Drosophila as a Model for Developmental Biology: Stem Cell-Fate Decisions in the Developing Nervous System
Abstract
1. Introduction
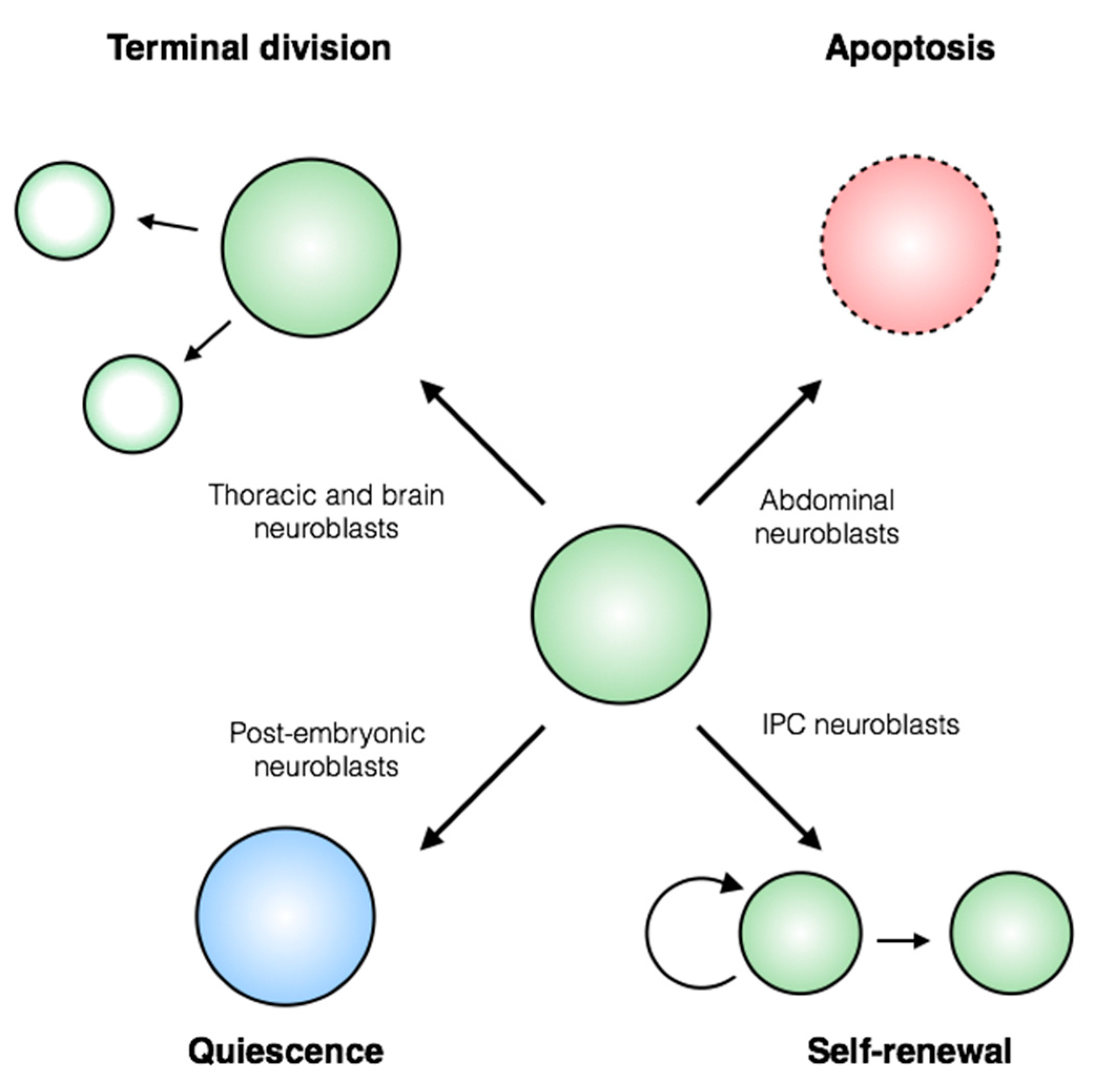
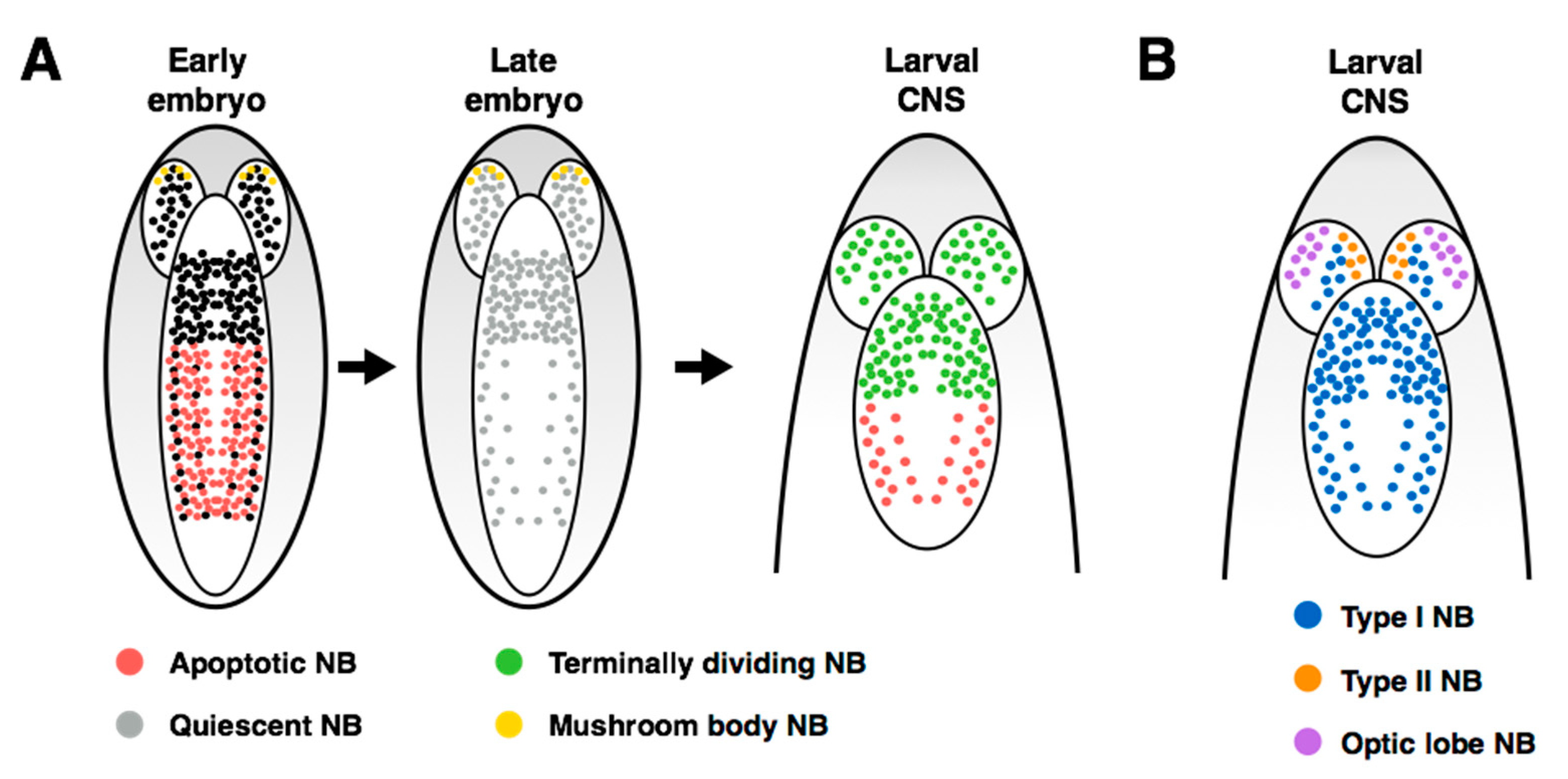
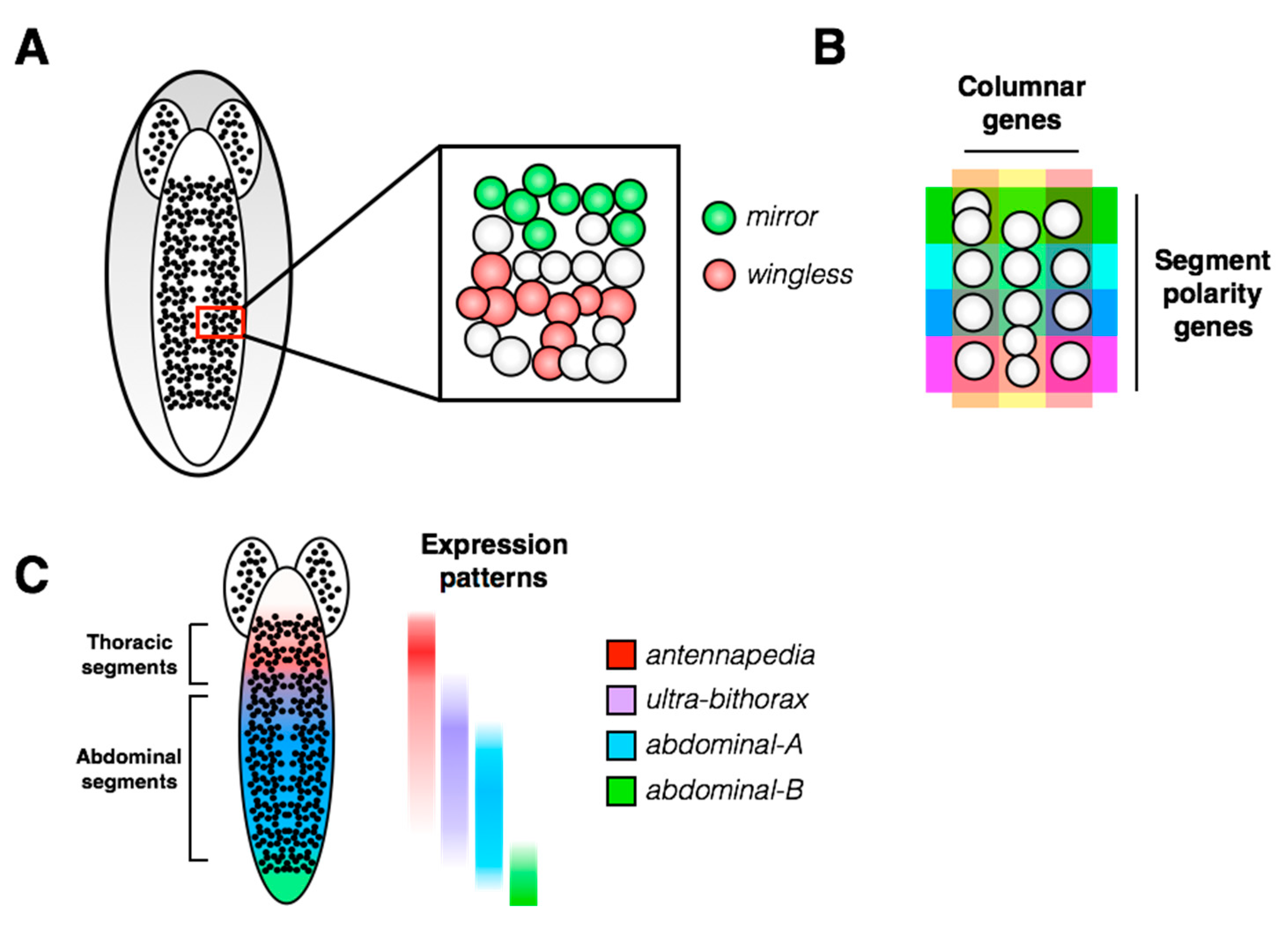
2. Overview of the Fly Nervous System and Its Stem Cell Populations
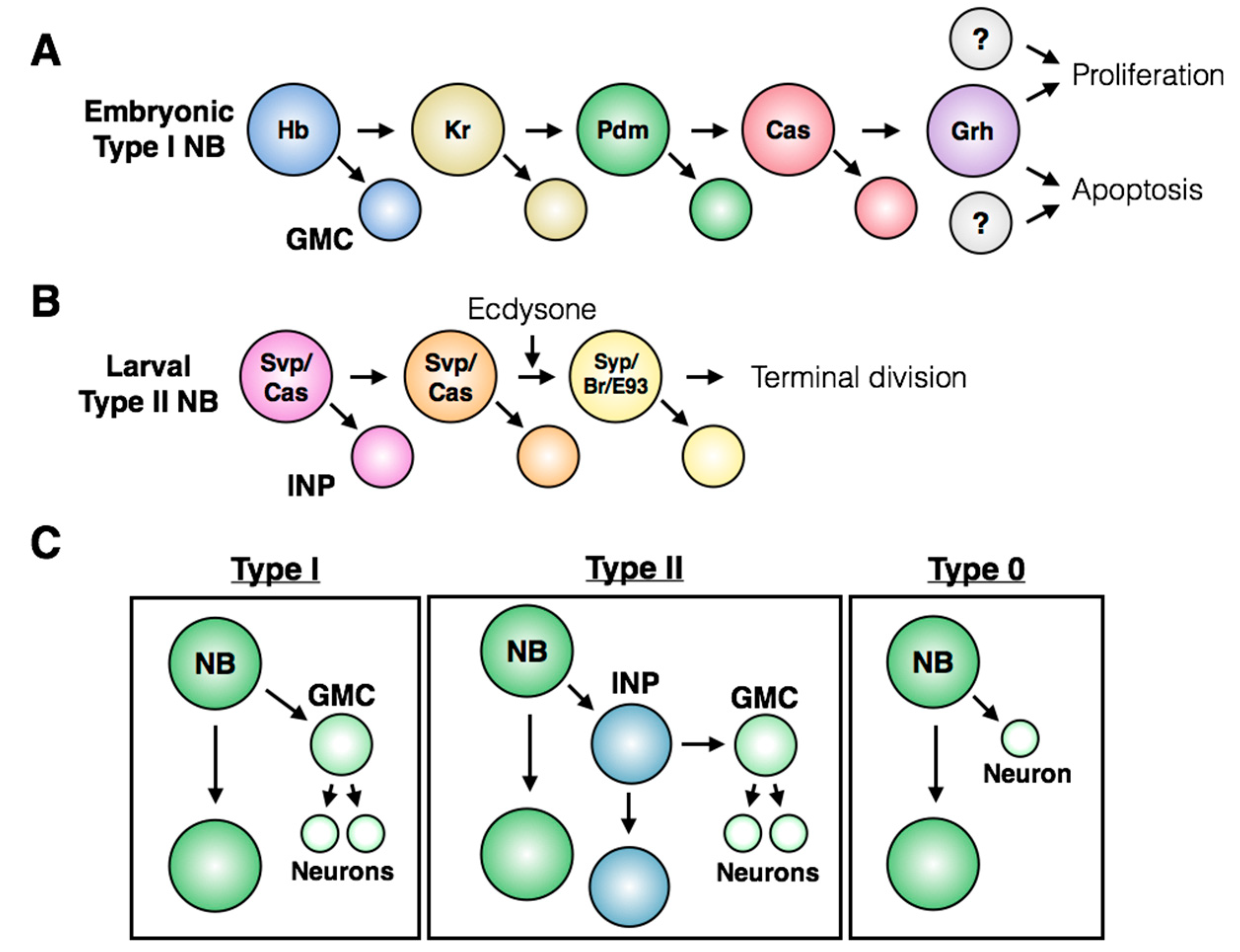
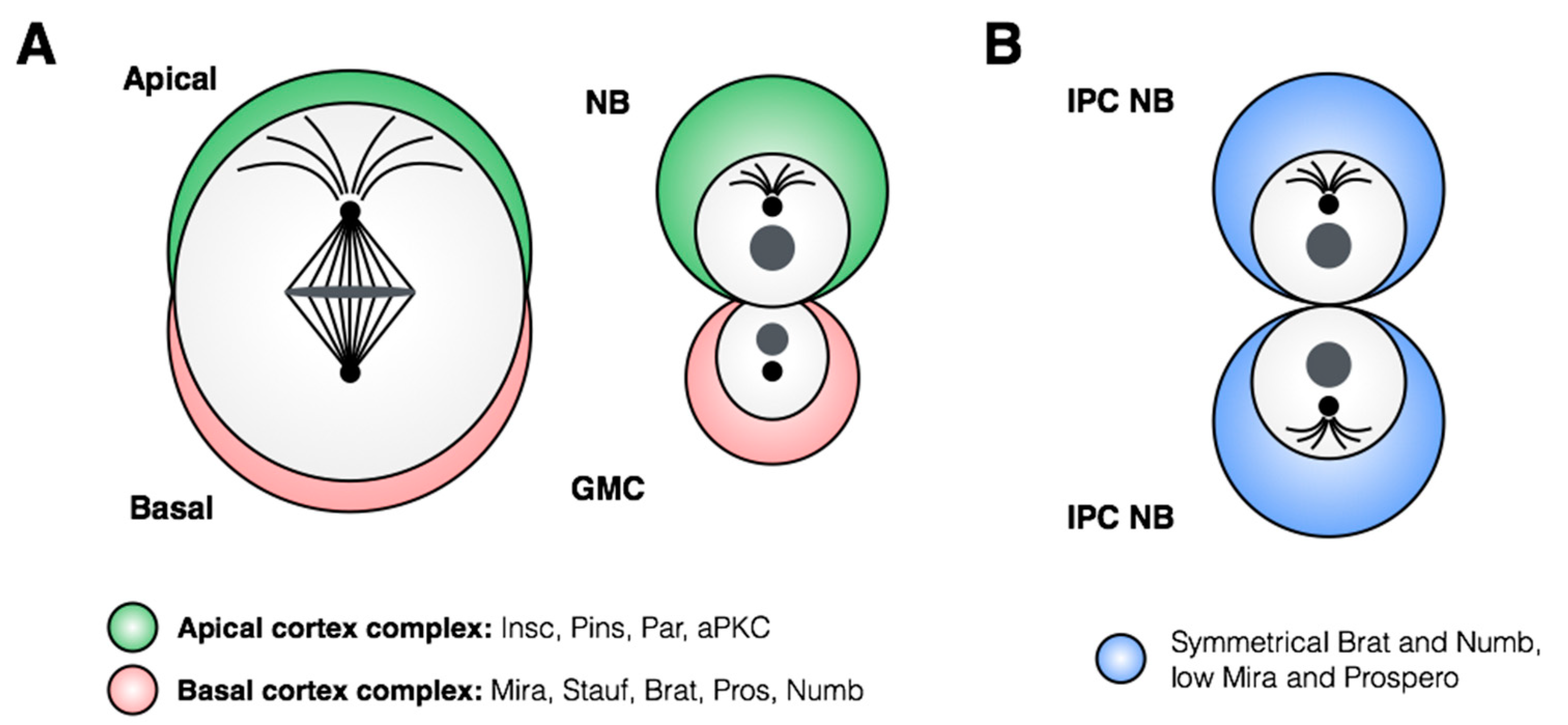
3. Control of Stem Cell Proliferation: Self-Renewal and Amplification
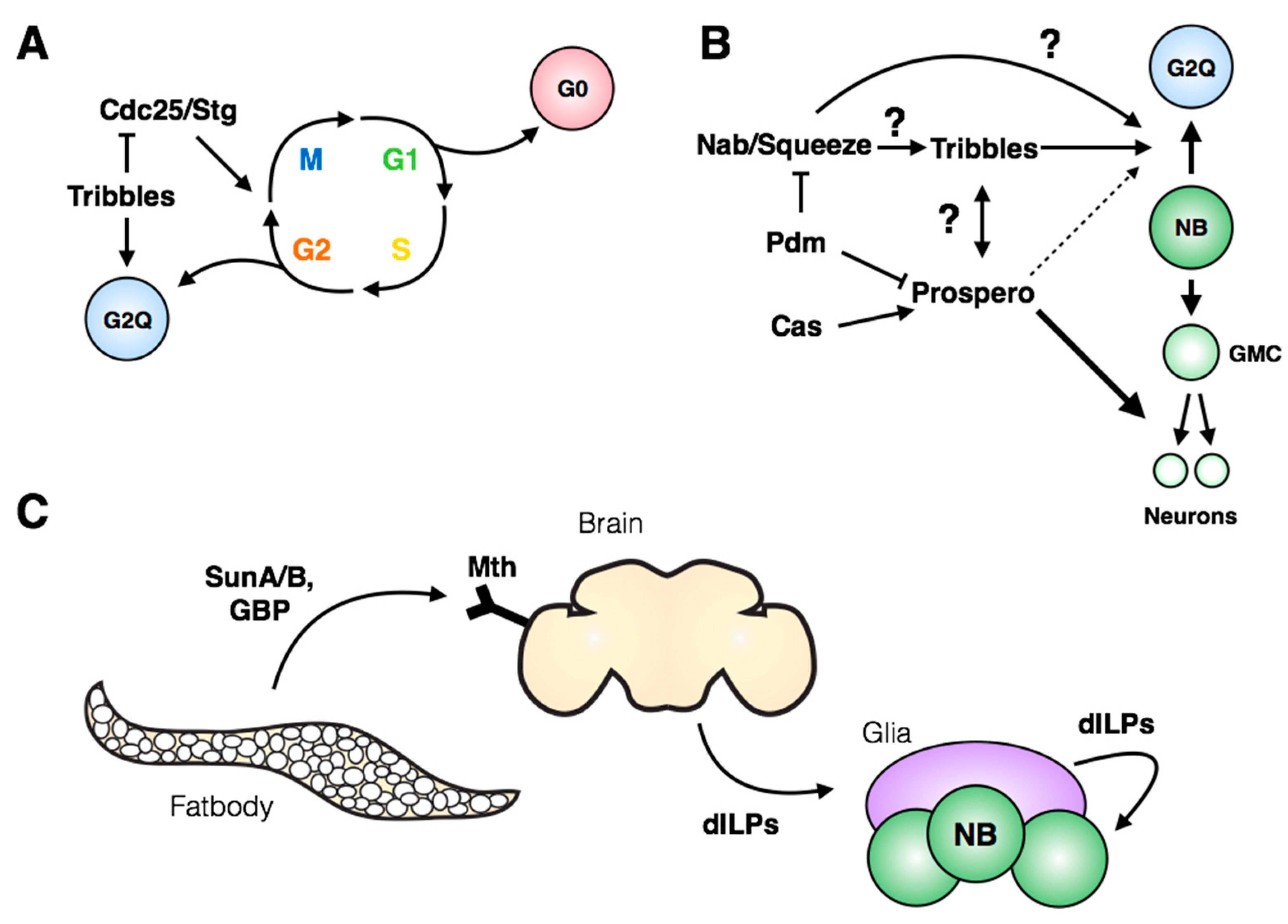
4. Regulation of Stem Cell Quiescence
5. Induction of Neuroblast Cell Cycle Re-Entry
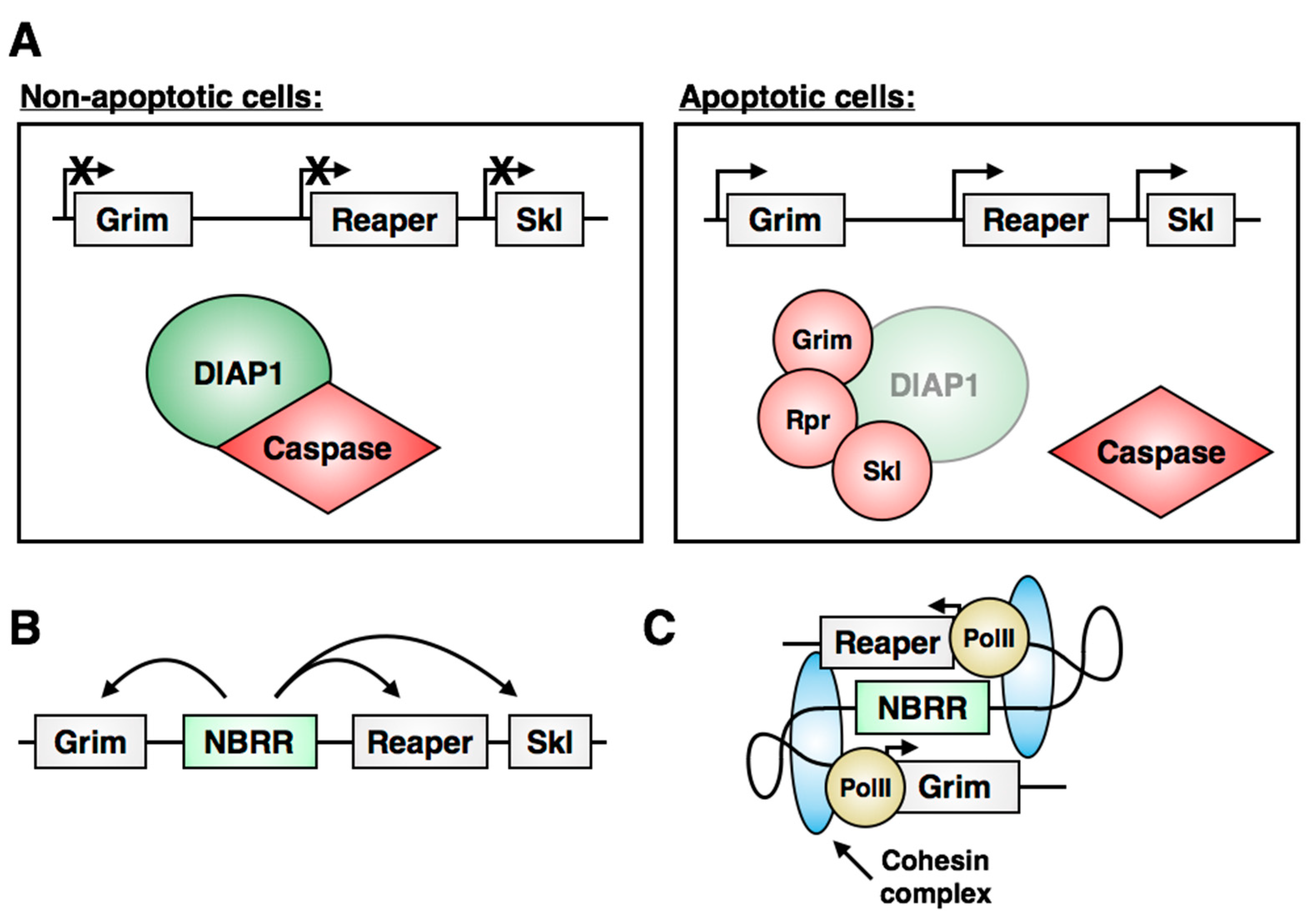
6. Control of Stem Cell Apoptosis
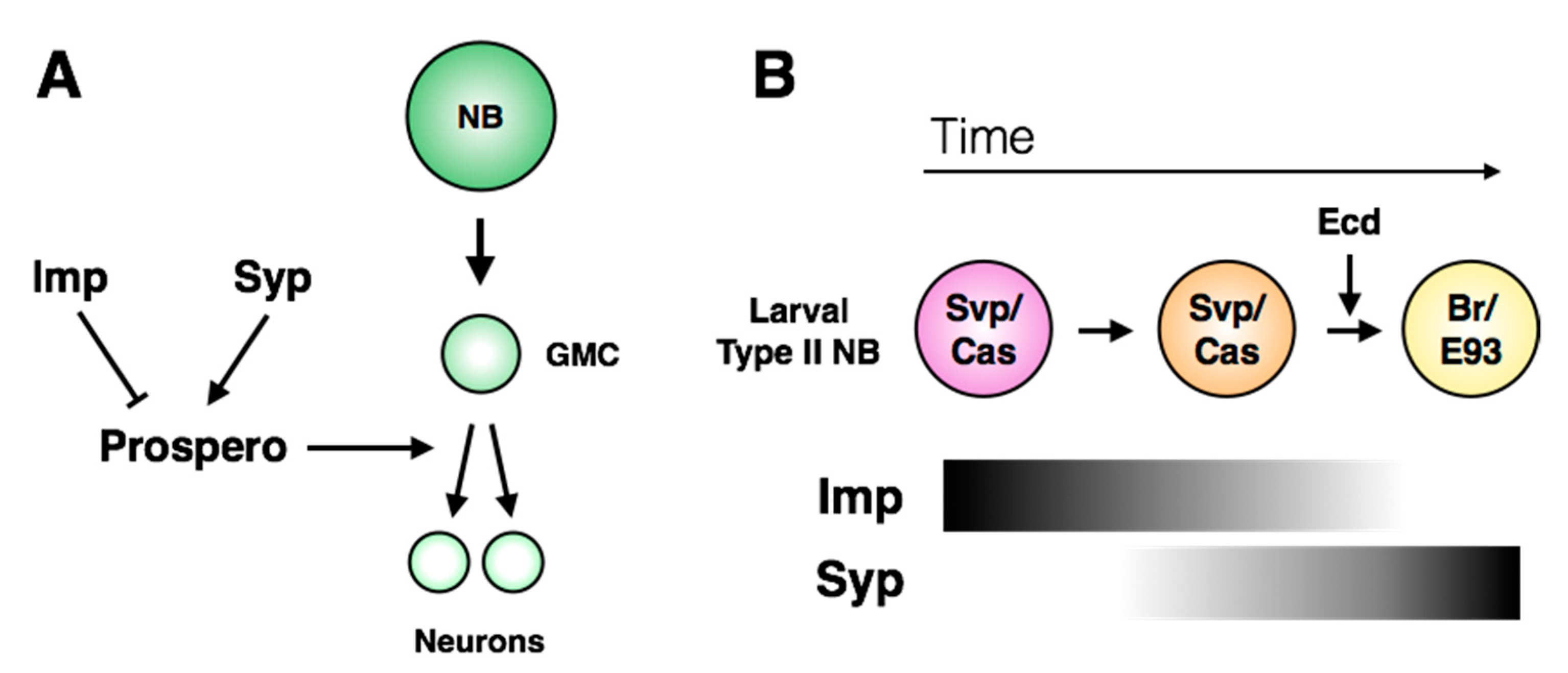
7. Regulation of Terminal Division
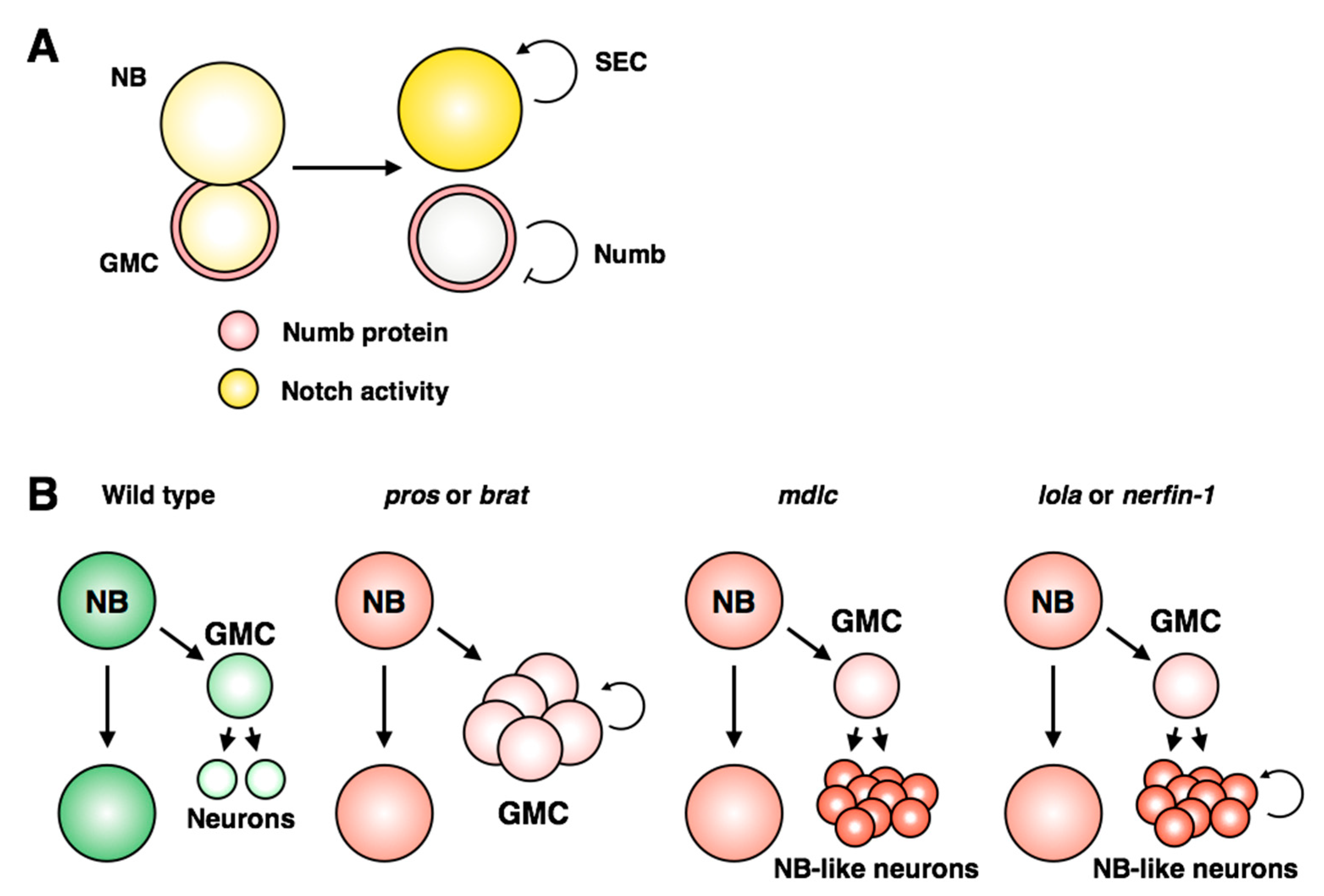
8. Misregulation of Stem Cell and Progeny Identity
9. Concluding Thoughts
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Truman, J.W.; Bate, M. Spatial and Temporal Patterns of Neurogenesis in the Central Nervous System of Drosophila melanogaster. Dev. Biol. 1988, 125, 145–157. [Google Scholar] [CrossRef]
- White, K.; Grether, M.E.; Abrams, J.M.; Young, L.; Farrell, K.; Steller, H. Genetic Control of Programmed Cell Death in Drosophila. Science 1994, 264, 677–683. [Google Scholar] [CrossRef] [PubMed]
- Bello, B.C.; Hirth, F.; Gould, A.P. A Pulse of the Drosophila Hox Protein Abdominal-A Schedules the End of Neural Proliferation via Neuroblast Apoptosis. Neuron 2003, 37, 209–219. [Google Scholar] [CrossRef]
- Maurange, C.; Cheng, L.; Gould, A.P. Temporal Transcription Factors and Their Targets Schedule the End of Neural Proliferation in Drosophila. Cell 2008, 133, 891–902. [Google Scholar] [CrossRef] [PubMed]
- Abrams, J.M.; White, K.; Fessier, L.I.; Steller, H. Programmed cell death during Drosophila embryogenesis. Development 1993, 117, 29–43. [Google Scholar] [PubMed]
- Campos-Ortega, J.A.; Jan, Y.N. Genetic and Molecular Bases of Neurogenesis in Drosophila melanogaster. Ann. Rev. Neurosci. 1991, 14, 399–420. [Google Scholar] [CrossRef] [PubMed]
- Nambu, P.A.; Nambu, J.R. The Drosophila fish-hook gene encodes a HMG domain protein essential for segmentation and CNS development. Development 1996, 122, 3467–3475. [Google Scholar] [PubMed]
- Overton, P.M.; Meadows, L.A.; Urban, J.; Russell, S. Evidence for differential and redundant function of the Sox genes Dichaete and SoxN during CNS development in Drosophila. Development 2002, 129, 4219–4228. [Google Scholar] [PubMed]
- Zhao, G.; Skeath, J.B. The Sox-domain containing gene Dichaete/fish-hook acts in concert with vnd and ind to regulate cell fate in the Drosophila neuroectoderm. Development 2002, 129, 1165–1174. [Google Scholar] [PubMed]
- Buescher, M.; Hing, F.S.; Chia, W. Formation of neuroblasts in the embryonic central nervous system of Drosophila melanogaster is controlled by SoxNeuro. Development 2002, 129, 4193–4203. [Google Scholar] [PubMed]
- Aleksic, J.; Ferrero, E.; Fischer, B.; Shen, S.P.; Russell, S. The role of Dichaete in transcriptional regulation during Drosophila embryonic development. BMC Genom. 2013, 14, 861–878. [Google Scholar] [CrossRef] [PubMed]
- Ashraf, S.I.; Ip, Y.T. The Snail protein family regulates neuroblast expression of inscuteable and string, genes involved in asymmetry and cell division in Drosophila. Development 2001, 128, 4757–4767. [Google Scholar] [PubMed]
- Cai, Y.; Chia, W.; Yang, X. A family of snail-related zinc finger proteins regulates two distinct and parallel mechanisms that mediate Drosophila neuroblast asymmetric divisions. EMBO J. 2001, 20, 1704–1714. [Google Scholar] [CrossRef] [PubMed]
- Bahrampour, S.; Gunnar, E.; Jonsson, C.; Ekman, H.; Thor, S. Neural Lineage Progression Controlled by a Temporal Proliferation Program. Dev. Cell 2017, 43, 332–348.e4. [Google Scholar] [CrossRef] [PubMed]
- Doe, C.Q. Molecular markers for identified neuroblasts and ganglion mother cells in the Drosophila central nervous system. Development 1992, 116, 855–863. [Google Scholar] [PubMed]
- Skeath, J.B. At the nexus between pattern formation and cell-type specification: The generation of individual neuroblast fates in the Drosophila embryonic central nervous system. BioEssays 1999, 21, 922–931. [Google Scholar] [CrossRef]
- Becker, T.; Technau, G.M. Single cell transplantation reveals interspecific cell communication in Drosophila chimeras. Development 1990, 109, 821–832. [Google Scholar] [PubMed]
