Extracts from Chlorella vulgaris Protect Mesenchymal Stromal Cells from Oxidative Stress Induced by Hydrogen Peroxide
Abstract
1. Introduction
2. Results
2.1. Biomass Profile of the Commercially Available and Laboratory Cultivated C. vulgaris
2.2. Bioactive Compound Recovery and Antioxidant Activity Measurement of the C. vulgaris Extracts
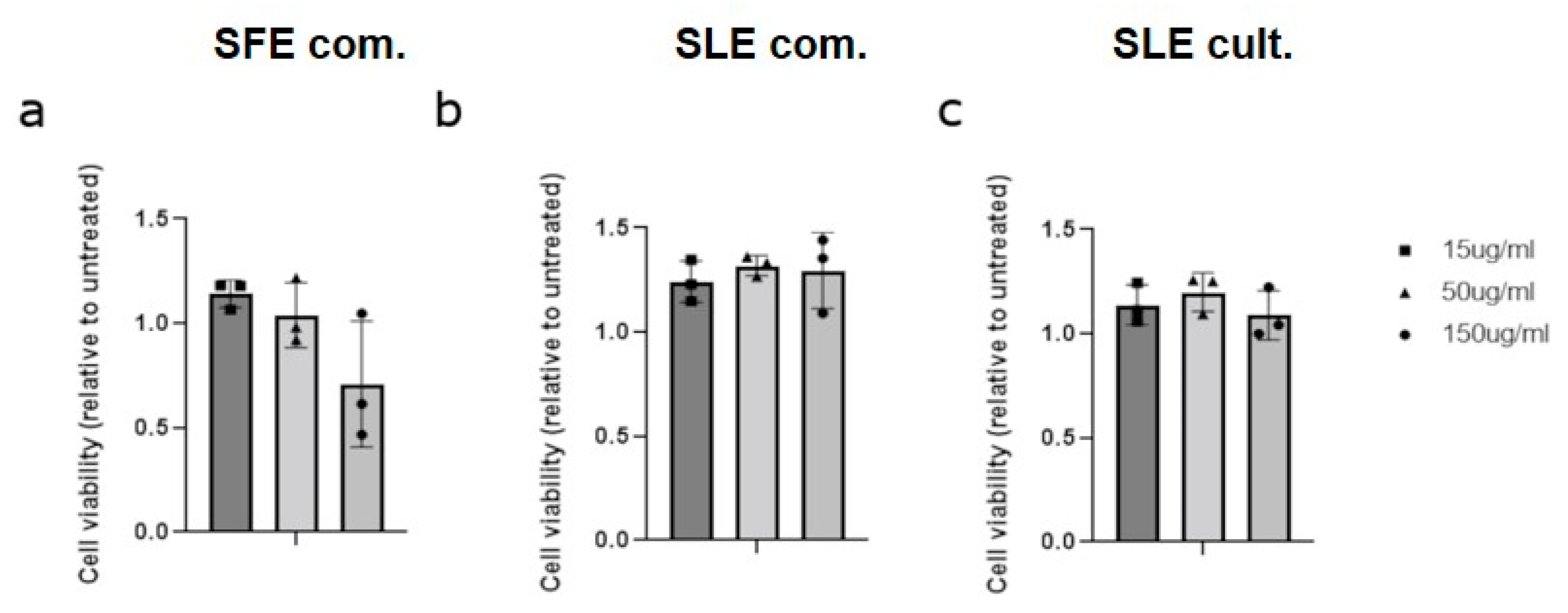
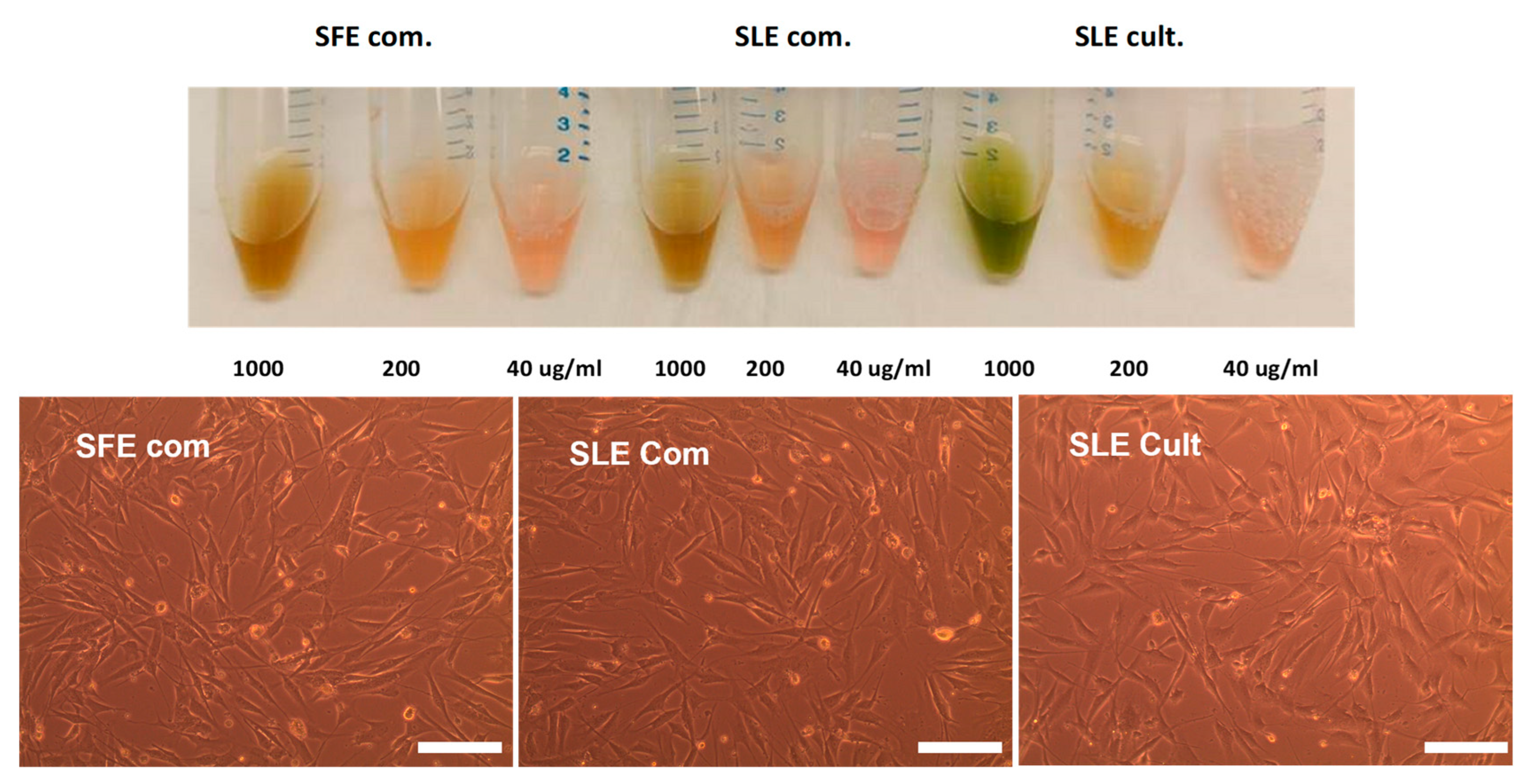
2.3. Effect of C. vulgaris Extracts Obtained by Different Extraction Methods on Cell Viability
2.4. C. vulgaris Extracts Mediate the Inhibition of H2O2-Induced ROS Generation in MSCs
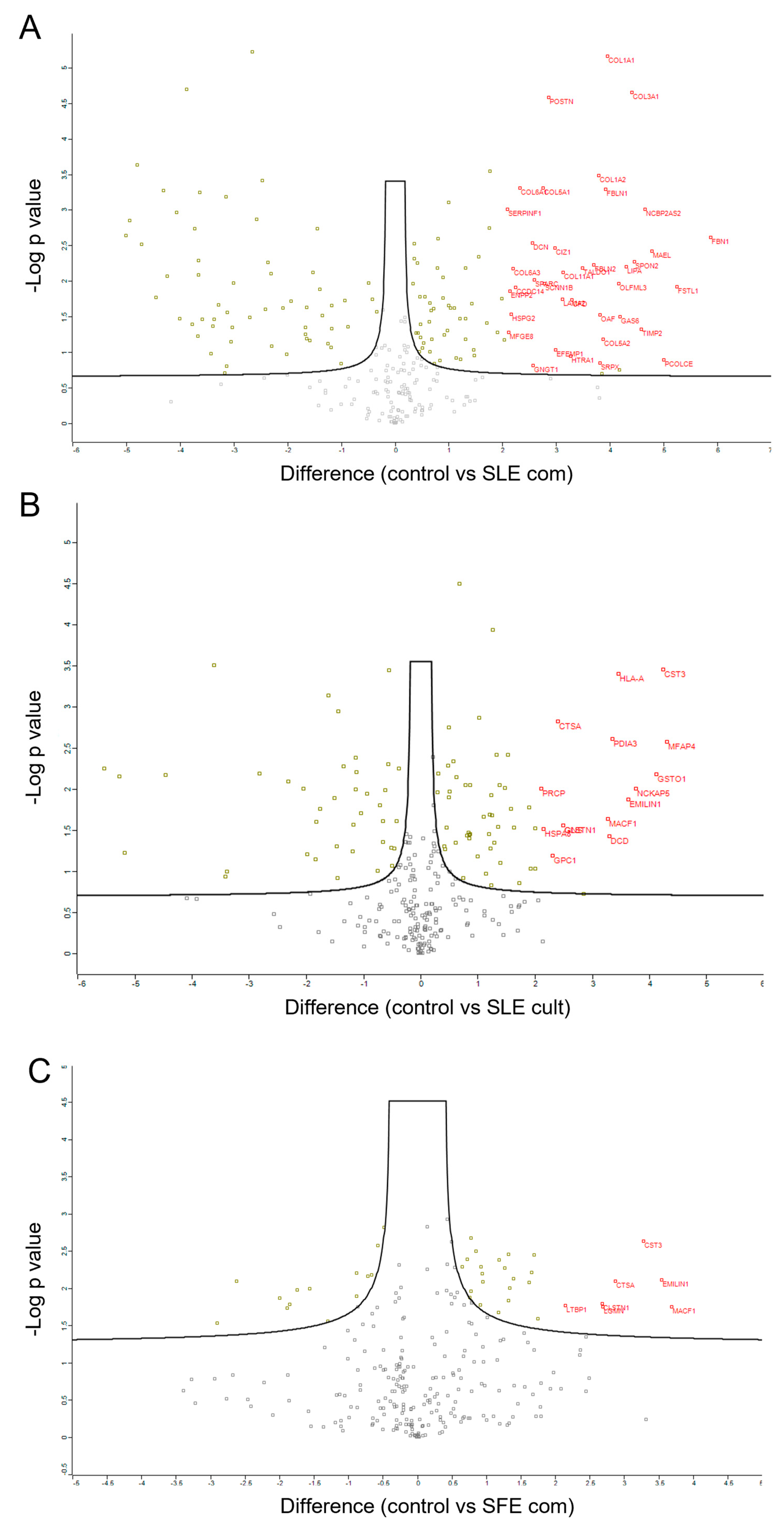
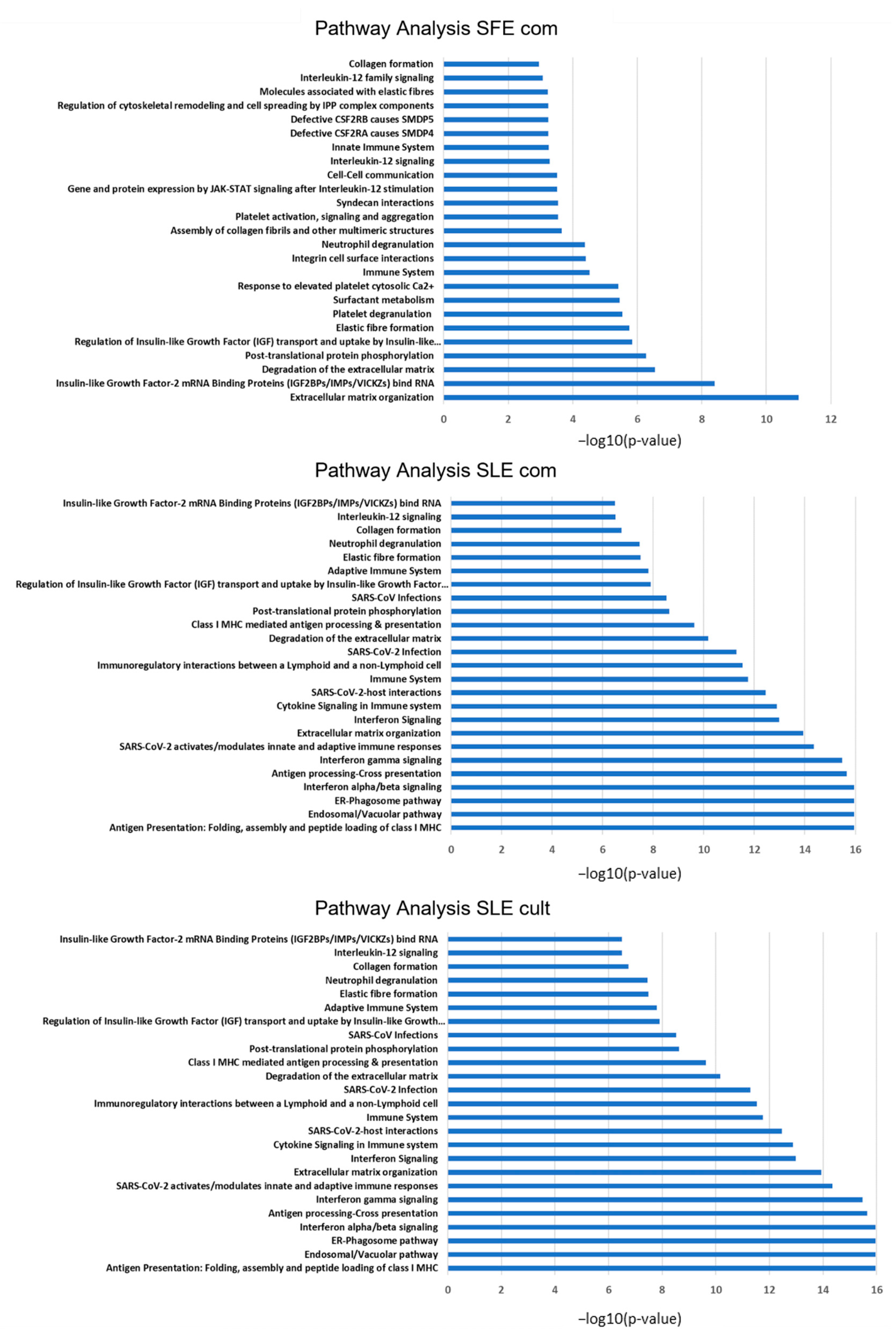
2.5. Proteomic Analysis of MSC Secretomes after Pre-Incubation with Algal Extracts
3. Discussion
4. Materials and Methods
4.1. Materials/Chemicals
4.2. Instrumentation
4.3. Methods
4.3.1. Cell Culture and Reagents
4.3.2. Biomass Characterization
4.3.3. Biomass Extraction
Supercritical Fluid Extraction (SFE)
Solid-Liquid Extraction (SLE)
4.3.4. Extract Characterization
4.3.5. Solubility of C. vulgaris Extracts Obtained by Different Extraction Methods
4.3.6. Cell Viability Assay
4.3.7. Measurement of Intracellular Reactive Oxygen Species
4.3.8. Proteomics Analysis
4.3.9. Statistical Analysis
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
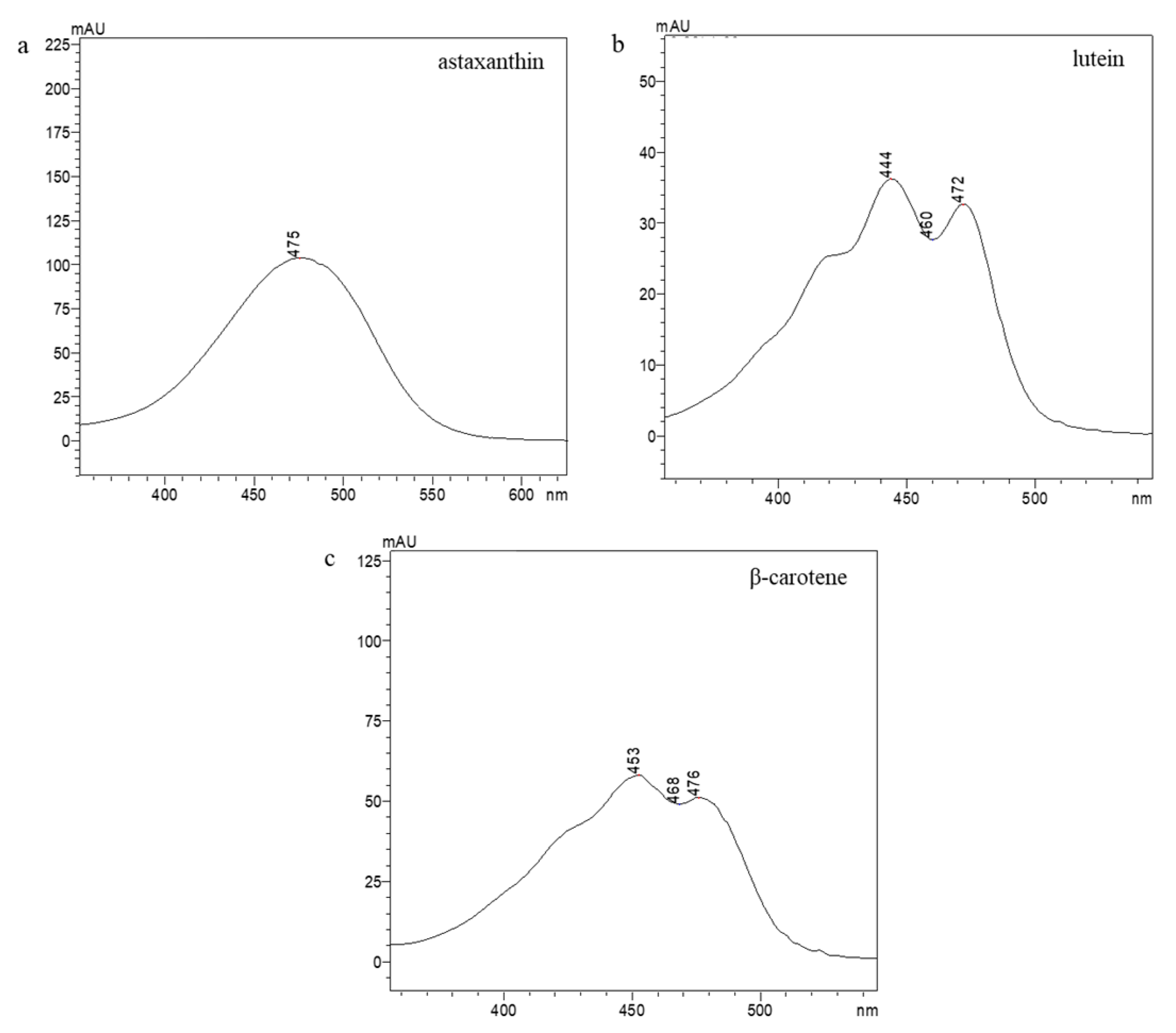
Appendix A. Identification of the Selected Carotenoids through RP-HPLC
Appendix B. Operational Conditions of SLE and SFE
| Parameter | SLE cult. SLE com. [25] | SFE com. [26] |
|---|---|---|
| Solvent | Ethanol 90% v/v | CO2 |
| Biomass | Laboratory cultivated/ Commercially available | Commercially available |
| Solvent-to-biomass (kg/kgbiom) | 30 | 100 |
| Stirring (rpm) | 500 | - |
| Duration (h) | 24 | 3.3 |
| Temperature (°C) | 30 | 60 |
| Pressure (bar) | 1 | 250 |
| Solvent flow rate (g/min) | - | 40 |
Appendix C. Supplementary Data of Total Chlorophyll and Carotenoid Determination
References
- Borowitzka, M.A. Microalgae in Medicine and Human Health: A Historical Perspective. In Microalgae in Health and Disease Prevention; Elsevier: Amsterdam, The Netherlands, 2018; pp. 195–210. [Google Scholar]
