The Effect of Different Agrotechnical Treatments on the Establishment of Miscanthus Hybrids in Soil Contaminated with Trace Metals
Abstract
1. Introduction
2. Results
3. Discussion
4. Materials and Methods
4.1. Trial Design
4.2. Soil and Plants Properties
4.3. Statistical Analysis
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Kalemba, E.M.; Bagniewska-Zadworna, A.; Suszka, J.; Pukacka, S. Dehydration sensitivity at the early seedling establishment stages of the European beech (Fagus sylvatica L.). Forests 2019, 10, 900. [Google Scholar] [CrossRef]
- Clifton-Brown, J.; Robson, P.; Sanderson, R.; Hastings, A.; Valentine, J.; Donnison, I. Thermal requirements for seed germination in Miscanthus compared with Switchgrass (Panicum virgatum), Reed canary grass (Phalaris arundinaceae), Maize (Zea mays) and perennial ryegrass (Lolium perenne). Glob. Change Biol. Bioenergy 2011, 3, 375–386. [Google Scholar] [CrossRef]
- Heaton, E.A.; Dohleman, F.G.; Miguez, A.F.; Juvik, J.A.; Lozovaya, V.; Widholm, J.; Zabotina, O.A.; McIsaac, G.F.; David, M.B.; Voigt, T.B.; et al. Chapter 3—Miscanthus: A Promising Biomass Crop. In Advances in Botanical Research; Kader, J.-C., Delseny, M., Eds.; Academic Press: Cambridge, MA, USA, 2010; Volume 56, pp. 75–137. [Google Scholar] [CrossRef]
- Panoutsou, C.; Chiaramonti, D. Socio-Economic Opportunities from Miscanthus Cultivation in Marginal Land for Bioenergy. Energies 2020, 13, 2741. [Google Scholar] [CrossRef]
- Pogrzeba, M.; Krzyżak, J.; Rusinowski, S.; McCalmont, J.P.; Jensen, E. Energy Crop at Heavy Metal-Contaminated Arable Land as an Alternative for Food and Feed Production: Biomass Quantity and Quality. In Plant Metallomics and Functional Omics: A System-Wide Perspective; Sablok, G., Ed.; Springer: Cham, Switzerland, 2019; pp. 1–21. [Google Scholar] [CrossRef]
- Cadoux, S.; Riche, A.B.; Yates, N.E.; Machet, J.-M. Nutrient Requirements of Miscanthus x Giganteus: Conclusions from a Review of Published Studies. Biomass Bioenergy 2012, 38, 14–22. [Google Scholar] [CrossRef]
- Magenau, E.; Clifton-Brown, J.; Awty-Carroll, D.; Ashman, C.; Ferrarini, A.; Kontek, M.; Martani, E.; Roderick, K.; Amaducci, S.; Davey, C.; et al. Site Impacts Nutrient Translocation Efficiency in Intraspecies and Interspecies Miscanthus Hybrids on Marginal Lands. Glob. Change Biol. Bioenergy 2022, 14, 1035–1054. [Google Scholar] [CrossRef]
- Nakajima, T.; Yamada, T.; Anzoua, K.G.; Kokubo, R.; Noborio, K. Carbon Sequestration and Yield Performances of Miscanthus × Giganteus and Miscanthus Sinensis. Carbon Manag. 2018, 9, 415–423. [Google Scholar] [CrossRef]
- Shafique, F.; Ali, Q.; Malik, A. Effects of Heavy Metal Toxicity on Maze Seedlings Growth Traits. Biol. Clin. Sci. Res. J. 2020, 2020, 1–5. [Google Scholar] [CrossRef]
- Yun, Y.; Liang, L.; Wei, Y.; Luo, Z.; Yuan, F.; Li, G.; Sang, N. Exposure to Nitro-PAHs Interfere with Germination and Early Growth of Hordeum Vulgare via Oxidative Stress. Ecotoxicol. Environ. Saf. 2019, 180, 756–761. [Google Scholar] [CrossRef]
- Clifton-Brown, J.C.; Lewandowski, I. Overwintering Problems of Newly Established Miscanthus Plantations Can Be Overcome by Identifying Genotypes with Improved Rhizome Cold Tolerance. New Phytol. 2000, 148, 287–294. [Google Scholar] [CrossRef]