- Prokop, A.; Technau, G.M. Early tagma-specific commitment of Drosophila CNS progenitor NB1-1. Development 1994, 120, 2557–2578. [Google Scholar]
- Udolph, G.; Luer, K.; Bossing, T.; Technau, G. Commitment of CNS progenitors along the dorsoventral axis of Drosophila neuroectoderm. Science 1995, 269, 1278–1281. [Google Scholar] [CrossRef] [PubMed]
- Prokop, A.; Bray, S.; Harrison, E.; Technau, G.M. Homeotic regulation of segment-specific differences in neuroblast numbers and proliferation in the Drosophila central nervous system. Mech. Dev. 1998, 74, 99–110. [Google Scholar] [CrossRef]
- Furst, A.; Mahowald, A.P. Cell division cycle of cultured neural precursor cells from Drosophila. Dev. Biol. 1985, 112, 467–476. [Google Scholar] [CrossRef]
- Birkholz, O.; Rickert, C.; Berger, C.; Urbach, R.; Technau, G.M. Neuroblast pattern and identity in the Drosophila tail region and role of doublesex in the survival of sex-specific precursors. Development 2013, 140, 1830–1842. [Google Scholar] [CrossRef] [PubMed]
- Isshiki, T.; Pearson, B.; Holbrook, S.; Doe, C.Q. Drosophila Neuroblasts Sequentially Express Transcription Factors which Specify the Temporal Identity of Their Neuronal Progeny. Cell 2001, 106, 511–521. [Google Scholar] [CrossRef]
- Grosskortenhaus, R.; Pearson, B.J.; Marusich, A.; Doe, C.Q. Regulation of Temporal Identity Transitions in Drosophila Neuroblasts. Dev. Cell 2005, 8, 193–202. [Google Scholar] [CrossRef] [PubMed]
- Doe, C.Q. Temporal Patterning in the Drosophila CNS. Annu. Rev. Cell Dev. Biol. 2017, 33, 219–240. [Google Scholar] [CrossRef] [PubMed]
- Syed, M.H.; Mark, B.; Doe, C.Q. Steroid hormone induction of temporal gene expression in Drosophila brain neuroblasts generates neuronal and glial diversity. eLife 2017, 6, e26287. [Google Scholar] [CrossRef] [PubMed]
- Ren, Q.; Yang, C.P.; Liu, Z.; Sugino, K.; Mok, K.; He, Y.; Ito, M.; Nern, A.; Otsuna, H.; Lee, T. Stem Cell-Intrinsic, Seven-up-Triggered Temporal Factor Gradients Diversify Intermediate Neural Progenitors. Curr. Biol. 2017, 27, 1303–1313. [Google Scholar] [CrossRef] [PubMed]
- Homem, C.C.F.; Repic, M.; Knoblich, J.A. Proliferation control in neural stem and progenitor cells. Nat. Rev. Neurosci. 2015, 16, 647. [Google Scholar] [CrossRef] [PubMed]
- Bello, B.C.; Izergina, N.; Caussinus, E.; Reichert, H. Amplification of neural stem cell proliferation by intermediate progenitor cells in Drosophila brain development. Neural Dev. 2008, 3, 5. [Google Scholar] [CrossRef] [PubMed]
- Bowman, S.K.; Rolland, V.; Betschinger, J.; Kinsey, K.A.; Emery, G.; Knoblich, J.A. The Tumor Suppressors Brat and Numb Regulate Transit-Amplifying Neuroblast Lineages in Drosophila. Dev. Cell 2008, 14, 535–546. [Google Scholar] [CrossRef] [PubMed]
- Boone, J.Q.; Doe, C.Q. Identification of Drosophila type II neuroblast lineages containing transit amplifying ganglion mother cells. Dev. Neurobiol. 2008, 68, 1185–1195. [Google Scholar] [CrossRef] [PubMed]
- Homem, C.C.F.; Steinmann, V.; Burkard, T.R.; Jais, A.; Esterbauer, H.; Knoblich, J.A. Ecdysone and Mediator Change Energy Metabolism to Terminate Proliferation in Drosophila Neural Stem Cells. Cell 2014, 158, 874–888. [Google Scholar] [CrossRef] [PubMed]
- Baumgardt, M.; Karlsson, D.; Salmani, B.Y.; Bivik, C.; MacDonald, R.B.; Gunnar, E.; Thor, S. Global programmed switch in neural daughter cell proliferation mode triggered by a temporal gene cascade. Dev. Cell 2014, 30, 192–208. [Google Scholar] [CrossRef] [PubMed]
- Baumgardt, M.; Karlsson, D.; Terriente, J.; Díaz-Benjumea, F.J.; Thor, S. Neuronal Subtype Specification within a Lineage by Opposing Temporal Feed-Forward Loops. Cell 2009, 139, 969–982. [Google Scholar] [CrossRef] [PubMed]
- Karcavich, R.; Doe, C.Q. Drosophila neuroblast 7-3 cell lineage: A model system for studying programmed cell death, Notch/Numb signaling, and sequential specification of ganglion mother cell identity. J. Comp. Neurol. 2005, 481, 240–251. [Google Scholar] [CrossRef] [PubMed]
- Sousa-Nunes, R.; Somers, W.G. Mechanisms of asymmetric progenitor divisions in the Drosophila central nervous system. Adv. Exp. Med. Biol. 2013, 786, 79–102. [Google Scholar] [CrossRef] [PubMed]
- Yu, F.; Kuo, C.T.; Jan, Y.N. Drosophila Neuroblast Asymmetric Cell Division: Recent Advances and Implications for Stem Cell Biology. Neuron 2006, 51, 13–20. [Google Scholar] [CrossRef] [PubMed]
