Immunogenicity of Recombinant-Deficient Lactobacillus casei with Complementary Plasmid Expressing Alanine Racemase Gene and Core Neutralizing Epitope Antigen against Porcine Epidemic Diarrhea Virus
Abstract
1. Introduction
2. Materials and Methods
2.1. Bacterial Strains, Virus, and Plasmids
2.2. Construction of Recombinant L. casei Strains
2.3. Identification of the Expression of the Protein of Interest
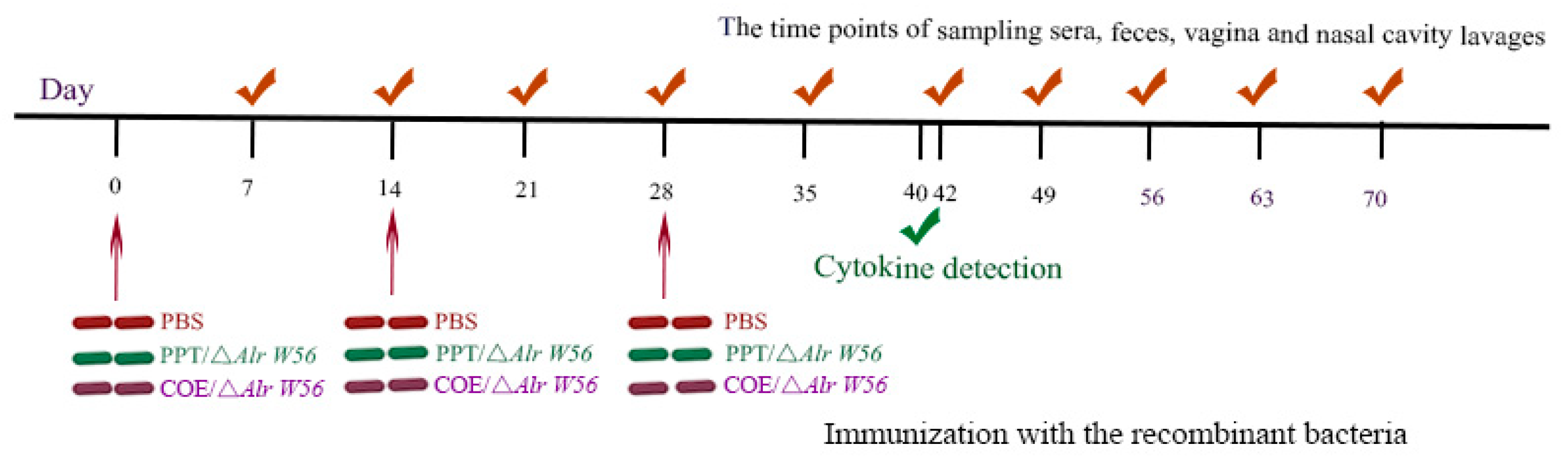
2.4. Immunization
2.5. Sample Collection
2.6. Enzyme-Linked Immunosorbent Assay (ELISA) Analysis
2.7. Neutralizing Activity of the Samples
2.8. Statistical Analysis
3. Results
3.1. Construction of Recombinant Lactobacillus Casei Strains and Demand Verification
3.2. Detection of Expression of the Protein of Interest
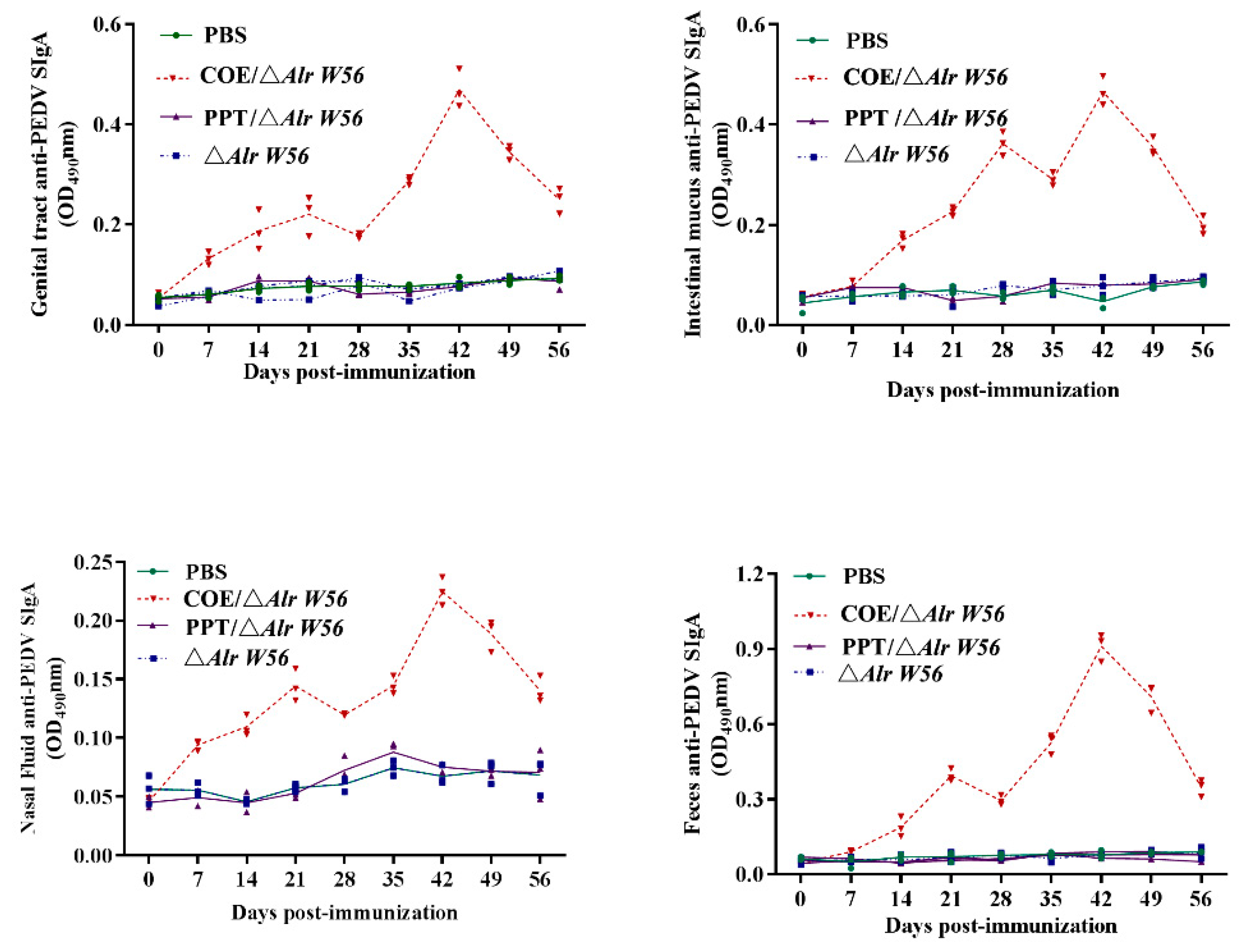
3.3. SIgA Levels Induced by Oral Recombinant Strains
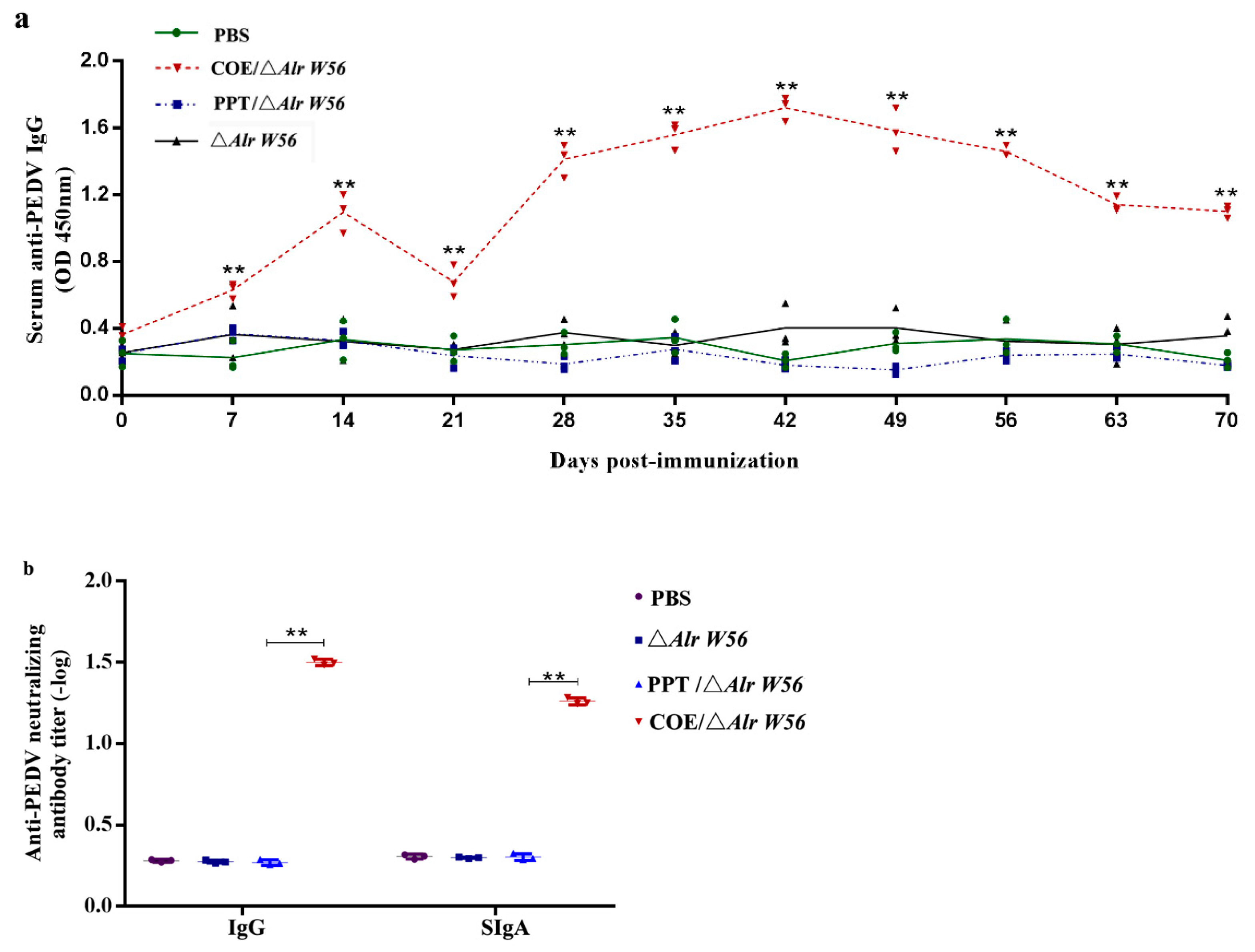
3.4. IgG Levels and Neutralizing Antibody Activity Induced by Oral Recombinant Strains
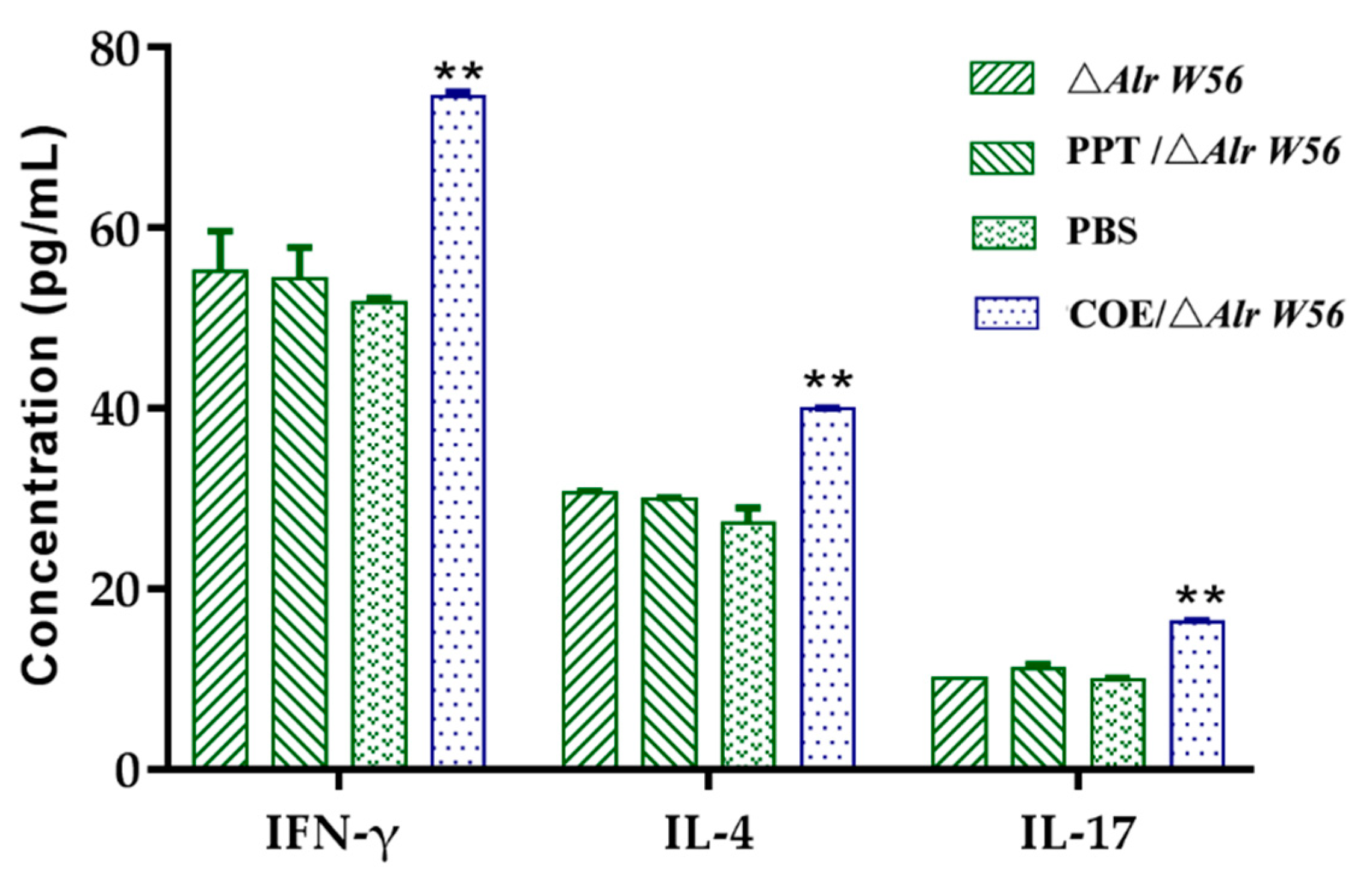
3.5. Production of Cytokines Excited by Oral Recombinant Strains
4. Discussion
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Conflicts of Interest
References
- Song, D.; Park, B. Porcine epidemic diarrhoea virus: A comprehensive review of molecular epidemiology, diagnosis, and vaccines. Virus Genes 2012, 44, 167–175. [Google Scholar] [CrossRef]
- Barman, N.N.; Barman, B.; Sarma, D.K.; Pensaert, M.B. Prevalence of rotavirus, transmissible gastroenteritis virus and porcine epidemic diarrhoea virus antibodies in pigs of Assam, India. Indian J. Anim. Sci. 2003, 73, 576–578. [Google Scholar]
- Cavanagh, D.; Brian, D.A.; Brinton, M.A.; Enjuanes, L.; Holmes, K.V.; Horzinek, M.C.; Lai, M.M.C.; Laude, H.; Plagemann, P.G.W.; Siddell, S.G.; et al. The Coronaviridae Now Comprises Two Genera, Coronavirus and Torovirus: Report of the Coronaviridae Study Group. Adv. Exp. Med. Biol. 1993, 342, 255–257. [Google Scholar] [PubMed]
- Kim, S.Y.; Song, D.S.; Park, B.K. Investigation, Differential detection of transmissible gastroenteritis virus and porcine epidemic diarrhea virus by duplex RT-PCR. J. Vet. Diagn. Investig. 2001, 13, 516–520. [Google Scholar] [CrossRef] [PubMed]
