A Novel Approach to Assessing Infant and Child Mental Rotation
Abstract
1. Introduction
2. Materials and Methods
2.1. Participants
2.2. Stimuli
2.3. Apparatus
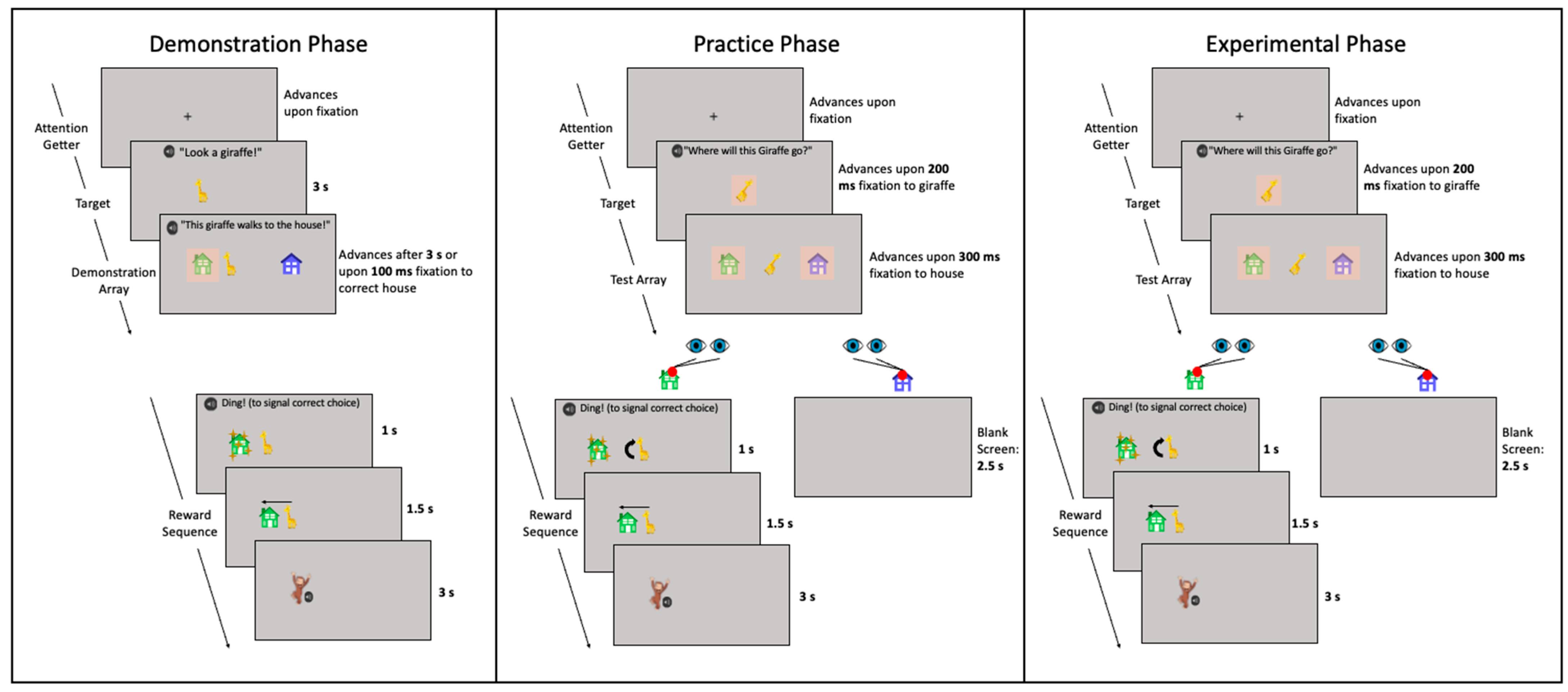
2.4. Procedure
2.4.1. Demonstration Phase
2.4.2. Practice Phase
2.4.3. Experimental Phase
2.4.4. Data Processing and Analytic Approach
3. Results
3.1. Task Engagement and Comprehension
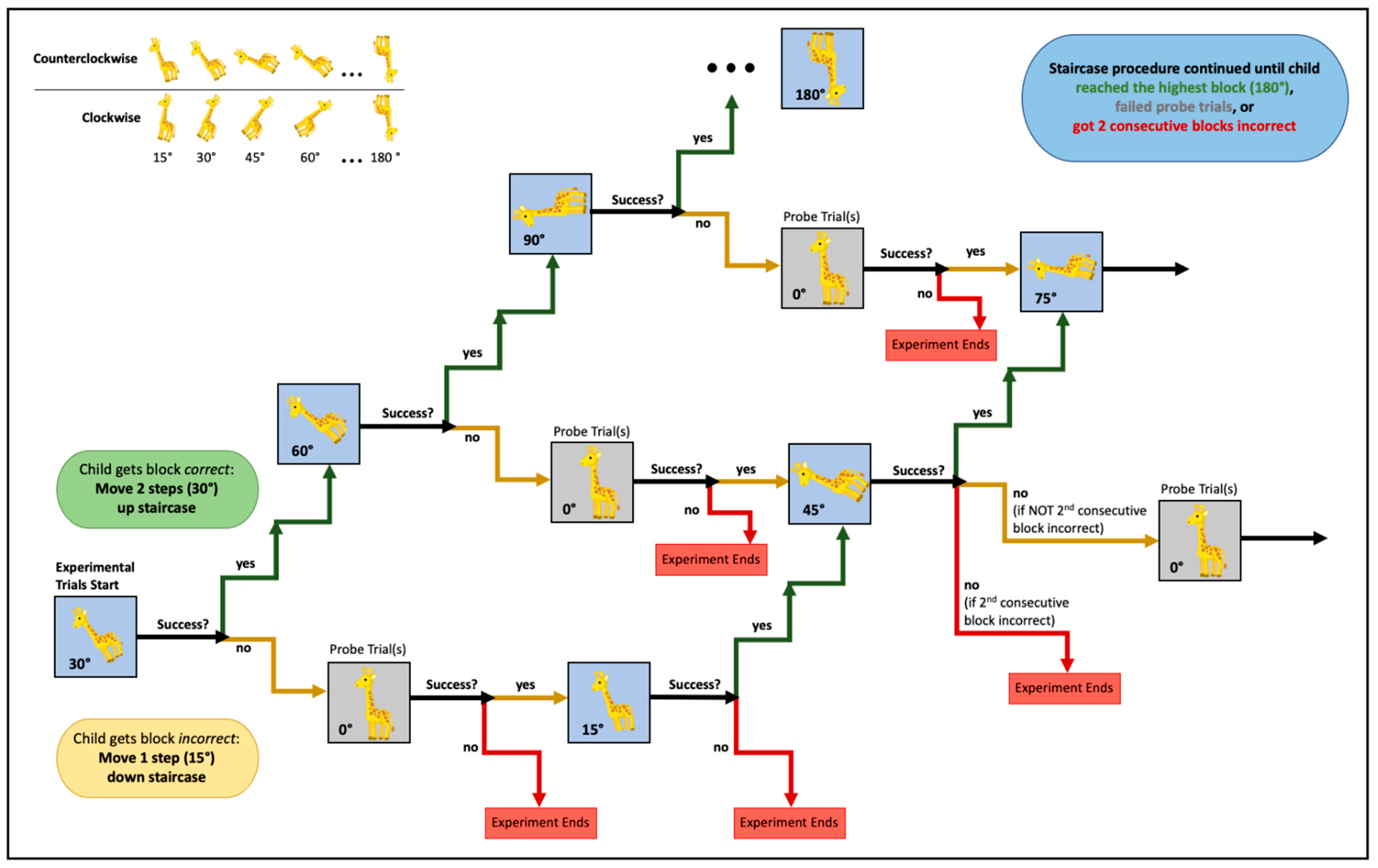
3.2. Staircase Analysis
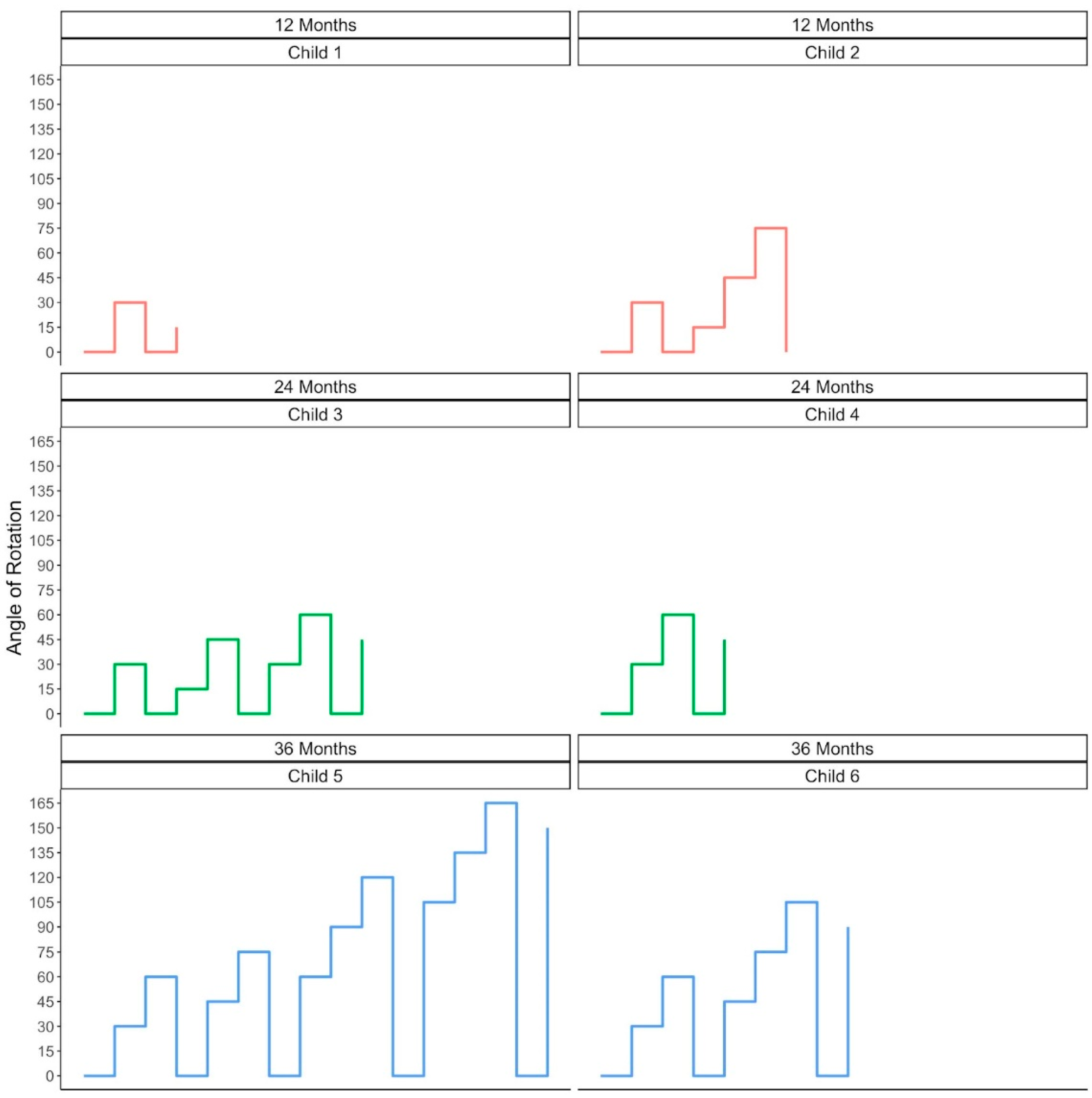
3.3. Exploring Variation in Children’s Pathways to Achieving Their Max Angle
4. Discussion
Limitations
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
| 1 | Angular disparity effects can be examined between-subject in infant paradigms, but such manipulations are challenging to implement and we are only aware of one study that examined angular disparity effects in infants (Gerhard and Schwarzer 2018). |
References
- Aguilar Ramirez, Daniela E., Jarrod Blinch, and Claudia L. R. Gonzalez. 2021. One brick at a time: Building a developmental profile of spatial abilities. Developmental Psychobiology 63: e22155. [Google Scholar] [CrossRef]
- Beckner, Aaron G., Annika T. Voss, Lindsey Phillips, Kathryn King, Marianella Casasola, and Lisa M. Oakes. 2023. An investigation of mental rotation in infancy using change detection. Infant Behavior and Development 71: 101834. [Google Scholar] [CrossRef]
- Bradski, Gary. 2000. The openCV library. Dr. Dobb’s Journal: Software Tools for the Professional Programmer 25: 120–23. [Google Scholar]
- Breslow, Norman E. 1975. Analysis of Survival Data under the Proportional Hazards Model. International Statistical Review 43: 45–57. [Google Scholar] [CrossRef]
- Brooks, Rechele, and Andrew N. Meltzoff. 2005. The development of gaze following and its relation to language. Developmental Science 8: 535–43. [Google Scholar] [CrossRef]
