Survey about the Quality of Life of Italian Patients with Fabry Disease
Abstract
1. Introduction
2. Patients and Methods
3. Results of the Study
4. Disease-Related Problems
5. Concerns about the Future
6. Health-Related Quality of Life
7. Discussion and Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Germain, D.P. Fabry disease. Orphanet J. Rare Dis. 2010, 5, 30. [Google Scholar] [CrossRef] [PubMed]
- Eng, C.M.; Germain, D.P.; Banikazemi, M.; Warnock, D.G.; Wanner, C.; Hopkin, R.J.; Bultas, J.; Lee, P.; Sims, K.; Brodie, S.E.; et al. Fabry disease: Guidelines for the evaluation and management of multi-organ involvement. Genet. Med. 2006, 8, 539–548. [Google Scholar] [CrossRef]
- Arends, M.; Hollak, C.E.; Biegstraaten, M. Quality of life in patients with Fabry disease: A systematic review of the literature. Orphanet J. Rare Dis. 2015, 10, 77. [Google Scholar] [CrossRef] [PubMed]
- Gold, K.F.; Pastores, G.M.; Botteman, M.F.; Yeh, J.M.; Sweeney, S.; Aliski, W.; Pashos, C.L. Quality of life of patients with Fabry disease. Qual. Life Res. 2002, 11, 317–327. [Google Scholar] [CrossRef] [PubMed]
- Tuttolomondo, A.; Pecoraro, R.; Simonetta, I.; Miceli, S.; Arnao, V.; Licata, G.; Pinto, A. Neurological complications of Anderson-Fabry disease. Curr. Pharm. Des. 2013, 19, 6014–6030. [Google Scholar] [CrossRef] [PubMed]
- Ali, N.; Gillespie, S.; Laney, D. Treatment of depression in adults with Fabry disease. JIMD Rep. 2018, 38, 13–21. [Google Scholar]
- Körver, S.; Geurtsen, G.J.; Hollak, C.E.M.; van Schaik, I.N.; Longo, M.G.F.; Lima, M.R.; Vedolin, L.; Dijkgraaf, M.G.W.; Langeveld, M. Depressive symptoms in Fabry disease: The importance of coping, subjective health perception and pain. Orphanet J. Rare Dis. 2020, 15, 28. [Google Scholar] [CrossRef]
- Bolsover, F.E.; Murphy, E.; Cipolotti, L.; Werring, D.J.; Lachmann, R.H. Cognitive dysfunction and depression in Fabry disease: A systematic review. J. Inherit. Metab. Dis. 2014, 37, 177–187. [Google Scholar] [CrossRef]
- Lelieveld, I.M.; Böttcher, A.; Hennermann, J.B.; Beck, M.; Fellgiebel, A. Eight-year follow-up of neuropsychiatric symptoms and brain structural changes in Fabry disease. PLoS ONE 2015, 10, e0137603. [Google Scholar] [CrossRef]
- Cole, A.L.; Lee, P.J.; Hughes, D.A.; Deegan, P.B.; Waldek, S.; Lachmann, R.H. Depression in adults with Fabry disease: A common and under-diagnosed problem. J. Inherit. Metab. Dis. 2007, 30, 943–951. [Google Scholar] [CrossRef]
- Wang, R.Y.; Lelis, A.; Mirocha, J.; Wilcox, W.R. Heterozygous Fabry women are not just carriers, but have a significant burden of disease and impaired quality of life. Genet. Med. 2007, 9, 34–45. [Google Scholar] [CrossRef]
- Sigmundsdottir, L.; Tchan, M.C.; Knopman, A.A.; Menzies, G.C.; Batchelor, J.; Sillence, D.O. Cognitive and psychological functioning in Fabry disease. Arch. Clin. Neuropsychol. 2014, 29, 642–650. [Google Scholar] [CrossRef] [PubMed]
- Scalone, L.; Cortesi, P.A.; Ciampichini, R.; Belisari, A.; D’Angiolella, L.S.; Cesana, G.; Mantovani, L.G. Italian population-based values of EQ-5D health states. Value Health 2013, 16, 814–822. [Google Scholar] [CrossRef]
- Rabin, R.; de Charro, F. EQ-5D: A measure of health status from the EuroQol Group. Ann. Med. 2001, 33, 337–343. [Google Scholar] [CrossRef]
- Scalone, L.; Fagiuoli, S.; Ciampichini, R.; Gardini, I.; Bruno, R.; Pasulo, L.; Lucà, M.G.; Fusco, F.; Gaeta, L.; Del Prete, A.; et al. The societal burden of chronic liver diseases: Results from the COME study. BMJ Open Gastroenterol. 2015, 2, e000025. [Google Scholar] [CrossRef] [PubMed]
- Dolan, P.; Gudex, C.; Kind, P.; Williams, A. A Social Tariff for EuroQol: Results from a UK General Population Survey—Discussion Paper 138. 1995. Available online: https://www.york.ac.uk/che/pdf/DP138.pdf (accessed on 9 October 2021).
