Polyacrylamide Hydrogel Enriched with Amber for In Vitro Plant Rooting
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Methods
2.2.1. Synthesis of PAAG
2.2.2. Modification of PAAG with Amber
2.2.3. Biotesting of Acrylamide Toxicity Using Pea and Chickpea Seeds
2.2.4. Bioassay Using Daphnia magna
2.2.5. Investigation of Hydrogel Structure and Rheological Properties
2.2.6. In Vitro Plant Rooting
3. Results
3.1. FTIR
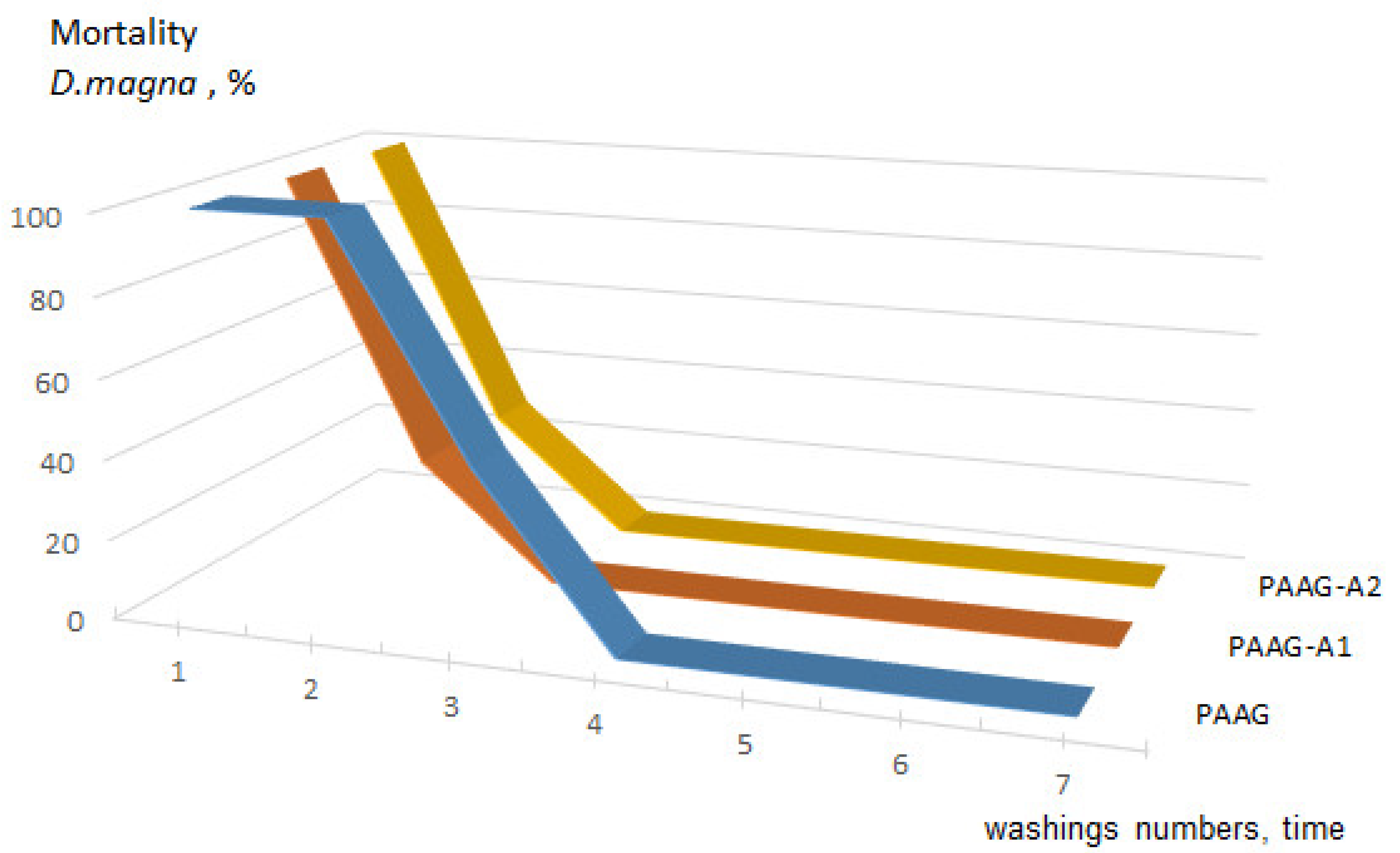
3.2. Acute Toxicity
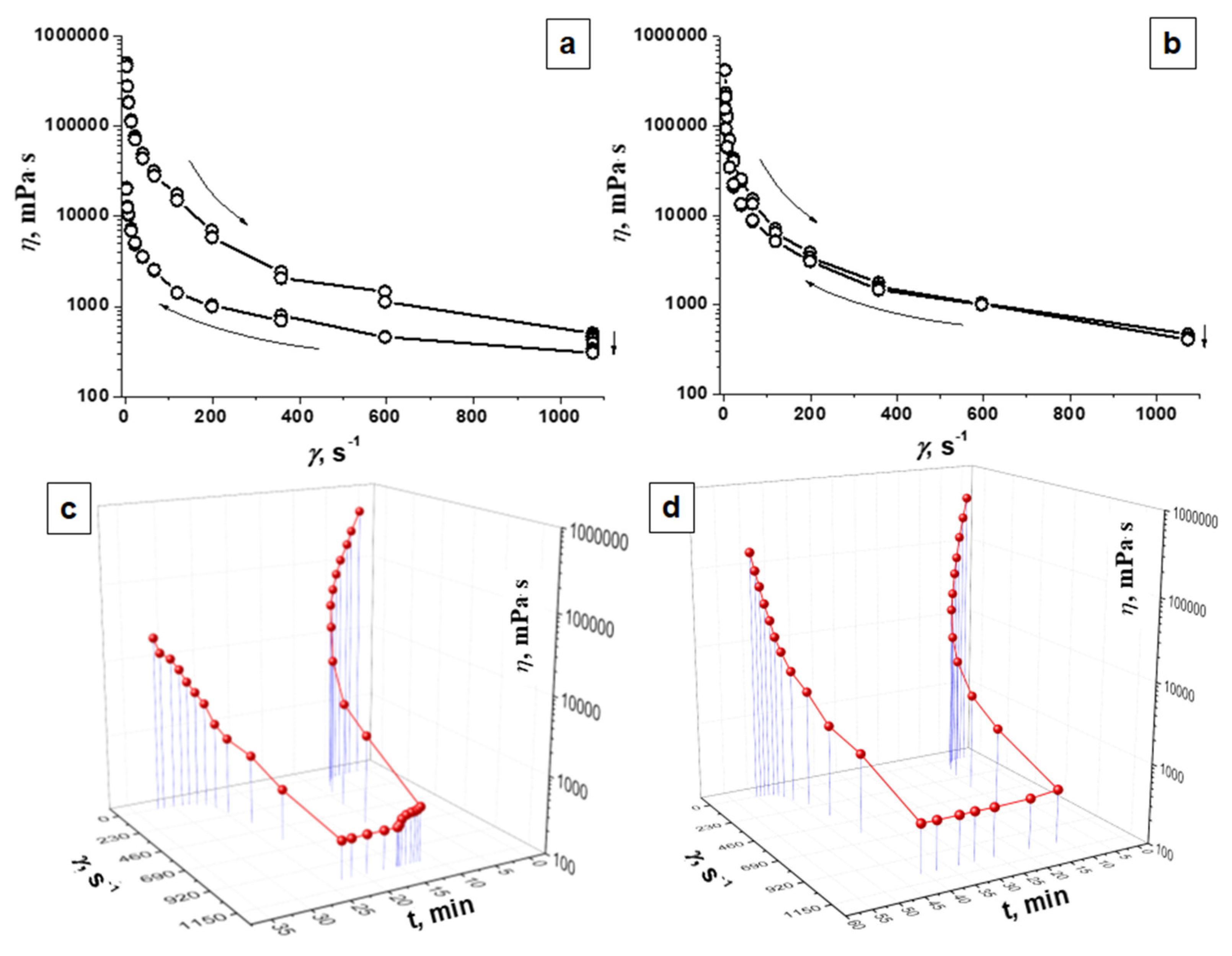
3.3. Rheological Properties
3.4. Plant Rooting
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Conflicts of Interest
References
- Kaur, J.; Mudgal, G. An efficient and quick protocol for in vitro multiplication of snake plant, Sansevieria trifasciata var. Laurentii [Prain]. Plant Cell. Tissue Organ Cult. 2021, 147, 405–411. [Google Scholar] [CrossRef]
- Kaçar, Y.A.; Biçen, B.; Varol; Mendi, Y.Y.; Serçe, S.; Çetiner, S. Gelling agents and culture vessels affect in vitro multiplication of banana plantlets. Genet. Mol. Res. 2010, 9, 416–424. [Google Scholar] [PubMed]
- Mood, K.; Jogam, P.; Sirikonda, A.; Shekhawat, M.S.; Rohela, G.K.; Manokari, M.; Allini, V.R. Micropropagation, morpho-anatomical characterization, and genetic stability studies in Lippia javanica (Burm.f.) Spreng: A multipurpose medicinal plant. Plant Cell. Tissue Organ Cult. 2022, 150, 427–437. [Google Scholar] [CrossRef]
- Lubell-Brand, J.D.; Kurtz, L.E.; Brand, M.H. An In Vitro–Ex Vitro Micropropagation System for Hemp. Horttechnology 2021, 31, 199–207. [Google Scholar] [CrossRef]
- Adhikary, D.; Kulkarni, M.; El-Mezawy, A.; Mobini, S.; Elhiti, M.; Gjuric, R.; Ray, A.; Polowick, P.; Slaski, J.J.; Jones, M.P.; et al. Medical Cannabis and Industrial Hemp Tissue Culture: Present Status and Future Potential. Front. Plant Sci. 2021, 12, 275. [Google Scholar] [CrossRef] [PubMed]
