Differences in Nectar Traits between Ornithophilous and Entomophilous Plants on Mount Cameroon
Abstract
1. Introduction
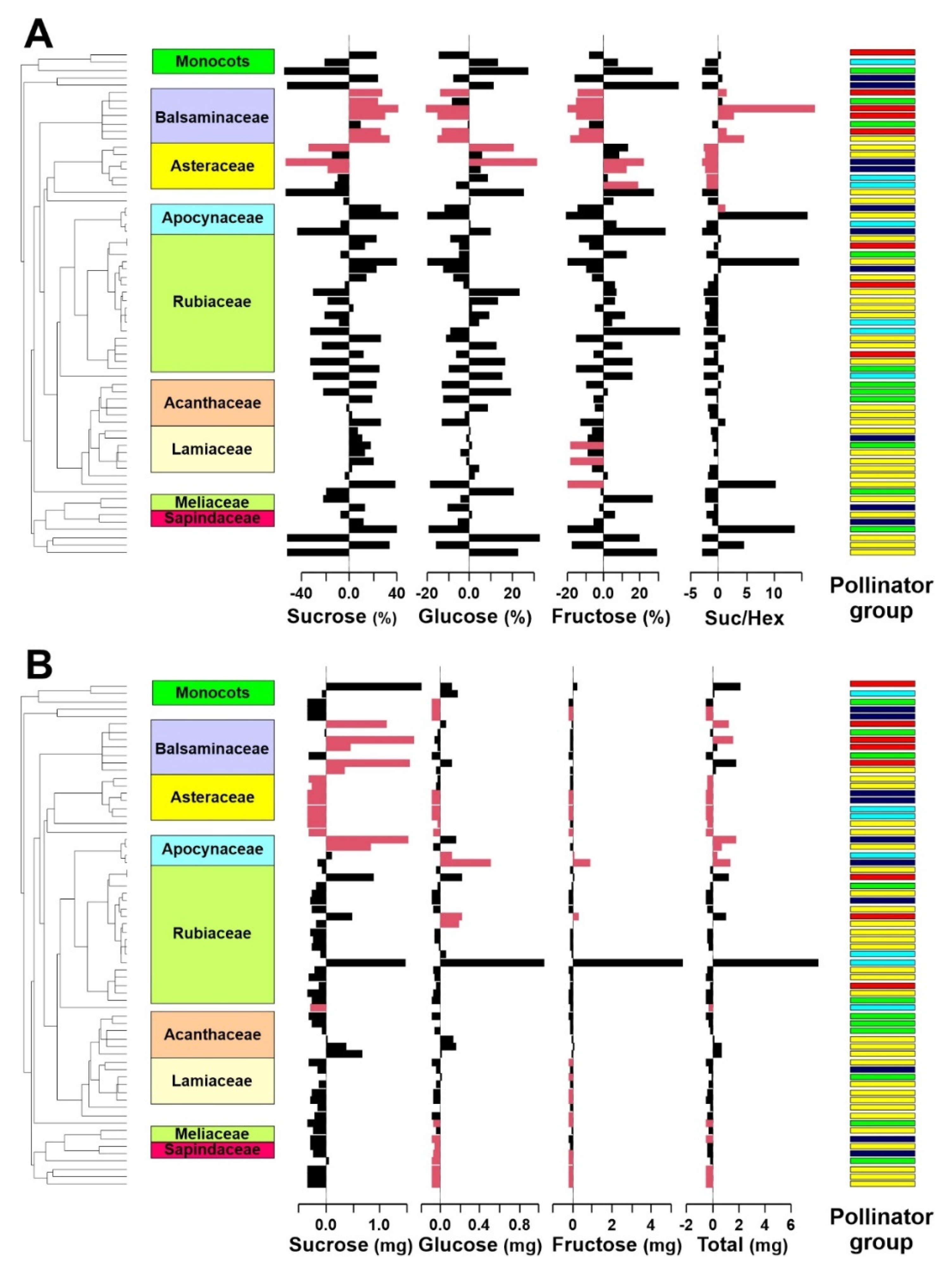
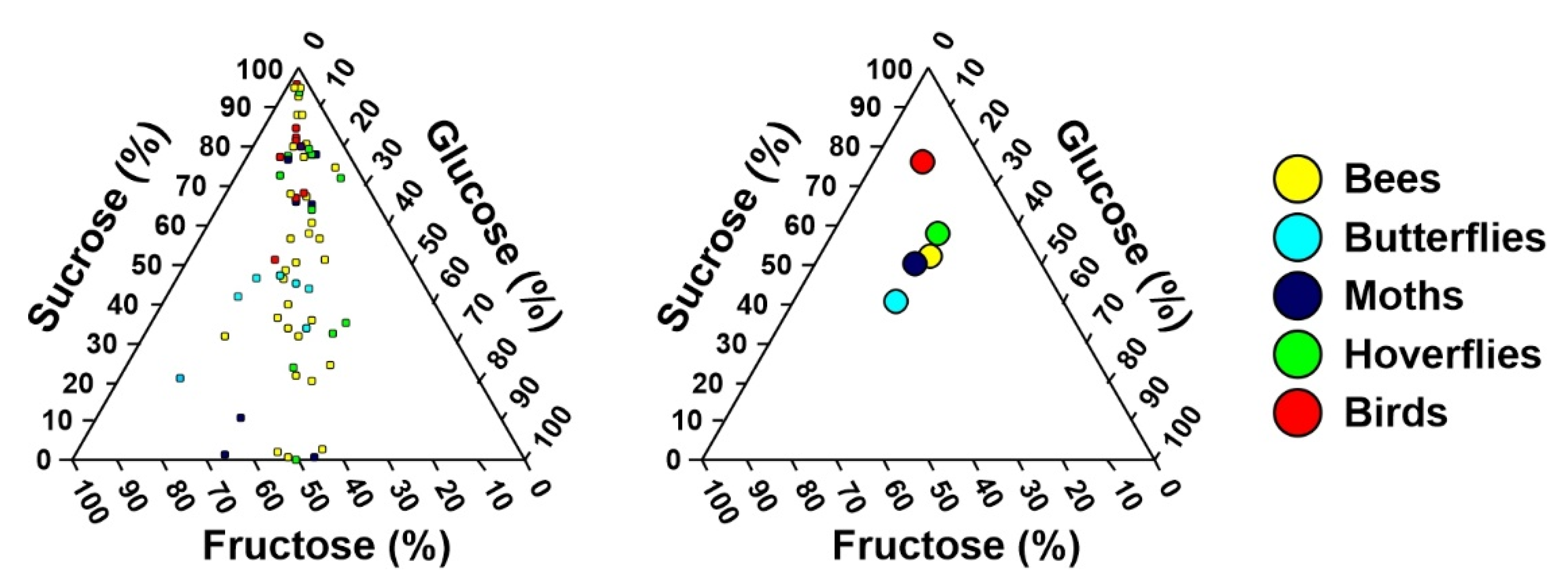
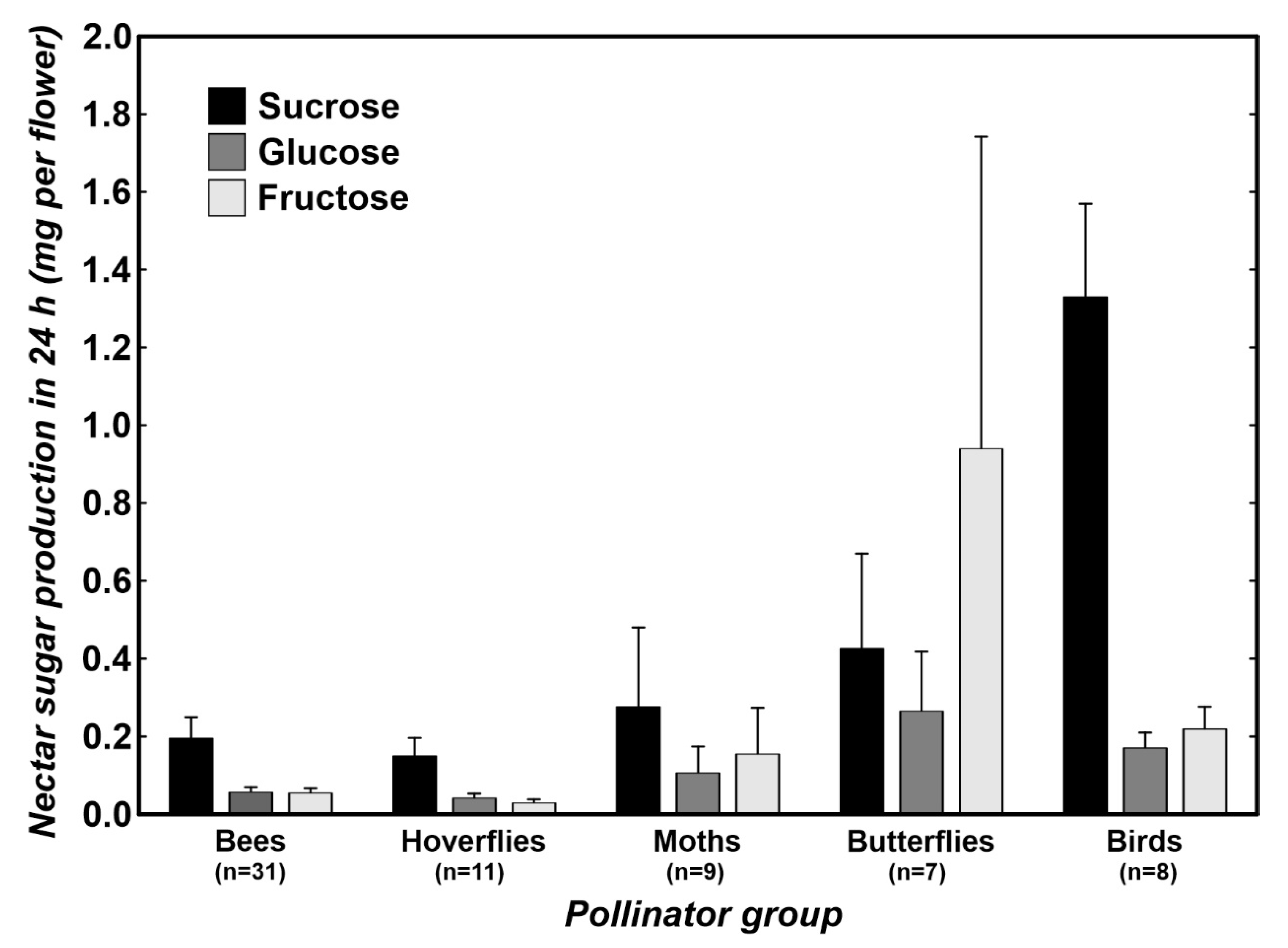
2. Results
3. Discussion
3.1. Differences in Nectar Properties in Relation to the Main Pollinator
3.2. Phylogenetic Signal
4. Materials and Methods
4.1. Study Site
4.2. Nectar Collection and Analyses
4.3. Recording Visitors
4.4. Phylogenetic and Statistical Analyses
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Endress, P.K. Floral structure and evolution of primitive angiosperms: Recent advances. Plant. Syst. Evol. 1994, 192, 79–97. [Google Scholar] [CrossRef]
- Labandeira, C.C.; Kvaček, J.; Mostovski, M.B. Pollination drops, pollen, and insect pollination of Mesozoic gymnosperms. TAXON 2007, 56, 663–695. [Google Scholar] [CrossRef]
- Lau, J.Y.Y.; Pang, C.; Ramsden, L.; Saunders, R.M.K. Stigmatic exudate in the Annonaceae: Pollinator reward, pollen germination medium or extragynoecial compitum? J. Integr. Plant Biol. 2017, 59, 881–894. [Google Scholar] [CrossRef] [PubMed]
- González-Teuber, M.; Heil, M. Nectar chemistry is tailored for both attraction of mutualists and protection from exploiters. Plant Signal. Behav. 2009, 4, 809–813. [Google Scholar] [CrossRef] [PubMed]
- Nicolson, S.W.; Thornburg, R.W. Nectar Chemistry. In Nectaries and Nectar; Nicolson, S.W., Nepi, M., Pacini, E., Eds.; Springer Science and Business Media LLC: Dordrecht, The Netherlands, 2007; pp. 215–264. [Google Scholar]
- Davis, A.R.; Pylatuik, J.D.; Paradis, J.C.; Low, N.H. Nectar-carbohydrate production and composition vary in relation to nectary anatomy and location within individual flowers of several species of Brassicaceae. Planta 1998, 205, 305–318. [Google Scholar] [CrossRef] [PubMed]
- Belmonte, E.; Cardemil, L.; Arroyo, M.T.K. Floral nectary structure and nectar composition in Eccremocarpus scaber (Bignoniaceae), a hummingbird-pollinated plant of central Chile. Am. J. Bot. 1994, 81, 493–503. [Google Scholar] [CrossRef]
- Lohaus, G.; Schwerdtfeger, M. Comparison of Sugars, Iridoid Glycosides and Amino Acids in Nectar and Phloem Sap of Maurandya barclayana, Lophospermum erubescens, and Brassica napus. PLoS ONE 2014, 9, e87689. [Google Scholar] [CrossRef]
- Heil, M. Nectar: Generation, regulation and ecological functions. Trends Plant Sci. 2011, 16, 191–200. [Google Scholar] [CrossRef] [PubMed]
- Wenzler, M.; Hölscher, D.; Oerther, T.; Schneider, B. Nectar formation and floral nectary anatomy of Anigozanthos flavidus: A combined magnetic resonance imaging and spectroscopy study. J. Exp. Bot. 2008, 59, 3425–3434. [Google Scholar] [CrossRef][Green Version]