- Knoblich, J.A.; Jan, Y.N. Asymmetric segregation of Numb and Prospero during cell division. Nature 1995, 377, 624–627. [Google Scholar] [CrossRef] [PubMed]
- Hirata, J.; Nakagoshi, H.; Nabeshima, Y.; Matsuzaki, F. Asymmetric segregation of the homeodomain protein Prospero during Drosophila development. Nature 1995, 377, 627–630. [Google Scholar] [CrossRef] [PubMed]
- Broadus, J.; Fuerstenberg, S.; Doe, C.Q. Staufen-dependent localization of prospero mRNA contributes to neuroblast daughter-cell fate. Nature 1998, 391, 792–795. [Google Scholar] [CrossRef] [PubMed]
- Schuldt, A.J.; Adams, J.H.; Davidson, C.M.; Micklem, D.R.; Haseloff, J.; St Johnston, D.; Brand, A.H. Miranda mediates asymmetric protein and RNA localization in the developing nervous system. Genes Dev. 1998, 12, 1847–1857. [Google Scholar] [CrossRef] [PubMed]
- Shen, C.P.; Knoblich, J.A.; Chan, Y.M.; Jiang, M.M.; Jan, L.Y.; Jan, Y.N. Miranda as a multidomain adapter linking apically localized Inscuteable and basally localized Staufen and Prospero during asymmetric cell division in Drosophila. Genes Dev. 1998, 12, 1837–1846. [Google Scholar] [CrossRef] [PubMed]
- Ikeshima-Kataoka, H.; Skeath, J.B.; Nabeshima, Y.; Doe, C.Q.; Matsuzaki, F. Miranda directs Prospero to a daughter cell during Drosophila asymmetric divisions. Nature 1997, 390, 625–629. [Google Scholar] [CrossRef] [PubMed]
- Matsuzaki, F.; Ohshiro, T.; Ikeshima-Kataoka, H.; Izumi, H. miranda localizes staufen and prospero asymmetrically in mitotic neuroblasts and epithelial cells in early Drosophila embryogenesis. Development 1998, 125, 4089–4098. [Google Scholar] [PubMed]
- Wodarz, A.; Ramrath, A.; Kuchinke, U.; Knust, E. Bazooka provides an apical cue for Inscuteable localization in Drosophila neuroblasts. Nature 1999, 402, 544–547. [Google Scholar] [CrossRef] [PubMed]
- Wodarz, A.; Ramrath, A.; Grimm, A.; Knust, E. Drosophila atypical protein kinase C associates with Bazooka and controls polarity of epithelia and neuroblasts. J. Cell Biol. 2000, 150, 1361–1374. [Google Scholar] [CrossRef] [PubMed]
- Li, P.; Yang, X.; Wasser, M.; Cai, Y.; Chia, W. Inscuteable and Staufen mediate asymmetric localization and segregation of prospero RNA during Drosophila neuroblast cell divisions. Cell 1997, 90, 437–447. [Google Scholar] [CrossRef]
- Schober, M.; Schaefer, M.; Knoblich, J.A. Bazooka recruits Inscuteable to orient asymmetric cell divisions in Drosophila neuroblasts. Nature 1999, 402, 548–551. [Google Scholar] [CrossRef] [PubMed]
- Kraut, R.; Chia, W.; Jan, L.Y.; Jan, Y.N.; Knoblich, J.A. Role of inscuteable in orienting asymmetric cell divisions in Drosophila. Nature 1996, 383, 50–55. [Google Scholar] [CrossRef] [PubMed]
- Kuchinke, U.; Grawe, F.; Knust, E. Control of spindle orientation in Drosophila by the Par-3-related PDZ-domain protein Bazooka. Curr. Biol. 1998, 8, 1357–1365. [Google Scholar] [CrossRef]
- Mora, N.; Oliva, C.; Fiers, M.; Ejsmont, R.; Soldano, A.; Zhang, T.T.; Yan, J.; Claeys, A.; De Geest, N.; Hassan, B.A. A Temporal Transcriptional Switch Governs Stem Cell Division, Neuronal Numbers, and Maintenance of Differentiation. Dev. Cell 2018, 45, 53–66.e5. [Google Scholar] [CrossRef] [PubMed]
- Skeath, J.B.; Carroll, S.B. Regulation of proneural gene expression and cell fate during neuroblast segregation in the Drosophila embryo. Development 1992, 114, 939–946. [Google Scholar] [PubMed]
- Cabrera, C.V. Lateral inhibition and cell fate during neurogenesis in Drosophila: The interactions between scute, Notch and Delta. Development 1990, 110, 733–742. [Google Scholar] [PubMed]
- Martin-Bermudo, M.D.; Martinez, C.; Rodriguez, A.; Jimenez, F. Distribution and function of the lethal of scute gene product during early neurogenesis in Drosophila. Development 1991, 113, 445–454. [Google Scholar] [PubMed]
- Egger, B.; Boone, J.Q.; Stevens, N.R.; Brand, A.H.; Doe, C.Q. Regulation of spindle orientation and neural stem cell fate in the Drosophila optic lobe. Neural Dev. 2007, 2, 1. [Google Scholar] [CrossRef] [PubMed]
- Hakes, A.E.; Otsuki, L.; Brand, A.H. A newly discovered neural stem cell population is generated by the optic lobe neuroepithelium during embryogenesis in Drosophila melanogaster. Development 2018, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Walsh, K.T.; Doe, C.Q. Drosophila embryonic type II neuroblasts: Origin, temporal patterning, and contribution to the adult central complex. Development 2017, 144, 4552–4562. [Google Scholar] [CrossRef] [PubMed]
