Characterization of Two BAHD Acetyltransferases Highly Expressed in the Flowers of Jasminum sambac (L.) Aiton
Abstract
:1. Introduction
2. Results
2.1. Profiling Floral Scent Volatiles in J. sambac
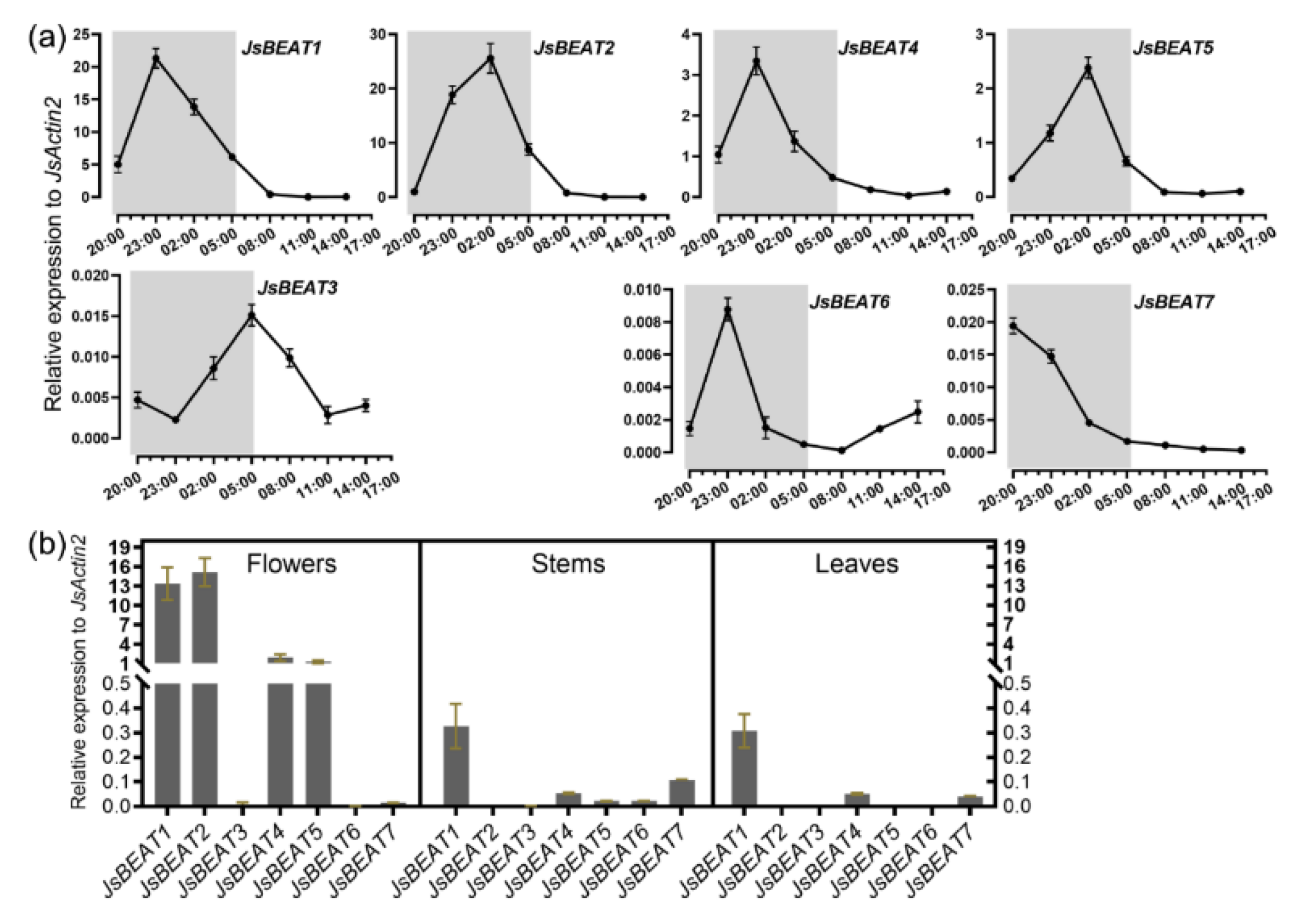
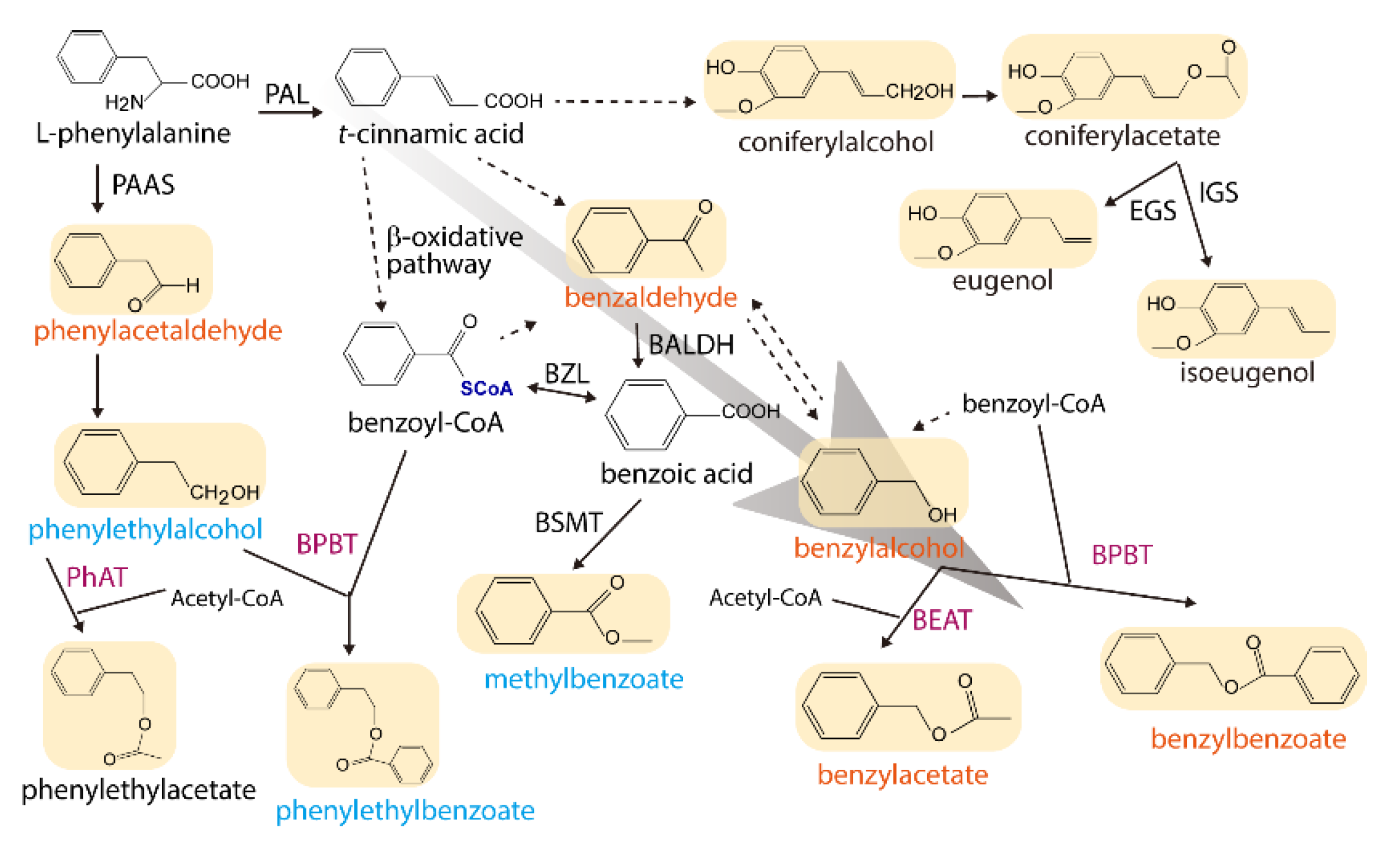
2.2. Cloning of Petal-Specific BAHD-Like cDNAs
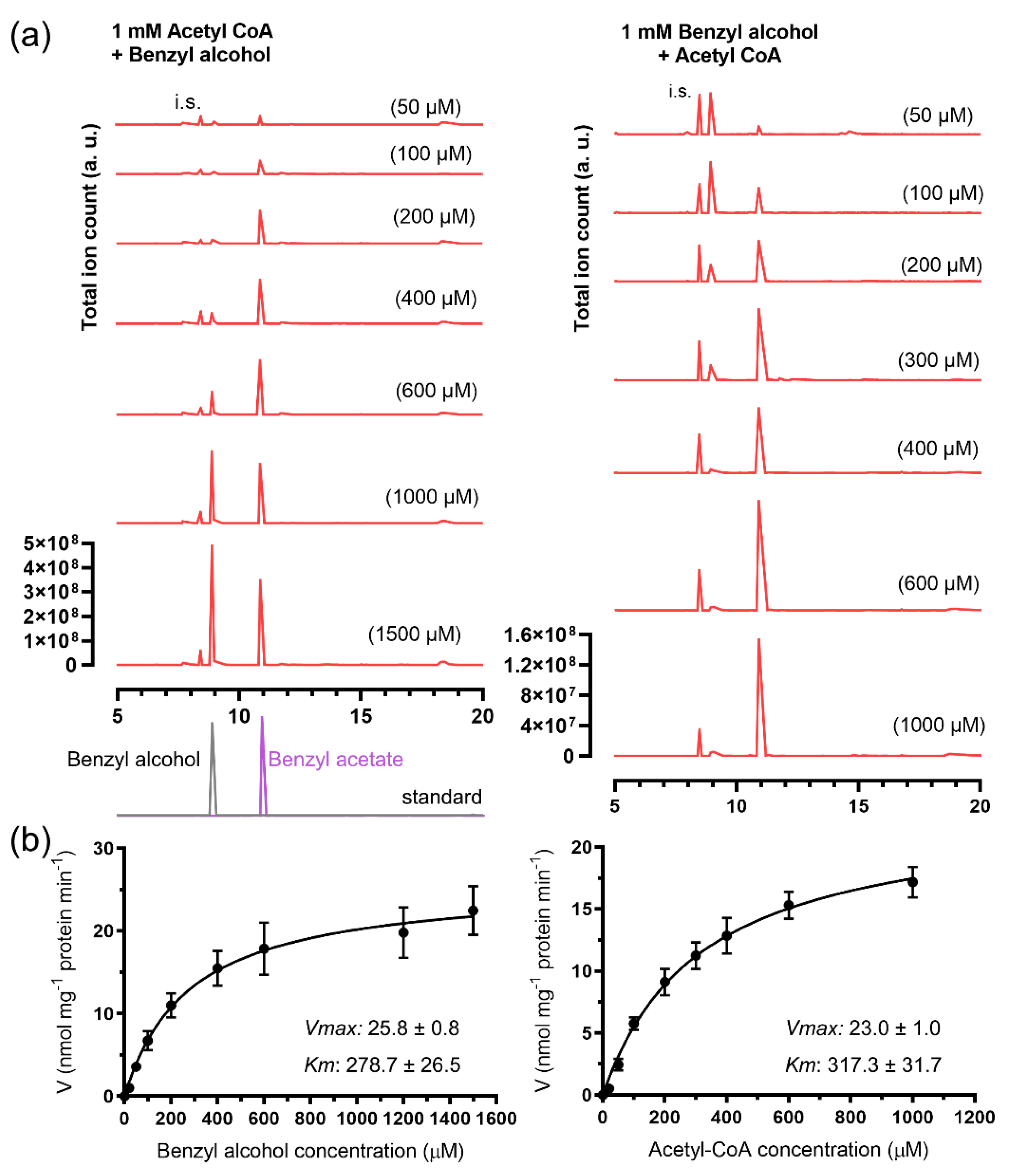
2.3. Enzymatic Characterization of the Recombinant JsBEAT1 and JsBEAT2
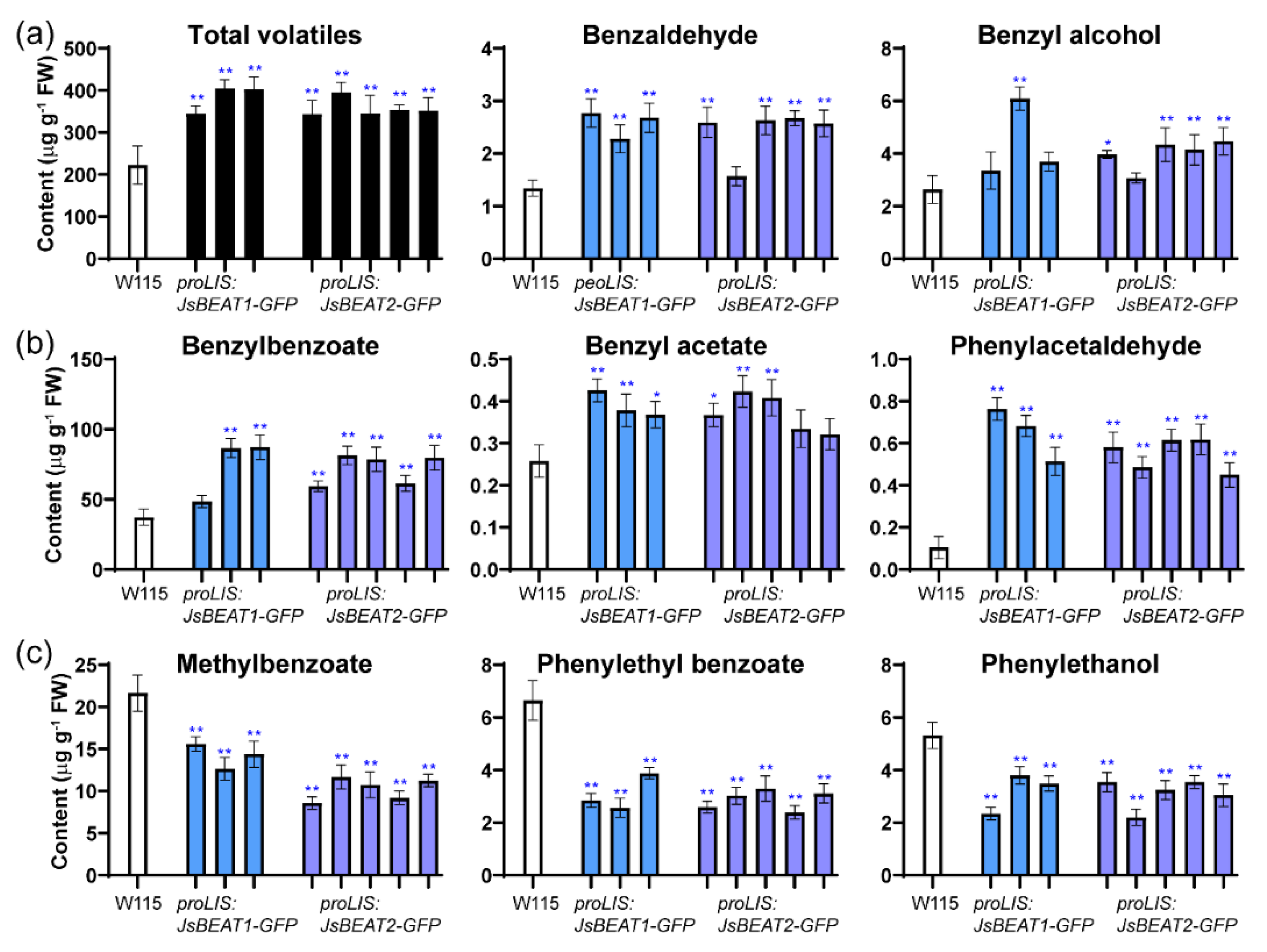
2.4. Ectopic Expression of JsBEAT1 and JsBEAT2 in Flowers of Transgenic Petunia hybrida Induced More Benzylbenzoate and Benzylacetate
3. Discussion
4. Materials and Methods
4.1. Plants and Culture Conditions
4.2. Volatile Extration and GC-MS Determination
4.3. RNA Isolation, Real-Time RT-PCR, Full-Length cDNA Cloning, and Vector Construction
4.4. Recombinant Proteins and In Vitro Enzyme Reaction
4.5. Transient Expression and Stable Transformation
4.6. Confocal Fluorescence Microscopy
4.7. Statistical Analysis
4.8. Accession Number
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Leong, B.J.; Last, R.L. Promiscuity, impersonation and accommodation: Evolution of plant specialized metabolism. Curr. Opin. Struct. Biol. 2017, 47, 105–112. [Google Scholar] [CrossRef]
- Pichersky, E.; Lewinsohn, E. Convergent evolution in plant specialized metabolism. Annu. Rev. Plant Biol. 2011, 62, 549–566. [Google Scholar] [CrossRef] [Green Version]
- Pichersky, E.; Noel, J.P.; Dudareva, N. Biosynthesis of plant volatiles: Nature’s diversity and ingenuity. Science 2006, 311, 808–811. [Google Scholar] [CrossRef] [Green Version]