- Chen, J.; Liu, X.; Shi, D.; Shi, H.; Zhang, X.; Li, C.; Chi, Y.; Feng, L. Detection and Molecular Diversity of Spike Gene of Porcine Epidemic Diarrhea Virus in China. Viruses 2013, 5, 2601–2613. [Google Scholar] [CrossRef] [PubMed]
- Ge, J.W.; Liu, D.Q.; Li, Y.J. Construction of recombinant lactobacilli expressing the core neutralizing epitope (COE) of porcine epidemic diarrhea virus and a fusion protein consisting of COE and Escherichia coli heat-labile enterotoxin B, and comparison of the immune responses by orogastric immunization. Can. J. Microbiol. 2012, 58, 1258–1267. [Google Scholar] [PubMed]
- Yu, M.; Wang, L.; Ma, S.; Wang, X.; Wang, Y.; Xiao, Y.; Jiang, Y.; Qiao, X.; Tang, L.; Xu, Y.; et al. Immunogenicity of eGFP-Marked Recombinant Lactobacillus casei against Transmissible Gastroenteritis Virus and Porcine Epidemic Diarrhea Virus. Viruses 2017, 9, 274. [Google Scholar] [CrossRef]
- Su, Y.; Liu, Y.; Chen, Y.; Zhao, B.; Ji, P.; Xing, G.; Jiang, D.; Liu, C.; Song, Y.; Wang, G.; et al. Detection and phylogenetic analysis of porcine epidemic diarrhea virus in central China based on the ORF3 gene and the S1 gene. Virol. J. 2016, 13, 192. [Google Scholar] [CrossRef] [PubMed]
- Shima, H.; Watanabe, T.; Fukuda, S.; Fukuoka, S.-I.; Ohara, O.; Ohno, H. A novel mucosal vaccine targeting Peyer’s patch M cells induces protective antigen-specific IgA responses. Int. Immunol. 2014, 26, 619–625. [Google Scholar] [CrossRef] [PubMed]
- Pouwels, P.H.; Leer, R.J.; Boersma, W.J. The potential of Lactobacillus as a carrier for oral immunization: Development and preliminary characterization of vector systems for targeted delivery of antigens. J. Biotechnol. 1996, 44, 183–192. [Google Scholar] [CrossRef]
- Tsai, Y.-T.; Cheng, P.-C.; Pan, T.-M. The immunomodulatory effects of lactic acid bacteria for improving immune functions and benefits. Appl. Microbiol. Biotechnol. 2012, 96, 853–862. [Google Scholar] [CrossRef]
- Gibson, G.R.; Probert, H.M.; Van Loo, J.; Rastall, R.A.; Roberfroid, M.B. Dietary modulation of the human colonic microbiota: Updating the concept of prebiotics. Nutr. Res. Rev. 2004, 17, 259–275. [Google Scholar] [CrossRef]