- Álvarez, J.-A.; Díaz-Benjumea, F.J. Origin and specification of type II neuroblasts in the Drosophila embryo. Development 2018, 145, dev158394. [Google Scholar] [CrossRef] [PubMed]
- Xie, Y.; Li, X.; Deng, X.; Hou, Y.; O’Hara, K.; Urso, A.; Peng, Y.; Chen, L.; Zhu, S. The Ets protein Pointed prevents both premature differentiation and dedifferentiation of Drosophila intermediate neural progenitors. Development 2016, 143, 3109–3118. [Google Scholar] [CrossRef] [PubMed]
- Zhu, S.; Barshow, S.; Wildonger, J.; Jan, L.Y.; Jan, Y.N. Ets transcription factor Pointed promotes the generation of intermediate neural progenitors in Drosophila larval brains. Proc. Natl. Acad. Sci. USA 2011, 108, 20615–20620. [Google Scholar] [CrossRef] [PubMed]
- An, H.; Ge, W.; Xi, Y.; Yang, X. Inscuteable maintains type I neuroblast lineage identity via Numb/Notch signaling in the Drosophila larval brain. J. Genet. Genom. 2017, 44, 151–162. [Google Scholar] [CrossRef] [PubMed]
- Udolph, G.; Prokop, A.; Bossing, T.; Technau, G.M. A common precursor for glia and neurons in the embryonic CNS of Drosophila gives rise to segment-specific lineage variants. Development 1993, 118, 765–775. [Google Scholar] [PubMed]
- Bossing, T.; Udolph, G.; Doe, C.Q.; Technau, G.M. The Embryonic Central Nervous System Lineages of Drosophila melanogaster: I. Neuroblast Lineages Derived from the Ventral Half of the Neuroectoderm. Dev. Biol. 1996, 179, 41–64. [Google Scholar] [CrossRef] [PubMed]
- Schmid, A.; Chiba, A.; Doe, C.Q. Clonal analysis of Drosophila embryonic neuroblasts: Neural cell types, axon projections and muscle targets. Development 1999, 126, 4653–4689. [Google Scholar] [PubMed]
- Cobeta, I.M.; Salmani, B.Y.; Thor, S. Anterior-Posterior Gradient in Neural Stem and Daughter Cell Proliferation Governed by Spatial and Temporal Hox Control. Curr. Biol. 2017, 27, 1161–1172. [Google Scholar] [CrossRef] [PubMed]
- Salmani, B.Y.; Cobeta, I.M.; Rakar, J.; Bauer, S.; Curt, J.R.; Starkenberg, A.; Thor, S. Evolutionarily conserved anterior expansion of the central nervous system promoted by a common PcG-Hox program. Development 2018, 145, dev160747. [Google Scholar] [CrossRef] [PubMed]
- Berger, C.; Pallavi, S.K.; Prasad, M.; Shashidhara, L.S.; Technau, G.M. A critical role for cyclin E in cell fate determination in the central nervous system of Drosophila melanogaster. Nat. Cell Biol. 2005, 7, 56–62. [Google Scholar] [CrossRef] [PubMed]
- Berger, C.; Kannan, R.; Myneni, S.; Renner, S.; Shashidhara, L.S.; Technau, G.M. Cell cycle independent role of Cyclin E during neural cell fate specification in Drosophila is mediated by its regulation of Prospero function. Dev. Biol. 2010, 337, 415–424. [Google Scholar] [CrossRef] [PubMed]
- Shen, Y.; Wang, L.; Hirose, S.; Zhou, Z.; Liu, Q. The transcriptional factor Apt regulates neuroblast differentiation through activating CycE expression. Biochem. Biophys. Res. Commun. 2018, 499, 889–894. [Google Scholar] [CrossRef] [PubMed]
- Hartenstein, V.; Rudloff, E.; Ortega, J.A.C. The pattern of proliferation of the neuroblasts in the wild-type embryo of Drosophila melanogaster. Rouxs Arch. Dev. Biol. 1987, 196, 473–485. [Google Scholar] [CrossRef] [PubMed]
- Ito, K.; Hotta, Y. Proliferation pattern of postembryonic neuroblasts in the brain of Drosophila melanogaster. Dev. Biol. 1992, 149, 134–148. [Google Scholar] [CrossRef]
- Sousa-Nunes, R.; Yee, L.L.; Gould, A.P. Fat cells reactivate quiescent neuroblasts via TOR and glial insulin relays in Drosophila. Nature 2011, 471, 508–512. [Google Scholar] [CrossRef] [PubMed]
- Otsuki, L.; Brand, A.H. Cell cycle heterogeneity directs the timing of neural stem cell activation from quiescence. Science 2018, 360, 99–102. [Google Scholar] [CrossRef] [PubMed]
- Mata, J.; Curado, S.; Ephrussi, A.; Rørth, P. Tribbles coordinates mitosis and morphogenesis in Drosophila by regulating string/CDC25 proteolysis. Cell 2000, 101, 511–522. [Google Scholar] [CrossRef]
- Grosshans, J.; Wieschaus, E. A genetic link between morphogenesis and cell division during formation of the ventral furrow in Drosophila. Cell 2000, 101, 523–531. [Google Scholar] [CrossRef]
