A Simple and High-Throughput ELISA-Based Neutralization Assay for the Determination of Anti-Flavivirus Neutralizing Antibodies
Abstract
1. Introduction
2. Materials and Methods
2.1. Cells and Viruses
2.2. Serum Samples and Monoclonal Antibodies
2.3. Plaque Reduction Neutralization Test
2.4. ELISA-Based Microneutralization Test
2.5. Single-Round Infectious Particle Production and Neutralization Test
2.6. Data Analysis
2.7. Ethics Statement
3. Results
3.1. Development of the ELISA-Based Microneutralization Test
3.2. Determination of EMNT Titers Using Monoclonal Antibodies
3.3. Determination of EMNT Titers Using Clinical Samples
3.4. Comparison of EMNT to the PRNT
3.5. EMNT on a 384-Well Plate Format
3.6. EMNT Using Single-Round Infectious Particles (SRIPs)
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Bhatt, S.; Gething, P.W.; Brady, O.J.; Messina, J.P.; Farlow, A.W.; Moyes, C.L.; Drake, J.M.; Brownstein, J.S.; Hoen, A.G.; Sankoh, O.; et al. The Global distribution and burden of dengue. Nature 2013, 496, 504–507. [Google Scholar] [CrossRef] [PubMed]
- Brady, O.J.; Gething, P.W.; Bhatt, S.; Messina, J.P.; Brownstein, J.S.; Hoen, A.G.; Moyes, C.L.; Farlow, A.W.; Scott, T.W.; Hay, S.I. Refining the global spatial limits of dengue virus transmission by evidence-based consensus. PLoS Negl. Trop. Dis. 2012, 6, e1760. [Google Scholar] [CrossRef] [PubMed]
- Boldescu, V.; Behnam, M.A.M.; Vasilakis, N.; Klein, C.D. Broad-spectrum agents for flaviviral infections: dengue, Zika and beyond. Nat. Rev. Drug Discov. 2017, 16, 565–586. [Google Scholar] [CrossRef] [PubMed]
- Collins, M.H.; Metz, S.W. Progress and works in progress: Update on flavivirus vaccine development. Clin. Ther. 2017, 39, 1519–1536. [Google Scholar] [CrossRef] [PubMed]
- Pierson, T.C.; Fremont, D.H.; Kuhn, R.J.; Diamond, M.S. Structural insights into the mechanisms of antibody-mediated neutralization of flavivirus infection: Implications for vaccine development. Cell Host Microbe. 2008, 4, 229–238. [Google Scholar] [CrossRef] [PubMed]
- Katzelnick, L.C.; Montoya, M.; Gresh, L.; Balmaseda, A.; Harris, E. Neutralizing antibody titers against dengue virus correlate with protection from symptomatic infection in a longitudinal cohort. Proc. Natl. Acad. Sci. USA 2016, 113, 728–733. [Google Scholar] [CrossRef] [PubMed]
- Katzelnick, L.C.; Baric, R.; Coller, B.A.; Coloma, J.; Crowe, J.E.; Cummings, D.A.T.; Dean, H.; de Silva, A.; Diamond, M.S.; Durbin, A.; et al. Immune correlates of protection for dengue: State of the art and research agenda. Vaccine 2017, 35, 1659–1669. [Google Scholar] [CrossRef] [PubMed]
- Buddhari, D.; Aldstadt, J.; Endy, T.P.; Srikiatkhachorn, A.; Thaisomboonsuk, B.; Klungthong, C.; Nisalak, A.; Khuntirat, B.; Jarman, R.G.; Fernandez, S.; et al. Dengue virus neutralizing antibody levels associated with protection from infection in Thai cluster studies. PLoS Negl. Trop. Dis. 2014, 8, e3230. [Google Scholar] [CrossRef]
- Russell, P.K.; Nisalak, A.; Sukhavachana, P.; Vivona, S. A plaque reduction test for dengue virus neutralizing antibodies. J. Immunol. 1967, 99, 285–290. [Google Scholar] [PubMed]
