Piper nigrum CYP719A37 Catalyzes the Decisive Methylenedioxy Bridge Formation in Piperine Biosynthesis
Abstract
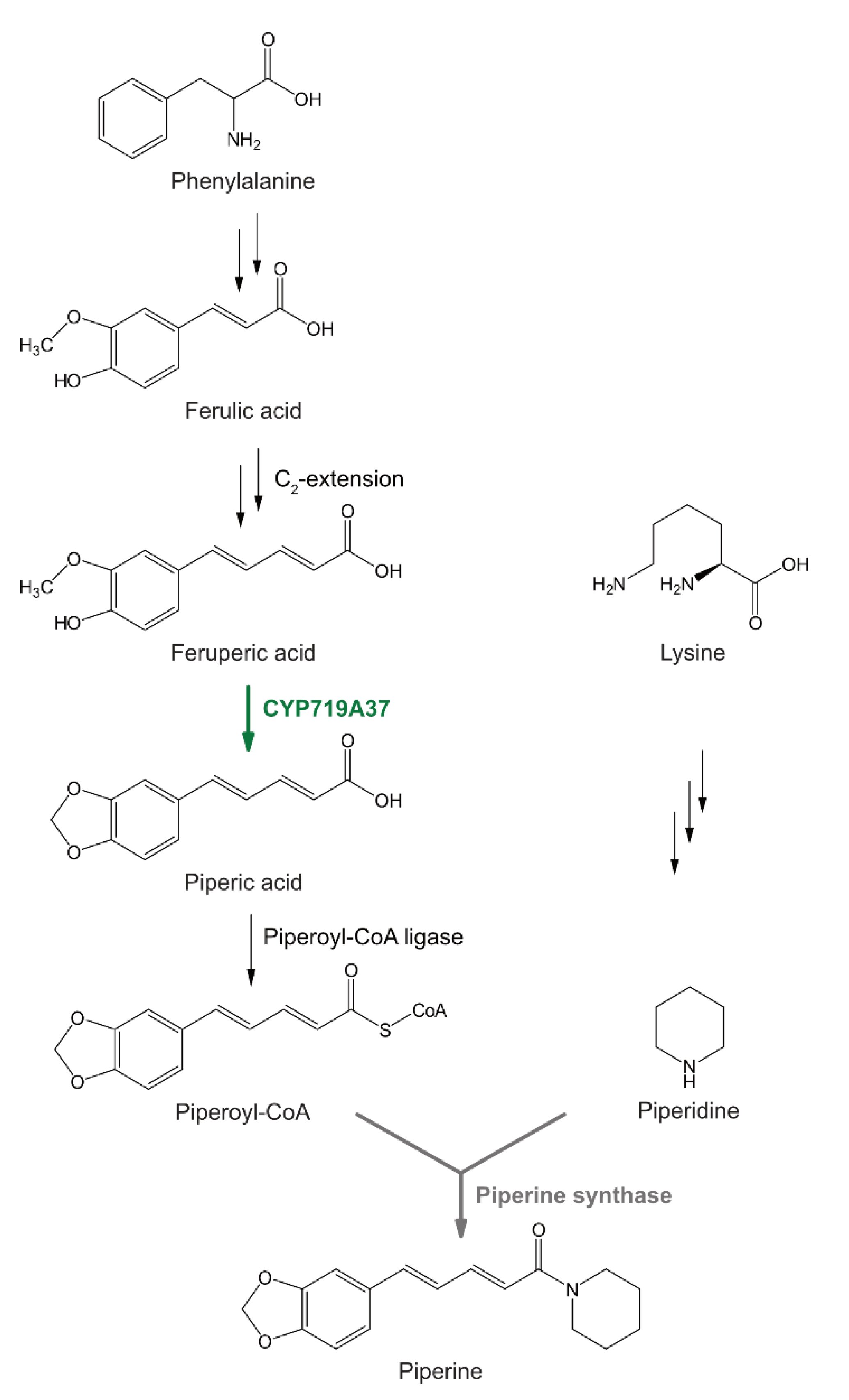
1. Introduction
2. Results
2.1. Identification and Cloning of PnCYP719 and PnCPR
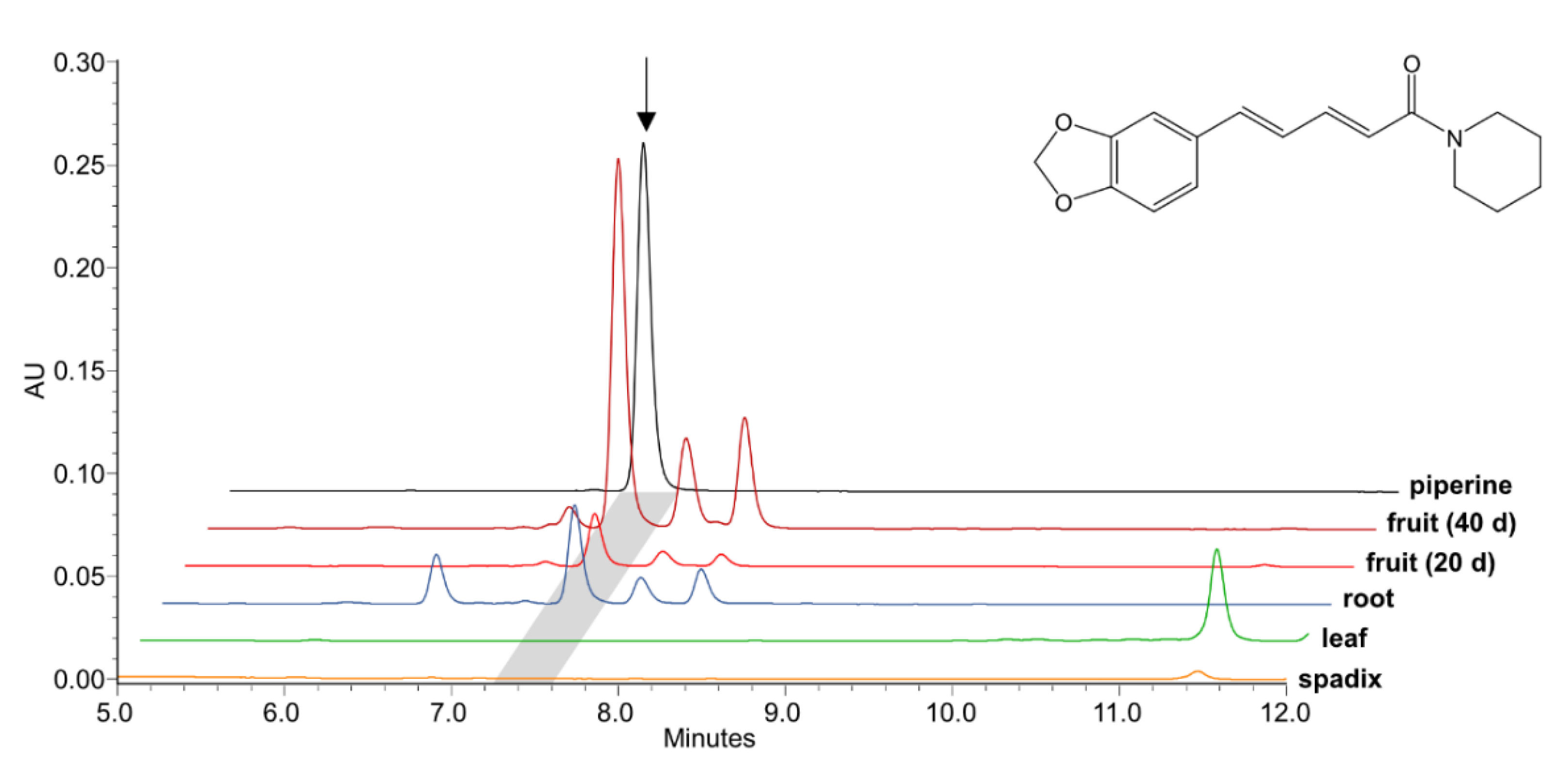
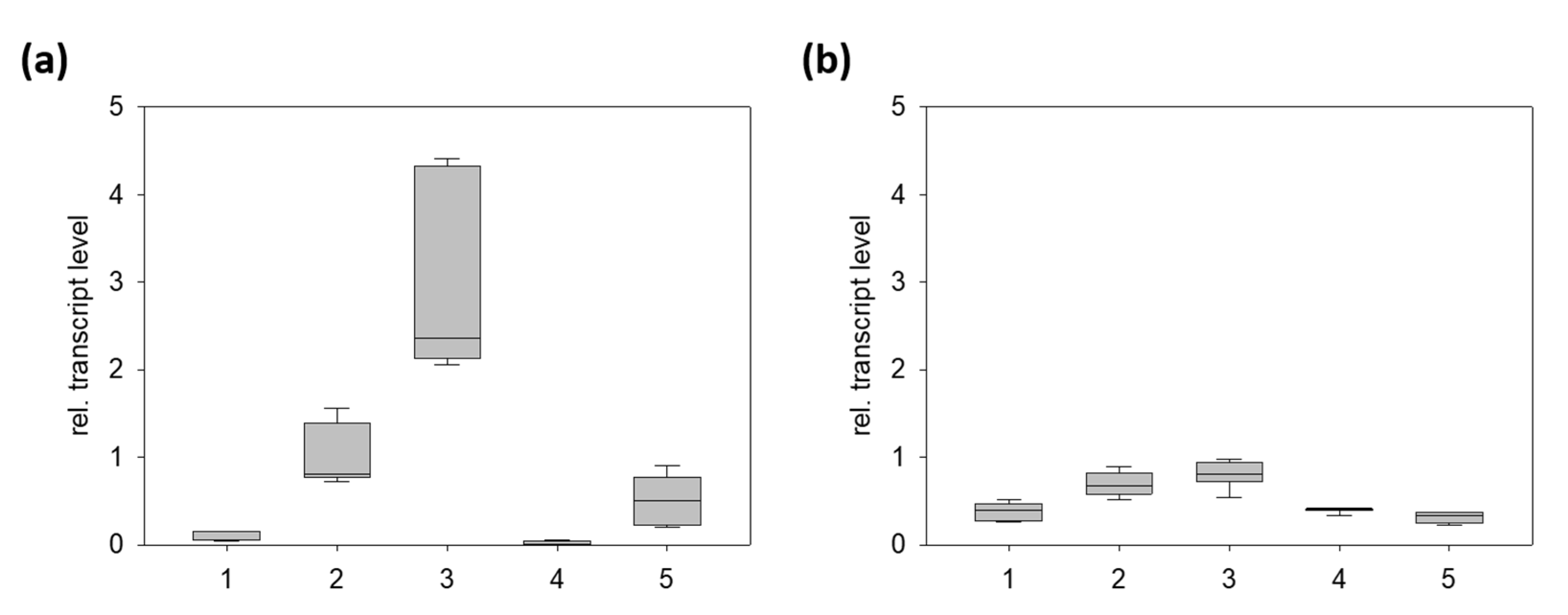
2.2. PnCYP719 Is Differentially Expressed in Black Pepper Organs
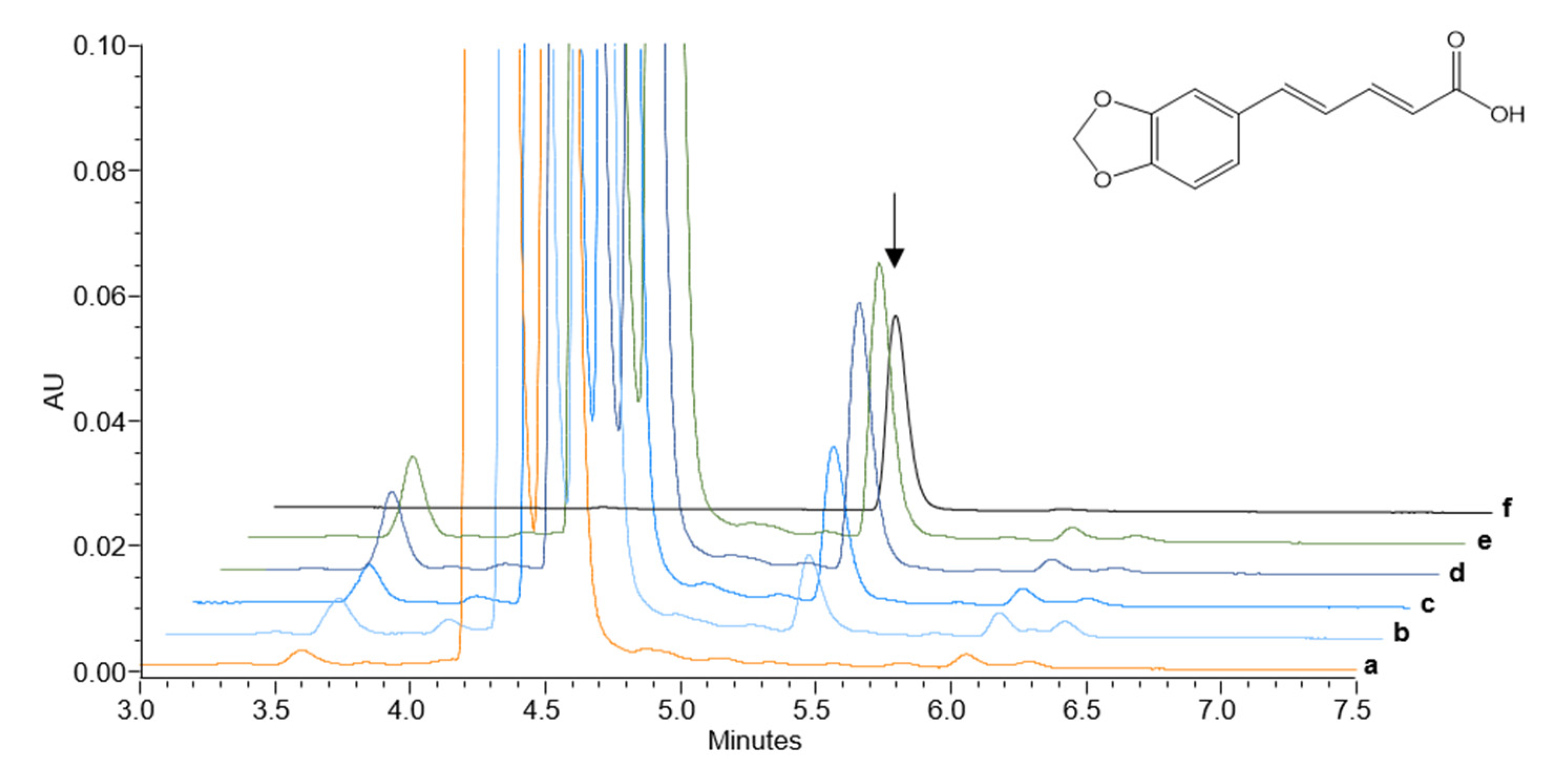
2.3. Heterologously Expressed PnCYP719 Is Substrate Specific
2.4. Phylogeny and Structural Aspects of PnCYP719 and PnCPR
3. Discussion
4. Materials and Methods
4.1. Plant Material, RNA Extraction and cDNA Synthesis
4.2. Chemical Synthesis of Feruperic Acid and Feruperine
4.3. Preparation of Black Pepper Methanolic Extracts
4.4. Cloning of PnCYP719A and PnCPR Genes
4.5. Transient Gene Expression in S. Cerevisiae INVSc1
4.6. Characterization of Enzyme Activities in Transient Yeast Cell Assays
4.7. In Vitro CYP Assay with Microsomal Fractions
4.8. RT-qPCR of PnCYPA37 and PnCPR Transcripts
4.9. Data Availability and Deposition
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Appendix A
References
- Schilmiller, A.L.; Pichersky, E.; Last, R.L. Taming the hydra of specialized metabolism: How systems biology and comparative approaches are revolutionizing plant biochemistry. Curr. Opin. Plant Biol. 2012, 15, 338–344. [Google Scholar] [CrossRef]
- Mizutani, M.; Ohta, D. Evolution of P450 genes during land plant evolution. Ann. Rev. Plant Biol. 2010, 61, 291–315. [Google Scholar] [CrossRef] [PubMed]
- Nelson, D.; Werck-Reichhart, D. A P450-centric view of plant evolution. Plant J. 2011, 66, 194–211. [Google Scholar] [CrossRef] [PubMed]
- Nielson, K.A.; Møller, B.L. Cytochrome P450s in plants. In Cytochrome P450, Structure, Mechanism and Biochemistry, 3rd ed.; Ortiz De Montellano, P.R., Ed.; Kluwer Academic/Plenum Publishers: New York, NY, USA, 2005; pp. 553–583. [Google Scholar]
- Mizutani, M.; Sato, F. Unusual P450 reactions in plant secondary metabolism. Arch. Biochem. Biophys. 2011, 507, 194–203. [Google Scholar] [CrossRef] [PubMed]
