First Report of Phaeoacremonium iranianum Causing Olive Twig and Branch Dieback
Abstract
1. Introduction
2. Materials and Methods
2.1. Sampling and Fungal Isolation
2.2. Morphological and Molecular Identification
2.3. Pathogenicity Tests of Isolate
3. Results
3.1. Sampling and Fungal Isolation
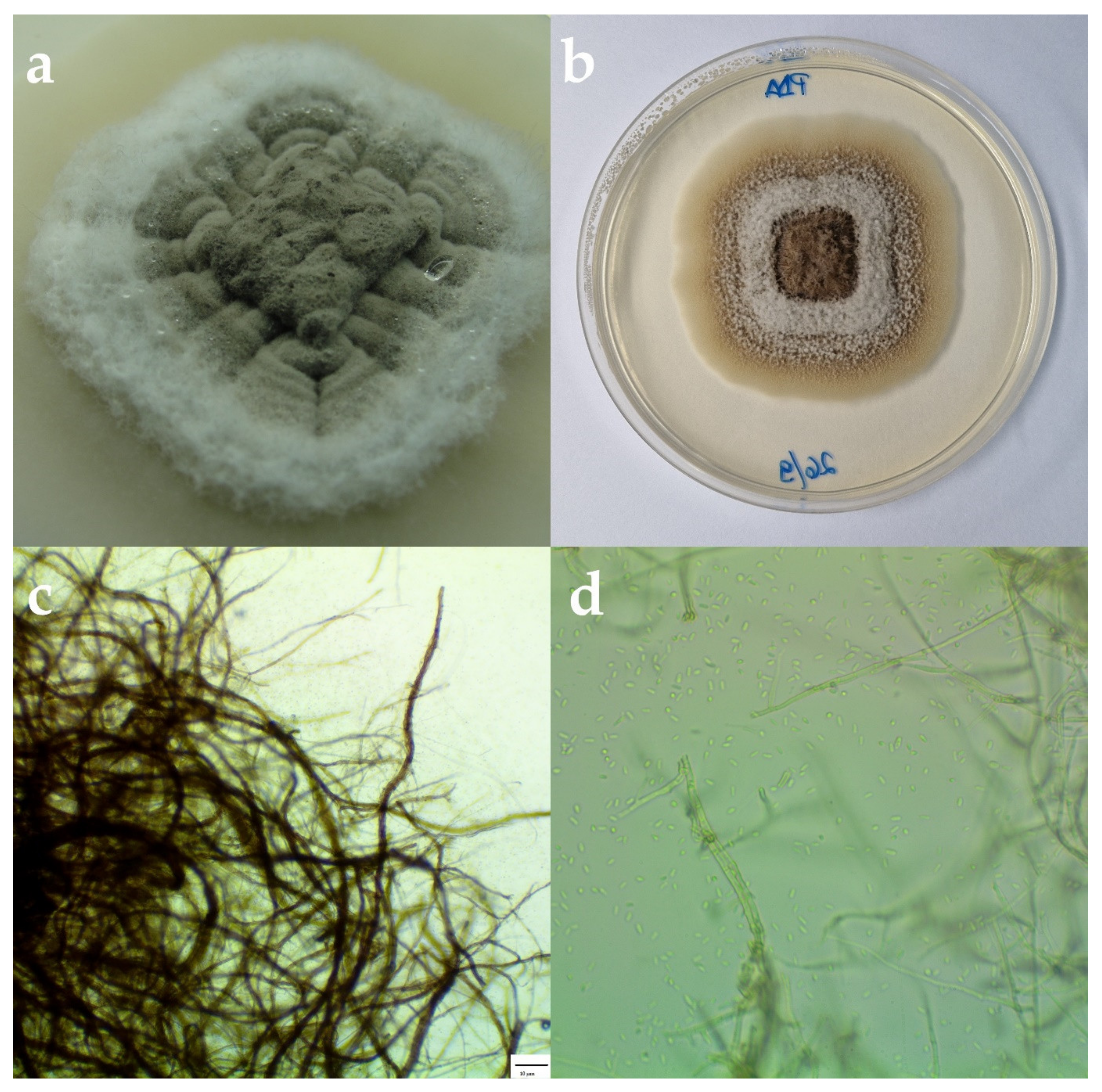