- Knudsen, J.T.; Eriksson, R.; Gershenzon, J.; Stahl, B. Diversity and distribution of floral scent. Bot. Rev. 2006, 72, 1–120. [Google Scholar] [CrossRef]
- Muhlemann, J.K.; Klempien, A.; Dudareva, N. Floral volatiles: From biosynthesis to function. Plant Cell Env. 2014, 37, 1936–1949. [Google Scholar] [CrossRef]
- Schiestl, F.P. Ecology and evolution of floral volatile-mediated information transfer in plants. New Phytol. 2015, 206, 571–577. [Google Scholar] [CrossRef]
- Kantsa, A.; Raguso, R.A.; Dyer, A.G.; Olesen, J.M.; Tscheulin, T.; Petanidou, T. Disentangling the role of floral sensory stimuli in pollination networks. Nat. Commun. 2018, 9, 1041. [Google Scholar] [CrossRef]
- de Groot, A.C.; Schmidt, E. Essential Oils, Part VI: Sandalwood Oil, Ylang-Ylang Oil, and Jasmine Absolute. Dermatitis 2017, 28, 14–21. [Google Scholar] [CrossRef]
- Bera, P.; Kotamreddy, J.N.; Samanta, T.; Maiti, S.; Mitra, A. Inter-specific variation in headspace scent volatiles composition of four commercially cultivated jasmine flowers. Nat. Prod. Res. 2015, 29, 1328–1335. [Google Scholar] [CrossRef]
- Barman, M.; Mitra, A. Temporal relationship between emitted and endogenous floral scent volatiles in summer- and winter-blooming Jasminum species. Physiol. Plant. 2019, 166, 946–959. [Google Scholar] [CrossRef]
- Yu, Y.; Lyu, S.; Chen, D.; Lin, Y.; Chen, J.; Chen, G.; Ye, N. Volatiles Emitted at Different Flowering Stages of Jasminum sambac and Expression of Genes Related to α-Farnesene Biosynthesis. Molecules 2017, 22, 546. [Google Scholar] [CrossRef] [Green Version]
- Pragadheesh, V.S.; Chanotiya, C.S.; Rastogi, S.; Shasany, A.K. Scent from Jasminum grandiflorum flowers: Investigation of the change in linalool enantiomers at various developmental stages using chemical and molecular methods. Phytochemistry 2017, 140, 83–94. [Google Scholar] [CrossRef]