- Koyande, A.K.; Chew, K.W.; Rambabu, K.; Tao, Y.; Chu, D.-T.; Show, P.-L. Microalgae: A potential alternative to health supplementation for humans. Food Sci. Hum. Wellness 2019, 8, 16–24. [Google Scholar] [CrossRef]
- Savvidou, M.G.; Boli, E.; Logothetis, D.; Lymperopoulou, T.; Ferraro, A.; Louli, V.; Mamma, D.; Kekos, D.; Magoulas, K.; Kolisis, F.N. A study on the effect of macro-and micro-nutrients on Nannochloropsis oceanica growth, fatty acid composition and magnetic harvesting efficiency. Plants 2020, 9, 660. [Google Scholar] [CrossRef] [PubMed]
- Savvidou, M.G.; Lymperopoulou, T.V.; Mamma, D.; Balta-Brouma, K.P.; Sotiroudis, T.G.; Kekos, D.; Kolisis, F.N. A study on the combined effects of carbon and nitrogen source on high added value products synthesis by Nannochloropsis oceanica CCMP1779 using response surface methodology. Biocatal. Agric. Biotechnol. 2017, 10, 298–307. [Google Scholar] [CrossRef]
- Savvidou, M.; Banis, G.; Ferraro, A.; Molino, A.; Karatza, D.; Chianese, S.; Musmarra, D.; Kolisis, F.; Hristoforou, E. Production of magnetic modified microalgae using iron oxide nanoparticles and electroporation technique. Chem. Eng. Trans. 2019, 73, 13–18. [Google Scholar]
- Savvidou, M.G.; Ferraro, A.; Hristoforou, E.; Mamma, D.; Kekos, D.; Kolisis, F.N. Incorporation of Magnetic Nanoparticles into Protoplasts of Microalgae Haematococcus pluvialis: A Tool for Biotechnological Applications. Molecules 2020, 25, 5068. [Google Scholar] [CrossRef] [PubMed]
- Savvidou, M.G.; Ferraro, A.; Schinas, P.; Mamma, D.; Kekos, D.; Hristoforou, E.; Kolisis, F.N. Magnetic Immobilization and Growth of Nannochloropsis oceanica and Scenedasmus almeriensis. Plants 2021, 11, 72. [Google Scholar] [CrossRef]
- Boli, E.; Savvidou, M.; Logothetis, D.; Louli, V.; Pappa, G.; Voutsas, E.; Kolisis, F.; Magoulas, K. Magnetic harvesting of marine algae Nannochloropsis oceanica. Sep. Sci. Technol. 2021, 56, 730–737. [Google Scholar] [CrossRef]
- Savvidou, M.G.; Dardavila, M.M.; Georgiopoulou, I.; Louli, V.; Stamatis, H.; Kekos, D.; Voutsas, E. Optimization of microalga Chlorella vulgaris magnetic harvesting. Nanomaterials 2021, 11, 1614. [Google Scholar] [CrossRef]
