Identification of Genetic Markers and Genes Putatively Involved in Determining Olive Fruit Weight
Abstract
1. Introduction
2. Results
2.1. GMs Obtained by GWAS of Fruit Weight
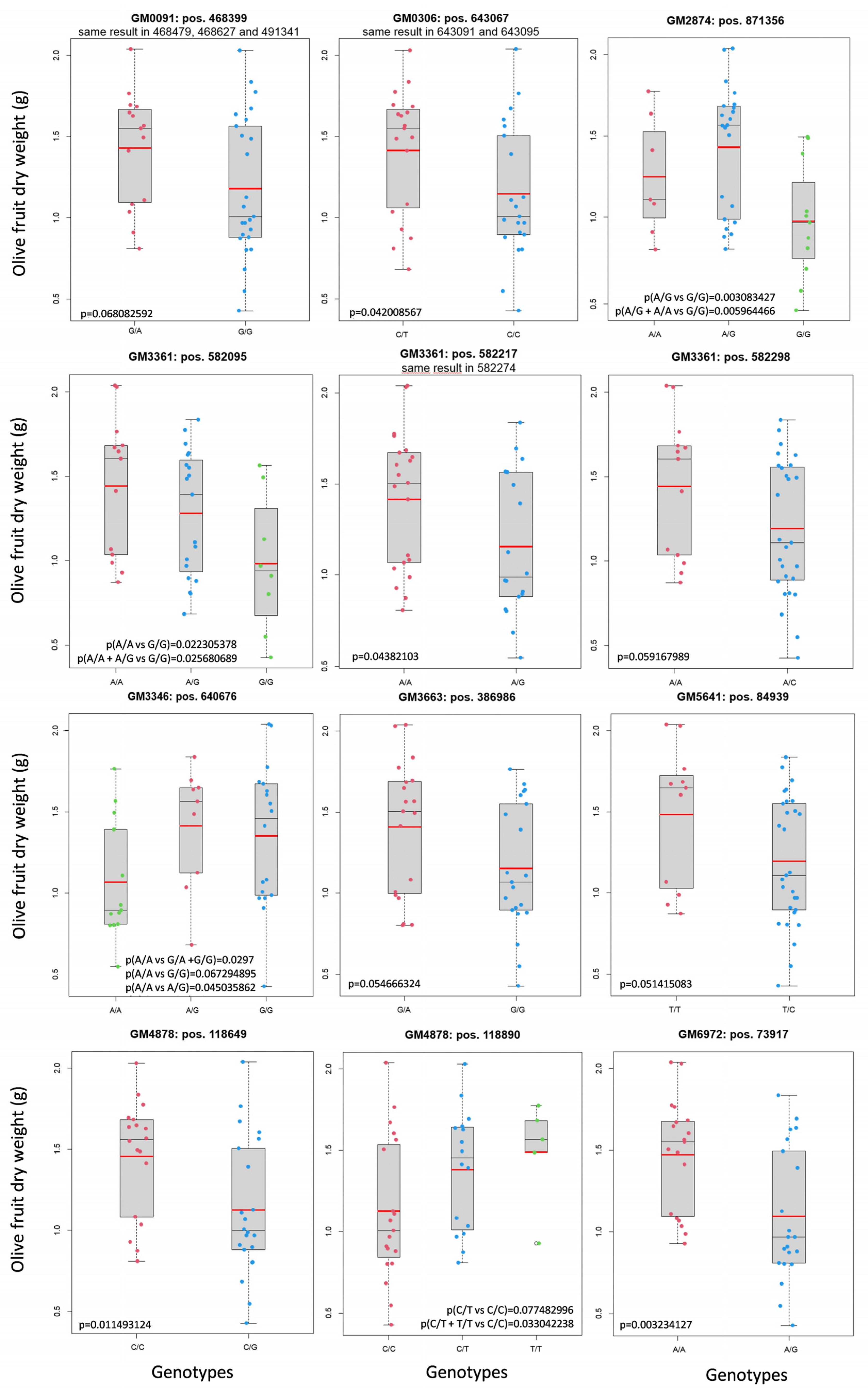
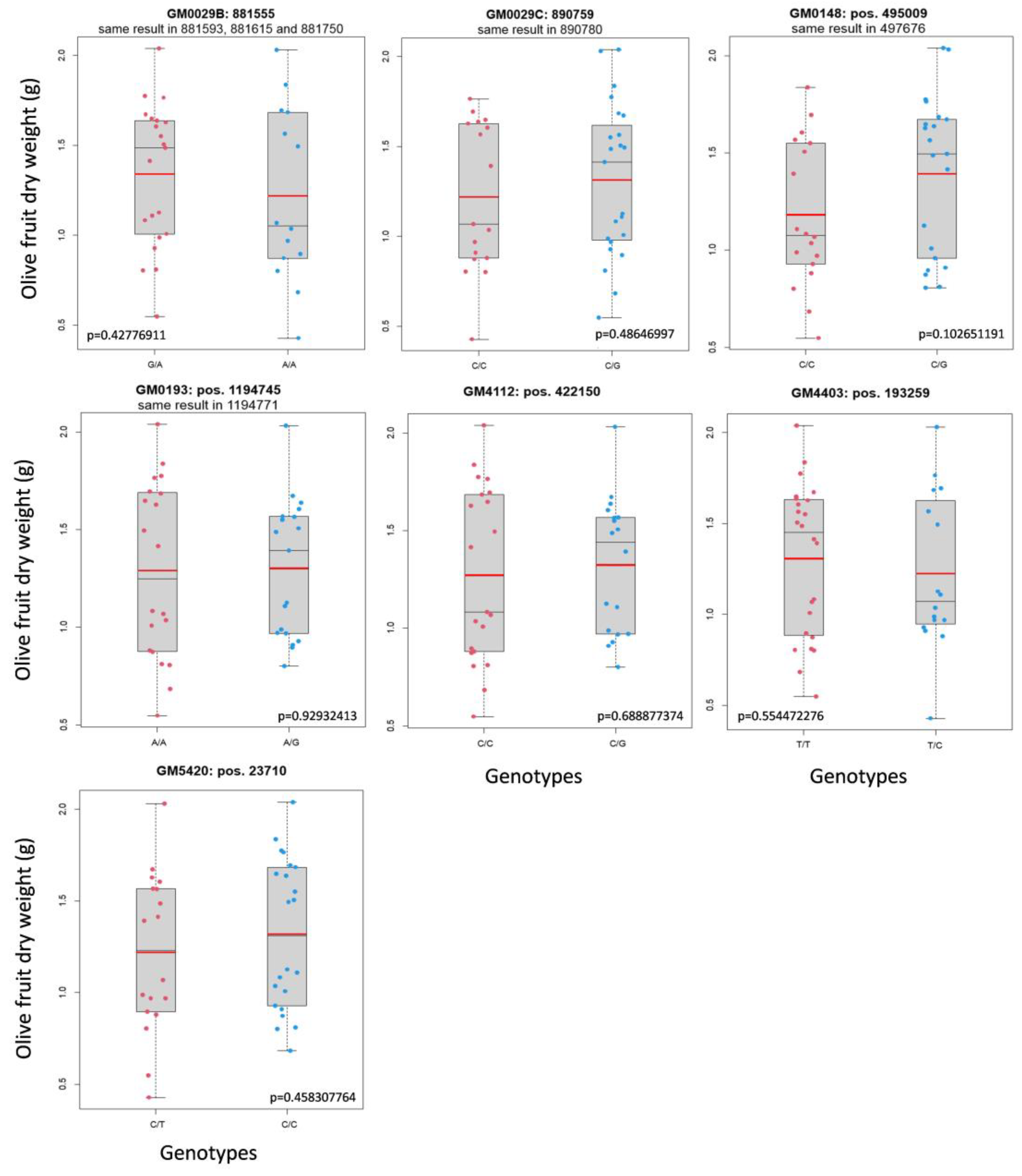
2.2. Analysis of GMs’ Segregation in a Phenotyped Progeny
2.3. Inheritance Model
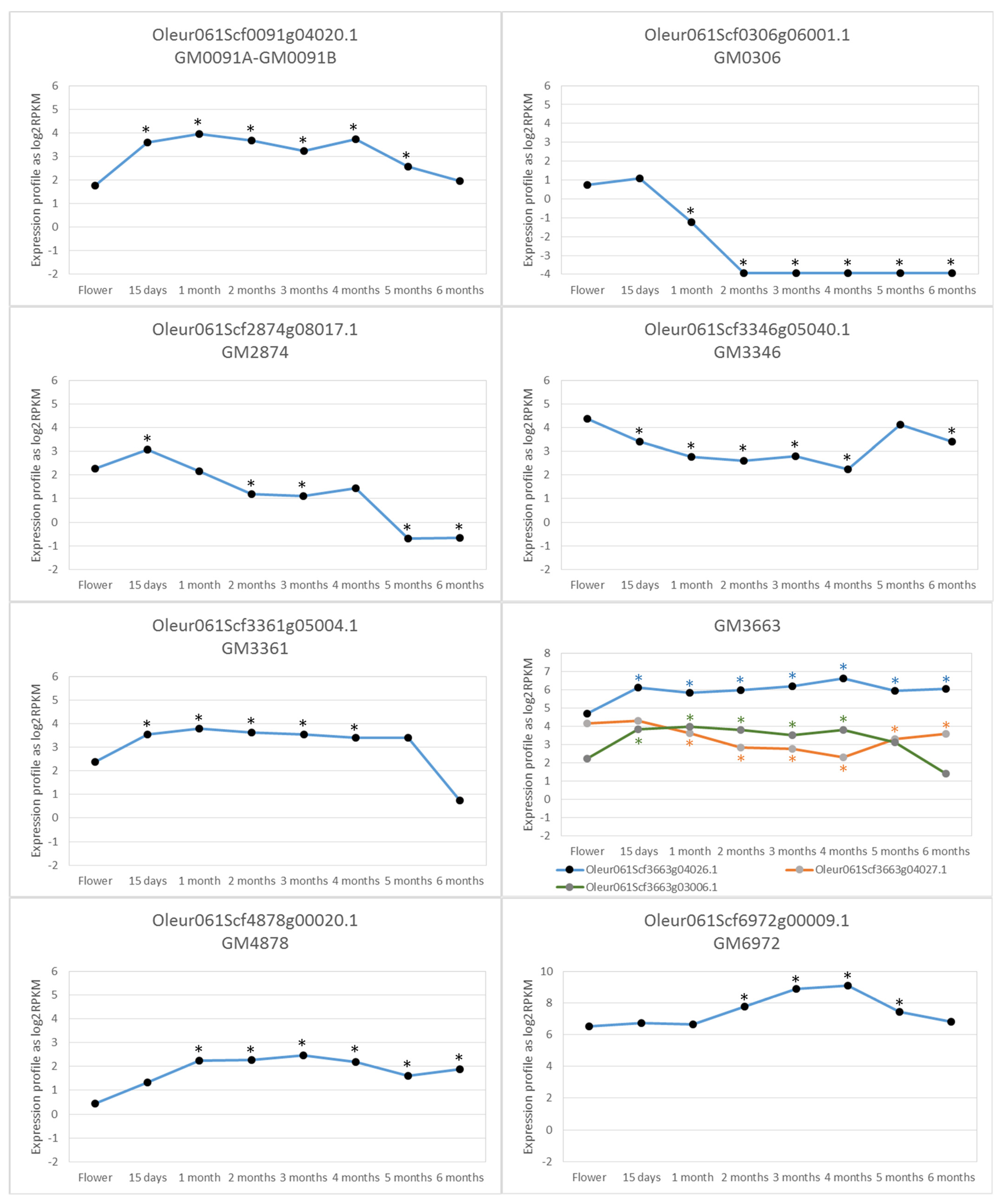
2.4. Putative Genes Associated with the Fruit Size Trait
2.4.1. GM0091
2.4.2. GM0306
2.4.3. GM2874
2.4.4. GM3346
2.4.5. GM3361
2.4.6. GM3663
2.4.7. GM4878
2.4.8. GM5641
2.4.9. GM6972
3. Discussion
4. Materials and Methods
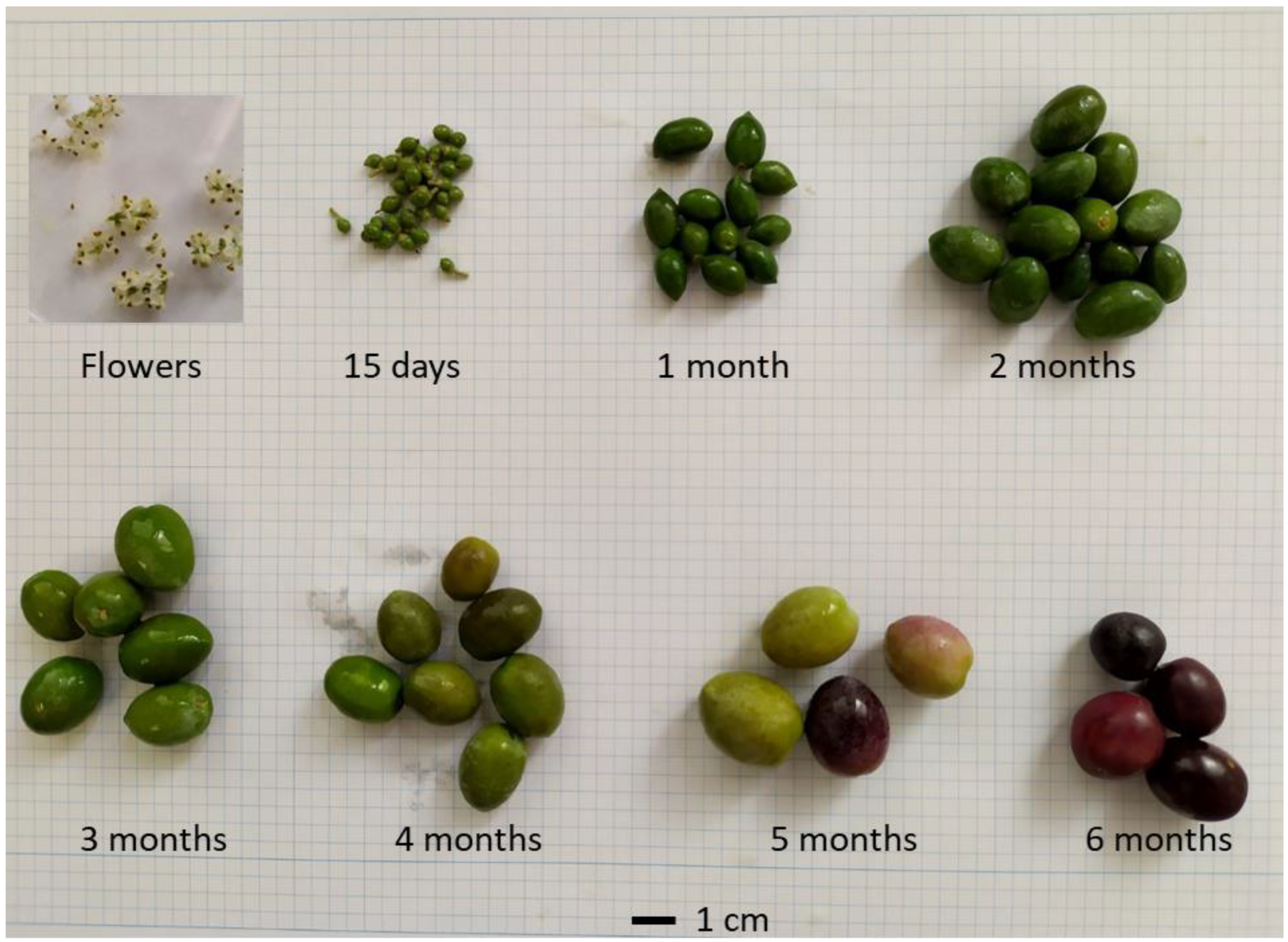
4.1. Plant Materials and Fruit Weight Determination
4.2. GWAS Analysis
4.3. Genotyping the Segregating Progeny
4.4. Statistical Analysis
4.5. Transcriptomic Analysis
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Besnard, G.; Terral, J.F.; Cornille, A. On the origins and domestication of the olive: A review and perspectives. Ann. Bot. 2017, 121, 385–403. [Google Scholar] [CrossRef] [PubMed]
- Hewitt, G.M. Genetic consequences of climatic oscillations in the Quaternary. Philos. Trans. R. Soc. London. Ser. B Biol. Sci. 2004, 359, 183–195. [Google Scholar] [CrossRef] [PubMed]
- Zohary, D.; Hopf, M.; Weiss, A. Domestication of Plants in the Old World- The Origin and Spread of Domesticated Plants in South-West Asia, Europe and the Mediterranean Basin; Oxford University Press: Oxford, UK, 2012. [Google Scholar] [CrossRef]
- Kislev, M.E.; Nadel, D.; Carmi, I. Epipalaeolithic (19,000 BP) cereal and fruit diet at Ohalo II, Sea of Galilee, Israel. Rev. Palaeobot. Palynol. 1992, 73, 161–166. [Google Scholar] [CrossRef]
- Noy, T.; Legge, A.J.; Higgs, E.S. Recent Excavations at Nahal Oren, Israel. P Prehist Soc 1973, 39, 75–99. [Google Scholar] [CrossRef]
- Galili, E.; Stanley, J.-D.; Sharvit, J.; Mina, W.-E. Evidence for Earliest OliveOil Production in Submerged Settlements off the Carmel Coast, Israel. J. Archaeol. Sci. 1997, 24, 1141–1150. [Google Scholar] [CrossRef]
- Rugini, E. The Olive Tree Genome; Springer: Berlin, Germany, 2016. [Google Scholar] [CrossRef]
- León, L.; de la Rosa, R.; Velasco, L.; Belaj, A. Using wild olives in breeding programs: Implications on oil quality composition. Front. Plant Sci. 2018, 9, 232. [Google Scholar] [CrossRef]
