Short-Term Effects of Trans-Cinnamic Acid on the Metabolism of Zea mays L. Roots
Abstract
1. Introduction
2. Results
2.1. Xylem Measurements
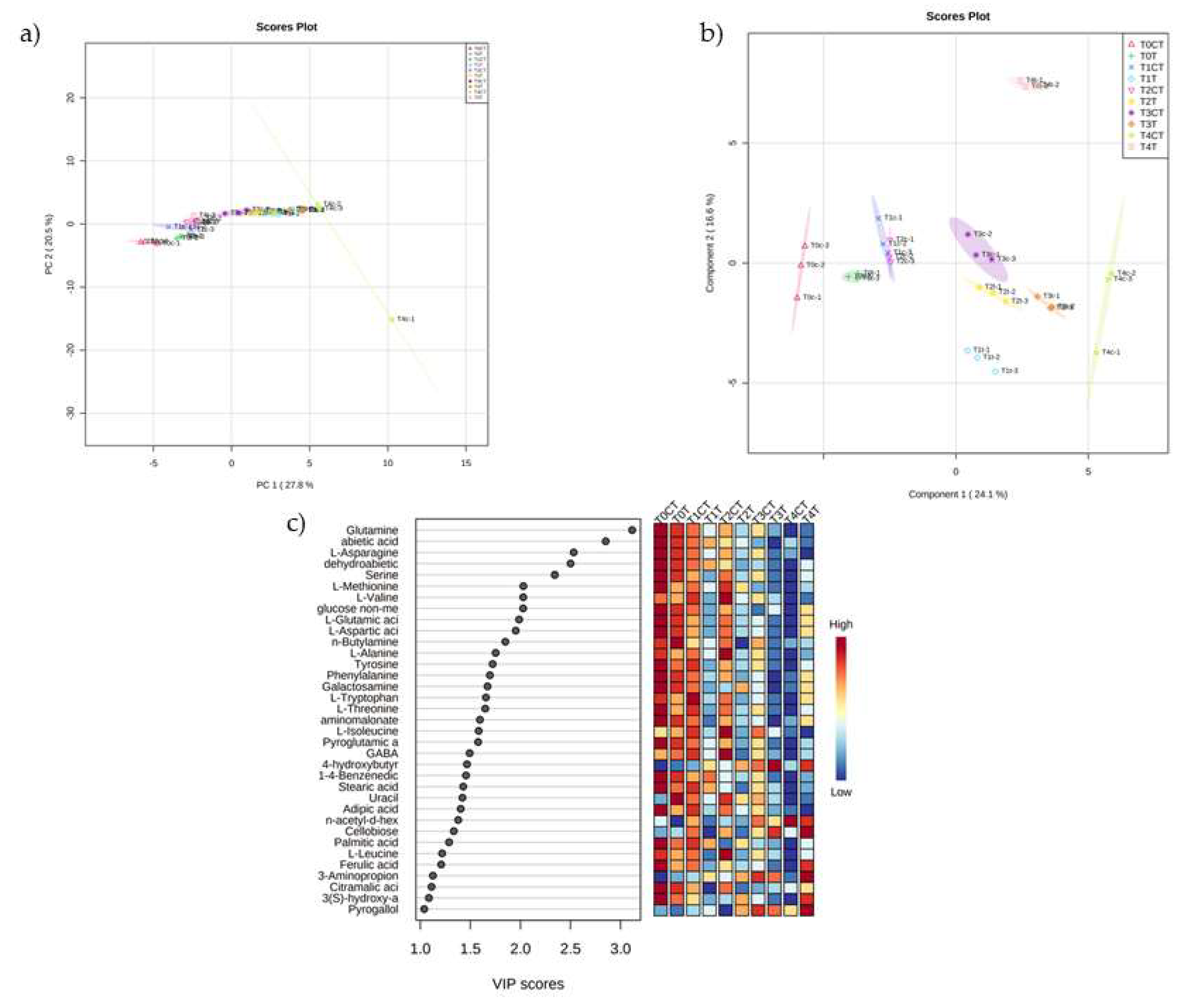
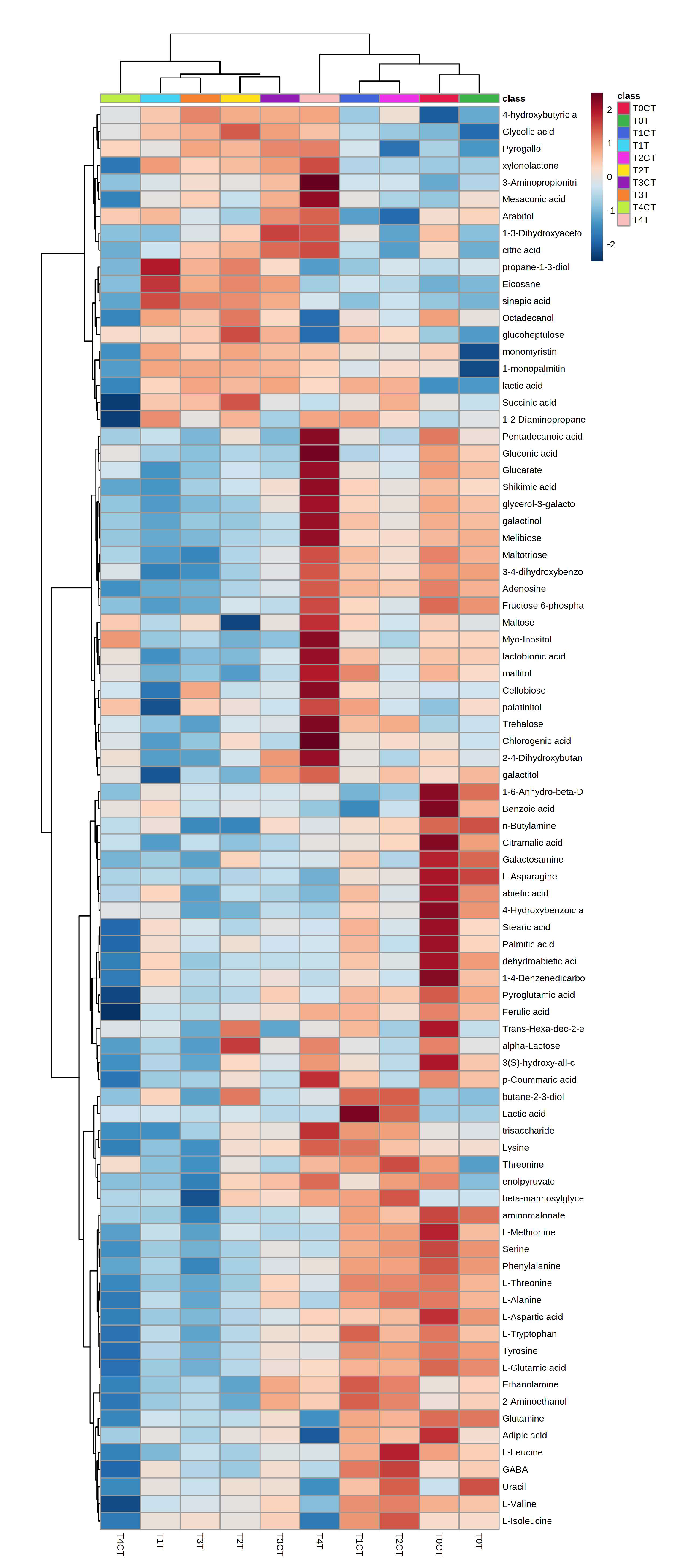
2.2. Untargeted Metabolomic Analysis
3. Discussion
4. Conclusions
5. Materials and Methods
5.1. Plant Material, Growth Conditions and Treatment
5.2. Measurement of Xylem Area
5.3. Untargeted Metabolomic Analysis
5.4. GC-Quadrupole/MS Analysis
5.5. Analysis of GS-MS Data by MS-DIAL
5.6. Statistical Analysis
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Farooq, M.; Jabran, K.; Cheema, Z.A.; Wahid, A.; Siddique, K.H. The role of allelopathy in agricultural pest management. Pest. Manag. Sci. 2011, 67, 493–506. [Google Scholar] [CrossRef]
- Shirgapure, K.H.; Ghosh, P. Allelopathy a tool for sustainable weed management. Arch. Curr. Res. Int. 2020, 20, 17–25. [Google Scholar] [CrossRef]
- Wink, M. Modes of action of herbal medicines and plant secondary metabolites. Medicines 2015, 2, 251–286. [Google Scholar] [CrossRef] [PubMed]
- Duke, S.O. Why have no new herbicide modes of action appeared in recent years? Pest. Manag. Sci. 2012, 68, 505–512. [Google Scholar] [CrossRef] [PubMed]
- Álvarez-Rodríguez, S.; López-González, D.; Reigosa, M.J.; Araniti, F.; Sánchez-Moreiras, A.M. Ultrastructural and hormonal changes related to harmaline-induced treatment in Arabidopsis thaliana (L.) Heynh. root meristem. Plant Physiol. Biochem. 2022, 179, 78–89. [Google Scholar] [CrossRef] [PubMed]
- López-González, D.; Costas-Gil, A.; Reigosa, M.J.; Araniti, F.; Sánchez-Moreiras, A.M. A natural indole alkaloid, norharmane, affects PIN expression patterns and compromises root growth in arabidopsis thaliana. Plant Physiol. Biochem. 2020, 151, 378–390. [Google Scholar] [CrossRef]
- López-González, D.; Ledo, D.; Cabeiras-Freijanes, L.; Verdeguer, M.; Reigosa, M.J.; Sánchez-Moreiras, A.M. Phytotoxic activity of the natural compound norharmane on crops, weeds and model plants. Plants 2020, 9, 1328. [Google Scholar] [CrossRef]
- Graña, E.; Sotelo, T.; Díaz-Tielas, C.; Reigosa, M.J.; Sánchez-Moreiras, A.M. The phytotoxic potential of the terpenoid citral on seedlings and adult plants. Weed Sci. 2013, 61, 469–481. [Google Scholar] [CrossRef]