- Bera, P.; Mukherjee, C.; Mitra, A. Enzymatic production and emission of floral scent volatiles in Jasminum sambac. Plant Sci. Int. J. Exp. Plant Biol. 2017, 256, 25–38. [Google Scholar] [CrossRef]
- Pragadheesh, V.S.; Yadav, A.; Chanotiya, C.S.; Rout, P.K.; Uniyal, G.C. Monitoring the Emission of Volatile Organic Compounds from Flowers of Jasminum sambac Using Solid-Phase Micro-extraction Fibers and Gas Chromatography with Mass Spectrometry Detection. Nat. Prod. Commun. 2011, 6, 1333–1338. [Google Scholar] [CrossRef] [Green Version]
- D’Auria, J.C. Acyltransferases in plants: A good time to be BAHD. Curr. Opin. Plant Biol. 2006, 9, 331–340. [Google Scholar] [CrossRef]
- St-Pierre, B.; Laflamme, P.; Alarco, A.M.; De Luca, V. The terminal O-acetyltransferase involved in vindoline biosynthesis defines a new class of proteins responsible for coenzyme A-dependent acyl transfer. Plant J. 1998, 14, 703–713. [Google Scholar] [CrossRef]
- Dudareva, N.; D’Auria, J.C.; Nam, K.H.; Raguso, R.A.; Pichersky, E. Acetyl-CoA:benzylalcohol acetyltransferase--an enzyme involved in floral scent production in Clarkia breweri. Plant J. 1998, 14, 297–304. [Google Scholar] [CrossRef] [Green Version]
- Yang, Q.; Reinhard, K.; Schiltz, E.; Matern, U. Characterization and heterologous expression of hydroxycinnamoyl/benzoyl-CoA:anthranilate N-hydroxycinnamoyl/benzoyltransferase from elicited cell cultures of carnation, Dianthus caryophyllus L. Plant Mol. Biol. 1997, 35, 777–789. [Google Scholar] [CrossRef]