- Belaj, A.; León, L.; Satovic, Z.; De la Rosa, R. Variability of wild olives (Olea europaea subsp. europaea var. sylvestris) analysed by agromorphological traits and SSR markers. Sci. Hortic. 2011, 129, 561–569. [Google Scholar] [CrossRef]
- Rey, P.J.; Gutiérrez, J.E.; Alcántara, J.; Valera, F. Fruit size in wild olives: Implications for avian seed dispersal. Funct. Ecol. 1997, 11, 611–618. [Google Scholar] [CrossRef]
- Zohary, D.; Hopf, M. Domestication of plants in the Old World. Ann. Bot. 2000, 88, 666. [Google Scholar] [CrossRef]
- Carrión, Y.; Ntinou, M.; Badal, E. Olea europaea L. in the North Mediterranean Basin during the Pleniglacial and the Early-Middle Holocene. Quat. Sci. Rev. 2010, 29, 952–968. [Google Scholar] [CrossRef]
- Haouane, H.; Khadari, B. Olive diversification process in southwetern mediterranean tradicional agro-ecosystems. Acta Hortic. 2011, 918, 807–812. [Google Scholar] [CrossRef]
- León, L.; Velasco, L.; De La Rosa, R. Initial selection steps in olive breeding programs. Euphytica 2015, 201, 453–462. [Google Scholar] [CrossRef]
- León, L.; de la Rosa, R.; Barranco, D.; Rallo, L. Selection for fruit removal force and related characteristics in olive breeding progenies. Aust. J. Exp. Agric. 2005, 45, 1643–1647. [Google Scholar] [CrossRef]
- Klepo, T.; De La Rosa, R.; Satovic, Z.; León, L.; Belaj, A. Utility of wild germplasm in olive breeding. Sci. Hortic. 2013, 152, 92–101. [Google Scholar] [CrossRef]
- Santos-Antunes, F.; León, L.; de la Rosa, R.; Alvarado, J.; Mohedo, A.; Trujillo, I.; Rallo, L. The length of the juvenile period in olive as influenced by vigor of the seedlings and the precocity of the parents. HortScience 2005, 40, 1213–1215. [Google Scholar] [CrossRef]
- Minamikawa, M.F.; Nonaka, K.; Kaminuma, E.; Kajiya-Kanegae, H.; Onogi, A.; Goto, S.; Yoshioka, T.; Imai, A.; Hamada, H.; Hayashi, T.; et al. Genome-wide association study and genomic prediction in citrus: Potential of genomics-assisted breeding for fruit quality traits. Sci. Rep. 2017, 7, 4721. [Google Scholar] [CrossRef]
- Zhang, M.-Y.; Xue, C.; Hu, H.; Li, J.; Xue, Y.; Wang, R.; Fan, J.; Zou, C.; Tao, S.; Qin, M.; et al. Genome-wide association studies provide insights into the genetic determination of fruit traits of pear. Nat. Commun. 2021, 12, 1144. [Google Scholar] [CrossRef]
