Healthy Zerumbone: From Natural Sources to Strategies to Improve Its Bioavailability and Oral Administration
Abstract
1. Introduction
2. Natural Sources of Zerumbone
3. Zerumbone Derivative
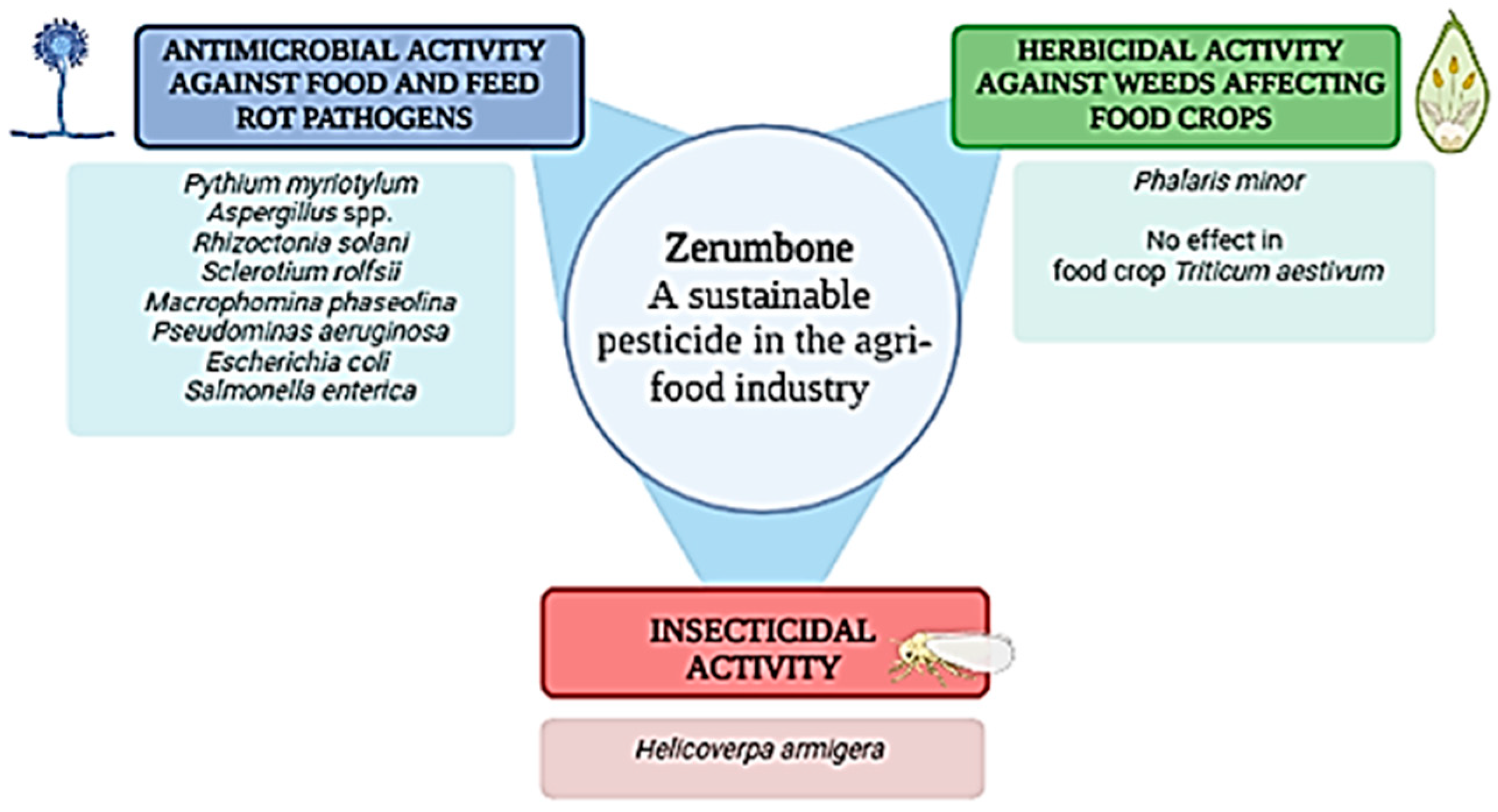
4. Further Uses of Zerumbone
5. Formulation Strategies to Enhance the Solubility and Bioavailability of Zerumbone for Oral Administration
6. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Kitayama, T. Attractive reactivity of a natural product, zerumbone. Biosci. Biotechnol. Biochem. 2011, 75, 199–207. [Google Scholar] [CrossRef]
- Padalia, R.C.; Verma, R.S.; Chauhan, A.; Singh, V.R.; Goswami, P.; Singh, S.; Verma, S.K.; Luqman, S.; Chanotiya, C.S.; Darokar, M.P. Zingiber zerumbet (L.) Roscoe ex Sm. from northern India: Potential source of zerumbone rich essential oil for antiproliferative and antibacterial applications. Ind. Crops Prod. 2018, 112, 749–754. [Google Scholar] [CrossRef]
- Moreira Da Silva, T.; Pinheiro, C.D.; Puccinelli Orlandi, P.; Pinheiro, C.C.; Soares Pontes, G. Zerumbone from Zingiber zerumbet (L.) smith: A potential prophylactic and therapeutic agent against the cariogenic bacterium Streptococcus mutans. BMC Complement. Altern. Med. 2018, 18, 301. [Google Scholar] [CrossRef] [PubMed]
- Girisa, S.; Shabnam, B.; Monisha, J.; Fan, L.; Halim, C.E.; Arfuso, F.; Ahn, K.S.; Sethi, G.; Kunnumakkara, A.B. Potential of zerumbone as an anti-cancer agent. Molecules 2019, 24, 734. [Google Scholar] [CrossRef] [PubMed]
- Deng, M.; Yun, X.; Ren, S.; Qing, Z.; Luo, F. Plants of the genus Zingiber: A review of their ethnomedicine, phytochemistry and pharmacology. Molecules 2022, 27, 2826. [Google Scholar] [CrossRef] [PubMed]
- Tian, M.; Wu, X.; Hong, Y.; Wang, H.; Deng, G.; Zhou, Y. Comparison of chemical composition and bioactivities of essential oils from fresh and dry rhizomes of Zingiber zerumbet (L.) Smith. Biomed Res. Int. 2020, 2020, 9. [Google Scholar] [CrossRef] [PubMed]
- Sharifi-Rad, M.; Varoni, E.M.; Salehi, B.; Sharifi-Rad, J.; Matthews, K.R.; Ayatollahi, S.A.; Kobarfard, F.; Ibrahim, S.A.; Mnayer, D.; Zakaria, Z.A.; et al. Plants of the genus Zingiber as a source of bioactive phytochemicals: From tradition to pharmacy. Molecules 2017, 22, 2145. [Google Scholar] [CrossRef]
- Akhtar, N.M.Y.; Jantan, I.; Arshad, L.; Haque, M.A. Standardized ethanol extract, essential oil and zerumbone of Zingiber zerumbet rhizome suppress phagocytic activity of human neutrophils. BMC Complement. Altern. Med. 2019, 19, 331. [Google Scholar] [CrossRef] [PubMed]
- Rana, V.S.; Verdeguer, M.; Blazquez, M.A. Chemical composition of the essential oil of Zingiber zerumbet var darcyi. Nat. Prod. Commun. 2012, 7, 1369–1370. [Google Scholar] [CrossRef]
- Wu, Y.; Guo, S.S.; Huang, D.Y.; Wang, C.F.; Wei, J.Y.; Li, Z.H.; Sun, J.S.; Bai, J.F.; Tian, Z.F.; Wang, P.J.; et al. Contact and repellant activities of zerumbone and its analogues from the essential oil of Zingiber zerumbet (L.) smith against Lasioderma serricorne. J. Oleo Sci. 2017, 66, 399–405. [Google Scholar] [CrossRef]
- Veena, K.S.; Gopalan, G.; Madhukrishnan, M.; Varughese, S.; Radhakrishnan, K.V.; Lankalapalli, R.S. Putative biomimetic route to 8-oxabicyclo[3.2.1]octane motif from a humulene sesquiterpenoid zerumbone. Org. Lett. 2020, 22, 6409–6413. [Google Scholar] [CrossRef] [PubMed]
- Zhang, C.; Liu, J.; Zhao, F.; Lu, C.; Zhao, G.R.; Lu, W. Production of sesquiterpenoid zerumbone from metabolic engineered Saccharomyces cerevisiae. Metab. Eng. 2018, 49, 28–35. [Google Scholar] [CrossRef] [PubMed]
- Albaayit, S.F.A.; Maharjan, R.; Abdullah, R.; Noor, M.H.M. Evaluation of anti-methicillin-resistant Staphylococcus aureus property of zerumbone. J. Appl. Biomed. 2022, 20, 15–21. [Google Scholar] [CrossRef] [PubMed]
- Woo, H.J.; Yang, J.Y.; Lee, P.; Kim, J.B.; Kim, S.H. Zerumbone inhibits Helicobacter pylori urease activity. Molecules 2021, 26, 2663. [Google Scholar] [CrossRef]
- Li, J.; Wang, L.; Sun, Y.; Wang, Z.; Qian, Y.; Duraisamy, V.; Antary, T.M.A.I. Zerumbone-induced reactive oxygen species-mediated oxidative stress re-sensitizes breast cancer cells to paclitaxel. Biotechnol. Appl. Biochem. 2022. [Google Scholar] [CrossRef] [PubMed]
- Ashraf, S.M.; Sebastian, J.; Rathinasamy, K. Zerumbone, a cyclic sesquiterpene, exerts antimitotic activity in HeLa cells through tubulin binding and exhibits synergistic activity with vinblastine and paclitaxel. Cell Prolif. 2019, 52, e12558. [Google Scholar] [CrossRef] [PubMed]
- Schnoell, J.; Stanisz, I.; Jank, B.J.; Stanek, V.; Schmid, R.; Brunner, M.; Heiduschka, G.; Kotowski, U. Zerumbone acts as a radiosensitizer in head and neck squamous cell carcinoma. Investig. New Drugs 2022, 40, 224–231. [Google Scholar] [CrossRef]