- Mäkelä, P.S.A.; Wasonga, D.O.; Solano Hernandez, A.; Santanen, A. Seedling Growth and Phosphorus Uptake in Response to Different Phosphorus Sources. Agronomy 2020, 10, 1089. [Google Scholar] [CrossRef]
- Song, X.; Wan, F.; Chang, X.; Zhang, J.; Sun, M.; Liu, Y. Effects of Nutrient Deficiency on Root Morphology and Nutrient Allocation in Pistacia Chinensis Bunge Seedlings. Forests 2019, 10, 1035. [Google Scholar] [CrossRef]
- O’Loughlin, J.; Finnan, J.; McDonnell, K. Accelerating Early Growth in Miscanthus with the Application of Plastic Mulch Film. Biomass Bioenergy 2017, 100, 52–61. [Google Scholar] [CrossRef]
- Krzyżak, J.; Pogrzeba, M.; Rusinowski, S.; Clifton-Brown, J.; McCalmont, J.P.; Kiesel, A.; Mangold, A.; Mos, M. Heavy Metal Uptake by Novel Miscanthus Seed-Based Hybrids Cultivated in Heavy Metal Contaminated Soil. Civ. Environ. Eng. Rep. 2017, 26, 121–132. [Google Scholar] [CrossRef][Green Version]
- Xue, S.; Kalinina, O.; Lewandowski, I. Present and Future Options for Miscanthus Propagation and Establishment. Renew. Sustain. Energy Rev. 2015, 49, 1233–1246. [Google Scholar] [CrossRef]
- Clifton-Brown, J.; Hastings, A.; Mos, M.; McCalmont, J.P.; Ashman, C.; Awty-Carroll, D.; Cerazy, J.; Chiang, Y.-C.; Cosentino, S.; Cracroft-Eley, W.; et al. Progress in Upscaling Miscanthus Biomass Production for the European Bio-Economy with Seed-Based Hybrids. Glob. Change Biol. Bioenergy 2017, 9, 6–17. [Google Scholar] [CrossRef]
- Hassan, M.A.; Xiang, C.; Farooq, M.; Muhammad, N.; Yan, Z.; Hui, X.; Yuanyuan, K.; Bruno, A.K.; Lele, Z.; Jincai, L. Cold Stress in Wheat: Plant Acclimation Responses and Management Strategies. Front. Plant Sci. 2021, 12, 676884. [Google Scholar] [CrossRef] [PubMed]
- Teixeira, G.C.M.; de Mello Prado, R.; Rocha, A.M.S.; dos Santos, L.C.N.; dos Santos Sarah, M.M.; Gratão, P.L.; Fernandes, C. Silicon in Pre-Sprouted Sugarcane Seedlings Mitigates the Effects of Water Deficit after Transplanting. J. Soil Sci. Plant Nutr. 2020, 20, 849–859. [Google Scholar] [CrossRef]
- Baruah, N.; Mondal, S.C.; Farooq, M.; Gogoi, N. Influence of Heavy Metals on Seed Germination and Seedling Growth of Wheat, Pea, and Tomato. Water Air Soil Pollut. 2019, 230, 273. [Google Scholar] [CrossRef]
- Ashman, C.; Awty-Carroll, D.; Mos, M.; Robson, P.; Clifton-Brown, J. Assessing Seed Priming, Sowing Date, and Mulch Film to Improve the Germination and Survival of Direct-Sown Miscanthus Sinensis in the United Kingdom. Glob. Change Biol. Bioenergy 2018, 10, 612–627. [Google Scholar] [CrossRef]
- Rusinowski, S.; Krzyżak, J.; Sitko, K.; Kalaji, H.M.; Jensen, E.; Pogrzeba, M. Cultivation of C4 Perennial Energy Grasses on Heavy Metal Contaminated Arable Land: Impact on Soil, Biomass, and Photosynthetic Traits. Environ. Pollut. 2019, 250, 300–311. [Google Scholar] [CrossRef]
- Rusinowski, S.; Krzyżak, J.; Clifton-Brown, J.; Jensen, E.; Mos, M.; Webster, R.; Sitko, K.; Pogrzeba, M. New Miscanthus Hybrids Cultivated at a Polish Metal-Contaminated Site Demonstrate High Stomatal Regulation and Reduced Shoot Pb and Cd Concentrations. Environ. Pollut. 2019, 252, 1377–1387. [Google Scholar] [CrossRef] [PubMed]
- Korzeniowska, J.; Stanislawska-Glubiak, E. Phytoremediation Potential of Miscanthus × Giganteus and Spartina Pectinata in Soil Contaminated with Heavy Metals. Environ. Sci. Pollut. Res. 2015, 22, 11648–11657. [Google Scholar] [CrossRef] [PubMed]
