A Consent Support Resource with Benefits and Harms of Vaccination Does Not Increase Hesitancy in Parents—An Acceptability Study
Abstract
1. Introduction
2. Materials and Methods
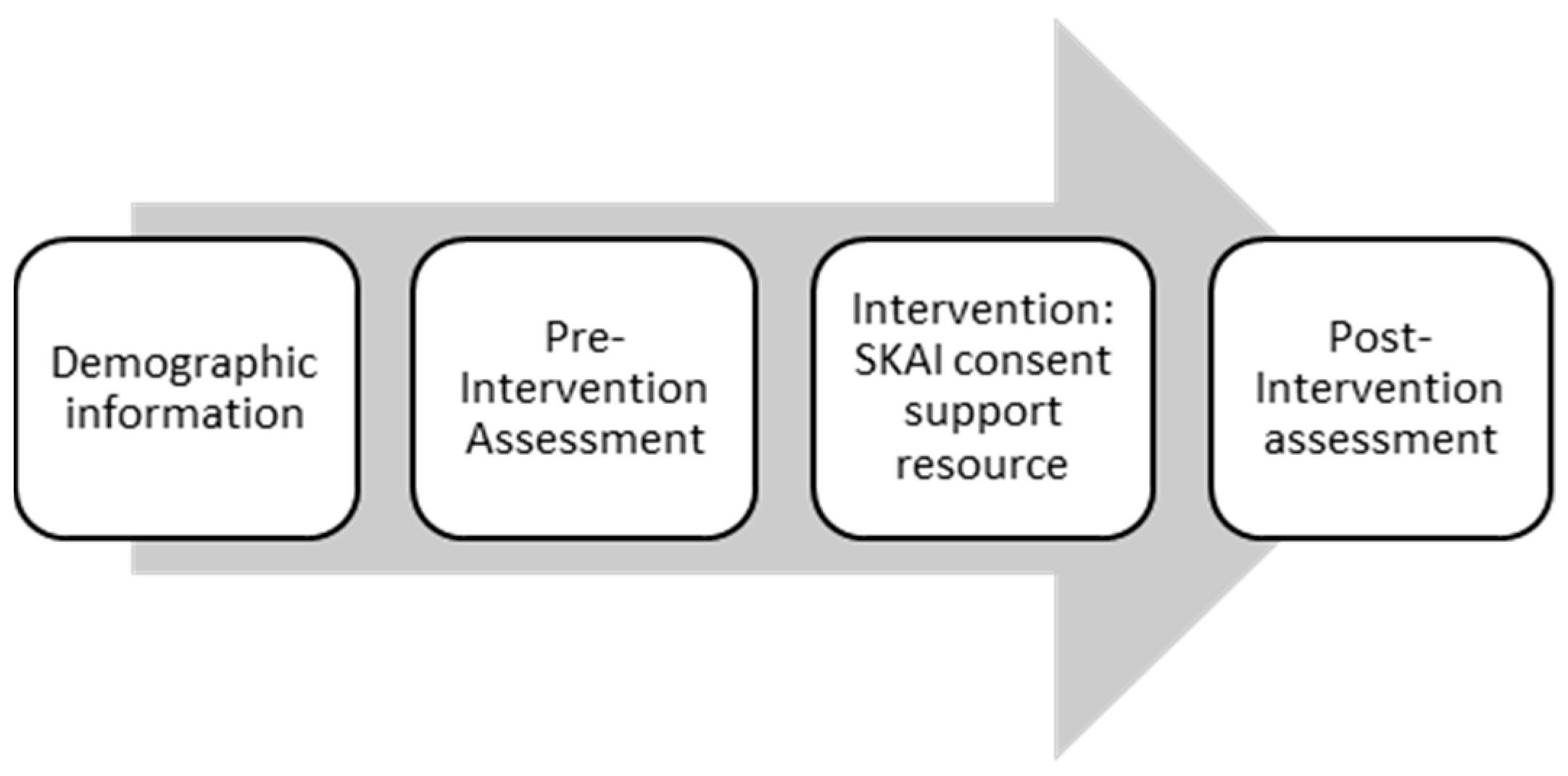
2.1. Study Design
2.2. Ethics
2.3. Intervention
2.4. Recruitment and Data Collection
2.5. Sample Size
2.6. Survey Items and Outcome Measures
2.7. Analysis
3. Results
3.1. Baseline Characteristics
3.2. Outcomes
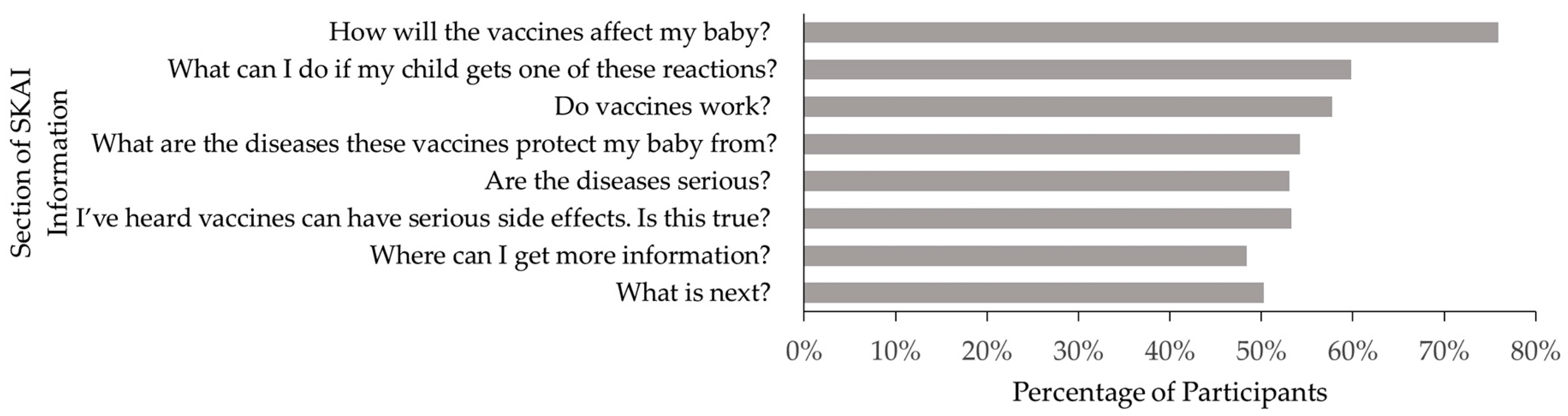
3.2.1. Intervention Fidelity
3.2.2. Primary Outcome: Hesitancy and Informed Decision Making
3.2.3. Secondary Outcomes: Stages of Decision Making, Vaccine Hesitancy Category, Attitude to Vaccination and Concerns about Side Effects and Safety
3.3. Subgroup Analyses
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Chan, M. The Power of Vaccines: Still Not fully Utilised; World Health Organization: Geneva, Switzerland, 2017; pp. 81–90. [Google Scholar]
- Orenstein, W.A.; Ahmed, R. Simply put: Vaccination saves lives. Proc. Natl. Acad. Sci. USA 2017, 114, 4031–4033. [Google Scholar] [CrossRef] [PubMed]
- Leask, J.; Braunack-Mayer, A.; Kerridge, I. Consent and public engagement in an era of expanded childhood immunisation. J. Paediatr. Child Health 2011, 47, 603–607. [Google Scholar] [CrossRef] [PubMed]
- Schwarz, N.; Sanna, L.J.; Skurnik, I.; Yoon, C. Metacognitive experiences and the intricacies of setting people straight: Implications for debiasing and public information campaigns. In Advances in Experimental Social Psychology; Elsevier Academic Press: San Diego, CA, USA, 2007; Volume 39, pp. 127–161. [Google Scholar] [CrossRef]
- Brewer, N.T.; Chapman, G.B.; Rothman, A.J.; Leask, J.; Kempe, A. Increasing Vaccination: Putting Psychological Science into Action. Psychol. Sci. Public Interest 2017, 18, 149–207. [Google Scholar] [CrossRef] [PubMed]
- MacDonald, N.E. Vaccine hesitancy: Definition, scope and determinants. Vaccine 2015, 33, 4161–4164. [Google Scholar] [CrossRef]
- WHO. Ten Threats to Global Health. Available online: https://www.who.int/news-room/feature-stories/ten-threats-to-global-health-in-2019 (accessed on 31 March 2020).
