Understanding Molecular Plant–Nematode Interactions to Develop Alternative Approaches for Nematode Control
Abstract
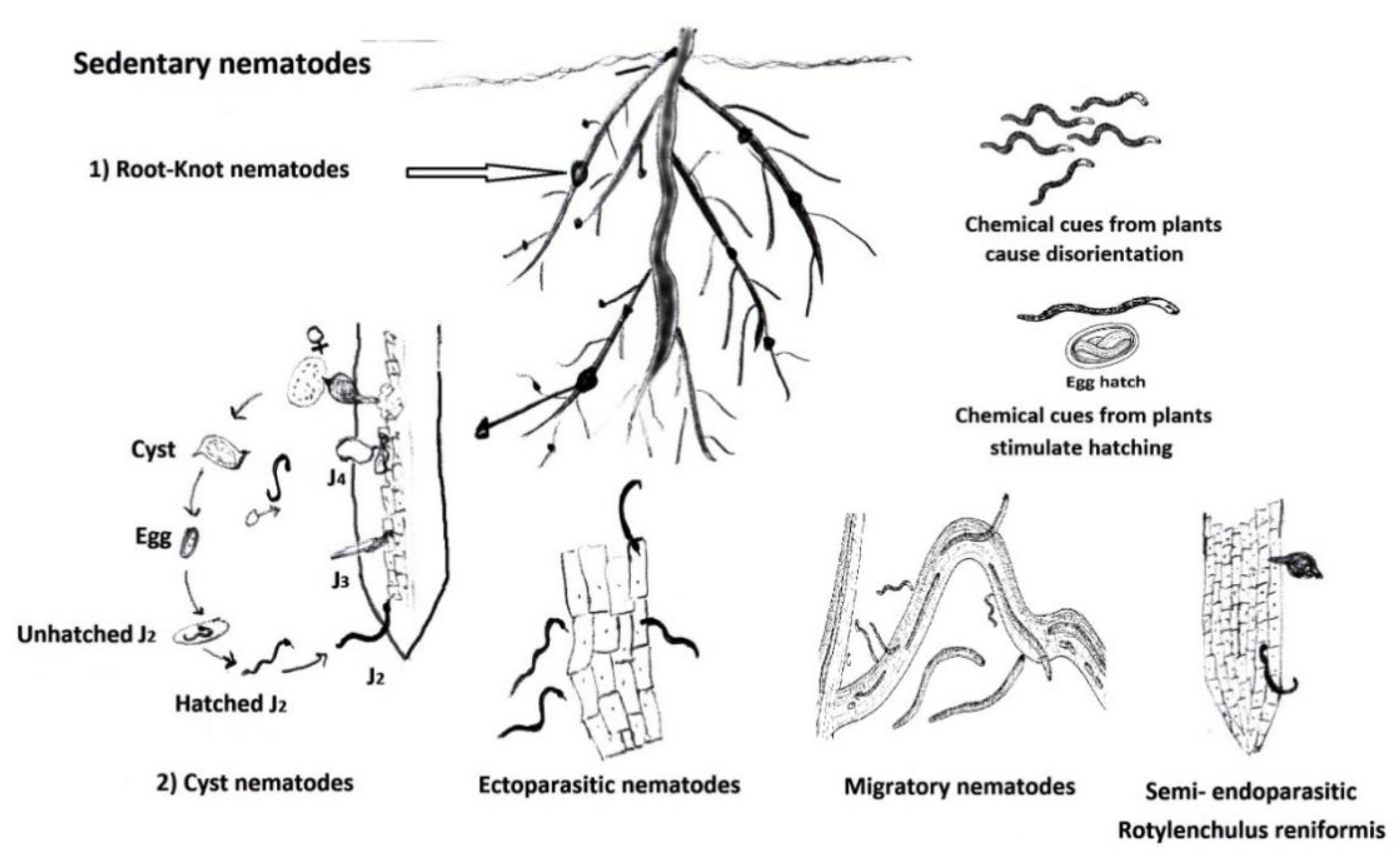
:1. Introduction
2. General PPN Categories and Management Measures
3. The Mechanism of Natural Resistance
4. Successes and Difficulties in Using R-Genes
5. Common Issues of Natural Plant Resistance
5.1. Resistance Breaking Nematode Pathotypes
5.2. Genetics of Virulence in Nematodes
5.3. The Temperature Factor
5.4. Improper Research Methods and Tools
6. General Approaches to Solve the Related Issues
7. Approaches to Strengthen Molecular PPN Control
7.1. Expanding the Use of Marker-Assisted Selection
| Crop | Nematode Species | Resistance Genes | Marker Type | References |
|---|---|---|---|---|
| Tomato | Meloidogyne incognita | Mi 3 | RAPD and RFLP | [92] |
| Eggplant | Meloidogyne javanica | Mi-1.2 | RT-PCR | [93] |
| Wheat | Heterodera avenae | CreX and CreY | SCAR | [94] |
| Pepper | M. incognita, M. arenaria, M. javanica | Me3 and Me4 | RAPD and AFLP | [95] |
| Potato | Globodera rostochinensis | H1 | RFLP | [96] |
| Soybean | Heterodera glycines | Rhg1 and Rhg4 | SNPs | [97] |
| Cucumber | M. javanica | mj | AFLP | [98] |
| Cotton | M. incognita | qMi-C14 | SSR | [99] |
| Cotton | Rotylenchulus reniformis | Renari | SSR | [100] |
| Peanut | Meloidogyne arenaria | Rma | CAPS, SSR, AFLP | [101] |
7.2. Utilizing Proteinase Inhibitor Coding Genes
7.3. Use of RNA Interference
7.4. Nematicidal Proteins
7.5. Chemodisruptive Peptides
7.6. Employing Plant Resistance Mechanisms
7.7. Related Molecular Tools
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Abd-Elgawad, M.M.M.; Askary, T.H. Impact of phytonematodes on agriculture economy. In Biocontrol Agents of Phytonematodes; Askary, T.H., Martinelli, P.R.P., Eds.; CABI: Wallingford, UK, 2015; pp. 3–49. [Google Scholar]
- Forghani, F.; Hajihassani, A. Recent advances in the development of environmentally benign treatments to control root-knot nematodes. Front. Plant Sci. 2020, 11, 1125. [Google Scholar] [CrossRef] [PubMed]
- Abd-Elgawad, M.M.M. Optimizing safe approaches to manage plant-parasitic nematodes. Plants 2021, 10, 1911. [Google Scholar] [CrossRef] [PubMed]
- Ochola, J.; Cortada, L.; Ng’ang’a, M.; Hassanali, A.; Coyne, D.; Torto, B. Mediation of potato–potato cyst nematode, G. rostochiensis interaction by specific root exudate compounds. Front. Plant Sci. 2020, 11, 649. [Google Scholar] [CrossRef] [PubMed]
- Sikora, R.A.; Coyne, D.; Hallmann, J.; Timper, P. Plant Parasitic Nematodes in Subtropical and Tropical Agriculture, 3rd ed.; CABI: Cambridge, UK, 2018. [Google Scholar]
- Quentin, M.; Abad, P.; Favery, B.; Rivas, S.; Reymond, P. Plant parasitic nematode effectors target host defense and nuclear functions to establish feeding cells. Front. Plant Sci. 2013, 4, 53. [Google Scholar] [CrossRef] [PubMed]
- Molinari, S. Can the plant immune system be activated against animal parasites such as nematodes? Nematology 2020, 22, 481–492. [Google Scholar] [CrossRef]
- Fuller, V.L.; Lilley, C.J.; Urwin, P.E. Nematode resistance. New Phytol. 2008, 180, 27–44. [Google Scholar] [CrossRef]
- El-Sappah, A.H.; Islam, M.M.; El-Awady, H.H.; Yan, S.; Qi, S.; Liu, J.; Cheng, G.-T.; Liang, Y. Tomato natural resistance genes in controlling the root-knot nematode. Genes 2019, 10, 925. [Google Scholar] [CrossRef]
- Bobardt, S.D.; Dillman, A.R.; Nair, M.G. The two faces of nematode infection: Virulence and immunomodulatory molecules from nematode parasites of mammals, insects and plants. Front. Microbiol. 2020, 11, 577846. [Google Scholar] [CrossRef]
- Habash, S.S.; Brass, H.U.C.; Klein, A.S.; Klebl, D.P.; Weber, T.M.; Classen, T.; Pietruszka, J.; Grundler, F.M.W.; Schleker, A.S.S. Novel prodiginine derivatives demonstrate bioactivities on plants, nematodes, and fungi. Front. Plant Sci. 2020, 11, 579807. [Google Scholar] [CrossRef]
- Cabral, D.; Forero Ballesteros, H.; de-Melo, B.P.; Lourenço-Tessutti, I.T.; Simões de Siqueira, K.M.; Obicci, L.; Grossi-de-Sa, M.F.; Hemerly, A.S.; de Almeida Engler, J. The armadillo BTB protein ABAP1 is a crucial player in DNA replication and transcription of nematode-induced galls. Front. Plant Sci. 2020, 12, 636663. [Google Scholar] [CrossRef]
