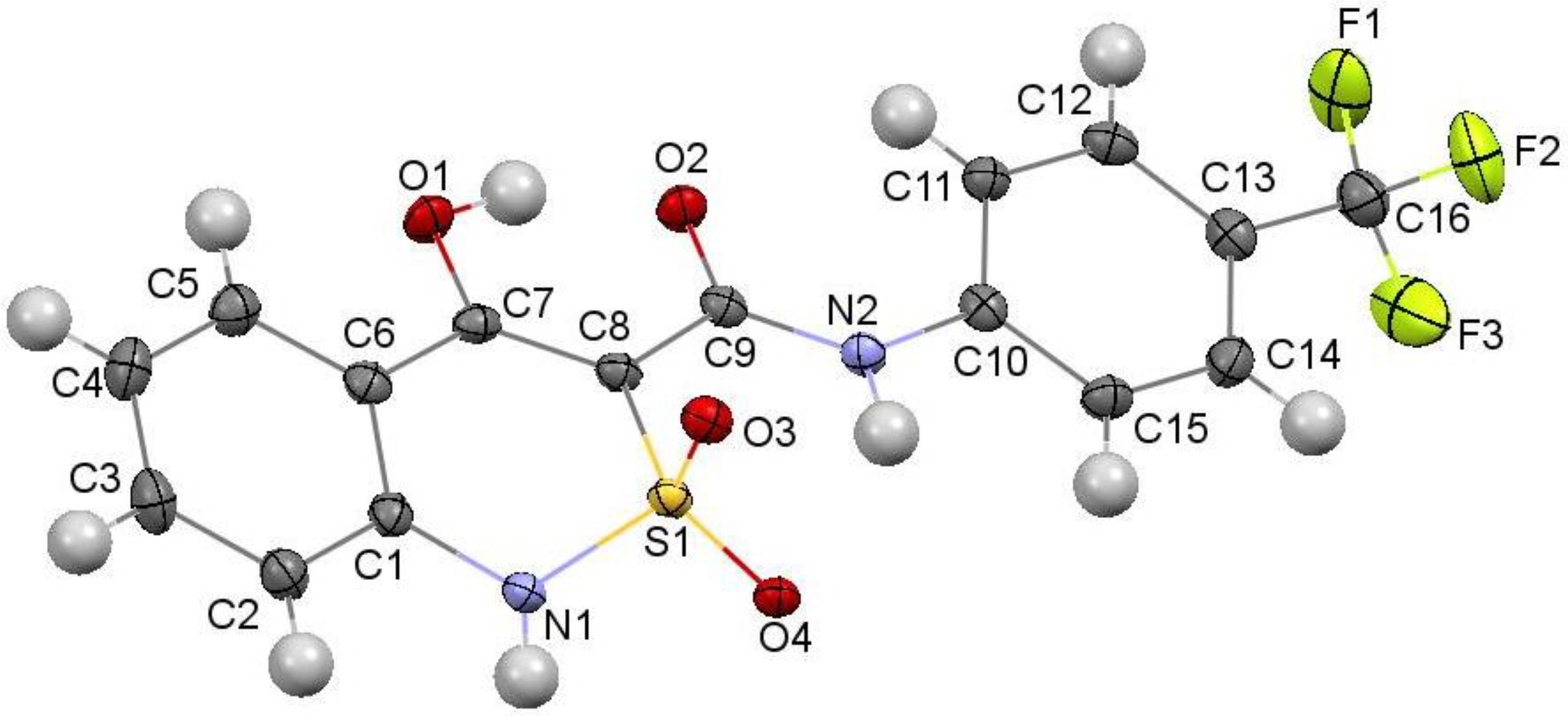
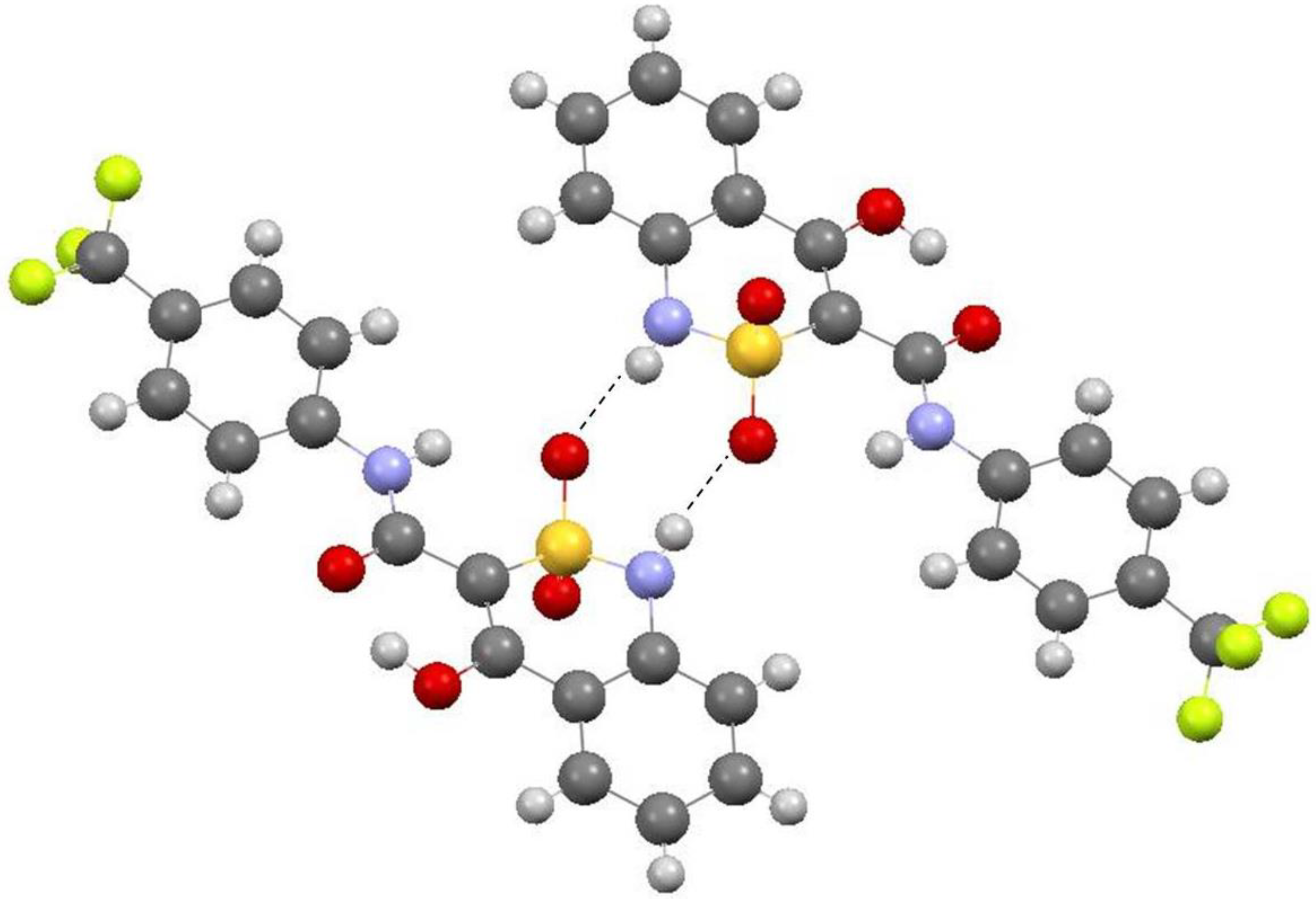
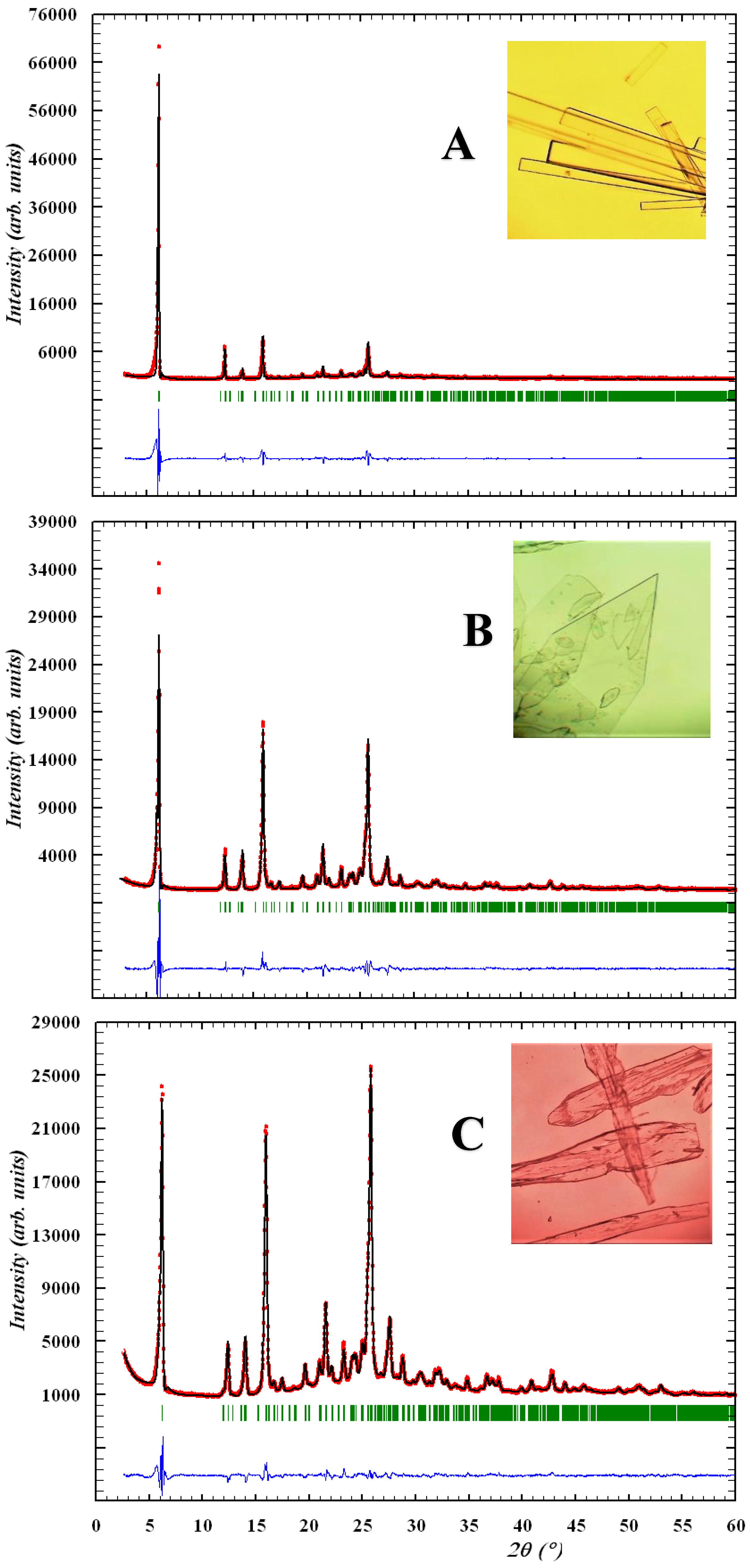
2.2. X-ray Structural Analysis of N-(4-Trifluoromethylphenyl)-4-Hydroxy-2,2-Dioxo-1H-2λ6,1-Benzothiazine-3-Carboxamide Crystalline Forms A, B and C
The crystals of form A were monoclinic, colorless sticks, C16H11N2O4F3S at 273 K, a 28.782(2), b 7.3709(4), c 14.933(1) Å, β 96.323(7)°, V 3148.6(4) Å3, Mr 384.33, Z 4, space group C2/c, dcalc 1.622 g/cm3, µ(MoKα) 0.266 mm−1, F(000) 1568.
The crystals of form B were monoclinic, colorless plates, C16H11N2O4F3S, at 100 K, a 28.697(2), b 7.3421(4), c 14.613(9) Å, β 96.112(9)°, V 3061.6(3) Å3, Mr 384.33, Z 4, space group C2/c, dcalc 1.668 g/cm3, µ(MoKα) 0.273 mm−1, F(000) 1568.
The crystals of form C were monoclinic, colorless blocks,·C16H11N2O4F3S, at 273 K, a 28.791(4), b 7.366(1), c 14.929(2) Å, β 96.37(1)°, V 3146.5(8) Å3, Mr 384.33, Z 4, space group C2/c, dcalc 1.623 g/cm3, µ(MoKα) 0.266 mm−1, F(000) 1568.