- Hasnain, A.; Naqvi, S.A.H.; Ayesha, S.I.; Khalid, F.; Ellahi, M.; Iqbal, S.; Hassan, M.Z.; Abbas, A.; Adamski, R.; Markowska, D.; et al. Plants in vitro propagation with its applications in food, pharmaceuticals and cosmetic industries; current scenario and future approaches. Front. Plant Sci. 2022, 13, 3894. [Google Scholar] [CrossRef]
- Hesami, M.; Pepe, M.; Monthony, A.S.; Baiton, A.; Phineas Jones, A.M. Modeling and optimizing in vitro seed germination of industrial hemp (Cannabis sativa L.). Ind. Crops Prod. 2021, 170, 113753. [Google Scholar] [CrossRef]
- Stephen, C.; Zayas, V.A.; Galic, A.; Bridgen, M.P. Micropropagation of Hemp (Cannabis sativa L.). HortScience 2023, 58, 307–316. [Google Scholar] [CrossRef]
- Smýkalová, I.; Vrbová, M.; Cvečková, M.; Plačková, L.; Žukauskaitė, A.; Zatloukal, M.; Hrdlička, J.; Plíhalová, L.; Doležal, K.; Griga, M. The effects of novel synthetic cytokinin derivatives and endogenous cytokinins on the in vitro growth responses of hemp (Cannabis sativa L.) explants. Plant Cell. Tissue Organ Cult. 2019, 139, 381–394. [Google Scholar] [CrossRef]
- Espinosa-Leal, C.A.; Puente-Garza, C.A.; García-Lara, S. In vitro plant tissue culture: Means for production of biological active compounds. Planta 2018, 248, 1–18. [Google Scholar] [CrossRef]
- Henderson, W.E.; Kinnersley, A.M. Corn starch as an alternative gelling agent for plant tissue culture. Plant Cell Tissue Organ Cult. 1988 151 1988, 15, 17–22. [Google Scholar] [CrossRef]
- Jain, N.; Babbar, S.B. Gum katira—A cheap gelling agent for plant tissue culture media. Plant Cell Tissue Organ Cult. 2002, 71, 223–229. [Google Scholar] [CrossRef]
- Goncharuk, O.; Samchenko, Y.; Kernosenko, L.; Korotych, O.; Poltoratska, T.; Pasmurtseva, N.; Oranska, O.; Sternik, D.; Mamyshev, I. Thermoresponsive hydrogels physically crosslinked with magnetically modified LAPONITE® nanoparticles. Soft Matter 2020, 16, 5689–5701. [Google Scholar] [CrossRef]