- Bontpart, T.; Cheynier, V.; Ageorges, A.; Terrier, N. BAHD or SCPL acyltransferase? What a dilemma for acylation in the world of plant phenolic compounds. New Phytol. 2015, 208, 695–707. [Google Scholar] [CrossRef]
- Ma, X.Y.; Koepke, J.; Panjikar, S.; Fritzsch, G.; Stockigt, J. Crystal structure of vinorine synthase, the first representative of the BAHD superfamily. J. Biol. Chem. 2005, 280, 13576–13583. [Google Scholar] [CrossRef] [Green Version]
- Tuominen, L.K.; Johnson, V.E.; Tsai, C.J. Differential phylogenetic expansions in BAHD acyltransferases across five angiosperm taxa and evidence of divergent expression among Populus paralogues. BMC Genom. 2011, 12, 236. [Google Scholar] [CrossRef] [Green Version]
- Wang, P.P.; Wei, P.N.; Niu, F.F.; Liu, X.F.; Zhang, H.L.; Lyu, M.L.; Yuan, Y.; Wu, B.H. Cloning and Functional Assessments of Floral-Expressed SWEET Transporter Genes from Jasminum sambac. Int. J. Mol. Sci. 2019, 20, 4001. [Google Scholar] [CrossRef] [Green Version]
- D’Auria, J.C.; Chen, F.; Pichersky, E. Characterization of an Acyltransferase Capable of Synthesizing Benzylbenzoate and Other Volatile Esters in Flowers and Damaged Leaves of Clarkia breweri. Plant Physiol. 2002, 130, 466–476. [Google Scholar] [CrossRef] [Green Version]
- Boatright, J.; Negre, F.; Chen, X.; Kish, C.M.; Wood, B.; Peel, G.; Orlova, I.; Gang, D.; Rhodes, D.; Dudareva, N. Understanding in vivo benzenoid metabolism in petunia petal tissue. Plant Physiol. 2004, 135, 1993–2011. [Google Scholar] [CrossRef] [Green Version]
- Chedgy, R.J.; Kollner, T.G.; Constabel, C.P. Functional characterization of two acyltransferases from Populus trichocarpa capable of synthesizing benzyl benzoate and salicyl benzoate, potential intermediates in salicinoid phenolic glycoside biosynthesis. Phytochemistry 2015, 113, 149–159. [Google Scholar] [CrossRef]