- Kong, W.; Liu, N.; Zhang, J.; Yang, Q.; Hua, S.; Song, H.; Xia, C. Optimization of ultrasound-assisted extraction parameters of chlorophyll from Chlorella vulgaris residue after lipid separation using response surface methodology. J. Food Sci. Technol. 2014, 51, 2006–2013. [Google Scholar] [CrossRef] [PubMed]
- Milani, A.; Basirnejad, M.; Shahbazi, S.; Bolhassani, A. Carotenoids: Biochemistry, pharmacology and treatment. Br. J. Pharmacol. 2017, 174, 1290–1324. [Google Scholar] [CrossRef] [PubMed]
- Lakey-Beitia, J.; Kumar, D.J.; Hegde, M.L.; Rao, K.S. Carotenoids as novel therapeutic molecules against neurodegenerative disorders: Chemistry and molecular docking analysis. Int. J. Mol. Sci. 2019, 20, 5553. [Google Scholar] [CrossRef]
- Khavari, F.; Saidijam, M.; Taheri, M.; Nouri, F. Microalgae: Therapeutic potentials and applications. Mol. Biol. Rep. 2021, 48, 4757–4765. [Google Scholar] [CrossRef]
- Zuluaga, M.; Gueguen, V.; Pavon-Djavid, G.; Letourneur, D. Carotenoids from microalgae to block oxidative stress. BioImpacts BI 2017, 7, 1–3. [Google Scholar] [CrossRef]
- Coulombier, N.; Jauffrais, T.; Lebouvier, N. Antioxidant compounds from microalgae: A review. Mar. Drugs 2021, 19, 549. [Google Scholar] [CrossRef] [PubMed]
- Ru, I.T.K.; Sung, Y.Y.; Jusoh, M.; Wahid, M.E.A.; Nagappan, T. Chlorella vulgaris: A perspective on its potential for combining high biomass with high value bioproducts. Appl. Phycol. 2020, 1, 2–11. [Google Scholar] [CrossRef]
- Khan, M.I.; Shin, J.H.; Kim, J.D. The promising future of microalgae: Current status, challenges, and optimization of a sustainable and renewable industry for biofuels, feed, and other products. Microb. Cell Factories 2018, 17, 36. [Google Scholar] [CrossRef]
- El-Fayoumy, E.A.; Shanab, S.M.; Gaballa, H.S.; Tantawy, M.A.; Shalaby, E.A. Evaluation of antioxidant and anticancer activity of crude extract and different fractions of Chlorella vulgaris axenic culture grown under various concentrations of copper ions. BMC Complement. Med. Ther. 2021, 21, 51. [Google Scholar] [CrossRef] [PubMed]
- Abdel-Karim, O.H.; Gheda, S.F.; Ismail, G.A.; Abo-Shady, A.M. Phytochemical Screening and antioxidant activity of Chlorella vulgaris. Delta J. Sci. 2020, 41, 81–91. [Google Scholar] [CrossRef]