- Kocoń, A.; Jurga, B. The Evaluation of Growth and Phytoextraction Potential of Miscanthus x Giganteus and Sida Hermaphrodita on Soil Contaminated Simultaneously with Cd, Cu, Ni, Pb, and Zn. Environ. Sci. Pollut. Res. 2017, 24, 4990–5000. [Google Scholar] [CrossRef]
- Pogrzeba, M.; Rusinowski, S.; Krzyżak, J. Macroelements and Heavy Metals Content in Energy Crops Cultivated on Contaminated Soil under Different Fertilization—Case Studies on Autumn Harvest. Environ. Sci. Pollut. Res. 2018, 25, 12096–12106. [Google Scholar] [CrossRef] [PubMed]
- Li, H.; Wu, W.; Min, X.; Zhan, W.; Fang, T.; Dong, X.; Shi, Y. Immobilization and Assessment of Heavy Metals in Chicken Manure Compost Amended with Rice Straw-Derived Biochar. Environ. Pollut. Bioavailab. 2021, 33, 1–10. [Google Scholar] [CrossRef]
- Wasilkowski, D.; Nowak, A.; Michalska, J.; Mrozik, A. Ecological Restoration of Heavy Metal-Contaminated Soil Using Na-Bentonite and Green Compost Coupled with the Cultivation of the Grass Festuca arundinacea. Ecol. Eng. 2019, 138, 420–433. [Google Scholar] [CrossRef]
- Pavel, P.-B.; Puschenreiter, M.; Wenzel, W.W.; Diacu, E.; Barbu, C.H. Aided Phytostabilization Using Miscanthus Sinensis × giganteus on Heavy Metal-Contaminated Soils. Sci. Total Environ. 2014, 479–480, 125–131. [Google Scholar] [CrossRef]
- Pogrzeba, M.; Rusinowski, S.; Krzyżak, J.; Szada-Borzyszkowska, A.; McCalmont, J.P.; Zieleźnik-Rusinowska, P.; Słaboń, N.; Sas-Nowosielska, A. Dactylis Glomerata L. Cultivation on Mercury Contaminated Soil and Its Physiological Response to Granular Sulphur Aided Phytostabilization. Environ. Pollut. 2019, 255, 113271. [Google Scholar] [CrossRef]
- Amirahmadi, E.; Ghorbani, M.; Moudrý, J. Effects of Zeolite on Aggregation, Nutrient Availability, and Growth Characteristics of Corn (Zea Mays L.) in Cadmium-Contaminated Soils. Water Air Soil Pollut. 2022, 233, 436. [Google Scholar] [CrossRef]
- Awasthi, M.K.; Duan, Y.; Awasthi, S.K.; Liu, T.; Chen, H.; Pandey, A.; Zhang, Z.; Taherzadeh, M.J. Emerging Applications of Biochar: Improving Pig Manure Composting and Attenuation of Heavy Metal Mobility in Mature Compost. J. Hazard. Mater. 2020, 389, 122116. [Google Scholar] [CrossRef]
- Lebrun, M.; Miard, F.; Nandillon, R.; Scippa, G.S.; Bourgerie, S.; Morabito, D. Biochar Effect Associated with Compost and Iron to Promote Pb and As Soil Stabilization and Salix Viminalis L. Growth. Chemosphere 2019, 222, 810–822. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Liu, Y.; Zhan, W.; Zheng, K.; Wang, J.; Zhang, C.; Chen, R. Stabilization of Heavy Metal-Contaminated Soils by Biochar: Challenges and Recommendations. Sci. Total Environ. 2020, 729, 139060. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Wang, S. Preparation, modification and environmental application of biochar: A review. J. Clean. Prod. 2019, 227, 1002–1022. [Google Scholar] [CrossRef]
- Ghorbani, M.; Konvalina, P.; Neugschwandtner, R.W.; Kopecký, M.; Amirahmadi, E.; Moudrý, J.; Menšík, L. Preliminary Findings on Cadmium Bioaccumulation and Photosynthesis in Rice (Oryza Sativa L.) and Maize (Zea Mays L.) Using Biochar Made from C3- and C4-Originated Straw. Plants 2022, 11, 1424. [Google Scholar] [CrossRef] [PubMed]
