Overexpression of a Gene Encoding Trigonelline Synthase from Areca catechu L. Promotes Drought Resilience in Transgenic Arabidopsis
Abstract
:1. Introduction
2. Results
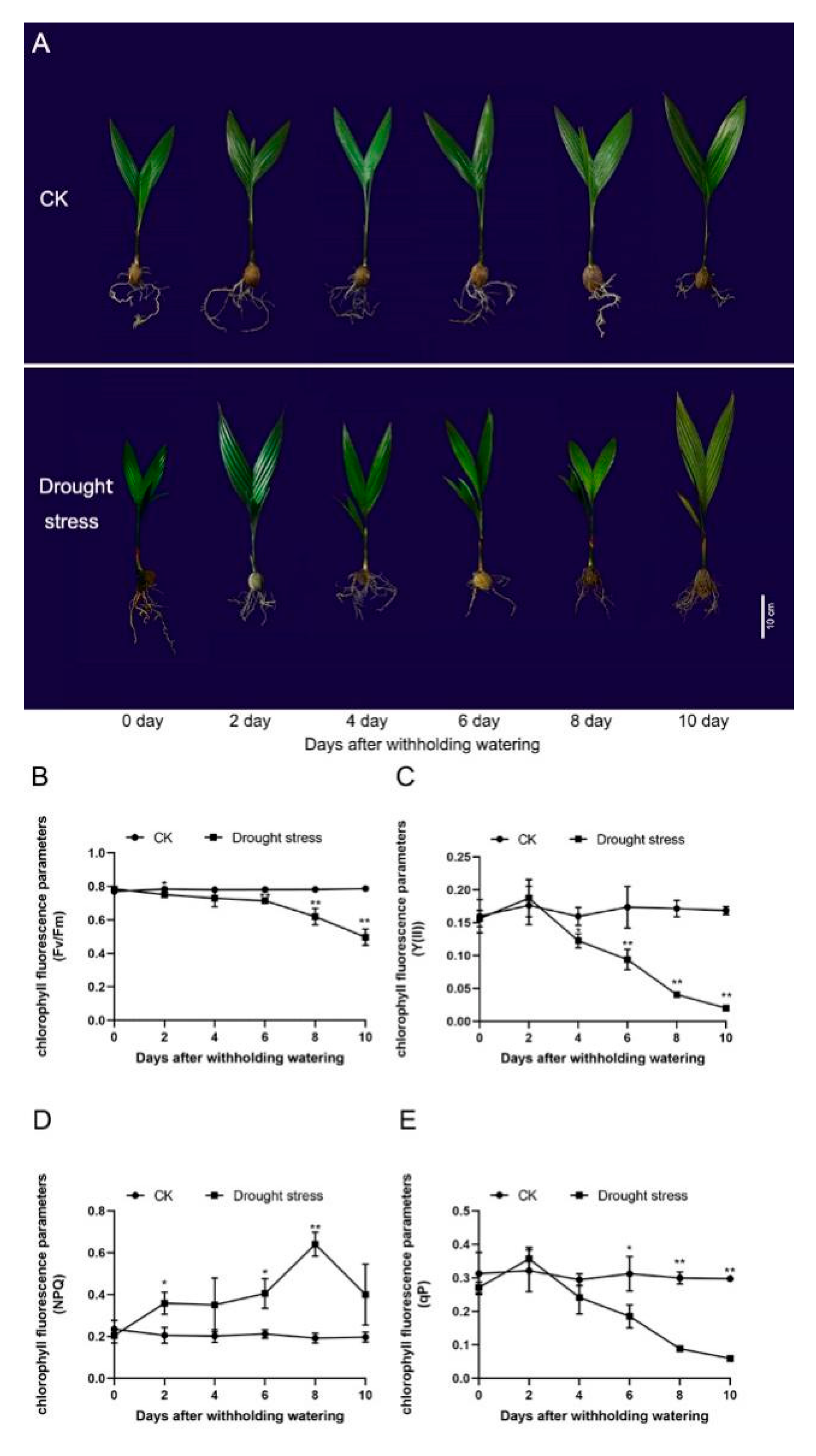
2.1. Drought Stress Response of A. catechu Seedlings
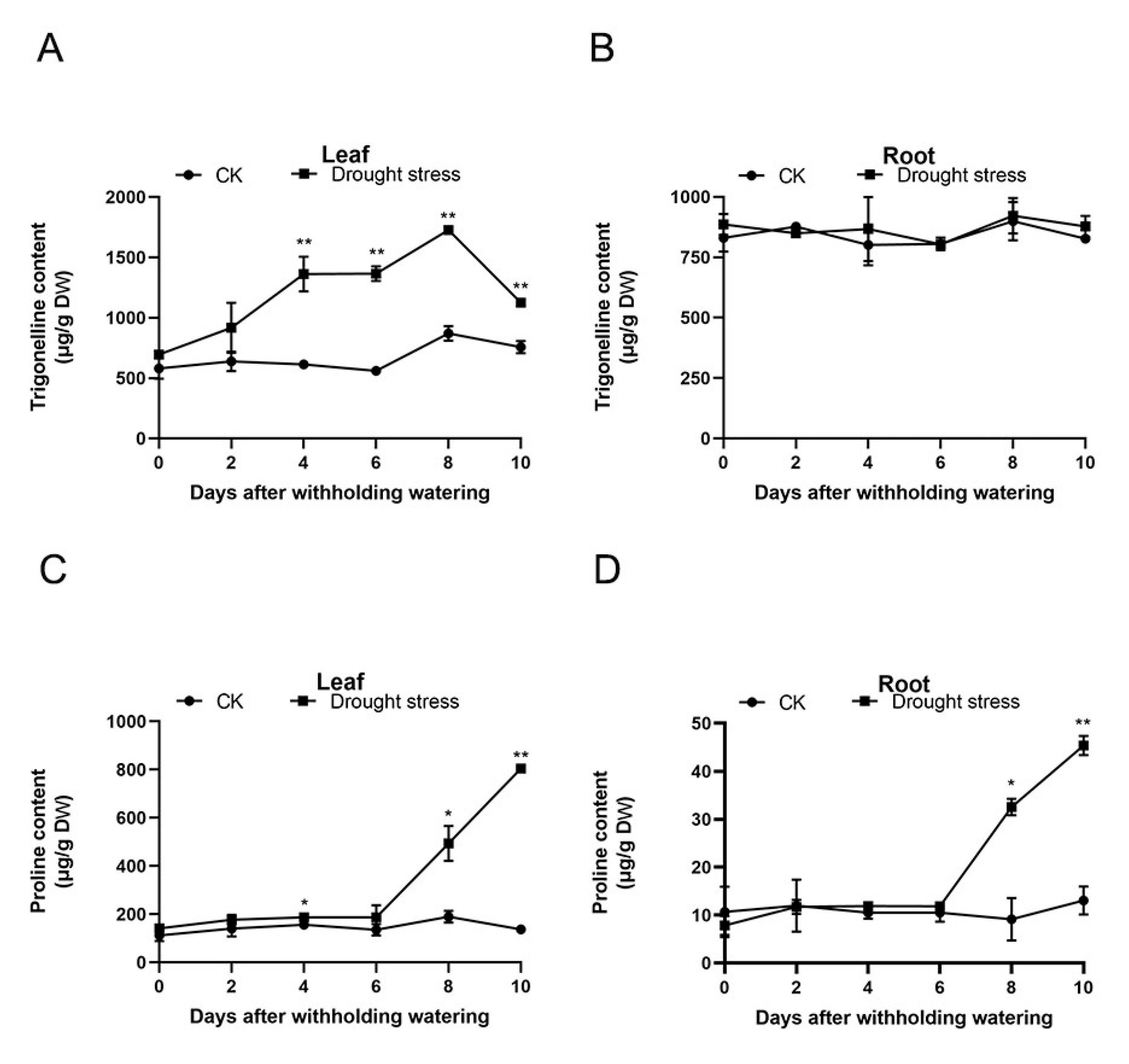
2.2. Changes in Trigonelline and Proline in A. catechu Seedlings under Drought Stress
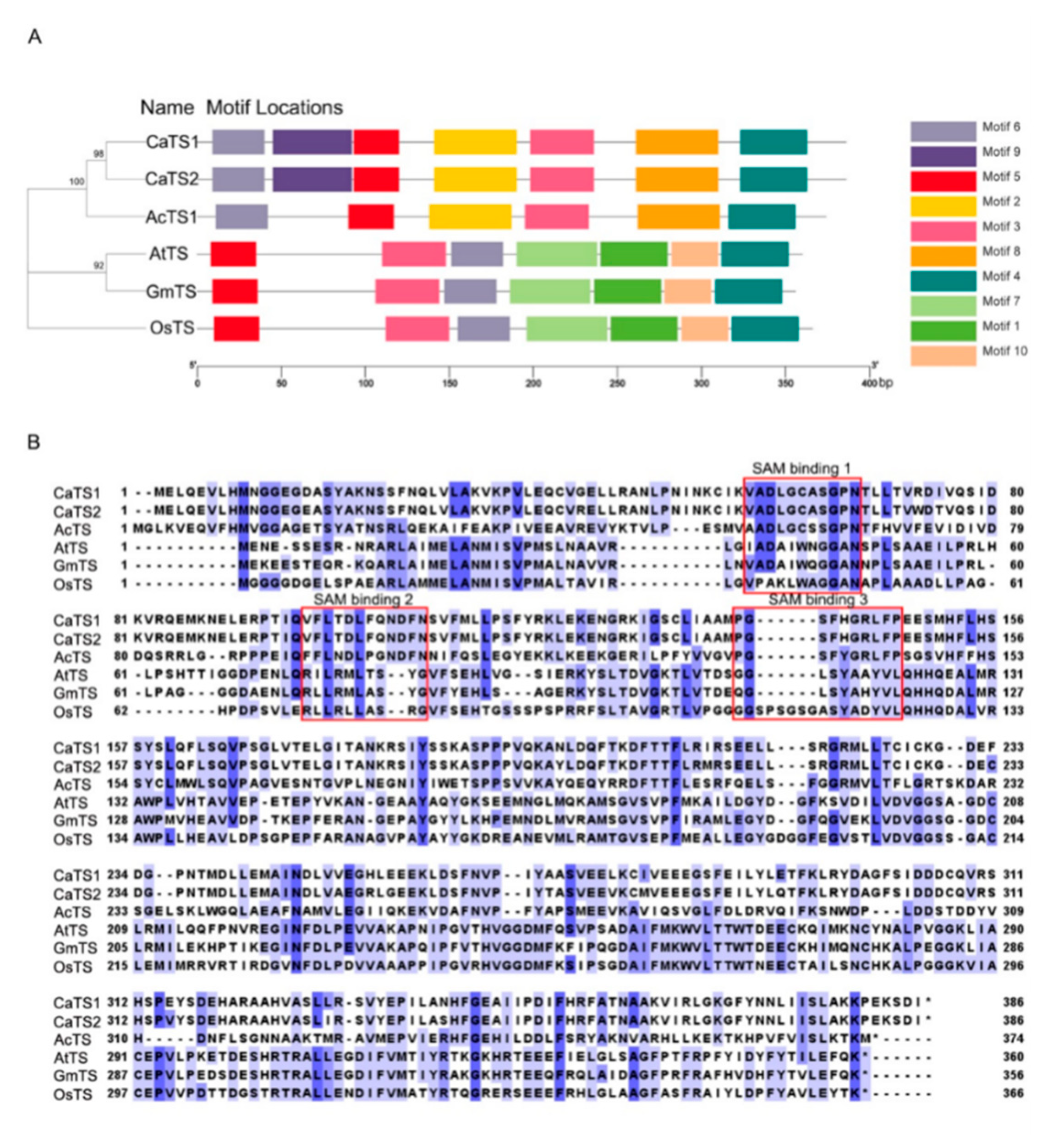
2.3. Defining the Trigonelline Synthesis Gene from the A. catechu Genome
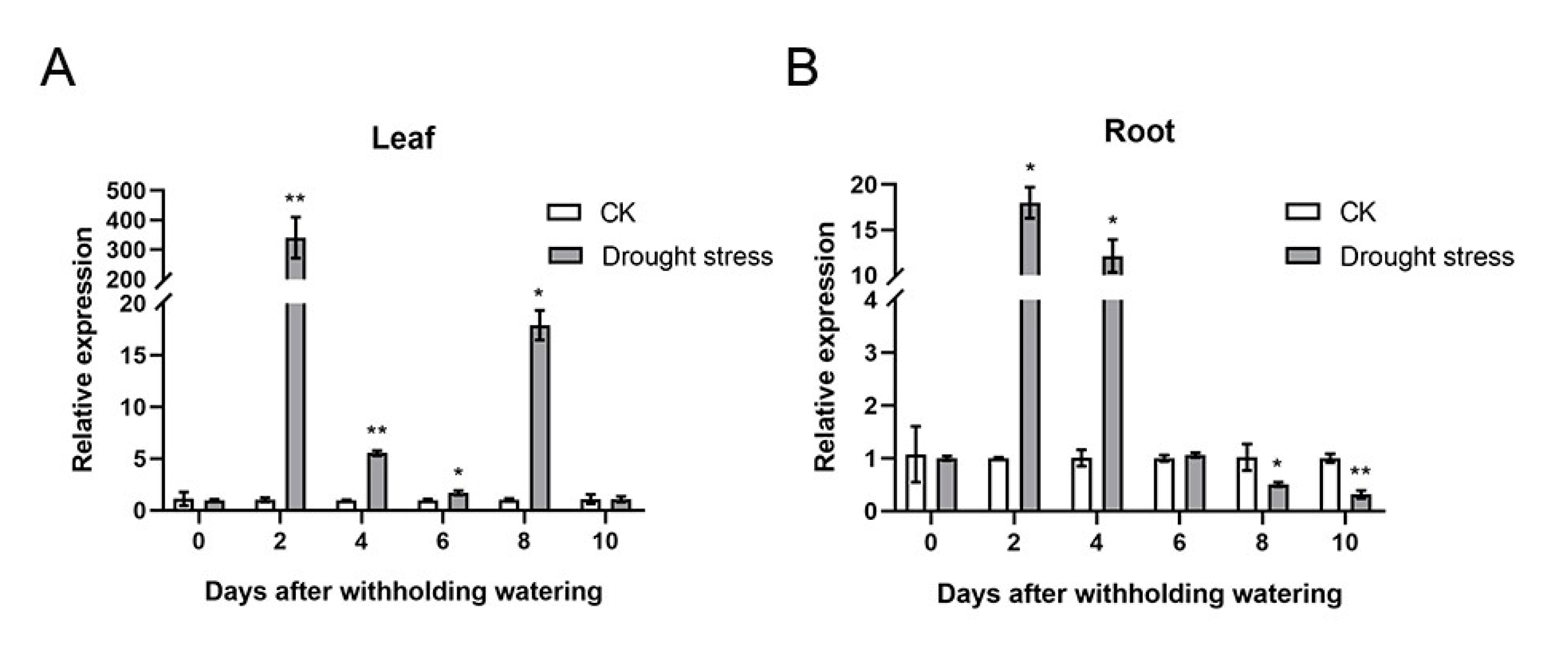
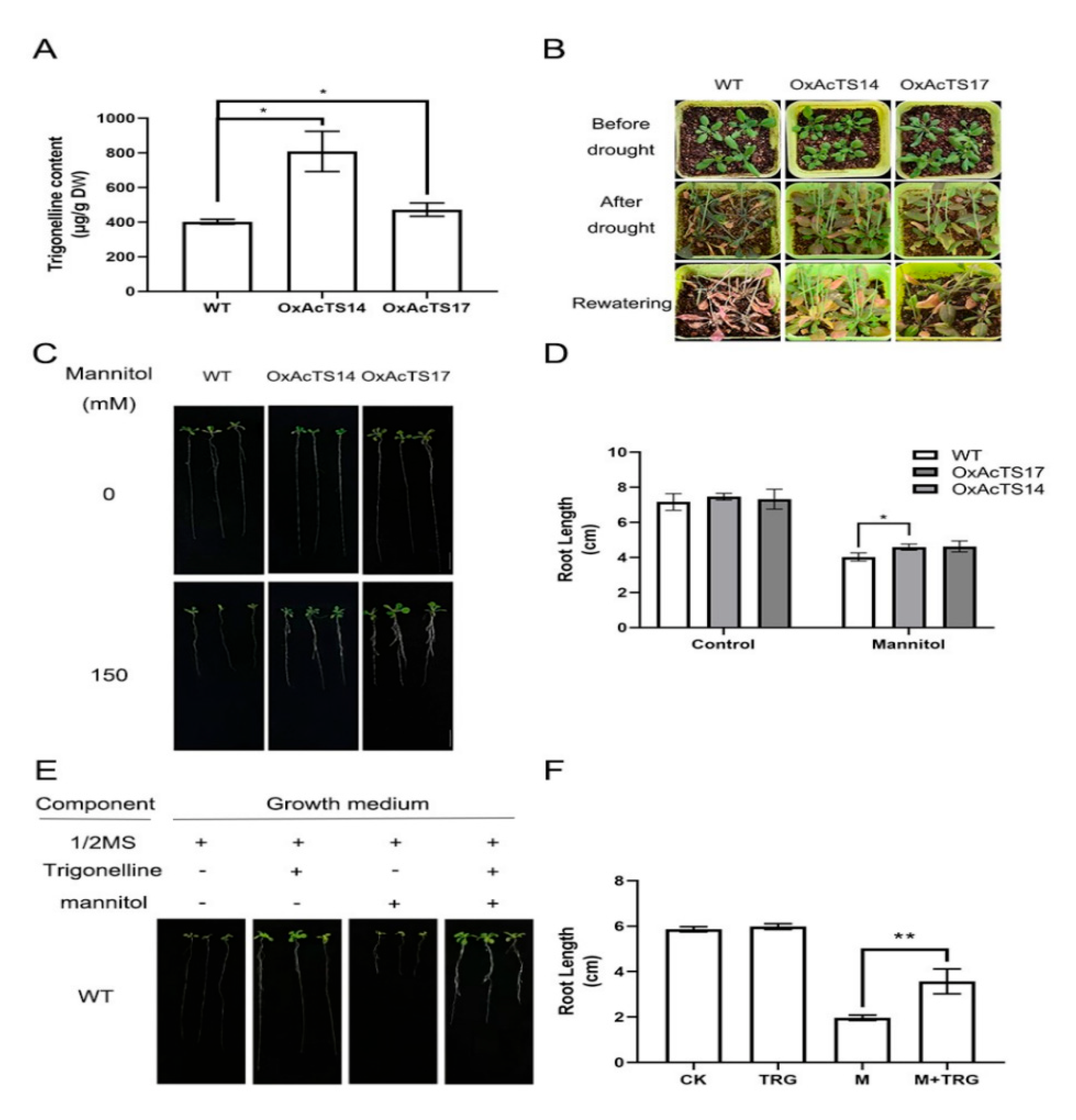
2.4. Overexpression of AcTS Increased the Drought Tolerance of Arabidopsis
3. Discussion
4. Materials and Methods
4.1. Plant Materials and Treatment
4.2. Determination of Chlorophyll Fluorescence Parameters
4.3. Determination of Trigonelline Content
4.4. Determination of Proline Content
4.5. Sequence Analysis of the Gene Encoding Trigonelline Synthase
4.6. Extraction of Total RNA
4.7. Cloning of the Full-Length AcTS CDS
4.8. Analysis of Quantitative Real Time PCR (qRT-PCR)
4.9. Generation of Transgenic Arabidopsis Overexpressing AcTS
4.10. Statistical Analysis
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Zhu, J.K. Salt and drought stress signal transduction in plants. Annu. Rev. Plant Biol. 2002, 53, 247–273. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ahuja, I.; de Vos, R.C.H.; Bones, A.M.; Hall, R.D. Plant molecular stress responses face climate change. Trends Plant Sci. 2010, 15, 664–674. [Google Scholar] [CrossRef]
- Gupta, A.; Rico-Medina, A.; Caño-Delgado, A.I. The physiology of plant responses to drought. Science 2020, 368, 266–269. [Google Scholar] [CrossRef] [PubMed]
