Phosphorus Use Efficiency of Leafy Brassica sp. Grown in Three Contrasting Soils: Growth, Enzyme Activity and Phosphorus Fractionation
Abstract
1. Introduction
2. Results
2.1. Plant Growth and P Accumulation
2.2. P use Efficiency Parameters
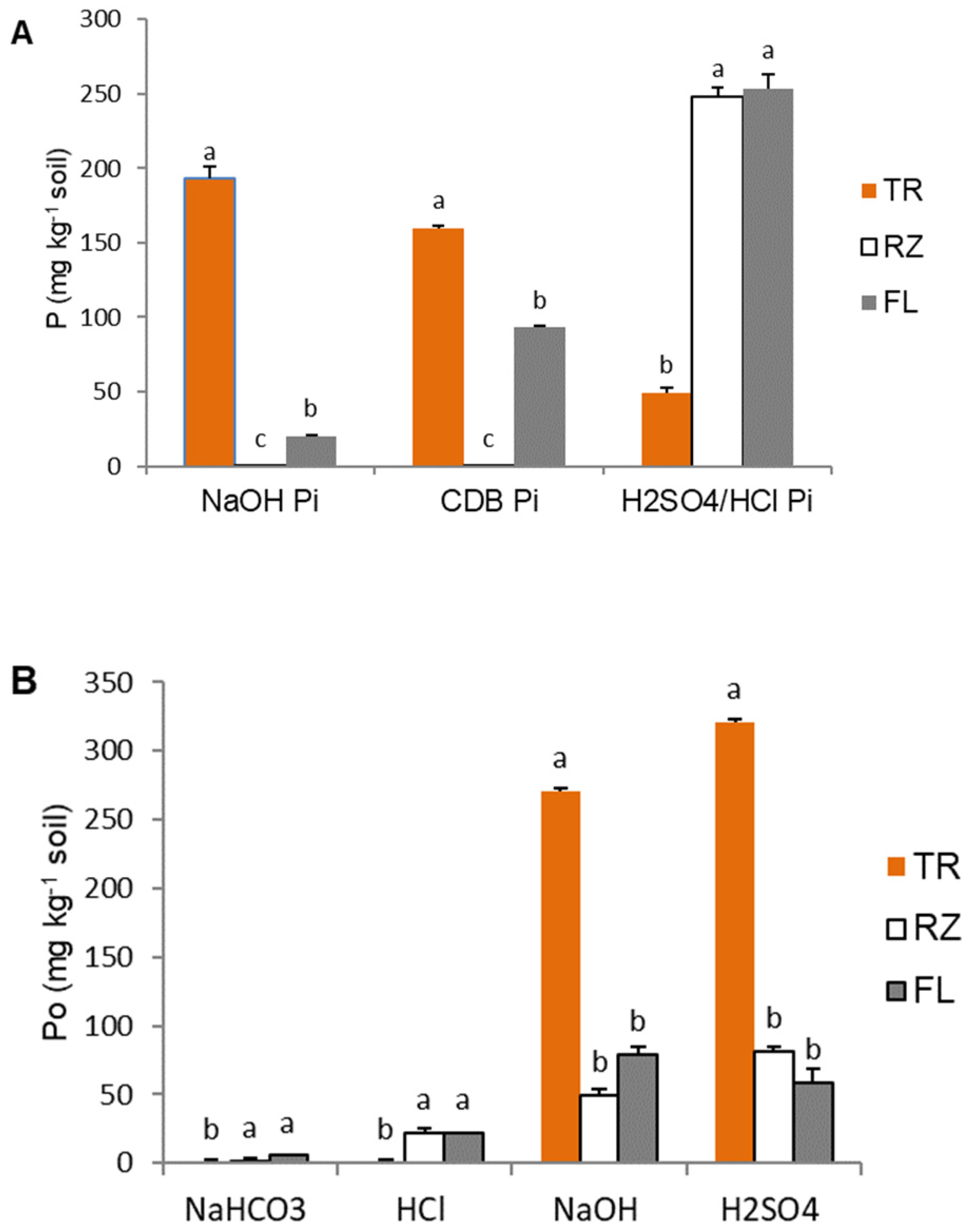
2.3. Soil P Fractions
3. Discussion
4. Materials and Methods
4.1. Plant and Soil Materials
4.2. Experiment Set Up
4.3. Plant Harvest and Measurements
- Specific root length (SRL) (m root g−1 root DW);
- Specific phosphorus uptake (SPU)—total plant P/total root length (mg P m−1).
