The Genetic Complexity of Type-IV Trichome Development Reveals the Steps towards an Insect-Resistant Tomato
Abstract
:1. Introduction
2. Results
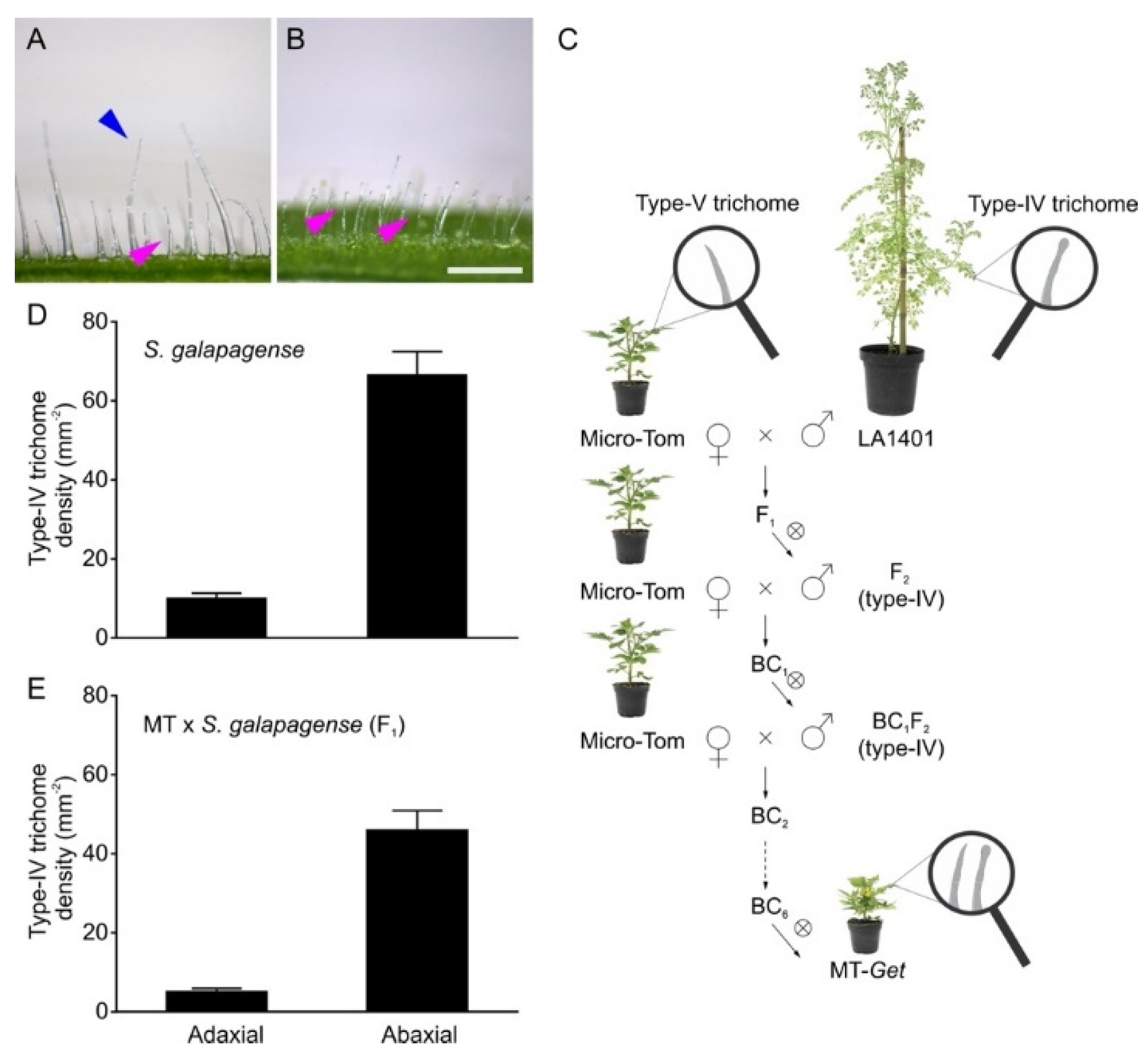
2.1. Introgression of the Genetic Determinants Controlling Type-IV Trichome Development on Adult Leaves from S. galapagense LA1401 into Tomato (S. lycopersicum cv. Micro-Tom)
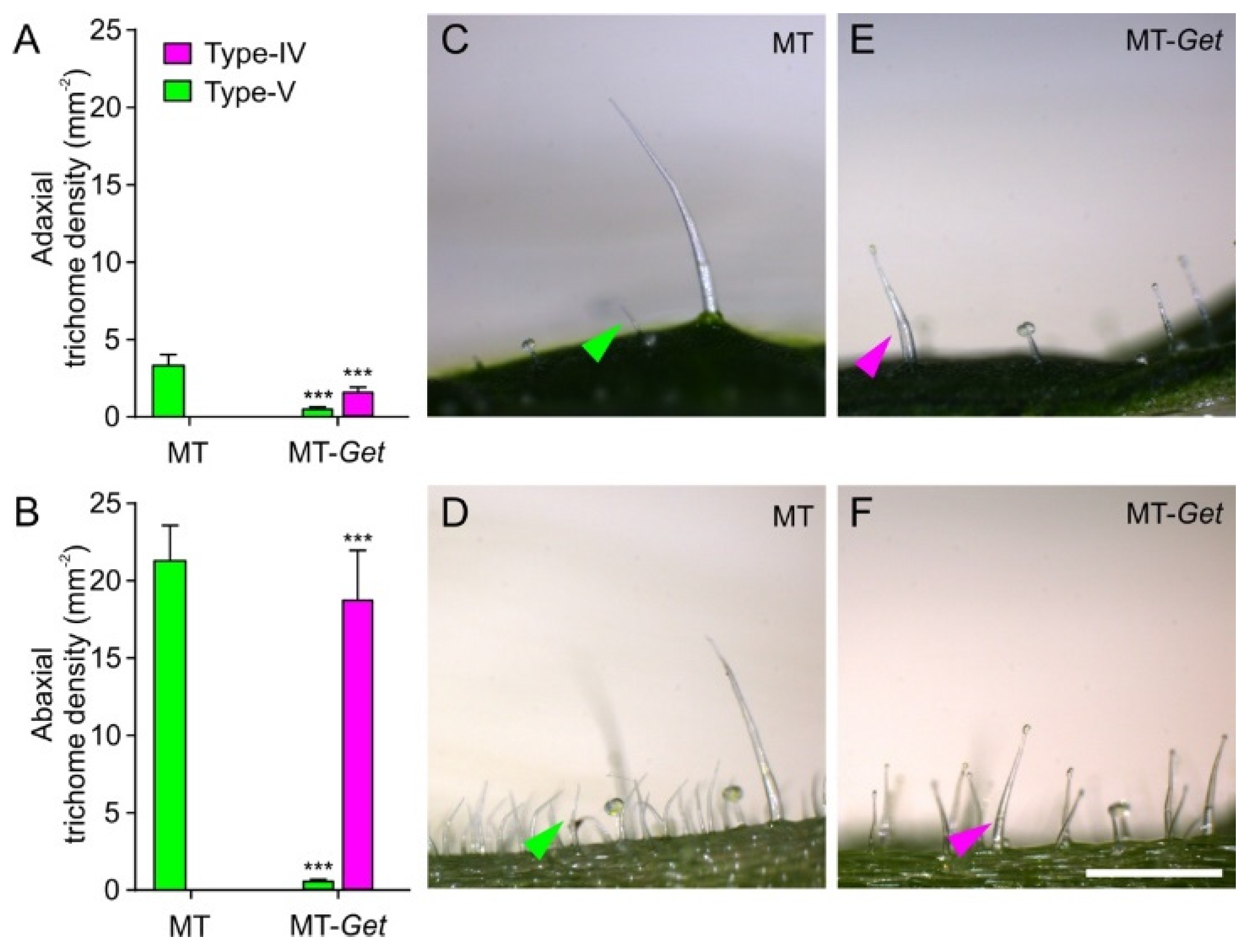
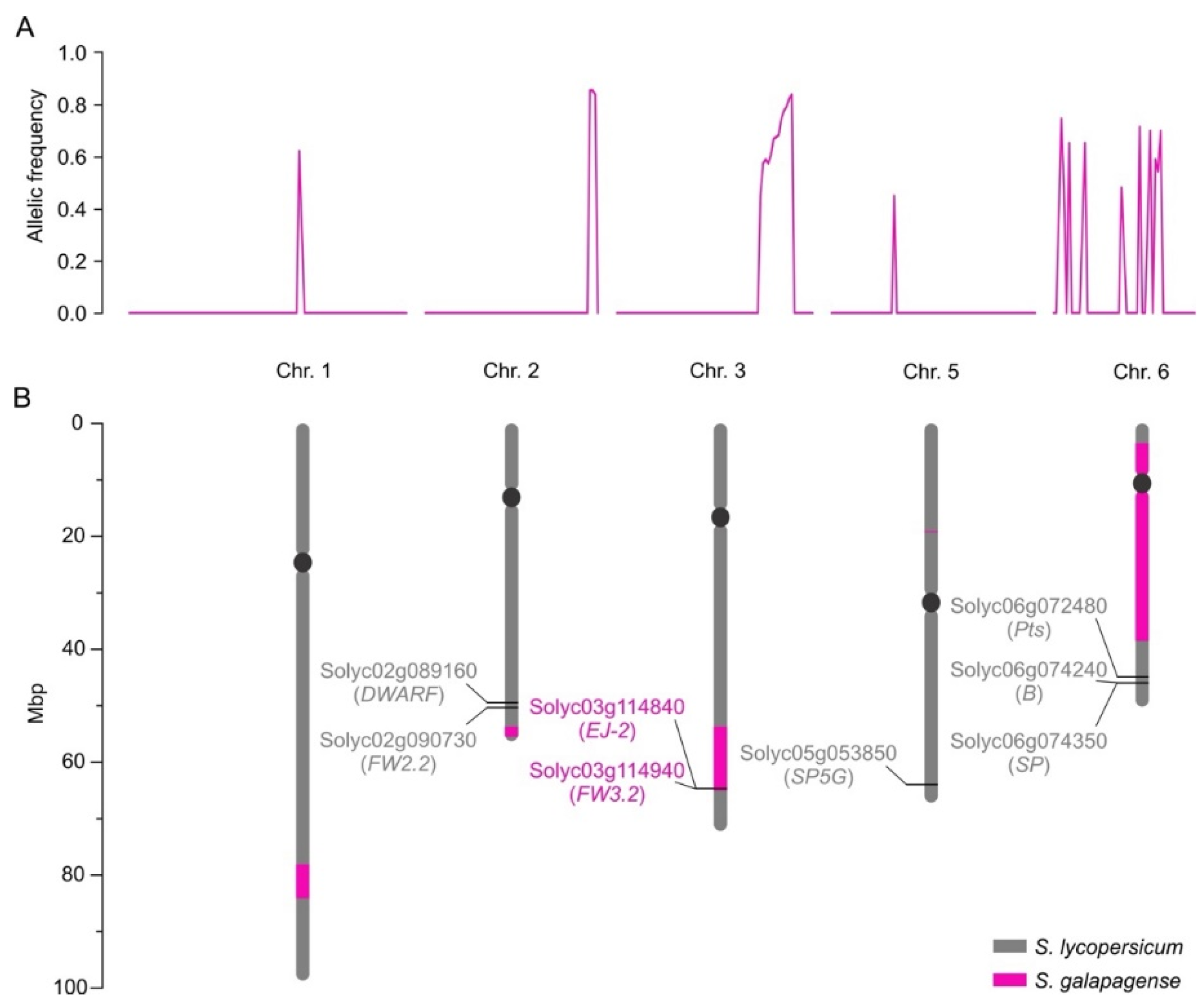
2.2. Phenotypic Characterization and Genetic Mapping of the “Galapagos-enhanced trichomes” (MT-Get) Introgression Line
2.3. Whitefly Resistance Test and Trichome Gland Exudation in MT-Get Plants
2.4. Acylsugar Accumulation and Related Gene Expression in MT-Get
3. Discussion
4. Materials and Methods
4.1. Plant Material, Growth Conditions, and Breeding Scheme
4.2. Plant Genetic Transformation
4.3. Trichome Morphometry
4.4. Scanning Electron Microscopy
4.5. Fluorescence Microscopy
4.6. Mapping-by-Sequencing Analysis
4.7. Micro-Tom and S. galapagense Allele Genotyping
4.8. Plant Phenotyping
4.9. Herbivory Test with BEMISIA Tabaci
4.10. Rhodamine B Assay for Acylsugar Staining
4.11. LC-MS/MS Analysis of Surface Extracts
4.12. GC-MS Acylsugar Quantification
4.13. Gene Expression Analyses
4.14. Statistical Analyses
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Schilmiller, A.L.; Last, R.L.; Pichersky, E. Harnessing Plant Trichome Biochemistry for the Production of Useful Compounds. Plant J. 2008, 54, 702–711. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Tissier, A. Glandular Trichomes: What Comes after Expressed Sequence Tags? Glandular Trichomes: What Comes after ESTs? Plant J. 2012, 70, 51–68. [Google Scholar] [CrossRef]
- Aharoni, A.; Jongsma, M.; Bouwmeester, H. Volatile Science? Metabolic Engineering of Terpenoids in Plants. Trends Plant Sci. 2005, 10, 594–602. [Google Scholar] [CrossRef] [PubMed]
- Maes, L.; Van Nieuwerburgh, F.C.W.; Zhang, Y.; Reed, D.W.; Pollier, J.; Vande Casteele, S.R.F.; Inzé, D.; Covello, P.S.; Deforce, D.L.D.; Goossens, A. Dissection of the Phytohormonal Regulation of Trichome Formation and Biosynthesis of the Antimalarial Compound Artemisinin in Artemisia annua Plants. New Phytol. 2011, 189, 176–189. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kang, J.-H.; Liu, G.; Shi, F.; Jones, A.D.; Beaudry, R.M.; Howe, G.A. The Tomato Odorless-2 Mutant Is Defective in Trichome-Based Production of Diverse Specialized Metabolites and Broad-Spectrum Resistance to Insect Herbivores. Plant Physiol. 2010, 154, 262–272. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Glas, J.; Schimmel, B.; Alba, J.; Escobar-Bravo, R.; Schuurink, R.; Kant, M. Plant Glandular Trichomes as Targets for Breeding or Engineering of Resistance to Herbivores. Int. J. Mol. Sci. 2012, 13, 17077–17103. [Google Scholar] [CrossRef] [Green Version]