- Araniti, F.; Bruno, L.; Sunseri, F.; Pacenza, M.; Forgione, I.; Bitonti, M.B.; Abenavoli, M.R. The allelochemical farnesene affects arabidopsis thaliana root meristem altering auxin distribution. Plant Physiol. Biochem. 2017, 121, 14–20. [Google Scholar] [CrossRef]
- Gelsomino, A.; Araniti, F.; Lupini, A.; Princi, G.; Petrovičová, B.; Abenavoli, M.R. Phenolic acids in plant-soil interactions: A microcosm experiment. J. Allelochem. Inter. 2015, 1, 25–38. [Google Scholar]
- Kumar, S.; Abedin, M.M.; Singh, A.K.; Das, S. Role of phenolic compounds in plant-defensive mechanisms. In Plant Phenolics in Sustainable Agriculture; Lone, R., Shuab, R., Kamili, A.N., Eds.; Springer: Singapore, 2020; Volume 1, pp. 517–532. [Google Scholar]
- Arvaniti, O.S.; Samaras, Y.; Gatidou, G.; Thomaidis, N.S.; Stasinakis, A.S. Review on fresh and dried figs: Chemical analysis and occurrence of phytochemical compounds, antioxidant capacity and health effects. Food Res. J. 2019, 119, 244–267. [Google Scholar] [CrossRef] [PubMed]
- Kähkönen, M.P.; Hopia, A.I.; Vuorela, H.J.; Rauha, J.P.; Pihlaja, K.; Kujala, T.S.; Heinonen, M. Antioxidant activity of plant extracts containing phenolic compounds. J. Agric. Food Chem. 1999, 47, 3954–3962. [Google Scholar] [CrossRef] [PubMed]
- Silva, V.; Igrejas, G.; Falco, V.; Santos, T.P.; Torres, C.; Oliveira, A.M.P.; Pereira, J.E.; Amaral, J.S.; Poeta, P. Chemical composition, antioxidant and antimicrobial activity of phenolic compounds extracted from wine industry by-products. Food Control 2018, 92, 516–522. [Google Scholar] [CrossRef]
- Silva, V.; Falco, V.; Dias, M.I.; Barros, L.; Silva, A.; Capita, R.; Alonso-Calleja, C.; Amaral, J.S.; Igrejas, G.; Ferreira, I.C.F.R.; et al. Evaluation of the phenolic profile of castanea sativa mill. By-products and their antioxidant and antimicrobial activity against multiresistant bacteria. Antioxidants 2020, 9, 87. [Google Scholar] [CrossRef]
- Lin, H.Y.; Chang, T.C.; Chang, S.T. A review of antioxidant and pharmacological properties of phenolic compounds in acacia confusa. J. Tradit. Complement. Med. 2018, 8, 443–450. [Google Scholar] [CrossRef] [PubMed]
- Beserra, F.P.; Gushiken, L.F.S.; Hussni, M.F.; Ribeiro, V.P.; Bonamin, F.; Jackson, C.J.; Pellizzon, C.H.; Bastos, J.K. Artepillin C as an outstanding phenolic compound of brazilian green propolis for disease treatment: A review on pharmacological aspects. Phytother. Res. 2021, 35, 2274–2286. [Google Scholar] [CrossRef]
