2000, 3, 84–93. [Google Scholar] [CrossRef]
- Deeb, D.; Jiang, H.; Gao, X.; Hafner, M.S.; Wong, H.; Divine, G.; Chapman, R.A.; Dulchavsky, S.A.; Gautam, S.C. Curcumin sensitizes prostate cancer cells to tumor necrosis factor-related apoptosis-inducing ligand/Apo2L by inhibiting nuclear factor-kappaB through suppression of IkappaBalpha phosphorylation. Mol. Cancer Ther. 2004, 3, 803–812. [Google Scholar] [CrossRef]
- Klinger, N.V.; Mittal, S. Therapeutic potential of curcumin for the treatment of brain tumors. Oxid. Med. Cell Longev. 2016, 2016, 9324085. [Google Scholar] [CrossRef]
- Weathers, S.P.; Gilbert, M.R. Advances in treating glioblastoma. F1000Prime Rep. 2014, 6, 46. [Google Scholar] [CrossRef]
- Lathia, J.D.; Mack, S.C.; Mulkearns-Hubert, E.E.; Valentim, C.L.; Rich, J.N. Cancer stem cells in glioblastoma. Genes Dev. 2015, 29, 1203–1217. [Google Scholar] [CrossRef]
- Anand, P.; Kunnumakkara, A.B.; Newman, R.A.; Aggarwal, B.B. Bioavailability of curcumin: Problems and promises. Mol. Pharm. 2007, 4, 807–818. [Google Scholar] [CrossRef]
- Wong, S.C.; Kamarudin, M.N.A.; Naidu, R. Anticancer Mechanism of Curcumin on Human Glioblastoma. Nutrients 2021, 13, 950. [Google Scholar] [CrossRef]
- Zanotto-Filho, A.; Braganhol, E.; Klafke, K.; Figueiro, F.; Terra, S.R.; Paludo, F.J.; Morrone, M.; Bristot, I.J.; Battastini, A.M.; Forcelini, C.M.; et al. Autophagy inhibition improves the efficacy of curcumin/temozolomide combination therapy in glioblastomas. Cancer Lett. 2015, 358, 220–231. [Google Scholar] [CrossRef]
- Zhao, J.; Zhu, J.; Lv, X.; Xing, J.; Liu, S.; Chen, C.; Xu, Y. Curcumin potentiates the potent antitumor activity of ACNU against glioblastoma by suppressing the PI3K/AKT and NF-kappaB/COX-2 signaling pathways. Onco. Targets Ther. 2017, 10, 5471–5482. [Google Scholar] [CrossRef]
- Bojko, A.; Cierniak, A.; Adamczyk, A.; Ligeza, J. Modulatory Effects of Curcumin and Tyrphostins (AG494 and AG1478) on Growth Regulation and Viability of LN229 Human Brain Cancer Cells. Nutr. Cancer 2015, 67, 1170–1182. [Google Scholar] [CrossRef] [PubMed]
- Oettle, H. Progress in the knowledge and treatment of advanced pancreatic cancer: From benchside to bedside. Cancer Treat. Rev. 2014, 40, 1039–1047. [Google Scholar] [CrossRef] [PubMed]
- Masamune, A.; Suzuki, N.; Kikuta, K.; Satoh, M.; Satoh, K.; Shimosegawa, T. Curcumin blocks activation of pancreatic stellate cells. J. Cell Biochem. 2006, 97, 1080–1093. [Google Scholar] [CrossRef] [PubMed]
- Singh, A.; Settleman, J. EMT, cancer stem cells and drug resistance: An emerging axis of evil in the war on cancer. Oncogene 2010, 29, 4741–4751. [Google Scholar] [CrossRef]
- Bao, B.; Ali, S.; Banerjee, S.; Wang, Z.; Logna, F.; Azmi, A.S.; Kong, D.; Ahmad, A.; Li, Y.; Padhye, S.; et al. Curcumin analogue CDF inhibits pancreatic tumor growth by switching on suppressor microRNAs and attenuating EZH2 expression. Cancer Res. 2012, 72, 335–345. [Google Scholar] [CrossRef] [PubMed]
- Zhao, Z.; Li, C.; Xi, H.; Gao, Y.; Xu, D. Curcumin induces apoptosis in pancreatic cancer cells through the induction of forkhead box O1 and inhibition of the PI3K/Akt pathway. Mol. Med. Rep. 2015, 12, 5415–5422. [Google Scholar] [CrossRef]