- Jiang, X.; Yu, M.; Qiao, X.; Liu, M.; Tang, L.; Jiang, Y.; Cui, W.; Li, Y. Up-regulation of MDP and tuftsin gene expression in Th1 and Th17 cells as an adjuvant for an oral Lactobacillus casei vaccine against anti-transmissible gastroenteritis virus. Appl. Microbiol. Biotechnol. 1940, 98, 8301–8312. [Google Scholar] [CrossRef]
- Wang, X.N.; Wang, L.; Zheng, D.Z.; Chen, S.; Shi, W.; Qiao, X.Y.; Jiang, Y.P.; Tang, L.J.; Xu, Y.G.; Li, Y.J. Oral immunization with a Lactobacillus casei-based anti-porcine epidemic diarrhoea virus (PEDV) vaccine expressing microfold cell-targeting peptide Co1 fused with the COE antigen of PEDV. J. Appl. Microbiol. 2018, 124, 368–378. [Google Scholar] [CrossRef]
- Cho, C.-S.; Islam, M.A.; Firdous, J.; Choi, Y.-J.; Yun, C.-H. Design and application of chitosan microspheres as oral and nasal vaccine carriers: An updated review. Int. J. Nanomed. 2012, 7, 6077–6093. [Google Scholar] [CrossRef]
- Bron, P.A.; Benchimol, M.G.; Lambert, J.; Palumbo, E.; Deghorain, M.; Delcour, J.; de Vos, W.M.; Kleerebezem, M.; Hols, P. Use of the alr Gene as a Food-Grade Selection Marker in Lactic Acid Bacteria. Appl. Environ. Microbiol. 2002, 68, 5663–5670. [Google Scholar] [CrossRef]
- Ye, M.; Li, G.; Liang, W.Q.; Liu, Y.H. Molecular cloning and characterization of a novel metagenome-derived multicopper oxidase with alkaline laccase activity and highly soluble expression. Appl. Microbiol. Biotechnol. 2010, 87, 1023–1031. [Google Scholar] [CrossRef]
- Heaton, M.P.; Johnston, R.B.; Thompson, T.L. Controlled lysis of bacterial cells utilizing mutants with defective synthesis of D-alanine. Can. J. Microbiol. 1988, 34, 256–261. [Google Scholar] [CrossRef] [PubMed]
- Palumbo, E.; Favier, C.F.; Deghorain, M.; Cocconcelli, P.S.; Grangette, C.; Mercenier, A.; Vaughan, E.E.; Hols, P. Knockout of the alanine racemase gene in Lactobacillus plantarum results in septation defects and cell wall perforation. FEMS Microbiol. Lett. 2010, 233, 131–138. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Choi, S.; Kwon, S.; Kim, K. Generation of a temperature-sensitive Edwardsiella tarda mutant and its potential as a prophylactic vaccine in olive flounder (Paralichthys olivaceus). J. Appl. Microbiol. 2012, 113, 248–255. [Google Scholar] [CrossRef] [PubMed]