- Zheng, Q.; Putker, V.; Goverse, A. Molecular and cellular mechanisms involved in host-specific resistance to cyst nematodes in crops. Front. Plant Sci. 2021, 12, 641582. [Google Scholar] [CrossRef]
- Blanc-Mathieu, R.; Perfus-Barbeoch, L.; Aury, J.-M.; Da Rocha, M.; Gouzy, J.; Sallet, E. Hybridization and polyploidy enable genomic plasticity without sex in the most devastating plant-parasitic nematodes. PLoS Genet. 2017, 13, e1006777. [Google Scholar] [CrossRef]
- Jaubert-Possamai, S.; Noureddine, Y.; Favery, B. MicroRNAs, new players in the plant–nematode interaction. Front. Plant Sci. 2019, 10, 1180. [Google Scholar] [CrossRef]
- Ibrahim, H.M.M.; Ahmad, E.M.; Martínez-Medina, A.; Aly, M.A.M. Effective approaches to study the plant-root knot nematode interaction. Plant Physiol. Biochem. 2019, 141, 332–342. [Google Scholar] [CrossRef]
- Hada, A.; Kumari, C.; Phani, V.; Singh, D.; Chinnusamy, V.; Rao, U. Host-induced silencing of FMR Famide-like peptide genes, flp-1 and flp-12, in rice impairs reproductive fitness of the root-knot nematode Meloidogyne graminicola. Front. Plant Sci. 2020, 11, 894. [Google Scholar] [CrossRef]
- Hada, A.; Singh, D.; Papolu, P.K.; Banakar, P.; Raj, A.; Rao, U. Host-mediated RNAi for simultaneous silencing of different functional groups of genes in Meloidogyne incognita using fusion cassettes in Nicotiana tabacum. Plant Cell Rep. 2021, 40, 2287–2302. [Google Scholar] [CrossRef]
- Hada, A.; Patil, B.L.; Bajpai, A.; Kesiraju, K.; Dinesh-Kumar, S.; Paraselli, B.; Sreevathsa, R.; Rao, U. Micro RNA-induced gene silencing strategy for the delivery of siRNAs targeting Meloidogyne incognita in a model plant Nicotiana benthamiana. Pest Manag. Sci. 2021, 77, 3396–3405. [Google Scholar] [CrossRef]
- Shinya, R.; Kirino, H.; Morisaka, H.; Takeuchi-Kaneko, Y.; Futai, K.; Ueda, M. Comparative secretome and functional analyses reveal glycoside hydrolase family 30 and cysteine peptidase as virulence determinants in the pinewood nematode Bursaphelenchus xylophilus. Front. Plant Sci. 2021, 12, 640459. [Google Scholar] [CrossRef]
- Chitwood, D.J. Nematicides. In Encyclopedia of Agrochemicals; Plimmer, J.R., Ed.; John Wiley & Sons: New York, NY, USA, 2003; Volume 3, pp. 1104–1115. [Google Scholar]
- Abd-Elgawad, M.M.M.; Kour, F.F.H.; Montasser, S.A.; Hammam, M.M.A. Distribution and losses of Tylenchulus semipenetrans in citrus orchards on reclaimed land in Egypt. Nematology 2016, 18, 1141–1150. [Google Scholar] [CrossRef]
- Abd-Elgawad, M.M.M.; Askary, T.H. Factors affecting success of biological agents used in controlling plant-parasitic nematodes. Egypt. J. Biol. Pest. Cont. 2020, 30, 17. [Google Scholar] [CrossRef]
- Timper, P. Conserving and enhancing biological control of nematodes. J. Nematol. 2014, 46, 75–89. [Google Scholar]
- Ntalli, N.; Adamski, Z.; Doula, M.; Monokrousos, N. Nematicidal amendments and soil remediation. Plants 2020, 9, 429. [Google Scholar] [CrossRef]
- Dutta, T.K.; Khan, M.R.; Phani, V. Plant-parasitic nematode management via biofumigation using brassica and non-brassica plants: Current status and future prospects. Curr. Plant Biol. 2019, 17, 17–32. [Google Scholar] [CrossRef]
- Ali, M.A.; Azeem, F.; Abbas, A.; Joyia, F.A.; Li, H.; Dababat, A.A. Transgenic strategies for enhancement of nematode resistance in plants. Front. Plant Sci. 2017, 8, 750. [Google Scholar] [CrossRef]
- Sasanelli, N.; Konrat, A.; Migunova, V.; Toderas, I.; Iurcu-Straistaru, E.; Rusu, S.; Bivol, A.; Andoni, C.; Veronico, P. Review on control methods against plant parasitic nematodes applied in southern member states (C Zone) of the European Union. Agriculture 2021, 11, 602. [Google Scholar] [CrossRef]
- Abd-Elgawad, M.M.M. Optimizing sampling and extraction methods for plant-parasitic and entomopathogenic nematodes. Plants 2021, 10, 629. [Google Scholar] [CrossRef] [PubMed]
- Sato, K.; Kadota, Y.; Shirasu, K. Plant immune responses to parasitic nematodes. Front. Plant Sci. 2019, 10, 1165. [Google Scholar] [CrossRef] [PubMed]