- Wang, L.; Zhang, J.; Wan, L.; Zhou, X.; Wang, Z.; Wei, W. Targeting Cdc20 as a novel cancer therapeutic strategy. Pharmacol. Ther. 2015, 151, 141–151. [Google Scholar] [CrossRef]
- Diaz Osterman, C.J.; Gonda, A.; Stiff, T.; Sigaran, U.; Valenzuela, M.M.; Ferguson Bennit, H.R.; Moyron, R.B.; Khan, S.; Wall, N.R. Curcumin Induces Pancreatic Adenocarcinoma Cell Death Via Reduction of the Inhibitors of Apoptosis. Pancreas 2016, 45, 101–109. [Google Scholar] [CrossRef]
- Rugge, M.; Fassan, M.; Graham, D.Y. Epidemiology of Gastric Cancer. In Gastric Cancer; Strong, V.E., Ed.; Springer: Cham, Switzerland, 2015; pp. 23–34. [Google Scholar]
- Yamashita, K.; Sakuramoto, S.; Watanabe, M. Genomic and epigenetic profiles of gastric cancer: Potential diagnostic and therapeutic applications. Surg. Today 2011, 41, 24–38. [Google Scholar] [CrossRef] [PubMed]
- Kasi, P.D.; Tamilselvam, R.; Skalicka-Wozniak, K.; Nabavi, S.F.; Daglia, M.; Bishayee, A.; Pazoki-Toroudi, H.; Nabavi, S.M. Molecular targets of curcumin for cancer therapy: An updated review. Tumour. Biol. 2016, 37, 13017–13028. [Google Scholar] [CrossRef]
- Basnet, P.; Skalko-Basnet, N. Curcumin: An anti-inflammatory molecule from a curry spice on the path to cancer treatment. Molecules 2011, 16, 4567–4598. [Google Scholar] [CrossRef] [PubMed]
- Jin, H.; Lian, N.; Zhang, F.; Chen, L.; Chen, Q.; Lu, C.; Bian, M.; Shao, J.; Wu, L.; Zheng, S. Activation of PPARgamma/P53 signaling is required for curcumin to induce hepatic stellate cell senescence. Cell Death Dis. 2016, 7, e2189. [Google Scholar] [CrossRef]
- Fu, H.; Wang, C.; Yang, D.; Wei, Z.; Xu, J.; Hu, Z.; Zhang, Y.; Wang, W.; Yan, R.; Cai, Q. Curcumin regulates proliferation, autophagy, and apoptosis in gastric cancer cells by affecting PI3K and P53 signaling. J. Cell Physiol. 2018, 233, 4634–4642. [Google Scholar] [CrossRef]
- Liu, W.H.; Yuan, J.B.; Zhang, F.; Chang, J.X. Curcumin inhibits proliferation, migration and invasion of gastric cancer cells via Wnt3a/beta-catenin/EMT signaling pathway. Zhongguo Zhong Yao Za Zhi 2019, 44, 3107–3115. [Google Scholar] [CrossRef]
- Rafiq, S.; Raza, M.H.; Younas, M.; Naeem, F.; Adeeb, R.; Iqbal, J.; Anwar, P.; Sajid, U.; Manzoor, H.M. Molecular Targets of Curcumin and Future Therapeutic Role in Leukemia. J. Biosci. Med. 2018, 06, 33–50. [Google Scholar] [CrossRef]
- Jabbour, E.; Kantarjian, H. Chronic myeloid leukemia: 2016 update on diagnosis, therapy, and monitoring. Am. J. Hematol. 2016, 91, 252–265. [Google Scholar] [CrossRef] [PubMed]
- Wu, L.X.; Xu, J.H.; Wu, G.H.; Chen, Y.Z. Inhibitory effect of curcumin on proliferation of K562 cells involves down-regulation of p210(bcr/abl) initiated Ras signal transduction pathway. Acta Pharmacol. Sin. 2003, 24, 1155–1160. [Google Scholar] [PubMed]
- Mukherjee, A.; Sarkar, R.; Mukherjee, S.; Biswas, J.; Roy, M. Curcumin boosts up the efficacy of imatinib mesylate in chronic myelogenic leukemia cell line K-562 by modulation of various markers. Int. J. Curr. Microbiol. Appl. Sci. 2016, 5, 240–255. [Google Scholar] [CrossRef]
- Gupta, S.C.; Sundaram, C.; Reuter, S.; Aggarwal, B.B. Inhibiting NF-κB activation by small molecules as a therapeutic strategy. Biochim. Biophys. Acta (BBA)-Gene Regul. Mech. 2010, 1799, 775–787. [Google Scholar] [CrossRef]