- Kim, H.R.; Rhee, K.J.; Eom, Y. Bin Anti-biofilm and antimicrobial effects of zerumbone against Bacteroides fragilis. Anaerobe 2019, 57, 99–106. [Google Scholar] [CrossRef]
- Kim, H.R.; Shin, D.S.; Jang, H.I.; Eom, Y.B. Anti-biofilm and anti-virulence effects of zerumbone against Acinetobacter baumannii. Microbiol. 2020, 166, 717–726. [Google Scholar] [CrossRef]
- Shin, D.S.; Eom, Y.B. Efficacy of zerumbone against dual-species biofilms of Candida albicans and Staphylococcus aureus. Microb. Pathog. 2019, 137, 103768. [Google Scholar] [CrossRef] [PubMed]
- Cho, H.; Rhee, K.; Eom, Y. Composition in ETBF Colonized AOM/DSS. J. Microbiol. Biotechnol. 2020, 30, 1640–1650. [Google Scholar] [CrossRef] [PubMed]
- Li, L.; Kong, L.; Song, H. The therapeutic effect of zerumbone on chronic gastritis via antioxidant mechanisms. Exp. Ther. Med. 2017, 14, 2505–2510. [Google Scholar] [CrossRef] [PubMed]
- Zulazmi, N.A.; Gopalsamy, B.; Omar Farouk, A.A.; Sulaiman, M.R.; Bharatham, B.H.; Perimal, E.K. Antiallodynic and antihyperalgesic effects of zerumbone on a mouse model of chronic constriction injury-induced neuropathic pain. Fitoterapia 2015, 105, 215–221. [Google Scholar] [CrossRef] [PubMed]
- Chia, J.S.M.; Omar Farouk, A.A.; Mohamad, A.S.; Sulaiman, M.R.; Perimal, E.K. Zerumbone alleviates chronic constriction injury-induced allodynia and hyperalgesia through serotonin 5-HT receptors. Biomed. Pharmacother. 2016, 83, 1303–1310. [Google Scholar] [CrossRef]
- Gopalsamy, B.; Chia, J.S.M.; Farouk, A.A.O.; Sulaiman, M.R.; Perimal, E.K. Zerumbone-induced analgesia modulated via potassium channels and opioid receptors in chronic constriction injury-induced neuropathic pain. Molecules 2020, 25, 3880. [Google Scholar] [CrossRef]
- Chia, J.S.M.; Farouk, A.A.O.; Mohamad, T.A.S.T.; Sulaiman, M.R.; Zakaria, H.; Hassan, N.I.; Perimal, E.K. Zerumbone ameliorates neuropathic pain symptoms via cannabinoid and ppar receptors using in vivo and in silico models. Molecules 2021, 26, 3849. [Google Scholar] [CrossRef]
- Liu, W.Y.; Tzeng, T.F.; Liu, I.M. Zerumbone, a bioactive sesquiterpene, ameliorates diabetes-induced retinal microvascular damage through inhibition of phospho-p38 mitogen-activated protein kinase and nuclear factor-κB pathways. Molecules 2016, 21, 1708. [Google Scholar] [CrossRef] [PubMed]
- Hwang, J.; Youn, K.; Ji, Y.; Lee, S.; Lim, G. Biological and computational studies for dual cholinesterases inhibitory effect of zerumbone. Nutrients 2020, 12, 1215. [Google Scholar] [CrossRef] [PubMed]
- Mohamad, N.E.; Abu, N.; Rahman, H.S.; Ky, H.; Ho, W.Y.; Lim, K.L.; How, C.W.; Rasedee, A.; Alitheen, N.B.; Yeap, S.K. Nanostructured lipid carrier improved in vivo anti-tumor and immunomodulatory effect of zerumbone in 4T1 challenged mice. RSC Adv. 2015, 5, 22066–22074. [Google Scholar] [CrossRef]
- Albaayit, S.F.A.; Rasedee, A.; Abdullah, N. Zerumbone-loaded nanostructured lipid carrier gel facilitates wound healing in rats. Rev. Bras. Farmacogn. 2020, 30, 272–278. [Google Scholar] [CrossRef]
- Rahman, H.S.; Rasedee, A.; How, C.W.; Abdul, A.B.; Zeenathul, N.A.; Othman, H.H.; Saeed, M.I.; Yeap, S.K. Zerumbone-loaded nanostructured lipid carriers: Preparation, characterization, and antileukemic effect. Int. J. Nanomed. 2013, 8, 2769–2781. [Google Scholar] [CrossRef] [PubMed]
- Md, S.; Kit, B.C.M.; Jagdish, S.; David, D.J.P.; Pandey, M.; Chatterjee, L.A. Development and in vitro evaluation of a zerumbone loaded nanosuspension drug delivery system. Crystals 2018, 8, 286. [Google Scholar]
- Hassan, M.M.; Mohammed, A.F.A.; Elamin, K.M.; Devkota, H.P.; Ohno, Y.; Motoyama, K.; Higashi, T.; Imai, T. Improvement of pharmaceutical properties of zerumbone, a multifunctional compound, using cyclodextrin derivatives. Chem. Pharm. Bull. 2020, 68, 1117–1120. [Google Scholar] [CrossRef] [PubMed]
- Li, P.; Shu, X. Zerumbone with hydroxypropyl-β-cyclodextrin inclusion complex as a potential treatment for gastritis. Int. J. Pharmacol. 2017, 13, 1071–1078. [Google Scholar] [CrossRef]
- Zakaria, Z.A.; Yob, N.J.; Jofrry, S.M.; Affandi, M.M.R.M.M.; Teh, L.K.; Salleh, M.Z. Zingiber zerumbet (L.) Smith: A review of its ethnomedicinal, chemical, and pharmacological uses. Evid. Based Complement. Altern. Med. 2011, 2011, 543216. [Google Scholar]
- Koga, A.Y.; Beltrame, F.L.; Pereira, A.V. Several aspects of Zingiber zerumbet: A review. Rev. Bras. Farmacogn. 2016, 26, 385–391. [Google Scholar] [CrossRef]
- Datiles, M.J.; Acevedo-Rodríguez, P. Datasheet Report for Zingiber zerumbet (Shampoo Ginger). Available online: https://www.cabi.org/isc/datasheet/57539 (accessed on 21 July 2022).