- Seher, T.C.; Leptin, M. Tribbles a cell-cycle brake that coordinates proliferation and morphogenesis during Drosophila gastrulation. Curr. Biol. 2000, 10, 623–629. [Google Scholar] [CrossRef]
- Choksi, S.P.; Southall, T.D.; Bossing, T.; Edoff, K.; de Wit, E.; Fischer, B.E.; van Steensel, B.; Micklem, G.; Brand, A.H. Prospero Acts as a Binary Switch between Self-Renewal and Differentiation in Drosophila Neural Stem Cells. Dev. Cell 2006, 11, 775–789. [Google Scholar] [CrossRef] [PubMed]
- Lai, S.-L.; Doe, C.Q. Transient nuclear Prospero induces neural progenitor quiescence. eLife 2014, 3, 379–12. [Google Scholar] [CrossRef] [PubMed]
- Tsuji, T.; Hasegawa, E.; Isshiki, T. Neuroblast entry into quiescence is regulated intrinsically by the combined action of spatial Hox proteins and temporal identity factors. Development 2008, 135, 3859–3869. [Google Scholar] [CrossRef] [PubMed]
- Chell, J.M.; Brand, A.H. Nutrition-Responsive Glia Control Exit of Neural Stem Cells from Quiescence. Cell 2010, 143, 1161–1173. [Google Scholar] [CrossRef] [PubMed]
- Britton, J.S.; Edgar, B.A. Environmental control of the cell cycle in Drosophila: Nutrition activates mitotic and endoreplicative cells by distinct mechanisms. Development 1998, 125, 2149–2158. [Google Scholar] [PubMed]
- Lin, S.; Marin, E.C.; Yang, C.P.; Kao, C.F.; Apenteng, B.A.; Huang, Y.; O’Connor, M.B.; Truman, J.W.; Lee, T. Extremes of lineage plasticity in the Drosophila brain. Curr. Biol. 2013, 23, 1908–1913. [Google Scholar] [CrossRef] [PubMed]
- Sipe, C.W.; Siegrist, S.E. Eyeless uncouples mushroom body neuroblast proliferation from dietary amino acids in Drosophila. eLife 2017, 6, e26343. [Google Scholar] [CrossRef] [PubMed]
- Ebens, A.J.; Garren, H.; Cheyette, B.N.R.; Zipursky, S.L. The Drosophila anachronism Locus: A Glycoprotein Secreted by Glia Inhibits Neuroblast Proliferation. Cell 1993, 74, 15–27. [Google Scholar] [CrossRef]
- Datta, S. Control of proliferation activation in quiescent neuroblasts of the Drosophila central nervous system. Development 1995, 121, 1173–1182. [Google Scholar] [PubMed]
- Voigt, A.; Pflanz, R.; Schäfer, U.; Jäckle, H. Perlecan participates in proliferation activation of quiescent Drosophila neuroblasts. Dev. Dyn. 2002, 224, 403–412. [Google Scholar] [CrossRef] [PubMed]
- Kanai, M.I.; Kim, M.J.; Akiyama, T.; Takemura, M.; Wharton, K.; O’Connor, M.B.; Nakato, H. Regulation of neuroblast proliferation by surface glia in the Drosophila larval brain. Sci. Rep. 2018, 8, 3730. [Google Scholar] [CrossRef] [PubMed]
- Weng, R.; Cohen, S.M. Drosophila miR-124 regulates neuroblast proliferation through its target anachronism. Development 2012, 139, 1427–1434. [Google Scholar] [CrossRef] [PubMed]
- Droujinine, I.A.; Perrimon, N. Interorgan Communication Pathways in Physiology: Focus on Drosophila. Annu. Rev. Genet. 2016, 50, 539–570. [Google Scholar] [CrossRef] [PubMed]
- Delanoue, R.; Meschi, E.; Agrawal, N.; Mauri, A.; Tsatskis, Y.; McNeill, H.; Léopold, P. Drosophila insulin release is triggered by adipose Stunted ligand to brain Methuselah receptor. Science 2016, 353, 1553–1556. [Google Scholar] [CrossRef] [PubMed]
- Koyama, T.; Mirth, C.K. Growth-Blocking Peptides as Nutrition-Sensitive Signals for Insulin Secretion and Body Size Regulation. PLoS Biol. 2016, 14, e1002392. [Google Scholar] [CrossRef]
- Sung, E.J.; Ryuda, M.; Matsumoto, H.; Uryu, O.; Ochiai, M.; Cook, M.E.; Yi, N.Y.; Wang, H.; Putney, J.W.; Bird, G.S.; et al. Cytokine signaling through Drosophila Mthl10 ties lifespan to environmental stress. Proc. Natl. Acad. Sci. USA 2017, 114, 13786–13791. [Google Scholar] [CrossRef] [PubMed]
- Li, S.; Koe, C.T.; Tay, S.T.; Tan, A.L.; Zhang, S.; Zhang, Y.; Tan, P.; Sung, W.K.; Wang, H. An intrinsic mechanism controls reactivation of neural stem cells by spindle matrix proteins. Nat. Commun. 2017, 8, 122. [Google Scholar] [CrossRef] [PubMed]
- Wasser, M.; Osman, Z.B.; Chia, W. EAST and Chromator control the destruction and remodeling of muscles during Drosophila metamorphosis. Dev. Biol. 2007, 307, 380–393. [Google Scholar] [CrossRef] [PubMed]
- Guruharsha, K.G.; Rual, J.F.; Zhai, B.; Mintseris, J.; Vaidya, P.; Vaidya, N.; Beekman, C.; Wong, C.; Rhee, D.Y.; Cenaj, O.; et al. A Protein Complex Network of Drosophila melanogaster. Cell 2011, 147, 690–703. [Google Scholar] [CrossRef] [PubMed]