- World Health Organization Department of Immunization Vaccines and Biologicals. Guidelines for Plaque Reduction Neutralization Testing of Human Antibodies to Dengue Viruses; World Health Organization: Geneva, Switzerland, 2007; p. 2. [Google Scholar]
- Rodrigo, W.W.S.I.; Alcena, D.C.; Rose, R.C.; Jin, X.; Schlesinger, J.J. Short report: An automated dengue virus microneutralization plaque assay performed in human Fcγ receptor-expressing CV-1 cells. Am. J. Trop. Med. Hyg. 2009, 80, 61–65. [Google Scholar] [PubMed]
- Moi, M.L.; Lim, C.K.; Chua, K.B.; Takasaki, T.; Kurane, I. Dengue virus infection-enhancing activity in serum samples with neutralizing activity as determined by using FcγR-expressing cells. PLoS Neg. Trop. Dis. 2012, 6, e1536. [Google Scholar] [CrossRef] [PubMed]
- Sabchareon, A.; Wallace, D.; Sirivichayakul, C.; Limkittikul, K.; Chanthavanich, P.; Suvannadabba, S.; Jiwariyavej, V.; Dulyachai, W.; Pengsaa, K.; Wartel, T.A.; et al. Protective efficacy of the recombinant, live-attenuated, CYD tetravalent dengue vaccine in Thai schoolchildren: A randomised, controlled phase 2b trial. Lancet 2012, 380, 1559–1567. [Google Scholar] [CrossRef]
- Capeding, M.R.; Tran, N.H.; Hadinegoro, S.R.S.; Ismail, H.I.H.M.; Chotpitayasunondh, T.; Chua, M.N.; Luong, C.Q.; Rusmil, K.; Wirawan, D.N.; Nallusamy, R.; et al. Clinical efficacy and safety of a novel tetravalent dengue vaccine in healthy children in Asia: A phase 3, randomised, observer-masked, placebo-controlled trial. Lancet 2014, 384, 1358–1365. [Google Scholar] [CrossRef]
- Villar, L.; Dayan, G.H.; Arredondo-García, J.L.; Rivera, D.M.; Cunha, R.; Deseda, C.; Reynales, H.; Costa, M.S.; Morales-Ramírez, J.O.; Carrasquilla, G.; et al. Efficacy of a tetravalent dengue vaccine in children in Latin America. N. Engl. J. Med. 2015, 372, 113–123. [Google Scholar] [CrossRef]
- Chan, K.R.; Wang, X.; Saron, W.A.A.; Gan, E.S.; Tan, H.C.; Mok, D.Z.L.; Zhang, S.L.-X.; Lee, Y.H.; Liang, C.; Wijaya, L.; et al. Cross-reactive antibodies enhance live attenuated virus infection for increased immunogenecity. Nat. Microbiol. 2016, 1, 16164. [Google Scholar] [CrossRef] [PubMed]
- Dejnirattisai, W.; Supasa, P.; Wongwiwat, W.; Rouvinski, A.; Barba-Spaeth, G.; Duangchinda, T.; Sakuntabhai, A.; Cao-Lormeau, V.-M.; Malasit, P.; Rey, f.A.; et al. Dengue virus sero-cross-reactivity drives antibody-dependent enhancement of infection with Zika virus. Nat. Immunol. 2016, 17, 1102–1108. [Google Scholar] [CrossRef]
- Chan, K.R.; Ong, E.Z.; Tan, H.C.; Zhang, S.L.-X.; Zhang, Q.; Tang, K.F.; Kaliaperumal, N.; Lim, A.P.C.; Hibberd, M.L.; Chan, S.H.; et al. Leukocyte immunoglobulin-like receptor B1 is critical for antibody-dependent dengue. Proc. Natl. Acad. Sci. USA 2013, 111, 2722–2727. [Google Scholar] [CrossRef]
- Rodrigo, W.W.S.I.; Jin, X.; Blackley, S.D.; Rose, R.C.; Schlesinger, J.J. Differential enhancement of dengue virus immune complex infectivity mediated by signaling-competent and signaling-incompetent human FcgammaRIA (CD64) or FcgammaRIIA (CD32). J. Virol. 2006, 80, 10128–10138. [Google Scholar] [CrossRef] [PubMed]