- Ames, H.M.; Glenton, C.; Lewin, S. Parents’ and informal caregivers’ views and experiences of communication about routine childhood vaccination: A synthesis of qualitative evidence. Cochrane Database Syst. Rev. 2017, 2. [Google Scholar] [CrossRef]
- My, C.; Danchin, M.; Willaby, H.W.; Pemberton, S.; Leask, J. Parental attitudes, beliefs, behaviours and concerns towards childhood vaccinations in Australia: A national online survey. Aust. Fam. Physician 2017, 46, 145–151. [Google Scholar]
- Brown, K.; Kroll, J.; Hudson, M.; Ramsay, M.; Green, J.; Long, S.; Vincent, C.; Fraser, G.; Sevdalis, N. Factors underlying parental decisions about combination childhood vaccinations including MMR: A systematic review. Vaccine 2010, 28, 4235–4248. [Google Scholar] [CrossRef]
- Handy, L.K.; Maroudi, S.; Powell, M.; Nfila, B.; Moser, C.; Japa, I.; Monyatsi, N.; Tzortzi, E.; Kouzeli, I.; Luberti, A.; et al. The impact of access to immunization information on vaccine acceptance in three countries. PLoS ONE 2017, 12, e0180759. [Google Scholar] [CrossRef]
- Costa-Pinto, J.C.; Willaby, H.W.; Leask, J.; Hoq, M.; Schuster, T.; Ghazarian, A.; O’Keefe, J.; Danchin, M.H. Parental Immunisation Needs and Attitudes Survey in paediatric hospital clinics and community maternal and child health centres in Melbourne, Australia. J. Paediatr. Child Health 2018, 54, 522–529. [Google Scholar] [CrossRef]
- Dubé, E.; Gagnon, D.; Ouakki, M.; Bettinger, J.A.; Witteman, H.O.; MacDonald, S.; Fisher, W.; Saini, V.; Greyson, D. Measuring vaccine acceptance among Canadian parents: A survey of the Canadian Immunization Research Network. Vaccine 2018, 36, 545–552. [Google Scholar] [CrossRef]
- Berry, N.J.; Danchin, M.; Trevena, L.; Witteman, H.O.; Kinnersley, P.; Snelling, T.; Robinson, P.; Leask, J. Sharing knowledge about immunisation (SKAI): An exploration of parents’ communication needs to inform development of a clinical communication support intervention. Vaccine 2018, 36, 6480–6490. [Google Scholar] [CrossRef] [PubMed]
- Dube, E.; Gagnon, D.; MacDonald, N.; Bocquier, A.; Peretti-Watel, P.; Verger, P. Underlying factors impacting vaccine hesitancy in high income countries: A review of qualitative studies. Expert Rev. Vaccines 2018, 17, 989–1004. [Google Scholar] [CrossRef] [PubMed]
- Wiley, K.E.; Steffens, M.; Berry, N.; Leask, J. An audit of the quality of online immunisation information available to Australian parents. BMC Public Health 2017, 17, 76. [Google Scholar] [CrossRef] [PubMed]
- Wheeler, M.; Buttenheim, A.M. Parental vaccine concerns, information source, and choice of alternative immunization schedules. Hum. Vaccin Immunother. 2013, 9, 1782–1789. [Google Scholar] [CrossRef]
- Connors, J.T.; Slotwinski, K.L.; Hodges, E.A. Provider-parent Communication When Discussing Vaccines: A Systematic Review. J. Pediatr. Nurs. 2017, 33, 10–15. [Google Scholar] [CrossRef]
- Berry, N.J.; Henry, A.; Danchin, M.; Trevena, L.J.; Willaby, H.W.; Leask, J. When parents won’t vaccinate their children: A qualitative investigation of Australian Primary Care providers’ experiences. BMC Pediatr. 2017, 17, 19. [Google Scholar] [CrossRef]