- Bohla, D.; Herold, M.; Panzer, I.; Buxa, M.K.; Ali, T.; Demmers, J.; Krüger, M.; Scharfe, M.; Jarek, M.; Bartkuhn, M.; et al. A Functional Insulator Screen Identifies NURF and dREAM Components to Be Required for Enhancer-Blocking. PLoS ONE 2014, 9, e107765. [Google Scholar] [CrossRef] [PubMed]
- Vogelmann, J.; Le Gall, A.; Dejardin, S.; Allemand, F.; Gamot, A.; Labesse, G.; Cuvier, O.; Nègre, N.; Cohen-Gonsaud, M.; Margeat, E.; et al. Chromatin Insulator Factors Involved in Long-Range DNA Interactions and Their Role in the Folding of the Drosophila Genome. PLoS Genet. 2014, 10, e1004544. [Google Scholar] [CrossRef] [PubMed]
- Rhee, D.Y. Transcription Factor Networks in Drosophila melanogaster. Cell Rep. 2014, 8, 2031–2043. [Google Scholar] [CrossRef] [PubMed]
- Chen, P.; Nordstrom, W.; Gish, B.; Abrams, J.M. grim, a novel cell death gene in Drosophila. Genes Dev. 1996, 10, 1773–1782. [Google Scholar] [CrossRef] [PubMed]
- Grether, M.E.; Abrams, J.M.; Agapite, J.; White, K.; Steller, H. The head involution defective gene of Drosophila melanogaster functions in programmed cell death. Genes Dev. 1995, 9, 1694–1708. [Google Scholar] [CrossRef] [PubMed]
- Peterson, C.; Carney, G.E.; Taylor, B.J.; White, K. reaper is required for neuroblast apoptosis during Drosophila development. Development 2002, 129, 1467–1476. [Google Scholar] [PubMed]
- Tan, Y.; Yamada-Mabuchi, M.; Arya, R.; St Pierre, S.; Tang, W.; Tosa, M.; Brachmann, C.; White, K. Coordinated expression of cell death genes regulates neuroblast apoptosis. Development 2011, 138, 2197–2206. [Google Scholar] [CrossRef] [PubMed]
- Yang, Y.; Fang, S.; Jensen, J.P.; Weissman, A.M.; Ashwell, J.D. Ubiquitin Protein Ligase Activity of IAPs and Their Degradation in Proteasomes in Response to Apoptotic Stimuli. Science 2000, 288, 874–877. [Google Scholar] [CrossRef] [PubMed]
- Suzuki, Y.; Nakabayashi, Y.; Takahashi, R. Ubiquitin-protein ligase activity of X-linked inhibitor of apoptosis protein promotes proteasomal degradation of caspase-3 and enhances its anti-apoptotic effect in Fas-induced cell death. Proc. Natl. Acad. Sci. USA 2001, 98, 8662–8667. [Google Scholar] [CrossRef] [PubMed]
- Chai, J.; Yan, N.; Huh, J.R.; Wu, J.W.; Li, W.; Hay, B.A.; Shi, Y. Molecular mechanism of Reaper-Grim-Hid-mediated suppression of DIAP1-dependent Dronc ubiquitination. Nat. Struct. Biol. 2003, 10, 892–898. [Google Scholar] [CrossRef] [PubMed]
- Lisi, S.; Mazzon, I.; White, K. Diverse Domains of THREAD/DIAP1 Are Required to Inhibit Apoptosis Induced by REAPER and HID in Drosophila. Genetics 2000, 154, 669–678. [Google Scholar] [PubMed]
- Wing, J.P.; Schwartz, L.M.; Nambu, J.R. The RHG motifs of Drosophila Reaper and Grim are important for their distinct cell death-inducing abilities. Mech. Dev. 2001, 102, 193–203. [Google Scholar] [CrossRef]
- Zachariou, A.; Tenev, T.; Goyal, L.; Agapite, J.; Steller, H.; Meier, P. IAP-antagonists exhibit non-redundant modes of action through differential DIAP1 binding. EMBO J. 2003, 22, 6642–6652. [Google Scholar] [CrossRef] [PubMed]
- Yokokura, T.; Dresnek, D.; Huseinovic, N.; Lisi, S.; Abdelwahid, E.; Bangs, P.; White, K. Dissection of DIAP1 Functional Domains via a Mutant Replacement Strategy. J. Biol. Chem. 2004, 279, 52603–52612. [Google Scholar] [CrossRef] [PubMed]
- Ryoo, H.D.; Bergmann, A.; Gonen, H.; Ciechanover, A.; Steller, H. Regulation of Drosophila IAP1 degradation and apoptosis by reaper and ubcD1. Nat. Cell Biol. 2002, 4, 432–438. [Google Scholar] [CrossRef] [PubMed]
- Yoo, S.J.; Huh, J.R.; Muro, I.; Yu, H.; Wang, L.; Wang, S.L.; Feldman, R.R.; Clem, R.J.; Müller, H.A.; Hay, B.A. Hid, Rpr and Grim negatively regulate DIAP1 levels through distinct mechanisms. Nat. Cell Biol. 2002, 4, 416–424. [Google Scholar] [CrossRef] [PubMed]
- Arya, R.; White, K. Cell death in development: Signaling pathways and core mechanisms. Semin. Cell Dev. Biol. 2015, 39, 12–19. [Google Scholar] [CrossRef] [PubMed]
- Pinto-Teixeira, F.; Konstantinides, N.; Desplan, C. Programmed cell death acts at different stages of Drosophila neurodevelopment to shape the central nervous system. FEBS Lett. 2016, 590, 2435–2453. [Google Scholar] [CrossRef] [PubMed]
- Arya, R.; Sarkissian, T.; Tan, Y.; White, K. Neural stem cell progeny regulate stem cell death in a Notch and Hox dependent manner. Cell Death Differ. 2015, 22, 1378–1387. [Google Scholar] [CrossRef] [PubMed]