- Chane-Ming, J.; Vera, R.; Chalchat, J.C. Chemical composition of the essential oil from rhizomes, leaves and flowers of Zingiber zerumbet smith from Reunion Island. J. Essent. Oil Res. 2003, 15, 202–205. [Google Scholar] [CrossRef]
- Azelan, N.A. Optimization of Hydrodistillation Extraction Conditions of Zerumbone from Zingiber zerumbet Rhizome. Master’s Thesis, Universiti Teknologi Malaysia, Johor, Malaysia, 2017. [Google Scholar]
- Rana, V.S.; Ahluwalia, V.; Shakil, N.A.; Prasad, L. Essential oil composition, antifungal, and seedling growth inhibitory effects of zerumbone from Zingiber zerumbet Smith. J. Essent. Oil Res. 2017, 29, 320–329. [Google Scholar] [CrossRef]
- Raina, A.P.; Misra, R.C. Zingiber zerumbet (L.) Roscoe ex Smith: A potential source of zerumbone rich essential oil. Med. Plants-Int. J. Phytomedicines Relat. Ind. 2022, 14, 162–168. [Google Scholar]
- Chavan, J.J.; Kshirsagar, P.R.; Pai, S.R.; Pawar, N.V. Micropropagation, metabolite profiling, antioxidant activities and chromatographic determination of bioactive molecules across in vitro conditions and subsequent field cultivation stages of ‘Shampoo Ginger’ (Zingiber zerumbet L. Roscoe ex Sm). Biocatal. Agric. Biotechnol. 2018, 16, 79–89. [Google Scholar] [CrossRef]
- Dash, B.; Sahoo, A.; Ray, A.; Jena, S.; Nayak, S. Identification of chemical constituents of Zingiber zerumbet rhizome extract using GC/MS. J. Biol. Act. Prod. Nat. 2020, 10, 411–417. [Google Scholar] [CrossRef]
- Lal, M.; Begum, T.; Munda, S.; Pandey, S.K. Identification of high rhizome and essential oil yielding variety (Jor Lab ZB-103) of Zingiber zerumbet (L.) Roscoe ex Sm. J. Essent. Oil-Bear. Plants 2021, 24, 1010–1025. [Google Scholar] [CrossRef]
- Dash, B.; Ray, A.; Sahoo, A.; Kar, B.; Chatterjee, T.; Halder, T.; Chandra Panda, P.; Patnaik, J.; Ghosh, B.; Nayak, S. A combined approach using ISSR and volatile compound analysis for assessment of genetic and phytochemical diversity in Zingiber zerumbet (L.) from Eastern India. J. Essent. Oil-Bear. Plants 2019, 22, 31–49. [Google Scholar] [CrossRef]
- Baby, S.; Dan, M.; Thaha, A.R.M.; Johnson, A.J.; Kurup, R.; Balakrishnapillai, P.; Lim, C.K. High content of zerumbone in volatile oils of Zingiber zerumbet from southern India and Malaysia. Flavour Fragr. J. 2009, 24, 301–308. [Google Scholar] [CrossRef]
- Bhavya, M.L.; Ravi, R.; Madhava Naidu, M. Development and validation of headspace Solid-Phase microextraction coupled with gas chromatography (HS-SPME-GC) method for the analysis of Zingiber zerumbet L. Nat. Prod. Res. 2019, 35, 1221–1225. [Google Scholar] [CrossRef]
- Ghasemzadeh, A.; Jaafar, H.Z.E.; Rahmat, A.; Swamy, M.K. Optimization of microwave-assisted extraction of zerumbone from Zingiber zerumbet L. rhizome and evaluation of antiproliferative activity of optimized extracts. Chem. Cent. J. 2017, 11, 5. [Google Scholar] [CrossRef]
- Paramita, A.; Wibowo, I.; Insanu, M. A study of genus Zingiber: The role of condiments in science. Curr. Res. Biosci. Biotechnol. 2021, 3, 186–195. [Google Scholar] [CrossRef]
- Sivasothy, Y.; Awang, K.; Ibrahim, H.; Thong, K.L.; Fitrah, N.; Koh, X.P.; Tan, L.K. Chemical composition and antibacterial activities of essential oils from Zingiber spectabile Griff. J. Essent. Oil Res. 2012, 24, 305–313. [Google Scholar] [CrossRef]
- Sirat, H.M.; Leh, N.H.N. The rhizome oil of Zingiber spectabile valet. J. Essent. Oil Res. 2001, 13, 256–257. [Google Scholar] [CrossRef]
- Mahardika, R.W.; Ibrahim, H.; Nurulhusna, A.H.; Awang, K. Efficacy of four species of Zingiberaceae extract against vectors of dengue, chikungunya and filariasis. Trop. Biomed. 2017, 34, 375–387. [Google Scholar]
- Sukari, M.A.; Mohd Sharif, N.W.; Yap, A.L.C.; Tang, S.W.; Neoh, B.K.; Rahmani, M.; Ee, G.C.L.; Taufiq-Yap, Y.H.; Yusof, U.K. Chemical constituent variations of essential oils from rhizomes of four Zingiberaceae species. Malays. J. Anal. Sci. 2008, 12, 638–644. [Google Scholar]
- Ariani, S.R.D.; Nurul, S.; Falentina, S. Isolation and identification of essential oils from bitter ginger (Zingiber amaricans BL.), fragrant ginger (Zingiber aromaticum Val.) and shampoo ginger (Zingiber zerumbet (L.) Smith) rhizomes grown in Jumapolo Karaganyar Central Java Indonesia. J. Kim. Dan Pendidik. Kim. 2021, 6, 242–251. [Google Scholar] [CrossRef]
- Devkota, H.P.; Paudel, K.R.; Hassan, M.M.; Dirar, A.I.; Das, N.; Adhikari-Devkota, A.; Echeverría, J.; Logesh, R.; Jha, N.K.; Singh, S.K.; et al. Bioactive compounds from Zingiber montanum and their pharmacological activities with focus on zerumbone. Appl. Sci. 2021, 11, 10205. [Google Scholar] [CrossRef]
- Bora, P.K.; Saikia, J.; Kemprai, P.; Saikia, S.P.; Banik, D.; Haldar, S. Evaluation of postharvest drying, key odorants, and phytotoxins in Plai (Zingiber montanum) essential oil. J. Agric. Food Chem. 2021, 69, 5500–5509. [Google Scholar] [CrossRef]
- Rajkumari, S.; Sanatombi, K. Secondary metabolites content and essential oil composition of in vitro cultures of Zingiber montanum (Koenig) Link ex A. Dietr. Biotechnol. Lett. 2020, 42, 1237–1245. [Google Scholar] [CrossRef] [PubMed]
- Hassan, M.M.; Adhikari-Devkota, A.; Imai, T.; Devkota, H.P. Zerumbone and kaempferol derivatives from the rhizomes of Zingiber montanum (J. Koenig) Link ex A. Dietr. from Bangladesh. Separations 2019, 6, 31. [Google Scholar] [CrossRef]
- Thitinarongwate, W.; Mektrirat, R.; Nimlamool, W.; Khonsung, P.; Pikulkaew, S.; Okonogi, S.; Kunanusorn, P. Phytochemical and safety evaluations of Zingiber ottensii Valeton essential oil in zebrafish embryos and rats. Toxics 2021, 9, 102. [Google Scholar] [CrossRef]
- Ruttanapattanakul, J.; Wikan, N.; Chinda, K.; Jearanaikulvanich, T.; Krisanuruks, N.; Muangcha, M.; Okonogi, S.; Potikanond, S.; Nimlamool, W. Essential oil from Zingiber ottensii induces human cervical cancer cell apoptosis and inhibits MAPK and PI3K/AKT signaling cascades. Plants 2021, 10, 1419. [Google Scholar] [CrossRef]
- Panyajai, P.; Chueahongthong, F.; Viriyaadhammaa, N.; Nirachonkul, W.; Tima, S.; Chiampanichayakul, S.; Anuchapreeda, S.; Okonogi, S. Anticancer activity of Zingiber ottensii essential oil and its nanoformulations. PLoS ONE 2022, 17, e0262335. [Google Scholar] [CrossRef]