- Scherer, L.D.; Shaffer, V.A.; Patel, N.; Zikmund-Fisher, B.J. Can the vaccine adverse event reporting system be used to increase vaccine acceptance and trust? Vaccine 2016, 34, 2424–2429. [Google Scholar] [CrossRef]
- Shen, S.C.; Dubey, V. Addressing vaccine hesitancy: Clinical guidance for primary care physicians working with parents. Can. Fam. Phys. 2019, 65, 175–181. [Google Scholar]
- Ryan, R.; Hill, S. Supporting implementation of Cochrane methods in complex communication reviews: Resources developed and lessons learned for editorial practice and policy. Health Res. Policy Syst. 2019, 17, 32. [Google Scholar] [CrossRef]
- Craig, P.; Dieppe, P.; Macintyre, S.; Michie, S.; Nazareth, I.; Petticrew, M. Developing and evaluating complex interventions: The new Medical Research Council guidance. BMJ 2008, 337, a1655. [Google Scholar] [CrossRef]
- O’Cathain, A.; Croot, L.; Duncan, E.; Rousseau, N.; Sworn, K.; Turner, K.M.; Yardley, L.; Hoddinott, P. Guidance on how to develop complex interventions to improve health and healthcare. BMJ Open 2019, 9, e029954. [Google Scholar] [CrossRef] [PubMed]
- Opel, D.J.; Taylor, J.A.; Mangione-Smith, R.; Solomon, C.; Zhao, C.; Catz, S.; Martin, D. Validity and reliability of a survey to identify vaccine-hesitant parents. Vaccine 2011, 29, 6598–6605. [Google Scholar] [CrossRef] [PubMed]
- Bowen, D.J.; Kreuter, M.; Spring, B.; Cofta-Woerpel, L.; Linnan, L.; Weiner, D.; Bakken, S.; Kaplan, C.P.; Squiers, L.; Fabrizio, C.; et al. How we design feasibility studies. Am. J. Prev. Med. 2009, 36, 452–457. [Google Scholar] [CrossRef] [PubMed]
- Danchin, M.H.; Costa-Pinto, J.; Attwell, K.; Willaby, H.; Wiley, K.; Hoq, M.; Leask, J.; Perrett, K.P.; O’Keefe, J.; Giles, M.L.; et al. Vaccine decision-making begins in pregnancy: Correlation between vaccine concerns, intentions and maternal vaccination with subsequent childhood vaccine uptake. Vaccine 2018, 36, 6473–6479. [Google Scholar] [CrossRef]
- Opel, D.J.; Mangione-Smith, R.; Taylor, J.A.; Korfiatis, C.; Wiese, C.; Catz, S.; Martin, D.P. Development of a survey to identify vaccine-hesitant parents: The parent attitudes about childhood vaccines survey. Hum. Vaccines 2011, 7, 419–425. [Google Scholar] [CrossRef]
- Corben, P.; Leask, J. Vaccination hesitancy in the antenatal period: A cross-sectional survey. BMC Public Health 2018, 18, 566. [Google Scholar] [CrossRef]
- SKAI. Sharing Knowledge about Immunisation. Available online: www.talkingaboutimmunisation.org.au (accessed on 31 August 2020).
- Leask, J.; Kinnersley, P.; Jackson, C.; Cheater, F.; Bedford, H.; Rowles, G. Communicating with parents about vaccination: A framework for health professionals. BMC Pediatr. 2012, 12, 154. [Google Scholar] [CrossRef]
- Raosoft Sample Size Calculator. Available online: http://www.raosoft.com/samplesize.html (accessed on 31 August 2020).
- Parrella, A.; Gold, M.; Marshall, H.; Braunack-Mayer, A.; Baghurst, P. Parental perspectives of vaccine safety and experience of adverse events following immunisation. Vaccine 2013, 31, 2067–2074. [Google Scholar] [CrossRef]
- O’Connor, A.M. Validation of a decisional conflict scale. Med. Decis. Mak. 1995, 15, 25–30. [Google Scholar] [CrossRef]
- O’Connor, A. User Manual—Stage of Decision Making. Available online: https://decisionaid.ohri.ca/eval_stage.html (accessed on 31 August 2020).