4.4. P Fractionation
4.5. Soil Enzymatic Analyses
4.6. P use Efficiency Parameters
- PupE = plant P content/soil available P (mg mg−1);
- PutE = plant DW/P content (g DW mg P−1);
4.7. Statistical Analyses
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Cordell, D.; Drangert, J.O.; White, S. The story of phosphorus: Global food security and food for thought. Glob. Environ. Chang. 2009, 19, 292–305. [Google Scholar] [CrossRef]
- Nadeem, M.; Wu, J.; Ghaffari, H.; Kedir, A.J.; Saleem, S.; Mollier, A.; Singh, J.; Cheema, M. Understanding the adaptive mechanisms of plants to enhance phosphorus use efficiency on podzolic soils in boreal agroecosystem. Front. Plant Sci. 2022, 13, 804058. [Google Scholar] [CrossRef]
- Celi, L.; Barberis, E. Abiotic Stabilization of Organic Phosphorus in the Environment. In Organic Phosphorus in the Environment; Turner, B.L., Frossard, E., Baldwin, D.S., Eds.; CABI Publishing: Cambridge, MA, USA, 2005; pp. 113–132. [Google Scholar]
- Machado, C.T.D.T.; Furlani, Â.M.C. Root phosphatase activity, plant growth and phosphorus accumulation of maize genotypes. Sci. Agric. 2004, 61, 216–223. [Google Scholar] [CrossRef]
- Hedley, M.J.; Stewart, J.W.B.; Chauhan, B.C. Changes in inorganic and organic soil phosphorus fractions induce by cultivation practices and by laboratory incubation. Soil Sci. Soc. Am. J. 1982, 46, 970–976. [Google Scholar] [CrossRef]
- Cabeza, R.A.; Myint, K.; Steingrobe, B.; Stritsis, C.; Schulze, J.; Claassen, N. Phosphorus fractions depletion in the rhizosphere of young and adult maize and oilseed rape plants. J. Soil Sci. Plant Nutr. 2017, 17, 824–838. [Google Scholar] [CrossRef]
- Castillo, M.S.; Wright, A.L. Soil phosphorus pools for Histosols under sugarcane and pasture in the Everglades, USA. Geoderma 2008, 145, 130–135. [Google Scholar] [CrossRef]
- Richardson, A.E.; Hocking, P.J.; Simpson, R.J.; George, T.S. Plant mechanisms to optimise access to soil phosphorus. Crop Pasture Sci. 2009, 60, 124–143. [Google Scholar] [CrossRef]
- Manzoor, A.; Dippold, M.A.; Loeppmann, S.; Blagodatskaya, E. Two-phase conceptual framework of phosphatase activity and phosphorus bioavailability. Front. Plant Sci. 2022, 13, 935829. [Google Scholar] [CrossRef]
- Fernández, M.C.; Belinque, H.; Boem, F.G.; Rubio, G. Compared phosphorus efficiency in soybean, sunflower and maize. J. Plant Nutr. 2009, 32, 2027–2043. [Google Scholar] [CrossRef]
- Marschner, P.; Solaiman, Z.; Rengel, Z. Brassica genotypes differ in growth, phosphorus uptake and rhizosphere properties under P-limiting conditions. Soil Biol. Biochem. 2007, 39, 87–98. [Google Scholar] [CrossRef]
- Schwerdtner, U.; Lacher, U.; Spohn, M. Soy and mustard effectively mobilize phosphorus from inorganic and organic sources. Nutr. Cycl. Agroecosyst. 2022, 124, 211–226. [Google Scholar] [CrossRef]
- Urlić, B.; Dumičić, G.; Goreta Ban, S.; Romić, M. Phosphorus-use efficiency of kale genotypes from coastal Croatia. J. Plant Nutr. 2016, 39, 389–398. [Google Scholar] [CrossRef]
- Durn, G. Terra Rossa in the Mediterranean Region: Parent Materials, Composition and Origin. Geol. Croat. 2003, 56, 83–100. [Google Scholar] [CrossRef]
- FAO. World Reference Base for Soil Resources; Food and Agriculture Organization of United Nations: Rome, Italy, 2014; pp. 148–149. [Google Scholar]
- Romić, D.; Romić, M.; Zovko, M.; Bakić, H.; Ondrašek, G. Trace metals in the coastal soils developed from estuarine floodplain sediments in the Croatian Mediterranean region. Environ. Geochem. Health 2012, 34, 399–416. [Google Scholar] [CrossRef] [PubMed]
- Bolland, M.D.A.; Kumar, V.; Gilkes, R.J. A comparison of five soil phosphorus tests for five crop species for soil previously fertilized with superphosphate and rock phosphate. Fertil. Res. 1994, 37, 125–132. [Google Scholar] [CrossRef]