3.2. Morphological and Molecular Identification
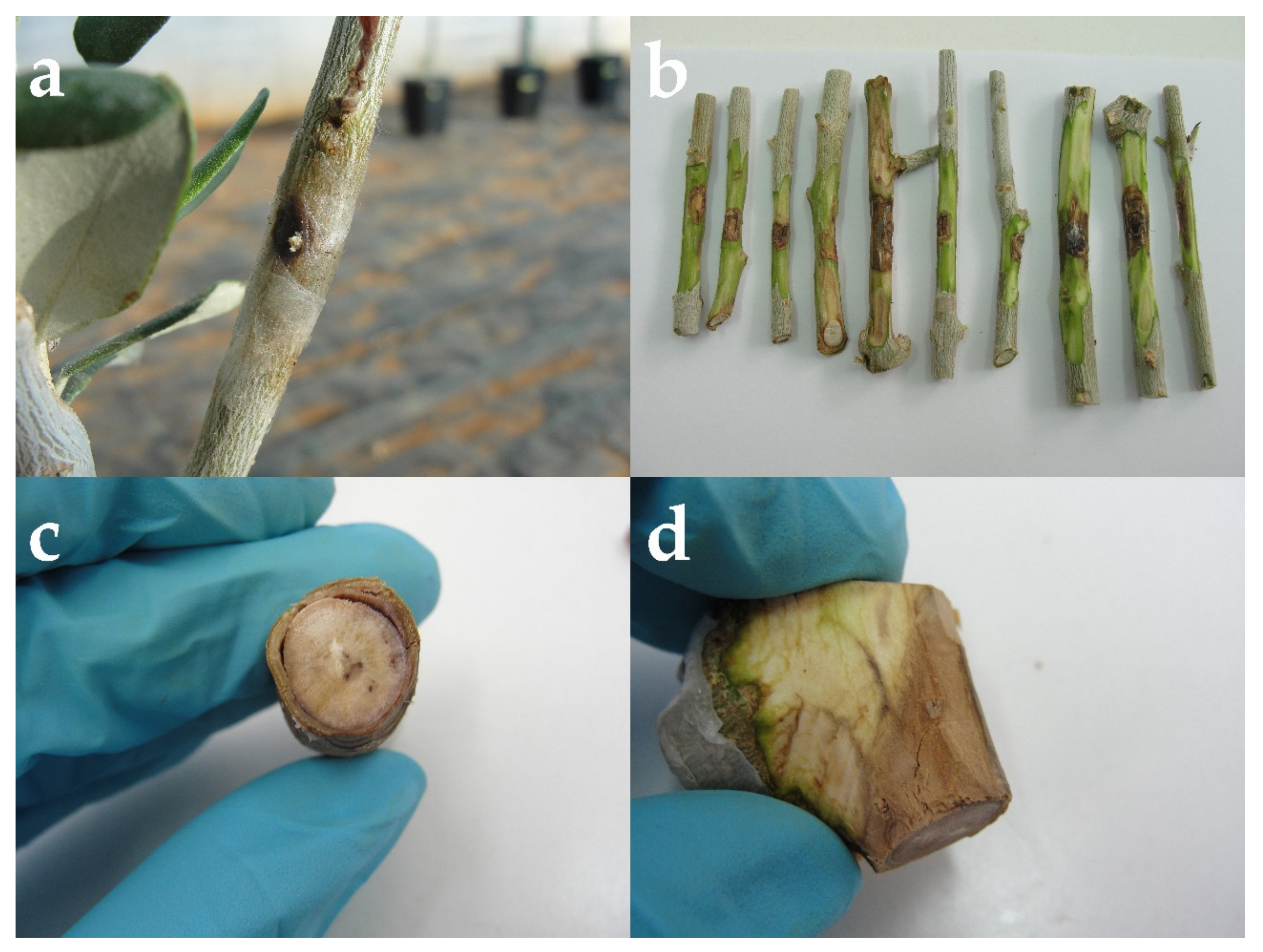
3.3. Pathogenicity Tests of Isolate
4. Discussion
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Croatian Bureau of Statistics. Available online: https://podaci.dzs.hr/2021/hr/10118 (accessed on 25 October 2022).