- Williamson, L.; Glaab, H. Addressing vaccine hesitancy requires an ethically consistent health strategy. BMC Med. Ethics 2018, 19, 84. [Google Scholar] [CrossRef]
- Randall, S.; Leask, J.; Robinson, P.; Danchin, M.; Kinnersley, P.; Witteman, H.; Trevena, L.; Berry, N. Underpinning of the sharing knowledge about immunisation (SKAI) communication approach: A qualitative study using recorded observations. Patient Educ. Couns. 2020, 103, 1118–1124. [Google Scholar] [CrossRef] [PubMed]
- Beard, F.H.; Hull, B.P.; Leask, J.; Dey, A.; McIntyre, P.B. Trends and patterns in vaccination objection, Australia, 2002–2013. Med. J. Aust. 2016, 204, 275. [Google Scholar] [CrossRef] [PubMed]
- Fielding, J.; Bolam, B.; Danchin, M. Immunisation coverage and socioeconomic status—Questioning inequity in the ‘No Jab, No Pay’ policy. Aust. N. Z. J. Public Health 2017, 41, 455–457. [Google Scholar] [CrossRef] [PubMed][Green Version]
| Survey Item | Grouping/Score | |
|---|---|---|
| Stage of Decision Making At this Current Time, Regarding the Decision about Vaccinating Your Baby, Would You Say You: | ||
| haven’t begun to think about the choices |  | Not firmly decided |
| haven’t begun to think about the choices, but am interested in doing so | ||
| are considering the options now | ||
| are close to selecting an option | ||
| have already made a decision, but am willing to reconsider | ||
| have already made a decision and am unlikely to change my mind |  | Firmly decided |
| Decisional Conflict Scale–Informed Subscale (DCS-IS) When thinking about vaccinating your baby: (Answer Yes, Unsure, No) Do you know which options are available to you? Do you know the benefits of each option? Do you know the risks and side effects of each option? Parental Attitude to Childhood Vaccines Short Scale (PACV-SS) (Answer Agree, Unsure, Disagree) I trust the information I receive about vaccinations It is better for my child to develop immunity by getting sick than to get a vaccine It is better for my child to get fewer vaccines at the same time Children get more vaccines than are good for them (Answer Not hesitant, Unsure, Hesitant) Overall, how hesitant about childhood vaccines would you consider yourself to be? Positive Attitude Assessment I would give my child any new vaccine available, even if it was not on the National Immunisation Schedule | ||
| Strongly agree |  | Agree |
| Agree | ||
| Somewhat agree | ||
| Neither agree nor disagree | ||
| Somewhat disagree |  | Disagree |
| Disagree | ||
| Strongly disagree | ||
| Side-effect Concerns How concerned are you that your child might have a serious side effect from a vaccine? | ||
| Very concerned |  | Concerned |
| Somewhat concerned | ||
| Not sure |  | Not concerned |
| Not too concerned | ||
| Not at all concerned | ||
| Safety Concerns How concerned are you that any one of the childhood vaccines might not be safe? | ||
| Very concerned |  | Concerned |
| Somewhat concerned | ||
| Not sure |  | Not concerned |
| Not too concerned | ||
| Not at all concerned | ||
| Vaccine Communication Framework (VCF) Question Which one of the following statements best applies to you at this moment? | ||
| I will allow my baby to have all of their recommended vaccines and have NO concerns about the safety of those vaccines |  | Accepting |
| I will allow my baby to have all of their recommended vaccines and have A FEW MINOR concerns about their safety | ||
| I will allow my baby to have all of their recommended vaccines but have A LOT OF concerns about their safety |  | Not accepting |
| I will allow my baby to have some vaccines on time but there are some I prefer to delay or not have at all | ||
| I will not allow my baby to have any vaccines | ||
| I am unsure about vaccinating my baby | ||
| Characteristic | N (%) |
|---|---|
| Age (mean ± SD) | 31.6 (±4.8) years |
| Gender (Female) | 321 (77.2) |
| Level of Education | |
| Postgraduate | 85 (20.4) |