- Chawla, T.; Chan, K.R.; Zhang, S.L.; Tan, H.C.; Lim, A.P.C.; Hanson, B.J.; Ooi, E.E. Dengue virus neutralization in cells expressing Fc gamma receptors. PLoS ONE 2013, 8, e65231. [Google Scholar] [CrossRef]
- Goncalvez, A.P.; Engle, R.E.; St Claire, M.; Purcell, R.H.; Lai, C.J. Monoclonal antibody-mediated enhancement of dengue virus infection in vitro and in vivo and strategies for prevention. Proc. Natl. Acad. Sci. USA 2007, 104, 9422–9427. [Google Scholar] [CrossRef] [PubMed]
- Ng, J.K.W.; Zhang, S.L.; Tan, H.C.; Yan, B.; Martinez, J.M.; Tan, W.Y.; Lam, J.H.; Tan, G.K.X.; Ooi, E.E.; Alonso, S. First experimental in vivo model of enhanced dengue disease severity through maternally acquired heterotypic dengue antibodies. PLoS Pathog. 2014, 10, e1004031. [Google Scholar] [CrossRef] [PubMed]
- Halstead, S.B. In vivo enhancement of dengue virus infection in Rhesus monkeys by passively transferred antibody. J. Infect. Dis. 1979, 140, 527–533. [Google Scholar] [CrossRef] [PubMed]
- Katzelnick, L.C.; Gresh, L.; Halloran, M.E.; Mercado, J.C.; Kuan, G.; Gordon, A.; Balmaseda, A.; Harris, E. Antibody-dependent enhancement of severe dengue disease in humans. Science 2017, 358, 929–932. [Google Scholar] [CrossRef] [PubMed]
- Gan, E.S.; Ting, D.H.R.; Chan, K.R. The mechanistic role of antibodies to dengue virus in protection and disease pathogenesis. Expert Rev. Anti. Infect. Ther. 2017, 15, 111–119. [Google Scholar] [CrossRef] [PubMed]
- Vorndam, V.; Beltran, M. Enzyme-linked immunosorbent assay-format microneutralization test for dengue viruses. Am. J. Trop. Med. Hyg. 2002, 66, 208–212. [Google Scholar] [CrossRef] [PubMed]
- Whiteman, M.C.; Bogardus, L.; Giacone, D.G.; Rubinstein, L.J.; Antonello, J.M.; Sun, D.; Daijogo, S.; Gurney, K.B. Virus reduction neutralization test: A single-cell imaging high-throughput virus neutralization assay for dengue. Am. J. Trop. Med. Hyg. 2018, 99, 1430–1439. [Google Scholar] [CrossRef] [PubMed]
- Matsuda, M.; Yamanaka, A.; Yato, K.; Yoshii, K.; Watashi, K.; Aizaki, H.; Konishi, E.; Takasaki, T.; Kato, T.; Muramatsu, M.; et al. High-throughput neutralization assay for multiple flaviviruses based on single-round infectious particles using dengue virus type 1 reporter replicon. Sci. Rep. 2018, 8, 4–7. [Google Scholar] [CrossRef] [PubMed]
- Liu, L.; Wen, K.; Li, J.; Hu, D.; Huang, Y.; Qiu, L.; Cai, J.; Che, X. Comparison of plaque- and enzyme-linked immunospot-based assays to measure the neutralizing activities of monoclonal antibodies specific to domain III of dengue virus envelope protein. Clin. Vaccine Immunol. 2012, 19, 73–78. [Google Scholar] [CrossRef] [PubMed]
- Wilson, H.L.; Tran, T.; Druce, J.; Dupont-Rouzeyrol, M.; Catton, M. Neutralization assay for Zika and dengue viruses by use of real-time-PCR-based endpoint assessment. Clin. Microbiol. 2017, 55, 3104–3112. [Google Scholar] [CrossRef] [PubMed]