- Adebesin, F.; Widhalm, J.R.; Boachon, B.; Lefevre, F.; Pierman, B.; Lynch, J.H.; Alam, I.; Junqueira, B.; Benke, R.; Ray, S.; et al. Emission of volatile organic compounds from petunia flowers is facilitated by an ABC transporter. Science 2017, 356, 1386–1388. [Google Scholar] [CrossRef] [Green Version]
- Dudareva, N.; Cseke, L.; Blanc, V.M.; Pichersky, E. Evolution of floral scent in Clarkia: Novel patterns of S-linalool synthase gene expression in the C. breweri flower. Plant Cell 1996, 8, 1137–1148. [Google Scholar] [CrossRef] [Green Version]
- Klempien, A.; Kaminaga, Y.; Qualley, A.; Nagegowda, D.A.; Widhalm, J.R.; Orlova, I.; Shasany, A.K.; Taguchi, G.; Kish, C.M.; Cooper, B.R.; et al. Contribution of CoA ligases to benzenoid biosynthesis in petunia flowers. Plant Cell 2012, 24, 2015–2030. [Google Scholar] [CrossRef] [Green Version]
- Colquhoun, T.A.; Marciniak, D.M.; Wedde, A.E.; Kim, J.Y.; Schwieterman, M.L.; Levin, L.A.; Van Moerkercke, A.; Schuurink, R.C.; Clark, D.G. A peroxisomally localized acyl-activating enzyme is required for volatile benzenoid formation in a Petunia x hybrida cv. ‘Mitchell Diploid’ flower. J. Exp. Bot. 2012, 63, 4821–4833. [Google Scholar] [CrossRef] [Green Version]
- Dexter, R.; Qualley, A.; Kish, C.M.; Ma, C.J.; Koeduka, T.; Nagegowda, D.A.; Dudareva, N.; Pichersky, E.; Clark, D. Characterization of a petunia acetyltransferase involved in the biosynthesis of the floral volatile isoeugenol. Plant J. 2007, 49, 265–275. [Google Scholar] [CrossRef] [Green Version]