- Heil, M.; Rattke, J.; Boland, W. Postsecretory Hydrolysis of Nectar Sucrose and Specialization in Ant/Plant Mutualism. Science 2005, 308, 560–563. [Google Scholar] [CrossRef]
- Pozo, M.I.; Van Kemenade, G.; Van Oystaeyen, A.; Aledón-Catalá, T.; Benavente, A.; Ende, W.V.D.; Wäckers, F.; Jacquemyn, H. The impact of yeast presence in nectar on bumble bee behavior and fitness. Ecol. Monogr. 2020, 90. [Google Scholar] [CrossRef]
- Jackson, S.; Nicolson, S.W. Xylose as a nectar sugar: From biochemistry to ecology. Comp. Biochem. Physiol. Part B Biochem. Mol. Biol. 2002, 131, 613–620. [Google Scholar] [CrossRef]
- Baker, H.G.; Baker, I. Floral nectar constituents in relation to pollinator type. In Handbook of Experimental Pollination Biology; Jones, C.E., Little, R.J., Eds.; Van Nostrand Reinhold: New York, NY, USA, 1983; pp. 117–141. [Google Scholar]
- Abrahamczyk, S.; Kessler, M.; Hanley, D.; Karger, D.N.; Müller, M.P.J.; Knauer, A.C.; Keller, F.; Schwerdtfeger, M.; Humphreys, A.M. Pollinator adaptation and the evolution of floral nectar sugar composition. J. Evol. Biol. 2016, 30, 112–127. [Google Scholar] [CrossRef]
- Adler, L.S.; Bronstein, J.L. Attracting antagonists: Does floral nectar increase leaf herbivory? Ecology 2004, 85, 1519–1526. [Google Scholar] [CrossRef]
- Chalcoff, V.R.; Gleiser, G.; Ezcurra, C.; Aizen, M.A. Pollinator type and secondarily climate are related to nectar sugar composition across the angiosperms. Evol. Ecol. 2017, 31, 585–602. [Google Scholar] [CrossRef]
- Percival, M.S. Types of nectar in angiosperms. New Phytol. 1961, 60, 235–281. [Google Scholar] [CrossRef]
- Mitchell, R.J. Heritability of nectar traits: Why do we know so little? Ecology 2004, 85, 1527–1533. [Google Scholar] [CrossRef]
- Parachnowitsch, A.L.; Manson, J.S.; Sletvold, N. Evolutionary ecology of nectar. Ann. Bot. 2018, 123, 247–261. [Google Scholar] [CrossRef] [PubMed]
- Faegri, K.; van der Pijl, L. The Principles of Pollination Ecology; Pergamon Press: Oxford, UK, 1979. [Google Scholar]
- Petanidou, T. Sugars in Mediterranean Floral Nectars: An Ecological and Evolutionary Approach. J. Chem. Ecol. 2005, 31, 1065–1088. [Google Scholar] [CrossRef]
- Schmidt-Lebuhn, A.N.; Schwerdtfeger, M.; Kessler, M.; Lohaus, G. Phylogenetic constraints vs. ecology in the nectar composition of Acanthaceae. Flora Morphol. Distrib. Funct. Ecol. Plants 2007, 202, 62–69. [Google Scholar] [CrossRef]
- Perret, M.; Chautems, A.; Spichiger, R.; Peixoto, M.; Savolainen, V. Nectar Sugar Composition in Relation to Pollination Syndromes in Sinningieae (Gesneriaceae). Ann. Bot. 2001, 87, 267–273. [Google Scholar] [CrossRef] [PubMed]
- Galetto, L.; Bernardello, G. Nectar sugar composition in angiosperms from Chaco and Patagonia (Argentina): An animal visitor’s matter? Plant Syst. Evol. 2003, 238, 69–86. [Google Scholar] [CrossRef]