- Seah, S.; Telleen, A.C.; Williamson, V.M. Introgressed and endogenous Mi-1 gene clusters in tomato differ by complex rearrangements in flanking sequences and show sequence exchange and diversifying selection among homologues. Theoret. Appl. Gen. 2007, 114, 1289–1302. [Google Scholar] [CrossRef]
- Ammiraju, J.S.S.; Veremis, J.C.; Huang, X.; Roberts, P.A.; Kaloshian, I. The heat-stable, root-knot nematode resistance gene Mi-9 from Lycopersicum peruvianum is localized on the short arm of chromosome 6. Theoret. Appl. Gen. 2003, 106, 478–484. [Google Scholar] [CrossRef]
- Molinari, S. Resistance and virulence in plant-nematode interactions. In Nematodes; Boeri, F., Chung, J.A., Eds.; Nova Science Publisher, Inc.: New York, NY, USA, 2012; pp. 59–82. [Google Scholar]
- Finkers-Tomczak, A.; Bakker, E.; de Boer, J.; van der Vossen, E.; Achenbach, U.; Golas, T. Comparative sequence analysis of the potato cyst nematode resistance locus H1 reveals a major lack of co-linearity between three haplotypes in potato (Solanum tuberosum ssp.). Theor. Appl. Genet. 2011, 122, 595–608. [Google Scholar] [CrossRef]
- Dropkin, V.H.; Helgeson, J.P.; Upper, C.D. The hypersensitive reaction of tomatoes resistant to Meloidogyne incognita: Reversal by cytokinins. J. Nematol. 1969, 1, 55–60. [Google Scholar]
- Janssen, R.; Bakker, J.; Gommers, F. Mendelian proof for a gene-for-gene relationship between virulence of Globodera rostochiensis and the H1 resistance gene in Solanum tuberosum spp. andigena CPC 1673. Rev. Nematol. 1991, 14, 213–219. [Google Scholar]
- Dangl, J.L.; Jones, J.D. Plant pathogens and integrated defence responses to infection. Nature 2001, 411, 826–833. [Google Scholar] [CrossRef]
- Bakker, E.; Dees, R.; Bakker, J.; Goverse, A. Mechanisms involved in plant resistance to nematodes. In Multi-Genic and Induced Systemic Resistance in Plants; Tuzun, S., Bent, E., Eds.; Springer Science+Business Media, Inc.: Secaucus, NJ, USA, 2006; pp. 314–334. [Google Scholar]
- Abd-Elgawad, M.M.M. Biological control agents in the integrated nematode management of pepper in Egypt. Egypt. J. Biol. Pest Cont. 2020, 30, 70. [Google Scholar] [CrossRef]
- Williamson, V.M.; Roberts, P.A. Mechanisms and genetics of resistance. In Root-Knot Nematodes; Perry, R.N., Moens, M., Starr, J.L., Eds.; CAB International: Wallingford, UK, 2009; pp. 301–325. [Google Scholar]
- Evans, K.; Trudgill, D.L.; Webster, J.M. Plant Parasitic Nematodes in Temperate Agriculture; CABI International: Wallingford, UK, 1993. [Google Scholar]
- Sato, K.; Uehara, T.; Holbein, J.; Sasaki-Sekimoto, Y.; Gan, P.; Bino, T.; Yamaguchi, K.; Ichihashi, Y.; Maki, N.; Shigenobu, S.; et al. Transcriptomic analysis of resistant and susceptible responses in a new model root-knot nematode infection system using Solanum torvum and Meloidogyne arenaria. Front. Plant Sci. 2021, 12, 680151. [Google Scholar] [CrossRef]
- Seifi, A.; Kaloshian, I.; Vossen, J.; Che, D.; Bhattarai, K.K.; Fan, J. Linked, if not the same, Mi-1 homologues confer resistance to tomato powdery mildew and root-knot nematodes. Mol. Plant-Microbe Interact. 2011, 24, 441–450. [Google Scholar] [CrossRef]
- Ndeve, N.D.; Matthews, W.C.; Santos, J.R.P.; Huynh, B.L.; Roberts, P.A. Broad-based root-knot nematode resistance identified in cowpea gene-pool two. J. Nematol. 2018, 50, 545–558. [Google Scholar] [CrossRef]
- Bhuiyan, S.A.; Garlick, K. Evaluation of root-knot nematode resistance assays for sugarcane accession lines in Australia. J. Nematol. 2021, 53, 1–11. [Google Scholar] [CrossRef]
- Khallouk, S.; Voisin, R.; Van Ghelder, C.; Engler, G.; Amiri, S.; Esmenjaud, D. Histological mechanisms of the resistance conferred by the Ma gene against Meloidogyne incognita in Prunus spp. Phytopathology 2011, 101, 945–951. [Google Scholar] [CrossRef]
- Zhang, L.Y.; Zhang, Y.Y.; Chen, R.G.; Zhang, J.H.; Wang, T.T.; Li, H.X.; Ye, Z.B. Ectopic expression of the tomato Mi-1 gene confers resistance to root knot nematodes in lettuce (Lactuca sativa). Plant Mol. Biol. Rep. 2010, 28, 204–211. [Google Scholar] [CrossRef]