| Bachelor Degree | 183 (44.0) |
| Diploma or Certificate | 82 (19.7) |
| Secondary School | 60 (14.4) |
| Index of Relative Socio-economic Advantage and Disadvantage (IRSAD) Ranking in Deciles, By Postcode (10 represents most advantaged) | |
| 10 | 116 (27.9) |
| 9 | 105 (25.2) |
| 8 | 52 (12.5) |
| 7 | 26 (6.3) |
| 6 | 26 (6.3) |
| 5 | 21 (5.0) |
| 4 | 27 (6.5) |
| 3 | 17 (4.1) |
| 2 | 10 (2.4) |
| 1 | 14 (3.4) |
| Currently pregnant | |
| Yes | 353 (84.9) |
| First Trimester | 30 (7.2) |
| Second Trimester | 170 (40.9) |
| Third Trimester | 153 (36.8) |
| Have other children? (Yes) | 152 (36.5) |
| Youngest child has received birth vaccines? (Yes) | 62/152 (40.8) |
| Attitude towards vaccination | |
| Strongly oppose | 2 (0.5) |
| Generally oppose | 6 (1.4) |
| Neither oppose nor support | 11 (2.6) |
| Generally support | 57 (13.7) |
| Strongly support | 340 (81.7) |
| Previous childhood vaccination experiences | |
| Own child experienced adverse event | 13 (3.1) |
| Knows someone with adverse event | 73 (17.5) |
| Delayed vaccination (other than illness or allergy) | 13 (3.1) |
| Not vaccinated (other than illness or allergy) | 4 (1.0) |
| Survey Item | Mean (SE) Pre | Mean (SE) Post | Difference | 95% CI | p-Value | |
|---|---|---|---|---|---|---|
| Lower Limit | Upper Limit | |||||
| PACV-SS Score (Hesitancy Measure) a | - | - | - | |||
| All participants | 1.97 (0.10) | 1.94 (0.11) | −0.02 | −0.10 | 0.15 | 0.71 |
| Subgroup–VCF c “Not accepting” | 5.31 (0.44) | 4.84 (0.45) | −0.47 | −1.15 | 1.09 | 0.13 |
| Subgroup–Concerns raised | 3.82 (0.43) | 4.26 (0.50) | 0.44 | 0.24 | −1.12 | 0.20 |
| Subgroup-Non-viewers excluded | 1.96 (0.12) | 1.87 (0.13) | −0.09 | −0.63 | 0.23 | 0.26 |
| DCS-IS Score (Informed Decision Making) b | - | - | - | |||
| All participants | 29.05 (1.73) | 7.41 (0.90) | −21.63 | −24.71 | −18.56 | <0.0001 * |
| Subgroup–VCF “Not accepting” | 50.88 (5.62) | 15.79 (3.77) | −35.09 | −46.75 | −23.42 | <0.0001 * |
| Subgroup–Concerns raised | 47.06 (6.15) | 14.22 (3.86) | −32.84 | −45.69 | −20.00 | <0.0001 * |
| Subgroup–Non-viewers excluded | 30.83 (1.96) | 6.39 (0.91) | −24.44 | −28.80 | −20.80 | <0.0001 * |
| Survey Item a | Response Group a | Pre | Post | Difference | 95% CI | p-Value | |
|---|---|---|---|---|---|---|---|
| Lower Limit | Upper Limit | ||||||
| Positive Attitude Assessment | Agree | 43.8% | 50.4% | 6.5% | 3.0% | 10.0% | 0.001 * |
| Side-effect Concerns | Concerned | 37.0% | 29.0% | −8.0% | −4.0% | −12.0% | <0.001 * |
| Safety Concerns | Concerned | 28.5% | 23.0% | −5.6% | −2.3% | −8.8% | 0.001 * |
| Stage of Decision Making | Firmly decided | 75.4% | 72.5% | −2.8% | −5.9% | 0.2% | 0.097 |
| VCF Question | Accepting | 90.1% | 92.0% | 1.9% | −0.1% | 3.9% | 0.096 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
McDonald, C.; Leask, J.; Chad, N.; Danchin, M.; Fethney, J.; Trevena, L. A Consent Support Resource with Benefits and Harms of Vaccination Does Not Increase Hesitancy in Parents—An Acceptability Study. Vaccines 2020, 8, 500. https://doi.org/10.3390/vaccines8030500
McDonald C, Leask J, Chad N, Danchin M, Fethney J, Trevena L. A Consent Support Resource with Benefits and Harms of Vaccination Does Not Increase Hesitancy in Parents—An Acceptability Study. Vaccines. 2020; 8(3):500. https://doi.org/10.3390/vaccines8030500
Chicago/Turabian StyleMcDonald, Ciara, Julie Leask, Nina Chad, Margie Danchin, Judith Fethney, and Lyndal Trevena. 2020. "A Consent Support Resource with Benefits and Harms of Vaccination Does Not Increase Hesitancy in Parents—An Acceptability Study" Vaccines 8, no. 3: 500. https://doi.org/10.3390/vaccines8030500
APA StyleMcDonald, C., Leask, J., Chad, N., Danchin, M., Fethney, J., & Trevena, L. (2020). A Consent Support Resource with Benefits and Harms of Vaccination Does Not Increase Hesitancy in Parents—An Acceptability Study. Vaccines, 8(3), 500. https://doi.org/10.3390/vaccines8030500