- Wolff, D. Nectar Sugar Composition and Volumes of 47 Species of Gentianales from a Southern Ecuadorian Montane Forest. Ann. Bot. 2006, 97, 767–777. [Google Scholar] [CrossRef]
- Galetto, L.; Bernardello, G.; Sosa, C.A. The relationship between floral nectar composition and visitors in Lycium (Solanaceae) from Argentina and Chile: What does it reflect? Flora Morphol. Distrib. Funct. Ecol. Plants 1998, 193, 303–314. [Google Scholar] [CrossRef]
- Rodríguez-Riaño, T.; Ortega-Olivencia, A.; López, J.; Pérez-Bote, J.L.; Navarro-Pérez, M.L. Main sugar composition of floral nectar in three species groups of Scrophularia (Scrophulariaceae) with different principal pollinators. Plant Biol. 2014, 16, 1075–1086. [Google Scholar] [CrossRef]
- Johnson, S.D.; Nicolson, S. Evolutionary associations between nectar properties and specificity in bird pollination systems. Biol. Lett. 2007, 4, 49–52. [Google Scholar] [CrossRef] [PubMed]
- Bartoš, M.; Janeček, Š.; Padyšáková, E.; Patáčová, E.; Altman, J.; Pešata, M.; Kantorová, J.; Tropek, R. Nectar properties of the sunbird-pollinated plant Impatiens sakeriana: A comparison with six other co-flowering species. South. Afr. J. Bot. 2012, 78, 63–74. [Google Scholar] [CrossRef]
- Cheke, R.A.; Mann, C.F.; Allen, R. Sunbirds: A Guide to the Sunbirds, Flowerpeckers, Spiderhunters and Sugarbirds of the World; Christopher Helm: London, UK, 2001. [Google Scholar]
- Elisens, W.J.; Freeman, C.E. Floral Nectar Sugar Composition and Pollinator Type Among New World Genera in Tribe Antirrhineae (Scrophulariaceae). Am. J. Bot. 1988, 75, 971. [Google Scholar] [CrossRef]
- Van Wyk, B.E.; Whitehead, C.S.; Glen, H.F.; Hardy, D.S.; Van Jaarsveld, E.J.; Smith, G.F. Nectar sugar composition in the subfamily Alooideaec (Asphodelaceae). Biochem. Syst. Ecol. 1993, 21, 249–253. [Google Scholar] [CrossRef]
- Nicolson, S.W.; Van Wyk, B.-E. Nectar Sugars in Proteaceae: Patterns and Processes. Aust. J. Bot. 1998, 46, 489–504. [Google Scholar] [CrossRef]
- Silva, F.D.A.; Chatt, E.C.; Mohd-Fadzil, N.-A.; Guirgis, A.; Guo, X.; Nettleton, D.; Nikolau, B.J.; Thornburg, R. Metabolomic Profiling of Nicotiana Spp. Nectars Indicate That Pollinator Feeding Preference Is a Stronger Determinant than Plant Phylogenetics in Shaping Nectar Diversity. Metabolites 2020, 10, 214. [Google Scholar] [CrossRef]
- Göttlinger, T.; Schwerdtfeger, M.; Tiedge, K.; Lohaus, G. What Do Nectarivorous Bats Like? Nectar Composition in Bromeliaceae With Special Emphasis on Bat-Pollinated Species. Front. Plant Sci. 2019, 10, 205. [Google Scholar] [CrossRef]