- Hammond, J.P.; Broadley, M.R.; White, P.J.; King, G.J.; Bowen, H.C.; Hayden, R.; Meacham, M.C.; Mead, A.; Overs, T.; Spracklen, W.P.; et al. Shoot yield drives phosphorus use efficiency in Brassica oleracea and correlates with root architecture traits. J. Exp. Bot. 2009, 60, 1953–1968. [Google Scholar] [CrossRef] [PubMed]
- Niu, Y.F.; Chai, R.S.; Jin, G.L.; Wang, H.; Tang, C.X.; Zhang, Y.S. Responses of root architecture development to low phosphorus availability: A review. Ann. Bot. 2013, 112, 391–408. [Google Scholar] [CrossRef]
- Vengavasi, K.; Pandey, R.; Soumya, P.R.; Hawkesford, M.J.; Siddique, K.H. Below-ground physiological processes enhancing phosphorus acquisition in plants. Plant Physiol. Rep. 2021, 26, 600–613. [Google Scholar] [CrossRef]
- Duan, X.; Jin, K.; Ding, G.; Wang, C.; Cai, H.; Wang, S.; White, P.J.; Xu, F.; Shi, L. The impact of different morphological and biochemical root traits on phosphorus acquisition and seed yield of Brassica napus. Field Crops Res. 2020, 258, 107960. [Google Scholar] [CrossRef]
- Robles-Aguilar, A.A.; Pang, J.; Postma, J.A.; Schrey, S.D.; Lambers, H.; Jablonowski, N.D. The effect of pH on morphological and physiological root traits of Lupinus angustifolius treated with struviteg as a recycled phosphorus source. Plant Soil 2019, 434, 65–78. [Google Scholar] [CrossRef]
- Erel, R.; Bérard, A.; Capowiez, L.; Doussan, C.; Arnal, D.; Souche, G.; Gavaland, A.; Fritz, C.; Eric, J.; Visser, W.; et al. Soil type determines how root and rhizosphere traits relate to phosphorus acquisition in field-grown maize genotypes. Plant Soil 2017, 412, 115–132. [Google Scholar] [CrossRef]
- Morrow de la Riva, L. Root Etiolation as a Strategy for Phosphorus Acquisition in Common Bean. Master’s Thesis, Pennsylvania State University, University Park, PA, USA, 2010. [Google Scholar]
- Lyu, Y.; Tang, H.; Li, H.; Zhang, F.; Rengel, Z.; Whalley, W.R.; Shen, J. Major crop species show differential balance between root morphological and physiological responses to variable phosphorus supply. Front. Plant Sci. 2016, 7, 1939. [Google Scholar] [CrossRef]
- Fageria, N.K.; Baligar, V.C.; Li, Y.C. The role of nutrient efficient plants in improving crop yields in the twenty first century. J. Plant Nutr. 2008, 31, 1121–1157. [Google Scholar] [CrossRef]
- Wang, X.; Shen, J.; Liao, H. Acquisition or utilization, which is more critical for enhancing phosphorus efficiency in modern crops? Plant Sci. 2010, 179, 302–306. [Google Scholar] [CrossRef]
- Moll, R.H.; Kamprath, E.J.; Jackson, W.A. Analysis and interpretation of factors which contribute to efficiency of nitrogen utilization. Agron. J. 1982, 74, 562–564. [Google Scholar] [CrossRef]
- Manske, G.G.B.; Ortiz-Monasterio, J.I.; van Ginkel, M.; González, R.M.; Fischer, R.A.; Rajaram, S.; Vlek, P.L.G. Importance of P uptake efficiency versus P utilization for wheat yield in acid and calcareous soils in Mexico. Eur. J. Agron. 2001, 14, 261–274. [Google Scholar] [CrossRef]
- Daoui, K.; Karrou, M.; Mrabet, R.; Fatemi, Z.; Draye, X.; Ledent, J.F. Genotypic variation of phosphorus use efficiency among Moroccan faba bean varieties (Vicia faba major) under rainfed conditions. J. Plant Nutr. 2012, 35, 34–48. [Google Scholar] [CrossRef]
- Parentoni, S.N.; Souza Júnior, C.L.D. Phosphorus acquisition and internal utilization efficiency in tropical maize genotypes. Pesqui. Agropecu. Bras. 2008, 43, 893–901. [Google Scholar] [CrossRef]
- Wang, Y.; Marschner, P.; Zhang, F. Phosphorus pools and other soil properties in the rhizosphere of wheat and legumes growing in three soils in monoculture or as a mixture of wheat and legume. Plant Soil. 2012, 354, 283–298. [Google Scholar] [CrossRef]