- Luckwill, L.C. The Genus Lycopersicon: An Historical, Biological, and Taxonomic Survey of the Wild and Cultivated Tomatoes; Aberdeen University Studies; Aberdeen University Press: Aberdeen, UK, 1943. [Google Scholar]
- Kim, J.; Kang, K.; Gonzales-Vigil, E.; Shi, F.; Jones, A.D.; Barry, C.S.; Last, R.L. Striking Natural Diversity in Glandular Trichome Acylsugar Composition Is Shaped by Variation at the Acyltransferase2 Locus in the Wild Tomato Solanum habrochaites. Plant Physiol. 2012, 160, 1854–1870. [Google Scholar] [CrossRef] [Green Version]
- Ghosh, B.; Westbrook, T.C.; Jones, A.D. Comparative Structural Profiling of Trichome Specialized Metabolites in Tomato (Solanum lycopersicum) and S. habrochaites: Acylsugar Profiles Revealed by UHPLC/MS and NMR. Metabolomics 2014, 10, 496–507. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ning, J.; Moghe, G.; Leong, B.; Kim, J.; Ofner, I.; Wang, Z.; Adams, C.; Jones, A.D.; Zamir, D.; Last, R.L. A Feedback Insensitive Isopropylmalate Synthase Affects Acylsugar Composition in Cultivated and Wild Tomato. Plant Physiol. 2015, 169, 1821–1835. [Google Scholar] [CrossRef] [Green Version]
- Schilmiller, A.L.; Moghe, G.D.; Fan, P.; Ghosh, B.; Ning, J.; Jones, A.D.; Last, R.L. Functionally Divergent Alleles and Duplicated Loci Encoding an Acyltransferase Contribute to Acylsugar Metabolite Diversity in Solanum Trichomes. Plant Cell 2015, 27, 1002–1017. [Google Scholar] [CrossRef] [Green Version]
- Fan, P.; Miller, A.M.; Schilmiller, A.L.; Liu, X.; Ofner, I.; Jones, A.D.; Zamir, D.; Last, R.L. In Vitro Reconstruction and Analysis of Evolutionary Variation of the Tomato Acylsucrose Metabolic Network. Proc. Natl. Acad. Sci. USA 2016, 113, E239–E248. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Schilmiller, A.L.; Charbonneau, A.L.; Last, R.L. Identification of a BAHD Acetyltransferase That Produces Protective Acyl Sugars in Tomato Trichomes. Proc. Natl. Acad. Sci. USA 2012, 109, 16377–16382. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Fan, P.; Leong, B.J.; Last, R.L. Tip of the Trichome: Evolution of Acylsugar Metabolic Diversity in Solanaceae. Curr. Opin. Plant Biol. 2019, 49, 8–16. [Google Scholar] [CrossRef] [PubMed]
- Luu, V.T.; Weinhold, A.; Ullah, C.; Dressel, S.; Schoettner, M.; Gase, K.; Gaquerel, E.; Xu, S.; Baldwin, I.T. O-Acyl Sugars Protect a Wild Tobacco from Both Native Fungal Pathogens and a Specialist Herbivore. Plant Physiol. 2017, 174, 370–386. [Google Scholar] [CrossRef] [Green Version]
- Goffreda, J.C.; Szymkowiak, E.J.; Sussex, I.M.; Mutschler, M.A. Chimeric Tomato Plants Show That Aphid Resistance and Triacylglucose Production Are Epidermal Autonomous Characters. Plant Cell 1990, 2, 643–649. [Google Scholar] [CrossRef] [Green Version]