- Shanab, S.M.; Mostafa, S.S.; Shalaby, E.A.; Mahmoud, G.I. Aqueous extracts of microalgae exhibit antioxidant and anticancer activities. Asian Pac. J. Trop. Biomed. 2012, 2, 608–615. [Google Scholar] [CrossRef]
- Yu, M.; Chen, M.; Gui, J.; Huang, S.; Liu, Y.; Shentu, H.; He, J.; Fang, Z.; Wang, W.; Zhang, Y. Preparation of Chlorella vulgaris polysaccharides and their antioxidant activity in vitro and in vivo. Int. J. Biol. Macromol. 2019, 137, 139–150. [Google Scholar] [CrossRef]
- Hu, C.; Zhao, L.; Peng, C.; Li, L. Regulation of the mitochondrial reactive oxygen species: Strategies to control mesenchymal stem cell fates ex vivo and in vivo. J. Cell. Mol. Med. 2018, 22, 5196–5207. [Google Scholar] [CrossRef] [PubMed]
- Atashi, F.; Modarressi, A.; Pepper, M.S. The role of reactive oxygen species in mesenchymal stem cell adipogenic and osteogenic differentiation: A review. Stem Cells Dev. 2015, 24, 1150–1163. [Google Scholar] [CrossRef] [PubMed]
- Stavely, R.; Nurgali, K. The emerging antioxidant paradigm of mesenchymal stem cell therapy. Stem Cells Transl. Med. 2020, 9, 985–1006. [Google Scholar] [CrossRef] [PubMed]
- Georgiopoulou, I.; Tzima, S.; Pappa, G.D.; Louli, V.; Voutsas, E.; Magoulas, K. Experimental Design and Optimization of Recovering Bioactive Compounds from Chlorella vulgaris through Conventional Extraction. Molecules 2021, 27, 29. [Google Scholar] [CrossRef]
- Georgiopoulou, I.; Tzima, S.; Louli, V.; Magoulas, K. Supercritical CO2 Extraction of High-Added Value Compounds from Chlorella vulgaris: Experimental Design, Modelling and Optimization. Molecules 2022, 27, 5884. [Google Scholar] [CrossRef]
- González-González, A.; García-Sánchez, D.; Dotta, M.; Rodríguez-Rey, J.C.; Pérez-Campo, F.M. Mesenchymal stem cells secretome: The cornerstone of cell-free regenerative medicine. World J. Stem Cells 2020, 12, 1529. [Google Scholar] [CrossRef]
- Firuzi, O.; Miri, R.; Tavakkoli, M.; Saso, L. Antioxidant therapy: Current status and future prospects. Curr. Med. Chem. 2011, 18, 3871–3888. [Google Scholar] [CrossRef]