- Kahn, T.W.; Duck, N.B.; McCarville, M.T.A. Bacillus thuringiensis Cry protein controls soybean cyst nematode in transgenic soybean plants. Nat. Commun. 2021, 12, 3380. [Google Scholar] [CrossRef]
- Chen, Y.L.; He, Y.; Hsiao, T.T.; Wang, C.J.; Tian, Z.; Yeh, K.W. Pyramiding taro cystatin and fungal chitinase genes driven by a synthetic promoter enhances resistance in tomato to root-knot nematode Meloidogyne incognita. Plant Sci. 2015, 231, 74–81. [Google Scholar] [CrossRef]
- Glazebrook, J. Contrasting mechanisms of defense against biotrophic and nectrophic pathogens. Annu. Rev. Phytopathol. 2005, 43, 205–227. [Google Scholar] [CrossRef]
- Uehara, T.; Sugiyama, S.; Matsuura, H.; Arie, T.; Masuta, C. Resistant and susceptible responses in tomato to cyst nematodes are differentially regulated by salicylic acid. Plant Cell Physiol. 2010, 51, 1524–1536. [Google Scholar] [CrossRef]
- Zipfel, C.; Oldoryd, G.E.D. Plant signalling in symbiosis and immunity. Nature 2017, 543, 328–336. [Google Scholar] [CrossRef]
- El-Nagdi, W.M.A.; Youssef, M.A.M.; Abd-El-Khair, H.; Abd-Elgawad, M.M.M. Effect of certain organic amendments and Trichoderma species on the root-knot nematode, Meloidogyne incognita infecting pea (Pisum sativum L.) plants. Egypt. J. Biol. Pest Control 2019, 29, 75. [Google Scholar] [CrossRef]
- Gao, X.; Guo, H.; Zhang, Q. Arbuscular mycorrhizal fungi (AMF) enhanced the growth, yield, fiber quality and phosphorus regulation in upland cotton (Gossypium hirsutum L.). Sci. Rep. 2020, 10, 2084. [Google Scholar] [CrossRef]
- Janssen, R.; Bakker, J.; Gommers, F. Selection of virulent and avirulent lines of Globodera rostochiensis for the H1 resistance gene in Solanum tuberosum ssp. andigena CPC 1673. Rev. Nématol. 1990, 13, 265–268. [Google Scholar]
- Castagnone-Sereno, P. Genetic variability of nematodes: A threat to the durability of plant resistance genes? Euphytica 2002, 124, 193–199. [Google Scholar] [CrossRef]
- Xu, J.; Narabu, T.; Mizukubo, T.; Hibi, T. A molecular marker correlated with selected virulence against the tomato resistance gene Mi in Meloidogyne incognita, M. javanica and M. arenaria. Phytopathology 2001, 91, 377–382. [Google Scholar] [CrossRef]
- Molinari, S. Antioxidant enzymes in (a)virulent populations of root-knot nematodes. Nematology 2009, 11, 689–697. [Google Scholar] [CrossRef]
- Kaloshian, I.; Teixeira, M. Advances in plant−nematode interactions with emphasis on the notorious nematode genus Meloidogyne. Phytopathology 2019, 109, 1988–1996. [Google Scholar] [CrossRef]
- Padilla-Hurtado, B.; Morillo-Coronado, Y.; Tarapues, S.; Burbano, S.; Soto-Suárez, M.; Urrea, R.; Ceballos-Aguirre, N. Evaluation of root-knot nematodes (Meloidogyne spp.) population density for disease resistance screening of tomato germplasm carrying the gene Mi-1. Chil. J. Agric. Res. 2022, 82, 157–166. [Google Scholar] [CrossRef]
- Zhang, L.; Gleason, C. Transcriptome analyses of pre-parasitic and parasitic Meloidogyne chitwoodi race 1 to identify putative effector genes. J. Nematol. 2021, 53, e2021-84. [Google Scholar] [CrossRef] [PubMed]
- Hussey, R.S.; Mims, C.W. Ultrastructure of esophageal glands and their secretory granules in the root-knot nematode Meloidogyne incognita. Protoplasma 1990, 156, 9–18. [Google Scholar] [CrossRef]
- Li, X.Q.; Wei, J.Z.; Tan, A.; Aroian, R.V. Resistance to root-knot nematode in tomato roots expressing a nematicidal Bacillus thuringiensis crystal protein. Plant Biotechnol. J. 2007, 5, 455–464. [Google Scholar] [CrossRef] [PubMed]
- Li, X.-Q.; Tan, A.; Voegtline, M.; Bekele, S.; Chen, C.-S.; Aroian, R.V. Expression of Cry5B protein from Bacillus thuringiensis in plant roots confers resistance to root-knot nematode. Biol. Control 2008, 47, 97–102. [Google Scholar] [CrossRef]
- Urwin, P.E.; Mcpherson, M.J.; Atkinson, H.J. Enhanced transgenic plant resistance to nematodes by dual proteinase inhibitor constructs. Planta 1998, 204, 472–479. [Google Scholar] [CrossRef]
- Orlando, V.; Grove, I.G.; Edwards, S.G.; Prior, T.; Roberts, D.; Neilson, R.; Back, M. Root-lesion nematodes of potato: Current status of diagnostics, pathogenicity and management. Plant Pathol. 2020, 69, 405–417. [Google Scholar] [CrossRef]