- Hawthorne, D.J.; Shapiro, J.A.; Tingey, W.M.; Mutschler, M.A. Trichome-Borne and Artificially Applied Acylsugars of Wild Tomato Deter Feeding and Oviposition of the Leafminer Liriomyza trifolii. Entomol. Exp. Appl. 1992, 65, 65–73. [Google Scholar] [CrossRef]
- Liedl, B.E.; Lawson, D.M.; White, K.K.; Shapiro, J.A.; Cohen, D.E.; Carson, W.G.; Trumble, J.T.; Mutschler, M.A. Acylsugars of Wild Tomato Lycopersicon pennellii Alters Settling and Reduces Oviposition of Bemisia argentifolii (Homoptera: Aleyrodidae). J. Econ. Entomol. 1995, 88, 742–748. [Google Scholar] [CrossRef]
- Maluf, W.R.; Maciel, G.M.; Gomes, L.A.A.; Cardoso, M.D.G.; Gonçalves, L.D.; da Silva, E.C.; Knapp, M. Broad-Spectrum Arthropod Resistance in Hybrids between High- and Low-Acylsugar Tomato Lines. Crop Sci. 2010, 50, 439–450. [Google Scholar] [CrossRef]
- Weinhold, A.; Baldwin, I.T. Trichome-Derived O-Acyl Sugars Are a First Meal for Caterpillars That Tags Them for Predation. Proc. Natl. Acad. Sci. USA 2011, 108, 7855–7859. [Google Scholar] [CrossRef] [Green Version]
- Xia, J.; Guo, Z.; Yang, Z.; Han, H.; Wang, S.; Xu, H.; Yang, X.; Yang, F.; Wu, Q.; Xie, W.; et al. Whitefly Hijacks a Plant Detoxification Gene That Neutralizes Plant Toxins. Cell 2021, 184, 1693–1705.e17. [Google Scholar] [CrossRef]
- Simmons, A.T.; Gurr, G.M. Trichomes of Lycopersicon Species and Their Hybrids: Effects on Pests and Natural Enemies. Agric. For. Entomol. 2005, 7, 265–276. [Google Scholar] [CrossRef]
- Kortbeek, R.W.J.; Galland, M.D.; Muras, A.; van der Kloet, F.M.; André, B.; Heilijgers, M.; van Hijum, S.A.F.T.; Haring, M.A.; Schuurink, R.C.; Bleeker, P.M. Natural Variation in Wild Tomato Trichomes; Selecting Metabolites That Contribute to Insect Resistance Using a Random Forest Approach. BMC Plant Biol. 2021, 21, 315. [Google Scholar] [CrossRef] [PubMed]
- Fobes, J.F.; Mudd, J.B.; Marsden, M.P.F. Epicuticular Lipid Accumulation on the Leaves of Lycopersicon pennellii (Corr.) D’Arcy and Lycopersicon esculentum Mill. Plant Physiol. 1985, 77, 567–570. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Schilmiller, A.; Shi, F.; Kim, J.; Charbonneau, A.L.; Holmes, D.; Daniel Jones, A.; Last, R.L. Mass Spectrometry Screening Reveals Widespread Diversity in Trichome Specialized Metabolites of Tomato Chromosomal Substitution Lines: Solanum Trichome Chemistry. Plant J. 2010, 62, 391–403. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Mutschler, M.A.; Doerge, R.W.; Liu, S.C.; Kuai, J.P.; Liedl, B.E.; Shapiro, J.A. QTL Analysis of Pest Resistance in the Wild Tomato Lycopersicon pennellii: QTLs Controlling Acylsugar Level and Composition. Theor. Appl. Genet. 1996, 92, 709–718. [Google Scholar] [CrossRef]
- Momotaz, A.; Scott, J.W.; Schuster, D.J. Identification of Quantitative Trait Loci Conferring Resistance to Bemisia Tabaci in an F2 Population of Solanum lycopersicum × Solanum habrochaites Accession LA1777. J. Am. Soc. Hortic. Sci. 2010, 135, 134–142. [Google Scholar] [CrossRef]