- Kocak, G.; Tuncer, C.; Bütün, V. pH-Responsive polymers. Polym. Chem. 2016, 8, 144–176. [Google Scholar] [CrossRef]
- Schmaljohann, D. Thermo- and pH-responsive polymers in drug delivery. Adv. Drug Deliv. Rev. 2006, 58, 1655–1670. [Google Scholar] [CrossRef] [PubMed]
- Samchenko, Y.; Korotych, O.; Kernosenko, L.; Kryklia, S.; Litsis, O.; Skoryk, M.; Poltoratska, T.; Pasmurtseva, N. Stimuli-responsive hybrid porous polymers based on acetals of polyvinyl alcohol and acrylic hydrogels. Colloids Surf. A Physicochem. Eng. Asp. 2018, 544, 91–104. [Google Scholar] [CrossRef]
- Sinha, V.; Chakma, S. Advances in the preparation of hydrogel for wastewater treatment: A concise review. J. Environ. Chem. Eng. 2019, 7, 103295. [Google Scholar] [CrossRef]
- Stadniy, I.A.; Konovalova, V.V.; Samchenko, Y.M.; Pobigay, G.A.; Burban, A.F.; Ulberg, Z.R.; Stadniy, I.A.; Konovalova, V.V.; Samchenko, Y.M.; Pobigay, G.A.; et al. Development of Hydrogel Polyelectrolyte Membranes with Fixed Sulpho-Groups via Radical Copolymerization of Acrylic Monomers. Mater. Sci. Appl. 2011, 2, 270–275. [Google Scholar] [CrossRef]
- Kosenko, O.O.; Lukash, L.L.; Samchenko, Y.M.; Ruban, T.A.; Ulberg, Z.R.; Lukash, S.I. Copolymeric hydrogel membranes for immobilization and cultivation of human stem cells. Biopolym. Cell 2006, 22, 143–148. [Google Scholar] [CrossRef]
- Milani, P.; França, D.; Balieiro, A.G.; Faez, R. Polymers and its applications in agriculture. Polimeros 2017, 27, 256–266. [Google Scholar] [CrossRef]
- Ramli, R.A. Slow release fertilizer hydrogels: A review. Polym. Chem. 2019, 10, 6073–6090. [Google Scholar] [CrossRef]
- Singh, N.; Agarwal, S.; Jain, A.; Khan, S. 3-Dimensional cross linked hydrophilic polymeric network “hydrogels”: An agriculture boom. Agric. Water Manag. 2021, 253, 106939. [Google Scholar] [CrossRef]