- Ikezawa, N.; Iwasa, K.; Sato, F. Molecular cloning and characterization of methylene bridge-forming enzymes involved in stylopine biosynthesis in Eschscholzia californica. FEBS J. 2007, 274, 1019–1035. [Google Scholar] [CrossRef] [PubMed]
- Chávez, D.M.L.; Rolf, M.; Gesell, A.; Kutchan, T.M. Characterization of two methylenedioxy bridge-forming cytochrome P450-dependent enzymes of alkaloid formation in the Mexican prickly poppy Argemone mexicana. Arch. Biochem. Biophys. 2011, 507, 186–193. [Google Scholar] [CrossRef] [PubMed]
- Gesell, A.; Rolf, M.; Ziegler, J.; Chávez, D.M.L.; Huang, F.C.; Kutchan, T.M. CYP719B1 is salutaridine synthase, the C-C phenol-coupling enzyme of morphine biosynthesis in opium poppy. J. Biol. Chem. 2009, 284, 24432–24442. [Google Scholar] [CrossRef]
- Nelson, D.R.; Schuler, M. Cytochrome P450 genes from the sacred lotus genome. Tropic. Plant Biol. 2013, 6, 138–151. [Google Scholar] [CrossRef]
- Simmons, N.W.; Stevens, R. Occurrence of the methylenedioxy bridge in the phenolic components of plants. Nature 1956, 178, 752–753. [Google Scholar] [CrossRef]
- Ikezawa, N.; Tanaka, M.; Nagayoshi, M.; Shinkyo, R.; Sasaki, T.; Inouye, K.; Sato, F. Molecular cloning and characterization of CYP719, a methylenedioxy bridge-forming enzyme that belongs to a novel P450 family, from cultured Coptis japonica cells. J. Biol. Chem. 2003, 278, 38557–38565. [Google Scholar] [CrossRef]
- Yahyazadeh, M.; Ratmoyo, P.; Bittner, F.; Sato, F.; Selmar, D. Cloning and characterization of cheilanthifoline and stylopine synthase genes from Chelidonium majus. Plant Cell Physiol. 2017, 58, 1421–1430. [Google Scholar] [CrossRef] [PubMed]
- Bauer, W.; Zenk, M.H. Two methylenedioxy bridge forming cytochrome P450-dependent enzymes are involved in (S)-stylopine formation. Phytochemistry 1991, 30, 2953–2961. [Google Scholar] [CrossRef]
- Beaudoin, B.A.W.; Facchini, P.J. Benzylisoquinoline alkaloid biosynthesis in opium poppy. Planta 2014, 40, 19–32. [Google Scholar] [CrossRef] [PubMed]
- Dastmalchi, M.; Park, M.R.; Morris, J.S.; Facchini, P.J. Family portraits: The enzymes behind benzylisoquinoline alkaloid biosynthesis. Phytochem. Rev. 2018, 17, 249–277. [Google Scholar] [CrossRef]
