1. Introduction
Antimicrobial resistance has increased exponentially in recent decades and is considered one of the greatest health challenges of the current century. It is estimated that 700,000 individuals die worldwide every year due to infections caused by resistant strains, and this number could increase to approximately 10 million worldwide by 2050 if there is no intervention [
1].
Fungal infections are consequences of the health problems often associated with immunosuppression. Clinical success depends on the response to the antifungal therapy, presenting high mortality. Unfortunately, treatment options are restricted due to the limited availability of antifungals [
2].
Candida sp. are opportunistic agents responsible for several diseases, ranging from skin and mucosal infections to systemic infections.
Candida albicans accounts for more than 70% of all
Candida infections; however, there has been an increase in clinical cases related to species such as
Candida parapsilosis and
Candida krusei [
3].
Several mechanisms of resistance to antifungals have been described; among them is the formation of biofilm, which can lead to limitations in the use of available drugs. Furthermore, the indiscriminate use of these drugs increases the resistance, which has stimulated the use of gradually higher doses and broad-spectrum antifungals, which has increased the reporting of adverse reactions [
4].
Considering the increased microbial resistance to multiple drugs, such as antifungals, synergistic combinations between natural molecules and commercial drugs may be good alternatives for the control of these microorganisms, reducing the doses and consequently the adverse effects associated with the treatment [
5].
Lippia alba (
Verbenaceae), popularly known as lemon balm or lemon grass, is widely used by the population in the form of tea, macerated leaves, in compresses, baths or as an alcoholic extract, due to its calming effects, as well as for the treatment of skin diseases and gastrointestinal problems [
6,
7]. Several studies on the biological activities of
L. alba essential oil (LaEO) have demonstrated anxiolytic, antioxidant, anesthetic, antigenotoxic, antiviral, antibacterial and antiparasitic activities, thus supporting its use in the treatment of several diseases [
8,
9,
10,
11]. In northeastern Brazil, different chemotypes of
L. alba can be found, named according to the major constituents: chemotype I (citral, b-myrcene and limonene), chemotype II (citral and limonene) and chemotype III (carvone and limonene) [
12,
13]. Thus, the present study aimed to investigate the antifungal activity of LaEO against planctonic cell and
Candida sp. biofilm and its modulatory effect on the activity of clinically used antifungal agents.
4. Discussion
The present study showed that different concentrations of LaEO were able to inhibit the growth of Candida sp. strains associated with the inhibition of the biofilm formation capacity in the early hours of incubation. Furthermore, LaEO eradicated the already formed biofilm, although at higher concentrations. In this sense, LaEO presented a modulatory effect on the activity of clinically used antifungal agents, leading to a possible reduction in the concentration of these substances with better effects.
Several studies have reported the antimicrobial activity of the species
L. alba and that it has antifungal properties due to the presence of compounds such as coumarins, terpenes and flavonoids [
25]. The main presence of limonene and carvone in this oil allows its classification as chemotype III [
26].
Of the compounds most found in essential oils, carvone has shown antibacterial and antifungal activity [
27]. The mechanism of action of carvone involves structural destabilization of the phospholipid and interaction with membrane proteins, in addition to acting as a proton exchanger, altering the pH gradient across the membrane [
9].
Recent work has shown that monoterpenes, such as carvone, have been described as having activity against
Candida. Among the tested monoterpenes, (±)-citronellol was the most potent compound followed by (+)-α-pinene and menthol [
28]. It has already been shown that carvone interferes with the serum-induced formation of filamentous structures
Candida albicans at substantially lower concentrations than those causing significant inhibition of growth [
29].
Limonene, another major component of LaEO, inhibited the growth of
C. albicans by causing oxidative stress in the cell envelope, also leading to DNA damage, resulting in cell-cycle modulation and apoptosis induction through nucleolar stress [
28]. Indeed, it has already been described that limonene, a component generally recognized as safe (GRAS), inhibits
Candida albicans growth by inducing apoptosis through downregulation of Tps3 and activation of caspase (CaMca1) [
30].
For natural products, MIC values between 50 and 500, 600 and 1500 or above 1600.0 μg/mL are considered to have strong, moderate and weak activity, respectively [
31]. In the present study, LaEO showed an antifungal effect against eight
Candida strains, with MIC ranging from 0.078 mg/mL to 1.250 mg/mL, with values similar to those of MLC. This demonstrates that LaEO has antifungal effects rated from strong to weak, depending on the strain tested. Previous studies showed that LaEO showed fungicidal and fungistatic effects against saprophytic fungus at concentrations around 1.0 mg/mL, even with higher antifungal activity due to a synergistic effect with some active components [
32].