- Chávez-Arias, C.C.; Ligarreto-Moreno, G.A.; Ramírez-Godoy, A.; Restrepo-Díaz, H. Maize responses challenged by drought, elevated daytime temperature and arthropod herbivory stresses: A physiological, biochemical and molecular view. Front. Plant Sci. 2021, 12, 702841. [Google Scholar] [CrossRef] [PubMed]
- Schroeder, J.I.; Kwak, J.M.; Allen, G.J. Guard cell abscisic acid signalling and engineering drought hardiness in plants. Nature 2001, 410, 327–330. [Google Scholar] [CrossRef] [PubMed]
- Djebbar, R.; Rzigui, T.; Pétriacq, P.; Mauve, C.; Priault, P.; Fresneau, C.; De Paepe, M.; Florez-Sarasa, I.; Benhassaine-Kesri, G.; Streb, P.; et al. Respiratory complex I deficiency induces drought tolerance by impacting leaf stomatal and hydraulic conductances. Planta 2012, 235, 603–614. [Google Scholar] [CrossRef] [PubMed]
- Mega, R.; Abe, F.; Kim, J.S.; Tsuboi, Y.; Tanaka, K.; Kobayashi, H.; Sakata, Y.; Hanada, K.; Tsujimoto, H.; Kikuchi, J.; et al. Tuning water-use efficiency and drought tolerance in wheat using abscisic acid receptors. Nat. Plants 2019, 5, 153–159. [Google Scholar] [CrossRef] [PubMed]
- Yeats, T.H.; Rose, J.K.C. The formation and function of plant cuticles. Physiol. Plant. 2013, 163, 5–20. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Xue, D.W.; Zhang, X.Q.; Lu, X.L.; Chen, G.; Chen, Z.H. Molecular and Evolutionary Mechanisms of Cuticular Wax for Plant Drought Tolerance. Front. Plant Sci. 2017, 8, 621. [Google Scholar] [CrossRef]