- Zakaria, Z.A.; Mohamad, A.S.; Ahmad, M.S.; Mokhtar, A.F.; Israf, D.A.; Lajis, N.H.; Sulaiman, M.R. Preliminary analysis of the anti-inflammatory activity of essential oils of Zingiber Zerumbet. Biol. Res. Nurs. 2011, 13, 425–432. [Google Scholar] [CrossRef] [PubMed]
- Batubara, I.; Suparto, I.H.; Sadiah, S.; Matsuoka, R.; Mitsunaga, T. Effect of Zingiber zerumbet essential oils and zerumbone inhalation on body weight of Sprague dawley rat. Pak. J. Biol. Sci. 2013, 16, 1028–1033. [Google Scholar] [CrossRef]
- Madegowda, B.H.; Rameshwaran, P.; Nagaraju, N.P.; Murthy, P.S. In-vitro mycological activity of essential oil from Zingiber zerumbet rhizomes. J. Essent. Oil Res. 2015, 28, 81–88. [Google Scholar] [CrossRef]
- Sulaiman, M.R.; Tengku Mohamad, T.A.S.; Shaik Mossadeq, W.M.; Moin, S.; Yusof, M.; Mokhtar, A.F.; Zakaria, Z.A.; Israf, D.A.; Lajis, N. Antinociceptive activity of the essential oil of Zingiber Zerumbet. Planta Med. 2010, 76, 107–112. [Google Scholar] [CrossRef]
- Fusi, F.; Durante, M.; Sgaragli, G.; Khanh, P.N.; Son, N.T.; Huong, T.T.; Huong, V.N.; Cuong, N.M. In vitro vasoactivity of zerumbone from Zingiber Zerumbet. Planta Med. 2015, 81, 298–304. [Google Scholar] [CrossRef] [PubMed]
- Huong, L.T.; Chinh, H.V.; An, N.T.G.; Viet, N.T.; Hung, N.H.; Thuong, N.T.H.; Giwa-Ajeniya, A.O.; Ogunwande, I.A. Zingiber zerumbet rhizome essential oil: Chemical composition, antimicrobial and mosquito larvicidal activities. Eur. J. Med. Plants 2020, 3, 2019. [Google Scholar] [CrossRef]
- Sithara, T.; Dhanya, B.P.; Arun, K.B.; Sini, S.; Dan, M.; Kokkuvayil Vasu, R.; Nisha, P. Zerumbone, a cyclic sesquiterpene from Zingiber zerumbet induces apoptosis, cell cycle arrest, and antimigratory effects in SW480 colorectal cancer cells. J. Agric. Food Chem. 2018, 66, 602–612. [Google Scholar] [CrossRef] [PubMed]
- Jantan, I.; Haque, M.A.; Ilangkovan, M.; Arshad, L. Zerumbone from Zingiber zerumbet inhibits innate and adaptive immune responses in Balb/C mice. Int. Immunopharmacol. 2019, 73, 552–559. [Google Scholar] [CrossRef]
- Zhang, L.; Yang, Z.; Wei, J.; Su, P.; Chen, D.; Pan, W.; Zhou, W.; Zhang, K.; Zheng, X.; Lin, L.; et al. Contrastive analysis of chemical composition of essential oil from twelve Curcuma species distributed in China. Ind. Crops Prod. 2017, 108, 17–25. [Google Scholar] [CrossRef]
- Xiang, H.; Zhang, L.; Xi, L.; Yang, Y.; Wang, X.; Lei, D.; Zheng, X.; Liu, X. Phytochemical profiles and bioactivities of essential oils extracted from seven Curcuma herbs. Ind. Crops Prod. 2018, 111, 298–305. [Google Scholar] [CrossRef]
- Thambi, M.; Shafi, M.P. Rhizome essential oil composition of Costus speciosus and its antimicrobial properties. Int. J. Pharm. Res. Allied Sci. 2015, 4, 28–32. [Google Scholar]
- Benelli, G.; Govindarajan, M.; Rajeswary, M.; Vaseeharan, B.; Alyahya, S.A.; Alharbi, N.S.; Kadaikunnan, S.; Khaled, J.M.; Maggi, F. Insecticidal activity of camphene, zerumbone and α-humulene from Cheilocostus speciosus rhizome essential oil against the Old-World bollworm, Helicoverpa armigera. Ecotoxicol. Environ. Saf. 2018, 148, 781–786. [Google Scholar] [CrossRef] [PubMed]
- Arambewela, L.S.R.; Arawwawala, M.; Owen, N.L.; Jarvis, B. Volatile oil of Alpinia galanga Willd. of Sri Lanka. J. Essent. Oil Res. 2007, 19, 455–456. [Google Scholar] [CrossRef]
- Huong, L.T.; Sam, L.N.; Dai, D.N.; Ogunwande, I.A. Investigation into the chemical compositions and antimicrobial activity of essential oil from the rhizomes of Boesenbergia quangngaiensis N.S. Lý from Vietnam. J. Essent. Oil-Bear. Plants 2021, 24, 1125–1133. [Google Scholar] [CrossRef]
- Huong, L.T.; Hung, N.V.; Chung, M.V.; Dai, D.N.; Ogunwande, I.A. Essential oils constituents of the leaves of Amomum gagnepainii and Amomum repoense. Nat. Prod. Res. 2018, 32, 316–321. [Google Scholar] [CrossRef] [PubMed]
- Chang, K.S.; Jeon, J.H.; Kim, G.H.; Jang, C.W.; Jeong, S.J.; Ju, Y.R.; Ahn, Y.J. Repellency of zerumbone identified in Cyperus rotundus rhizome and other constituents to Blattella germanica. Sci. Rep. 2017, 7, 16643. [Google Scholar] [CrossRef] [PubMed]
- Huong, L.T.; Chung, N.T.; Huong, T.T.; Sam, L.N.; Hung, N.H.; Ogunwande, I.A.; Dai, D.N.; Linh, L.D.; Setzer, W.N. Essential oils of Zingiber species from Vietnam: Chemical compositions and biological activities. Plants 2020, 9, 1269. [Google Scholar] [CrossRef]
- Bin Jantan, I.; Mohd Yassin, M.S.; Chin, C.B.; Chen, L.L.; Sim, N.L. Antifungal activity of the essential oils of nine Zingiberaceae species. Pharm. Biol. 2003, 41, 392–397. [Google Scholar] [CrossRef]
- Bucur, L.; Ionus, E.; Moise, G.; Gîrd, C.; Schröder, V. GC-MS analysis and bioactive properties of Zingiberis rhizoma essential oil. Farmacia 2020, 68, 280–287. [Google Scholar] [CrossRef]
- Chairgulprasert, V.; Prasertsongskun, S.; Wichaporn, W. Chemical constituents of the essential oil and anti-bacterial activity of Zingiber wrayi var. halabala. Songklanakarin J. Sci. Technol. 2005, 27, 813–818. [Google Scholar]
- Pintatum, A.; Laphookhieo, S.; Maneerat, W.; Logie, E.; Berghe, W. Vanden Chemical composition of essential oils from different parts of Zingiber kerrii Craib and their antibacterial, antioxidant, and tyrosinase inhibitory activities. Biomolecules 2020, 10, 228. [Google Scholar] [CrossRef]
- Shukurova, M.K.; Asikin, Y.; Chen, Y.; Kusano, M.; Watanabe, K.N. Profiling of volatile organic compounds in wild indigenous medicinal ginger (Zingiber barbatum Wall.) from Myanmar. Metabolites 2020, 10, 248. [Google Scholar] [CrossRef] [PubMed]
- Mahboubi, M. Zingiber officinale Rosc. essential oil, a review on its composition and bioactivity. Clin. Phytosci. 2019, 5, 6. [Google Scholar] [CrossRef]
- Sommano, S.R.; Tangpao, T. Aromatic profile of rhizomes from the ginger family used in food. In Aromatic Herbs in Food. Bioactive Compounds, Processing, and Applications; Galanakis, C.M., Ed.; Elsevier: London, UK, 2021; pp. 123–165. ISBN 9780128227169. [Google Scholar]
- CAB. Curcuma. Available online: https://www.cabi.org/ISC/datasheet/17013 (accessed on 23 July 2022).