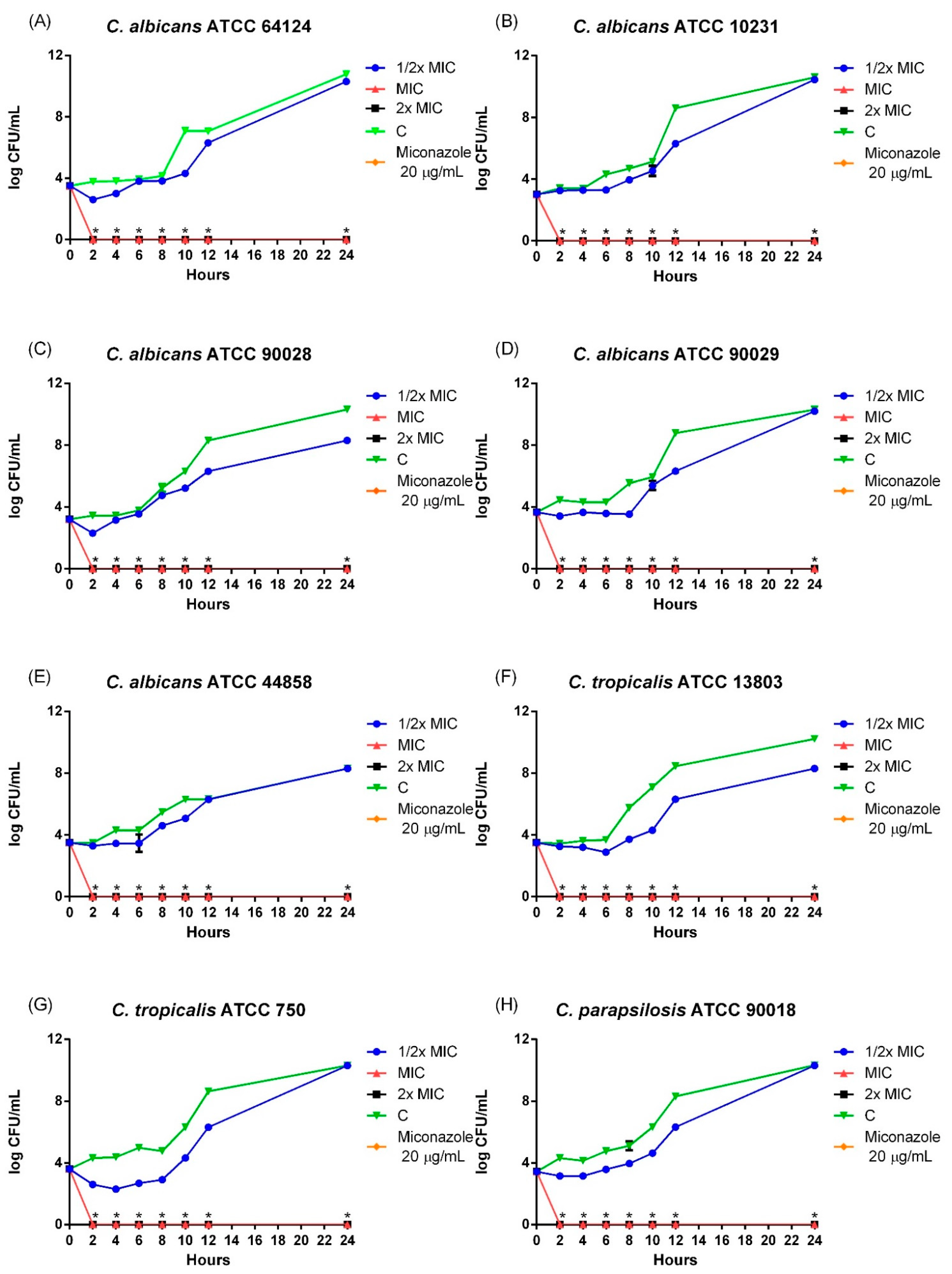
The evaluation of microbial growth kinetics is an important factor, as it provides valuable information for conducting microbiological experiments and determining the mechanism of action. Among the mechanisms evaluated, the inhibitory and lethal activity of substances can be determined by temporal killing assays, which allow the identification of the speed of antimicrobial action [
33,
34].
Importantly, the effects observed for LaEO occurred within the first hours of incubation, reducing microbial growth even at half MIC, indicating potent antifungal activity. Essential oil extracted from
Lippia sp. species has been shown to reduce growth by preventing fungal recovery [
35].