- Pesakhov, S.; Nachliely, M.; Barvish, Z.; Aqaqe, N.; Schwartzman, B.; Voronov, E.; Sharoni, Y.; Studzinski, G.P.; Fishman, D.; Danilenko, M. Cancer-selective cytotoxic Ca2+ overload in acute myeloid leukemia cells and attenuation of disease progression in mice by synergistically acting polyphenols curcumin and carnosic acid. Oncotarget 2016, 7, 31847–31861. [Google Scholar] [CrossRef]
- Rao, J.; Xu, D.-R.; Zheng, F.-M.; Long, Z.-J.; Huang, S.-S.; Wu, X.; Zhou, W.-H.; Huang, R.-W.; Liu, Q. Curcumin reduces expression of Bcl-2, leading to apoptosis in daunorubicin-insensitive CD34+ acute myeloid leukemia cell lines and primary sorted CD34+ acute myeloid leukemia cells. J. Transl. Med. 2011, 9, 71. [Google Scholar] [CrossRef]
- Tima, S.; Ichikawa, H.; Ampasavate, C.; Okonogi, S.; Anuchapreeda, S. Inhibitory effect of turmeric curcuminoids on FLT3 expression and cell cycle arrest in the FLT3-overexpressing EoL-1 leukemic cell line. J. Nat. Prod. 2014, 77, 948–954. [Google Scholar] [CrossRef]
- Hallek, M. Chronic lymphocytic leukemia: 2013 update on diagnosis, risk stratification and treatment. Am. J. Hematol. 2013, 88, 803–816. [Google Scholar] [CrossRef]
- Gonzalez-Rodriguez, A.P.; Contesti, J.; Huergo-Zapico, L.; Lopez-Soto, A.; Fernandez-Guizan, A.; Acebes-Huerta, A.; Gonzalez-Huerta, A.J.; Gonzalez, E.; Fernandez-Alvarez, C.; Gonzalez, S. Prognostic significance of CD8 and CD4 T cells in chronic lymphocytic leukemia. Leuk Lymphoma 2010, 51, 1829–1836. [Google Scholar] [CrossRef]
- Ghosh, A.K.; Kay, N.E.; Secreto, C.R.; Shanafelt, T.D. Curcumin inhibits prosurvival pathways in chronic lymphocytic leukemia B cells and may overcome their stromal protection in combination with EGCG. Clin. Cancer Res. 2009, 15, 1250–1258. [Google Scholar] [CrossRef] [PubMed]
- Mishra, D.; Singh, S.; Narayan, G. Curcumin induces apoptosis in Pre-B acute lymphoblastic leukemia cell lines Via PARP-1 cleavage. Asian Pac. J. Cancer Prev. 2016, 17, 3865–3869. [Google Scholar]
- Guo, Y.; Li, Y.; Shan, Q.; He, G.; Lin, J.; Gong, Y. Curcumin potentiates the anti-leukemia effects of imatinib by downregulation of the AKT/mTOR pathway and BCR/ABL gene expression in Ph+ acute lymphoblastic leukemia. Int. J. Biochem. Cell Biol. 2015, 65, 1–11. [Google Scholar] [CrossRef]
- Zoi, V.; Galani, V.; Lianos, G.D.; Voulgaris, S.; Kyritsis, A.P.; Alexiou, G.A. The role of curcumin in cancer treatment. Biomedicines 2021, 9, 1086. [Google Scholar] [CrossRef] [PubMed]
- Bayet-Robert, M.; Kwiatkowski, F.; Leheurteur, M.; Gachon, F.; Planchat, E.; Abrial, C.; Mouret-Reynier, M.A.; Durando, X.; Barthomeuf, C.; Chollet, P. Phase I dose escalation trial of docetaxel plus curcumin in patients with advanced and metastatic breast cancer. Cancer Biol. Ther. 2010, 9, 8–14. [Google Scholar] [CrossRef] [PubMed]
- Ghalaut, V.S.; Sangwan, L.; Dahiya, K.; Ghalaut, P.S.; Dhankhar, R.; Saharan, R. Effect of imatinib therapy with and without turmeric powder on nitric oxide levels in chronic myeloid leukemia. J. Oncol. Pharm. Pract. 2012, 18, 186–190. [Google Scholar] [CrossRef]
- Ledda, A.; Belcaro, G.; Dugall, M.; Luzzi, R.; Scoccianti, M.; Togni, S.; Appendino, G.; Ciammaichella, G. Meriva®, a lecithinized curcumin delivery system, in the control of benign prostatic hyperplasia: A pilot, product evaluation registry study. Panminerva Med. 2012, 54, 17. [Google Scholar] [PubMed]