- Khandelwal, R.; Sipani, R.; Rajan, S.G.; Kumar, R.; Joshi, R. Combinatorial action of Grainyhead, Extradenticle and Notch in regulating Hox mediated apoptosis in Drosophila larval CNS. PLoS Genet. 2017, 13, e1007043. [Google Scholar] [CrossRef] [PubMed]
- Arya, R.; Gyonjyan, S.; Harding, K.; Sarkissian, T.; Li, Y.; Zhou, L.; White, K. A Cut/cohesin axis alters the chromatin landscape to facilitate neuroblast death. bioRxiv 2018, 299164. [Google Scholar] [CrossRef]
- Cenci, C.; Gould, A. Drosophila Grainyhead specifies late programmes of neural proliferation by regulating the mitotic activity and Hox-dependent apoptosis of neuroblasts. Development 2005, 132, 3835–3845. [Google Scholar] [CrossRef] [PubMed]
- Almeida, M.S.; Bray, S.J. Regulation of post-embryonic neuroblasts by Drosophila Grainyhead. Mech. Dev. 2005, 122, 1282–1293. [Google Scholar] [CrossRef] [PubMed]
- Ulvklo, C.; MacDonald, R.; Bivik, C.; Baumgardt, M.; Karlsson, D.; Thor, S. Control of neuronal cell fate and number by integration of distinct daughter cell proliferation modes with temporal progression. Development 2012, 139, 678–689. [Google Scholar] [CrossRef] [PubMed]
- Chai, P.C.; Liu, Z.; Chia, W.; Cai, Y. Hedgehog Signaling Acts with the Temporal Cascade to Promote Neuroblast Cell Cycle Exit. PLoS Biol. 2013, 11, e1001494. [Google Scholar] [CrossRef] [PubMed]
- Yang, C.P.; Samuels, T.J.; Huang, Y.; Yang, L.; Ish-Horowicz, D.; Davis, I.; Lee, T. Imp and Syp RNA-binding proteins govern decommissioning of Drosophila neural stem cells. Development 2017, 144, 3454–3464. [Google Scholar] [CrossRef] [PubMed]
- Liu, Z.; Yang, C.P.; Sugino, K.; Fu, C.C.; Liu, L.Y.; Yao, X.; Lee, L.P.; Lee, T. Opposing intrinsic temporal gradients guide neural stem cell production of varied neuronal fates. Science 2015, 350, 317–320. [Google Scholar] [CrossRef] [PubMed]
- McDermott, S.M.; Yang, L.; Halstead, J.M.; Hamilton, R.S.; Meignin, C.; Davis, I. Drosophila Syncrip modulates the expression of mRNAs encoding key synaptic proteins required for morphology at the neuromuscular junction. RNA 2014, 20, 1593–1606. [Google Scholar] [CrossRef] [PubMed]
- Siegrist, S.E.; Haque, N.S.; Chen, C.H.; Hay, B.A.; Hariharan, I.K. Inactivation of Both foxo and reaper Promotes Long-Term Adult Neurogenesis in Drosophila. Curr. Biol. 2010, 20, 643–648. [Google Scholar] [CrossRef] [PubMed]
- Fuse, N.; Hisata, K.; Katzen, A.L.; Matsuzaki, F. Heterotrimeric G proteins regulate daughter cell size asymmetry in Drosophila neuroblast divisions. Curr. Biol. 2003, 13, 947–954. [Google Scholar] [CrossRef]
- Wang, H.; Somers, G.W.; Bashirullah, A.; Heberlein, U.; Yu, F.; Chia, W. Aurora-A acts as a tumor suppressor and regulates self-renewal of Drosophila neuroblasts. Genes Dev. 2006, 20, 3453–3463. [Google Scholar] [CrossRef] [PubMed]
- Weng, M.; Golden, K.; Lee, C.-Y. dFezf/Earmuff Maintains the Restricted Developmental Potential of Intermediate Neural Progenitors in Drosophila. Dev. Cell 2010, 18, 126–135. [Google Scholar] [CrossRef] [PubMed]
- Liu, K.; Shen, D.; Shen, J.; Gao, S.M.; Li, B.; Wong, C.; Feng, W.; Song, Y. The Super Elongation Complex Drives Neural Stem Cell Fate Commitment. Dev. Cell 2017, 40, 537–551.e6. [Google Scholar] [CrossRef] [PubMed]
- Zacharioudaki, E.; Magadi, S.S.; Delidakis, C. bHLH-O proteins are crucial for Drosophila neuroblast self-renewal and mediate Notch-induced overproliferation. Development 2012, 139, 1258–1269. [Google Scholar] [CrossRef] [PubMed]
- Bivik, C.; MacDonald, R.B.; Gunnar, E.; Mazouni, K.; Schweisguth, F.; Thor, S. Control of Neural Daughter Cell Proliferation by Multi-level Notch/Su(H)/E(spl)-HLH Signaling. PLoS Genet. 2016, 12, e1005984. [Google Scholar] [CrossRef] [PubMed]
- Guo, M.; Jan, L.Y.; Jan, Y.N. Control of daughter cell fates during asymmetric division: Interaction of Numb and Notch. Neuron 1996, 17, 27–41. [Google Scholar] [CrossRef]
- Spana, E.P.; Doe, C.Q. Numb antagonizes Notch signaling to specify sibling neuron cell fates. Neuron 1996, 17, 21–26. [Google Scholar] [CrossRef]
- Rhyu, M.S.; Jan, L.Y.; Jan, Y.N. Asymmetric Distribution of Numb Protein during Division of the Sensory Organ Precursor Cell Confers Distinct Fates to Daughter Cells. Cell 1994, 76, 477–491. [Google Scholar] [CrossRef]
- Wissel, S.; Harzer, H.; Bonnay, F.; Burkard, T.R.; Neumüller, R.A.; Knoblich, J.A. Time-resolved transcriptomics in neural stem cells identifies a v-ATPase/Notch regulatory loop. J. Cell Biol. 2018, 217, 3285–3300. [Google Scholar] [CrossRef] [PubMed]