- Xia, D.; Qiu, W.; Wang, X.; Liu, J. Recent advancements and future perspectives of microalgae-derived pharmaceuticals. Mar. Drugs 2021, 19, 703. [Google Scholar] [CrossRef]
- Araujo, G.S.; Matos, L.J.; Fernandes, J.O.; Cartaxo, S.J.; Gonçalves, L.R.; Fernandes, F.A.; Farias, W.R. Extraction of lipids from microalgae by ultrasound application: Prospection of the optimal extraction method. Ultrason. Sonochem. 2013, 20, 95–98. [Google Scholar] [CrossRef]
- Agrawal, A.; Chakraborty, S. A kinetic study of pyrolysis and combustion of microalgae Chlorella vulgaris using thermo-gravimetric analysis. Bioresour. Technol. 2013, 128, 72–80. [Google Scholar] [CrossRef]
- Safi, C.; Zebib, B.; Merah, O.; Pontalier, P.-Y.; Vaca-Garcia, C. Morphology, composition, production, processing and applications of Chlorella vulgaris: A review. Renew. Sustain. Energy Rev. 2014, 35, 265–278. [Google Scholar] [CrossRef]
- Tokuşoglu, Ö.; Üunal, M.K. Biomass nutrient profiles of three microalgae: Spirulina platensis, Chlorella vulgaris, and Isochrisis galbana. J. Food Sci. 2003, 68, 1144–1148. [Google Scholar] [CrossRef]
- Wan Mahmood, W.M.A.; Lorwirachsutee, A.; Theodoropoulos, C.; Gonzalez-Miquel, M. Polyol-based deep eutectic solvents for extraction of natural polyphenolic antioxidants from Chlorella vulgaris. ACS Sustain. Chem. Eng. 2019, 7, 5018–5026. [Google Scholar] [CrossRef]
- Zainan, N.H.; Srivatsa, S.C.; Li, F.; Bhattacharya, S. Quality of bio-oil from catalytic pyrolysis of microalgae Chlorella vulgaris. Fuel 2018, 223, 12–19. [Google Scholar] [CrossRef]
- Pavia, D.L.; Lampman, G.M.; Kriz, G.S.; Engel, R.G. Isolation of chlorophyll and carotenoid pigments from spinach. Introd. Org. Lab. Tech. Microscale Approach 1999, 1–7. [Google Scholar]
- Vidussi, F.; Claustre, H.; Bustillos-Guzmàn, J.; Cailliau, C.; Marty, J.-C. Determination of chlorophylls and carotenoids of marine phytoplankton: Separation of chlorophyll a from divinylchlorophyll a and zeaxanthin from lutein. J. Plankton Res. 1996, 18, 2377–2382. [Google Scholar] [CrossRef]
- Ransy, C.; Vaz, C.; Lombès, A.; Bouillaud, F. Use of H2O2 to cause oxidative stress, the catalase issue. Int. J. Mol. Sci. 2020, 21, 9149. [Google Scholar] [CrossRef]