- Patwari, P.; Salewski, V.; Gutbrod, K.; Kreszies, T.; Dresen-Scholz, B.; Peisker, H.; Steiner, U.; Meyer, A.J.; Schreiber, L.; Dormann, P.; et al. Surface wax esters contribute to drought tolerance in Arabidopsis. Plant J. 2019, 98, 727–744. [Google Scholar] [CrossRef]
- Bhaskarla, V.; Zinta, G.; Ford, R.; Jain, M.; Varshney, R.K.; Mantri, N. Comparative root transcriptomics provide insights into drought adaptation strategies in chickpea (Cicer arietinum L.). Int. J. Mol. Sci. 2020, 21, 1781. [Google Scholar] [CrossRef] [Green Version]
- Boguszewska-Mańkowska, D.; Zarzyńska, K.; Nosalewicz, A. Drought differentially affects root system size and architecture of potato cultivars with differing drought tolerance. Am. J. Potato Res. 2020, 97, 54–62. [Google Scholar] [CrossRef]
- Blum, A. Osmotic adjustment is a prime drought stress adaptive engine in support of plant production. Plant Cell Environ. 2017, 40, 4–10. [Google Scholar] [CrossRef]
- Turner, N.C. Turgor maintenance by osmotic adjustment: 40 years of progress. J. Exp. Bot. 2018, 69, 3223–3233. [Google Scholar] [CrossRef] [Green Version]
- Ashihara, H. Trigonelline (N-methylnicotinic acid) biosynthesis and its biological role in plants. Nat. Prod. Commun. 2008, 3, 1423–1428. [Google Scholar] [CrossRef] [Green Version]
- Loukehaich, R.; Wang, T.T.; Ouyang, B.; Ziaf, K.; Li, H.X.; Zhang, J.H.; Lu, Y.E.; Ye, Z.B. SpUSP, an annexin-interacting universal stress protein, enhances drought tolerance in tomato. J. Exp. Bot. 2012, 63, 5593–5606. [Google Scholar] [CrossRef] [Green Version]