- Wang, J.; De Luca, V. The biosynthesis and regulation of biosynthesis of Concord grape fruit esters, including ‘foxy’ methylanthranilate. Plant J. 2005, 44, 606–619. [Google Scholar] [CrossRef]
- D’Auria, J.C.; Reichelt, M.; Luck, K.; Svatoš, A.; Gershenzon, J. Identification and characterization of the BAHD acyltransferase malonyl CoA:anthocyanidin 5-O-glucoside-6″-O-malonyltransferase (At5MAT) in Arabidopsis thaliana. FEBS Lett. 2007, 581, 872–878. [Google Scholar] [CrossRef] [Green Version]
- Oshikiri, H.; Watanabe, B.; Yamamoto, H.; Yazaki, K.; Takanashi, K. Two BAHD Acyltransferases Catalyze the Last Step in the Shikonin/Alkannin Biosynthetic Pathway. Plant Physiol. 2020, 184, 753–761. [Google Scholar] [CrossRef]
- Werner, V.; Petersen, M. A BAHD hydroxycinnamoyltransferase from Actaea racemosa catalyses the formation of fukinolic and cimicifugic acids. Planta 2019, 250, 475–485. [Google Scholar] [CrossRef]
- Legrand, G.; Delporte, M.; Khelifi, C.; Harant, A.; Vuylsteker, C.; Mörchen, M.; Hance, P.; Hilbert, J.L.; Gagneul, D. Identification and Characterization of Five BAHD Acyltransferases Involved in Hydroxycinnamoyl Ester Metabolism in Chicory. Front. Plant Sci. 2016, 7, 741. [Google Scholar] [CrossRef]
- Colquhoun, T.A.; Verdonk, J.C.; Schimmel, B.C.J.; Tieman, D.M.; Underwood, B.A.; Clark, D.G. Petunia floral volatile benzenoid/phenylpropanoid genes are regulated in a similar manner. Phytochemistry 2010, 71, 158–167. [Google Scholar] [CrossRef]
- Karimi, M.; Inze, D.; Depicker, A. GATEWAY vectors for Agrobacterium-mediated plant transformation. Trends Plant Sci. 2002, 7, 193–195. [Google Scholar] [CrossRef]