- Zhou, X.; Hong, Y.; Zhang, H.; Li, X. Mesenchymal stem cell senescence and rejuvenation: Current status and challenges. Front. Cell Dev. Biol. 2020, 8, 364. [Google Scholar] [CrossRef]
- Liao, N.; Shi, Y.; Zhang, C.; Zheng, Y.; Wang, Y.; Zhao, B.; Zeng, Y.; Liu, X.; Liu, J. Antioxidants inhibit cell senescence and preserve stemness of adipose tissue-derived stem cells by reducing ROS generation during long-term in vitro expansion. Stem Cell Res. Ther. 2019, 10, 306. [Google Scholar] [CrossRef]
- Weiss, A.R.R.; Dahlke, M.H. Immunomodulation by mesenchymal stem cells (MSCs): Mechanisms of action of living, apoptotic, and dead MSCs. Front. Immunol. 2019, 10, 1191. [Google Scholar] [CrossRef]
- Denu, R.A.; Hematti, P. Effects of oxidative stress on mesenchymal stem cell biology. Oxid. Med. Cell. Longev. 2016, 2016, 2989076. [Google Scholar] [CrossRef] [PubMed]
- Goiris, K.; Muylaert, K.; Fraeye, I.; Foubert, I.; De Brabanter, J.; De Cooman, L. Antioxidant potential of microalgae in relation to their phenolic and carotenoid content. J. Appl. Phycol. 2012, 24, 1477–1486. [Google Scholar] [CrossRef]
- Ni, Z.; Zhou, S.; Li, S.; Kuang, L.; Chen, H.; Luo, X.; Quyang, J.; He, M.; Du, X.; Chen, L. Exosomes: Roles and therapeutic potential in osteoarthritis. Bone Res. 2020, 8, 25. [Google Scholar] [CrossRef] [PubMed]
- Papamichail, I.; Louli, V.; Magoulas, K. Supercritical fluid extraction of celery seed oil. J. Supercrit. Fluids 2000, 18, 213–226. [Google Scholar] [CrossRef]
- Folch, J.; Lees, M.; Sloane Stanley, G.H. A simple method for the isolation and purification of total lipids from animal tissues. J. Biol. Chem. 1957, 226, 497–509. [Google Scholar] [CrossRef]
- Moheimani, N.R.; Borowitzka, M.A.; Isdepsky, A.; Sing, S. Standard Methods for Measuring Growth of Algae and Their Composition. In Algae for Biofuels and Energy; Springer: Berlin/Heidelberg, Germany, 2013; pp. 265–284. [Google Scholar]
- Kjeldahl, J.G.C.T. Neue methode zur bestimmung des stickstoffs in organischen körpern. Z. Anal. Chem. 1883, 22, 366–382. [Google Scholar] [CrossRef]
- Bremner, J.M. Determination of nitrogen in soil by the Kjeldahl method. J. Agric. Sci. 1960, 55, 11–33. [Google Scholar] [CrossRef]
