1. Introduction
The Intergovernmental Panel on Climate Change (IPCC) estimates an increase in average temperatures from 2 to 4.5 °C within the 21st century [
1]. In this scenario of global warming, agriculture is expected to be one of the most affected sectors [
2], with negative impacts on crop productivity due to the effects of high temperatures on plant development [
3,
4]. The major losses due to heat stress are expected to occur in low-latitude regions (temperate and tropical areas), with temperatures exceeding even the most extreme seasonal temperatures recorded to date [
5]. In fact, yield reductions due to heat stress have already been documented in many crops from these geographical areas, such as wheat, rice, barley, sorghum, maize, chickpea, canola and more [
6,
7,
8].
Tomato (
Solanum lycopersicum L.) is one of the most extensively cultivated crops in temperate regions worldwide [
9]. The cultivation of this crop is affected by high temperature, which can cause different physiological or morphological injuries, such as reductions in plant growth and development or damage to reproductive organs and pollen [
10,
11,
12,
13]. As a consequence, agronomic yield reductions have already been reported due to heat stress [
6].
Thus, there is an urgent need to increase the knowledge of the genetic responses to heat stress, considering that heat tolerance is a complex trait affected by numerous factors [
14], and to develop plant breeding strategies to face this problem. Reproductive traits have been commonly used to study the responses to heat stress in tomato plants. The flower number per inflorescence decreases due to heat stress [
15,
16], and the ability of plants to set fruit after the exposure to high temperatures is the most commonly studied trait. This trait is quite complex, involving physiology, biochemistry and gene regulation pathways [
3], affecting aspects ranging from the pollen viability [
12,
14,
17], photosynthesis and respiration [
18] to activation or silencing of genes [
19,
20]. Additionally, the fruit set is directly correlated with the final agronomic yield. Therefore, the fruit set at high temperature is considered a good indicator of heat tolerance in tomatoes, and it has been widely studied in previous works [
14,
16,
21,
22,
23]. Only a very limited number of cultivated varieties or accessions have been identified as heat-tolerant [
10,
23,
24]. On the other hand, several wild
Solanum spp. are well adapted to marginal environments, being more tolerant to biotic and abiotic stresses than cultivated varieties [
25], which could be potential sources of tolerance. In tomatoes, the use of these wild relative species has been extensively exploited in plant breeding strategies for different characteristics [
26], including biotic stress [
25] or abiotic stress, such as salinity tolerance [
27,
28,
29]. Regarding heat tolerance, accessions from wild
Solanum spp. such as
S. pimpinellifolium, L.,
S. pennellii L.,
S. habrochaites L. and
S. chmielewskii L. have been found to be tolerant to high temperatures [
14,
17,
23,
30,
31,
32,
33,
34], confirming the value of the wild germplasm as a source of thermotolerance.
Currently, our understanding of the genetic control of the heat tolerance in tomatoes is still very limited, with few reports addressing this topic. Quantitative trait loci (QTLs) involved in heat tolerance have been previously identified in tomatoes [
22,
24,
35,
36,
37,
38,
39]. In most of these studies, the identification of QTLs associated with heat tolerant traits in tomatoes was achieved using simple mapping populations (F
2), with a limited sample size or without a dense marker coverage. Therefore, the number of reported QTLs involved in heat tolerance is relatively low, and the stability of their effects still needs to be verified. More recently, powerful mapping populations such as recombinant inbred lines (RILs), introgression lines (ILs) [
22], multiparent advanced generation intercross (MAGIC) panels and genome-wide association study (GWAS) panels [
39] have been used to dissect the genetic control of heat tolerance, providing the foundation for marker-assisted selection programs for the development of tolerant cultivars.
Along with the temperature, other environmental factors such as salinity and drought affect plant growth [
40]. Furthermore, in real field conditions, the occurrence of different stresses is common. The response to the combination of stresses is specific and independent of the individual response to each stress [
41], although cross-tolerance can occur [
42]. For example, the effect of heat stress has been reported to increase tolerance to various abiotic stresses [
43]. In the current climate change scenario, the concurrence of various stresses simultaneously is expected. Thus, the search for plants with tolerance to different stresses will be a target in plant breeding programs. In the same way, increased knowledge of the genetic control of tolerance to high temperature has become necessary, together with the development of new varieties of tomato tolerant to heat and to different abiotic stresses. Therefore, taking these perspectives into account, the main objective of the present work is the identification of QTLs that are mainly related to reproductive traits involved in the responses to different temperatures. Two very different populations of inbred lines from independent crosses between tomato accessions (fresh market and processing tomato cultivars) and two
S. pimpinellifolium accessions were studied to expand the genetic variability, and to genetically analyze the responses of reproductive traits to heat stress.