- Casey, Beth M., Elizabeth Pezaris, Bonnie Fineman, Amanda Pollock, Lindsay Demers, and Eric Dearing. 2015. A longitudinal analysis of early spatial skills compared to arithmetic and verbal skills as predictors of fifth-grade girls’ math reasoning. Learning and Individual Differences 40: 90–100. [Google Scholar] [CrossRef]
- Cheng, Yi-Ling, and Kelly S. Mix. 2014. Spatial Training Improves Children’s Mathematics Ability. Journal of Cognition and Development: Official Journal of the Cognitive Development Society 15: 2–11. [Google Scholar] [CrossRef]
- Christodoulou, Joan, Scott P. Johnson, Dawn M. Moore, and David S. Moore. 2016. Seeing double: 5-month-olds’ mental rotation of dynamic, 3D block stimuli presented on dual monitors. Infant Behavior & Development 45: 64–70. [Google Scholar]
- Constantinescu, Mihaela, David S. Moore, Scott P. Johnson, and Melissa Hines. 2018. Early contributions to infants’ mental rotation abilities. Developmental Science 21: e12613. [Google Scholar] [CrossRef]
- Cooper, Lynn A., and Roger N. Shepard. 1973. Chronometric studies of the rotation of mental images. In Visual Information Processing. Edited by W. G. Chase. Oxford: Academic Press, pp. 75–176. [Google Scholar]
- Ebersbach, Mirjam, and Christian Nawroth. 2018. Shape but not color facilitates two-year-Olds’ search performance in a spatial rotation task. Infancy: The Official Journal of the International Society on Infant Studies 23: 820–32. [Google Scholar] [CrossRef]
- Erdmann, Kathrin, Michael Kavšek, and Martin Heil. 2018. Infants’ looking times in a dynamic mental rotation task: Clarifying inconsistent results. Cognitive Development 48: 279–85. [Google Scholar] [CrossRef]
- Estes, David. 1998. Young children’s awareness of their mental activity: The case of mental rotation. Child Development 69: 1345–60. [Google Scholar] [CrossRef]
- Farroni, Teresa, Stefano Massaccesi, Donatella Pividori, and Mark H. Johnson. 2004. Gaze Following in Newborns. Infancy 5: 39–60. [Google Scholar] [CrossRef]