- Nguyen, T.T.; Mathiesen, G.; Fredriksen, L.; Kittl, R.; Nguyen, T.H.; Eijsink, V.G.; Haltrich, D.; Peterbauer, C.K. A Food-Grade System for Inducible Gene Expression in Lactobacillus plantarum Using an Alanine Racemase-Encoding Selection Marker. J. Agric. Food Chem. 2011, 59, 5617–5624. [Google Scholar] [CrossRef]
- Hols, P.; Defrenne, C.; Ferain, T.; Derzelle, S.; Delplace, B.; Delcour, J. The alanine racemase gene is essential for growth of Lactobacillus plantarum. J. Bacteriol. 1997, 179, 3804–3807. [Google Scholar] [CrossRef]
- Song, B.-F.; Ju, L.-Z.; Li, Y.-J.; Tang, L.-J. Chromosomal Insertions in the Lactobacillus caseiupp Gene That Are Useful for Vaccine Expression. Appl. Environ. Microbiol. 2014, 80, 3321–3326. [Google Scholar] [CrossRef] [PubMed]
- Zhang, N.; Hou, X.; Yu, L.; Wang, G.; Zhao, Z.; Gao, Y. Colonization and distribution of recombinant Lactobacillus casei with green fluorescent protein in mice intestine. Acta Microbiol. Sin. 2010, 50, 1232–1238. [Google Scholar]
- Robinson, K.; Chamberlain, L.M.; Schofield, K.M.; Wells, J.M.; Le Page, R.W. Oral vaccination of mice against tetanus with recombinant Lactococcus lactis. Nat. Biotechnol. 1997, 15, 653–657. [Google Scholar] [CrossRef] [PubMed]
- Langel, S.; Paim, F.C.; Lager, K.M.; Vlasova, A.N.; Saif, L.J. Lactogenic immunity and vaccines for porcine epidemic diarrhea virus (PEDV): Historical and current concepts. Virus Res. 2016, 226, 93–107. [Google Scholar] [CrossRef] [PubMed]
- Boniotti, M.B.; Papetti, A.; Lavazza, A.; Alborali, G.; Sozzi, E.; Chiapponi, C.; Faccini, S.; Bonilauri, P.; Cordioli, P.; Marthaler, D. Porcine Epidemic Diarrhea Virus and Discovery of a Recombinant Swine Enteric Coronavirus, Italy. Emerg. Infect. Dis. 2016, 22, 83–87. [Google Scholar] [CrossRef] [PubMed]