- Hoffmann, B.; Garcia de Lorenzo, A.; Mehta, A.; Beck, M.; Widmer, U.; Ricci, R.; FOS European Investigators. Effects of enzyme replacement therapy on pain and health related quality of life in patients with Fabry disease: Data from FOS (Fabry Outcome Survey). J. Med. Genet. 2005, 42, 247–252. [Google Scholar] [CrossRef]
- Mozzi, A.; Meregaglia, M.; Lazzaro, C.; Tornatore, V.; Belfiglio, M.; Fattore, G. A comparison of EuroQol 5-Dimension health-related utilities using Italian, UK, and US preference weights in a patient sample. Clinicoecon. Outcomes Res. 2016, 8, 267–274. [Google Scholar] [CrossRef] [PubMed]
- Battaglia, M.; Kobelt, G.; Ponzio, M.; Berg, J.; Capsa, D.; Dalén, J.; European Multiple Sclerosis Platform. New insights into the burden and costs of multiple sclerosis in Europe: Results for Italy. Mult. Scler. 2017, 23, 104–116. [Google Scholar] [CrossRef] [PubMed]
- Devlin, N.J.; Parkin, D.; Browne, J. Patient-reported outcome measures in the NHS: New methods for analysing and reporting EQ-5D data. Health Econ. 2010, 19, 886–905. [Google Scholar] [CrossRef]
- Drummond, M.F.; Sculpher, M.J.; Torrance, G.W.; O’Brien, B.J.; Stoddart, G.L. Methods for the Economic Evaluation of Health Care Programme, 3rd ed.; Oxford University Press: Oxford, UK, 2005. [Google Scholar]
- Hays, R.D.; Reeve, B.B. Measurement and Modeling of Health-Related Quality of Life; San Diego Academic Press: San Diego, CA, USA, 2008; Volume 4, pp. 241–252. [Google Scholar]
- Platt, F.M.; Boland, B.; van der Spoel, A.C. The cell biology of disease: Lysosomal storage disorders: The cellular impact of lysosomal dysfunction. J. Cell Biol. 2012, 199, 723–734. [Google Scholar] [CrossRef]
- Mikosch, P.; Hughes, D. An overview on bone manifestations in Gaucher disease. Wien Med. Wochenschr. 2010, 160, 609–624. [Google Scholar] [CrossRef]
- Rigante, D.; Segni, G. Cardiac structural involvement in mucopolysaccharidoses. Cardiology 2002, 98, 18–20. [Google Scholar] [CrossRef]
- Giugliani, R.; Harmatz, P.; Lin, S.P.; Scarpa, M. Assessing the impact of the five senses on quality of life in mucopolysaccharidoses. Orphanet J. Rare Dis. 2020, 15, 97. [Google Scholar] [CrossRef]