3. Discussion
The threat of climate change has resulted in the urgent need to understand the genetic control of abiotic stress responses in order to overcome the effects of environmental changes. Identifying sources of genetic diversity for heat tolerance may contribute towards the development of new varieties that are better adapted to the new environmental conditions by offering novel solutions to the problem. In the present study, we studied the genetic control of reproductive traits under different temperature regimes in two tomato inbred line populations derived from crosses between cultivated and wild relative accessions to search for genetic variability that may be useful for the development of new cultivars that might adapt to changing environmental conditions.
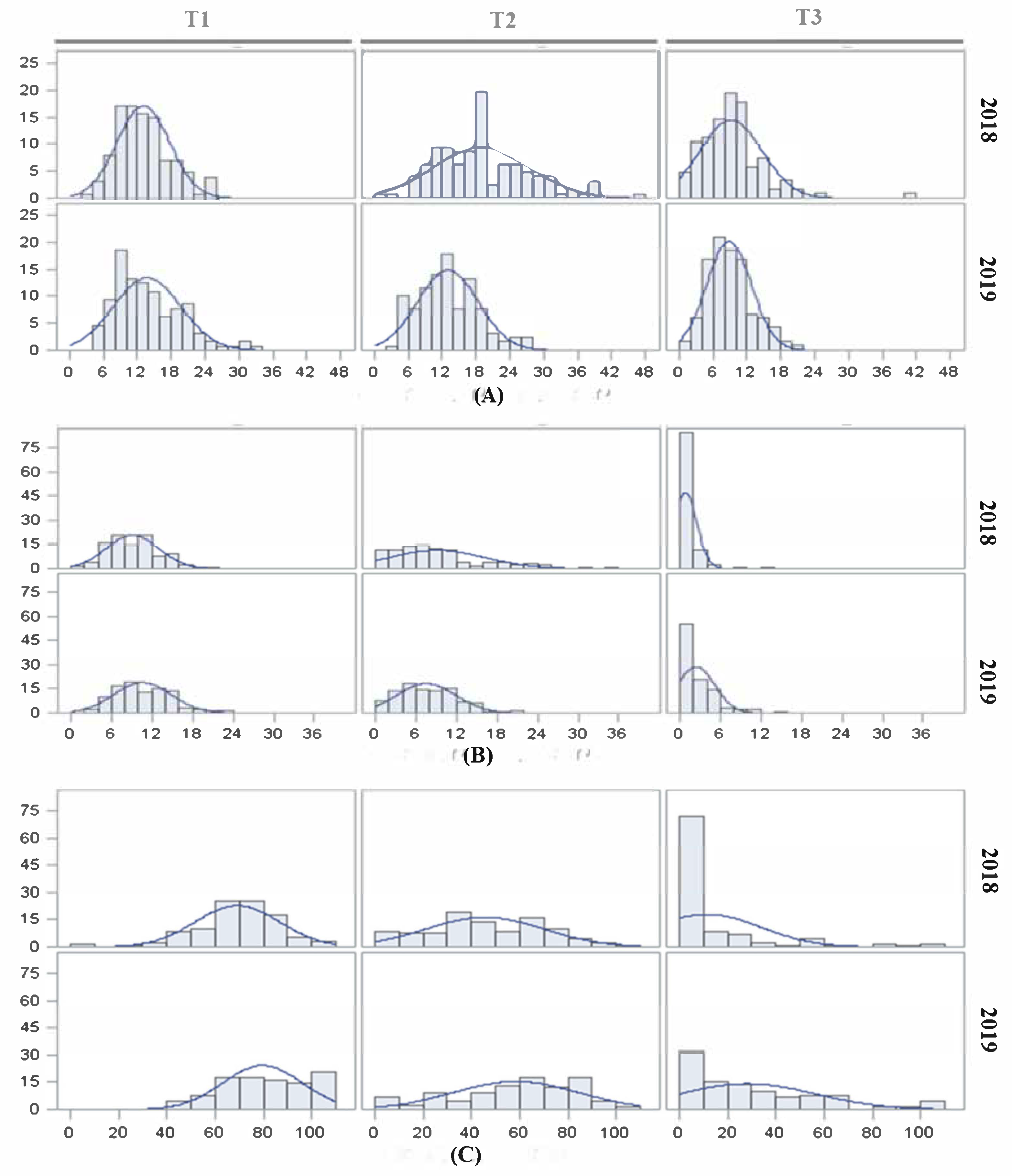
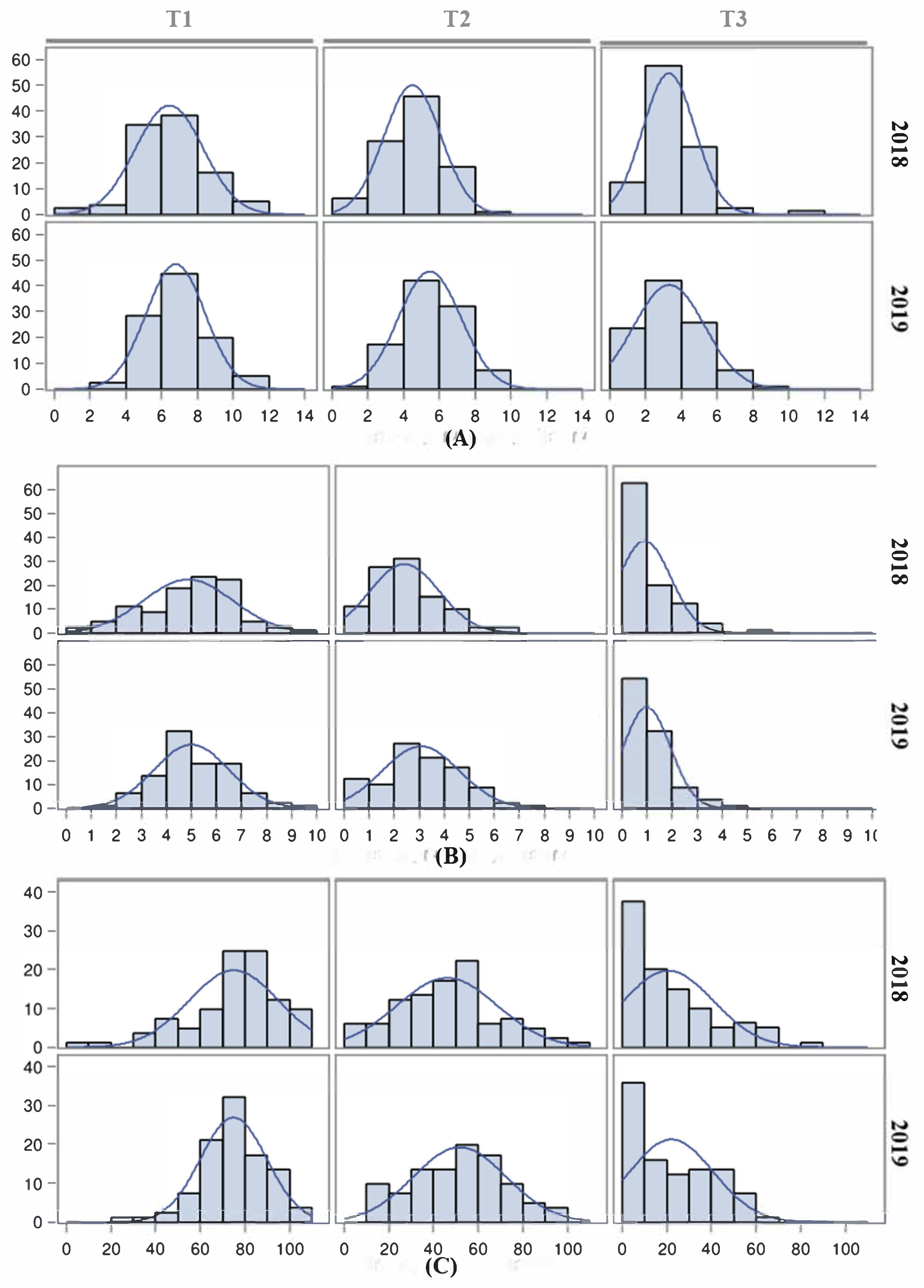
The effect of high temperatures on reproductive traits is evident when night temperatures are maintained over 25 °C [
46], which may be frequently reached in the Mediterranean basin fields during summer. Thus, both inbred line populations were subjected to three temperature regimes (control T1, moderate stress T2 and extreme stress T3) to evaluate their responses to temperature changes and to identify QTLs involved in these responses. The effect of heat stress on FLN was more evident under T3, although the magnitude was not as drastic as reported previously [
22,
23,
24,
46,
47], especially in the E9×L5 population, suggesting a better performance of this population under heat stress for this trait.