- Kim, S.G.; Veena, M.S.; Basak, S.K.; Han, E.; Tajima, T.; Gjertson, D.W.; Starr, J.; Eidelman, O.; Pollard, H.B.; Srivastava, M.; et al. Curcumin treatment suppresses IKKbeta kinase activity of salivary cells of patients with head and neck cancer: A pilot study. Clin. Cancer Res. 2011, 17, 5953–5961. [Google Scholar]
- Plummer, S.M.; Hill, K.A.; Festing, M.F.; Steward, W.P.; Gescher, A.J.; Sharma, R.A. Clinical development of leukocyte cyclooxygenase 2 activity as a systemic biomarker for cancer chemopreventive agents. Cancer Epidemiol. Prev. Biomark. 2001, 10, 1295–1299. [Google Scholar]
- Garcea, G.; Berry, D.P.; Jones, D.J.; Singh, R.; Dennison, A.R.; Farmer, P.B.; Sharma, R.A.; Steward, W.P.; Gescher, A.J. Consumption of the putative chemopreventive agent curcumin by cancer patients: Assessmentof curcumin levels in the colorectum and their pharmacodynamic consequences. Cancer Epidemiol. Prev. Biomark. 2005, 14, 120–125. [Google Scholar] [CrossRef]
- Sharma, R.A.; Euden, S.A.; Platton, S.L.; Cooke, D.N.; Shafayat, A.; Hewitt, H.R.; Marczylo, T.H.; Morgan, B.; Hemingway, D.; Plummer, S.M.; et al. Phase I clinical trial of oral curcumin: Biomarkers of systemic activity and compliance. Clin. Cancer Res. 2004, 10, 6847–6854. [Google Scholar] [CrossRef] [PubMed]
- Irving, G.R.; Howells, L.M.; Sale, S.; Kralj-Hans, I.; Atkin, W.S.; Clark, S.K.; Britton, R.G.; Jones, D.J.; Scott, E.N.; Berry, D.P.; et al. Prolonged biologically active colonic tissue levels of curcumin achieved after oral administration–a clinical pilot study including assessment of patient acceptability. Cancer Prev. Res. 2013, 6, 119–128. [Google Scholar] [CrossRef] [PubMed]
- He, Z.Y.; Shi, C.B.; Wen, H.; Li, F.L.; Wang, B.L.; Wang, J. Upregulation of p53 expression in patients with colorectal cancer by administration of curcumin. Cancer Investig. 2011, 29, 208–213. [Google Scholar] [CrossRef] [PubMed]
- Carroll, R.E.; Benya, R.V.; Turgeon, D.K.; Vareed, S.; Neuman, M.; Rodriguez, L.; Kakarala, M.; Carpenter, P.M.; McLaren, C.; Meyskens, F.L., Jr.; et al. Phase IIa clinical trial of curcumin for the prevention of colorectal neoplasia. Cancer Prev. Res. 2011, 4, 354–364. [Google Scholar] [CrossRef] [PubMed]
- Ide, H.; Tokiwa, S.; Sakamaki, K.; Nishio, K.; Isotani, S.; Muto, S.; Hama, T.; Masuda, H.; Horie, S. Combined inhibitory effects of soy isoflavones and curcumin on the production of prostate-specific antigen. Prostate 2010, 70, 1127–1133. [Google Scholar] [CrossRef] [PubMed]
- Hejazi, J.; Rastmanesh, R.; Taleban, F.A.; Molana, S.H.; Hejazi, E.; Ehtejab, G.; Hara, N. Effect of Curcumin Supplementation During Radiotherapy on Oxidative Status of Patients with Prostate Cancer: A Double Blinded, Randomized, Placebo-Controlled Study. Nutr. Cancer 2016, 68, 77–85. [Google Scholar] [CrossRef]
- Kanai, M.; Otsuka, Y.; Otsuka, K.; Sato, M.; Nishimura, T.; Mori, Y.; Kawaguchi, M.; Hatano, E.; Kodama, Y.; Matsumoto, S.; et al. A phase I study investigating the safety and pharmacokinetics of highly bioavailable curcumin (Theracurmin) in cancer patients. Cancer Chemother. Pharmacol. 2013, 71, 1521–1530. [Google Scholar] [CrossRef]