- Perchat, N.; Saaidi, P.L.; Darii, E.; Pellé, C.; Petit, J.L.; Besnard-Gonnet, M.; de Berardinis, V.; Dupont, M.; Gimbernat, A.; Salanoubat, M.; et al. Elucidation of the trigonelline degradation pathway reveals previously undescribed enzymes and metabolites. Proc. Nat. Acad. Sci. USA 2018, 115, E4358–E4367. [Google Scholar] [CrossRef] [Green Version]
- Ozturk, M.; Unal, B.T.; García-Caparrós, P.; Khursheed, A.; Gul, A.; Hasanuzzaman, M. Osmoregulation and its actions during the drought stress in plants. Physiol. Plant. 2021, 172, 1321–1335. [Google Scholar] [CrossRef]
- Mizuno, K.; Matsuzaki, M.; Kanazawa, S.; Tokiwano, T.; Yoshizawa, Y.; Kato, M. Conversion of nicotinic acid to trigonelline is catalyzed by N-methyltransferase belonged to motif B′ methyltransferase family in Coffea arabica. Biochem. Biophys. Res. Commun. 2014, 452, 1060–1066. [Google Scholar] [CrossRef]
- Minorsky, P.V. The Hot and the Classic. Plant Physiol. 2003, 131, 6–7. [Google Scholar] [CrossRef]
- Sasamoto, H.; Ashihara, H. Effect of nicotinic acid, nicotinamide and trigonelline on the proliferation of lettuce cells derived from protoplasts. Phytochem. Lett. 2014, 7, 38–41. [Google Scholar] [CrossRef]
- Chen, W.; Wang, W.S.; Peng, M.; Gong, L.; Gao, Y.Q.; Wan, J.; Wang, S.C.; Shi, L.; Zhou, B.; Li, Z.M.; et al. Comparative and parallel genome-wide association studies for metabolic and agronomic traits in cereals. Nat. Commun. 2016, 7, 12767. [Google Scholar] [CrossRef] [PubMed]
- Mazzuca, S.; Bitonti, M.B.; Innocenti, A.M.; Francis, D. Inactivation of DNA replication origins by the cell cycle regulator, trigonelline, in root meristems of Lactuca sativa. Planta 2000, 211, 127–132. [Google Scholar] [CrossRef] [PubMed]