- Rodríguez-López, M.J.; Garzo, E.; Bonani, J.P.; Fereres, A.; Fernández-Muñoz, R.; Moriones, E. Whitefly Resistance Traits Derived from the Wild Tomato Solanum pimpinellifolium Affect the Preference and Feeding Behavior of Bemisia tabaci and Reduce the Spread of Tomato Yellow Leaf Curl Virus. Phytopathology 2011, 101, 1191–1201. [Google Scholar] [CrossRef] [Green Version]
- Leckie, B.M.; De Jong, D.M.; Mutschler, M.A. Quantitative Trait Loci Increasing Acylsugars in Tomato Breeding Lines and Their Impacts on Silverleaf Whiteflies. Mol. Breed. 2012, 30, 1621–1634. [Google Scholar] [CrossRef]
- McDowell, E.T.; Kapteyn, J.; Schmidt, A.; Li, C.; Kang, J.-H.; Descour, A.; Shi, F.; Larson, M.; Schilmiller, A.; An, L.; et al. Comparative Functional Genomic Analysis of Solanum Glandular Trichome Types. Plant Physiol. 2011, 155, 524–539. [Google Scholar] [CrossRef] [Green Version]
- Vendemiatti, E.; Zsögön, A.; e Silva, G.F.F.; de Jesus, F.A.; Cutri, L.; Figueiredo, C.R.F.; Tanaka, F.A.O.; Nogueira, F.T.S.; Peres, L.E.P. Loss of Type-IV Glandular Trichomes Is a Heterochronic Trait in Tomato and Can Be Reverted by Promoting Juvenility. Plant Sci. 2017, 259, 35–47. [Google Scholar] [CrossRef]
- Smeda, J.R.; Schilmiller, A.L.; Anderson, T.; Ben-Mahmoud, S.; Ullman, D.E.; Chappell, T.M.; Kessler, A.; Mutschler, M.A. Combination of Acylglucose QTL Reveals Additive and Epistatic Genetic Interactions and Impacts Insect Oviposition and Virus Infection. Mol. Breed. 2018, 38, 3. [Google Scholar] [CrossRef]
- Nadakuduti, S.S.; Uebler, J.B.; Liu, X.; Jones, A.D.; Barry, C.S. Characterization of Trichome-Expressed BAHD Acyltransferases in Petunia Axillaris Reveals Distinct Acylsugar Assembly Mechanisms within the Solanaceae. Plant Physiol. 2017, 175, 36–50. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lou, Y.-R.; Anthony, T.M.; Fiesel, P.D.; Arking, R.E.; Christensen, E.M.; Jones, A.D.; Last, R.L. It Happened Again: Convergent Evolution of Acylglucose Specialized Metabolism in Black Nightshade and Wild Tomato. Sci. Adv. 2021, 7, eabj8726. [Google Scholar] [CrossRef] [PubMed]
- Channarayappa, S.G.; Muniyappa, V.; Frist, R.H. Resistance of Lycopersicon Species to Bemisia tabaci, a Tomato Leaf Curl Virus Vector. Can. J. Bot. 1992, 70, 2184–2192. [Google Scholar] [CrossRef]
- Garcia, V.; Bres, C.; Just, D.; Fernandez, L.; Tai, F.W.J.; Mauxion, J.-P.; Le Paslier, M.-C.; Bérard, A.; Brunel, D.; Aoki, K.; et al. Rapid Identification of Causal Mutations in Tomato EMS Populations via Mapping-by-Sequencing. Nat. Protoc. 2016, 11, 2401–2418. [Google Scholar] [CrossRef] [Green Version]
- Bishop, G.J.; Harrison, K.; Jones, J.D. The Tomato Dwarf Gene Isolated by Heterologous Transposon Tagging Encodes the First Member of a New Cytochrome P450 Family. Plant Cell 1996, 8, 959–969. [Google Scholar] [CrossRef] [Green Version]