- Ramaswami, U.; Stull, D.E.; Parini, R.; Pintos-Morell, G.; Whybra, C.; Kalkum, G.; Rohrbach, M.; Raluy-Callado, M.; Beck, M.; Chen, W.H.; et al. Measuring patient experiences in Fabry disease: Validation of the Fabry-specific Pediatric Health and Pain Questionnaire (FPHPQ). Health Qual. Life Outcomes 2012, 10, 116. [Google Scholar] [CrossRef] [PubMed]
- Beck, M.; Hughes, D.; Kampmann, C.; Pintos-Morell, G.; Ramaswami, U.; West, M.L.; Giugliani, R. Long-term outcomes with agalsidase alfa enzyme replacement therapy: Analysis using deconstructed composite events. Mol. Genet. Metab. Rep. 2017, 14, 31–35. [Google Scholar] [CrossRef]
- Bähner, F.; Kampmann, C.; Whybra, C.; Miebach, E.; Wiethoff, C.M.; Beck, M. Enzyme replacement therapy in heterozygous females with Fabry disease: Results of a phase IIIB study. J. Inherit. Metab. Dis. 2003, 26, 617–627. [Google Scholar] [CrossRef] [PubMed]
- Petta, S.; Cabibbo, G.; Enea, M.; Macaluso, F.S.; Plaia, A.; Bruno, R.; Gasbarrini, A.; Craxì, A.; Cammà, C.; WEF Study Group. Cost-effectiveness of sofosbuvir-based triple therapy for untreated patients with genotype 1 chronic hepatitis C. Hepatology 2014, 59, 1692–1705. [Google Scholar] [CrossRef] [PubMed]
- Patti, F.; Amato, M.P.; Trojano, M.; Solaro, C.; Pappalardo, A.; Zipoli, V.; Portaccio, E.; Paolicelli, D.; Paolillo, A.; Mennini, F.S.; et al. Multiple sclerosis in Italy: Cost-of-illness study. Neurol. Sci. 2011, 32, 787–794. [Google Scholar] [CrossRef] [PubMed]
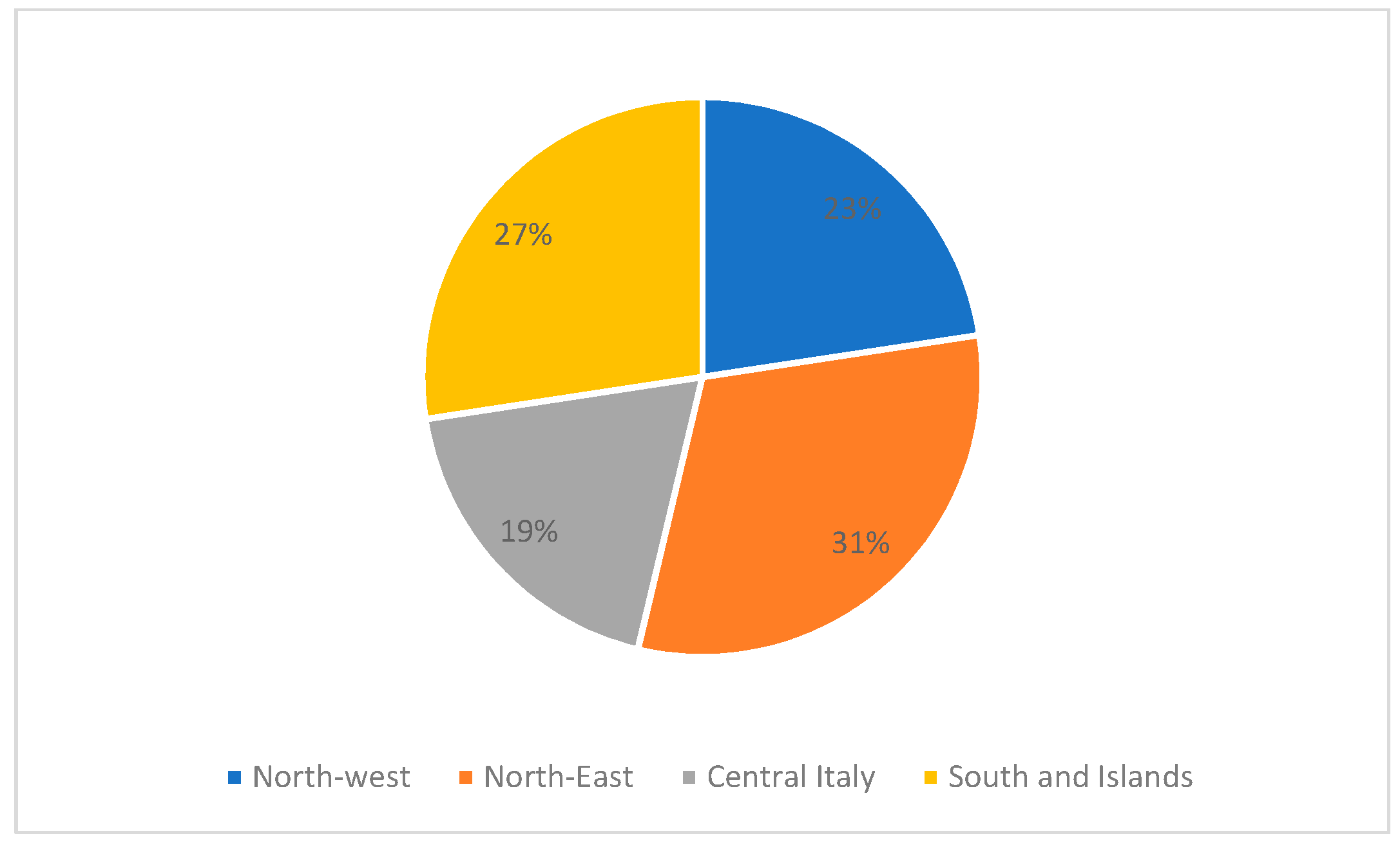
| Variable | M + F | M | F |
|---|---|---|---|
| Sample | 106 | 43 | 63 |
| Average age (years) | 42 | 37 | 45 |
| Minimum age (years) | 5 | 7 | 5 |
| Maximum age (years) | 77 | 67 | 77 |
| Average age at diagnosis (years) | 32 | 27 | 35 |
| % of females | 59.5% | ||
| Status | |||
| Married | 54.7% | 44.2% | 61.9% |
| Unmarried | 33.0% | 48.8% | 22.2% |
| Divorced | 8.5% | 7.0% | 9.5% |
| Separated | 3.8% | 0.0% | 6.3% |
| Widow/widower | 54.7% | 44.2% | 61.9% |
| School Level and Qualifications | |||
| Degree | 20.8% | 18.6% | 22.2% |
| High school diploma | 44.3% | 48.8% | 41.3% |