- Jiménez-Cárceles, F.J.; Álvarez-Rogel, J. Phosphorus fractionation and distribution in salt marsh soils affected by mine wastes and eutrophicated water: A case study in SE Spain. Geoderma 2008, 144, 299–309. [Google Scholar] [CrossRef]
- Adhami, E.; Owliaie, H.R.; Molavi, R.; Rezaei Rashti, M.; Esfandbod, M. Effects of soil properties on phosphorus fractions in subtropical soils of Iran. J. Soil Sci. Plant Nutr. 2013, 13, 11–21. [Google Scholar] [CrossRef]
- Williams, J.D.H.; Syers, J.K.; Harris, R.F.; Armstrong, D.E. Fractionation of inorganic phosphate in calcareous lake sediments. Soil Sci. Soc. Am. Proc. 1971, 35, 250–255. [Google Scholar] [CrossRef]
- Rose, T.J.; Hardiputra, B.; Rengel, Z. Wheat, canola and grain legume access to soil phosphorus fractions differs in soils with contrasting phosphorus dynamics. Plant Soil 2010, 326, 159–170. [Google Scholar] [CrossRef]
- Nuruzzaman, M.; Lambers, H.; Bolland, M.D.; Veneklaas, E.J. Distribution of carboxylates and acid phosphatase and depletion of different phosphorus fractions in the rhizosphere of a cereal and three grain legumes. Plant Soil 2006, 281, 109–120. [Google Scholar] [CrossRef]
- Vu, D.T.; Tang, C.; Armstrong, R.D. Changes and availability of P fractions following 65 years of P application to a calcareous soil in a Mediterranean climate. Plant Soil 2008, 304, 21–33. [Google Scholar] [CrossRef]
- Ruiz, J.M.; Delgado, A.; Torrent, J. Iron-related phosphorus in over fertilized European soils. J. Environ. Qual. 1997, 26, 1548–1554. [Google Scholar] [CrossRef]
- Shen, J.; Li, R.; Zhang, F.; Fan, J.; Tang, C.; Rengel, Z. Crop yields, soil fertility and phosphorus fractions in response to long-term fertilization under the rice monoculture system on a calcareous soil. Field Crops Res. 2004, 86, 225–238. [Google Scholar] [CrossRef]
- Hoffland, E. Quantitative evaluation of the role of organic acid exudation in the mobilization of rock phosphate by rape. Plant Soil 1992, 140, 279–289. [Google Scholar] [CrossRef]
- Solaiman, Z.; Marschner, P.; Wang, D.; Rengel, Z. Growth, P uptake and rhizosphere properties of wheat and canola genotypes in an alkaline soil with low P availability. Biol. Fertil. Soils 2007, 44, 143–153. [Google Scholar] [CrossRef]
- Neumann, G.; Romheld, V. Root excretion of carboxylic acids and protons in phosphorus-deficient plants. Plant Soil 1999, 211, 121–130. [Google Scholar] [CrossRef]
- Marschner, P.; Solaiman, Z.; Rengel, Z. Growth, phosphorus uptake, and rhizosphere microbial community composition of a phosphorus defficient wheat cultivar in soils differing in pH. J. Plant Nutr. Soil Sci. 2005, 168, 343–351. [Google Scholar] [CrossRef]
- Tabatabai, M.A. Soil enzymes. In Methods of Soil Analysis, Part 2—Microbiological and Biochemical Properties; Soil Science Society of America Book Series; Madison, WI, USA, 1994; pp. 775–833. [Google Scholar]
- Criquet, S.; Braud, A.; Nèble, S. Short-term effects of sewage sludge application on phosphatase activities and available P fractions in Mediterranean soils. Soil Biol. Biochem. 2007, 39, 921–929. [Google Scholar] [CrossRef]
- Turner, B.L.; Haygarth, P.M. Phosphatase activity in temperate pasture soils: Potential regulation of labile organic phosphorus turnover by phosphodiesterase activity. Sci. Total Environ. 2005, 344, 27–36. [Google Scholar] [CrossRef] [PubMed]
- Halajnia, A.; Haghnia, G.H.; Fotovat, A.; Khorasani, R. Phosphorus fractions in calcareous soils amended with P fertilizer and cattle manure. Geoderma 2009, 150, 209–213. [Google Scholar] [CrossRef]
- ISO 11466; Soil Quality—Extraction of Trace Elements Soluble in Aqua Regia. International Organi Sation for Standardization: Geneva, Switzerland, 1995.