- Kanai, M.; Yoshimura, K.; Asada, M.; Imaizumi, A.; Suzuki, C.; Matsumoto, S.; Nishimura, T.; Mori, Y.; Masui, T.; Kawaguchi, Y.; et al. A phase I/II study of gemcitabine-based chemotherapy plus curcumin for patients with gemcitabine-resistant pancreatic cancer. Cancer Chemother. Pharmacol. 2011, 68, 157–164. [Google Scholar] [CrossRef]
- Epelbaum, R.; Schaffer, M.; Vizel, B.; Badmaev, V.; Bar-Sela, G. Curcumin and gemcitabine in patients with advanced pancreatic cancer. Nutr. Cancer 2010, 62, 1137–1141. [Google Scholar] [CrossRef]
- Dhillon, N.; Aggarwal, B.B.; Newman, R.A.; Wolff, R.A.; Kunnumakkara, A.B.; Abbruzzese, J.L.; Ng, C.S.; Badmaev, V.; Kurzrock, R. Phase II trial of curcumin in patients with advanced pancreatic cancer. Clin. Cancer Res. 2008, 14, 4491–4499. [Google Scholar] [CrossRef]
- Cruz-Correa, M.; Hylind, L.M.; Marrero, J.H.; Zahurak, M.L.; Murray-Stewart, T.; Casero, R.A., Jr.; Montgomery, E.A.; Iacobuzio-Donahue, C.; Brosens, L.A.; Offerhaus, G.J.; et al. Efficacy and Safety of Curcumin in Treatment of Intestinal Adenomas in Patients with Familial Adenomatous Polyposis. Gastroenterology 2018, 155, 668–673. [Google Scholar] [CrossRef] [PubMed]
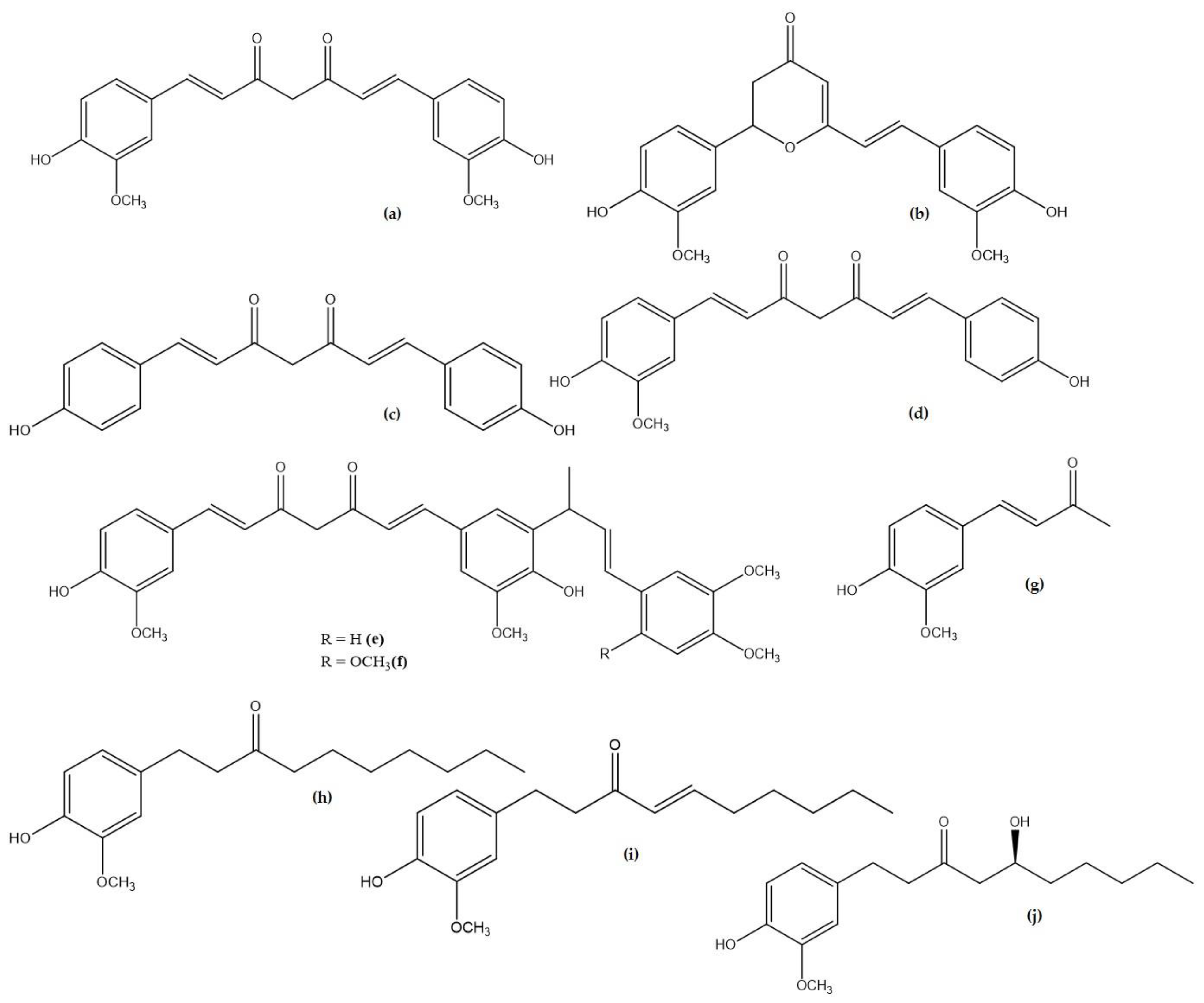
| Compound Name | Plant Origin | Molecular Formula | Pubchem ID |
|---|---|---|---|
| Curcumin (a) | Curcuma longa (Turmeric) | C21H20O6 | 969516 |
| Cyclocurcumin (b) | Curcuma longa (Turmeric) | C21H20O6 | 69879809 |
| Bisdemethoxycurcumin (c) | Curcuma longa (Turmeric) | C19H16O4 | 5315472 |
| Demethoxycurcumin (d) | Curcuma longa (Turmeric) | C20H18O5 | 5469424 |
| Cassumunin A (e) | Zingiber cassumunar (Ginger) | C33H34O8 | 10460395 |
| Cassumunin B (f) | Zingiber cassumunar (Ginger) | C34H36O9 | 10054109 |
| Dehydrozingerone (g) | Zingiber officinale Roscoe (Ginger) | C11H12O3 | 5354238 |
| 6-Paradol (h) | Zingiber officinale Roscoe (Ginger) | C17H26O3 | 94378 |
| 6-Shogaol (i) | Zingiber officinale (Ginger) | C17H24O3 | 5281794 |
| 6-Gingerol (j) | Zingiber officinale Roscoe (Ginger) | C17H26O4 | 442793 |
| Formula | C21H20O6 |
|---|---|
| Chemical name | [(1E,6E)-1,7-bis(4-hydroxy-3-methoxyphenyl)hepta-1,6-diene-3,5-dione] |
| Molecular weight | 368.38 g/mol |
| pKa values | First (pKa 7.7–8.5) Second (pKa 8.5–10.4) Third (pKa 9.5–10.7) |
| Stable at pH | Between 1–6 |
| Num. heavy atoms | 27 |
| Num. rotatable bonds | 8 |
| Num. H-bond acceptors | 6 |
| Num. H-bond donors | 2 |
| Molar refractivity | 102.80 |
| Melting temperature | 176 °C to 183 °C |
| Water solubility | 0.4 mg/mL |
| Bioavailability score | 0.55 |
| Gastrointestinal absorption | High |
| Blood–brain barrier (BBB) permeant | No |
| Formulations | Curcumin Dose Administered | Plasma Levels of Curcumin |
|---|---|---|
| Use of lipid particles | 650 mg | 22.4 ng/mL at 2.4 h |
| From 2 to 4 g | 30–40 ng/mL between 2 to 4 h | |
| Use of micelles | 500 mg | 1189 ng/mL at 1.1 h |
| 210 mg/day per 4 days | 253 ng/mL (total curcuminoids) | |
| Use of piperine | 2 g + 5 mg | 6.92 ng/mL (mean) |
| 4 g + 24 mg | 136–176 ng/mL (range) | |
| 2 g/kg + 20 mg/kg | 180 ng/mL at 0.75 h | |
| Use of hydrophilic nanoparticles | 30 mg | 1.8 ± 2.8 ng/mL |
| 376 mg | 27.3 ± 6.4 ng/mL at 1.4 h | |
| 30 mg | 25.5 ± 12.2 ng/mL | |
| Multiple doses of 200 or 400 mg/day | 324 ng/mL with a dose of 200 mg of Theracurmin® and 440 ng/mL with a dose of 400 mg | |