- Pnueli, L.; Carmel-Goren, L.; Hareven, D.; Gutfinger, T.; Alvarez, J.; Ganal, M.; Zamir, D.; Lifschitz, E. The SELF-PRUNING Gene of Tomato Regulates Vegetative to Reproductive Switching of Sympodial Meristems and Is the Ortholog of CEN and TFL1. Development 1998, 125, 1979–1989. [Google Scholar] [CrossRef]
- Carvalho, R.F.; Campos, M.L.; Pino, L.E.; Crestana, S.L.; Zsögön, A.; Lima, J.E.; Benedito, V.A.; Peres, L.E. Convergence of Developmental Mutants into a Single Tomato Model System: “Micro-Tom” as an Effective Toolkit for Plant Development Research. Plant Methods 2011, 7, 18. [Google Scholar] [CrossRef] [Green Version]
- Soyk, S.; Müller, N.A.; Park, S.J.; Schmalenbach, I.; Jiang, K.; Hayama, R.; Zhang, L.; Van Eck, J.; Jiménez-Gómez, J.M.; Lippman, Z.B. Variation in the Flowering Gene SELF PRUNING 5G Promotes Day-Neutrality and Early Yield in Tomato. Nat. Genet. 2017, 49, 162–168. [Google Scholar] [CrossRef]
- Kimura, S.; Koenig, D.; Kang, J.; Yoong, F.Y.; Sinha, N. Natural Variation in Leaf Morphology Results from Mutation of a Novel KNOX Gene. Curr. Biol. 2008, 18, 672–677. [Google Scholar] [CrossRef] [Green Version]
- Ronen, G.; Carmel-Goren, L.; Zamir, D.; Hirschberg, J. An Alternative Pathway to β-Carotene Formation in Plant Chromoplasts Discovered by Map-Based Cloning of Beta and Old-Gold Color Mutations in Tomato. Proc. Natl. Acad. Sci. USA 2000, 97, 11102–11107. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Soyk, S.; Lemmon, Z.H.; Oved, M.; Fisher, J.; Liberatore, K.L.; Park, S.J.; Goren, A.; Jiang, K.; Ramos, A.; van der Knaap, E.; et al. Bypassing Negative Epistasis on Yield in Tomato Imposed by a Domestication Gene. Cell 2017, 169, 1142–1155.e12. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chakrabarti, M.; Zhang, N.; Sauvage, C.; Muños, S.; Blanca, J.; Cañizares, J.; Diez, M.J.; Schneider, R.; Mazourek, M.; McClead, J.; et al. A Cytochrome P450 Regulates a Domestication Trait in Cultivated Tomato. Proc. Natl. Acad. Sci. USA 2013, 110, 17125–17130. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Frary, A.; Nesbitt, T.C.; Frary, A.; Grandillo, S.; van der Knaap, E.; Cong, B.; Liu, J.; Meller, J.; Elber, R.; Alpert, K.B.; et al. Fw2.2: A Quantitative Trait Locus Key to the Evolution of Tomato Fruit Size. Science 2000, 289, 85–88. [Google Scholar] [CrossRef] [Green Version]
- Firdaus, S.; van Heusden, A.W.; Hidayati, N.; Supena, E.D.J.; Mumm, R.; de Vos, R.C.H.; Visser, R.G.F.; Vosman, B. Identification and QTL Mapping of Whitefly Resistance Components in Solanum galapagense. Theor. Appl. Genet. 2013, 126, 1487–1501. [Google Scholar] [CrossRef]
- Firdaus, S.; van Heusden, A.W.; Hidayati, N.; Supena, E.D.J.; Visser, R.G.F.; Vosman, B. Resistance to Bemisia tabaci in Tomato Wild Relatives. Euphytica 2012, 187, 31–45. [Google Scholar] [CrossRef] [Green Version]