- Frick, Andrea. 2019. Spatial transformation abilities and their relation to later mathematics performance. Psychological Research 83: 1465–84. [Google Scholar] [CrossRef]
- Frick, Andrea, and Wenke Möhring. 2013. Child Mental object rotation and motor development in 8- and 10-month-old infants. Journal of Experimental Child Psychology 115: 708–20. [Google Scholar] [CrossRef]
- Frick, Andrea, Melissa A. Hansen, and Nora S. Newcombe. 2013. Development of mental rotation in 3- to 5-year-old children. Cognitive Development 28: 386–99. [Google Scholar] [CrossRef]
- Geer, Elyssa A., Jamie M. Quinn, and Colleen M. Ganley. 2019. Relations between spatial skills and math performance in elementary school children: A longitudinal investigation. Developmental Psychology 55: 637–52. [Google Scholar] [CrossRef]
- Gerhard, Theresa M., and Gudrun Schwarzer. 2018. Impact of rotation angle on crawling and non-crawling 9-month-old infants’ mental rotation ability. Journal of Experimental Child Psychology 170: 45–56. [Google Scholar] [CrossRef]
- Gilligan, Katie A., Eirini Flouri, and Emily K. Farran. 2017. The contribution of spatial ability to mathematics achievement in middle childhood. Journal of Experimental Child Psychology 163: 107–25. [Google Scholar]
- Gredebäck, Gustaf, Kim Astor, and Christine Fawcett. 2018. Gaze Following Is Not Dependent on Ostensive Cues: A Critical Test of Natural Pedagogy. Child Development 89: 2091–98. [Google Scholar] [CrossRef]
- Hawes, Zachary, Jo-Anne LeFevre, Chang Xu, and Catherine D. Bruce. 2015. Mental rotation with tangible three-dimensional objects: A new measure sensitive to developmental differences in 4- to 8-year-old children. Mind, Brain and Education: The Official Journal of the International Mind, Brain, and Education Society 9: 10–18. [Google Scholar] [CrossRef]
- Hegarty, Mary, and Maria Kozhevnikov. 1999. Types of visual–spatial representations and mathematical problem solving. Journal of Educational Psychology 91: 684–89. [Google Scholar] [CrossRef]