- Chiou, H.Y.; Huang, Y.L.; Deng, M.C.; Chang, C.Y.; Jeng, C.R.; Tsai, P.S.; Yang, C.; Pang, V.F.; Chang, H.W. Phylogenetic Analysis of the Spike (S) Gene of the New Variants of Porcine Epidemic Diarrhoea Virus in Taiwan. Transbound. Emerg. Dis. 2017, 64, 157–166. [Google Scholar] [CrossRef]
- Lee, S.; Lee, C. Outbreak-Related Porcine Epidemic Diarrhea Virus Strains Similar to US Strains, South Korea, 2013. Emerg. Infect. Dis. 2014, 20, 1223–1226. [Google Scholar] [CrossRef]
- Sun, D.; Wang, X.; Wei, S.; Chen, J.; Feng, L. Epidemiology and vaccine of porcine epidemic diarrhea virus in China: A mini-review. J. Vet. Med. Sci. 2016, 78, 355–363. [Google Scholar] [CrossRef]
- Jiang, X.; Hou, X.; Tang, L.; Jiang, Y.; Ma, G.; Li, Y. A phase trial of the oral Lactobacillus casei vaccine polarizes Th2 cell immunity against transmissible gastroenteritis coronavirus infection. Appl. Microbiol. Biotechnol. 2016, 100, 7457–7469. [Google Scholar] [CrossRef]
- Xu, Y.; Li, Y. Induction of Immune Responses in Mice after Intragastric Administration of Lactobacillus casei Producing Porcine Parvovirus VP2 Protein. Appl. Environ. Microbiol. 2007, 73, 7041–7047. [Google Scholar] [CrossRef]
- Daniel, C.; Roussel, Y.; Kleerebezem, M.; Pot, B. Recombinant lactic acid bacteria as mucosal biotherapeutic agents. Trends Biotechnol. 2011, 29, 499–508. [Google Scholar] [CrossRef] [PubMed]
- Esvaran, M.; Conway, P.L. Factors that Influence the Immunological Adjuvant Effect of Lactobacillus fermentum PC1 on Specific Immune Responses in Mice to Orally Administered Antigens. Vaccines 2016, 4, 24. [Google Scholar] [CrossRef]
- Wells, J.M.; Mercenier, A. Mucosal delivery of therapeutic and prophylactic molecules using lactic acid bacteria. Nat. Rev. Microbiol. 2008, 6, 349–362. [Google Scholar] [CrossRef] [PubMed]
- Eun, C.S.; Kim, Y.S.; Han, D.S.; Choi, J.H.; Lee, A.R.; Park, Y.K. Lactobacillus casei prevents impaired barrier function in intestinal epithelial cells. APMIS 2010, 119, 49–56. [Google Scholar] [CrossRef]
- Mohamadzadeh, M.; Duong, T.; Sandwick, S.J.; Hoover, T.; Klaenhammer, T.R. Dendritic cell targeting of Bacillus anthracis protective antigen expressed by Lactobacillus acidophilus protects mice from lethal challenge. Proc. Natl. Acad. Sci. USA 2009, 106, 4331–4336. [Google Scholar] [CrossRef] [PubMed]