- Flutre, T.; Le Cunff, L.; Fodor, A.; Launay, A.; Romieu, C.; Berger, G.; Bertrand, Y.; Terrier, N.; Beccavin, I.; Bouckenooghe, V.; et al. A genome-wide association and prediction study in grapevine deciphers the genetic architecture of multiple traits and identifies genes under many new QTLs. G3 Genes|Genomes|Genetics 2022, 12, jkac103. [Google Scholar] [CrossRef]
- Zahid, G.; Aka Kaçar, Y.; Dönmez, D.; Küden, A.; Giordani, T. Perspectives and recent progress of genome-wide association studies (GWAS) in fruits. Mol. Biol. Rep. 2022, 49, 5341–5352. [Google Scholar] [CrossRef]
- Kaya, H.B.; Akdemir, D.; Lozano, R.; Cetin, O.; Kaya, H.S.; Sahin, M.; Smith, J.L.; Tanyolac, B.; Jannink, J.-L. Genome wide association study of 5 agronomic traits in olive (Olea europaea L.). Sci. Rep. 2019, 9, 18764. [Google Scholar] [CrossRef]
- Jiménez-Ruiz, J.; Ramírez-Tejero, J.A.; Fernández-Pozo, N.; Leyva-Pérez, M.d.l.O.; Yan, H.; Rosa, R.d.l.; Belaj, A.; Montes, E.; Rodríguez-Ariza, M.O.; Navarro, F.; et al. Transposon activation is a major driver in the genome evolution of cultivated olive trees (Olea Europaea L.). Plant Genome 2020, 13, e20010. [Google Scholar] [CrossRef] [PubMed]
- Unver, T.; Wu, Z.; Sterck, L.; Turktas, M.; Lohaus, R.; Li, Z.; Yang, M.; He, L.; Deng, T.; Escalante, F.J.; et al. Genome of wild olive and the evolution of oil biosynthesis. Proc. Natl. Acad. Sci. USA 2017, 114, E9413–E9422. [Google Scholar] [CrossRef] [PubMed]
- Luhua, S.; Hegie, A.; Suzuki, N.; Shulaev, E.; Luo, X.; Cenariu, D.; Ma, V.; Kao, S.; Lim, J.; Gunay, M.B.; et al. Linking genes of unknown function with abiotic stress responses by high-throughput phenotype screening. Physiol. Plant. 2013, 148, 322–333. [Google Scholar] [CrossRef] [PubMed]
- Coates, J.C. Armadillo Repeat Proteins: Versatile Regulators of Plant Development and Signalling. In Plant Growth Signaling; Bögre, L., Beemster, G., Eds.; Plant Cell Monographs; Springer: Berlin, Germany, 2007; Volume 10. [Google Scholar] [CrossRef]
- Ilgenfritz, H.; Bouyer, D.; Schnittger, A.; Mathur, J.; Kirik, V.; Schwab, B.; Chua, N.-H.; Jürgens, G.; Hülskamp, M. The Arabidopsis STICHEL gene is a regulator of trichome branch number and encodes a novel protein. Plant Physiol. 2003, 131, 643–655. [Google Scholar] [CrossRef]