- Marques, J.V.; Kim, K.W.; Lee, C.; Costa, M.A.; May, G.-D.; Crow, J.A.; Davin, L.B.; Lewis, N.G. Next generation sequencing in predicting gene function in podophyllotoxin biosynthesis. J. Biol. Chem. 2013, 288, 466–479. [Google Scholar] [CrossRef]
- Ono, E.; Nakai, M.; Fukui, Y.; Tomimori, N.; Fukuchi-Mizutani, M.; Saito, M.; Satake, H.; Tanaka, T.; Katsuta, M.; Umezuwa, T.; et al. Formation of two methylenedioxy bridges by a Sesamum CYP81Q protein yielding a furofuran lignan, (+)-sesamin. Proc. Natl. Acad. Sci. USA 2006, 103, 10116–10121. [Google Scholar] [CrossRef]
- Ono, E.; Murata, H.; Toyonaga, J.; Nakayasu, M.; Mizutani, M.; Yamamoto, M.; Umezawa, T.; Orikawa, M. Formation of a methylenedioxy bridge in (+)-epipinoresinol by CYP81Q3 corroborates with diastereomeric specialization in sesame lignans. Plant Cell Physiol. 2018, 59, 2278–2287. [Google Scholar] [CrossRef]
- Pluskal, T.; Torrens-Spence, M.P.; Fallon, T.R.; De Abreu, A.; Shi, C.H.; Weng, J.K. The biosynthetic origin of psychoactive kavalactones in kava. Nat. Plants 2019, 5, 867–878. [Google Scholar] [CrossRef]
- Meghwal, M.; Goswami, T. Piper nigrum and piperine: An update. Phytother. Res. 2013, 27, 1121–1130. [Google Scholar] [CrossRef]
- Li, H.; Kristin, S.; Wang, S.; Wink, M. Capsaicin and piperine can overcome multidrug resistance to doxorubicine. Molecules 2018, 23, 557. [Google Scholar] [CrossRef]
- McNamara, F.N.; Randall, A.; Gunthorpe, M.J. Effects of piperine, the pungent component of black pepper, at the human vanilloid receptor (TRPV1). Br. J. Pharmacol. 2006, 144, 781–790. [Google Scholar] [CrossRef] [PubMed]
- Correa, E.A.; Högestett, E.D.; Sterner, O.; Echeverri, F.; Zygmundt, P.M. In vitro TRPV-activity of piperine derived amides. Bioorg. Med. Chem. 2010, 18, 3299–3308. [Google Scholar] [CrossRef] [PubMed]
- Gupta, R.N.; Spenser, I.D. Biosynthesis of the piperidine nucleus: The occurrence of two pathways from lysine. Phytochemistry 1970, 9, 2329–2334. [Google Scholar] [CrossRef]
- Leistner, E.; Spenser, I.D. Biosynthesis of the piperidine nucleus. Incorporation of chirally labelled cadaverine-1-3H. J. Am. Chem. Soc. 1973, 95, 4715–4725. [Google Scholar] [CrossRef] [PubMed]