- Vosman, B.; Kashaninia, A.; van’t Westende, W.; Meijer-Dekens, F.; van Eekelen, H.; Visser, R.G.F.; de Vos, R.C.H.; Voorrips, R.E. QTL Mapping of Insect Resistance Components of Solanum galapagense. Theor. Appl. Genet. 2019, 132, 531–541. [Google Scholar] [CrossRef] [Green Version]
- Goffreda, J.C.; Mutschler, M.A.; Avé, D.A.; Tingey, W.M.; Steffens, J.C. Aphid Deterrence by Glucose Esters in Glandular Trichome Exudate of the Wild Tomato, Lycopersicon pennellii. J. Chem. Ecol. 1989, 15, 2135–2147. [Google Scholar] [CrossRef]
- Blauth, S.L.; Churchill, G.A.; Mutschler, M.A. Identification of Quantitative Trait Loci Associated with Acylsugar Accumulation Using Intraspecific Populations of the Wild Tomato, Lycopersicon pennellii. Theor. Appl. Genet. 1998, 96, 458–467. [Google Scholar] [CrossRef]
- Schilmiller, A.L.; Gilgallon, K.; Ghosh, B.; Jones, A.D.; Last, R.L. Acylsugar Acylhydrolases: Carboxylesterase-Catalyzed Hydrolysis of Acylsugars in Tomato Trichomes. Plant Physiol. 2016, 170, 1331–1344. [Google Scholar] [CrossRef] [Green Version]
- Mandal, S.; Ji, W.; McKnight, T.D. Candidate Gene Networks for Acylsugar Metabolism and Plant Defense in Wild Tomato Solanum pennellii. Plant Cell 2020, 32, 81–99. [Google Scholar] [CrossRef] [Green Version]
- Mirnezhad, M.; Romero-González, R.R.; Leiss, K.A.; Choi, Y.H.; Verpoorte, R.; Klinkhamer, P.G.L. Metabolomic Analysis of Host Plant Resistance to Thrips in Wild and Cultivated Tomatoes: Metabolomic Analysis of Host Plant Resistance. Phytochem. Anal. 2010, 21, 110–117. [Google Scholar] [CrossRef] [PubMed]
- Schuurink, R.; Tissier, A. Glandular Trichomes: Micro-organs with Model Status? New Phytol. 2020, 225, 2251–2266. [Google Scholar] [CrossRef] [Green Version]
- Pesch, M.; Hülskamp, M. Role of TRIPTYCHON in Trichome Patterning in Arabidopsis. BMC Plant Biol. 2011, 11, 130. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Rodríguez-López, M.J.; Garzo, E.; Bonani, J.P.; Fernández-Muñoz, R.; Moriones, E.; Fereres, A. Acylsucrose-Producing Tomato Plants Forces Bemisia tabaci to Shift Its Preferred Settling and Feeding Site. PLoS ONE 2012, 7, e33064. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bitew, M.K. Significant Role of Wild Genotypes of Tomato Trichomes for Tuta absoluta Resistance. J. Plant Genet. Breed. 2018, 2, 104. [Google Scholar]
- Mata-Nicolás, E.; Montero-Pau, J.; Gimeno-Paez, E.; García-Pérez, A.; Ziarsolo, P.; Blanca, J.; van der Knaap, E.; Díez, M.J.; Cañizares, J. Discovery of a Major QTL Controlling Trichome IV Density in Tomato Using K-Seq Genotyping. Genes 2021, 12, 243. [Google Scholar] [CrossRef]
- Alonge, M.; Wang, X.; Benoit, M.; Soyk, S.; Pereira, L.; Zhang, L.; Suresh, H.; Ramakrishnan, S.; Maumus, F.; Ciren, D.; et al. Major Impacts of Widespread Structural Variation on Gene Expression and Crop Improvement in Tomato. Cell 2020, 182, 145–161.e23. [Google Scholar] [CrossRef]