Eradicating the formation of these fungal biofilms is a challenge. Biofilm formation is an important strategy for survival, resistance and pathogenicity. Terpenes and flavonoid-rich extracts have been shown to reduce the expression of adhesion and aggregation molecules, reducing biofilm viability [
36].
Biofilms are usually composed of one or several species, forming so-called mono-species or multi-species biofilms, respectively, which also show increased resistance to antimicrobial and antifungal agents and may involve spoilage and pathogenic microorganisms and also involve sessile microbial cells that are different from their planktonic form as they have an altered phenotype and different metabolic characteristics [
37,
38].
Clinically used antifungals are generally ineffective against certain
C. albicans biofilms or require high doses to eradicate them, which can result in serious adverse effects. In general, antifungals eliminate free planktonic cells but are often ineffective against biofilms, mainly due to their incomplete removal, which can result in resistant reinfections [
39].
The prevention of
Candida biofilms is an important assay since they are associated with severe infections and the fact that microorganisms with high resistance to antifungal drugs and immune defenses occur in these structures [
40], which justifies the search for essential oils and other bioactive compounds with antibiotic activity.
The use of phytochemicals to treat biofilm-forming pathogens is a breakthrough, bringing with it new prospects for product development to prevent or eradicate the biofilm-forming microbial community [
41]. Several studies have demonstrated the inhibition of bacterial and fungal biofilm formation by phytoconstituents such as essential oils and flavonoids [
16,
42,
43,
44,
45] as in the case of LaEO, which was able to inhibit all strains tested.
In fact, EOs can act on the mechanisms involved in biofilm formation, thus making it possible to eradicate and control this process. The effect exerted depends both on the chemical characteristics associated with its chemical composition and on the target microorganism. It is noteworthy that both hydrophilic and hydrophobic compounds in essential oils can contribute to antibiofilm activity [
46].
In order to reduce the dosage of antifungal agents, preventing toxicity, adverse effects, collateral effects and drug interactions, several studies have developed strategies to combine bioactive molecules, which can interact and determine synergistic, additive or antagonistic effects. Synergism can be defined as the occurrence of a greater effect when combined than the sum of the individual effects of the assessed substances [
47]. Thus, synergistic interactions can increase efficacy and lead to a reduction in effective doses for the treatment of infectious diseases [
48].
Combination therapies that incorporate substances such as natural products can enhance the activity of an antimicrobial agent and are suggested as strategies to combat microbial resistance [
49]. LaEO showed different modulation profiles, with a predominance of synergistic and additive effects for the tested strains when associated with standard commercial antifungal agents.
Previously, a synergistic potential of limonene-rich
Citrus aurantium L. essential oil with antibiotics against
Candida albicans was demonstrated, indicating a potential interaction of this compound with the enzymes N-myristoyl transferase (NMT) and cytochrome P450 14-α sterol demethylase (CYP51), essential for the redox metabolism of the fungus [
50].
In in vitro experiments using LaEO (chemotype—geranium, neral and myrcene), a positive modulation of the action of ciprofloxacin, cefepime and ceftriaxone on Salmonella and Shigella strains was observed, also reducing the biofilm mass of both strains when exposed to the combination LaEO–ciprofloxacin [
45]. In another study using LaEO (chemotype—sabinene, (E)-caryophyllene and limonene), a positive modulatory activity (synergism) was observed in combinations with standard antifungal drugs, resulting in the inhibition of all
Trichophyton rubrum strains tested and of two strains of
C. tropicalis and one of
C. parapsilosis [
51].
Synergism assays have played a leading role in the search for new antimicrobial and antifungal compounds to improve the use of already known substances. One study showed antibacterial and antibiofilm activities and synergism with florfenicol from the essential oils of
Lippia sidoides and
Cymbopogon citratus against
Aeromonas hydrophila, a Gram-negative bacterium that has a high potential for resistance [
52].
Extracts from species of the genus Lippia have already been studied as modulators of antibiotic activity on
S. aureus. A study performed with
Lippia origanoides showed a modulating effect on antibacterial activity at subinhibitory concentrations of neomycin and amikacin. A similar modulation was found when the natural products were switched to chlorpromazine, an inhibitor of bacterial efflux pumps, suggesting the involvement of resistance mediated by the efflux system in MRSA [
53].
Altogether, our results demonstrate the good antifungal potential of LaEO. Interestingly, the oil is promising against planktonic fungus and in biofilms. It is a valuable finding, opening a field of study aiming at the development of a pharmacological and biotechnological tool, being a candidate for the development of a new antifungal agent for clinical use.