In general, both populations showed a good response to moderate heat stress under T2, with an average FRS close to 50% but with a wider range compared with T1. However, FRS was dramatically reduced under T3, with a low proportion of inbred lines being capable of setting fruits at this temperature, as reported previously [
21,
22,
23,
24,
46,
47,
48]. The current results confirm that the heat stress mostly affects the fertility of the flowers and not their production. The lack of parents, due to different agronomical problems, made the confirmation of transgressive segregation impossible, although the wide range of values obtained under T2 and the extreme values under T3 suggest the existence of transgressive phenotypes, which are commonly reported in crosses of cultivated tomatoes with exotic germplasm [
49,
50,
51]. The variability of responses under heat stress observed in both populations confirmed the complexity of the trait.
The strong and positive correlations, and the relatively high repeatability observed for FLN and FRN between the 2018 and 2019 under T1 and T2 experiments, indicated a significant hereditability of the tolerance under moderate heat stress. The correlation and repeatability of FLN and FRN under T3 were in general low and non-significant, suggesting no or low genetic variance for these traits at that heat stress. On the other hand, the low repeatability for FRS indicates a low genetic variability for this trait in the studied populations.
The genetic map generated for the E6203×LA1589 population had a total length of 1502 cM, similar to other published maps for this species, ranging from 1091 cM [
52] to 2023 cM [
53]. Moreover, the collinearity between the physical and genetic maps was very high, validating the robustness of the map to be used in QTL analysis.
Most of the detected QTLs were associated with FLN and FRN under T1 and T2 temperature regimes, coinciding with the heritability estimated in those trait and temperature regime combinations. Therefore, the results obtained in the current report may have impacted those conditions. On the other hand, additional QTLs were detected using log-transformed data or MQM analysis, which were remarkable for FRS under T3 in the E9×L5 population. This observation reinforces the need to optimize the data analysis for such complex traits to obtain the maximum information and to not miss potentially valuable QTLs such as
frslog5.2_T3_2E. This low QTL detection, as well as the modest single QTL effects at high temperature regimes, was concomitant with the low genetic variability estimated via repeatability analysis, as found in previous studies [
21,
22,
24,
37,
38,
39], reflecting the genetic complexity of the response to heat stress. Even though an important proportion of the QTLs were population-specific, as previously found [
39], some genomic regions contained QTLs clusters detected in several studies. Thus, FRN QTLs were detected in both E9×L5 and E6203×LA1589 at the bottom of chromosome 1. Additionally, IL SP_1–4, which harbors an introgression from the
S. pimpinellifolium TO-937 accession on the same genomic region in the genetic background of the Moneymaker cultivar, very consistently showed high FRS and FRN under T3 in previous works [
22], while a plasticity QTL for FRN also mapped to that region [
39]. Another interesting QTL cluster was found at the bottom of chromosome 2, whereby
BCfln2.1_T3_2E and FLN plasticity QTLs (in the population E6203×LA1589) co-located with FLN QTLs were detected in the Moneymaker×TO937 RIL population [
22], GWAS studies [
24,
54] and MAGIC populations [
39]. Another QTL cluster was found in the central region of chromosome 2, with QTLs involved in reproductive traits in several populations, for example,
fln2.1_T2_2E (E9×L5),
BCfrn2.1_T1_2E (E6203×LA1589),
fln2.1_T3_2E (Moneymaker×TO-937, [
22]),
nflw2.2,
nflw2.3 (GWAS panel, [
39]) and
nflw2 (MAGIC, [
39]). These results suggest that those genomic regions are strong candidates for harboring useful genetic diversity for the study of the responses of reproductive traits under different temperature regimes. Other examples of co-localization include
flnlog6.1_T3_2E and
flnlog10.1_T2_2E, with QTLs identified in Moneymaker×TO-937 [
22],
fln5.1_T2_2E with nflw5.1 in the MAGIC population [
39] and
fln2.1_T2_2E,
pfln6.1_T31_2E and
BCfln9.1_T2_2E with associations from a GWAS panel [
39]. Furthermore, the unexpected differences found in the QTL detection depending on the software could be due to differences in sensitivity to deviations from normality, as well as in the tools and algorithms used in each software for the selection of cofactors during multiple QTL mapping or composite interval mapping procedures, although simulation studies are needed for confirmation. In any case, the IciMapping software allows for multi-environmental QTL analysis, and in fact was able to detect 6 QTLs that were not detected at all when analyzing the years separately.