- Wang, X.; Aguirre, L.; Rodríguez-Leal, D.; Hendelman, A.; Benoit, M.; Lippman, Z.B. Dissecting Cis-Regulatory Control of Quantitative Trait Variation in a Plant Stem Cell Circuit. Nat. Plants 2021, 7, 419–427. [Google Scholar] [CrossRef]
- Demissie, Z.A.; Tarnowycz, M.; Adal, A.M.; Sarker, L.S.; Mahmoud, S.S. A Lavender ABC Transporter Confers Resistance to Monoterpene Toxicity in Yeast. Planta 2019, 249, 139–144. [Google Scholar] [CrossRef]
- Kandra, L.; Wagner, G.J. Studies of the Site and Mode of Biosynthesis of Tobacco Trichome Exudate Components. Arch. Biochem. Biophys. 1988, 265, 425–432. [Google Scholar] [CrossRef]
- Balcke, G.U.; Bennewitz, S.; Bergau, N.; Athmer, B.; Henning, A.; Majovsky, P.; Jiménez-Gómez, J.M.; Hoehenwarter, W.; Tissier, A. Multi-Omics of Tomato Glandular Trichomes Reveals Distinct Features of Central Carbon Metabolism Supporting High Productivity of Specialized Metabolites. Plant Cell 2017, 29, 960–983. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Pino, L.E.; Lombardi-Crestana, S.; Azevedo, M.S.; Scotton, D.C.; Borgo, L.; Quecini, V.; Figueira, A.; Peres, L.E. The Rg1 Allele as a Valuable Tool for Genetic Transformation of the Tomato “Micro-Tom” Model System. Plant Methods 2010, 6, 23. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Fulton, T.M.; Chunwongse, J.; Tanksley, S.D. Microprep Protocol for Extraction of DNA from Tomato and Other Herbaceous Plants. Plant Mol. Biol. Rep. 1995, 13, 207–209. [Google Scholar] [CrossRef]
- Li, H.; Durbin, R. Fast and Accurate Short Read Alignment with Burrows-Wheeler Transform. Bioinformatics 2009, 25, 1754–1760. [Google Scholar] [CrossRef] [Green Version]
- Li, H.; Handsaker, B.; Wysoker, A.; Fennell, T.; Ruan, J.; Homer, N.; Marth, G.; Abecasis, G.; Durbin, R.; 1000 Genome Project Data Processing Subgroup. The Sequence Alignment/Map Format and SAMtools. Bioinformatics 2009, 25, 2078–2079. [Google Scholar] [CrossRef] [Green Version]
- Mascher, M.; Jost, M.; Kuon, J.-E.; Himmelbach, A.; Aßfalg, A.; Beier, S.; Scholz, U.; Graner, A.; Stein, N. Mapping-by-Sequencing Accelerates Forward Genetics in Barley. Genome Biol. 2014, 15, R78. [Google Scholar] [CrossRef] [Green Version]
- Hopfgartner, G.; Tonoli, D.; Varesio, E. High-Resolution Mass Spectrometry for Integrated Qualitative and Quantitative Analysis of Pharmaceuticals in Biological Matrices. Anal. Bioanal. Chem. 2012, 402, 2587–2596. [Google Scholar] [CrossRef] [Green Version]
- Leckie, B.M.; De Jong, D.M.; Mutschler, M.A. Quantitative Trait Loci Regulating Sugar Moiety of Acylsugars in Tomato. Mol. Breed. 2013, 31, 957–970. [Google Scholar] [CrossRef]