- Albuquerque, B.R.; Heleno, S.A.; Oliveira, M.B.P.P.; Barros, L.; Ferreira, I.C.F.R. Phenolic compounds: Current industrial applications, limitations and future challenges. Food Funct. 2021, 12, 14–29. [Google Scholar] [CrossRef]
- Macias-Garbett, R.; Serna-Hernández, S.O.; Sosa-Hernández, J.E.; Parra-Saldívar, R. Phenolic compounds from brewer’s spent grains: Toward green recovery methods and applications in the cosmetic industry. Front. Sustain. Food Syst. 2021, 5, 681684. [Google Scholar] [CrossRef]
- Caporaso, N.; Formisano, D.; Genovese, A. Use of phenolic compounds from olive mill wastewater as valuable ingredients for functional foods. Crit. Rev. Food Sci. Nutr. 2018, 58, 2829–2841. [Google Scholar] [CrossRef]
- Mark, R.; Lyu, X.; Lee, J.J.L.; Parra-Saldívar, R.; Chen, W.N. Sustainable production of natural phenolics for functional food applications. J. Funct. Foods 2019, 57, 233–254. [Google Scholar] [CrossRef]
- Whitehead, D.C. Identification of P-hydroxybenzoic, vanillic, p-coumaric and ferulic acids in soils. Nature 1964, 202, 418. [Google Scholar] [CrossRef] [PubMed]
- Yin, Z.; Wong, W.; Ye, W.; Li, N. Biologically active cis-cinnamic acid occurs naturally in brassica parachinensis. Chin. Sci. Bull. 2003, 48, 555. [Google Scholar] [CrossRef]
- Steenackers, W.; Klíma, P.; Quareshy, M.; Cesarino, I.; Kumpf, R.P.; Corneillie, S.; Araújo, P.; Viaene, T.; Goeminne, G.; Nowack, M.K.; et al. Cis-cinnamic acid is a novel, natural auxin efflux inhibitor that promotes lateral root formation. Plant Physiol. 2017, 173, 552–565. [Google Scholar] [CrossRef]
- Salum, M.L.; Erra-Balsells, R. High purity cis-cinnamic acid preparation for studying physiological of trans-cinnamic and cis-cinnamic acids in higher plants. Environ. Control Biol. 2013, 52, 1–10. [Google Scholar] [CrossRef][Green Version]
- Baziramakenga, R.; Leroux, I.G.D.; Simard, R.R. Effects of benzoic and cinnamic acids on membrane permeability of soybean roots. J. Chem. Ecol. 1995, 21, 1271–1285. [Google Scholar] [CrossRef]
- Lupini, A.; Araniti, F.; Sunseri, F.; Abenavoli, M.R. Coumarin interacts with auxin polar transport to modify root system architecture in arabidopsis thaliana. Plant Growth Regul. 2014, 74, 23–31. [Google Scholar] [CrossRef]
- Ye, S.F.; Zhou, Y.H.; Sun, Y.; Zou, L.Y.; Yu, J.Q. Cinnamic acid causes oxidative stress in cucumber roots, and promotes incidence of fusarium wilt. Environ. Exp. Bot. 2006, 56, 255–262. [Google Scholar] [CrossRef]