- Jacob, V.; Chernyavskaya, Y.; Chen, X.T.; Tan, P.S.; Kent, B.; Hoshida, Y.; Sadler, K.C. DNA hypomethylation induces a DNA replication-associated cell cycle arrest to block hepatic outgrowth in uhrf1 mutant zebrafish embryos. Development 2015, 142, 510–521. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Berglund, T.; Wallström, A.; Nguyen, T.V.; Laurell, C.; Ohlsson, A.B. Nicotinamide; antioxidative and DNA hypomethylation effects in plant cells. Plant Physiol. Bioch. 2017, 118, 551–560. [Google Scholar] [CrossRef] [PubMed]
- Cho, Y.; Njiti, V.N.; Chen, X.; Lightfoot, D.A.; Wood, A.J. Trigonelline concentration in field-grown soybean in response to irrigation. Biol. Plant. 2003, 46, 405–410. [Google Scholar] [CrossRef]
- Schwartz, L.M.; Wood, A.J.; Gibson, D.J. Trigonelline accumulation in leaves of panicum virgatum seedlings. Nat. Prod. Commun. 2014, 58, 1750–1759. [Google Scholar] [CrossRef] [Green Version]
- Li, W.; Zhang, F.X.; Wu, R.; Jia, L.J.; Li, G.S.; Guo, Y.L.; Liu, C.M.; Wang, G.D. A novel N-methyltransferase in Arabidopsis appears to feed a conserved pathway for nicotinate detoxification among land plants and is associated with lignin biosynthesis. Plant Physiol. 2017, 174, 1492–1504. [Google Scholar] [CrossRef]
- Chen, X.; Wood, A.J. Purification and characterization of S-adenosyl-L-methionine nicotinic acid-N-methyltransferase from leaves of Glycine max. Biol. Plant. 2004, 48, 531–535. [Google Scholar] [CrossRef]
- Ali, N.S.; Khuwaja, A.K. Chapter 23—Betel nut (Areca catechu) usage and its effects on health. In Nuts and Seeds in Health and Disease Prevention; Academic Press: San Diego, CA, USA, 2011; pp. 197–204. [Google Scholar] [CrossRef]