- Khan, F.; Atif, M.; Haseen, M.; Kamal, S.; Khan, M.S.; Shahid, S.; Nami, S.A.A. Synthesis, classification and properties of hydrogels: Their applications in drug delivery and agriculture. J. Mater. Chem. B 2022, 10, 170–203. [Google Scholar] [CrossRef] [PubMed]
- Levchyck, N.Y.; Liubinska, A.V.; Skrypchenko, N.V.; Dziuba, O.I.; Kachalova, N.M.; Mironov, O.L. Biological activity of aqueous solution of amber. Biotechnol. Acta 2017, 10, 53–60. [Google Scholar] [CrossRef]
- Mironov, O.L.; Kachalova, N.M.; Dzyuba, O.I.; Levchyk, N.Y.; Bogza, S.L. Research of biological activity of Ukrainian amber. In Physico-Organic Chemistry, Pharmacology and Pharmaceutical Technology of Biologically Active Substances: Coll, Science, Work; Popov, A., Ed.; KNUTD: Kyiv, Ukraine, 2018; pp. 100–120. [Google Scholar]
- Tumiłowicz, P.; Synoradzki, L.; Sobiecka, A.; Arct, J.; Pytkowska, K.; Safarzyński, S. Bioactivity of Baltic amber—Fossil resin. Polimery 2016, 61, 347–356. [Google Scholar] [CrossRef]
- Shimizu, E.; Shimoda, N.; Kawamura, T.; Ueda, N.; Kimura, K.; Shimizu, E.; Shimoda, N.; Kawamura, T.; Ueda, N.; Kimura, K. Comparison of the Biological Activity and Constituents in Japanese Ambers. Adv. Biol. Chem. 2020, 10, 99–112. [Google Scholar] [CrossRef]
- Kim, H.J.; Koedrith, P.; Seo, Y.R. Ecotoxicogenomic Approaches for Understanding Molecular Mechanisms of Environmental Chemical Toxicity Using Aquatic Invertebrate, Daphnia Model Organism. Int. J. Mol. Sci. 2015, 16, 12261. [Google Scholar] [CrossRef]
- Olkova, A.; Zimonina, N. Assessment of the Toxicity of the Natural and Technogenic Environment for Motor Activity of Daphnia magna. J. Ecol. Eng. 2020, 21, 11–16. [Google Scholar] [CrossRef]
- Test No. 202: Daphnia sp. Acute Immobilisation Test. Available online: https://www.oecd-ilibrary.org/environment/test-no-202-daphnia-sp-acute-immobilisation-test_9789264069947-en (accessed on 12 August 2004).