- Kristianto, S.; Isrianto, P.L.; Marmi, M.; Chamidah, D.; Khan, A.U. Screening phytochemical and study insilico of family Zingiberaceae as anti-inflammatory. J. Biota 2022, 8, 95–114. [Google Scholar] [CrossRef]
- Peng, W.; Li, P.; Ling, R.; Wang, Z.; Feng, X.; Liu, J.; Yang, Q.; Yan, J. Diversity of volatile compounds in ten varieties of Zingiberaceae. Molecules 2022, 27, 565. [Google Scholar] [CrossRef]
- Abdelkader, N.F.; Moustafa, P.E. Rhizomatous plants: Curcuma longa and Zingiber officinale in affording immunity. In Plants and Phytomolecules for Immunomodulation; Sangwan, N.S., Farag, M.A., Modolo, L.V., Eds.; Springer Nature: Singapore, 2022; pp. 361–390. ISBN 9789811681172. [Google Scholar]
- Kshirsagar, P.R.; Pawar, N.V.; Patil, S.P.; Nimbalkar, M.S.; Pai, S.R. Quantification of zerumbone in eleven accessions of Curcuma longa using RP-HPLC. Indian J. Pharm. Sci. 2018, 80, 1159–1165. [Google Scholar] [CrossRef]
- Duong, L.; Mentreddy, S.R.; Satyal, R.; Satyal, P.; Setzer, W.N. Essential oil chemotypes of four vietnamese Curcuma species cultivated in North Alabama. Horticulturae 2022, 8, 360. [Google Scholar] [CrossRef]
- Jena, S.; Ray, A.; Sahoo, A.; Panda, P.C.; Nayak, S. Deeper insight into the volatile profile of essential oil of two Curcuma species and their antioxidant and antimicrobial activities. Ind. Crops Prod. 2020, 155, 112830. [Google Scholar] [CrossRef]
- Gharge, S.; Hiremath, S.I.; Kagawad, P.; Jivaje, K.; Palled, M.S.; Suryawanshi, S.S. Curcuma zedoaria Rosc (Zingiberaceae): A review on its chemical, pharmacological and biological activities. Future J. Pharm. Sci. 2021, 7, 166. [Google Scholar] [CrossRef]
- Hamdi, O.A.A.; Ye, L.J.; Kamarudin, M.N.A.; Hazni, H.; Paydar, M.; Looi, C.Y.; Shilpi, J.A.; Kadir, H.A.; Awang, K. Neuroprotective and antioxidant constituents from Curcuma zedoaria rhizomes. Rec. Nat. Prod. 2015, 9, 349–355. [Google Scholar]
- Al-Amin, M.; Eltayeb, N.M.; Khairuddean, M.; Salhimi, S.M. Bioactive chemical constituents from Curcuma caesia Roxb. rhizomes and inhibitory effect of curcuzederone on the migration of triple-negative breast cancer cell line MDA-MB-231. Nat. Prod. Res. 2021, 35, 3166–3170. [Google Scholar] [CrossRef] [PubMed]
- Rangsiruji, A.; Newman, M.F.; Cronk, Q.C.B. Origin and relationships of Alpinia galanga (Zingiberaceae) based on molecular data. Edinb. J. Bot. 2000, 57, 9–37. [Google Scholar] [CrossRef]
- Kress, W.J.; Liu, A.Z.; Newman, M.; Qing-Jun, L.I. The molecular phylogeny of Alpinia (Zingiberaceae): A complex and polyphyletic genus of gingers. Am. J. Bot. 2005, 92, 167–178. [Google Scholar] [CrossRef] [PubMed]
- Van, H.T.; Thang, T.D.; Luu, T.N.; Doan, V.D. An overview of the chemical composition and biological activities of essential oils from: Alpinia genus (Zingiberaceae). RSC Adv. 2021, 11, 37767–37783. [Google Scholar] [CrossRef] [PubMed]
- Ramanunny, A.K.; Wadhwa, S.; Gulati, M.; Vishwas, S.; Khursheed, R.; Paudel, K.R.; Gupta, S.; Porwal, O.; Alshahrani, S.M.; Jha, N.K.; et al. Journey of Alpinia galanga from kitchen spice to nutraceutical to folk medicine to nanomedicine. J. Ethnopharmacol. 2022, 291, 115144. [Google Scholar] [CrossRef]
- Mallavarapu, G.R.; Rao, L.; Ramesh, S.; Dimri, B.P.; Bhaskaruni, R.; Rajeswara, R.; Kaul, P.N.; Bhattacharya, A.K. Composition of the volatile oils of Alpina galanga rhizomes and leaves from India. J. Essent. Oil Res. 2002, 14, 397–399. [Google Scholar] [CrossRef]
- Rana, V.S.; Verdeguer, M.; Blazquez, M.A. GC and GC/MS analysis of the volatile constituents of the oils of Alpinia galanga (L.) Willd and A. officinarum Hance rhizomes. J. Essent. Oil Res. 2010, 22, 521–524. [Google Scholar] [CrossRef]
- Huang, G.C.; Kao, C.L.; Li, W.J.; Huang, S.T.; Li, H.T.; Chen, C.Y. A new phenylalkanoid from the rhizomes of Alpinia galanga. Chem. Nat. Compd. 2018, 54, 1072–1075. [Google Scholar] [CrossRef]
- Bhatt, G.; Nagarkoti, R.S.; Kholiya, S.; Tiwari, A.; Verma, S.K.; Verma, R.S.; Darokar, M.P.; Padalia, R.C. Chemical and antibacterial activity evaluation of Alpinia calcarata and Alpinia zerumbet grown in foothills agroclimatic conditions of Northern India. Open Bioact. Compd. J. 2021, 9, 15–19. [Google Scholar] [CrossRef]
- Feng, Y.X.; Zhang, X.; Wang, Y.; Chen, Z.Y.; Lu, X.X.; Du, Y.S.; Du, S.S. The potential contribution of cymene isomers to insecticidal and repellent activities of the essential oil from Alpinia zerumbet. Int. Biodeterior. Biodegrad. 2021, 157, 105138. [Google Scholar] [CrossRef]
- Vanijajiva, O.; Sirirugsa, P.; Suvachittanont, W. Confirmation of relationships among Boesenbergia (Zingiberaceae) and related genera by RAPD. Biochem. Syst. Ecol. 2005, 33, 159–170. [Google Scholar] [CrossRef]
- Nor, N.A.; Ibrahim, H. Chemical constituents of essential oils of Boesenbergia armeniaca. Pak. J. Biotechnol. 2018, 50, 1917–1922. [Google Scholar]
- Zubair, M.S.; Khairunisa, S.Q.; Widodo, A.; Nasronudin; Pitopang, R. Antiviral screening on Alpinia eremochlamys, Etlingera flexuosa, and Etlingera acanthoides extracts against HIV-infected MT-4 cells. Heliyon 2021, 7, e06710. [Google Scholar] [CrossRef] [PubMed]
- Lim, T.K. Edible Medicinal and Non-Medicinal Plants; Springer: Dordrecht, The Netherlands, 2014; Volume 7, Flowers; ISBN 978-94-007-7394-3. [Google Scholar]
- Kashiwazaki, G.; Watanabe, R.; Tsuzuki, T.; Yamamoto, C.; Nishikawa, A.; Ohtomo, S.; Yoshikawa, T.; Kitamura, Y.; Utaka, Y.; Kawai, Y.; et al. Brønsted acid-induced transannulation of the phytochemical zerumbone. Org. Biomol. Chem. 2021, 19, 10444–10454. [Google Scholar] [CrossRef]
- Kitayama, T.; Okamoto, T.; Hill, R.K.; Kawai, Y.; Takahashi, S.; Yonemori, S.; Yamamoto, Y.; Ohe, K.; Uemura, S.; Sawada, S. Chemistry of Zerumbone. 1. Simplified isolation, conjugate addition reactions, and a unique ring contracting transannular reaction of its dibromide. J. Org. Chem. 1999, 64, 2667–2672. [Google Scholar] [CrossRef] [PubMed]
- Vu, V.V.; Vu, T.K. Synthesis of new zerumbone hydrazones and their in-vitro anticancer activity. Curr. Bioact. Compd. 2020, 17, 331–338. [Google Scholar] [CrossRef]
- Santosh Kumar, S.C.; Srinivas, P.; Negi, P.S.; Bettadaiah, B.K. Antibacterial and antimutagenic activities of novel zerumbone analogues. Food Chem. 2013, 141, 1097–1103. [Google Scholar] [CrossRef]