- Hespos, Susan J., and Philippe Rochat. 1996. Tracking invisible spatial transformations by 4- and 6-month-old infants. Infant Behavior & Development 19: 504. [Google Scholar]
- Hunter, John D. 2007. Matplotlib: A 2D Graphics Environment. Computing in Science & Engineering 9: 90–95. [Google Scholar]
- Jansen, Petra, Jennifer Lehmann, and Jessica Van Doren. 2012. Mental rotation performance in male soccer players. PLoS ONE 7: e48620. [Google Scholar] [CrossRef]
- Johnson, Wendy, and Thomas J. Bouchard, Jr. 2005. The structure of human intelligence: It is verbal, perceptual, and image rotation (VPR), not fluid and crystallized. Intelligence 33: 393–416. [Google Scholar]
- Kail, Robert. 1986. The impact of extended practice on rate of mental rotation. Journal of Experimental Child Psychology 42: 378–91. [Google Scholar] [CrossRef]
- Kaplan, Edward L., and Paul Meier. 1958. Nonparametric Estimation from Incomplete Observations. Journal of the American Statistical Association 53: 457–81. [Google Scholar] [CrossRef]
- Krause, Daniel, Benjamin Richert, and Matthias Weigelt. 2021. Neurophysiology of embodied mental rotation: Event-related potentials in a mental rotation task with human bodies as compared to alphanumeric stimuli. The European Journal of Neuroscience 54: 5384–403. [Google Scholar] [CrossRef]
- Krüger, Markus, Marlen Kaiser, Kristin Mahler, Wolfgang Bartels, and Horst Krist. 2014. Analogue mental transformations in 3-year-Olds: Introducing a new mental rotation paradigm suitable for young children. Infant and Child Development 23: 123–38. [Google Scholar] [CrossRef]
- Lauer, Jillian E., and Stella F. Lourenco. 2016. Spatial Processing in Infancy Predicts Both Spatial and Mathematical Aptitude in Childhood. Psychological Science 27: 1291–98. [Google Scholar] [CrossRef]