- Shan, C.; Ortiz, D.A.; Yang, Y.; Wong, S.J.; Kramer, L.D.; Shi, P.Y.; Loeffelholz, M.J.; Ren, P. Evaluation of a novel reporter virus neutralization test for serological diagnosis of Zika and dengue virus infection. J. Clin. Microbiol. 2017, 55, 3028–3036. [Google Scholar] [CrossRef] [PubMed]
- Li, S.; Zhao, H.; Yang, H.; Hou, W.; Cruz-Cosme, R.; Cao, R.; Chen, C.; Wang, W.; Xu, L.; Zhang, J.; et al. Rapid neutralization testing system for Zika virus based on an enzyme-linked immunospot assay. ACS Infect. Dis. 2020, 6, 811–819. [Google Scholar] [CrossRef] [PubMed]
- Shan, C.; Xie, X.; Ren, P.; Loeffelholz, M.J.; Yang, Y.; Furuya, A.; Dupuis, A.P.; Kramer, L.D.; Wong, S.J.; Shi, P.Y. A Rapid Zika diagnostic assay to measure neutralizing antibodies in patients. eBioMedicine 2017, 17, 157–162. [Google Scholar] [CrossRef] [PubMed]
- Koishi, A.C.; Suzukawa, A.A.; Zanluca, C.; Camacho, D.E.; Comach, G.; Duarte dos Santos, C.N. Development and evaluation of a novel high-throughput image-based fluorescent neutralization test for detection of Zika virus infection. PLoS Negl. Trop. Dis. 2018, 12, e0006342. [Google Scholar] [CrossRef] [PubMed]
- Nascimento, E.J.M.; Bonaparte, M.I.; Luo, P.; Vincent, T.S.; Hu, B.; George, J.K.; Áñez, G.; Noriega, F.; Zheng, L.; Huleatt, J.W. Use of a blockade-of-binding ELISA and microneutralization assay to evaluate Zika virus serostatus in dengue-endemic areas. Am. J. Trop. Med. Hyg. 2019, 101, 708–715. [Google Scholar] [CrossRef] [PubMed]
- Moi, M.L.; Lim, C.K.; Kotaki, A.; Takasaki, T.; Kurane, I. Development of an antibody-dependent enhancement assay for dengue virus using stable BHK-21 cell lines expressing FcγRIIA. J. Virol. Methods 2010, 163, 205–209. [Google Scholar] [CrossRef] [PubMed]
- Kinoshita, H.; Matumbi Mathenge, E.G.; Hung, N.T.; Que Huong, V.T.; Kumatori, A.; Yu, F.; Parquet, M.D.C.; Inoue, S.; Matias, R.R.; Natividad, F.F.; et al. Isolation and characterization of two phenotypically distinct dengue type-2 virus isolates from the same dengue hemorrhagic fever patient. Jpn. J. Infect. Dis. 2009, 62, 343–350. [Google Scholar] [PubMed]
- Gross, F.L.; Bai, Y.; Jefferson, S.; Holiday, C.; Levine, M.Z. Measuring influenza neutralizing antibody responses to A(H3N2) viruses in human sera by microneutralization assays using MDCK-SIAT1 cells. J. Vis. Exp. 2017, 129, 56448. [Google Scholar] [CrossRef] [PubMed]
- Yamanaka, A.; Moi, M.L.; Takasaki, T.; Kurane, I.; Matsuda, M.; Suzuki, R.; Konishi, E. Utility of Japanese encephalitis virus subgenomic replicon-based single-round infectious particles as antigens in neutralization tests for Zika virus and three other flaviviruses. J. Virol. Methods 2017, 243, 164–171. [Google Scholar] [CrossRef] [PubMed]
- Suzuki, R.; Ishikawa, T.; Konishi, E.; Matsuda, M.; Watashi, K.; Aizaki, H.; Takasaki, T.; Wakita, T. Production of single-round infectious chimeric flaviviruses with DNA-based Japanese encephalitis virus replicon. J. Gen. Virol. 2014, 95, 60–65. [Google Scholar] [CrossRef] [PubMed]