- Biji, M.; Prabha, B.; Lankalapalli, R.S.; Radhakrishnan, K.V. Transition metal/Lewis acid catalyzed reactions of zerumbone for diverse molecular motifs. Chem. Rec. 2021, 21, 3943–3953. [Google Scholar] [CrossRef] [PubMed]
- Nandagopal, K.; Halder, M.; Dash, B.; Nayak, S.; Jha, S. Biotechnological approaches for production of anti-cancerous compounds resveratrol, podophyllotoxin and zerumbone. Curr. Med. Chem. 2018, 25, 4693–4717. [Google Scholar] [CrossRef]
- Keerthi, D.; Geethu, C.; Nair, R.A.; Pillai, P. Metabolic profiling of Zingiber zerumbet following Pythium myriotylum infection: Investigations on the defensive role of the principal secondary metabolite, zerumbone. Appl. Biochem. Biotechnol. 2014, 172, 2593–2603. [Google Scholar] [CrossRef]
- Doehlemann, G.; Ökmen, B.; Zhu, W.; Sharon, A. Plant pathogenic fungi. Microbiol. Spectr. 2017, 5, FUNK-0023-2016. [Google Scholar] [CrossRef]
- Salvatore, M.M.; Andolfi, A. Phytopathogenic fungi and toxicity. Toxins 2021, 13, 689. [Google Scholar] [CrossRef] [PubMed]
- Sheikh-Ali, S.I.; Ahmad, A.; Mohd-Setapar, S.H.; Zakaria, Z.A.; Abdul-Talib, N.; Khamis, A.K.; Hoque, M.E. The potential hazards of Aspergillus sp. in foods and feeds, and the role of biological treatment: A review. J. Microbiol. 2014, 52, 807–818. [Google Scholar] [CrossRef] [PubMed]
- Ráduly, Z.; Szabó, L.; Madar, A.; Pócsi, I.; Csernoch, L. Toxicological and medical aspects of Aspergillus-derived mycotoxins entering the feed and food chain. Front. Microbiol. 2020, 10, 2908. [Google Scholar] [CrossRef] [PubMed]
- Navale, V.; Vamkudoth, K.R.; Ajmera, S.; Dhuri, V. Aspergillus derived mycotoxins in food and the environment: Prevalence, detection, and toxicity. Toxicol. Rep. 2021, 8, 1008–1030. [Google Scholar] [CrossRef] [PubMed]
- Dousset, X.; Jaffrès, E.; Zagorec, M. Spoilage: Bacterial spoilage. Encycl. Food Health 2015, 48, 106–112. [Google Scholar]
- Lorenzo, J.M.; Munekata, P.E.; Dominguez, R.; Pateiro, M.; Saraiva, J.A.; Franco, D. Main groups of microorganisms of relevance for food safety and stability: General aspects and overall description. In Innovative Technologies for Food Preservation: Inactivation of Spoilage and Pathogenic Microorganisms; Barba, F.J., Sant’Ana, A.S., Orlien, V., Koubaa, M., Eds.; Elsevier Inc.: Amsterdam, The Netherlands, 2018; pp. 53–107. ISBN 9780128110324. [Google Scholar]
- Pandey, A.K.; Silva, A.S.; Varshney, R.; Chávez-González, M.L.; Singh, P. Curcuma-based botanicals as crop protectors: From knowledge to application in food crops. Curr. Res. Biotechnol. 2021, 3, 235–248. [Google Scholar] [CrossRef]
- Arnason, J.T.; Sims, S.R.; Scott, I.M. Natural Products from Plants as Insecticides. UNESCO—Encyclopedia Life Support Systems (UNESCO-EOLSS). Available online: http://www.eolss.net/sample-chapters/c06/e6-151-13.pdf (accessed on 3 November 2022).
- Pavela, R. History, presence and perspective of using plant extracts as commercial botanical insecticides and farm products for protection against insects-a review. Czech Acad. Agric. Sci. 2016, 52, 229–241. [Google Scholar]
- Isman, M.B. Botanical Insecticides in the twenty-first century-fulfilling their. Annu. Rev. Entomol. 2020, 65, 233–249. [Google Scholar] [CrossRef]
- Shaaya, E.; Kostjukovski, M.; Eilberg, J.; Sukprakarn, C. Plant oils as fumigants and contact insecticides for the control of stored-product insects. J. Stored Prod. Res. 1997, 33, 7–15. [Google Scholar] [CrossRef]
- Pavela, R. Possibilities of botanical insecticide exploitation in plant protection. Pest Technol. 2007, 1, 47–52. [Google Scholar]
- Chauhan, B.S. Grand challenges in weed management. Front. Agron. 2020, 1, 3. [Google Scholar] [CrossRef]
- Vilà, M.; Beaury, E.M.; Blumenthal, D.M.; Bradley, B.A.; Early, R.; Laginhas, B.B.; Trillo, A.; Dukes, J.S.; Sorte, C.J.B.; Ibáñez, I. Understanding the combined impacts of weeds and climate change on crops. Environ. Res. Lett. 2021, 16, 034043. [Google Scholar] [CrossRef]
- Tataridas, A.; Kanatas, P.; Chatzigeorgiou, A.; Zannopoulos, S.; Travlos, I. Sustainable crop and weed management in the era of the EU green deal: A survival guide. Agronomy 2022, 12, 589. [Google Scholar] [CrossRef]
- Fierascu, R.C.; Fierascu, I.C.; Dinu-Pirvu, C.E.; Fierascu, I.; Paunescu, A. The application of essential oils as a next-generation of pesticides: Recent developments and future perspectives. Z. Naturforsch. Sect. C J. Biosci. 2020, 75, 183–204. [Google Scholar] [CrossRef] [PubMed]
- Bhovi, V.K. Plants based materials as the antifungal and antibacterial agents. Int. J. Plant Based Pharm. 2022, 2, 98–110. [Google Scholar]
- Subhash, S.; Raghavendra, K.V.; Balodi, R.; Deepika; Dubey, N.K. Use of green chemicals in pest and disease management. In Sustainable Management of Potato Pests and Diseases; Kumar Chakrabarti, S., Sharma, S., Shah, M.A., Eds.; Springer: Berlin/Heidelberg, Germany, 2022; pp. 495–524. ISBN 9789811676949. [Google Scholar]
- Alwakil, N.H.; Mohamad Annuar, M.S.; Jalil, M. Synergistic effects of plant growth regulators and elicitors on α-humulene and zerumbone production in Zingiber zerumbet Smith adventitious root cultures. Molecules 2022, 27, 4744. [Google Scholar] [CrossRef]
- Gupta, T.; Singh, J.; Kaur, S.; Sandhu, S.; Singh, G.; Kaur, I.P. Enhancing bioavailability and stability of curcumin using solid lipid nanoparticles (CLEN): A covenant for its effectiveness. Front. Bioeng. Biotechnol. 2020, 8, 879. [Google Scholar] [CrossRef]
- Kurien, B.T.; Scofield, R.H. Increasing aqueous solubility of curcumin for improving bioavailability. Trends Pharmacol. Sci. 2009, 30, 334–335. [Google Scholar] [CrossRef]
- Kesharwani, S.S.; Jayarama Bhat, G. Formulation and nanotechnology-based approaches for solubility and bioavailability enhancement of zerumbone. Medicina 2020, 56, 557. [Google Scholar] [CrossRef]
- Eid, E.E.M.; Abdul, A.B.; Suliman, F.E.O.; Sukari, M.A.; Rasedee, A.; Fatah, S.S. Characterization of the inclusion complex of zerumbone with hydroxypropyl-β-cyclodextrin. Carbohydr. Polym. 2011, 83, 1707–1714. [Google Scholar] [CrossRef]