- Laina, K.M.; Eleni, P.N.; Tsitseli, K.G.; Krokida, M.K. Process Design for the Extraction of Bioactive Compounds from Several Mediterranean Medicinal Plants. Chem. Eng. Trans. 2021, 86, 1327–1332. [Google Scholar]
- Drosou, C.; Kyriakopoulou, K.; Bimpilas, A.; Tsimogiannis, D.; Krokida, M. A comparative study on different extraction techniques to recover red grape pomace polyphenols from vinification byproducts. Ind. Crops Prod. 2015, 75, 141–149. [Google Scholar] [CrossRef]
- Jeffrey, S.W.; Humphrey, G.F. New spectrophotometric equations for determining chlorophylls a, b, c1 and c2 in higher plants, algae and natural phytoplankton. Biochem. Physiol. Pflanz. 1975, 167, 191–194. [Google Scholar] [CrossRef]
- Jeffrey, S.W.; Mantoura, R.F.C.; Wright, S.W. Phytoplankton Pigments in Oceanography: Monographs on Oceanographic Methodology; United Nations Educational, Scientific and Cultural Organizations: Paris, France, 1997. [Google Scholar]
- Stramarkou, M.; Papadaki, S.; Kyriakopoulou, K.; Krokida, M. Effect of drying and extraction conditions on the recovery of bioactive compounds from Chlorella vulgaris. J. Appl. Phycol. 2017, 29, 2947–2960. [Google Scholar] [CrossRef]
- Marazioti, A.; Papadia, K.; Kannavou, M.; Spella, M.; Basta, A.; de Lastic, A.L.; Rodi, M.; Mouzaki, A.; Samiotaki, M.; Panayotou, G.; et al. Cellular Vesicles: New insights in engineering methods, interaction with cells and potential for brain targeting. J. Pharmacol. Exp. Ther. 2019, 370, 772–785. [Google Scholar] [CrossRef] [PubMed]


| Primary Composition (% 1) | Commercially Available [25] | Laboratory Cultivated |
|---|---|---|
| Lipid | 22.17 ± 0.46 | 17.64 ± 0.13 |
| Carbohydrate | 33.84 ± 1.33 | 19.51 ± 0.90 |
| Protein | 44.48 ± 0.77 | 39.70 ± 0.18 |
| Ash | 5.63 ± 0.06 | 10.70 ± 0.01 |
| Moisture | 2.32 ± 0.12 | 1.15 ± 0.09 |
| Response | SLE cult. | SLE com. [25] | SFE com. [26] |
|---|---|---|---|
| Yield (% w/w) | 17.53 ± 0.54 | 15.39 ± 0.54 | 3.37 ± 0.07 |
| Total phenolic content (mgGA/gextr) | 14.88 ± 3.61 | 18.23 ± 3.61 | 18.29 ± 2.05 |
| Total chlorophyll content (mg/gextr) | 87.27 ± 2.48 | 53.47 ± 2.48 | 32.55 ± 1.54 |
| Chlorophyll a (mg/gextr) | 32.49 ± 1.77 | 36.61 ± 1.77 | 32.55 ± 1.54 |