- Lauer, Jillian E., Hallie B. Udelson, Sung O. Jeon, and Stella F. Lourenco. 2015. An early sex difference in the relation between mental rotation and object preference. Frontiers in Psychology 6: 558. [Google Scholar] [CrossRef]
- Lee, Kang, Michelle Eskritt, Lawrence A. Symons, and Darwin Muir. 1998. Children’s use of triadic eye gaze information for “mind reading”. Developmental Psychology 34: 525–39. [Google Scholar] [CrossRef][Green Version]
- Levine, Susan C., Janellen Huttenlocher, Amy Taylor, and Adela Langrock. 1999. Early sex differences in spatial skill. Developmental Psychology 35: 940–49. [Google Scholar] [CrossRef] [PubMed]
- Marmor, Gloria Strauss. 1975. Development of kinetic images: When does the child first represent movement in mental images? Cognitive Psychology 7: 548–59. [Google Scholar] [CrossRef]
- Marmor, Gloria S. 1977. Mental rotation and number conservation: Are they related? Developmental Psychology 13: 320–25. [Google Scholar] [CrossRef]
- Mix, Kelly S., Susan C. Levine, Yi-Ling Cheng, Chris Young, D. Zachary Hambrick, Raedy Ping, and Spyros Konstantopoulos. 2016. Separate but correlated: The latent structure of space and mathematics across development. Journal of Experimental Psychology General 145: 1206–27. [Google Scholar] [CrossRef]
- Möhring, Wenke, and Andrea Frick. 2013. Touching up mental rotation: Effects of manual experience on 6-month-old infants’ mental object rotation. Child Development 84: 1554–65. [Google Scholar] [CrossRef] [PubMed]
- Möhring, Wenke, Andrew D. Ribner, Robin Segerer, Melissa E. Libertus, Tobias Kahl, Larissa Maria Troesch, and Alexander Grob. 2021. Developmental trajectories of children’s spatial skills: Influencing variables and associations with later mathematical thinking. Learning and Instruction 75: 101515. [Google Scholar] [CrossRef]
- Moore, David S., and Scott P. Johnson. 2008. Mental rotation in human infants: A sex difference. Psychological Science 19: 1063–66. [Google Scholar] [CrossRef]