- Cui, L.C.; Guan, X.T.; Liu, Z.M.; Tian, C.Y.; Xu, Y.G. Recombinant lactobacillus expressing G protein of spring viremia of carp virus (SVCV) combined with ORF81 protein of koi herpesvirus (KHV): A promising way to induce protective immunity against SVCV and KHV infection in cyprinid fish via oral vaccination. Vaccine 2015, 33, 3092–3099. [Google Scholar] [CrossRef] [PubMed]
- Ma, S.; Wang, L.; Huang, X.; Wang, X.; Chen, S.; Shi, W.; Qiao, X.; Jiang, Y.; Tang, L.; Xu, Y.; et al. Oral recombinant Lactobacillus vaccine targeting the intestinal microfold cells and dendritic cells for delivering the core neutralizing epitope of porcine epidemic diarrhea virus. Microb. Cell Fact. 2018, 17, 20. [Google Scholar] [CrossRef]
- Xu, Y.; Cui, L.; Tian, C.; Zhang, G.; Huo, G.; Tang, L.; Li, Y. Immunogenicity of Recombinant Classic Swine Fever Virus CD8+ T Lymphocyte Epitope and Porcine Parvovirus VP2 Antigen Coexpressed by Lactobacillus casei in Swine via Oral Vaccination. Clin. Vaccine Immunol. 2011, 18, 1979–1986. [Google Scholar] [CrossRef]
- Qiao, X.; Li, G.; Wang, X.; Li, X.; Liu, M.; Li, Y. Recombinant porcine rotavirus VP4 and VP4-LTB expressed in Lactobacillus casei induced mucosal and systemic antibody responses in mice. BMC Microbiol. 2009, 9, 249. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.; Xia, T.; Guo, T.; Ru, Y.; Jiang, Y.; Cui, W.; Zhou, H.; Qiao, X.; Tang, L.; Xu, Y.; et al. Recombinant Lactobacillus casei expressing capsid protein vp60 can serve as vaccine against rabbit hemorrhagic disease virus in rabbits. Vaccines 2019, 7, 172. [Google Scholar] [CrossRef] [PubMed]
- O’Sullivan, D.J.; Klaenhammer, T.R. High- and low-copy-number Lactococcus shuttle cloning vectors with features for clone screening. Gene 1993, 137, 227–231. [Google Scholar] [CrossRef]
- Ferrari, E.; Henner, D.J.; Yang, M.Y. Isolation of an Alanine Racemase Gene from Bacillus subtilis and Its Use for Plasmid Maintenance in B. subtilis. Nat. Biotechnol. 1985, 3, 1003–1007. [Google Scholar] [CrossRef]