| 150 or 210 mg | 189 ± 48 ng/mL with a dose of 150 mg and 275 ± 7 ng/mL with a dose of 210 mg |
| Type of Cancer | Cell Signalling Pathway | Effect | Model Used | Dose Administered | References |
|---|---|---|---|---|---|
| Lung Cancer | Wnt/β-catenin | Downregulation/inhibition | Human cell line A549 | 60 µM | [10,11,12,13,14,15,16,17,18,19] |
| VEGF | Downregulation/inhibition | Nude mice | 100 mg/kg | ||
| NF-κB | Downregulation/inhibition | Nude mice | 100 mg/kg | ||
| Notch 1 | Downregulation/inhibition | Human lung cancer cell lines | 6 µM | ||
| ERK 1/2 | Downregulation/inhibition | Human NCI-H1975 line | 10 ng/mL | ||
| Breast Cancer | Akt/mTOR | Downregulation/inhibition | Human breast cell lines | 10 or 30 µM | [20,21,22,23,24,25,26,27,28] |
| NF-κB | Downregulation/inhibition | Human breast cell lines | 20 or 25 µM | ||
| MDR-1 | Downregulation/inhibition | MCF-7 breast cancer cell line | 1.3 µM | ||
| Bcl-2 and Bcl- xL | Downregulation/inhibition | T47D human breast cells | 20 µM | ||
| FEN1 | Downregulation/inhibition | MCF-7 breast cancer cell line | 0–50 µM | ||
| Autocrine GH | Downregulation/inhibition | T47D human breast cells | 20 µM | ||
| Prostate Cancer | Androgen receptor-dependent | Downregulation/inhibition | LNCaP cell line | 0.25 µM and 0.5 µM | [29,30,31,32,33,34,35,36,37,38,39] |
| Brain Cancer | STAT3 | Downregulation/inhibition | Human GBM stem cells | 25 µM | [40,41,42,43,44,45,46,47] |
| IAP | Downregulation/inhibition | Human GBM stem cells | 25 µM | ||
| MAPK | Upregulation/activation | Human GBM stem cells | 25 µM | ||
| Pancreatic cancer | Platelet-derived growth factor | Downregulation/inhibition | Rat pancreatic stellate cells | 25 µM | [48,49,50,51,52,53,54] |
| PI3 K/Akt | Downregulation/inhibition | Panc-1 human pancreatic cells | 20 µM | ||
| Cdc20 | Downregulation/inhibition | Patu8988 and Panc-1 human cell lines | 10 or 20 µM | ||
| IAP | Downregulation/inhibition | PANC-1 human cells | 10/50/100 µM | ||
| Gastric cancer | PI3K | Downregulation/inhibition | Human SGC-7901 and BGC-823 cells | 10/20/40 µM | [55,56,57,58,59,60,61] |
| BCL-2 | Downregulation/inhibition | Human gastric cell lines | 20 µM | ||
| Wnt3 a/β-catenin/EMT | Downregulation/inhibition | Human gastric cell lines | 20 µM | ||
| Leukaemia-CML | MAPK | Downregulation/inhibition | Human K562 cell line | 5 or 10 mg/L | [62,63,64,65,66,67,68,69,70,71,72,73,74,75] |
| p210 BCR-ABL | Downregulation/inhibition | Human K562 cell line | 5 or 10 mg/L | ||
| Hsp90 | Downregulation/inhibition | Human K562 cell line | 30 µM | ||
| Leukaemia-CLL | AKT | Downregulation/inhibition | Human CLL B cells | 10–12.5 µM | |
| NF-κB | Downregulation/inhibition | Human CLL B cells | 10–12.5 µM | ||
| STAT3 | Downregulation/inhibition | Human CLL B cells | 10–12.5 µM | ||
| XIAP | Downregulation/inhibition | Human CLL B cells | 10–12.5 µM | ||
| Mcl-1 | Downregulation/inhibition | Human CLL B cells | 10–12.5 µM | ||
| Leukaemia-AML | MMP | Downregulation/inhibition | Human SHI-1 cells | 6.25–25 µM | |
| Bcl-2 | Downregulation/inhibition | Primary human CD34+ AML cells | 0–80 µM | ||
| MAPK | Downregulation/inhibition | Human SHI-1 cells | 6.25–25 µM | ||
| Leukaemia-ALL | AKT/mTOR | Downregulation/inhibition | Human ALL cell lines | 0–40 µM | |