- Moore, David S., and Scott P. Johnson. 2011. Mental rotation of dynamic, three-dimensional stimuli by 3-month-old infants. Infancy: The Official Journal of the International Society on Infant Studies 16: 435–45. [Google Scholar] [CrossRef]
- Moore, David S., and Scott P. Johnson. 2020. The development of mental rotation ability across the first year after birth. Advances in Child Development and Behavior 58: 1–33. [Google Scholar]
- Newcombe, Nora S., and Andrea Frick. 2010. Early education for spatial intelligence: Why, what, and how. Mind, Brain and Education: The Official Journal of the International Mind, Brain, and Education Society 4: 102–11. [Google Scholar] [CrossRef]
- Newcombe, Nora S., Julie L. Booth, and Elizabeth A. Gunderson. 2019. Spatial skills, reasoning, and mathematics. In The Cambridge Handbook of Cognition and Education. Cambridge: Cambridge University Press, pp. 100–23. [Google Scholar]
- Pedrett, Salome, Alain Chavaillaz, and Andrea Frick. 2023. Age-related changes in how 3.5- to 5.5-year-olds observe and imagine rotational object motion. Spatial Cognition and Computation 23: 83–111. [Google Scholar] [CrossRef]
- Pedrett, Salome, Lea Kaspar, and Andrea Frick. 2020. Understanding of object rotation between two and three years of age. Developmental Psychology 56: 261–74. [Google Scholar] [CrossRef] [PubMed]
- Perrucci, Vittore, Franca Agnoli, and Paolo Albiero. 2008. Children’s performance in mental rotation tasks: Orientation-free features flatten the slope. Developmental Science 11: 732–42. [Google Scholar] [CrossRef]
- Quaiser-Pohl, Claudia. 2003. The Mental Cutting Test “Schnitte” and the Picture Rotation Test-Two New Measures to Assess Spatial Ability. International Journal of Testing 3: 219–31. [Google Scholar] [CrossRef]
- Quaiser-Pohl, Claudia, Anna M. Rohe, and Tobias Amberger. 2010. The Solution Strategy as an Indicator of the Developmental Stage of Preschool Children’s Mental-Rotation Ability. Journal of Individual Differences 31: 95–100. [Google Scholar] [CrossRef]