- Khan, H.; Parks, N.; Kozera, C.; Curtis, B.A.; Parsons, B.J.; Bowman, S.; Archibald, J.M. Plastid genome sequence of the cryptophyte alga Rhodomonas salina CCMP1319: Lateral transfer of putative DNA replication machinery and a test of chromist plastid phylogeny. Mol. Biol. Evol. 2007, 24, 1832–1842. [Google Scholar] [CrossRef]
- He, X.; Hou, X.; Shen, Y.; Huang, Z. TaSRG, a wheat transcription factor, significantly affects salt tolerance in transgenic rice and Arabidopsis. FEBS Lett. 2011, 585, 1231–1237. [Google Scholar] [CrossRef]
- Rallo, P.; Rapoport, H.F. Early growth and development of the olive fruit mesocarp. J. Hortic. Sci. Biotech. 2001, 76, 408–412. [Google Scholar] [CrossRef]
- Del Rio, C.; Caballero, J.M.; Garcia-Fernandez, M.D. Rendimiento graso de la aceituna (Banco de Germoplasma de Córdoba). In Variedades de Olivo en España; de Andalucia, J., Ed.; M.A.P.A y Ediciones Mundi-Prensa: Madrid, Spain, 2005. [Google Scholar]
- Belaj, A.; del Carmen Dominguez-García, M.; Atienza, S.G.; Urdíroz, N.M.; De la Rosa, R.; Satovic, Z.; Martín, A.; Kilian, A.; Trujillo, I.; Valpuesta, V.; et al. Developing a core collection of olive (Olea europaea L.) based on molecular markers (DArTs, SSRs, SNPs) and agronomic traits. Tree Genet. Genomes 2012, 8, 365–378. [Google Scholar] [CrossRef]
- Friel, J.; Bombarely, A.; Fornell, C.D.; Luque, F.; Fernández-Ocaña, A.M. Comparative Analysis of Genotyping by Sequencing and Whole-Genome Sequencing Methods in Diversity Studies of Olea europaea L. Plants 2021, 10, 2514. [Google Scholar] [CrossRef]
- Arias-Calderón, R.; Rouiss, H.; Rodríguez-Jurado, D.; de la Rosa, R.; León, L. Variability and heritability of fruit characters in olive progenies from open-pollination. Sci. Hortic. 2014, 169, 94–98. [Google Scholar] [CrossRef]
- Yılmaz-Düzyaman, H.; de la Rosa, R.; León, L. Seedling Selection in Olive Breeding Progenies. Plants 2022, 11, 1195. [Google Scholar] [CrossRef] [PubMed]
- Rosati, A.; Caporali, S.; Hammami, S.B.M.; Moreno-Alías, I.; Rapoport, H.; Rosati, A.; Caporali, S.; Hammami, S.B.M.; Moreno-Alías, I.; Rapoport, H. Fruit growth and sink strength in olive (Olea europaea) are related to cell number, not to tissue size. Funct. Plant Biol. 2020, 47, 1098–1104. [Google Scholar] [CrossRef] [PubMed]
- Wang, G.; Zhang, W.A. Steganalysis-based approach to comprehensive identification and characterization of functional regulatory elements. Genome Biol. 2006, 7, R49. [Google Scholar] [CrossRef] [PubMed]