- Geisler, J.G.; Gross, G.G. The biosynthesis of piperine in Piper nigrum. Phytochemistry 1990, 29, 489–492. [Google Scholar] [CrossRef]
- Schnabel, A.; Cotinguiba, F.; Athmer, B.; Yang, C.; Westermann, B.; Schaks, A.; Porzel, A.; Brandt, W.; Schumacher, F.; Vogt, T. A piperic acid CoA ligase produces a putative precursor of piperine, the pungent principle from black pepper fruits. Plant J. 2020, 102, 569–581. [Google Scholar] [CrossRef] [PubMed]
- Jin, Z.; Wungsintaweekul, J.; Kim, S.H.; Kim, J.H.; Shin, Y.; Ro, D.K.; Kim, S.U. 4-coumarate:coenzyme A ligase isoform 3 from Piper nigrum (Pn4CL3) catalyzes the CoA thioester formation of 3,4-methylenedioxycinnamic and piperic acids. Biochem. J. 2020, 477, 61–74. [Google Scholar] [CrossRef]
- Engler, C.; Marillonnet, S. Golden Gate cloning. Methods Mol. Biol. 2000, 1116, 119–131. [Google Scholar]
- Schuler, M.A.; Werck-Reichart, D. Functional genomics of P450s. Ann. Rev. Plant Biol. 2003, 54, 629–667. [Google Scholar] [CrossRef]
- Sauveplane, V.; Kandel, S.; Kastner, P.E.; Ehlting, J.; Compagnon, V.; Werck-Reichhart, D.; Pinot, F. Arabidopsis thaliana CYP77A4 is the first cytochrome P450 able to catalyze the epoxidation of free fatty acids in plants. FEBS J. 2009, 276, 719–735. [Google Scholar] [CrossRef]
- Scheler, U.; Brandt, W.; Porzel, A.; Rothe, K.; Manzano, D.; Božić, D.; Papaefthimiou, D.; Balke, G.U.; Henning, A.; Lohse, S.; et al. Elucidation of the biosynthesis of carnosic acid and its reconstitution in yeast. Nat. Commun. 2016, 7, 12942. [Google Scholar] [CrossRef] [PubMed]
- Nakatani, N.; Inatani, R.; Ohta, H.; Nishioka, A. Chemical constituents of peppers (Piper spec.) and application to food preservation: Naturally occurring antioxidative compounds. Environ. Health Perspect. 1986, 67, 135–142. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Liang, Z.; Hou, J.; Bao, X.; Shen, Y. Identification and functional evaluation of the reductases and dehydrogenases from Saccharomyces cerevisiae involved in vanillin resistance. BMC Biotechnol. 2016, 16, 31. [Google Scholar] [CrossRef] [PubMed]