- Xia, Y.; Chen, W.; Fu, X.; Zhang, H.; Yang, S.; Ding, X. Construction of an integrative food-grade expression system for Bacillus subtilis. Food Res. Int. 2005, 38, 251–256. [Google Scholar] [CrossRef]
- Williams, R.C.; Gibbons, R.J. Inhibition of Bacterial Adherence by Secretory Immunoglobulin A: A Mechanism of Antigen Disposal. Science 1972, 177, 697–699. [Google Scholar] [CrossRef] [PubMed]
- Hou, X.; Jiang, X.; Jiang, Y.; Tang, L.; Xu, Y.; Qiao, X.; Liu, M.; Cui, W.; Ma, G.; Li, Y. Oral Immunization against PEDV with Recombinant Lactobacillus casei Expressing Dendritic Cell-Targeting Peptide Fusing COE Protein of PEDV in Piglets. Viruses 2018, 10, 106. [Google Scholar] [CrossRef] [PubMed]
- Wadolkowski, E.A.; Burris, J.A.; O’Brien, A.D. Mouse model for colonization and disease caused by enterohemorragic Escherichia coli O157: H7. Infect. Immun. 1990, 58, 2438–2445. [Google Scholar] [CrossRef]
- Yang, G.; Jiang, Y.; Tong, P.; Li, C.; Yang, W.; Hu, J.; Ye, L.; Gu, W.; Shi, C.; Shan, B.; et al. Alleviation of enterotoxigenic Escherichia coli challenge by recombinant Lactobacillus plantarum expressing a FaeG- and DC-targeting peptide fusion protein. Benef. Microbes 2017, 8, 379–391. [Google Scholar] [CrossRef]
- Yang, W.T.; Yang, G.L.; Zhao, L.; Jin, Y.B.; Jiang, Y.L.; Huang, H.B.; Shi, C.W.; Wang, J.Z.; Wang, G.; Kang, Y.H.; et al. Lactobacillus plantarum displaying conserved M2e and HA2 fusion antigens induces protection against influenza virus challenge. Appl. Microbiol. Biotechnol. 2018, 102, 5077–5088. [Google Scholar] [CrossRef]
- Ashiuchi, M.; Nawa, C.; Kamei, T.; Song, J.J.; Hong, S.P.; Sung, M.H.; Soda, K.; Misono, H. Physiological and biochemical characteristics of poly gamma-glutamate synthetase complex of Bacillus subtilis. Eur. J. Biochem. 2001, 268, 5321–5328. [Google Scholar] [CrossRef] [PubMed]
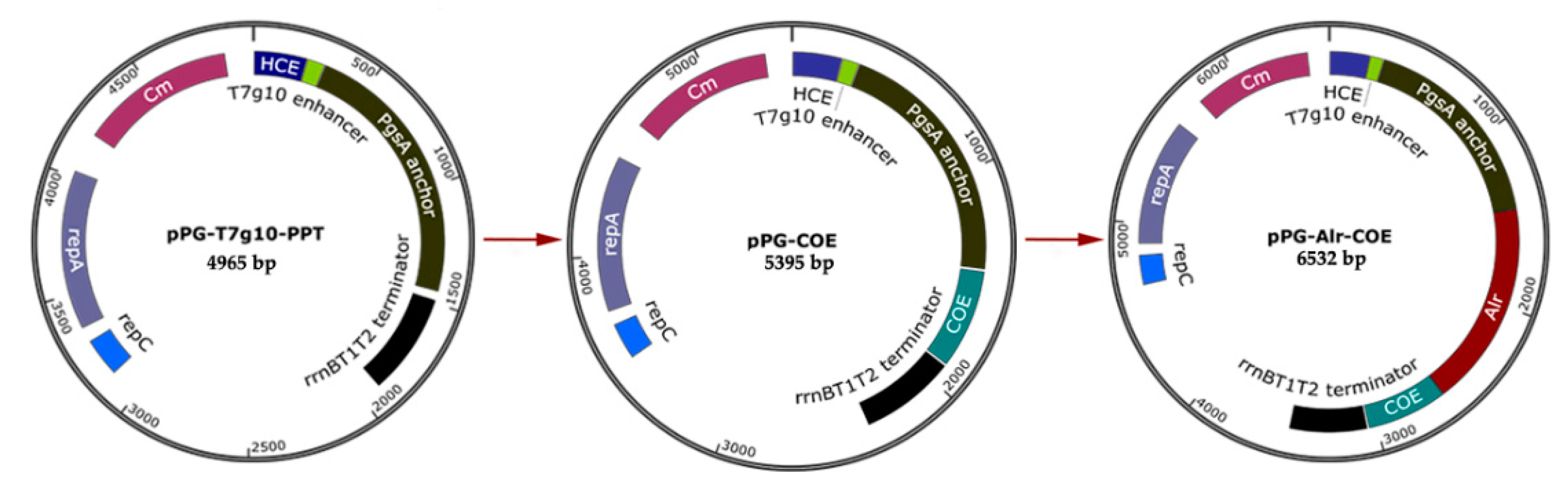


| Plasmids | Relevant Characteristics | Description/Reference |
|---|---|---|
| pMD19-Ts-COE | 3158bp; Ampr; the amplified DNA fragment core-neutralizing epitope (COE) was inserted into pMD19-Ts | This study |
| pMD19-Ts | 2692 bp; Ampr | Takara (Dalian, China) |
| pMD19-Ts-Alr | 3844 bp; Ampr; the amplified DNA fragment Alr was inserted into pMD19-Ts | This study |