| 2–3-year diploma | 5.7% | 4.7% | 6.3% |
| Middle school diploma | 20.8% | 18.6% | 22.2% |
| Primary school diploma | 6.6% | 77.0% | 6.3% |
| No diploma or degree | 1.9% | 2.3% | 1.6% |
| Professional Status | |||
| Employed | 53.8% | 60.5% | 49.2% |
| In search of his/her first job | 0.0% | 0.0% | 0.0% |
| Unemployed | 9.4% | 11.6% | 7.9% |
| Retired | 8.5% | 4.7 | 11.1 |
| Student | 11.3% | 14.0 | 9.5 |
| Housewife | 9.4% | 0.0% | 15.9% |
| Unable to work | 1.9% | 4.7% | 0.0% |
| Other | 5.7% | 4.7% | 6.3% |
| Mobility | |
| I have no problems in walking about. | 70.75% |
| I have some problems in walking about. | 28.30% |
| I am confined to bed. | 0.94% |
| Self-Care | |
| I have no problems with self-care. | 91.51% |
| I have some problems washing or dressing myself. | 7.55% |
| I am unable to wash or dress myself. | 0.94% |
| Usual Activities | |
| I have no problems with performing my usual activities. | 52.83% |
| I have some problems with performing my usual activities. | 45.28% |
| I am unable to perform my usual activities. | 1.89% |
| Pain/Discomfort | |
| I have no pain or discomfort. | 22.64% |
| I have moderate pain or discomfort. | 68.87% |
| I have extreme pain or discomfort. | 8.49% |
| Anxiety/Depression | |
| I am not anxious or depressed. | 42.45% |
| I am moderately anxious or depressed. | 50.94% |
| I am extremely anxious or depressed. | 6.60% |
| The Psychological Impact of the Disease (% of Patients) | ||||||||||||
| Loneliness and Isolation | Decision to Have Children | Anxiety | ||||||||||
| Not at all | Little | Quite | Much | Not at all | Little | Quite | Much | Not at all | Little | Quite | Much | |
| Total | 45.3 | 33.0 | 17.9 | 3.8 | 44.3 | 15.1 | 17.9 | 22.6 | 21.7 | 32.1 | 33.0 | 13.2 |
| Gender * | ||||||||||||
| Male | 51.2 | 30.2 | 16.3 | 2.3 | 39.5 | 16.3 | 23.3 | 20.9 | 27.9 | 32.6 | 32.6 | 7.0 |
| Female | 41.3 | 34.9 | 19.0 | 4.8 | 47.6 | 14.3 | 14.3 | 23.8 | 17.5 | 31.7 | 33.3 | 17.5 |
| Age (years) | ||||||||||||
| 18–29 | 55.6 | 33.3 | 11.1 | 0.0 | 11.1 | 11.1 | 55.6 | 22.2 | 22.2 | 11.1 | 55.6 | 11.1 |
| 30–39 | 45.5 | 40.9 | 13.6 | 0.0 | 9.1 | 27.3 | 27.3 | 36.4 | 22.7 | 31.8 | 31.8 | 13.6 |