- Nelson, D.R. The cytochrome P450 homepage. Hum. Genom. 2009, 4, 59–65. [Google Scholar] [CrossRef] [PubMed]
- Ikezawa, N.; Iwasa, K.; Sato, F. Molecular cloning and characterization of CYP80G2 that catalyzes and intramolecular phenol C-C coupling of S-reticuline in magniflorine biosynthesis from cultured Coptis japonica cells. J. Biol. Chem. 2008, 283, 8810–8821. [Google Scholar] [CrossRef] [PubMed]
- Hagel, J.M.; Morris, J.S.; Lee, E.J.; Desgagné-Penix, I.; Bross, C.D.; Chang, L.; Chen, X.; Farrow, S.C.; Zhang, Y.; Soh, Y.; et al. Transcriptome analysis of 20 taxonomically related benzylisoquinoline-producing plants. BMC Plant Biol. 2015, 15, 227. [Google Scholar] [CrossRef]
- Ehlting, J.; Sauveplane, V.; Olry, A.; Gingfinger, J.F.; Provart, N.; Werck-Reichart, D. An extensive (co-)expression analysis tool for the cytochrome P450 superfamily in Arabidopsis thaliana. BMC Plant Biol. 2008, 8, 47. [Google Scholar] [CrossRef]
- Hu, L.; Xu, Z.; Wang, M.; Fan, R.; Yuan, D.; Wu, B.; Jin, S. The chromosome-scale reference genome of black pepper provides insight into piperine biosynthesis. Nat. Comm. 2019, 10, 4702. [Google Scholar] [CrossRef]
- Schnabel, A.; Athmer, B.; Manke, K.; Schumacher, F.; Cotinguiba, F.; Vogt, T. Piperine synthase from black pepper, Piper nigrum. Commun. Biol. 2020. under revision. [Google Scholar]
- Fossati, E.; Ekins, A.; Narcross, L.; Zhu, Y.; Falgueyret, J.P.; Beaudoin, G.A.W.; Facchini, P.J.; Martin, V.J.J. Reconstitution of a 10-gene pathway for synthesis of the plant alkaloid dihydrosanguinarine in Saccharomyces cerevisiae. Nat. Comm. 2014, 5, 3283. [Google Scholar] [CrossRef]
- Scott, I.M.; Jensen, H.R.; Philogene, B.J.R.; Arnason, J.T. A review of Piper spp. (Piperaceae) phytochemistry, insecticidal activity, and mode of action. Phytochem. Rev. 2008, 7, 65–75. [Google Scholar] [CrossRef]
- Kajikawa, M.; Sierro, N.; Kawaguchi, H.; Bakaher, N.; Iwanov, N.V.; Hashimoto, T.; Shoji, T. Genomic insights into the evolution of the nicotine biosynthesis pathway in tobacco. Plant Physiol. 2017, 174, 999–1011. [Google Scholar] [CrossRef] [PubMed]