- Stiles, F.G.; Freeman, C.E. Patterns in Floral Nectar Characteristics of Some Bird-Visited Plant Species from Costa Rica. Biotropica 1993, 25, 191. [Google Scholar] [CrossRef]
- Baker, H.G.; Baker, I.; Hodges, S. Sugar Composition of Nectars and Fruits Consumed by Birds and Bats in the Tropics and Subtropics. Biotropica 1998, 30, 559–586. [Google Scholar] [CrossRef]
- Pender, R.J.; Morden, C.W.; Paull, R.E. Investigating the pollination syndrome of the Hawaiian lobeliad genus Clermontia (Campanulaceae) using floral nectar traits. Am. J. Bot. 2014, 101, 201–205. [Google Scholar] [CrossRef]
- Del Rio, C.M. Sugar Preferences in Hummingbirds: The Influence of Subtle Chemical Differences on Food Choice. Condor 1990, 92, 1022. [Google Scholar] [CrossRef]
- Chalcoff, V.R.; Aizen, M.A.; Galetto, L. Sugar preferences of the green-backed firecrown hummingbird (Sephanoides sephaniodes): A field experiment. Auk 2008, 125, 60–66. [Google Scholar] [CrossRef]
- Downs, C.T.; Perrin, M.R. Sugar preferences of some southern African nectarivorous birds. Ibis 1996, 138, 455–459. [Google Scholar] [CrossRef]
- Fleming, P.A.; Bakken, B.H.; Lotz, C.N.; Nicolson, S.W. Concentration and temperature effects on sugar intake and preferences in a sunbird and a hummingbird. Funct. Ecol. 2004, 18, 223–232. [Google Scholar] [CrossRef]
- Brown, M.; Downs, C.T.; Johnson, S.D. Sugar preferences of nectar feeding birds—A comparison of experimental techniques. J. Avian Biol. 2008, 39, 479–483. [Google Scholar] [CrossRef]
- Lotz, C.N.; Nicolson, S.W. Sugar Preferences of a Nectarivorus Passerine Bird, the Lesser Double-Collared Sunbird (Nectarinia Chalybea). Funct. Ecol. 1996, 10, 360. [Google Scholar] [CrossRef]
- Klomberg, Y.; Tropek, R.; Mertens, J.E.J.; Kobe, I.N.; Hodeček, J.; Raška, J.; Fominka, N.T.; Souto-Vilarós, D.; Janeček, Š. Spatiotemporal shifts in the role of floral traits in shaping tropical plant-pollinator interactions. biorXiv 2020. [Google Scholar] [CrossRef]
- Janeček, Š.; Riegert, J.; Sedláček, O.; Bartoš, M.; Hořák, D.; Reif, J.; Padyšáková, E.; Fainová, D.; Antczak, M.; Pešata, M.; et al. Food selection by avian floral visitors: An important aspect of plant-flower visitor interactions in West Africa. Biol. J. Linn. Soc. 2012, 107, 355–367. [Google Scholar] [CrossRef]
- Nsor, C.A.; Godsoe, W.; Chapman, H.M. Promiscuous pollinators—Evidence from an Afromontane sunbird–plant pollen transport network. Biotropica 2019, 51, 538–548. [Google Scholar] [CrossRef]
- Bachman, W.W.; Waller, G.D. Honeybee Responses to Sugar Solutions of Different Compositions. J. Apic. Res. 1977, 16, 165–169. [Google Scholar] [CrossRef]
- Mallinger, R.E.; Prasifka, J.R. Bee visitation rates to cultivated sunflowers increase with the amount and accessibility of nectar sugars. J. Appl. Entomol. 2017, 141, 561–573. [Google Scholar] [CrossRef]