- Lupini, A.; Sorgonà, A.; Princi, M.P.; Sunseri, F.; Abenavoli, M.R. Morphological and physiological effects of trans-cinnamic acid and its hydroxylated derivatives on maize root types. Plant Growth Regul. 2016, 78, 263–273. [Google Scholar] [CrossRef]
- Araniti, F.; Lupini, A.; Mauceri, A.; Zumbo, A.; Sunseri, F.; Abenavoli, M.R. The allelochemical trans-cinnamic acid stimulates salicylic acid production and galactose pathway in maize leaves: A potential mechanism of stress tolerance. Plant Physiol. Biochem. 2018, 128, 32–40. [Google Scholar] [CrossRef]
- Salvador, V.H.; Lima, R.B.; Dos Santos, W.D.; Soares, A.R.; Böhm, P.A.F.; Marchiosi, R.; Ferrarese, M.D.L.L.; Ferrarese-Filho, O. Cinnamic acid increases lignin production and inhibits soybean root growth. PLoS ONE 2013, 8, e69105. [Google Scholar] [CrossRef]
- Wang, Y.; Xu, L.; Shen, H.; Wang, J.; Liu, W.; Zhu, X.; Wang, R.; Sun, X.; Liu, L. Metabolomic analysis with Gc-Ms to reveal potential metabolites and biological pathways involved in pb &cd stress response of radish roots. Sci. Rep. 2015, 5, 18296. [Google Scholar] [CrossRef] [PubMed]
- Van Dam, N.M.; Bouwmeester, H.J. Metabolomics in the Rhizosphere: Tapping into belowground chemical communication. Trends Plant Sci. 2016, 21, 256–265. [Google Scholar] [CrossRef]
- Landi, M.; Misra, B.B.; Muto, A.; Bruno, L.; Araniti, F. Phytotoxicity, morphological, and metabolic effects of the sesquiterpenoid nerolidol on arabidopsis thaliana seedling roots. Plants 2020, 9, 1347. [Google Scholar] [CrossRef] [PubMed]
- Pérez Chaca, M.V.; Vigliocco, A.; Reinoso, H.; Molina, A.; Abdala, G.; Zirulnik, F.; Pedranzani, H. Effects of cadmium stress on growth, anatomy and hormone contents in glycine max (L.) Merr. Acta Physiol. Plant. 2014, 36, 2815–2826. [Google Scholar] [CrossRef]
- Silva, B.R.S.; Batista, B.L.; Lobato, A.K.S. Anatomical changes in stem and root of soybean plants submitted to salt stress. Plant Biol. 2021, 23, 57–65. [Google Scholar] [CrossRef]
- Zhang, R.; Ma, X.; Wang, M.; Lv, H.; Zhu, C. Effects of salinity and water stress on the physiological and ecological processes and plasticity of tamarix ramosissima seedlings. Acta Ecol. Sin. 2016, 36, 433–441. [Google Scholar] [CrossRef]