- Wu, J.; Zhang, H.; Wang, S.; Yuan, L.; Grünhofer, P.; Schreiber, L.; Wan, Y. Tissue-specific and maturity-dependent distribution of pyridine alkaloids in Areca triandra. J. Plant Res. 2019, 132, 531–540. [Google Scholar] [CrossRef]
- Chung, C.M.; Kuo, T.M.; Yeh, K.T.; Lee, C.H.; Ko, Y.C. Reduction in and preventive effects for oral-cancer risk with antidepressant treatment. J. Pers. Med. 2021, 11, 591. [Google Scholar] [CrossRef]
- Lichtenthaler, H.K.; Miehé, J.A. Fluorescence imaging as a diagnostic tool for plant stress. Trends Plant Sci. 1997, 2, 316–320. [Google Scholar] [CrossRef]
- Baker, N.R. Chlorophyll fluorescence: A probe of photosynthesis in vivo. Annu. Rev. Plant Biol. 2008, 59, 89–113. [Google Scholar] [CrossRef] [Green Version]
- Murchie, E.H.; Lawson, T. Chlorophyll fluorescence analysis: A guide to good practice and understanding some new applications. J. Exp. Bot. 2013, 64, 3983–3998. [Google Scholar] [CrossRef] [Green Version]
- De Sousa, C.A.F.; de Paiva, D.S.; Casari, R.A.D.N.; de Oliveira, N.G.; Molinari, H.B.C.; Kobayashi, A.K.; Magalhaes, P.C.; Gomide, R.L.; Souza, M.T. A procedure for maize genotypes discrimination to drought by chlorophyll fluorescence imaging rapid light curves. Plant Methods 2017, 13, 61. [Google Scholar] [CrossRef] [Green Version]
- Yousef, A.F.; Ali, M.M.; Rizwan, H.M.; Tadda, S.A.; Kalaji, H.M.; Yang, H.; Ahmed, M.A.A.; Wróbel, J.; Xu, Y.; Chen, F. Photosynthetic apparatus performance of tomato seedlings grown under various combinations of LED illumination. PLoS ONE 2021, 16, e0249373. [Google Scholar] [CrossRef]