- Rahman, H.S.; Rasedee, A.; Othman, H.H.; Chartrand, M.S.; Namvar, F.; Yeap, S.K.; Abdul Samad, N.; Andas, R.J.; Muhammad Nadzri, N.; Anasamy, T.; et al. Acute toxicity study of zerumbone-loaded nanostructured lipid carrier on BALB/c mice model. Biomed Res. Int. 2014, 2014, 15. [Google Scholar] [CrossRef] [PubMed]
- Salleh, N.; Mahat1, M.M.; Yahaya, S.M.; Rosmamuhamadani, R. Synthesis and characterization of cross-linked zerumbone loaded zeolite Y-gelatin for oral controlled release. AIP Conf. Proc. 2020, 2291, 020063. [Google Scholar]
- Salleh, N.; Jais, U.; Sarijo, S. Characterization of crosslinked zerumbone loaded gelatin–zeolite Y hybrid composites with glutaraldehyde for controlled release of natural anticancer drug. Malays. J. Anal. Sci. 2015, 19, 1290–1296. [Google Scholar]
- Brouwers, J.; Brewster, M.E.; Augustijns, P. Supersaturating drug delivery systems: The answer to solubility-limited oral bioavailability? J. Pharm. Sci. 2009, 98, 2549–2572. [Google Scholar] [CrossRef] [PubMed]
- Sidahmed, H.M.A.; Hashim, N.M.; Abdulla, M.A.; Ali, H.M.; Mohan, S.; Abdelwahab, S.I.; Taha, M.M.E.; Fai, L.M.; Vadivelu, J. Antisecretory, gastroprotective, antioxidant and anti-Helicobacter pylori activity of zerumbone from Zingiber zerumbet (L.) smith. PLoS ONE 2015, 10, e0121060. [Google Scholar] [CrossRef] [PubMed]
- Tian, M.; Hong, Y.; Wu, X.; Zhang, M.; Lin, B.; Zho, Y. Chemical constituents and cytotoxic activities of essential oils from the flowers, leaves and stems of Zingiber striolatum diels. Rec. Nat. Prod. 2019, 14, 144–149. [Google Scholar] [CrossRef]
- Bhatt, D.A.; Pethe, A. Lipid technology—A promising drug delivery system for poorly water soluble drugs. Int. J. Pharma Res. 2010, 3, 1748–1751. [Google Scholar]
- Hosseinpour, M.; Abdul, A.B.; Rahman, H.S.; Rasedee, A.; Yeap, S.K.; Ahmadi, N.; Othman, H.H.; Chartrand, M. Comparison of apoptotic inducing effect of zerumbone and zerumbone-loaded nanostructured lipid carrier on human mammary adenocarcinoma MDA-MB-231 cell line. J. Nanomater. 2014, 182, 742738. [Google Scholar] [CrossRef]
- Khan, A.A.; Mudassir, J.; Mohtar, N.; Yusrida, D. Advanced drug delivery to the lymphatic system: Lipid-based nanoformulations. Int. J. Nanomed. 2013, 8, 2733–2744. [Google Scholar]
- Abdul, A.B.H.; Al-Zubairi, A.S.; Tailan, N.D.; Wahab, S.I.A. Anticancer activity of natural compound (zerumbone) extracted from Zingiber zerumbet in human HeLa cervical cancer cells. Int. J. Pharmacol. 2008, 4, 160–168. [Google Scholar] [CrossRef]
- Rahman, H.S.; Rasedee, A.; Yeap, S.K.; Othman, H.H.; Chartrand, M.S.; Namvar, F.; Abdul, A.B.; How, C.W. Biomedical properties of a natural dietary plant metabolite, zerumbone, in cancer therapy and chemoprevention trials. BioMed Res. Int. 2014, 2014, 20. [Google Scholar] [CrossRef] [PubMed]

| Plant Species | Plant Part | Origin | Method of Extraction | Analysis | Yield | Main Components | Activity | Ref. |
|---|---|---|---|---|---|---|---|---|
| Zingiber zerumbet | Rhizome | India | Methanol extraction | RP-HPLC-PDA | Zerumbone (33.02 mg/g) | Antioxidant | [42] | |
| Rhizomes | India | GC/FID- GC/MS | Zerumbone (70.60%) α-Humulene (5.65%) Humulene epoxide I (5.21%) Humulene epoxide II (5.71%) Camphor (1.90%) Camphene (3.47%) Caryophyllene oxide (2.52%) | Antioxidant | [41] | |||
| Rhizomes | India | Hydrodistillation Clevenger | GC- GC/MS | 0.45% | Zerumbone (74.82%) Humulene (6.02%) β-Copaen-4α-ol (4.32%) | Antifungal | [40] | |
| Rhizomes | Brazil | Hydrodistillation Clevenger | GC/MS | 5% | Zerumbone (87.93%) | Antibacterial | [3] | |
| Rhizomes | Malaysia | Hydrodistillation | 0.25% | Anti-inflammatory | [62] | |||
| Rhizomes | Indonesia | Water distillation | GC/MS | 0.12% | Sabinene (32.96%) β-Myrcene (13.27%) Zerumbone (11.05%) | Increase body weight | [63] | |
| Rhizomes | Malaysia | Hydrodistillation Turbo Extractor Distillator | HPLC | 0.35% | Zerumbone (126.54 mg/mL) | Antibacterial | [39] | |
| Rhizomes | Reunion Island | Steam distillation | GC-GC/MS- GC/FTIR | 0.3–0.4% | Zerumbone (36%) α-Humulene (14.4%) Camphene (13.8%) Caryophyllene oxide (5.2%) Camphor (3.8%) 1,8-Cineole (3.2%) | [38] | ||
| Rhizomes | Malaysia | Ethanol extraction | HPLC | Zerumbone (242.73 mg/g) | Immunosuppressant | [8] | ||
| Rhizomes | Malaysia | Microwave | UHPLC | Zerumbone (4.82 mg/g DM) | Antiproliferative | [48] | ||
| Rhizomes | India | Methanol extraction | RP-HPLC | Zerumbone (15.598–30.143 mg/g) | Antioxidant | [42] | ||
| Fresh rhizomes | China | Hydrodistillation Clevenger | GC/MS– GC/FID | Zerumbone (40.2%) α-Caryophyllene (8.6%) Humulene epoxide II (7.3%) Camphene (5.9%) | Insecticidal, repellent | [10] | ||
| Fresh rhizomes | China | Hydrodistillation Clevenger | GC-FID/MS | 0.65% | Zerumbone (75.0%) α-Humulene (6.5%) Humulene oxide I (3.8%) Camphene (3.3%) Humulene oxide II (2.7%) Camphor (1.3%) Caryophyllene oxide (1.3%) 1,8-Cineole (1.2%) | Antibacterial, cytotoxic | [6] | |
| Fresh rhizomes | India | Hydrodistillation Clevenger | GC/MS | 1.12% | Zerumbone (49.3%) α-Caryophyllene (20.1%) Z-Caryophyllene (3.8%) | Antifungal, antimycotoxin | [64] | |
| Fresh rhizomes | Malaysia | Hydrodistillation | GC-FID/MS | 0.25% | Zerumbone (36.12%) Humulene (10.03%) Humulene oxide I (4.08%) Humulene oxide II (2.14%) Caryophyllene oxide II (1.66%) Caryophyllene oxide I (1.43%) | Antinociceptive | [65] | |
| Fresh rhizomes | Vietnam | Steam distillation | HPLC | 0.1% | Zerumbone (98%) | Weak in vitro vasodilator | [66] | |
| Fresh rhizomes | India | GC/MS | 0.75% | Zerumbone (32.79%) Camphene (19.41%) Eucalyptol (6.80%) | [44] | |||
| Dried rhizomes | China | Hydrodistillation Clevenger | GC-FID/MS | 0.39% | Zerumbone (41.9%) α-Humulene (29.4%) Humulene oxide I (6.0%) Humulene oxide II (3.9%) Camphene (3.9%) β-Caryophyllene (2.5%) Camphor (2.4%) Caryophyllene oxide (2.1%) 1,8-Cineole (1.2%) | Antimicrobial, cytotoxic | [6] | |
| Dried rhizomes | Malaysia | Hydrodistillation Clevenger | GC-GC/MS | 0.37% | Zerumbone (58.44%) α-Humulene (12.24%) Camphene (5.36%) | Immunosuppressant | [8] | |
| Air-dry rhizomes | Vietnam | Hydrodistillation Clevenger | GC/MS | 0.65% | Zerumbone (51.3%) Humulene epoxide I (6.4%) Humulene epoxide II (5.5%) α-Humulene (5.4%) Camphene (4.1%) 1,8-Cineole (3.2%) | Insecticidal, antifungal | [67] | |