- Lovisolo, C.; Schubert, A. Effects of water stress on vessel size and xylem hydraulic conductivity in Vitis vinifera L. J. Exp. Bot. 1998, 49, 693–700. [Google Scholar] [CrossRef]
- Hernandez-Espinoza, L.H.; Barrios-Masias, F.H. Physiological and anatomical changes in tomato roots in response to low water stress. Sci. Hortic. 2020, 265, 109208. [Google Scholar] [CrossRef]
- Prince, S.J.; Murphy, M.; Mutava, R.N.; Durnell, L.A.; Valliyodan, B.; Grover Shannon, J.; Nguyen, H.T. Root xylem plasticity to improve water use and yield in water-stressed soybean. J. Exp. Bot. 2017, 68, 2027–2036. [Google Scholar] [CrossRef]
- Purushothaman, R.; Zaman-Allah, M.; Mallikarjuna, N.; Pannirselvam, R.; Krishnamurthy, L.; Lakkegowda, C.; Gowda, L. Root anatomical traits and their possible contribution to drought tolerance in grain legumes. Plant Prod. Sci. 2013, 16, 1–8. [Google Scholar] [CrossRef]
- Moran-Zuloaga, D.; Dippold, M.; Glaser, B.; Kuzyakov, Y. Organic nitrogen uptake by plants: Reevaluation by position-specific labeling of amino acids: Reevaluation of organic n uptake by plants by position-specific labeling. Biogeochemistry 2015, 125, 359–374. [Google Scholar] [CrossRef]
- Novoa, R.; Loomis, R.S. Nitrogen and plant production. Plant Soil 1981, 58, 177–204. [Google Scholar] [CrossRef]
- Lea, P.J.; Azevedo, R.A. Nitrogen use efficiency. 2. Amino acid metabolism. Ann. Appl. Biol. 2007, 151, 269–275. [Google Scholar] [CrossRef]
- Perchlik, M.; Tegeder, M. Improving plant nitrogen use efficiency through alteration of amino acid transport processes. Plant Physiol. 2017, 175, 235–247. [Google Scholar] [CrossRef] [PubMed]
- Xin, W.; Zhang, L.; Zhang, W.; Gao, J.; Yi, J.; Zhen, X.; Li, Z.; Zhao, Y.; Peng, C.; Zhao, C. An integrated analysis of the rice transcriptome and metabolome reveals differential regulation of carbon and nitrogen metabolism in response to nitrogen availability. Int. J. Mol. Sci. 2019, 20, 2349. [Google Scholar] [CrossRef] [PubMed]
- Barros, J.; Serk, H.; Granlund, I.; Pesquet, E. The cell biology of lignification in higher plants. Ann. Bot. 2015, 115, 1053–1074. [Google Scholar] [CrossRef]
- Vanholme, R.; De Meester, B.; Ralph, J.; Boerjan, W. Lignin biosynthesis and its integration into metabolism. Curr. Opin. Biotechnol. 2019, 56, 230–239. [Google Scholar] [CrossRef]
- Dong, N.Q.; Lin, H.X. Contribution of phenylpropanoid metabolism to plant development and plant–environment interactions. J. Integr. Plant Biol. 2021, 63, 180–209. [Google Scholar] [CrossRef]