- Dritsoulas, A.; El-Borai, F.E.; Shehata, I.E.; Hammam, M.M.; El-Ashry, R.M.; Mohamed, M.M.; Abd-Elgawad, M.M.; Duncan, L.W. Reclaimed desert habitats favor entomopathogenic nematode and microarthropod abundance compared to ancient farmlands in the Nile Basin. J. Nematol. 2021, 53, 1–13. [Google Scholar] [CrossRef]
- Iqbal, S.; Fosu-Nyarko, J.; Jones, M.G.K. Attempt to silence genes of the RNAi pathways of the root-knot nematode, Meloidogyne incognita results in diverse responses including increase and no change in expression of some genes. Front. Plant Sci. 2020, 11, 328. [Google Scholar] [CrossRef]
- Rosso, M.N.; Dubrana, M.P.; Cimbolini, N.; Jaubert, S.; Abad, P. Application of RNA interference to root-knot nematode genes encoding esophageal gland proteins. Mol. Plant Microbe Interact. 2005, 18, 615–620. [Google Scholar] [CrossRef]
- Roze, E.; Hanse, B.; Mitreva, M.; Vanholme, B.; Bakker, J.; Smant, G. Mining the secretome of the root-knot nematode Meloidogyne chitwoodi for candidate parasitism genes. Mol. Plant Pathol. 2005, 9, 1–10. [Google Scholar] [CrossRef]
- Wubie, M.; Temesgen, Z. Resistance mechanisms of tomato (Solanum lycopersicum) to root-knot nematodes (Meloidogyne species). J. Plant Breed. Crop Sci. 2019, 11, 33–40. [Google Scholar]
- Pocurull, M.; Fullana, A.M.; Ferro, M.; Valero, P.; Escudero, N.; Saus, E.; Gabaldón, T.; Sorribas, F.J. Commercial formulates of Trichoderma induce systemic plant resistance to Meloidogyne incognita in tomato and the effect is additive to that of the mi-1.2 resistance gene. Front. Microbiol. 2020, 10, 3042. [Google Scholar] [CrossRef]
- Zhao, Y.-L.; Wu, Q.-L.; Li, Y.-P.; Wang, D.-Y. Translocation, transfer, and in vivo safety evaluation of engineered nanomaterials in the non-mammalian alternative toxicity assay model of nematode Caenorhabditis elegans. RSC Adv. 2013, 3, 5741–5757. [Google Scholar] [CrossRef]
- Szitenberg, A.; Salazar-Jaramillo, L.; Blok, V.C.; Laetsch, D.R.; Joseph, S.; Williamson, V.M.; Blaxter, M.L.; Lunt, D.H. Comparative genomics of apomictic root-knot nematodes: Hybridization, ploidy, and dynamic genome change. Genome Biol. Evol. 2017, 9, 2844–2861. [Google Scholar] [CrossRef]
- Sato, K.; Kadota, Y.; Gan, P.; Bino, T.; Uehara, T.; Yamaguchi, K.; Ichihashi, Y.; Maki, N.; Iwahori, H.; Suzuki, T.; et al. High-quality genome sequence of the root-knot nematode Meloidogyne arenaria genotype A2-O. Genome Announc. 2018, 6, e00519-18. [Google Scholar] [CrossRef]
- Mani, V.; Assefa, A.D.; Hahn, B.S. Transcriptome analysis and miRNA target profiling at various stages of root-knot nematode Meloidogyne incognita development for identification of potential regulatory networks. Int. J. Mol. Sci. 2001, 22, 7442. [Google Scholar] [CrossRef]
- Susič, N.; Koutsovoulos, G.D.; Riccio, C.; Danchin, E.G.J. Genome sequence of the root-knot nematode Meloidogyne luci. J. Nematol. 2020, 52, 1–5. [Google Scholar] [CrossRef]
- Koutsovoulos, G.D.; Poullet, M.; Elashry, A. Genome assembly and annotation of Meloidogyne enterolobii, an emerging parthenogenetic root-knot nematode. Sci. Data 2020, 7, 324. [Google Scholar] [CrossRef] [PubMed]
- Phan, N.T.; Besnard, G.; Ouazahrou, R.; Sánchez, W.S.; Gil, L.; Manzi, S.; Bellafiore, S. Genome sequence of the coffee root-knot nematode Meloidogyne exigua. J. Nematol. 2001, 53, 1–6. [Google Scholar] [CrossRef] [PubMed]
- Bali, S.; Hu, S.; Vining, K.; Brown, C.R.; Majtahedi, H.; Zhang, L.; Gleason, C.; Sathuvalli, V. Nematode genome announcement: Draft genome of Meloidogyne chitwoodi, an economically important pest of potato in the Pacific Northwest. Mol. Plant-Microbe Interact. 2021, 34, 981–986. [Google Scholar] [CrossRef] [PubMed]
- Somvanshi, V.S.; Dash, M.; Bhat, C.G.; Budhwar, R.; Godwin, J.; Shukla, R.N.; Patrignani, A.; Schlapbach, R.; Rao, U. An improved draft genome assembly of Meloidogyne graminicola IARI strain using long-read sequencing. Gene 2021, 793, 145748. [Google Scholar] [CrossRef] [PubMed]