- Wang, C.K.; Lee, W.H.; Peng, C.H. Contents of phenolics and alkaloids in Areca catechu Linn. during maturation. J. Agr. Food Chem. 1997, 45, 1185–1188. [Google Scholar] [CrossRef]
- Holdsworth, D.K.; Jones, R.A.; Self, R. Volatile alkaloids from Areca catechu. Phytochemistry 1998, 48, 581–582. [Google Scholar] [CrossRef]
- Peng, W.; Liu, Y.J.; Wu, N.; Sun, T.; He, X.Y.; Gao, Y.X.; Wu, C.J. Areca catechu L. (Arecaceae): A review of its traditional uses, botany, phytochemistry, pharmacology and toxicology. J. Ethnopharmacol. 2015, 164, 340–356. [Google Scholar] [CrossRef]
- Su, Z.; Ma, X.; Guo, H.H.; Sukiran, N.L.; Guo, B.; Assmann, S.M.; Ma, H. Flower development under drought stress: Morphological and transcriptomic analyses reveal acute responses and long-term acclimation in Arabidopsis. Plant Cell 2013, 25, 3785–3807. [Google Scholar] [CrossRef] [Green Version]
- Anjum, S.A.; Ashraf, U.; Tanveer, M.; Khan, I.; Hussain, S.; Shahzad, B.; Zohaib, A.; Abbas, F.; Saleem, M.F.; Ali, I.; et al. Drought induced changes in growth, osmolyte accumulation and antioxidant metabolism of three maize hybrids. Front. Plant Sci. 2017, 8, 69. [Google Scholar] [CrossRef]
- Szabados, L.; Savoure, A. Proline: A multifunctional amino acid. Trends Plant Sci. 2010, 15, 89–97. [Google Scholar] [CrossRef]
- Zulfiqar, F.; Akram, N.A.; Ashraf, M. Osmoprotection in plants under abiotic stresses: New insights into a classical phenomenon. Planta 2019, 251, 3. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wu, J.; Cui, C.; Zhang, H.; Liu, D.J.; Schreiber, L.; Qin, W.Q.; Wan, Y.L. Identifying new compounds with potential pharmaceutical and physiological activity in Areca catechu and Areca triandra via a non-targeted metabolomic approach. Phytochem. Anal. 2021, 32, 970–981. [Google Scholar] [CrossRef]