- Kelber, A. Sugar preferences and feeding strategies in the hawkmoth Macroglossum stellatarum. J. Comp. Physiol. A 2003, 189, 661–666. [Google Scholar] [CrossRef]
- Erhardt, A. Preferences and non-preferences for nectar constituents in Ornithoptera priamus poseidon (Lepidoptera, Papilionidae). Oecologia 1992, 90, 581–585. [Google Scholar] [CrossRef]
- Vandelook, F.; Janssens, S.B.; Gijbels, P.; Fischer, E.; Ende, W.V.D.; Honnay, O.; Abrahamczyk, S. Nectar traits differ between pollination syndromes in Balsaminaceae. Ann. Bot. 2019, 124, 269–279. [Google Scholar] [CrossRef]
- Torres, C.; Galetto, L. Are Nectar Sugar Composition and Corolla Tube Length Related to the Diversity of Insects that Visit Asteraceae Flowers? Plant Biol. 2002, 4, 360–366. [Google Scholar] [CrossRef]
- Petanidou, T.; Goethals, V.; Smets, E. Nectary structure of Labiatae in relation to their nectar secretion and characteristics in a Mediterranean shrub community? Does flowering time matter? Plant Syst. Evol. 2000, 225, 103–118. [Google Scholar] [CrossRef]
- Cable, S.; Cheek, M. The Plants of Mount Cameroon, a Conservation Checklist; Royal Botanic Gardens, Kew: London, UK, 1998. [Google Scholar]
- Ustjuzhanin, P.; Kovtunovich, V.; Sáfián, S.; Maicher, V.; Tropek, R. A newly discovered biodiversity hotspot of many-plumed moths in the Mount Cameroon area: First report on species diversity, with description of nine new species (Lepidoptera, Alucitidae). ZooKeys 2018, 777, 119–139. [Google Scholar] [CrossRef]
- Chlumská, Z.; Janeček, Š.; Doležal, J. How to Preserve Plant Samples for Carbohydrate Analysis? Test of Suitable Methods Applicable in Remote Areas. Folia Geobot. Phytotaxon. 2013, 49, 1–15. [Google Scholar] [CrossRef]
- Weinstein, B.G. Motion Meerkat: Integrating motion video detection and ecological monitoring. Methods Ecol. Evol. 2015, 6, 357–362. [Google Scholar] [CrossRef]
- Vogel, S. Blütenbiologische Typen als Elemente der Sippengliederung; Gustav Fischer: Jena, Germany, 1954. [Google Scholar]
- Willmer, P. Pollination and Floral Ecology; Princeton University Press: Princeton, New Jersey, USA, 2011. [Google Scholar]
- Smith, S.A.; Brown, J.W. Constructing a broadly inclusive seed plant phylogeny. Am. J. Bot. 2018, 105, 302–314. [Google Scholar] [CrossRef]
- Paradis, E.; Schliep, K. ape 5.0: An environment for modern phylogenetics and evolutionary analyses in R. Bioinformatics 2018, 35, 526–528. [Google Scholar] [CrossRef]
- R Core Team. R: A Language and Environment for Statistical Computing Computer Program, Version 3.6.1; R Foundation for Statistical Computing: Vienna, Austria, 2019; Newest Version; Available online: http://www.r-project.org (accessed on 15 May 2021).