| BCR/ABL | Downregulation/inhibition | Human ALL cell lines | 0–40 µM | ||
| ABL/STAT5 | Downregulation/inhibition | Human ALL cell lines | 0–40 µM |
| Cancer Type | Study Type | Number of Patients in the Study | Treatment | Endpoints | Results | References |
|---|---|---|---|---|---|---|
| Breast cancer | Clinical trial | 14 | Docetaxel + Curcumin (0.5–8 g/day for 7 days) | VEGF and tumour markers levels; Maximal tolerated dose of curcumin; Efficacy; Safety; Toxicity | Decreased levels of VEGF; No cancer progression; Low frequency of toxic effects; Partial response in some patients | [76] |
| Chronic Myeloid Leukaemia | Randomized controlled trial | 50 | Imatinib (400 mg twice daily) + Curcumin (5 g three times daily for 6 weeks) | Plasma nitric oxide levels | Reduced nitric oxide levels | [77] |
| Benign Prostatic Hypertrophy | Pilot project | 61 | Curcumin (1 g per day for 24 weeks) | Quality of life; Signs and symptoms | Improved quality of life; Reduced signs and symptoms of the disease | [78] |
| Head and Neck Squamous Cell Carcinoma | Pilot study | 21 | Single dose of curcumin (1 g) | Cytokine levels and Iκkβ kinase activity in saliva | Reduced IκKβ activity in salivary cells | [79] |
| Colorectal Cancer | Dose-escalation pilot study | 15 | Curcumin (40–200 mg per day for 29 days) | PGE2 levels and COX-2 activity | Dose-dependent decrease in PGE2 levels | [80] |
| Does-escalation trial (Phase I) | 12 | Curcumin (0.45 g, 1.8 g and 3.6 g per day for 7 days) | Concentrations of curcumin and its metabolites in plasma, and colorectal tissue | Concentrations of Biologically active curcumin in the colorectal tissue | [81] | |
| Does-escalation trial (Phase I) | 15 | Curcumin (0.45–3.6 g per day for 120 days) | PGE2 and glutathione S-transferase activity in blood; Concentration of curcumin and its metabolites in plasma, faeces and urine | Very low levels of curcumin and its metabolites in plasma and urine and dose-dependent decrease in PGE2 levels | [82] | |
| Pilot study | 26 | Curcumin (2.35 g per day for 14 days) | Tolerance, safety and levels of curcumin in the colonic mucosa | Prolonged biologically active levels of curcumin achieved in the colon. Safe and well tolerated | [83] | |
| Clinical trial (Phase I) | 126 | Curcumin (360 mg three times daily for 10–30 days) | p53 expression and TNF-α levels in serum and colorectal tissue | Increased expression of p53; Decreased levels of TNF-α in serum and tissue | [84] | |
| Clinical trial (Phase II) | 44 | Curcumin (2 and 4 g per day for 30 days) | Total number and concentration of 5-hydroxyeicosatetraenoic acid and PGE2 within aberrant crypt foci and normal mucosa | Reduced number of aberrant crypt foci with a dose of 4 g per day | [85] | |
| Prostate cancer | Randomized controlled trial | 85 | Soy isoflavones (40 mg) + curcumin (100 mg) for 180 days | Prostate-specific antigen levels in serum | Decreased levels of prostate-specific antigen | [86] |
| Randomized controlled trial | 40 | Radiotherapy + curcumin (3 g per day for 90 days) | Altered activity of antioxidant enzymes and biochemical and clinical progression-free survivals | Decreased levels of prostate-specific antigen and considerable antioxidant effect | [87] | |