- Jones, J.B. Laboratory Guide for Conducting Soil Tests and Plant Analysis; CRC Press: Boca Raton, FL, USA, 2001. [Google Scholar]
- Kuo, S. Phosphorus. In Methods of Soil Analysis; Sparks, D.L., Ed.; ASA-SSSA: Madison, WI, USA, 1994. [Google Scholar]
- Bowman, R.A.; Cole, C.V. An exploratory method for fractionation of organic phosphorus from grassland soils. Soil Sci. 1978, 125, 95–101. [Google Scholar] [CrossRef]
| Dry Weight (g Plant−1) | Root/Shoot Ratio | Leaf Area | Number of Leaves | P Concentration (mg g−1 DW) | P Content (mg Plant−1) | |||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Shoot | Root | cm2 Plant−1 | Plant−1 | Shoot | Root | Shoot | Root | |||||||||||
| Terra rossa | 2.00 ± 0.20 | b | 0.26 ± 0.02 | 0.14 ± 0.02 | a | 267 ± 36 | b | 8.2 ± 0.39 | b | 1.02 ± 0.08 | b | 1.25 ± 0.07 | b | 2.12 ± 0.35 | b | 0.33 ± 0.03 | b | |
| Soil | Rendzina | 1.60 ± 0.08 | b | 0.20 ± 0.02 | 0.13 ± 0.01 | a | 214 ± 20 | b | 7.7 ± 0.18 | b | 0.87 ± 0.04 | b | 1.29 ± 0.03 | b | 1.39 ± 0.10 | b | 0.26 ± 0.03 | b |
| Fluvisol | 3.45 ± 0.31 | a | 0.26 ± 0.04 | 0.07 ± 0.01 | b | 543 ± 59 | a | 10.5 ± 0.35 | a | 1.85 ± 0.11 | a | 2.77 ± 0.14 | a | 6.35 ± 0.74 | a | 0.71 ± 0.12 | a | |
| Genotype | RR | 2.10 ± 0.32 | 0.26 ± 0.02 | 0.14 ± 0.01 | a | 272 ± 41 | b | 9.0 ± 0.48 | 1.30 ± 0.13 | 1.67 ± 0.19 | 3.05 ± 0.70 | 0.44 ± 0.06 | ||||||
| IJK17 | 2.55 ± 0.26 | 0.22 ± 0.03 | 0.09 ± 0.01 | b | 410 ± 54 | a | 8.6 ± 0.39 | 1.24 ± 0.16 | 1.86 ± 0.25 | 3.52 ± 0.75 | 0.43 ± 0.10 | |||||||
| Enzyme Activity | ||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Root Length | Root Diameter | SRL | SPU | Acid Phosphatase | Alkaline Phosphatase | Phospho-Diesterase | ||||||||||
| m Plant−1 | mm | m g−1 | mg P m−1 | mg PNP kg−1 h−1 | ||||||||||||
| Terra rossa | 25.43 ± 2.2 | a | 0.38 ± 0.02 | a | 98.3 ± 7.5 | a | 0.11 ± 0.02 | b | 153 ± 9 | 51.7 ± 11.1 | b | 19.2 ± 3.3 | c | |||
| Soil | Rendzina | 17.01 ± 1.1 | b | 0.32 ± 0.01 | b | 89.8 ± 8.6 | ab | 0.10 ± 0.01 | b | 124 ± 26 | 280 ± 28.5 | a | 127 ± 9.9 | a | ||
| Fluvisol | 18.00 ± 1.9 | b | 0.34 ± 0.01 | ab | 68.3 ± 4.1 | b | 0.39 ± 0.05 | a | 82.7 ± 10 | 264 ± 13.1 | a | 87.7 ± 9.9 | b | |||
| Genotype | RR | 21.13 ± 1.7 | 0.35 ±0.02 | 81.0 ± 8.5 | 0.21 ± 0.06 | 116 ± 20 | 181 ± 31.3 | 78.9 ± 14 | ||||||||
| IJK17 | 19.16 ± 1.8 | 0.34 ±0.01 | 91.9 ± 4.7 | 0.20 ± 0.04 | 116 ± 15 | 234 ± 37.3 | 78.1 ± 15 | |||||||||
| PUpE | PUtE | PUE | |||||
|---|---|---|---|---|---|---|---|
| mg P mg−1P | g DW mg P−1 | ||||||
| Terra rossa | 0.55 ± 0.09 | b | 0.99 ± 0.08 | a | 0.51 ± 0.05 | b | |