- Papolu, P.K.; Gantasala, N.P.; Kamaraju, D.; Banakar, P.; Sreevathsa, R. Utility of host delivered RNAi of two FMRF amide like peptides, flp-14 and flp-18, for the management of root knot nematode, Meloidogyne incognita. PLoS ONE 2013, 8, e80603. [Google Scholar] [CrossRef]
- Shivakumara, T.N.; Papolu, P.K.; Dutta, T.K.; Kamaraju, D.; Rao, U. RNAi-induced silencing of an effector confers transcriptional oscillation in another group of effectors in the root-knot nematode, Meloidogyne incognita. Nematology 2016, 18, 857–870. [Google Scholar] [CrossRef]
- Caromel, B.; Gebhardt, C. Breeding for nematode resistance: Use of genomic information. In Genomics and Molecular Genetics of Plant-Nematode Interactions; Jones, J., Gheysen, G., Fenoll, C., Eds.; Springer: Dordrecht, The Netherlands, 2011; pp. 465–492. [Google Scholar] [CrossRef]
- Bak, R.O.; Gomez-Ospina, N.; Porteus, M.H. Gene editing on center stage. Trends Genet. 2018, 34, 600–611. [Google Scholar] [CrossRef]
- Rajput, M.; Choudhary, K.; Kumar, M.; Vivekanand, V.; Chawade, A.; Ortiz, R.; Pareek, N. RNA interference and CRISPR/Cas gene editing for crop improvement: Paradigm shift towards sustainable agriculture. Plants 2001, 10, 1914. [Google Scholar] [CrossRef]
- O’Halloran, D.M. CRISPR-PN2: A flexible and genome-aware platform for diverse CRISPR experiments in parasitic nematodes. BioTechniques 2001, 71, 495–498. [Google Scholar] [CrossRef]
- Banu, J.G.; Meena, K.S.; Selvi, C.; Manickam, S.; Jainullabudeen, C.; Banu, G. Molecular marker-assisted selection for nematode resistance in crop plants. J. Entomol. Zool. Stud. 2017, 5, 1307–1311. [Google Scholar]
- Simko, I.; Jia, M.; Venkatesh, J.; Kang, B.; Weng, Y.; Barcaccia, G.; Lanteri, S.; Bhattarai, G.; Foolad, M.R. Genomics and marker-assisted improvement of vegetable crops. Crit. Rev. Plant Sci. 2021, 40, 303–365. [Google Scholar] [CrossRef]
- Shi, A.; Gepts, P.; Song, Q.; Xiong, H.; Michaels, T.E.; Chen, S. Genome-wide association study and genomic prediction for soybean cyst nematode resistance in USDA common bean (Phaseolus vulgaris) core collection. Front. Plant Sci. 2021, 12, 624156. [Google Scholar] [CrossRef]
- Alekcevetch, J.C.; de Lima Passianotto, A.L.; Ferreira, E.G.C. Genome-wide association study for resistance to the Meloidogyne javanica causing root-knot nematode in soybean. Theoret. Appl. Gen. 2021, 134, 777–792. [Google Scholar] [CrossRef]
- Williamson, V.M.; Ho, J.Y.; Wu, F.F.; Miller, N.; Kaloshian, I. A PCR based marker tightly linked to the nematode resistance gene, Mi in tomato. Theoret. Appl. Gen. 1997, 87, 757–763. [Google Scholar] [CrossRef]
- Goggin, F.L.; Jia, L.L.; Shah, G.; Hebert, S.; Williamson, V.M.; Ullman, D.E. Heterologous expression of the Mi-1.2 gene from tomato confers resistance against nematodes but not aphids in eggplant. Mol. Plant-Microbe Interact. 2006, 19, 383–388. [Google Scholar] [CrossRef]
- Barloy, D.; Lemoine, J.; Abelard, P.; Tanguy, A.M.; Rivoal, R.; Jahier, J. Marker-assisted pyramiding of two cereal cyst nematode resistance genes from Aegilops variabilis in wheat. Mol. Breed. 2007, 20, 31–40. [Google Scholar] [CrossRef]
- Djian-Caporalino, C.; Pijarowski, L.; Fazari, A.; Samson, M.; Gaveau, L.; O’Byrne, C.; Lefebvre, V.; Caranta, C.; Palloix, A.; Abad, P. High-resolution genetic mapping of pepper (Capsicum annuum L.) resistance loci Me3 and Me4 conferring heat-stable resistance root-knot nematodes (Meloidogyne spp.). Theoret. Appl. Gen. 2001, 103, 592–600. [Google Scholar] [CrossRef]
- Gebhardt, C.; Mugniery, D.; Ritter, E.; Salamini, F.; Bonnel, E. Identification of RFLP markers closely linked to the H1 gene conferring resistance to Globodera rostochiensis in potato. Theoret. Appl. Gen. 1993, 85, 541–544. [Google Scholar] [CrossRef]
- Kadam, S.; Tri, D.; Vuonga, Q.D.; Clinton, G.; Meinhardta Deshmukha, L.S.R.; Patila, G. Genomic-assisted phylogenetic analysis and marker development for next generation soybean cyst nematode resistance breeding. Plant Sci. 2016, 242, 342–350. [Google Scholar] [CrossRef]
- Devran, Z.; Firat, A.F.; Tor, M.; Mutlu, N.; Elekçioglu, I.H. AFLP and SRAP markers linked to the Mj gene for root-knot nematode resistance in cucumber. Sci. Agric. 2011, 68, 115–119. [Google Scholar] [CrossRef]