- D’Apice, G.; Moschin, S.; Araniti, F.; Nigris, S.; Di Marzo, M.; Muto, A.; Banfi, C.; Bruno, L.; Colombo, L.; Baldan, B. The role of pollination in controlling ginkgo biloba ovule development. New Phytol. 2021, 232, 2353–2368. [Google Scholar] [CrossRef]
- Silva, N.; Mazzafera, P.; Cesarino, I. Should I stay or should I go: Are chlorogenic acids mobilized towards lignin biosynthesis? Phytochemistry 2019, 166, 112063. [Google Scholar] [CrossRef]
- Luo, Y.; Wang, Y.; Xie, Y.; Gao, Y.; Li, W.; Lang, S. Transcriptomic and metabolomic analyses of the effects of exogenous trehalose on heat tolerance in wheat. Int. J. Mol. Sci. 2022, 23, 5194. [Google Scholar] [CrossRef] [PubMed]
- Yu, M.; Wang, M.; Gyalpo, T.; Basang, Y. Stem lodging resistance in hulless barley: Transcriptome and metabolome analysis of lignin biosynthesis pathways in contrasting genotypes. Genomics 2021, 113, 935–943. [Google Scholar] [CrossRef] [PubMed]
- Deng, Y.; Wang, J.; Zhang, A.; Zhu, Z.; Ren, S.; Zhang, C.; Zhang, Q. Metabolomics mechanism and lignin response to laxogenin c, a natural regulator of plants growth. Int. J. Mol. Sci. 2022, 23, 2990. [Google Scholar] [CrossRef] [PubMed]
- Kopka, J.; Schauer, N.; Krueger, S.; Birkemeyer, C.; Usadel, B.; Bergmüller, E.; Dörmann, P.; Weckwerth, W.; Gibon, Y.; Stitt, M.; et al. GMD@ CSB. DB: The golm metabolome database. Bioinformatics 2005, 21, 1635–1638. [Google Scholar] [CrossRef]
- Horai, H.; Arita, M.; Kanaya, S.; Nihei, Y.; Ikeda, T.; Suwa, K.; Ojima, Y.; Tanaka, K.; Tanaka, S.; Aoshima, K.; et al. MassBank: A public repository for sharing mass spectral data for life sciences. J. Mass Spectrom. 2010, 45, 703–714. [Google Scholar] [CrossRef]
- Fiehn, O.; Robertson, D.; Griffin, J.; Vab Der Werf, M.; Nikolau, B.; Morrison, N.; Sumner, L.W.; Goodacre, R.; Hardy, N.W.; Taylor, C.; et al. The metabolomics standards initiative (MSI). Metabolomics 2007, 3, 175–178. [Google Scholar] [CrossRef]
- Pang, Z.; Chong, J.; Zhou, G.; De Lima Morais, D.A.; Chang, L.; Barrette, M.; Gauthier, C.; Jacques, P.É.; Li, S.; Xia, J. MetaboAnalyst 5.0: Narrowing the gap between raw spectra and functional insights. Nucleic Acids Res. 2021, 49, W388–W396. [Google Scholar] [CrossRef] [PubMed]

| Pathway | Total Cmpd | Hits | Impact | Raw p | FDR |
|---|---|---|---|---|---|