- Phillips, A.J.L.; Rumbos, I.C.; Alves, A.; Correia, A. Morphology and phylogeny of Botryosphaeria dothidea causing fruit rot of olives. Mycopathologia 2005, 159, 433–439. [Google Scholar] [CrossRef] [PubMed]
- White, T.J.; Bruns, T.D.; Lee, S.B.; Taylor, J.W. 38—Amplification and direct sequencing of fungal ribosomal RNA Genes for phylogenetics. In PCR—Protocols and Applications—A Laboratory Manual; Innis, M.A., Gelfand, D.H., Sninsky, J.J., White, T.J., Eds.; Academic Press, Inc.: New York, NY, USA, 1990; pp. 315–322. [Google Scholar]
- Glass, N.L.; Donaldson, G.C. Development of primer sets designed for use with the PCR to amplify conserved genes from filamentous ascomycetes. Appl. Environ. Microbiol. 1995, 61, 1323–1330. [Google Scholar] [CrossRef] [PubMed]
- Carbone, I.; Kohn, L.M. A Method for Designing Primer Sets for Speciation Studies in Filamentous Ascomycetes. Mycologia 1995, 91, 553–556. [Google Scholar] [CrossRef]
- Mostert, L.; Groenewald, J.Z.; Summerbell, R.C.; Gams, W.; Crous, P.W. Taxonomy and Pathology of Togninia (Diaporthales) and its Phaeoacremonium Anamorphs. Stud. Mycol. 2006, 54, 1–113. [Google Scholar] [CrossRef]
- European and Mediterranean Plant Protection Organization (EPPO). PM 7/129 (1) DNA barcoding as an identification tool for a number of regulated pests. Bull. OEPP/EPPO Bull. 2016, 46, 501–537. [Google Scholar] [CrossRef]
- Van Dyk, M.; Spies, C.F.J.; Mostert, L.; van der Rijst, M.; du Plessis, I.L.; Moyo, P.; van Jaarsveld, W.J.; Halleen, F. Pathogenicity Testing of Fungal Isolates Associated with Olive Trunk Diseases in South Africa. Plant Dis. 2022, 105, 4060–4073. [Google Scholar] [CrossRef]
- Spies, C.F.J.; Mostert, L.; Carlucci, A.; Moyo, P.; van Jaarsveld, W.J.; du Plessis, I.L.; van Dyk, M.; Halleen, F. Dieback and decline pathogens of olive trees in South Africa. Persoonia 2020, 45, 195–220. [Google Scholar] [CrossRef] [PubMed]
- Carlucci, A.; Lops, F.; Cibelli, F.; Raimondo, M.L. Phaeoacremonium species associated with olive wilt and decline in southern Italy. Eur. J. Plant Pathol. 2015, 141, 717–729. [Google Scholar] [CrossRef]
- Raimondo, M.L.; Lops, F.; Carlucci, A. First Report of Phaeoacremonium oleae and P. viticola Associated with Olive Trunk Diseases in Italy. Plant Dis. 2022, 106, 331. [Google Scholar] [CrossRef]
- Agusti-Brisach, C.; Jimenez-Urbano, J.P.; Raya, M.D.; Lopez-Moral, A.; Trapero, A. Vascular Fungi Associated with Branch Dieback of Olive in Super-High-Density Systems in Southern Spain. Plant Dis. 2021, 106, 797–818. [Google Scholar] [CrossRef]
- Carlucci, A.; Raimondo, M.L.; Cibelli, F.; Phillips, A.J.L.; Lops, F. Pleurostomophora richardsiae, Neofusicoccum parvum, and Phaeoacremonium aleophilum associated with a decline of olives in southern Italy. Phytopathol. Mediterr. 2013, 52, 517–527. [Google Scholar]
- Úrbez-Torres, J.R.; Peduto, F.; Vossen, P.M.; Krueger, W.H.; Gubler, W.D. Olive Twig and Branch Dieback: Etiology, Incidence, and Distribution in California. Plant Dis. 2013, 97, 231–244. [Google Scholar] [CrossRef] [PubMed]
- Van Dyk, M.; Spies, C.F.J.; Mostert, L.; Halleen, F. Survey of Trunk Pathogens in South African Olive Nurseries. Plant Dis. 2021, 105, 1630–1639. [Google Scholar] [CrossRef] [PubMed]
- Gramaje, D.; Agustí-Brisach, C.; Pérez-Sierra, A.; Moralejo, E.; Olmo, D.; Mostert, L.; Damm, U.; Armengol, J. Fungal trunk pathogens associated with wood decay of almond trees on Mallorca (Spain). Persoonia 2012, 28, 1–13. [Google Scholar] [CrossRef]
- Olmo, D.; Armengol, J.; Leon, M.; Gramaje, D. Pathogenicity testing of lesser-known fungal trunk pathogens associated with wood decay of almond trees. Eur. J. Plant Pathol. 2015, 143, 607–611. [Google Scholar] [CrossRef]
- Espargham, N.; Mohammadi, H.; Gramaje, D. A Survey of Trunk Disease Pathogens within Citrus Trees in Iran. Plant 2020, 9, 754. [Google Scholar] [CrossRef]
- Mohamadi, H.; Kazemi, S.; Farahmand, H. Phaeoacremonium and Botryosphaeriaceae species associated with cypress (Cupressus sempervirens L.) decline in Kerman province (Iran). Phytopathol. Mediterr. 2014, 53, 27–39. [Google Scholar]