- Tian, H.Z.; Zhu, C.Y.; Gao, J.J.; Cheng, K.; Hao, J.M.; Wang, K.; Hua, S.B.; Wang, Y.; Zhou, J.R. Quantitative Assessment of Atmospheric Emissions of Toxic Heavy Metals from Anthropogenic Sources in China: Historical Trend, Spatial Distribution, Uncertainties, and Control Policies. Atmos. Chem. Phys. 2015, 15, 10127–10147. [Google Scholar] [CrossRef]
- Romic, M.; Romic, D. Heavy Metals Distribution in Agricultural Topsoils in Urban Area. Env. Geol. 2003, 43, 795–805. [Google Scholar] [CrossRef]
- Pogrzeba, M.; Rusinowski, S.; Sitko, K.; Krzyżak, J.; Skalska, A.; Małkowski, E.; Ciszek, D.; Werle, S.; McCalmont, J.P.; Mos, M.; et al. Relationships between Soil Parameters and Physiological Status of Miscanthus x Giganteus Cultivated on Soil Contaminated with Trace Elements under NPK Fertilisation vs. Microbial Inoculation. Environ. Pollut. 2017, 225, 163–174. [Google Scholar] [CrossRef]
- Peijnenburg, W.J.G.M.; Zablotskaja, M.; Vijver, M.G. Monitoring Metals in Terrestrial Environments within a Bioavailability Framework and a Focus on Soil Extraction. Ecotoxicol. Environ. Saf. 2007, 67, 163–179. [Google Scholar] [CrossRef]
- Maathuis, F.J. Physiological Functions of Mineral Macronutrients. Curr. Opin. Plant Biol. 2009, 12, 250–258. [Google Scholar] [CrossRef]
- He, L.; Zhong, H.; Liu, G.; Dai, Z.; Brookes, P.C.; Xu, J. Remediation of Heavy Metal Contaminated Soils by Biochar: Mechanisms, Potential Risks and Applications in China. Environ. Pollut. 2019, 252, 846–855. [Google Scholar] [CrossRef]
- Mansoor, S.; Kour, N.; Manhas, S.; Zahid, S.; Wani, O.A.; Sharma, V.; Wijaya, L.; Alyemeni, M.N.; Alsahli, A.A.; El-Serehy, H.A.; et al. Biochar as a Tool for Effective Management of Drought and Heavy Metal Toxicity. Chemosphere 2021, 271, 129458. [Google Scholar] [CrossRef] [PubMed]
- Irfan, M.; Mudassir, M.; Khan, M.J.; Dawar, K.M.; Muhammad, D.; Mian, I.A.; Ali, W.; Fahad, S.; Saud, S.; Hayat, Z.; et al. Heavy Metals Immobilization and Improvement in Maize (Zea Mays L.) Growth Amended with Biochar and Compost. Sci. Rep. 2021, 11, 18416. [Google Scholar] [CrossRef] [PubMed]
- Zhang, R.-H.; Li, Z.-G.; Liu, X.-D.; Wang, B.; Zhou, G.-L.; Huang, X.-X.; Lin, C.-F.; Wang, A.; Brooks, M. Immobilization and Bioavailability of Heavy Metals in Greenhouse Soils Amended with Rice Straw-Derived Biochar. Ecol. Eng. 2017, 98, 183–188. [Google Scholar] [CrossRef]
- Rajkumar, M.; Prasad, M.N.V.; Swaminathan, S.; Freitas, H. Climate Change Driven Plant–Metal–Microbe Interactions. Environ. Int. 2013, 53, 74–86. [Google Scholar] [CrossRef]
- Nsanganwimana, F.; Al Souki, K.S.; Waterlot, C.; Douay, F.; Pelfrêne, A.; Ridošková, A.; Louvel, B.; Pourrut, B. Potentials of Miscanthus × Giganteus for Phytostabilization of Trace Element-Contaminated Soils: Ex Situ Experiment. Ecotoxicol. Environ. Saf. 2021, 214, 112125. [Google Scholar] [CrossRef]
- Eid, E.M.; Shaltout, K.H.; Alamri, S.A.; Alrumman, S.A.; Hussain, A.A.; Sewelam, N.; Ragab, G.A. Monitored Sewage Sludge Application Improves Soil Quality, Enhances Plant Growth, and Provides Evidence for Metal Remediation by Sorghum bicolor L. J. Soil Sci. Plant Nutr. 2021, 21, 2325–2338. [Google Scholar] [CrossRef]
- PN-ISO 11265:1997; Soil Quality—Determination of the Specific Electrical Conductivity. PKN: Warsaw, Poland, 1997.