- Kumar, P.; He, Y.J.; Singh, R.; Davis, R.F.; Guo, H.; Paterson, A.H. Fine mapping and identification of candidate genes for a QTL affecting Meloidogyne incognita reproduction in Upland cotton. BMC Genom. 2016, 17, 567. [Google Scholar] [CrossRef]
- Romano, G.B.; Sacks, E.J.; Stetina, S.R.; Robinson, A.F.; Fang, D.D.; Gutierrez, O.A.; Scheffler, J.A. Identification and genomic location of a reniform nematode (Rotylenchulus reniformis) resistance locus (Renari) introgressed from Gossypium aridum into upland cotton (G. hirsutum). Theoret. Appl. Gen. 2009, 120, 139–150. [Google Scholar] [CrossRef]
- Chu, Y.; Wu, C.L.; Holbrook, C.C.; Tillman, B.L.; Person, G.; Ozias-Akins, P. Marker-assisted selection to pyramid nematode resistance and the high oleic trait in peanut. Plant Genome 2011, 4, 110–117. [Google Scholar] [CrossRef]
- Abd-Elgawad, M.M.M.; Askary, T.H. Fungal and bacterial nematicides in integrated nematode management strategies. Egypt. J. Biol. Pest Control 2018, 28, 74. [Google Scholar] [CrossRef]
- Banakar, P.; Hada, A.; Papolu, P.K.; Rao, U. Simultaneous RNAi knockdown of three FMRF-amide-like peptide genes, Mi-flp1, Mi-flp12, and Mi-flp18 provides resistance to root-knot nematode, Meloidogyne incognita. Front. Microbiol. 2020, 11, 573916. [Google Scholar] [CrossRef] [PubMed]
- Abd-Elgawad, M.M.M. Biological control of nematodes infecting eggplant in Egypt. Bull. NRC 2021, 45, 6. [Google Scholar] [CrossRef]
- Njom, V.S.; Winks, T.; Diallo, O. The effects of plant cysteine proteinases on the nematode cuticle. Parasites Vectors 2021, 14, 302. [Google Scholar] [CrossRef] [PubMed]
- Tripathi, L.; Atkinson, H.; Roderick, H.; Kubiriba, J.; Tripathi, J.N. Genetically engineered bananas resistant to Xanthomonas wilt disease and nematodes. Food Energy Secur. 2017, 6, 37–47. [Google Scholar] [CrossRef]
- Huang, G.; Gao, B.; Maier, T.; Allen, R.; Davis, E.L.; Baum, T.J. A profile of putative parasitism genes expressed in the esophageal gland cells of the root-knot nematode Meloidogyne incognita. Mol. Plant-Microbe Interact. 2003, 16, 376–381. [Google Scholar] [CrossRef]
- Vasconcelos, I.M.; Oliveira, J.T. Antinutritional properties of plant lectins. Toxicon 2004, 44, 385–403. [Google Scholar] [CrossRef]
- Fioretti, L.; Porter, A.; Haydock, P.J.; Curtis, R. Monoclonal antibodies reactive with secreted-excreted products from the amphids and the cuticle surface of Globodera pallida affect nematode movement and delay invasion of potato roots. Int. J. Parasitol. 2002, 32, 1709–1718. [Google Scholar] [CrossRef]
- Ravari, S.B.; Moghaddam, E.M. Efficacy of Bacillus thuringiensis Cry14 toxin against root knot nematode, Meloidogyne javanica. Plant Prot. Sci. 2015, 51, 46–51. [Google Scholar] [CrossRef]
- Winter, M.D.; Mcpherson, M.J.; Atkinson, H.J. Neuronal uptake of pesticides disrupts chemosensory cells of nematodes. Parasitology 2002, 125, 561–565. [Google Scholar]
- Liu, B.; Hibbard, J.K.; Urwin, P.E.; Atkinson, H.J. The production of synthetic chemodisruptive peptides in planta disrupts the establishment of cyst nematodes. Plant Biotechnol. J. 2005, 3, 487–496. [Google Scholar] [CrossRef]
- Green, J.; Wang, D.; Lilley, C.J.; Urwin, P.E.; Atkinson, H.J. Transgenic potatoes for potato cyst nematode control can replace pesticide use without impact on soil quality. PLoS ONE 2012, 7, e30973. [Google Scholar] [CrossRef]
- Roderick, H.; Tripathi, L.; Babirye, A.; Wang, D.; Tripathi, J.; Urwin, P.E. Generation of transgenic plantain (Musa spp.) with resistance to plant pathogenic nematodes. Mol. Plant Pathol. 2012, 13, 842–851. [Google Scholar] [CrossRef]
- Tripathi, L.; Tripathi, J.N.; Roderick, H.; Atkinson, H.J. Engineering nematode resistant plantains for sub-Saharan Africa. Acta Hortic. 2013, 974, 99–107. [Google Scholar] [CrossRef]
- Papolu, P.K.; Dutta, T.K.; Hada, A.; Singh, D.; Rao, U. The production of a synthetic chemodisruptive peptide in planta precludes Meloidogyne incognita multiplication in Solanum melongena. Physiol. Mol. Plant Pathol. 2020, 112, 101542. [Google Scholar] [CrossRef]