- Quinn, Paul C., and Lynn S. Liben. 2008. A sex difference in mental rotation in young infants. Psychological Science 19: 1067–70. [Google Scholar] [CrossRef]
- Quinn, Paul C., and Lynn S. Liben. 2014. A sex difference in mental rotation in infants: Convergent evidence. Infancy: The Official Journal of the International Society on Infant Studies 19: 103–16. [Google Scholar] [CrossRef]
- R Core Team. 2013. R: A Language and Environment for Statistical Computing. Vienna: R Foundation for Statistical Computing. [Google Scholar]
- Rich, Jason T., J. Gail Neely, Randal C. Paniello, Courtney C. J. Voelker, Brian Nussenbaum, and Eric W. Wang. 2010. A practical guide to understanding Kaplan-Meier curves. Otolaryngology--Head and Neck Surgery: Official Journal of American Academy of Otolaryngology-Head and Neck Surgery 143: 331–36. [Google Scholar] [CrossRef]
- Schlegelmilch, Karola, and Annie E. Wertz. 2019. The Effects of Calibration Target, Screen Location, and Movement Type on Infant Eye-Tracking Data Quality. Infancy: The Official Journal of the International Society on Infant Studies 24: 636–62. [Google Scholar] [CrossRef]
- Senju, Atsushi, and Gergely Csibra. 2008. Gaze following in human infants depends on communicative signals. Current biology 18: 668–71. [Google Scholar]
- Shea, Daniel L., David Lubinski, and Camilla P. Benbow. 2001. Importance of assessing spatial ability in intellectually talented young adolescents: A 20-year longitudinal study. Journal of Educational Psychology 93: 604–14. [Google Scholar] [CrossRef]
- Shepard, Roger N., and Jacqueline Metzler. 1971. Mental rotation of three-dimensional objects. Science 171: 701–3. [Google Scholar] [CrossRef] [PubMed]
- Sjoberg, Daniel, Mark Baillie, Steven Haesendonckx, and Tim Treis. 2023. ggsurvfit: Flexible Time-to-Event Figures. Available online: http://www.danieldsjoberg.com/ggsurvfit/. (accessed on 1 March 2023).