- PNR-04032:1998; Soils and Mineral Soil Materials—Soil Sampling and Determination of Particle Size Distribution in Mineral Soil Material. (In Polish). PKN: Warsaw, Poland, 1998.
- PN-ISO 14235:2003; Soil Quality. Determination of Organic Carbon by Sulfochromic Oxidation. PKN: Warsaw, Poland, 2003.
- Egner, H.; Riehm, H.; Domingo, W.R. Investigations on the chemical soil analysis as a basis for assessing the soil nutrient status. II: Chemical extraction methods for phosphorus and potassium determination. Kungliga Lantbrukshügskolans Annaler 1960, 26, 199–215. [Google Scholar]
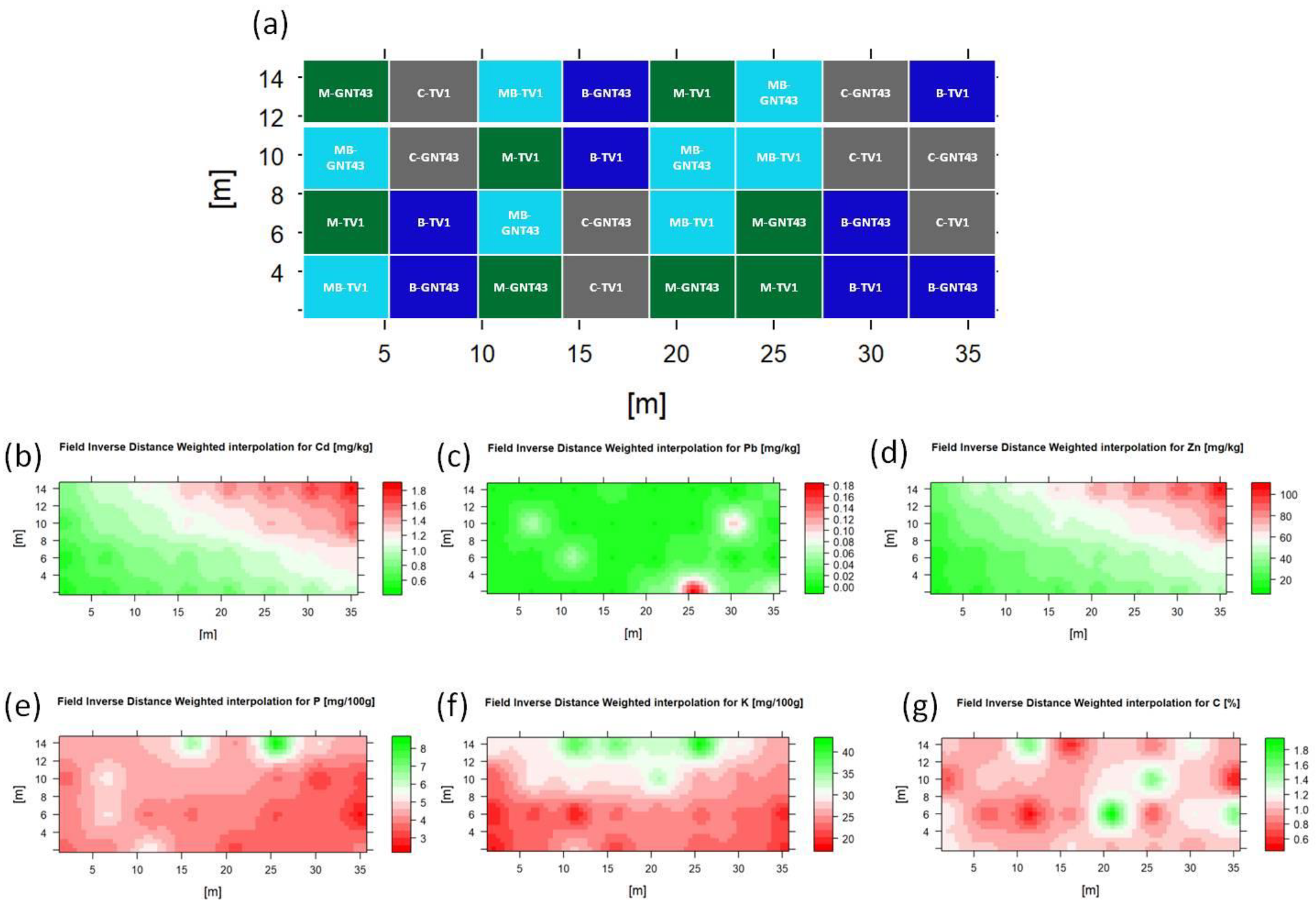
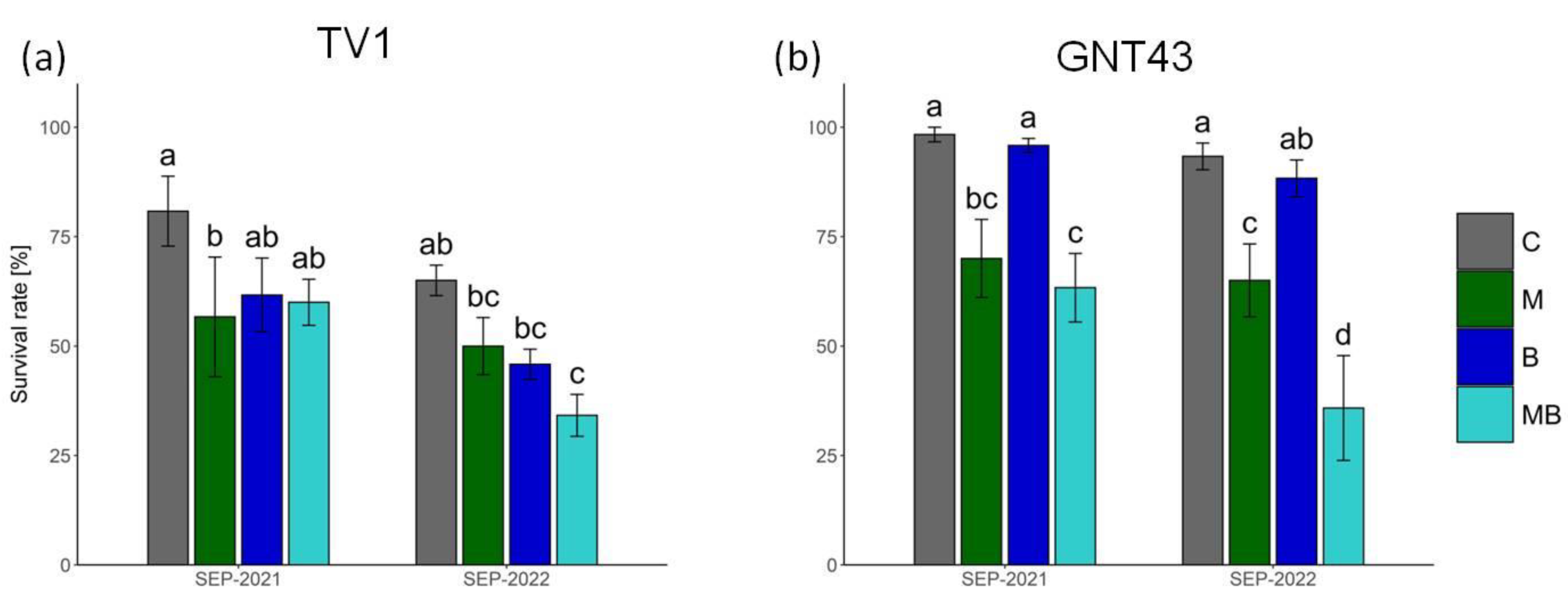


| Variety | TV1 | GNT43 | ||||||
|---|---|---|---|---|---|---|---|---|
| Treatment | C | M | B | MB | C | M | B | MB |
| Physicochemical Parameters | ||||||||
| pHH2O | 7.11 ± 0.06 a | 7.15 ± 0.04 a | 7.06 ± 0.04 a | 7.14 ± 0.04 a | 7.07 ± 0.06 a | 7.17 ± 0.06 a | 7.07 ± 0.06 a | 7.15 ± 0.06 a |
| pHKCl | 6.39 ± 0.14 a | 6.50 ± 0.11 a | 6.38 ± 0.09 a | 6.49 ± 0.08 a | 6.42 ± 0.06 a | 6.41 ± 0.15 a | 6.40 ± 0.11 a | 6.48 ± 0.06 a |
| EC [µS cm−1] | 75.7 ± 8.4 a | 74.8 ± 0.7 a | 74.4 ± 2.8 a | 76.9 ± 7.4 a | 67.2 ± 2.7 a | 79.4 ± 7.1 a | 73.5 ± 6.8 a | 72.4 ± 2.8 a |
| Pbbio [mg kg−1] | 0.007 ± 0.005 a | 0.024 ± 0.014 a | 0.032 ± 0.018 a | 0.003 ± 0.003 a | 0.011 ± 0.008 a | 0.001 ± 0.001 a | 0.033 ± 0.025 a | 0.045 ± 0.042 a |
| Cdbio [mg kg−1] | 1.18 ± 0.22 a | 0.95 ± 0.20 a | 1.12 ± 0.19 a | 0.94 ± 0.11 a | 0.98 ± 0.12 a | 1.13 ± 0.22 a | 1.25 ± 0.23 a | 0.82 ± 0.10 a |