Bineau et al. [
39] demonstrated the power of multiple parent populations in tomato plants to identify QTLs involved in response to heat stress. Compared with bi-parental population experiments (the current study and [
22]), some similar conclusions can be drawn, i.e., a large proportion of the QTLs were population-specific, and the individual effects of the QTLs were usually modest. Nevertheless, some QTLs seem to be mapped in the same regions among populations, which could represent major adaptation QTLs and targets for further investigations. Thus, these results confirm the need to continuously search for new genetic variability for stress tolerance to obtain a global picture of the QTLs involved in heat stress to identify those with robust effects that could be integrated efficiently in breeding programs and to identify the underlying genes.
Alleles from both parents were found to be associated with tolerance to heat stress in the current and previous studies [
22,
37,
38]. In the current report, wild alleles usually increased the traits in the E9×L5 population, while in the E6203×LA1589 population cultivated alleles were more frequently associated with a tolerant response in the reproductive trait. This variation in the direction of the additive effects, even within populations, might indicate that most of the studied accessions included alleles that may induce a positive response to heat stress, complementary to the alleles included in other accessions.
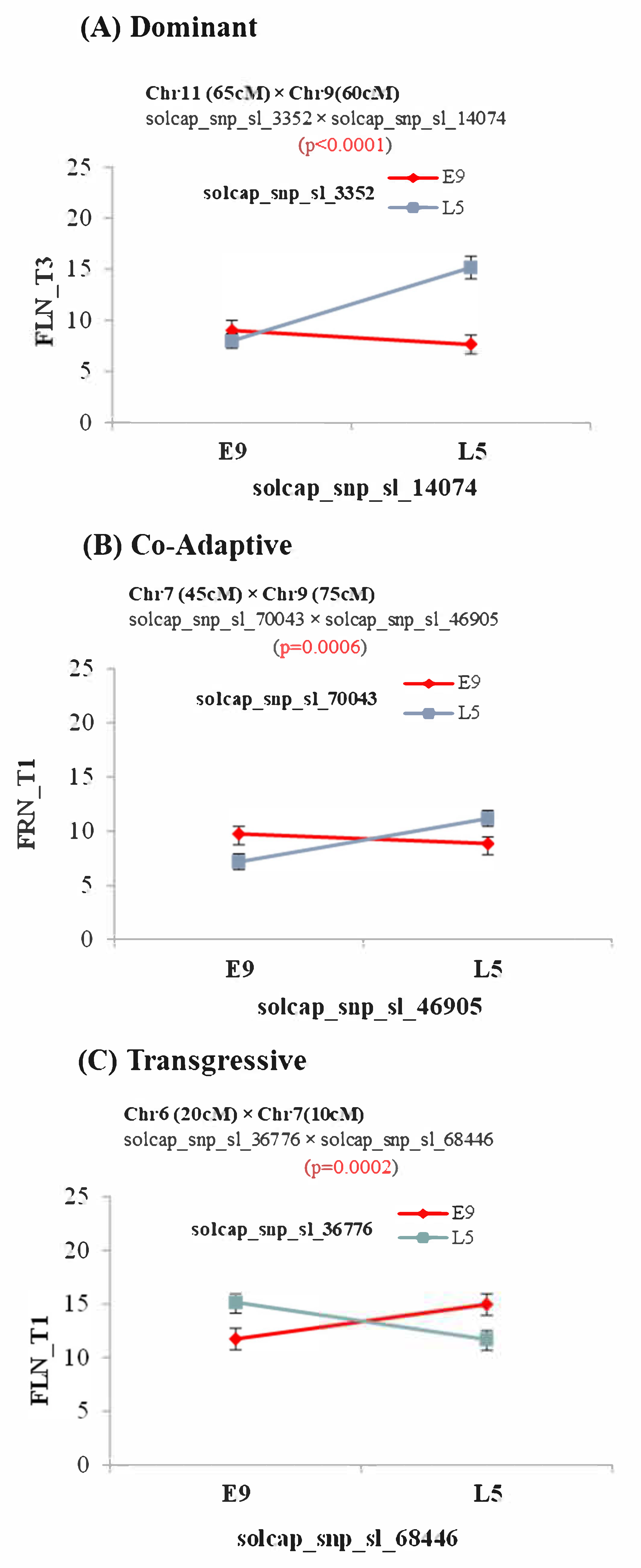
Interestingly, a relatively large number of epistatic interactions were detected in the E9×L5 population. Most of the interactions occurred in genomic regions that did not include any main effect QTL. The detection of epistatic interaction between QTLs is truly a challenge due to the low statistical power of the common experimental designs to detect these interactions [
55]. The E9×L5 population had a balanced allelic frequency (f(L5) = f(E9) = 0.5), whereas in the E6203×LA1589 population, the allelic frequencies were unbalanced (f(E6203) = 0.875, f(LA1589) = 0.125). The power to detect epistasis is maximal in balanced populations, and the unbalanced allele was certainly a factor that made the detection of interactions difficult in the E6203×LA1589 population. Thus, the possibility that the lack of detection was due to low statistical power in this population cannot be ruled out. Nevertheless, the high number of epistatic interactions in the E9×L5 population contrasts with the low number of detected main effect QTLs. Remarkably, epistatic interactions were detected under T3 for FRN, but no