| Clinical trial (Phase I) | 16 | Curcumin (200–400 mg per day for 270 days) | Safety, cytokine levels, pharmacokinetics, NF-κB activity, efficacy and quality of life | No noteworthy changes in NF-κB activity or cytokine levels, safe, good pharmacokinetics and improved quality of life | [88] | |
| Pancreatic cancer | Clinical trial (Phase I/II) | 21 | Gemcitabine + curcumin (8 g per day for 14 days) | Efficacy, patient compliance and toxicity | Median overall survival time of 161 days; Safe and well tolerated | [89] |
| Clinical trial (Phase II) | 17 | Curcumin (8 g per day for 30 days) | Toxicity profile and time to tumour progression | Tumour progression of 1–12 months and high frequency of side effects | [90] | |
| Clinical trial (Phase II) | 25 | Curcumin (8 g per day for 60 days) | Tumour markers, tumour response, and adverse effects | Biological response in only 2 patients, poor oral bioavailability, and no toxicity | [91] | |
| Intestinal Adenoma | Randomized controlled trial | 44 | Curcumin (1.5 g twice a day for 12 months) | Mean polyp size, total number of polyps and adverse effects | No significant clinical response Very few adverse effects | [92] |
| Cancer Type | Treatment | Project Title | NCT * | Phase | Estimated/Actual Completion Date |
|---|---|---|---|---|---|
| Breast Cancer | Curcumin ® (CUC-01)+ paclitaxel | Curcumin in Combination with Chemotherapy in Advanced Breast Cancer | NCT03072992 | 2 | 30 June 2019 |
| Colorectal Cancer | Avastin/FOLFIRI + curcumin | Avastin/FOLFIRI in Combination with Curcumin in Colorectal Cancer Patients with Unresectable Metastasis | NCT02439385 | 2 | 1 August 2019 |
| Prostate Cancer | Curcumin + radiation | Nanocurcumin for Prostate Cancer Patients Undergoing Radiotherapy (RT) | NCT02724618 | 2 | April 2022 |
| Breast Cancer | Curcumin | A ‘Window Trial” on Curcumin for Invasive Breast Cancer Primary Tumours | NCT03980509 | 1 | 30 December 2022 |
| Cervical Cancer | Curcumin | Curcumin in Advanced Cervical Cancer | NCT04294836 | 2 | 31 December 2023 |
| Prostate Cancer | Curcumin | Trial of Curcumin to Prevent Progression of Low-risk Prostate Cancer Under Active Surveillance | NCT03769766 | 3 | November 2026 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mishra, A.P.; Swetanshu; Singh, P.; Yadav, S.; Nigam, M.; Seidel, V.; Rodrigues, C.F. Role of the Dietary Phytochemical Curcumin in Targeting Cancer Cell Signalling Pathways. Plants 2023, 12, 1782. https://doi.org/10.3390/plants12091782
Mishra AP, Swetanshu, Singh P, Yadav S, Nigam M, Seidel V, Rodrigues CF. Role of the Dietary Phytochemical Curcumin in Targeting Cancer Cell Signalling Pathways. Plants. 2023; 12(9):1782. https://doi.org/10.3390/plants12091782
Chicago/Turabian StyleMishra, Abhay Prakash, Swetanshu, Pratichi Singh, Shikha Yadav, Manisha Nigam, Veronique Seidel, and Celia Fortuna Rodrigues. 2023. "Role of the Dietary Phytochemical Curcumin in Targeting Cancer Cell Signalling Pathways" Plants 12, no. 9: 1782. https://doi.org/10.3390/plants12091782
APA StyleMishra, A. P., Swetanshu, Singh, P., Yadav, S., Nigam, M., Seidel, V., & Rodrigues, C. F. (2023). Role of the Dietary Phytochemical Curcumin in Targeting Cancer Cell Signalling Pathways. Plants, 12(9), 1782. https://doi.org/10.3390/plants12091782