| Soil | Rendzina | 2.03 ± 0.14 | a | 1.13 ± 0.05 | a | 2.25 ± 0.10 | a |
| Fluvisol | 0.43 ± 0.04 | b | 0.55 ± 0.04 | b | 0.23 ± 0.02 | c | |
| Genotype | RR | 0.83 ± 0.20 | 0.93 ± 0.10 | 0.77 ± 0.23 | b | ||
| IJK17 | 1.01 ± 0.22 | 0.90 ± 0.08 | 0.91 ± 0.28 | a | |||
| PUE Component | Variability in Shoot DW Production | ||
|---|---|---|---|
| Soil | |||
| Terra rossa | Rendzina | Fluvisol | |
| PUpE | 0.96 | 0.65 | 0.40 |
| PUtE | 0.04 | 0.35 | 0.60 |
| Genotype | |||
| IJK 17 | RR | ||
| PUpE | 0.72 | 0.64 | |
| PUtE | 0.28 | 0.36 | |
| Acid Phosphatase | Alkaline Phosphatase | Phospho-Diesterase | NaHCO3-Po | HCl-Po | NaOH-Po | H2SO4-Po | Total Po | |
|---|---|---|---|---|---|---|---|---|
| Acid phosphatase | −0.06 | −0.16 | −0.37 | −0.52 * | 0.41 | 0.47 * | 0.43 | |
| Alkaline phosphatase | 0.75 *** | 0.3 | 0.69 ** | −0.88 *** | −0.88 *** | −0.89 *** | ||
| Phospho- diesterase | 0.36 | 0.54 * | −0.84 *** | −0.80 *** | −0.84 *** |
| Soil | pHH2O | pHKCl | CaCO3 | CaO | TOC | Total N | Available P * | Total P # | Available K ¥ | Texture |
|---|---|---|---|---|---|---|---|---|---|---|
| g kg−1 | mg kg−1 | |||||||||
| Terra rossa | 6.70 | 5.63 | 8.0 | - | 4.8 | 0.12 | 5.0 | 833 | 79 | Clay |
| Rendzina | 8.35 | 7.31 | 616 | 193 | 17.5 | 0.32 | 0.9 | 373 | 129 | Silt loam |
| Fluvisol | 8.16 | 7.70 | 356 | 486 | 32.7 | 0.69 | 18 | 408 | 198 | Sandy loam |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Urlić, B.; Dumičić, G.; Radić, T.; Goreta Ban, S.; Romić, M. Phosphorus Use Efficiency of Leafy Brassica sp. Grown in Three Contrasting Soils: Growth, Enzyme Activity and Phosphorus Fractionation. Plants 2023, 12, 1295. https://doi.org/10.3390/plants12061295
Urlić B, Dumičić G, Radić T, Goreta Ban S, Romić M. Phosphorus Use Efficiency of Leafy Brassica sp. Grown in Three Contrasting Soils: Growth, Enzyme Activity and Phosphorus Fractionation. Plants. 2023; 12(6):1295. https://doi.org/10.3390/plants12061295
Chicago/Turabian StyleUrlić, Branimir, Gvozden Dumičić, Tomislav Radić, Smiljana Goreta Ban, and Marija Romić. 2023. "Phosphorus Use Efficiency of Leafy Brassica sp. Grown in Three Contrasting Soils: Growth, Enzyme Activity and Phosphorus Fractionation" Plants 12, no. 6: 1295. https://doi.org/10.3390/plants12061295
APA StyleUrlić, B., Dumičić, G., Radić, T., Goreta Ban, S., & Romić, M. (2023). Phosphorus Use Efficiency of Leafy Brassica sp. Grown in Three Contrasting Soils: Growth, Enzyme Activity and Phosphorus Fractionation. Plants, 12(6), 1295. https://doi.org/10.3390/plants12061295