| Biosynthesis of secondary metabolites—unclassified | 5 | 1 | 1 | 0.00076429 | 0.0014331 |
| Alanine, aspartate and glutamate metabolism | 22 | 9 | 0.77698 | 2.88 × 10−5 | 0.00045239 |
| Isoquinoline alkaloid biosynthesis | 6 | 2 | 0.64705 | 0.00052097 | 0.0010779 |
| Phenylalanine metabolism | 12 | 1 | 0.42308 | 0.0035621 | 0.0048574 |
| Cyanoamino acid metabolism | 26 | 4 | 0.375 | 0.0013535 | 0.0021949 |
| Glycine, serine and threonine metabolism | 33 | 6 | 0.3547 | 0.00048176 | 0.0010323 |
| beta-Alanine metabolism | 18 | 4 | 0.3254 | 1.20 × 10−5 | 0.00045239 |
| Starch and sucrose metabolism | 22 | 5 | 0.32054 | 0.0016991 | 0.0025486 |
| Tyrosine metabolism | 18 | 4 | 0.27568 | 0.0014806 | 0.0023377 |
| Galactose metabolism | 27 | 8 | 0.26927 | 0.0009734 | 0.0016687 |
| Arginine biosynthesis | 18 | 6 | 0.25243 | 0.00011212 | 0.00051746 |
| Cutin, suberine and wax biosynthesis | 14 | 2 | 0.25 | 0.00071144 | 0.001377 |
| Glyoxylate and dicarboxylate metabolism | 29 | 7 | 0.23322 | 6.03 × 10−5 | 0.00045239 |
| Citrate cycle (TCA cycle) | 20 | 5 | 0.23269 | 0.019317 | 0.022726 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
López-González, D.; Bruno, L.; Díaz-Tielas, C.; Lupini, A.; Aci, M.M.; Talarico, E.; Madeo, M.L.; Muto, A.; Sánchez-Moreiras, A.M.; Araniti, F. Short-Term Effects of Trans-Cinnamic Acid on the Metabolism of Zea mays L. Roots. Plants 2023, 12, 189. https://doi.org/10.3390/plants12010189
López-González D, Bruno L, Díaz-Tielas C, Lupini A, Aci MM, Talarico E, Madeo ML, Muto A, Sánchez-Moreiras AM, Araniti F. Short-Term Effects of Trans-Cinnamic Acid on the Metabolism of Zea mays L. Roots. Plants. 2023; 12(1):189. https://doi.org/10.3390/plants12010189
Chicago/Turabian StyleLópez-González, David, Leonardo Bruno, Carla Díaz-Tielas, Antonio Lupini, Meriem Miyassa Aci, Emanuela Talarico, Maria Letizia Madeo, Antonella Muto, Adela M. Sánchez-Moreiras, and Fabrizio Araniti. 2023. "Short-Term Effects of Trans-Cinnamic Acid on the Metabolism of Zea mays L. Roots" Plants 12, no. 1: 189. https://doi.org/10.3390/plants12010189
APA StyleLópez-González, D., Bruno, L., Díaz-Tielas, C., Lupini, A., Aci, M. M., Talarico, E., Madeo, M. L., Muto, A., Sánchez-Moreiras, A. M., & Araniti, F. (2023). Short-Term Effects of Trans-Cinnamic Acid on the Metabolism of Zea mays L. Roots. Plants, 12(1), 189. https://doi.org/10.3390/plants12010189