| Znbio [mg kg−1] | 55 ± 14 a | 41 ± 14 a | 51 ± 13 a | 38 ± 6 a | 41 ± 8 a | 52 ± 13 a | 60 ± 16 a | 32 ± 6 a |
| OM [%] | 5.75 ± 0.12 ab | 5.75 ± 0.15 ab | 5.98 ± 0.15 ab | 6.08 ± 0.16 a | 5.83 ± 0.14 ab | 5.90 ± 0.24 ab | 6.00 ± 0.15 ab | 5.60 ± 0.16 b |
| P2O5 [mg 100g−1] | 4.34 ± 0.72 a | 4.73 ± 1.19 a | 4.46 ± 0.38 a | 4.38 ± 0.41 a | 3.90 ± 0.54 a | 4.05 ± 0.23 a | 3.97 ± 0.33 a | 3.97 ± 0.19 a |
| K2O [mg 100g−1] | 26.6 ± 3.4 a | 26.8 ± 5.1 a | 25.8 ± 2.6 a | 28.4 ± 2.3 a | 23.1 ± 2.0 a | 29.1 ± 4.3 a | 25.4 ± 1.8 a | 23.7 ± 2.4 a |
| Corg [%] | 0.85 ± 0.16 b | 0.77 ± 0.09 b | 1.06 ± 0.08 ab | 0.98 ± 0.09 ab | 1.04 ± 0.17 ab | 1.34 ± 0.13 a | 1.00 ± 0.04 ab | 1.27 ± 0.20 a |
| Pb [mg kg−1] | 450 ± 23 a | 442 ± 29 a | 460 ± 22 a | 463 ± 14 a | 438 ± 21 a | 454 ± 35 a | 483 ± 17 a | 423 ± 25 a |
| Cd [mg kg−1] | 17.9 ± 0.7 a | 17.8 ± 1.2 a | 18.4 ± 0.7 a | 18.6 ± 0.4 a | 17.3 ± 0.8 a | 18.1 ± 1.3 a | 19.2 ± 0.4 a | 16.8 ± 1.1 a |
| Zn [mg kg−1] | 1910 ± 60 a | 1880 ± 110 a | 1920 ± 70 a | 2000 ± 30 a | 1830 ± 70 a | 1910 ± 130 a | 2030 ± 20 a | 1800 ± 120 a |
| Mg [mg kg−1] | 2400 ± 390 a | 2410 ± 150 a | 2290 ± 170 a | 2750 ± 310 a | 2260 ± 110 a | 2410 ± 230 a | 2410 ± 270 a | 2410 ± 90 a |
| P [mg kg−1] | 767 ± 44 a | 790 ± 30 a | 801 ± 19 a | 811 ± 16 a | 740 ± 23 a | 774 ± 46 a | 811 ± 33 a | 769 ± 34 a |
| K [mg kg−1] | 1120 ± 50 ab | 1120 ± 20 ab | 1120 ± 30 ab | 1190 ± 40 a | 1060 ± 30 b | 1140 ± 60 ab | 1130 ± 40 ab | 1150 ± 30 ab |