| pPG-T7g10-PPT | 4965 bp; Cmr; HCE promoter; PgsA anchor; constitutive expression plasmid | Preserved in our Lab. |
| pPG-COE | 5395 bp; Cmr; COE was inserted into pPG-T7g10-PPT | This study |
| pPG-Alr-COE | 6532 bp; Cmr; Alr was inserted into pPG-COE | This study |
| Gene | ID | Primers Sequences |
|---|---|---|
| COE | COE-F COE-R | CGAGCTCATGGGTACCGATTATAAGGATGACGATGACAAGTAGAAACCTTCTGAGTCATG TAGGGCCCGTAATCAACTCACCCTTTGT |
| Alr | alr-F alr-R | CGAGCTCCTGAACGTGACGATCGGTAA TAGGTACCTCAATCGACCGGATTCACGC |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Li, F.; Wang, X.; Fan, X.; Sui, L.; Zhang, H.; Li, Y.; Zhou, H.; Wang, L.; Qiao, X.; Tang, L.; et al. Immunogenicity of Recombinant-Deficient Lactobacillus casei with Complementary Plasmid Expressing Alanine Racemase Gene and Core Neutralizing Epitope Antigen against Porcine Epidemic Diarrhea Virus. Vaccines 2021, 9, 1084. https://doi.org/10.3390/vaccines9101084
Li F, Wang X, Fan X, Sui L, Zhang H, Li Y, Zhou H, Wang L, Qiao X, Tang L, et al. Immunogenicity of Recombinant-Deficient Lactobacillus casei with Complementary Plasmid Expressing Alanine Racemase Gene and Core Neutralizing Epitope Antigen against Porcine Epidemic Diarrhea Virus. Vaccines. 2021; 9(10):1084. https://doi.org/10.3390/vaccines9101084
Chicago/Turabian StyleLi, Fengsai, Xiaona Wang, Xiaolong Fan, Ling Sui, Hailin Zhang, Yue Li, Han Zhou, Li Wang, Xinyuan Qiao, Lijie Tang, and et al. 2021. "Immunogenicity of Recombinant-Deficient Lactobacillus casei with Complementary Plasmid Expressing Alanine Racemase Gene and Core Neutralizing Epitope Antigen against Porcine Epidemic Diarrhea Virus" Vaccines 9, no. 10: 1084. https://doi.org/10.3390/vaccines9101084
APA StyleLi, F., Wang, X., Fan, X., Sui, L., Zhang, H., Li, Y., Zhou, H., Wang, L., Qiao, X., Tang, L., & Li, Y. (2021). Immunogenicity of Recombinant-Deficient Lactobacillus casei with Complementary Plasmid Expressing Alanine Racemase Gene and Core Neutralizing Epitope Antigen against Porcine Epidemic Diarrhea Virus. Vaccines, 9(10), 1084. https://doi.org/10.3390/vaccines9101084