- Kruse, L.H.; Stegemann, T.; Sievert, C.; Ober, D. Identification of a second site of pyrrolizidine alkaloid biosynthesis in comfrey to boost plant defense in floral stage. Plant Physiol. 2017, 174, 47–55. [Google Scholar] [CrossRef] [PubMed]
- Knudsen, C.; Gallage, N.J.; Hansen, C.C.; Møller, B.L.; Laurson, T. Dynamic metabolic solutions to the sessile lifestyle of plants. Nat. Prod. Rep. 2018, 35, 1140–1155. [Google Scholar] [CrossRef] [PubMed]
- Choi, Y.H.; van Spronsen, J.; Dai, Y.; Verberne, M.; Hollmann, F.; Arends, I.W.C.E.; Geert-Jan Witkamp, G.J.; Verpoorte, R. Are natural deep eutectic solvents the missing link in understanding cellular metabolism and physiology? Plant Physiol. 2011, 156, 1701–1705. [Google Scholar] [CrossRef] [PubMed]
- Knudsen, C.; Bavishi, K.; Viborg, K.M.; Drew, D.P.; Simonsen, H.T.; Motawia, M.S.; Møller, B.L.; Laursen, T. Stabilization of dhurrin biosynthetic enzymes from Sorghum bicolor using a natural deep eutectic solvent. Phytochemistry 2020, 170, 112214. [Google Scholar] [CrossRef]
- Rodziewicz, P.; Loroch, S.; Marczak, L.; Sickman, A.; Kayser, O. Cannabinoid synthases and osmoprotective metabolites accumulate in the exudates of Cannabis sativa L. glandular trichomes. Plant Sci. 2019, 284, 108–116. [Google Scholar] [CrossRef]
- Amborella Genome Project The Amborella genome and the evolution of flowering plants. Science 2013, 342, 1241089. [CrossRef]
- Wu, S.; Wilson, A.E.; Chang, E.; Tian, L. Exploring the phytochemical landscape of the early-diverging flowering plant Amborella trichopoda Baill. Molecules 2019, 24, 3814. [Google Scholar] [CrossRef]
- Yang, Y.; Sun, P.; Lv, L.; Wang, D.; Ru, D.; Li, Y.; Ma, T.; Zhang, L.; Shen, X.; Meng, F.; et al. Prickly water lily genome and rigid hornwort genomes shed light on early angiosperm evolution. Nat. Plants 2020, 6, 215–222. [Google Scholar] [CrossRef]
- Zhang, L.; Chen, F.; Zhang, X.; Li, Z.; Zhao, Y.; Lohaus, R.; Chang, X.; Dong, W.; Ho, S.Y.W.; Liu, X.; et al. The water lily genome and the early evolution of flowering plants. Nature 2020, 577, 79–84. [Google Scholar] [CrossRef]