- Zhang, X.R.; Henriques, R.; Lin, S.S.; Niu, Q.W.; Chua, N.H. Agrobacterium-mediated transformation of Arabidopsis thaliana using the floral dip method. Nat. Protoc. 2006, 1, 641–646. [Google Scholar] [CrossRef]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Li, Y.; Ding, M.; Cui, C.; An, Q.; Wu, J.; Zhou, G.; Wan, Y.; Bao, W. Overexpression of a Gene Encoding Trigonelline Synthase from Areca catechu L. Promotes Drought Resilience in Transgenic Arabidopsis. Plants 2022, 11, 487. https://doi.org/10.3390/plants11040487
Li Y, Ding M, Cui C, An Q, Wu J, Zhou G, Wan Y, Bao W. Overexpression of a Gene Encoding Trigonelline Synthase from Areca catechu L. Promotes Drought Resilience in Transgenic Arabidopsis. Plants. 2022; 11(4):487. https://doi.org/10.3390/plants11040487
Chicago/Turabian StyleLi, Yilin, Mengying Ding, Chuang Cui, Qiyuan An, Jiao Wu, Guangzhen Zhou, Yinglang Wan, and Wenlong Bao. 2022. "Overexpression of a Gene Encoding Trigonelline Synthase from Areca catechu L. Promotes Drought Resilience in Transgenic Arabidopsis" Plants 11, no. 4: 487. https://doi.org/10.3390/plants11040487
APA StyleLi, Y., Ding, M., Cui, C., An, Q., Wu, J., Zhou, G., Wan, Y., & Bao, W. (2022). Overexpression of a Gene Encoding Trigonelline Synthase from Areca catechu L. Promotes Drought Resilience in Transgenic Arabidopsis. Plants, 11(4), 487. https://doi.org/10.3390/plants11040487