| 40–49 | 44.0 | 32.0 | 20.0 | 4.0 | 48.0 | 12.0 | 12.0 | 28.0 | 16.0 | 36.0 | 24.0 | 24.0 |
| 50–59 | 40.9 | 36.4 | 18.2 | 4.5 | 50.0 | 27.3 | 4.5 | 18.2 | 9.1 | 45.5 | 45.5 | 0.0 |
| 60–69 | 25.0 | 41.7 | 25.0 | 8.3 | 83.3 | 0.0 | 0.0 | 16.7 | 25.0 | 25.0 | 33.3 | 16.7 |
| 70+ | 0.0 | 0.0 | 100.0 | 0.0 | 66.7 | 0.0 | 33.3 | 0.0 | 33.3 | 0.0 | 66.7 | 0.0 |
| Depression | Relational Problems | Fear of Other People’s Judgement | ||||||||||
| Not at all | Little | Quite | Much | Not at all | Few | Quite | Many | Not at all | Little | Quite | Much | |
| Total | 37.7 | 39.6 | 18.9 | 3.8 | 57.5 | 30.2 | 7.5 | 4.7 | 50.9 | 24.5 | 18.9 | 5.7 |
| Gender | ||||||||||||
| Male | 39.5 | 39.5 | 18.6 | 2.3 | 53.5 | 30.2 | 9.3 | 7.0 | 41.9 | 25.6 | 20.9 | 11.6 |
| Female | 36.5 | 39.7 | 19.0 | 4.8 | 60.3 | 30.2 | 6.3 | 3.2 | 57.1 | 23.8 | 17.5 | 1.6 |
| Age (years) | ||||||||||||
| 18–29 | 44.4 | 44.4 | 11.1 | 0.0 | 55.6 | 44.4 | 0.0 | 0.0 | 33.3 | 44.4 | 11.1 | 11.1 |
| 30–39 | 36.4 | 45.5 | 13.6 | 4.5 | 63.6 | 31.8 | 4.5 | 0.0 | 45.5 | 31.8 | 18.2 | 4.5 |
| 40–49 | 32.0 | 44.0 | 20.0 | 4.0 | 48.0 | 40.0 | 8.0 | 4.0 | 52.0 | 28.0 | 16.0 | 4.0 |
| 50–59 | 18.2 | 54.5 | 27.3 | 0.0 | 63.6 | 13.6 | 13.6 | 9.1 | 68.2 | 9.1 | 18.2 | 4.5 |
| 60–69 | 50.0 | 25.0 | 16.7 | 8.3 | 50.0 | 41.7 | 0.0 | 8.3 | 50.0 | 25.0 | 25.0 | 0.0 |
| 70+ | 33.3 | 0.0 | 66.7 | 0.0 | 33.3 | 0.0 | 66.7 | 0.0 | 33.3 | 0.0 | 66.7 | 0.0 |
| Disease-related Problems (% Patients) | ||||||||||||
| Chronic/Persistent Pain | Acute/Temporary Pain | Fatigue | ||||||||||
| Not at all | Little | Quite | Much | Not at all | Little | Quite | Much | Not at all | Little | Quite | Much | |
| Total | 27.4 | 33.0 | 30.2 | 9.4 | 20.8 | 32.1 | 38.7 | 8.5 | 7.5 | 29.2 | 40.6 | 22.6 |
| Gender | ||||||||||||
| Male | 27.9 | 32.6 | 25.6 | 14.0 | 11.6 | 32.6 | 41.9 | 14.0 | 14.0 | 27.9 | 37.2 | 20.9 |
| Female | 27.0 | 33.3 | 33.3 | 6.3 | 27.0 | 31.7 | 36.5 | 4.8 | 3.2 | 30.2 | 42.9 | 23.8 |
| Age (years) | ||||||||||||
| 18–29 | 33.3 | 44.4 | 11.1 | 11.1 | 33.3 | 11.1 | 44.4 | 11.1 | 22.2 | 22.2 | 33.3 | 22.2 |
| 30–39 | 36.4 | 27.3 | 22.7 | 13.6 | 27.3 | 18.2 | 50.0 | 4.5 | 4.5 | 45.5 | 31.8 | 18.2 |