| Ca [mg kg−1] | 3960 ± 730 a | 4050 ± 280 a | 3800 ± 320 a | 4720 ± 570 a | 3690 ± 210 a | 4000 ± 360 a | 4020 ± 500 a | 3930 ± 120 a |
| Fe [mg kg−1] | 15,500 ± 250 b | 16,200 ± 350 ab | 15,800 ± 150 ab | 16,500 ± 350 ab | 16,100 ± 350 ab | 16,000 ± 400 ab | 16,600 ± 60 a | 16,000 ± 500 ab |
| Variety | TV1 | GNT43 | ||||||
|---|---|---|---|---|---|---|---|---|
| Treatment | C | M | B | MB | C | M | B | MB |
| Pb [mg kg−1] | 30.8 ± 6.3 ab | 34.1 ± 4.6 a | 29.6 ± 1.5 ab | 33.3 ± 2.7 a | 16.5 ± 0.5 c | 19.5 ± 2.8 bc | 29.4 ± 6.3 ab | 31.0 ± 5.7 ab |
| Cd [mg kg−1] | 1.02 ± 0.07 b | 1.05 ± 0.17 b | 1.00 ± 0.07 b | 0.98 ± 0.12 b | 0.80 ± 0.03 b | 1.16 ± 0.23 ab | 1.17 ± 0.17 ab | 1.51 ± 0.14 a |
| Zn [mg kg−1] | 170 ± 20 b | 165 ± 17 b | 166 ± 12 b | 161 ± 13 b | 248 ± 16 a | 299 ± 28 a | 309 ± 35 a | 293 ± 48 a |
| Ca [mg kg−1] | 1510 ± 140 b | 1570 ± 160 ab | 1520 ± 130 b | 1490 ± 110 b | 1230 ± 50 b | 1240 ± 150 b | 1510 ± 250 b | 2060 ± 290 a |
| Mg [mg kg−1] | 890 ± 50 abc | 910 ± 50 abc | 950 ± 110 ab | 880 ± 20 bc | 680 ± 50 c | 760 ± 50 bc | 800 ± 110 bc | 1110 ± 130 a |
| N [%] | 0.93 ± 0.07 ab | 1.03 ± 0.03 ab | 1.01 ± 0.07 ab | 0.95 ± 0.03 ab | 0.87 ± 0.06 b | 1.00 ± 0.14 ab | 1.18 ± 0.16 a | 1.17 ± 0.10 a |
| P [mg kg−1] | 950 ± 80 b | 1210 ± 30 ab | 1190 ± 90 ab | 970 ± 90 b | 1090 ± 180 ab | 1020 ± 190 ab | 1130 ± 150 ab | 1390 ± 150 a |