- Rocha, R.O.; Morais, J.K.S.; Oliveira, J.T.A. Proteome of soybean seed exudates contains plant defense-related proteins active against the root-knot nematode Meloidogyne incognita. J. Agric. Food Chem. 2015, 63, 5335–5343. [Google Scholar] [CrossRef]
- Tsai, A.Y.L.; Higaki, T.; Nguyen, C.N.; Perfus-Barbeoch, L.; Favery, B.; Sawa, S. Regulation of root-knot nematode behavior by seed-coat mucilage-derived attractants. Mol. Plant. 2019, 12, 99–112. [Google Scholar] [CrossRef]
- Oosterbeek, M.; Lozano-Torres, J.L.; Bakker, J.; Goverse, A. Sedentary plant-parasitic nematodes alter auxin homeostasis via multiple strategies. Front. Plant Sci. 2021, 12, 668548. [Google Scholar] [CrossRef]
- Barbary, A.; Djian-Caporalino, C.; Palloix, A.; Castagnone-Sereno, P. Host genetic resistance to root-knot nematodes, Meloidogyne spp., in Solanaceae: From genes to the field. Pest Manag. Sci. 2015, 71, 1591–1598. [Google Scholar] [CrossRef]
- Hulbert, S.H.; Craig, A.W.; Shavannor, M.S.; Qing, S. Resistance gene complexes: Evolution and utilization. Annu. Rev. Phytopathol. 2001, 39, 285–312. [Google Scholar] [CrossRef]
- Abd-Elgawad, M.M.M. Can rational sampling maximise isolation and fix distribution measure of entomopathogenic nematodes? Nematology 2020, 22, 907–916. [Google Scholar] [CrossRef]
- Abd-Elgawad, M.M.M. Towards optimization of entomopathogenic nematodes for more service in the biological control of insect pests. Egypt. J. Biol. Pest Cont. 2019, 29, 77. [Google Scholar] [CrossRef]
- Abd-Elgawad, M.M.M. Optimizing biological control agents for controlling nematodes of tomato in Egypt. Egypt. J. Biol. Pest Cont. 2020, 30, 58. [Google Scholar] [CrossRef]
- Abd-Elgawad, M.M.M. Biological control agents in the integrated nematode management of potato in Egypt. Egypt. J. Biol. Pest Cont. 2020, 30, 121. [Google Scholar] [CrossRef]
- McCarter, J.P. Molecular approaches toward resistance to plant-parasitic nematodes. In Plant Cell Monographs: Cell Biology of Plant Nematode Parasitism; Berg, R.H., Taylor, C.G., Eds.; Springer: Berlin, Germany, 2008; pp. 239–268. [Google Scholar] [CrossRef]
- Guo, Y.; Fudali, S.; Gimeno, J.; Digennaro, P.; Chang, S.; Williamson, V.M.; Bird, D.M.; Nielsen, D. Networks underpinning symbiosis revealed through cross-species eQTL mapping. Genetics 2017, 206, 2175–2184. [Google Scholar] [CrossRef]
- Perry, R.N.; Moens, M. Introduction to plant-parasitic nematodes: Modes of parasitism. In Genomics and Molecular Genetics of Plant–Nematode Interactions; Jones, J.T., Gheysen, L., Fenoll, C., Eds.; Springer: Berlin/Heidelberg, Germany, 2011; pp. 3–20. [Google Scholar]
- Kaloshian, I.; Desmond, O.J.; Atamian, H.S. Disease resistance-genes and defense responses during incompatible interactions. In Genomics and Molecular Genetics of Plant–Nematode Interactions; Jones, J.T., Gheysen, L., Fenoll, C., Eds.; Springer: Berlin/Heidelberg, Germany, 2011; pp. 309–324. [Google Scholar] [CrossRef]
- Sun, B.; Zhang, X.; Song, L.; Zheng, L.; Wei, X.; Gu, X.; Cui, Y.; Hu, B.; Yoshiga, T.; Abd-Elgawad, M.M.; et al. Evaluation of indigenous entomopathogenic nematodes in Southwest China as potential biocontrol agents against Spodoptera litura (Lepidoptera: Noctuidae). J. Nematol. 2021, 53, 1–17. [Google Scholar] [CrossRef]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Abd-Elgawad, M.M.M. Understanding Molecular Plant–Nematode Interactions to Develop Alternative Approaches for Nematode Control. Plants 2022, 11, 2141. https://doi.org/10.3390/plants11162141
Abd-Elgawad MMM. Understanding Molecular Plant–Nematode Interactions to Develop Alternative Approaches for Nematode Control. Plants. 2022; 11(16):2141. https://doi.org/10.3390/plants11162141
Chicago/Turabian StyleAbd-Elgawad, Mahfouz M. M. 2022. "Understanding Molecular Plant–Nematode Interactions to Develop Alternative Approaches for Nematode Control" Plants 11, no. 16: 2141. https://doi.org/10.3390/plants11162141
APA StyleAbd-Elgawad, M. M. M. (2022). Understanding Molecular Plant–Nematode Interactions to Develop Alternative Approaches for Nematode Control. Plants, 11(16), 2141. https://doi.org/10.3390/plants11162141