- Rand, G.M.; Wells, P.G.; McCarty, L.S. Introduction to Aquatic Toxicology. In Fundamentals of Aquatic Toxicology; CRC Press: Boca Raton, FL, USA, 2020; pp. 3–67. [Google Scholar]
- Gertsiuk, M.; Samchenko, Y.M. Separation of nonreacted acrylamide from polyacrylamide gel for endoprothesing. Ars Separatoria Acta 2007, 5, 98–101. [Google Scholar]
- Seeds of Farm Crops. Methods for Determination of Viability; USSR State Standard: Moscow, Russia, 1982.
- ISO—ISO 10706:2000; Water Quality—Determination of Long Term Toxicity of Substances to Daphnia magna Straus (Cladocera, Crustacea). ISO Standards: Geneva, Switzerland. Available online: https://www.iso.org/standard/18795.html (accessed on 12 August 2022).
- Samchenko, Y.M.; Kernosenko, L.O.; Dziuba, O.I.; Mironov, O.L.; Kachalova, N.M. Method for Obtaining an Artificial Dense Nutrient Medium for Plant Growing. Patent No. 151754, 28 March 2022. [Google Scholar]
- Grodzinsky, A.M.; Golovko, E.A.; Gorobets, S.A. Experimental Allelopathy; Naukova Dumka: Kyiv, Ukraine, 1987. [Google Scholar]
- Bataineh, S.M.; Saadoun, I.; Hameed, K.M.; Ababneh, Q. Screening for soil streptomycetes from North Jordan that can produce herbicidal compounds. Polish J. Microbiol. 2008, 57, 297–305. [Google Scholar]
- Nakanishi, K. Infrared Absorption Spectroscopy, Practical; Nankodo: Tokyo, Japan; Holden-Day: San Francisco, CA, USA, 1962. [Google Scholar]
- Mänd, K.; Muehlenbachs, K.; McKellar, R.C.; Wolfe, A.P.; Konhauser, K.O. Distinct origins for Rovno and Baltic ambers: Evidence from carbon and hydrogen stable isotopes. Palaeogeogr. Palaeoclimatol. Palaeoecol. 2018, 505, 265–273. [Google Scholar] [CrossRef]
- Karolina, D.; Maja, M.S.; Magdalena, D.S.; Grażyna, Ż. Identification of treated Baltic amber by FTIR and FT-Raman—A feasibility study. Spectrochim. Acta. A. Mol. Biomol. Spectrosc. 2022, 279, 121404. [Google Scholar] [CrossRef] [PubMed]
- Boldeskul, I.E.; Samchenko, Y.M.; Sukhodub, L.B.; Ulberg, Z.R.; Sukhodub, L.F.; Bereza, L.I. Application of vibrational spectroscopy in express analysis of acrylic hydrogel and hydroxyapatite based composite materials. Methods Objects Chem. Anal. 2009, 4, 92–96. [Google Scholar]
- Tuson, H.H.; Renner, L.D.; Weibel, D.B. Polyacrylamide hydrogels as substrates for studying bacteria. Chem. Commun. 2012, 48, 1595–1597. [Google Scholar] [CrossRef] [PubMed]




| Sample | Water, g | AA, g | MBA, g | Amber, % |
|---|---|---|---|---|
| PAAG | 13.6 | 32 | 0.04 | - |
| PAAG-A1 | 11.1 | 32 | 0.04 | 5.8 |
| PAAG-A2 | 11.6 | 32 | 0.04 | 5.7 |
| Washing/AA Solution | Conc. AA, mmol/L | Death Toll, % | |||||
|---|---|---|---|---|---|---|---|