- Ming, R.; VanBuren, R.; Liu, Y.; Yang, M.; Han, Y.; Li, L.-T.; Zhang, Q.; Kim, M.-J.; Schatz, M.C.; Campbell, M.; et al. Genome of the long-lived sacred Lotus (Nelumbo nucifera Gartn.). Genome Biol. 2013, 14, R41. [Google Scholar] [CrossRef] [PubMed]
- Chaw, S.M.; Liu, Y.C.; Wu, Y.W.; Wang, H.Y.; Lin, C.Y.I.; Wu, C.S.; Ke, H.-M.; Chang, L.-Y.; Hsu, C.-Y.; Yang, H.-T.; et al. The stout camphor genome fills gaps in understanding of flowering plant genome evolution. Nat. Plants 2019, 5, 63–73. [Google Scholar] [CrossRef] [PubMed]
- Liu, X.; Liu, Y.; Huang, P.; Ma, Y.; Qing, Z.; Tang, Q.; Cao, H.; Cheng, P.; Zheng, Y.; Yuan, Z.; et al. The genome of medical plant Macleaya cordata provides new insights into benzylisoquinoline alkaloid metabolism. Mol. Plant 2017, 10, 975–989. [Google Scholar] [CrossRef] [PubMed]
- Lebot, V.; Lèbesque, J. Evidence for conspecificity of Piper methysticum forst. f. and Piper wichmannii C. DC. Biochem. Syst. Ecol. 1996, 24, 775–782. [Google Scholar]
- Negishi, M.; Uno, T.; Honkakoshi, P.; Sueyoshi, T.; Darden, D.A.; Petersen, L.P. The roles of individual amino acids in altering substrate specificity of the P450 2a4/2a5 enzymes. Biochimie 1996, 78, 685–694. [Google Scholar] [CrossRef]
- Schalk, M.; Croteau, R. A single amino acid substitution (F363I) converts the regiochemistry of the spearmint (−)-limonene hydroxylase from a C6- to a C3-hydroxylase. Proc. Natl. Acad. Sci. USA 2000, 97, 11948–11953. [Google Scholar] [CrossRef] [PubMed]
- Kato, M.J.; Furlan, M. Chemistry and Evolution of the Piperaceae. Pure Appl. Chem. 2007, 79, 529–538. [Google Scholar] [CrossRef]
- Wanke, S.; Jaramillo, A.; Borsch, T.; Samain, M.S.; Quandt, D.; Neinhuis, C. Evolution of Piperales—matK and trnK intron sequence data reveal lineage-specific resolution contrast. Mol. Phylogen. Evol. 2007, 42, 477–497. [Google Scholar] [CrossRef] [PubMed]
- DeLuca, V.; Salim, V.; Atsumi, S.M.; Yu, F. Mining the biodiversity of plants: A revolution in the making. Science 2012, 336, 1658–1662. [Google Scholar] [CrossRef]
- Wurzel, E.; Kutchan, T.M. Plant metabolism, the diverse chemistry set for the future. Science 2016, 353, 1232–1236. [Google Scholar] [CrossRef] [PubMed]