The unit cell parameters and intensities of 14,701 reflections (4596 independent reflections,
Rint 0.041) for
A, 15,594 reflections (4463 independent reflections,
Rint 0.072) for
B and 6611 reflections (2614 independent reflections,
Rint 0.085) for
C were measured on an Xcalibur-3 diffractometer (Oxford Diffraction Limited, Oxford, UK) using MoK
α radiation, a CCD detector, graphite monochromator, and ω-scanning to 2θ
max 60°. The structures were solved by the direct method using the SHELXTL program package (Institute of Inorganic Chemistry, Göttingen, Germany) [
13]. The positions of the hydrogen atoms were found from the electron density difference maps and refined using the “riding” model with
Uiso =
1.2Ueq for the nonhydrogen atom bonded to a given hydrogen atom. The hydrogen atoms involved in O–H…O and N–H…O hydrogen bond formation were refined using isotropic approximation. The structures were refined using
F2 full-matrix least-squares analysis in the anisotropic approximation for nonhydrogen atoms to
wR2 0.119 for 4577 reflections (
R1 0.045 for 3034 reflections with
F > 4σ (
F),
S = 0.965) for
A,
wR2 0.123 for 4451 reflections (
R1 0.051 for 3052 reflections with
F > 4σ (
F),
S = 0.966) for
B and
wR2 0.169 for 2599 reflections (
R1 0.070 for 1694 reflections with
F > 4σ (
F),
S = 0.979) for
C. The final atomic coordinates and the crystallographic data for the molecules of
N-(pyridin-2-yl)-4-methyl-2,2-dioxo-1
H-2λ
6,1-benzothiazine-3-carboxamide crystalline forms
A,
B and
C have been deposited with the Cambridge Crystallographic Data Centre, 12 Union Road, CB2 1EZ, UK (Fax: +44-1223-336033; e-mail:
deposit@ccdc.cam.ac.uk) and are available on request quoting the deposition numbers CCDC 1964379, and 1964377, respectively [
14,
15,
16].
2.4. Pharmacology
All biological experiments were carried out in full accord with the European Convention on the Protection of Vertebrate Animals Used for Experimental and Other Scientific Purposes and the Ukrainian Law No. 3447-IV “On protection of animals from severe treatment” [
20] (project ID 3410U14, approved 15 October 2015). The pharmacological research was carried out with the permission and under the supervision of the Commission on Bioethics (N.I. Pirogov Vinnitsa National Medical University, Vinnitsa, Ukraine).
All pharmacological experiments described in this article were performed on animals from the vivarium of the Institute of Pharmacology and Toxicology of the Academy of Medical Sciences of Ukraine (Kyiv, Ukraine). All animals received standard food for rodents and water. One day before the tests, the animals were transferred to a scientific laboratory for adaptation. At all times they were kept at 20–22 °C, a relative humidity of 40–60% and a 12 h light/12 h dark cycle.
The analgesic action was studied on white Wistar male rats weighing 19–22 g (10 animals for each test substance and way of administration) using the “acetic-acid-induced writhing” model [
21]. The nociceptive effect was reproduced by intraperitoneal injection of 0.6% aqueous solution of acetic acid in an amount of 0.1 mL per 10 g of animal weight in 30 min after the preliminary intraperitoneal or oral administration of the substances studied. Animals of the control group were intraperitoneally administered only diluted acetic acid in the specified concentration. After the introduction of acetic acid the animals were placed on a paper substrate and covered with a glass cap with a hole for free access of air. The number of “writhings” was counted from the 5th to the 20th min, inclusively. The analgesic effect was assessed by the ability of the test substances to reduce the number of “writhings” compared to the control and expressed as a percentage.
The analgesic activity was studied on white Wistar male rats weighing 160–190 g (10 animals for each test substance and way of administration) using the “hot plate” model [
21]. First, the experimental animals received the test samples intraperitoneally or orally or an equivalent amount of water and Tween-80 (control group). After an hour, the animals were placed on a metal surface surrounded by a cylinder heated to an average of 52 °C (Ugo Basile, Italy). The time from the moment of placement on a hot surface to the appearance of a behavioral response to the nociceptive stimulation was recorded: licking the hind legs (phase 1); jumping and/or the desire to jump out of the cylinder (phase 2). By summing up these indicators, the total duration of latent periods was determined. Their increase indicated the analgesic activity of the compounds studied compared to the control and was expressed as a percentage.
The antinociceptive effect was studied on white Wistar male rats weighing 160–190 g (10 animals for each test substance and way of administration) using the model of “thermal irritation of the tail tip” (tail-flick) [
21]. Thermal irritation was caused by a focused beam of light. First, the rats were placed in individual cells with their tails out, and the tail-flick analgesia meter (Ugo Basile, Italy) was used to determine the initial level of the latent period (the time of the attempt to get rid of the pain stimulus). Then, the substances studied were administered intraperitoneally or orally to the experimental animals; in an hour the experiment was repeated. The potency of the analgesic action was assessed by increasing the latency period compared to the baseline level taken as a control.