- Belaj, A.; Ninot, A.; Gómez-Gálvez, F.J.; El Riachy, M.; Gurbuz-Veral, M.; Torres, M.; Lazaj, A.; Klepo, T.; Paz, S.; Ugarte, J.; et al. Utility of EST-SNP markers for improving management and use of olive genetic resources: A case study at the Worldwide Olive Germplasm Bank of Córdoba. Plants 2022, 11, 921. [Google Scholar] [CrossRef] [PubMed]
- Langmead, B.; Salzberg, S. Fast gapped-read alignment with Bowtie 2. Nat. Methods 2012, 9, 357–359. [Google Scholar] [CrossRef]
- Li, H.; Handsaker, B.; Wysoker, A.; Fennell, T.; Ruan, J.; Homer, N.; Marth, G.; Abecasis, G.; Durbin, R. 1000 Genome Project Data Processing Subgroup. The Sequence Alignment/Map format and SAMtools. Bioinformatics 2009, 25, 2078–2079. [Google Scholar] [CrossRef]
- Danecek, P.; Auton, A.; Abecasis, G.; Albers, C.A.; Banks, E.; DePristo, M.A.; Handsaker, R.E.; Lunter, G.; Marth, G.T.; Sherry, S.T.; et al. 1000 Genomes Project Analysis Group. The variant call format and VCFtools. Bioinformatics 2011, 27, 2156–2158. [Google Scholar] [CrossRef]
- Thorvaldsdóttir, H.; Robinson, J.T.; Mesirov, J.P. Integrative Genomics Viewer (IGV): High-performance genomics data visualization and exploration. Brief. Bioinform. 2013, 14, 178–192. [Google Scholar] [CrossRef]

| GM Cluster | Scaffold | GM Positionin Scaffold | Alleles | GM Segregation with the Fruit Size Phenotype | Chromosome * |
|---|---|---|---|---|---|
| GM0014 | Oleur061Scf0014 | 300237 | A/G | - | 18 |
| GM0029A | Oleur061Scf0029 | 478377 | G/T | - | 15 |
| GM0029B | Oleur061Scf0029 | 881555 | G/A | NO | 15 |
| Oleur061Scf0029 | 881593 | C/A | NO | 15 | |
| Oleur061Scf0029 | 881615 | G/A | NO | 15 | |
| Oleur061Scf0029 | 881750 | A/C | NO | 15 | |
| Oleur061Scf0029 | 882112 | ATT/AT | - | 15 | |
| Oleur061Scf0029 | 882129 | C/T | - | 15 | |
| GM0029C | Oleur061Scf0029 | 889252 | A/C | - | 3 |
| Oleur061Scf0029 | 890759 | C/G | NO | 3 | |
| Oleur061Scf0029 | 890780 | A/G | NO | 3 | |
| GM0091 | Oleur061Scf0091 | 460570 | C/T | - | 3 |
| Oleur061Scf0091 | 465778 | A/G | - | 3 | |
| Oleur061Scf0091 | 465893 | A/G | - | 3 | |
| Oleur061Scf0091 | 466037 | T/C | - | 3 | |
| Oleur061Scf0091 | 466296 | T/C | - | 3 | |
| Oleur061Scf0091 | 468399 | G/A | YES | 3 | |
| Oleur061Scf0091 | 468479 | C/T | YES | 3 | |