| Powdered rhizomes | India | Acetone extraction | HPLC | Zerumbone (99.94%) | Anticancer | [68] | ||
| Dried powdered rhizomes | India | Acetone extraction Soxhlet | GC/MS | 2.86% | Zerumbone (64.58%) Diacetone alcohol (10.64%) α-Humulene (8.93%) Caryophyllene oxide (5.68%) Humulene epoxide (3.18%) | [43] | ||
| Powdered rhizomes | Malaysia | Ethanol extraction | HRESI/MS | Zerumbone (87.4 mg) | Immunosuppressant | [69] | ||
| Roots | India | Methanol extraction | RP-HPLC-PDA | Zerumbone (05.562 mg/g) | Antioxidant | [42] | ||
| Z. zerumbet var. darcyi | Fresh rhizomes | India | Hydrodistillation Clevenger | GC-GC/MS | 0.23% | Zerumbone (69.9%) α-Humulene (12.9%) Humulene epoxide II (2.5%) Caryophyllene oxide (1.1%) Camphene (1.9%) | [9] | |
| Z. ottensii | Fresh rhizome | Thailand | Hydrodistillation Clevenger | GC/MS | 0.24% | Zerumbone (24.73%) Terpinen-4-ol (18.75%) Sabinene (15.19%) β-Pinene (7.95%) | Cytotoxic | [59] |
| Fresh rhizomes | Thailand | Hydrodistillation | GC/MS | 0.21% | Zerumbone (25.21%) Sabinene (23.35%) Terpene-4-ol (15.97%) | Apoptotic | [61] | |
| Z. montanum | Dried rhizomes | Bangladesh | Ethanol extraction | 1H-NMR/13C-NMR | ZerumboneFlavonoid derivatives | [58] | ||
| Z. amaricans | Malaysia | Hydrodistillation | GC-MS | Zerumbone (40.70%) | [53] | |||
| Powdered dried rhizomes | Indonesia | Steam distillation | GC-MS | 1.6% | Zerumbone (65.06%) Humulene oxide (9.66%) α-Humulene (9.41%) | [54] | ||
| Z. aromaticum | Powdered dried rhizomes | Indonesia | Steam distillation | GC-MS | 2.8% | Zerumbone (31.45%) Cyclohexene (13.52%) Isogeraniol (10.52%) | [54] | |
| Curcuma rubescens | Fresh rhizomes | China | Steam distillation | GC/MS | 4.36% | Zerumbone (6.88%) Germacrone (4.99%) | [70] | |
| Powdered dried rhizomes | China | Hydrodistillation Clevenger | GC/MS | Zerumbone (15.45%) ar-Turmerone (13.80%) | Antimicrobial, antioxidant, anticancer, anti-inflammatory | [71] | ||
| Cheilocostus speciosus | Rhizomes | India | Steam distillation | GC-MS/FID | 0.18 g | Zerumbone (55.11%) α-Humulene (20.55%) | Anticancer, antibacterial | [72] |
| Fresh rhizomes | India | Hydrodistillation Clevenger | GC/MS | 1.9 mL/kg | Zerumbone (38.6%) α-Humulene (14.5%) Camphene (9.3%) | Insecticidal | [73] | |
| Alpinia galanga | Dried crushed rhizomes | Sri Lanka | Hydrodistillation Clevenger | GC/MS | 0.56% | Zerumbone (44.9%) | [74] | |
| Boesenbergia quangngaiensis | Rhizomes | Vietnam | Hydrodistillation | GC/MS | 0.16% | cis-β-Elemene (18.4%) Zerumbone (11.40%) Myrtenyl acetate (10.6%) | Antimicrobial | [75] |
| Amomum gagnepainii | Leaves | Vietnam | Hydrodistillation | GC-MS/FID | 0.20% | Farnesyl acetate (18.5%) Zerumbone (16.4%) β-Caryophyllene (10.5%) | [76] | |
| Cyperus rotundus | Air-dried rhizomes | South Korea | Methanolextraction | EI-MS/1H-NMR/13C-NMR | Zerumbone 1,8-Cineole | Insecticidal, repellent | [77] |
| Technique | Polymer/Matrix | Solubility Enhancement | Key Results | Reference |
|---|---|---|---|---|
| Inclusion complexes | HP-β-CyD | >30 fold (From 0.0053 mM for free zerumbone to 0.173 mM with 0.01 M HPβCyD at 20 °C) | Molecular modelling calculations showed that zerumbone penetrates completely into the cavity of the HPβCyD. Complexation efficiency zerumbone-HPβCyD was of 1.04. In vitro cell survival assay on human cervical cancer (Hela), breast cancer (MCF7 and MDA-MB 231) and human leukemic (CEMss) cell lines. | [139] |
| Inclusion complexes | HP-β-CyD | - | Clinical study conducted on three groups of 220 patients each) to treat acute and chronic gastritis. Patients injected with 20 µM of zerumbone. | [34] |
| Inclusion complexes | HP-β-CyD | 8 mM of zerumbone dissolved in 50 mM of HP-β-CyD 18 mM zerumbone dissolved in 18 mM of SBE-β-CyD | CyD derivatives are useful to maintain the supersaturation state of zerumbone | |
| SBE-β-CyD | [33] | |||
| Nanostructured lipid carrier | Palm oil:Olive oil:Lipoid S100 (7:3:3) | Entrapment efficiency of 99.03% Drug loading of 7.92%. | Half maximal inhibitory concentration (IC50) of ZER-NLC was 5.64 ± 0.38 μg/mL (free zerumbone IC50 was 5.39 ± 0.43 μg/mL) after 72 h of treatment. Zerumbone released after 48 h from NLC was 46.7% (0.95 mg) vs. 90.59% (1.81 mg) from pure drug dispersion. In vitro cytotoxicity tests in Jurkat T-cell line. | [31] |
| Nanostructured lipid carriers | Palm oil:Olive oil:Lipoid S100 (7:3:3) | Entrapment efficiency of 99% | Acute toxicity study for zerumbone-NLC conducted orally in BALB/c mice (single dose for 14 days. Oral doses of 100 and 200 mg/kg showed no sign of toxicity or mortality. LD50 is higher than 200 mg/kg therefore safe by oral administration. | [140] |
| Nanostructured lipid carriers | Palm oil:Olive oil:Lipoid S100 (7:3:3) | Entrapment efficiency of 99% | Release of environ 50% after 48 h. In vivo tests conducted on mice challenged with 4T1 breast cancer. | [29] |
| Pore Encapsulation | Zeolite Y-gelatin | Entrapment efficiency tests were here performed by soaking 2% w/v zeolite Y in 5, 10 and 15% aqueous solution of 100 µM zerumbone. EE% of 93.7, 97.3 and 99.2% for the zerumbone concentration of 5, 10 and 15% respectively. | In vitro controlled release tests confirm the interest of this system for oral administration | [141,142] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ibáñez, M.D.; Sánchez-Ballester, N.M.; Blázquez, M.A. Healthy Zerumbone: From Natural Sources to Strategies to Improve Its Bioavailability and Oral Administration. Plants 2023, 12, 5. https://doi.org/10.3390/plants12010005
Ibáñez MD, Sánchez-Ballester NM, Blázquez MA. Healthy Zerumbone: From Natural Sources to Strategies to Improve Its Bioavailability and Oral Administration. Plants. 2023; 12(1):5. https://doi.org/10.3390/plants12010005
Chicago/Turabian StyleIbáñez, María Dolores, Noelia M. Sánchez-Ballester, and María Amparo Blázquez. 2023. "Healthy Zerumbone: From Natural Sources to Strategies to Improve Its Bioavailability and Oral Administration" Plants 12, no. 1: 5. https://doi.org/10.3390/plants12010005
APA StyleIbáñez, M. D., Sánchez-Ballester, N. M., & Blázquez, M. A. (2023). Healthy Zerumbone: From Natural Sources to Strategies to Improve Its Bioavailability and Oral Administration. Plants, 12(1), 5. https://doi.org/10.3390/plants12010005