- Keck, F.; Rimet, F.; Bouchez, A.; François, K. phylosignal: An R package to measure, test, and explore the phylogenetic signal. Ecol. Evol. 2016, 6, 2774–2780. [Google Scholar] [CrossRef]
- Münkemüller, T.; Lavergne, S.; Bzeznik, B.; Dray, S.; Jombart, T.; Schiffers, K.; Thuiller, W. How to measure and test phylogenetic signal. Methods Ecol. Evol. 2012, 3, 743–756. [Google Scholar] [CrossRef]
- Garland, T.; Dickerman, A.W.; Janis, C.M.; Jones, J.A. Phylogenetic Analysis of Covariance by Computer Simulation. Syst. Biol. 1993, 42, 265–292. [Google Scholar] [CrossRef]
- Harmon, L.J.; Weir, J.T.; Brock, C.D.; Glor, R.E.; Challenger, W. GEIGER: Investigating evolutionary radiations. Bioinformatics 2007, 24, 129–131. [Google Scholar] [CrossRef]
- Schluter, D.; Price, T.; Mooers, A.Ø.; Ludwig, D. Likelihood of ancestor states in adaptive radiation. Evolution 1997, 51, 1699–1711. [Google Scholar] [CrossRef]
- Hansen, T.F. Stabilizing selection and the comparative analysis of adaptation. Evolution 1997, 51, 1341–1351. [Google Scholar] [CrossRef] [PubMed]
- Butler, M.A.; King, A. Phylogenetic Comparative Analysis: A Modeling Approach for Adaptive Evolution. Am. Nat. 2004, 164, 683–695. [Google Scholar] [CrossRef] [PubMed]
- Revell, L.J. phytools: An R package for phylogenetic comparative biology (and other things). Methods Ecol. Evol. 2011, 3, 217–223. [Google Scholar] [CrossRef]
- Beaulieu, J.M.; Jhwueng, D.-C.; Boettiger, C.; O’Meara, B.C. Modeling stabilizing selection: Expanding the Ornstein-Uhlenbeck model of adaptive evolution. Evolution 2012, 66, 2369–2383. [Google Scholar] [CrossRef] [PubMed]
| Species | No. of Samples | Con. (% w/w) | Sucrose (%) | Glucose (%) | Fructose (%) | Sugar Amount (µg/Flower) |
|---|---|---|---|---|---|---|
| Acanthonema strigosum Hook.f. | 5 | 8.97 | 93.0 ± 5.7 | 3.7 ± 2.6 | 3.3 ± 3.2 | 150.5 ± 76.5 |
| Acanthopale decempedalis C.B.Clarke | 15 | 6.93 | 73.1 ± 9.6 | 9.7 ± 4 | 17.2 ± 5.7 | 386.1 ± 220.6 |
| Aframomum sp. “purple” | 38 | 17.71 | 34.0 ± 25.3 | 35.0 ± 11.6 | 31.1 ± 15.6 | 763 ± 863 |
| Argocoffeopsis afzelii (Hiern) Robbr. | 4 | 35.5 ± 8.9 | 35.1 ± 1.1 | 29.4 ± 9.7 | 120.8 ± 90.7 | |
| Baccharoides calvoana (Hook.f.) “Isawumi, El-Ghazaly & B.Nord.” | 14 | 44.44 | 39.9 ± 6.4 | 27.9 ± 3.7 | 32.2 ± 3.7 | 189.9 ± 110.6 |
| Bertiera racemosa (G.Don) K.Schum. | 3 | 18.43 | 24.1 ± 4.7 | 45.1 ± 4.7 | 30.7 ± 7.8 | 628.3 ± 476.5 |
| Brillantaisia owariensis P.Beauv. | 24 | 30.2 | 56.9 ± 10.3 | 19.7 ± 8 | 23.4 ± 5.7 | 1295.3 ± 1024.4 |
| Calochone acuminata Keay | 4 | 20.75 | 21 ± 6.8 | 13.2 ± 0.2 | 65.7 ± 6.9 | 8742.7 ± 3191.5 |
| Chlorophytum comosum (Thunb.) Jacques | 6 | 0.2 ± 0.2 | 49.5 ± 7.2 | 50.3 ± 7.2 | 27.2 ± 19.7 | |
| Clematis simensis Fresen. | 3 | 78.1 ± 14.8 | 14.5 ± 10.6 | 7.3 ± 4.2 | 9.4 ± 1.6 | |
| Clerodendrum silvanum Henriq. | 17 | 11.69 | 65.3 ± 15.5 | 20.4 ± 8.3 | 14.2 ± 8.2 | 279.2 ± 210.2 |
| Costus dubius (Afzel.) K.Schum. | 7 | 18.6 | 77.2 ± 20 | 7.6 ± 9.2 | 15.3 ± 13.7 | 2755.6 ± 3435 |
| Crassocephalum montuosum (S.Moore) Milne-Redh. | 6 | 36.5 ± 26.6 | 27.5 ± 16.7 | 36 ± 15.1 | 13.5 ± 7.4 | |
| Cuviera longiflora Hiern | 3 | 51 ± 14.3 | 19.5 ± 5.7 | 29.4 ± 8.6 | 1627.3 ± 616.9 | |
| Deinbollia sp. 1 | 15 | 66.1 ± 21.7 | 16.4 ± 12.1 | 17.5 ± 10.3 | 164.5 ± 119.1 | |
| Dicranolepis vestita Engl. | 6 | 20.25 | 94.3 ± 2.9 | 2.8 ± 1.5 | 2.9 ± 1.5 | 422.2 ± 283.8 |
| Dioscoreophyllum cumminsii (Stapf) Diels | 4 | 1.7 ± 3.3 | 33.3 ± 17.7 | 65 ± 14.7 | 4.3 ± 2.4 | |
| Dischistocalyx strobilinus C.B.Clarke | 31 | 17.03 | 50.1 ± 19.7 | 30 ± 10.5 | 19.9 ± 13 | 617.4 ± 595.6 |