| 40–49 | 12.0 | 28.0 | 44.0 | 16.0 | 8.0 | 48.0 | 28.0 | 16.0 | 0.0 | 24.0 | 56.0 | 20.0 |
| 50–59 | 22.7 | 45.5 | 31.8 | 0.0 | 22.7 | 31.8 | 40.9 | 4.5 | 4.5 | 13.6 | 45.5 | 36.4 |
| 60–69 | 25.0 | 25.0 | 41.7 | 8.3 | 25.0 | 33.3 | 33.3 | 8.3 | 8.3 | 16.7 | 50.0 | 25.0 |
| 70+ | 0.0 | 33.3 | 66.7 | 0.0 | 33.3 | 33.3 | 33.3 | 0.0 | 0.0 | 0.0 | 66.7 | 33.3 |
| Motor Difficulties | ||||||||||||
| Not at all | Few | Quite | Many | |||||||||
| Total | 40.6 | 32.1 | 20.8 | 6.6 | ||||||||
| Gender | ||||||||||||
| Male | 37.2 | 32.6 | 23.3 | 7.0 | ||||||||
| Female | 42.9 | 31.7 | 19.0 | 6.3 | ||||||||
| Age (years) | ||||||||||||
| 18–29 | 55.6 | 33.3 | 11.1 | 0.0 | ||||||||
| 30–39 | 50.0 | 31.8 | 13.6 | 4.5 | ||||||||
| 40–49 | 28.0 | 40.0 | 32.0 | 0.0 | ||||||||
| 50–59 | 36.4 | 22.7 | 31.8 | 9.1 | ||||||||
| 60–69 | 41.7 | 33.3 | 8.3 | 16.7 | ||||||||
| 70+ | 0.0 | 0.0 | 66.7 | 33.3 | ||||||||
| Concerns About the Future (% Patients) | ||||||||||||
| Increasing Severity of Symptoms | Physical Disability | Physical Pain | ||||||||||
| Not at all | Little | Quite | Much | Not at all | Little | Quite | Much | Not at all | Little | Quite | Much | |
| Total | 5.7 | 19.8 | 50.0 | 24.5 | 12.3 | 18.9 | 45.3 | 23.6 | 6.6 | 30.2 | 42.5 | 20.8 |
| Gender | ||||||||||||
| Male | 4.7 | 14.0 | 53.5 | 27.9 | 14.0 | 11.6 | 48.8 | 25.6 | 7.0 | 30.2 | 39.5 | 23.3 |
| Female | 6.3 | 23.8 | 47.6 | 22.2 | 11.1 | 23.8 | 42.9 | 22.2 | 6.3 | 30.2 | 44.4 | 19.0 |
| Age (years) | ||||||||||||
| 18–29 | 11.1 | 11.1 | 44.4 | 33.3 | 33.3 | 11.1 | 44.4 | 11.1 | 11.1 | 22.2 | 44.4 | 22.2 |
| 30–39 | 4.5 | 22.7 | 40.9 | 31.8 | 4.5 | 27.3 | 40.9 | 27.3 | 0.0 | 40.9 | 31.8 | 27.3 |
| 40–49 | 4.0 | 24.0 | 64.0 | 8.0 | 8.0 | 20.0 | 52.0 | 20.0 | 8.0 | 24.0 | 60.0 | 8.0 |
| 50–59 | 4.5 | 18.2 | 50.0 | 27.3 | 13.6 | 18.2 | 40.9 | 27.3 | 4.5 | 36.4 | 31.8 | 27.3 |
| 60–69 | 8.3 | 16.7 | 41.7 | 33.3 | 8.3 | 16.7 | 50.0 | 25.0 | 16.7 | 8.3 | 50.0 | 25.0 |
| 70+ | 0.0 | 0.0 | 66.7 | 33.3 | 0.0 | 0.0 | 66.7 | 33.3 | 0.,0 | 0.0 | 66.7 | 33.3 |
| Impairment of Vital Organs | Possibility of Having to Give up/not Finding a Job | |||||||||||