| Oleur061Scf0091 | 468627 | A/G | YES | 3 | |
| Oleur061Scf0091 | 468749 | T/C | - | 3 | |
| Oleur061Scf0091 | 469150 | A/G | - | 3 | |
| Oleur061Scf0091 | 471744 | G/T | - | 3 | |
| Oleur061Scf0091 | 491025 | G/A | - | 1 | |
| Oleur061Scf0091 | 491341 | G/A | YES | 1 | |
| Oleur061Scf0091 | 491618 | T/C | - | 1 | |
| Oleur061Scf0091 | 492350 | C/G | - | 1 | |
| GM0122 | Oleur061Scf0122 | 385110 | C/T | - | 8 |
| GM0148 | Oleur061Scf0148 | 495009 | C/T | NO | 17 |
| Oleur061Scf0148 | 497676 | C/G | NO | 17 | |
| GM0193 | Oleur061Scf0193 | 1194745 | A/G | NO | US |
| Oleur061Scf0193 | 1194771 | ATTTTTTTG/ATTTTTTA | NO | US | |
| GM0306 | Oleur061Scf0306 | 640199 | C/T | - | 18 |
| Oleur061Scf0306 | 643067 | C/T | YES | 18 | |
| Oleur061Scf0306 | 643091 | C/A | YES | 18 | |
| Oleur061Scf0306 | 643095 | TT/TCT | YES | 18 | |
| GM0340 | Oleur061Scf0340 | 201854 | ATT/GTC | - | 6 |
| GM0360 | Oleur061Scf0360 | 830983 | A/T | - | US |
| GM0476 | Oleur061Scf0476 | 81329 | G/A | - | US |
| GM0503 | Oleur061Scf0503 | 499553 | GTT/CTC | - | 1 |
| GM0871 | Oleur061Scf0871 | 153993 | C/G | - | 11 |
| GM0960 | Oleur061Scf0960 | 26182 | A/G | - | US |
| Oleur061Scf0960 | 26749 | A/G | - | US | |
| Oleur061Scf0960 | 27393 | G/A | - | US | |
| GM1178 | Oleur061Scf1178 | 622380 | G/T | - | 5 |
| Oleur061Scf1178 | 622396 | G/T | - | 5 | |
| Oleur061Scf1178 | 624968 | TTT/CTA | - | 5 | |
| Oleur061Scf1178 | 628179 | A/G | - | 5 | |
| Oleur061Scf1178 | 628964 | GTATTA/AAATTC | - | 5 | |
| Oleur061Scf1178 | 629080 | T/C | - | 5 | |
| Oleur061Scf1178 | 629123 | TAGTG/TGGAC | - | 5 | |
| Oleur061Scf1178 | 629814 | TGTG/CGTA | - | 5 | |
| Oleur061Scf1178 | 630753 | GCGTGC/AAGTGT | - | 5 | |
| Oleur061Scf1178 | 631538 | TGA/TGGA | - | 5 | |
| Oleur061Scf1178 | 632869 | T/A | - | 5 | |
| GM1459 | Oleur061Scf1459 | 972705 | C/A | - | 3 |
| GM1787 | Oleur061Scf1787 | 58484 | A/C | - | 21 |
| Oleur061Scf1787 | 58605 | T/C | - | 21 | |
| GM2091 | Oleur061Scf2091 | 170031 | G/A | - | US |
| Oleur061Scf2091 | 170178 | C/G | - | US | |
| Oleur061Scf2091 | 170206 | T/C | - | US | |
| Oleur061Scf2091 | 172566 | T/G | - | US | |
| Oleur061Scf2091 | 173464 | T/A | - | US | |
| Oleur061Scf2091 | 173501 | T/C | - | US | |
| Oleur061Scf2091 | 173583 | T/C | - | US | |
| Oleur061Scf2091 | 173729 | A/G | - | US | |
| Oleur061Scf2091 | 174318 | C/T | - | US | |