- Kazemzadeh Chakusary, M.; Mohammadi, H.; Khodaparast, S.A. Decline-associated Phaeoacremonium species occurring on forest trees in the north of Iran. For. Pathol. 2017, 47, e12368. [Google Scholar] [CrossRef]
- Gramaje, D.; Armengol, J.; Colino, M.I.; Santiago, R.; Moralejo, E.; Olmo, D.; Luque, J.; Mostert, L. First Report of Phaeoacremonium inflatipes, P. iranianum, and P. sicilianum Causing Petri Disease of Grapevine in Spain. Plant Dis. 2009, 93, 964–965. [Google Scholar] [CrossRef]
- White, C.L.; Halleen, F.; Fischer, M.; Mostert, L. Characterisation of the fungi associated with esca diseased grapevines in South Africa. Phytopahologia Mediterr. 2011, 50, S204–S223. [Google Scholar]
- Úrbez-Torres, J.R.; Haag, P.; Bowen, P.; O’Gorman, D.T. Grapevine Trunk Diseases in British Columbia: Incidence and Characterization of the Fungal Pathogens Associated with Esca and Petri Diseases of Grapevine. Plant Dis. 2014, 98, 469–482. [Google Scholar] [CrossRef] [PubMed]
- Baloyi, M.A.; Mostert, L.; Halleen, F. Pathogenicity of ten Phaeoacremonium species associated with esca and Petri disease of Grapevine. Phytopathol. Mediterr. 2018, 57, 538–546. [Google Scholar]
- Agusti-Brisach, C.; Lopez-Moral, A.; Raya-Ortega, M.C.; Franco, R.; Roca-Castillo, L.F.; Trapero, A. Occurrence of grapevine trunk diseases affecting the native cultivar Pedro Ximenez in southern Spain. Eur. J. Plant Pathol. 2019, 153, 599–625. [Google Scholar] [CrossRef]
- Halleen, F.; Baloyi, M.A.; Bester, M.C.; Mostert, L. Aerial inoculum patterns of Petri disease pathogens in South African vineyards and rootstock mother blocks. Phytopathol. Mediterr. 2020, 59, 515–536. [Google Scholar]
- Aigoun-Mouhous, W.; Mahamedi, A.E.; León, M.; Chaouia, C.; Zitouni, A.; Barankova, K.; Eichmeier, A.; Armengol, J.; Gramaje, D.; Berraf-Tebbal, A. Cadophora sabaouae sp. nov. and Phaeoacremonium Species Associated with Petri Disease on Grapevine Propagation Material and Young Grapevines in Algeria. Plant Dis. 2021, 105, 3657–3668. [Google Scholar] [CrossRef] [PubMed]
- Sami, S.; Mohammadi, H.; Heydarnejad, J. Phaeoacremonium species associated with necrotic wood of pome fruit trees in Iran. J. Plant Pathol. 2014, 96, 487–495. [Google Scholar]
- Damm, U.; Mostert, L.; Crous, P.W.; Fourie, P.H. Novel Phaeoacremonium species associated with necrotic wood of Prunus trees. Persoonia 2008, 20, 87–102. [Google Scholar] [CrossRef] [PubMed]
- Alves, A.; Correia, A.; Phillips, A.J.L. Multi-gene genealogies and morphological data support Diplodia cupressi sp. nov., previouly recognized as D. pinea f. sp. cupressi, as a distinct species. Fungal Divers. 2006, 23, 1–15. [Google Scholar]
- Saitou, N.; Nei, M. The neighbor-joining method: A new method for reconstructing phylogenetic trees. Mol. Biol. Evol. 1987, 4, 406–425. [Google Scholar]
- Tamura, K.; Nei, M.; Kumar, S. Prospects for inferring very large phylogenies by using the neighbor-joining method. Proc. Natl. Acad. Sci. USA 2004, 101, 11030–11035. [Google Scholar] [CrossRef]
- Tamura, K.; Stecher, G.; Kumar, S. MEGA 11: Molecular Evolutionary Genetics Analysis Version 11. Mol. Biol. Evol. 2021, 38, 3022–3027. [Google Scholar] [CrossRef] [PubMed]

Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Petrović, E.; Vrandečić, K.; Ćosić, J.; Kanižai Šarić, G.; Godena, S. First Report of Phaeoacremonium iranianum Causing Olive Twig and Branch Dieback. Plants 2022, 11, 3578. https://doi.org/10.3390/plants11243578
Petrović E, Vrandečić K, Ćosić J, Kanižai Šarić G, Godena S. First Report of Phaeoacremonium iranianum Causing Olive Twig and Branch Dieback. Plants. 2022; 11(24):3578. https://doi.org/10.3390/plants11243578
Chicago/Turabian StylePetrović, Elena, Karolina Vrandečić, Jasenka Ćosić, Gabriella Kanižai Šarić, and Sara Godena. 2022. "First Report of Phaeoacremonium iranianum Causing Olive Twig and Branch Dieback" Plants 11, no. 24: 3578. https://doi.org/10.3390/plants11243578
APA StylePetrović, E., Vrandečić, K., Ćosić, J., Kanižai Šarić, G., & Godena, S. (2022). First Report of Phaeoacremonium iranianum Causing Olive Twig and Branch Dieback. Plants, 11(24), 3578. https://doi.org/10.3390/plants11243578