| Discoclaoxylon hexandrum (Müll.Arg.) Pax & K.Hoffm. | 8 | 88.2 ± 8 | 6.5 ± 4.8 | 5.4 ± 3.4 | 7.4 ± 5.3 | |
| Discopodium penninervium Hochst. | 5 | 23.5 ± 8.9 | 37.3 ± 4.2 | 39.3 ± 5.1 | 238.7 ± 162.2 | |
| Distephanus biafrae (Oliv. & Hiern) H.Rob. | 5 | 20.7 ± 12.3 | 42.7 ± 7.3 | 36.6 ± 6.7 | 154.7 ± 94.2 | |
| Gomphia flava Schumach. & Thonn. | 5 | 2.6 ± 2.1 | 54.4 ± 2.6 | 43 ± 3.3 | 7.7 ± 4.9 | |
| Heckeldora staudtii (Harms) Staner | 5 | 67.1 ± 11.8 | 11.9 ± 3.1 | 21 ± 8.9 | 74.7 ± 19.2 | |
| Heinsia crinita (Afzel.) G.Taylor | 3 | 23.5 | 46.8 ± 28.6 | 17.3 ± 14.8 | 35.9 ± 28.9 | 352.7 ± 143.3 |
| Hypoestes triflora (Forssk.) Roem. & Schult. | 20 | 17.88 | 32.6 ± 11.5 | 41.4 ± 9.1 | 26 ± 5.8 | 229.6 ± 164 |
| Ilex mitis (L.) Radlk. | 7 | 37.9 | 48.8 ± 10.7 | 22.6 ± 5 | 28.6 ± 6.3 | 78.2 ± 92.9 |
| Impatiens burtonii Hook.f. | 18 | 30.82 | 78.3 ± 19.4 | 14.1 ± 12.7 | 7.6 ± 7.2 | 423.3 ± 358.1 |
| Impatiens frithii Cheek | 11 | 16.12 | 85 ± 10.4 | 7.2 ± 5 | 7.8 ± 5.6 | 957.3 ± 645 |
| Impatiens hians Hook.f. | 20 | 19.75 | 95.3 ± 5.6 | 1.7 ± 1.9 | 2.9 ± 4.1 | 2107.7 ± 1758.9 |
| Impatiens macroptera Hook.f. | 25 | 26.01 | 88.2 ± 21 | 6.9 ± 13 | 4.9 ± 8.2 | 786.5 ± 691.9 |
| Impatiens mannii Hook.f. | 13 | 64 ± 35.5 | 21 ± 20.4 | 15.1 ± 15.1 | 59.1 ± 66.3 | |
| Impatiens niamniamensis Gilg | 46 | 15.95 | 82 ± 16.8 | 8.8 ± 8.5 | 9.2 ± 8.9 | 1819.7 ± 1530.4 |
| Impatiens sakeriana Hook. f. | 7 | 19.7 | 81.4 ± 14.9 | 9 ± 7.3 | 9.6 ± 7.8 | 2345.4 ± 1583.2 |
| Isodon ramosissimus (Hook.f.) Codd | 14 | 15.62 | 56.8 ± 10.6 | 26.3 ± 6.9 | 16.9 ± 4 | 100.5 ± 30.6 |
| Isoglossa glandulifera Lindau | 10 | 6.5 | 77.1 ± 9.1 | 9.2 ± 5 | 13.7 ± 6.3 | 43.4 ± 25.3 |
| Ixora foliosa Hiern | 9 | 13.88 | 94.6 ± 1.9 | 2.4 ± 1 | 3 ± 1.1 | 98 ± 34.2 |
| Ixora guineensis Benth. | 18 | 12.09 | 76.8 ± 17.7 | 9.7 ± 8.4 | 13.5 ± 11.1 | 59 ± 58.2 |
| Laccodiscus ferrugineus (Baker) Radlk. | 17 | 46.8 ± 36.4 | 23.5 ± 18.9 | 29.7 ± 21.3 | 137 ± 123.2 | |
| Melanthera scandens (Schumach. & Thonn.) Roberty | 13 | 44 ± 23.2 | 30.8 ± 16.9 | 25.2 ± 11 | 13.1 ± 14.1 | |
| Mikania cordata (Burm.f.) B.L.Rob. | 15 | 41.8 ± 20.2 | 15.9 ± 8.2 | 42.4 ± 14 | 5.6 ± 4.1 | |
| Nuxia congesta R.Br. ex Fresen. | 5 | 23.06 | 50.4 ± 4.5 | 24.4 ± 3 | 25.2 ± 1.9 | 386.3 ± 117.7 |
| Oncoba dentata Oliv. | 3 | 2.4 ± 2.1 | 44.5 ± 8.9 | 53 ± 9.1 | 7.6 ± 3.4 | |
| Pavetta hookeriana Hiern | 9 | 31.58 | 33.9 ± 13 | 31 ± 6.3 | 35.1 ± 7.3 | 244.4 ± 218.1 |
| Pavetta neurocarpa Benth. | 15 | 23.68 | 58 ± 12.4 | 23.5 ± 7.5 | 18.4 ± 5.9 | 185.1 ± 103.9 |
| Pavetta rigida Hiern | 19 | 12.36 | 45.2 ± 11.3 | 26.8 ± 5.6 | 28 ± 10 | 562.8 ± 549.5 |
| Plectranthus decurrens (Gürke) J.K.Morton | 20 | 19.21 | 74.6 ± 8.7 | 20.8 ± 6 | 4.7 ± 3.4 | 121 ± 71.3 |
| Plectranthus glandulosus Hook.f. | 15 | 36.05 | 67.4 ± 12.3 | 18.2 ± 5.3 | 14.4 ± 7.3 | 320.9 ± 160.7 |
| Plectranthus kamerunensis Gürke | 18 | 29.24 | 72.3 ± 8.8 | 23.3 ± 7.2 | 4.4 ± 2.4 | 478.5 ± 353.3 |
| Psychotria bifaria Hiern | 10 | 21.9 ± 32.2 | 38.8 ± 15.2 | 39.3 ± 17.6 | 58.3 ± 57.5 | |
| Psychotria leptophylla Hiern | 11 | 31.6 ± 35.3 | 34.6 ± 17.7 | 33.8 ± 17.7 | 101.6 ± 40.5 | |
| Psychotria peduncularis (Salisb.) Steyerm. | 16 | 14.13 | 66.7 ± 17 | 16 ± 6.8 | 17.3 ± 10.7 | 332 ± 324.5 |
| Psychotria thonneri (De Wild. & T. Durand) O. Lachenaud | 7 | 9.9 | 80.8 ± 16.8 | 11.4 ± 11.4 | 7.8 ± 5.7 | 182.4 ± 129.7 |
| Psydrax dunlapii (Hutch. & Dalziel) Bridson | 3 | 11.5 | 68.4 ± 2.9 | 14.5 ± 2 | 17.2 ± 1 | 102.1 ± 32.6 |
| Sabicea calycina Benth. | 15 | 24.35 | 77.2 ± 22.8 | 13 ± 8.7 | 9.9 ± 17.1 | 346.8 ± 234.7 |