| PAAG | PAAG-A1 | PAAG-A2 | |||||
| Daphnia | Peas/Chickpeas | Daphnia | Peas/Chickpeas | Daphnia | Peas/Chickpeas | ||
| AA solution | 1.4 | 100 * | 80/80 | 100 * | 80/80 | 100 * | 80/90 |
| AA solution | 0.14 | 100 * | 40/70 | 100 * | 40/80 | 100 * | 50/70 |
| AA solution | 0.014 | 100 * | 40/70 | 100 * | 40/80 | 100 * | 50/70 |
| 1 | 0.00125 | 100 * | 10/20 | 100 * | 10/20 | 100 * | 10/20 |
| 2 | 0.00096 | 100 ** | 10 | 28.6 ** | 10 | 28.6 ** | 10 |
| 3 | 0.00054 | 42.8 ** | 10 | 0 | 10 | 0 | 10 |
| 4 | 0.00007 | 0 | 0 | 0 | 0 | 0 | 0 |
| 5 | 0.00003 | 0 | 0 | 0 | 0 | 0 | 0 |
| 6 | 0.00002 | 0 | 0 | 0 | 0 | 0 | 0 |
| 7 | 0.00001 | 0 | 0 | 0 | 0 | 0 | 0 |
| Control (DV) | 0 | 0 | 0 | 0 | 0 | 0 | |
| Sample | η (at γ = 2.45 s−1), Pа∙s | η (at γ = 1073 s−1), Pа∙s | ||
|---|---|---|---|---|
| Initial | Final | Initial | Final | |
| Agar | 498.519 | 200.32 | 0.501 | 0.306 |
| PAAG | 422.986 | 156.398 | 0.471 | 0.410 |
| Experiment Techniques. | Cánnabis satíva Seedlings | Root Length (cm) | Stem Length (cm) | Root Weight (g) | Stem Weight (g) | Root and Stem Lengths (cm) | Root and Stem Weight (g) |
|---|---|---|---|---|---|---|---|
| In vitro | Agar | 3.5 ± 0.2 | 7.5 ± 0.4 | 0.3 ± 0.05 | 0.6 ± 0.02 | 11 ± 0.5 | 0.9 ± 0.07 |
| PAAG | 4 ± 0.3 | 8 ± 0.6 | 0.6 ± 0.03 | 0.7 ± 0.02 | 12 ± 0.6 | 1.3 ± 0.05 | |
| PAAG-A2 | 4.5 ± 0.3 | 9.5 ± 0.6 | 0.8 ± 0.07 | 1 ± 0.05 | 14 ± 0.7 | 1.8 ± 0.06 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kernosenko, L.; Samchenko, K.; Goncharuk, O.; Pasmurtseva, N.; Poltoratska, T.; Siryk, O.; Dziuba, O.; Mironov, O.; Szewczuk-Karpisz, K. Polyacrylamide Hydrogel Enriched with Amber for In Vitro Plant Rooting. Plants 2023, 12, 1196. https://doi.org/10.3390/plants12051196
Kernosenko L, Samchenko K, Goncharuk O, Pasmurtseva N, Poltoratska T, Siryk O, Dziuba O, Mironov O, Szewczuk-Karpisz K. Polyacrylamide Hydrogel Enriched with Amber for In Vitro Plant Rooting. Plants. 2023; 12(5):1196. https://doi.org/10.3390/plants12051196
Chicago/Turabian StyleKernosenko, Lyudmyla, Kateryna Samchenko, Olena Goncharuk, Natalya Pasmurtseva, Tetiana Poltoratska, Olena Siryk, Oksana Dziuba, Oleg Mironov, and Katarzyna Szewczuk-Karpisz. 2023. "Polyacrylamide Hydrogel Enriched with Amber for In Vitro Plant Rooting" Plants 12, no. 5: 1196. https://doi.org/10.3390/plants12051196
APA StyleKernosenko, L., Samchenko, K., Goncharuk, O., Pasmurtseva, N., Poltoratska, T., Siryk, O., Dziuba, O., Mironov, O., & Szewczuk-Karpisz, K. (2023). Polyacrylamide Hydrogel Enriched with Amber for In Vitro Plant Rooting. Plants, 12(5), 1196. https://doi.org/10.3390/plants12051196