- Katoh, K.; Toh, H. Recent developments in the MAFFT multiple sequence alignment program. Brief. Bioinform. 2008, 9, 286–298. [Google Scholar] [CrossRef] [Green Version]
- Larkin, M.A.; Blackshields, G.; Brown, N.P.; Chenna, R.; McGettigan, P.A.; McWilliam, H.; Valentin, F.; Wallace, I.M.; Wilm, A.; Lopez, R.; et al. Clustal W and Clustal X version 2.0. Bioinformatics 2007, 23, 2947–2948. [Google Scholar] [CrossRef] [Green Version]
- Horsch, R.B.; Fry, J.E.; Hoffmann, N.L.; Wallroth, M.; Eichholtz, D.; Rogers, S.G.; Fraley, R.T. A Simple and General Method for Transferring Genes into Plants. Science 1985, 227, 1229–1231. [Google Scholar] [CrossRef]
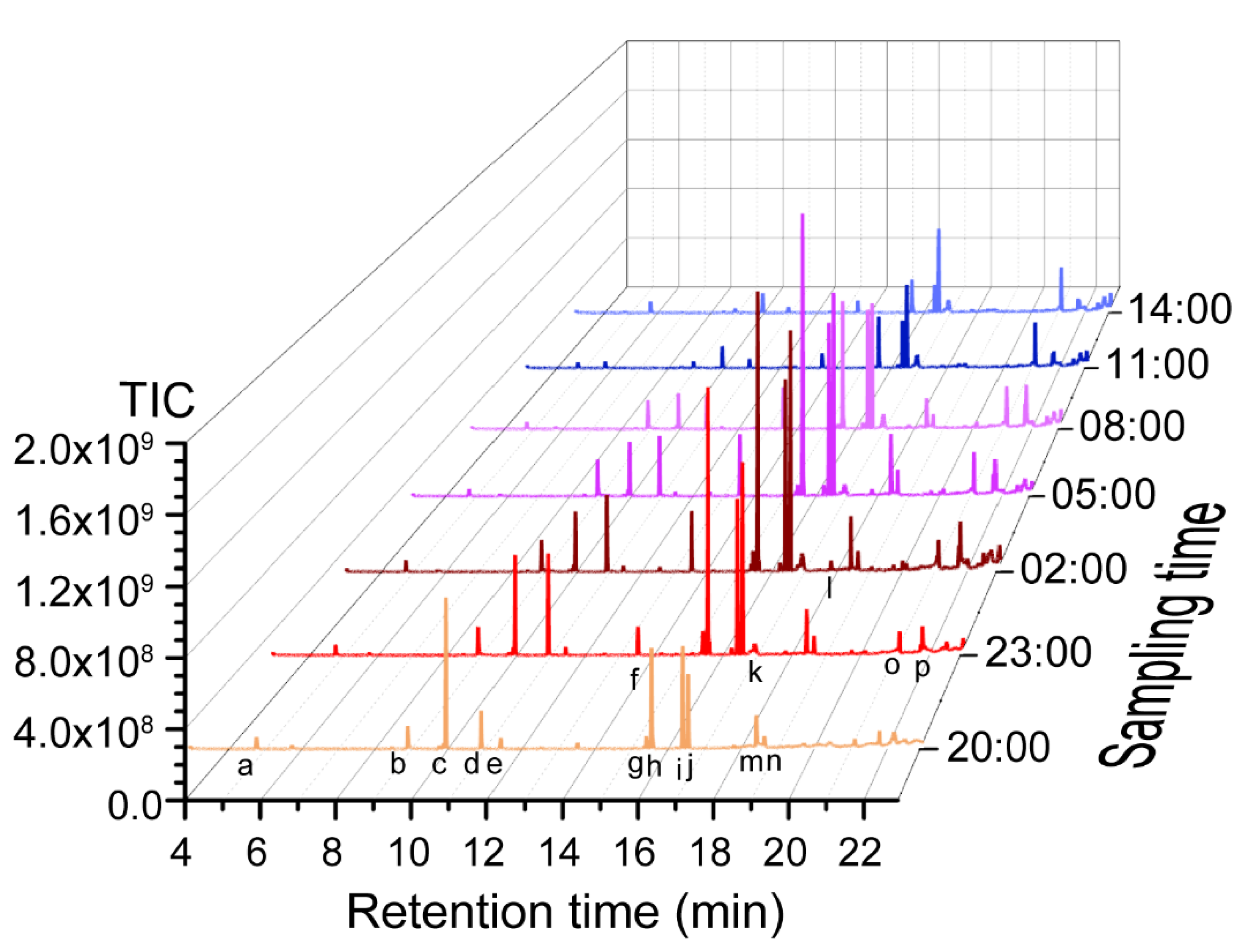

| Time | 3-Hexenal | Concentration (mg g−1 FW) * | Benzoic Acid-2-Propenyl Ester | ||||
|---|---|---|---|---|---|---|---|
| Benzyl Alcohol | Benzyl Acetate | Methyl Anthranilate | cis-3-Hexenyl Benzoate | Benzyl Benzoate | |||
| 20:00 | 16.5 ± 1.5 a | 41.3 ± 2.3 c | 34.2 ± 1.7 e | 10.7 ± 0.6 f | 140.8 ± 1.3 d | 14.3 ± 0.2 e | 50.4 ± 8.4 c |
| 23:00 | 15.3 ± 0.9 a | 48.3 ± 3.5 b | 199.4 ± 11.3 b | 50.1 ± 3.1 d | 208.9 ± 13.3 a | 23.9 ± 1.0 c | 69.0 ± 7.04 b |
| 02:00 | 14.2 ± 1.1 a | 48.3 ± 3.2 b | 227.2 ± 10.0 a | 87.2 ± 1. 7 b | 228.2 ± 7.8 a | 25.2 ± 0.62 b | 77.6 ± 2.5 a |
| 05:00 | 10.9 ± 0.6 c | 55.4 ± 3.4 a | 148.8 ± 5.9 c | 95. 4 ± 3.4 a | 170.2 ± 8.2 b | 29.7 ± 1.1 a | 80.9 ± 2.0 a |
| 08:00 | 11.1 ± 0.7 bc | 52.6 ± 1.2 a | 75.9 ± 1.3 d | 62.9 ± 2.9 c | 134.8 ± 2.5 c | 19.8 ± 0.6 d | 42. 5± 0.6 d |
| 11:00 | 12.5 ± 1.1 b | 42.8 ± 0.3 c | 33.4 ± 0. 8 ef | 47.9 ± 1.1 d | 68.7 ± 2.1 d | 10.1 ± 0.2 fg | 6.5 ± 0.2 e |
| 14:00 | 7.7 ± 1.3 d | 15.2 ± 1.2 d | 27.8 ± 0.5 fg | 26.6 ± 1.2 e | 44.5 ± 3.6 e | 8.9 ± 0.8 g | 8.2 ± 1.0 e |