- Engler, C.; Kandzia, R.; Marillonnet, S. A one pot, one step, precision cloning method with high throughput capability. PLoS ONE 2008, 8, e3647. [Google Scholar] [CrossRef] [PubMed]
- Werner, S.; Engler, C.; Weber, E.; Gruetzner, R.; Marillonnet, S. Fast track assembly of multigene constructs using Golden Gate cloning and the MoClo system. Bioengineering 2012, 3, 38–43. [Google Scholar] [CrossRef] [PubMed]

| Substrate [m/z] | Product [m/z] | Activity [pkat/mg Crude Protein] |
|---|---|---|
| Feruperic acid [219.1] | Piperic acid [217.1] | 0.92+/−0.05 |
| Feruperine [287.1] | Piperine [285.1] | n.d. 2 |
| Ferulic acid [194.1] | 3,4-methylenedioxycinnamic acid [192.1] | n.d. |
| Coniferylaldehyde [178.1] | 3,4-methylenedioxycinnamylaldehyde 1 [176.1] | n.d |
| Coniferylalcohol [180.1] | 3,4-methylenedioxycinnamylalcohol 1 [178.1] | n.d. |
| Vanillic acid [168.1] | Piperonylic acid [166.1] | n.d. |
| Isovanillic acid [168.1] | Piperonylic acid [166.1] | n.d. |
| Vanillylaldehyde [152.1] | Piperonal 1 [150.1] | n.d. |
| Vanillylalcohol [154.1] | Piperonylalcohol [152.1] | n.d. |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Schnabel, A.; Cotinguiba, F.; Athmer, B.; Vogt, T. Piper nigrum CYP719A37 Catalyzes the Decisive Methylenedioxy Bridge Formation in Piperine Biosynthesis. Plants 2021, 10, 128. https://doi.org/10.3390/plants10010128
Schnabel A, Cotinguiba F, Athmer B, Vogt T. Piper nigrum CYP719A37 Catalyzes the Decisive Methylenedioxy Bridge Formation in Piperine Biosynthesis. Plants. 2021; 10(1):128. https://doi.org/10.3390/plants10010128
Chicago/Turabian StyleSchnabel, Arianne, Fernando Cotinguiba, Benedikt Athmer, and Thomas Vogt. 2021. "Piper nigrum CYP719A37 Catalyzes the Decisive Methylenedioxy Bridge Formation in Piperine Biosynthesis" Plants 10, no. 1: 128. https://doi.org/10.3390/plants10010128
APA StyleSchnabel, A., Cotinguiba, F., Athmer, B., & Vogt, T. (2021). Piper nigrum CYP719A37 Catalyzes the Decisive Methylenedioxy Bridge Formation in Piperine Biosynthesis. Plants, 10(1), 128. https://doi.org/10.3390/plants10010128