| Not at all | Little | Quite | Much | Not at all | Little | Quite | Much | |||||
| Total | 4.7 | 11.3 | 48.1 | 35.8 | 24.5 | 22.6 | 30.2 | 22.6 | ||||
| Gender | ||||||||||||
| Male | 7.0 | 4.7 | 41.9 | 46.5 | 16.3 | 25.6 | 18.6 | 39.5 | ||||
| Female | 3.2 | 15.9 | 52.4 | 28.6 | 30.2 | 20.6 | 38.1 | 11.1 | ||||
| Age (years) | ||||||||||||
| 18–29 | 11.1 | 0.0 | 33.3 | 55.6 | 11.1 | 33.3 | 22.2 | 33.3 | ||||
| 30–39 | 0.0 | 4.5 | 45.5 | 50.0 | 0.0 | 31.8 | 36.4 | 31.8 | ||||
| 40–49 | 0.0 | 12.0 | 64.0 | 24.0 | 16.0 | 20.0 | 44.0 | 20.0 | ||||
| 50–59 | 0.0 | 27.3 | 45.5 | 27.3 | 31.8 | 27.3 | 22.7 | 18.2 | ||||
| 60–69 | 8.3 | 8.3 | 50.0 | 33.3 | 66.7 | 8.3 | 16.7 | 8.3 | ||||
| 70+ | 0.0 | 0.0 | 66.7 | 33.3 | 66.7 | 0.0 | 33.3 | 0.0 | ||||
| Italian Tariffs | UK Tariffs | |
|---|---|---|
| Total | 0.72 | 0.68 |
| Gender | ||
| Male | 0.72 | 0.68 |
| Female | 0.71 | 0.68 |
| Age | ||
| 18–29 | 0.86 | 0.77 |
| 30–39 | 0.82 | 0.74 |
| 40–49 | 0.79 | 0.63 |
| 50–59 | 0.77 | 0.64 |
| 60–69 | 0.64 | 0.64 |
| 70+ | 0.69 | 0.40 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Polistena, B.; Rigante, D.; Sicignano, L.L.; Verrecchia, E.; Manna, R.; d’Angela, D.; Spandonaro, F. Survey about the Quality of Life of Italian Patients with Fabry Disease. Diseases 2021, 9, 72. https://doi.org/10.3390/diseases9040072
Polistena B, Rigante D, Sicignano LL, Verrecchia E, Manna R, d’Angela D, Spandonaro F. Survey about the Quality of Life of Italian Patients with Fabry Disease. Diseases. 2021; 9(4):72. https://doi.org/10.3390/diseases9040072
Chicago/Turabian StylePolistena, Barbara, Donato Rigante, Ludovico Luca Sicignano, Elena Verrecchia, Raffaele Manna, Daniela d’Angela, and Federico Spandonaro. 2021. "Survey about the Quality of Life of Italian Patients with Fabry Disease" Diseases 9, no. 4: 72. https://doi.org/10.3390/diseases9040072
APA StylePolistena, B., Rigante, D., Sicignano, L. L., Verrecchia, E., Manna, R., d’Angela, D., & Spandonaro, F. (2021). Survey about the Quality of Life of Italian Patients with Fabry Disease. Diseases, 9(4), 72. https://doi.org/10.3390/diseases9040072