- Slone, Lauren K., David S. Moore, and Scott P. Johnson. 2018. Object exploration facilitates 4-month-olds’ mental rotation performance. PLoS ONE 13: e0200468. [Google Scholar] [CrossRef] [PubMed]
- Szumilas, Magdalena. 2010. Explaining odds ratios. Journal of the Canadian Academy of Child and Adolescent Psychiatry 19: 227–29. [Google Scholar]
- Thurstone, Thelma Gwinn. 1957. The Tests of Primary Mental Abilities. The Personnel and Guidance Journal 35: 569–76. [Google Scholar] [CrossRef]
- Uttal, David H., Nathaniel G. Meadow, Elizabeth Tipton, Linda L. Hand, Alison R. Alden, Christopher Warren, and Nora S. Newcombe. 2013. The malleability of spatial skills: A meta-analysis of training studies. Psychological Bulletin 139: 352–402. [Google Scholar] [CrossRef]
- van Tetering, Marleen, Marthe Van der Donk, Renate Helena Maria De Groot, and Jelle Jolles. 2019. Sex differences in the performance of 7–12 year olds on a mental rotation task and the relation with arithmetic performance. Frontiers in Psychology 10: 107. [Google Scholar] [CrossRef]
- Vandenberg, Steven G., and Allan R. Kuse. 1978. Mental rotations, a group test of three-dimensional spatial visualization. Perceptual and Motor Skills 47: 599–604. [Google Scholar] [CrossRef]
- Verdine, Brian N., Roberta M. Golinkoff, Kathryn Hirsh-Pasek, Nora S. Newcombe, Andrew T. Filipowicz, and Alicia Chang. 2014. Deconstructing building blocks: Preschoolers’ spatial assembly performance relates to early mathematical skills. Child Development 85: 1062–76. [Google Scholar]
- Wai, Jonathan, David Lubinski, and Camilla P. Benbow. 2009. Spatial ability for STEM domains: Aligning over 50 years of cumulative psychological knowledge solidifies its importance. Journal of Educational Psychology 101: 817–35. [Google Scholar] [CrossRef]
- Wickham, Hadley, Mara Averick, Jennifer Bryan, Winston Chang, Lucy D’Agostino McGowan, Romain François, Garrett Grolemund, Alex Hayes, Lionel Henry, Jim Hester, and et al. 2019. Welcome to the Tidyverse. Journal of Open Source Software 4: 1686. [Google Scholar] [CrossRef]
- Wimmer, Marina C., Elizabeth J. Robinson, and Martin J. Doherty. 2017. Are developments in mental scanning and mental rotation related? PLoS ONE 12: e0171762. [Google Scholar] [CrossRef] [PubMed]
- Xu, Shan, Yiying Song, and Jia Liu. 2023. The Development of Spatial Cognition and Its Malleability Assessed in Mass Population via a Mobile Game. Psychological Science 34: 345–57. [Google Scholar] [CrossRef] [PubMed]
- Yanaoka, Kaichi, Laura E. Michaelson, Ryan Mori Guild, Grace Dostart, Jade Yonehiro, Satoru Saito, and Yuko Munakata. 2022. Cultures Crossing: The Power of Habit in Delaying Gratification. Psychological Science 33: 1172–81. [Google Scholar] [CrossRef] [PubMed]


Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Beckner, A.G.; Katz, M.; Tompkins, D.N.; Voss, A.T.; Winebrake, D.; LoBue, V.; Oakes, L.M.; Casasola, M. A Novel Approach to Assessing Infant and Child Mental Rotation. J. Intell. 2023, 11, 168. https://doi.org/10.3390/jintelligence11080168
Beckner AG, Katz M, Tompkins DN, Voss AT, Winebrake D, LoBue V, Oakes LM, Casasola M. A Novel Approach to Assessing Infant and Child Mental Rotation. Journal of Intelligence. 2023; 11(8):168. https://doi.org/10.3390/jintelligence11080168
Chicago/Turabian StyleBeckner, Aaron G., Mary Katz, David N. Tompkins, Annika T. Voss, Deaven Winebrake, Vanessa LoBue, Lisa M. Oakes, and Marianella Casasola. 2023. "A Novel Approach to Assessing Infant and Child Mental Rotation" Journal of Intelligence 11, no. 8: 168. https://doi.org/10.3390/jintelligence11080168
APA StyleBeckner, A. G., Katz, M., Tompkins, D. N., Voss, A. T., Winebrake, D., LoBue, V., Oakes, L. M., & Casasola, M. (2023). A Novel Approach to Assessing Infant and Child Mental Rotation. Journal of Intelligence, 11(8), 168. https://doi.org/10.3390/jintelligence11080168