The analgesic activity was studied on white Wistar male rats weighing 160–190 g (seven animals for each test substance and way of administration) using the “tail electric stimulation” model [
21]. The rats were placed in cramped plexiglass cages with a copper plate floor that served as an electrode. Using an RS12 stimulator (TUR, Dresden, Germany), an electric stimulus was supplied in the form of sequences of constant current (rectangular pulses, 50 ms impulse width, 50 Hz, increment of 100 mA/s) on the lateral base of the tail through two needle electrodes made of stainless steel and located at a distance of 1 cm from each other. The minimum current strength that caused vocalization, immersion of the tail or paw flinches from the conductive surface of the floor was taken as the initial pain threshold. After that, the animals were treated with the substances studied (intraperitoneally or orally), and pain responses were measured again in 1, 2, 4 and 6 h after their administration. The analgesic effect was assessed by comparing the initial values of the pain threshold and its changes in 1, 2, 4 and 6 h after a single injection of the test substances.
The analgesic action was studied on white Wistar male rats weighing 180–200 g (seven animals for each test substance and way of administration) using the “neuropathic pain” model [
21,
22]. This model was reproduced under anesthesia by ligation of the sciatic nerve in the upper third of the thigh at the level of the popliteal fossa above the site of its trifurcation on nerve Tibialis, nerve Peroneus and nerve Suralis. The pathological process development lasted 14 days. The degree of hyperalgesia was determined using von Frey monofilaments from the Semmes Weinstein kit (Stoelting, Dale Wood, IL, USA) covering a wide range of pressure strength from 0.008 to 300 g, as well as a dolorimeter (Baseline, White Plains, NY, USA). To do this, we compared the pain threshold—the minimum pressure on the lower surface of the rat’s foot (g/mm
2) that caused a pain response (vocalization and/or paw flinches)—on the operated limb before surgery (baseline) and on the 14th day after surgery (pathology without treatment—control). The analgesic action of crystal forms
A,
B and
C was assessed 2 h after their single intraperitoneal or oral administration by determining changes in the pain threshold compared to the control.
All of the crystal forms (A, B and C), regardless of the experimental model and route of administration, were tested in the dose of 20 mg/kg in the form of aqueous suspensions stabilized with Tween-80.
Acute toxicity of
N-(4-trifluoromethylphenyl)-4-hydroxy-2,2-dioxo-1
H-2λ
6,1-benzothiazine-3-carboxamide crystal forms
A,
B and
C was studied on intact white mice weighing 18–22 g (seven animals with each dose). The substances studied were administered orally in the form of a thin aqueous suspension stabilized with Tween-80 (at high doses in 2–3 doses with an interval of 10 min). The number of surviving animals was recorded every 24 h for 14 days. Doses causing death in 16%, 50%, 84% and 100% of the test animals (LD
16, LD
50, LD
84 and LD
100, respectively) were calculated according to the Litchfield–Wilcoxon method [
23].
Gastric damage of crystal forms
A,
B and
C was studied on white Wistar male rats weighing 180–200 g (seven animals with each dose). Before the experiments, the animals were starved for 48 h, having access to drinking water ad libitum. During this time, they were housed in cages with raised bottoms of wide wire mesh in order to avoid coprophagy. The test compounds were administered orally through a gastric probe in the form of a thin aqueous suspension stabilized with Tween-80. Further studies of gastric damages were performed in 4 h according to the method described in detail in [
24]. Doses causing erosion in 16%, 50%, 84% and 100% of the test animals (UD
16, UD
50, UD
84 and UD
100, respectively) were calculated according to the Litchfield-Wilcoxon’s method [
23].
All results obtained during pharmacological tests were processed using the STATISTICA 6.1 software package (StatSoft Inc., Tulsa, OK, USA). Descriptive statistics included calculations of the arithmetic means (M) and standard errors of the mean (±m). The significance of differences within one group was assessed using the Wilcoxon nonparametric test. The reliability of intergroup differences was determined using the nonparametric Mann–Whitney U-criteria. The effects were considered statistically significant at p ≤ 0.05.