| Chlorophyll b (mg/gextr) | 44.45 ± 1.02 | 13.92 ± 1.02 | - |
| Chlorophyll c (mg/gextr) | 10.33 ± 0.32 | 2.94 ± 0.32 | - |
| Selected carotenoid content (mg/gextr) | 15.37 ± 0.13 | 4.12 ± 0.13 | 10.00 ± 0.30 |
| Astaxanthin (mg/gextr) | 0.503 ± 0.020 | 0.430 ± 0.020 | 0.155 ± 0.022 |
| Lutein (mg/gextr) | 13.92 ± 0.13 | 3.40 ± 0.13 | 8.78 ± 0.32 |
| β-carotene (mg/gextr) | 0.952 ± 0.011 | 0.290 ± 0.011 | 1.07 ± 0.24 |
| Total carotenoid content (mg/gextr) | 27.03 ± 0.52 | 9.92 ± 0.52 | 21.14 ± 1.39 |
| Antioxidant activity—IC50 (mgextr/mgDPPH) | 69.72 ± 5.52 | 52.58 ± 5.52 | 44.35 ± 4.32 |
| Identified Carotenoids | ||||
|---|---|---|---|---|
| Astaxanthin | Lutein | β-Carotene | ||
| SLE cult. | Retention time (min) | 6.3 | 9.9 | 23.9 |
| SLE com. | 7.1 | 9.6 | 24.0 | |
| SFE com. | 7.0 | 10.0 | 24.2 | |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Savvidou, M.G.; Georgiopoulou, I.; Antoniou, N.; Tzima, S.; Kontou, M.; Louli, V.; Fatouros, C.; Magoulas, K.; Kolisis, F.N. Extracts from Chlorella vulgaris Protect Mesenchymal Stromal Cells from Oxidative Stress Induced by Hydrogen Peroxide. Plants 2023, 12, 361. https://doi.org/10.3390/plants12020361
Savvidou MG, Georgiopoulou I, Antoniou N, Tzima S, Kontou M, Louli V, Fatouros C, Magoulas K, Kolisis FN. Extracts from Chlorella vulgaris Protect Mesenchymal Stromal Cells from Oxidative Stress Induced by Hydrogen Peroxide. Plants. 2023; 12(2):361. https://doi.org/10.3390/plants12020361
Chicago/Turabian StyleSavvidou, Maria G., Ioulia Georgiopoulou, Nasia Antoniou, Soultana Tzima, Maria Kontou, Vasiliki Louli, Chronis Fatouros, Kostis Magoulas, and Fragiskos N. Kolisis. 2023. "Extracts from Chlorella vulgaris Protect Mesenchymal Stromal Cells from Oxidative Stress Induced by Hydrogen Peroxide" Plants 12, no. 2: 361. https://doi.org/10.3390/plants12020361
APA StyleSavvidou, M. G., Georgiopoulou, I., Antoniou, N., Tzima, S., Kontou, M., Louli, V., Fatouros, C., Magoulas, K., & Kolisis, F. N. (2023). Extracts from Chlorella vulgaris Protect Mesenchymal Stromal Cells from Oxidative Stress Induced by Hydrogen Peroxide. Plants, 12(2), 361. https://doi.org/10.3390/plants12020361