| 17:00 | 2.3 ± 0.4 e | 9.0 ± 0.2 e | 24.1 ± 0.8 g | 23.1 ± 1.7 e | 13.8 ± 0.7 f | 11.2 ± 1.1 f | 7.5 ± 0.8 e |
| Km | kcat | kcat/Km | ||
|---|---|---|---|---|
| μM | s−1 | nM−1 s−1 | ||
| GST-JsBEAT1 | Benzyl alcohol (with acetyl-CoA) | 447.3 ± 107.8 | 201.5 ± 4.1 | 450.5 ± 241.8 |
| Acetyl-CoA (with benzyl alcohol) | 546.0 ± 94.5 | 316.8 ± 4.4 | 580.2 ± 173.6 | |
| 6xHis-JsBEAT2 | Benzyl alcohol (with acetyl-CoA) | 278.7 ± 26.5 | 36.8 ± 0.6 | 132.0 ± 96.5 |
| Acetyl-CoA (with benzyl alcohol) | 317.3 ± 31.7 | 25.7 ± 0.4 | 81.0 ± 67.4 | |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wang, Y.; Zhang, H.; Wan, C.; He, X.; Huang, J.; Lyu, M.; Yuan, Y.; Wu, B. Characterization of Two BAHD Acetyltransferases Highly Expressed in the Flowers of Jasminum sambac (L.) Aiton. Plants 2022, 11, 13. https://doi.org/10.3390/plants11010013
Wang Y, Zhang H, Wan C, He X, Huang J, Lyu M, Yuan Y, Wu B. Characterization of Two BAHD Acetyltransferases Highly Expressed in the Flowers of Jasminum sambac (L.) Aiton. Plants. 2022; 11(1):13. https://doi.org/10.3390/plants11010013
Chicago/Turabian StyleWang, Yuting, Hongliang Zhang, Chao Wan, Xian He, Jinfeng Huang, Meiling Lyu, Yuan Yuan, and Binghua Wu. 2022. "Characterization of Two BAHD Acetyltransferases Highly Expressed in the Flowers of Jasminum sambac (L.) Aiton" Plants 11, no. 1: 13. https://doi.org/10.3390/plants11010013
APA StyleWang, Y., Zhang, H., Wan, C., He, X., Huang, J., Lyu, M., Yuan, Y., & Wu, B. (2022). Characterization of Two BAHD Acetyltransferases Highly Expressed in the Flowers of Jasminum sambac (L.) Aiton. Plants, 11(1), 13. https://doi.org/10.3390/plants11010013