| Oleur061Scf2091 | 175015 | TAA/TA | - | US | |
| Oleur061Scf2091 | 175071 | T/A | - | US | |
| Oleur061Scf2091 | 175636 | A/G | - | US | |
| Oleur061Scf2091 | 175811 | C/G | - | US | |
| Oleur061Scf2091 | 175966 | T/A | - | US | |
| Oleur061Scf2091 | 176408 | A/C | - | US | |
| Oleur061Scf2091 | 176444 | T/C | - | US | |
| Oleur061Scf2091 | 176468 | G/A | - | US | |
| Oleur061Scf2091 | 176475 | A/C | - | US | |
| GM2874 | Oleur061Scf2874 | 871356 | A/G | YES | 5 |
| GM3270 | Oleur061Scf3270 | 126286 | TC/AA | - | 7 |
| GM3346 | Oleur061Scf3346 | 640676 | G/A | YES | 8 |
| GM3361 | Oleur061Scf3361 | 582095 | A/G | YES | 20 |
| Oleur061Scf3361 | 582217 | A/G | YES | 20 | |
| Oleur061Scf3361 | 582274 | TTGT/TT | YES | 20 | |
| Oleur061Scf3361 | 582298 | A/C | YES | 20 | |
| Oleur061Scf3361 | 582368 | T/A | - | 20 | |
| Oleur061Scf3361 | 582444 | T/A | - | 20 | |
| Oleur061Scf3361 | 582495 | A/T | - | 20 | |
| GM3663 | Oleur061Scf3663 | 386986 | G/A | YES | 8 |
| Oleur061Scf3663 | 387078 | T/C | - | 8 | |
| GM3825 | Oleur061Scf3825 | 171174 | T/C | - | 7 |
| GM4112 | Oleur061Scf4112 | 400436 | C/T | - | 13 |
| Oleur061Scf4112 | 400456 | C/A | - | 13 | |
| Oleur061Scf4112 | 400466 | G/A | - | 13 | |
| Oleur061Scf4112 | 422150 | C/G | NO | 3 | |
| Oleur061Scf4112 | 425723 | GA/TG | - | 3 | |
| GM4351 | Oleur061Scf4351 | 26325 | G/C | - | 15 |
| GM4403 | Oleur061Scf4403 | 193259 | T/C | NO | 14 |
| GM4462 | Oleur061Scf4462 | 60219 | T/C | - | 7 |
| GM4491 | Oleur061Scf4491 | 299627 | C/G | - | 18 |
| Oleur061Scf4491 | 300578 | C/G | - | 18 | |
| Oleur061Scf4491 | 300597 | G/A | - | 18 | |
| GM4878 | Oleur061Scf4878 | 118649 | C/G | YES | 4 |
| Oleur061Scf4878 | 118890 | T/C | YES | 4 | |
| GM4977 | Oleur061Scf4977 | 92722 | T/G | - | 1 |
| GM5420 | Oleur061Scf5420 | 23710 | C/T | NO | 16 |
| Oleur061Scf5420 | 27925 | C/T | - | 16 | |
| Oleur061Scf5420 | 29878 | C/T | - | 16 | |
| Oleur061Scf5420 | 220407 | A/G | - | 16 | |
| GM5641 | Oleur061Scf5641 | 83225 | A/C | YES | 11 |
| Oleur061Scf5641 | 84939 | T/C | - | 11 | |
| GM6972 | Oleur061Scf6972 | 73917 | A/G | YES | 21 |
| GM7206 | Oleur061Scf7206 | 20070 | CACG/CGCA | - | 16 |
| GM7731 | Oleur061Scf7731 | 48216 | TTT/CTC | - | 8 |
| Oleur061Scf7731 | 48236 | G/A | - | 8 | |
| GM8230 | Oleur061Scf8230 | 14454 | T/C | - | 15 |