- Portela, M.; Yang, L.; Paul, S.; Li, X.; Veraksa, A.; Parsons, L.M.; Richardson, H.E. Lgl reduces endosomal vesicle acidification and Notch signaling by promoting the interaction between Vap33 and the V-ATPase complex. Sci. Signal. 2018, 11, eaar1976. [Google Scholar] [CrossRef] [PubMed]
- Betschinger, J.; Mechtler, K.; Knoblich, J.A. Asymmetric Segregation of the Tumor Suppressor Brat Regulates Self-Renewal in Drosophila Neural Stem Cells. Cell 2006, 124, 1241–1253. [Google Scholar] [CrossRef] [PubMed]
- Lee, C.-Y.; Wilkinson, B.D.; Siegrist, S.E.; Wharton, R.P.; Doe, C.Q. Brat Is a Miranda Cargo Protein that Promotes Neuronal Differentiation and Inhibits Neuroblast Self-Renewal. Dev. Cell 2006, 10, 441–449. [Google Scholar] [CrossRef] [PubMed]
- Xiao, Q.; Komori, H.; Lee, C.-Y. klumpfuss distinguishes stem cells from progenitor cells during asymmetric neuroblast division. Development 2012, 139, 2670–2680. [Google Scholar] [CrossRef] [PubMed]
- Eroglu, E.; Burkard, T.R.; Jiang, Y.; Saini, N.; Homem, C.C.; Reichert, H.; Knoblich, J.A. SWI/SNF Complex Prevents Lineage Reversion and Induces Temporal Patterning in Neural Stem Cells. Cell 2014, 156, 1259–1273. [Google Scholar] [CrossRef] [PubMed]
- Koe, C.T.; Li, S.; Rossi, F.; Wong, J.J.; Wang, Y.; Zhang, Z.; Chen, K.; Aw, S.S.; Richardson, H.E.; Robson, P.; et al. The Brm-HDAC3-Erm repressor complex suppresses dedifferentiation in Drosophila type II neuroblast lineages. eLife 2014, 3, 2639. [Google Scholar] [CrossRef] [PubMed]
- Froldi, F.; Cheng, L.Y. Understanding how differentiation is maintained: Lessons from the Drosophila brain. Cell. Mol. Life Sci. 2016, 73, 1641–1644. [Google Scholar] [CrossRef] [PubMed]
- Carney, T.D.; Struck, A.J.; Doe, C.Q. midlife crisis encodes a conserved zinc-finger protein required to maintain neuronal differentiation in Drosophila. Development 2013, 140, 4155–4164. [Google Scholar] [CrossRef] [PubMed]
- Southall, T.D.; Davidson, C.M.; Miller, C.; Carr, A.; Brand, A.H. Dedifferentiation of Neurons Precedes Tumor Formation in lola Mutants. Dev. Cell 2014, 28, 685–696. [Google Scholar] [CrossRef] [PubMed]
- Froldi, F.; Szuperak, M.; Weng, C.-F.; Shi, W.; Papenfuss, A.T.; Cheng, L.Y. The transcription factor Nerfin-1 prevents reversion of neurons into neural stem cells. Genes Dev. 2015, 29, 129–143. [Google Scholar] [CrossRef] [PubMed]
- Pfeiffer, B.D.; Ngo, T.T.; Hibbard, K.L.; Murphy, C.; Jenett, A.; Truman, J.W.; Rubin, G.M. Refinement of Tools for Targeted Gene Expression in Drosophila. Genetics 2010, 186, 735–755. [Google Scholar] [CrossRef] [PubMed]
- Jenett, A.; Rubin, G.M.; Ngo, T.T.; Shepherd, D.; Murphy, C.; Dionne, H.; Pfeiffer, B.D.; Cavallaro, A.; Hall, D.; Jeter, J.; et al. A GAL4-driver line resource for Drosophila neurobiology. Cell Rep. 2012, 2, 991–1001. [Google Scholar] [CrossRef] [PubMed]
- Birkholz, O.; Rickert, C.; Nowak, J.; Coban, I.C.; Technau, G.M. Bridging the gap between postembryonic cell lineages and identified embryonic neuroblasts in the ventral nerve cord of Drosophila melanogaster. Biol. Open 2015, 4, 420–434. [Google Scholar] [CrossRef] [PubMed]
- Lacin, H.; Truman, J.W. Lineage mapping identifies molecular and architectural similarities between the larval and adult Drosophila central nervous system. eLife 2016, 5, e13399. [Google Scholar] [CrossRef] [PubMed]
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Harding, K.; White, K. Drosophila as a Model for Developmental Biology: Stem Cell-Fate Decisions in the Developing Nervous System. J. Dev. Biol. 2018, 6, 25. https://doi.org/10.3390/jdb6040025
Harding K, White K. Drosophila as a Model for Developmental Biology: Stem Cell-Fate Decisions in the Developing Nervous System. Journal of Developmental Biology. 2018; 6(4):25. https://doi.org/10.3390/jdb6040025
Chicago/Turabian StyleHarding, Katherine, and Kristin White. 2018. "Drosophila as a Model for Developmental Biology: Stem Cell-Fate Decisions in the Developing Nervous System" Journal of Developmental Biology 6, no. 4: 25. https://doi.org/10.3390/jdb6040025
APA StyleHarding, K., & White, K. (2018). Drosophila as a Model for Developmental Biology: Stem Cell-Fate Decisions in the Developing Nervous System. Journal of Developmental Biology, 6(4), 25. https://doi.org/10.3390/jdb6040025