main effect QTLs were detected at this temperature regime. This result suggests that an important proportion of the genetic variability for response to heat stress is non-additive in the E9×L5 population. This may also explain the relatively low number of detected QTLs under high temperature regimes, as the effects of the QTL alleles would depend on the allelic state of another locus at different chromosomes, so the main QTL effects may seem low or null when estimating single QTL effects, especially in cross-interactions, reinforcing the role of QTL interactions in important agronomic traits. Different types of epistasis were found, and transgressive epitasis, defined when the combination of double-homozygous alleles from different parents showed better performance than double-homozygous alleles from the same parent alleles (
Figure 2C and
Supplementary Figure S3), was interestingly frequent. This kind of interaction could be explained at the molecular level as interactions involving repressors or negative regulators that do not fit between species.
In summary, a low number of QTLs was found for each temperature regime and reproductive trait in both RIL populations. Among them, QTLs involved in FLN and FRN under optimal and moderate–high temperature regimes (T1 and T2) were mainly detected, meaning research must subscribe to these conditions. Low QTL detection rates in studies on heat tolerance of tomato were observed in previous works [
22,
24,
37,
38], corroborating the genetic complexity of heat tolerance in this crop. Studies involving multiple parent populations have been demonstrated to be very powerful for the detection of a larger number of QTLs, although the individual contribution of each QTL is relatively modest [
39]. The search for new sources of tolerance is still needed to enhance our knowledge on the genetic control of this abiotic stress. In the current report, the role of non-additive genetic variability was reinforced, undoubtedly complicating the practical application of the scientific results. Despite the low number of QTLs, four genomic regions presented common QTLs among the two studied populations. One region on chromosome 1 and two regions on chromosome 2 were also previously associated with heat tolerance [
22,
24,
39,
54], while two others on chromosomes 6 and 10 were also validated in [
22]. The concurrent identification of QTLs associated with reproductive traits under heat stress in the same localization area in different studies reinforces the importance of these regions in the adaptive response of reproductive traits to high temperatures in tomato plants.