- Silva, N.C.Q.; de Souza, G.A.; Pimenta, T.M.; Brito, F.A.L.; Picoli, E.A.T.; Zsögön, A.; Ribeiro, D.M. Salt Stress Inhibits Germination of Stylosanthes Humilis Seeds through Abscisic Acid Accumulation and Associated Changes in Ethylene Production. Plant Physiol. Biochem. 2018, 130, 399–407. [Google Scholar] [CrossRef]
- Pinto, M.D.S.; Abeyratne, C.R.; Benedito, V.A.; Peres, L.E.P. Genetic and Physiological Characterization of Three Natural Allelic Variations Affecting the Organogenic Capacity in Tomato (Solanum lycopersicum cv. Micro-Tom). Plant Cell Tiss Organ Cult 2017, 129, 89–103. [Google Scholar] [CrossRef]
- Livak, K.J.; Schmittgen, T.D. Analysis of Relative Gene Expression Data Using Real-Time Quantitative PCR and the 2−ΔΔCT Method. Methods 2001, 25, 402–408. [Google Scholar] [CrossRef]





| Predicted AS | m/z | Peak Area | S. galapagense/ MT-Get | ||
|---|---|---|---|---|---|
| S. galapagense | MT-Get | MT | |||
| S3:20 (5,5,10) | 709.368 | 675.914 a | 72.873 b | 7.192 c | 9.28 |
| S3:22 (5,5,12) | 737.396 | 9877.349 a | 1767.007 b | 208.375 c | 5.59 |
| S4:16 (2,4,5,5) | 667.282 | 18,907.925 a | 3835.183 b | 514.101 c | 4.93 |
| S4:17 (2,5,5,5) | 681.297 | 61,140.597 a | 22,592.369 b | 2509.929 c | 2.71 |
| S4:22 (2,5,5,10) | 751.375 | 5363.138 a | 125.538 b | 26.872 c | 42.72 |
| S4:23 (2,4,5,12) | 765.391 | 5864.023 a | 48.632 b | 26.809 c | 120.58 |
| S4:24 (2,5,5,12) | 779.407 | 26,335.227 a | 1446.972 b | 301.079 c | 18.20 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Vendemiatti, E.; Therezan, R.; Vicente, M.H.; Pinto, M.d.S.; Bergau, N.; Yang, L.; Bernardi, W.F.; Alencar, S.M.d.; Zsögön, A.; Tissier, A.; et al. The Genetic Complexity of Type-IV Trichome Development Reveals the Steps towards an Insect-Resistant Tomato. Plants 2022, 11, 1309. https://doi.org/10.3390/plants11101309
Vendemiatti E, Therezan R, Vicente MH, Pinto MdS, Bergau N, Yang L, Bernardi WF, Alencar SMd, Zsögön A, Tissier A, et al. The Genetic Complexity of Type-IV Trichome Development Reveals the Steps towards an Insect-Resistant Tomato. Plants. 2022; 11(10):1309. https://doi.org/10.3390/plants11101309
Chicago/Turabian StyleVendemiatti, Eloisa, Rodrigo Therezan, Mateus H. Vicente, Maísa de Siqueira Pinto, Nick Bergau, Lina Yang, Walter Fernando Bernardi, Severino M. de Alencar, Agustin Zsögön, Alain Tissier, and et al. 2022. "The Genetic Complexity of Type-IV Trichome Development Reveals the Steps towards an Insect-Resistant Tomato" Plants 11, no. 10: 1309. https://doi.org/10.3390/plants11101309
APA StyleVendemiatti, E., Therezan, R., Vicente, M. H., Pinto, M. d. S., Bergau, N., Yang, L., Bernardi, W. F., Alencar, S. M. d., Zsögön, A., Tissier, A., Benedito, V. A., & Peres, L. E. P. (2022). The Genetic Complexity of Type-IV Trichome Development Reveals the Steps towards an Insect-Resistant Tomato. Plants, 11(10), 1309. https://doi.org/10.3390/plants11101309