| Tree Reference Number Row Tree | Fruit Dry Weight Average of Seasons 2019–2020 (g) | |
|---|---|---|
| Frantoio | 1.24 | |
| Picual | 1.45 | |
| Heaviest fruit trees: | ||
| 279 | 59 | 2.04 |
| 281 | 4 | 2.03 |
| 281 | 29 | 1.84 |
| 280 | 39 | 1.77 |
| 283 | 12 | 1.77 |
| 282 | 35 | 1.70 |
| 281 | 15 | 1.68 |
| 281 | 13 | 1.67 |
| 281 | 17 | 1.65 |
| 282 | 32 | 1.64 |
| 279 | 42 | 1.63 |
| 281 | 55 | 1.60 |
| 280 | 47 | 1.57 |
| 282 | 46 | 1.56 |
| 282 | 11 | 1.55 |
| 282 | 33 | 1.51 |
| 280 | 7 | 1.50 |
| 280 | 13 | 1.49 |
| 279 | 19 | 1.41 |
| 280 | 45 | 1.39 |
| Lightest fruit trees: | ||
| 280 | 8 | 1.13 |
| 281 | 16 | 1.11 |
| 281 | 42 | 1.08 |
| 281 | 34 | 1.07 |
| 279 | 34 | 1.04 |
| 282 | 48 | 1.01 |
| 280 | 27 | 0.99 |
| 282 | 30 | 0.97 |
| 280 | 43 | 0.97 |
| 280 | 26 | 0.93 |
| 279 | 16 | 0.91 |
| 279 | 35 | 0.90 |
| 280 | 59 | 0.88 |
| 279 | 37 | 0.87 |
| 281 | 39 | 0.81 |
| 280 | 4 | 0.81 |
| 279 | 32 | 0.80 |
| 280 | 30 | 0.68 |
| 279 | 33 | 0.55 |
| 283 | 11 | 0.43 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Moret, M.; Ramírez-Tejero, J.A.; Serrano, A.; Ramírez-Yera, E.; Cueva-López, M.D.; Belaj, A.; León, L.; de la Rosa, R.; Bombarely, A.; Luque, F. Identification of Genetic Markers and Genes Putatively Involved in Determining Olive Fruit Weight. Plants 2023, 12, 155. https://doi.org/10.3390/plants12010155
Moret M, Ramírez-Tejero JA, Serrano A, Ramírez-Yera E, Cueva-López MD, Belaj A, León L, de la Rosa R, Bombarely A, Luque F. Identification of Genetic Markers and Genes Putatively Involved in Determining Olive Fruit Weight. Plants. 2023; 12(1):155. https://doi.org/10.3390/plants12010155
Chicago/Turabian StyleMoret, Martín, Jorge A. Ramírez-Tejero, Alicia Serrano, Elena Ramírez-Yera, María D. Cueva-López, Angjelina Belaj, Lorenzo León, Raúl de la Rosa, Aureliano Bombarely, and Francisco Luque. 2023. "Identification of Genetic Markers and Genes Putatively Involved in Determining Olive Fruit Weight" Plants 12, no. 1: 155. https://doi.org/10.3390/plants12010155
APA StyleMoret, M., Ramírez-Tejero, J. A., Serrano, A., Ramírez-Yera, E., Cueva-López, M. D., Belaj, A., León, L., de la Rosa, R., Bombarely, A., & Luque, F. (2023). Identification of Genetic Markers and Genes Putatively Involved in Determining Olive Fruit Weight. Plants, 12(1), 155. https://doi.org/10.3390/plants12010155