| K [mg kg−1] | 6430 ± 600 d | 7970 ± 530 cd | 7220 ± 570 cd | 6440 ± 650 d | 14,880 ± 1940 ab | 16,000 ± 3390 ab | 11,680 ± 1010 bc | 17,650 ± 2760 a |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Krzyżak, J.; Rusinowski, S.; Sitko, K.; Szada-Borzyszkowska, A.; Stec, R.; Jensen, E.; Clifton-Brown, J.; Kiesel, A.; Lewin, E.; Janota, P.; et al. The Effect of Different Agrotechnical Treatments on the Establishment of Miscanthus Hybrids in Soil Contaminated with Trace Metals. Plants 2023, 12, 98. https://doi.org/10.3390/plants12010098
Krzyżak J, Rusinowski S, Sitko K, Szada-Borzyszkowska A, Stec R, Jensen E, Clifton-Brown J, Kiesel A, Lewin E, Janota P, et al. The Effect of Different Agrotechnical Treatments on the Establishment of Miscanthus Hybrids in Soil Contaminated with Trace Metals. Plants. 2023; 12(1):98. https://doi.org/10.3390/plants12010098
Chicago/Turabian StyleKrzyżak, Jacek, Szymon Rusinowski, Krzysztof Sitko, Alicja Szada-Borzyszkowska, Radosław Stec, Elaine Jensen, John Clifton-Brown, Andreas Kiesel, Eva Lewin, Paulina Janota, and et al. 2023. "The Effect of Different Agrotechnical Treatments on the Establishment of Miscanthus Hybrids in Soil Contaminated with Trace Metals" Plants 12, no. 1: 98. https://doi.org/10.3390/plants12010098
APA StyleKrzyżak, J., Rusinowski, S., Sitko, K., Szada-Borzyszkowska, A., Stec, R., Jensen, E., Clifton-Brown, J., Kiesel, A., Lewin, E., Janota, P., & Pogrzeba, M. (2023). The Effect of Different Agrotechnical Treatments on the Establishment of Miscanthus Hybrids in Soil Contaminated with Trace Metals. Plants, 12(1), 98. https://doi.org/10.3390/plants12010098