| Sabicea pilosa Hiern | 16 | 18.34 | 68 ± 17.8 | 17.1 ± 9.3 | 14.9 ± 9.5 | 1817.5 ± 1771.8 |
| Schefflera abyssinica (Hochst. ex A.Rich.) Harms | 5 | 11.12 | 1 ± 0.9 | 47.4 ± 0.9 | 51.6 ± 1.2 | 127.9 ± 145.8 |
| Solanecio mannii (Hook.f.) C.Jeffrey | 5 | 0.8 ± 1 | 53.3 ± 3.5 | 46 ± 4.2 | 8.6 ± 4.6 | |
| Spermacoce princeae (K.Schum.) Verdc. | 5 | 11 | 79.8 ± 14.6 | 12.5 ± 7.8 | 7.7 ± 7.7 | 98.6 ± 38.4 |
| Stachys aculeolata Hook.f. | 13 | 61 ± 16.5 | 22.6 ± 9.2 | 16.4 ± 7.7 | 42.4 ± 28.9 | |
| Stellaria mannii Hook.f. | 14 | 35.3 ± 15.2 | 42.7 ± 9.1 | 22 ± 7.8 | 37 ± 27 | |
| Tabernaemontana brachyantha Stapf | 6 | 11.6 | 10.6 ± 18.1 | 32 ± 7.4 | 57.4 ± 12.9 | 1894.5 ± 1344.6 |
| Tabernaemontana ventricosa Hochst. ex A.DC. | 18 | 16.84 | 46.9 ± 29.5 | 22.4 ± 12.2 | 30.7 ± 20.6 | 980.6 ± 873.7 |
| Thunbergia fasciculata Lindau | 7 | 13.68 | 80.4 ± 9.4 | 8.9 ± 3.8 | 10.7 ± 7.5 | 1271.1 ± 2178.7 |
| Trichilia rubescens Oliv. | 22 | 22.02 | 32 ± 23 | 17.9 ± 13.9 | 50.1 ± 19.8 | 312.9 ± 305.8 |
| Voacanga africana Stapf ex Scott-Elliot | 15 | 14.7 | 95 ± 4.1 | 2.3 ± 1.9 | 2.7 ± 2.4 | 1239.4 ± 575.8 |
| Voacanga bracteata Stapf | 12 | 16.69 | 80.4 ± 21.5 | 10.6 ± 13.6 | 8.9 ± 8.3 | 2354 ± 2817.9 |
| Abouheif’s Cmean | Moran’s I | Blomberg’s K | Pagel’s λ | |
|---|---|---|---|---|
| Sucrose (%) | 0.132 * | 0.057 * | 0.250 ** | 0.596 ** |
| Glucose (%) | 0.082 | 0.034 † | 0.256 ** | 0.490 † |
| Fructose (%) | 0.133 * | 0.061 * | 0.195 * | 0.442 * |
| Sucrose amount | 0.185 * | 0.056 * | 0.080 | 0.329 * |
| Glucose amount | 0.091 † | 0.020 * | 0.055 | 0.209 |
| Fructose amount | −0.001 | −0.015 | 0.071 | 0.000 |
| Sucrose/hexose ratio | 0.038 | 0.009 | 0.097 | 0.000 |
| Sugar amount | 0.046 | −0.004 | 0.069 | 0.000 |
| Sucrose Pro. | Glucose Pro. | Fructose Pro. | Suc/Hex Ratio | Total Amount | Sucrose Amount | Glucose Amount | Fructose Amount | |
|---|---|---|---|---|---|---|---|---|
| BM1 | 57.51 | 8.06 | 35.84 | 462.94 | 311.64 | 200.03 | 69.37 | 238.24 |
| BMS | 47.04 | −21.31 | 12.35 | 442.54 | 301.15 | 180.20 | 60.74 | 211.51 |
| OU1 | 15.79 | −70.48 | −40.70 | 389.88 | 218.48 | 116.47 | −40.60 | 145.58 |
| OU-PG(5) | 16.76 | −64.36 | −46.73 | 398.28 | 215.98 | 117.30 | −39.36 | 152.53 |
| OU-PG(2) | 10.04 | −75.11 | −55.33 | 388.16 | 214.37 | 77.60 | −39.23 | 147.82 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Janeček, Š.; Chmel, K.; Ewome, F.L.; Hrubá, K.; Klomberg, Y.; Kobe, I.N.; Kouede, R.D.; Mertens, J.E.J.; Njie, M.M.; Tropek, R. Differences in Nectar Traits between Ornithophilous and Entomophilous Plants on Mount Cameroon. Plants 2021, 10, 1161. https://doi.org/10.3390/plants10061161
Janeček Š, Chmel K, Ewome FL, Hrubá K, Klomberg Y, Kobe IN, Kouede RD, Mertens JEJ, Njie MM, Tropek R. Differences in Nectar Traits between Ornithophilous and Entomophilous Plants on Mount Cameroon. Plants. 2021; 10(6):1161. https://doi.org/10.3390/plants10061161
Chicago/Turabian StyleJaneček, Štěpán, Kryštof Chmel, Francis Luma Ewome, Karolína Hrubá, Yannick Klomberg, Ishmeal N. Kobe, Raissa Dywou Kouede, Jan E. J. Mertens, Marcus Mokake Njie, and Robert Tropek. 2021. "Differences in Nectar Traits between Ornithophilous and Entomophilous Plants on Mount Cameroon" Plants 10, no. 6: 1161. https://doi.org/10.3390/plants10061161
APA StyleJaneček, Š., Chmel, K., Ewome, F. L., Hrubá, K., Klomberg, Y., Kobe, I. N., Kouede, R. D., Mertens, J. E. J., Njie, M. M., & Tropek, R. (2021). Differences in Nectar Traits between Ornithophilous and Entomophilous Plants on Mount Cameroon. Plants, 10(6), 1161. https://doi.org/10.3390/plants10061161