- Putnak, J.R.; de la Barrera, R.; Burgess, T.; Pardo, J.; Dessy, F.; Gheysen, D.; Lobet, Y.; Green, S.; Endy, T.P.; Thomas, S.J.; et al. Comparative evaluation of three assays for measurement of dengue virus neutralizing antibodies. Am. J. Trop. Med. Hyg. 2008, 79, 115–122. [Google Scholar] [CrossRef] [PubMed]
- Roehrig, J.T.; Hombach, J.; Barrett, A.D.T. Guidelines for plaque-reduction neutralization testing of human antibodies to dengue viruses. Viral Immunol. 2008, 21, 123–132. [Google Scholar] [CrossRef] [PubMed]
- Makino, Y.; Tadano, M.; Saito, M.; Fukunaga, T.; Maneekarn, N.; Sittisombut, N.; Sirisanthana, V.; Ponepraser, B. Studies on serological cross-reaction in sequential flavivirus infections. Microbiol. Immunol. 1994, 38, 951–955. [Google Scholar] [CrossRef] [PubMed]
- Nealon, J.; Taurel, A.-F.; Yoksan, S.; Moureau, A.; Bonaparte, M.; Quang, L.C.; Capeding, M.R.; Prayitno, A.; Hadinegoro, S.R.; Chansinghakul, D.; et al. Serological evidence of Japanese encephalitis virus circulation in Asian children from dengue-endemic countries. J. Infect. Dis. 2019, 219, 375–381. [Google Scholar] [CrossRef] [PubMed]
- Heffelfinger, J.D.; Li, X.; Batmunkh, N.; Grabovac, V.; Diorditsa, S.; Liyanage, J.B.; Pattamadilok, S.; Bahl, S.; Vannice, K.S.; Hyde, T.B.; et al. Japanese encephalitis surveillance and immunization – Asia and Western Pacific regions, 2016. MMWR Morb Mortal Wkly Rep. 2017, 66, 579–583. [Google Scholar] [CrossRef] [PubMed]
- Yen, N.T.; Duffy, M.R.; Hong, N.M.; Hien, N.T.; Fischer, M.; Hills, S.L. Surveillance for Japanese encephalitis in Vietnam, 1998–2007. Am. J. Trop. Med. Hyg. 2010, 83, 816–819. [Google Scholar] [CrossRef] [PubMed]
- Kou, Z.; Quinn, M.; Chen, H.; Rodrigo, W.W.S.I.; Rose, R.C.; Schlesinger, J.J.; Jin, X. Monocytes, but not T or B cells, are the principal target cells for dengue virus (DV) infection among human peripheral blood mononuclear cells. J. Med. Virol. 2008, 80, 134–146. [Google Scholar] [CrossRef] [PubMed]
- Pham, A.M.; Langlois, R.A.; tenOever, B.R. Replication in cells of hematopoietic origin is necessary for dengue virus dissemination. PLoS Pathog. 2012, 8, e1002465. [Google Scholar] [CrossRef]
- Ly, M.H.P.; Moi, M.L.; Vu, T.B.H.; Tun, M.M.N.; Saunders, T.; Nguyen, C.N.; Nguyen, A.K.T.; Nguyen, H.M.; Dao, T.H.; Pham, D.Q.; et al. Dengue virus infection-enhancement activity in neutralizing antibodies of healthy adults before dengue season as determined by using FcγR-expressing cells. BMC Infect. Dis. 2018, 18, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Moi, M.L.; Lim, C.K.; Kotaki, A.; Takasaki, T.; Kurane, I. Discrepancy in dengue virus neutralizing antibody titers between plaque reduction neutralizing tests with Fcγ receptor (FcγR)-negative and FcγR-expressing BHK-21 cells. Clin. Vaccine Immunol. 2010, 17, 402–407. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Moi, M.L.; Lim, C.K.; Kotaki, A.; Takasaki, T.; Kurane, I. Detection of higher levels of dengue viremia using FcγR-expressing BHK-21 cells than FcγR-negative cells in secondary infection but not in primary infection. J. Infect. Dis. 2011, 203, 1405–1414. [Google Scholar] [CrossRef] [PubMed]
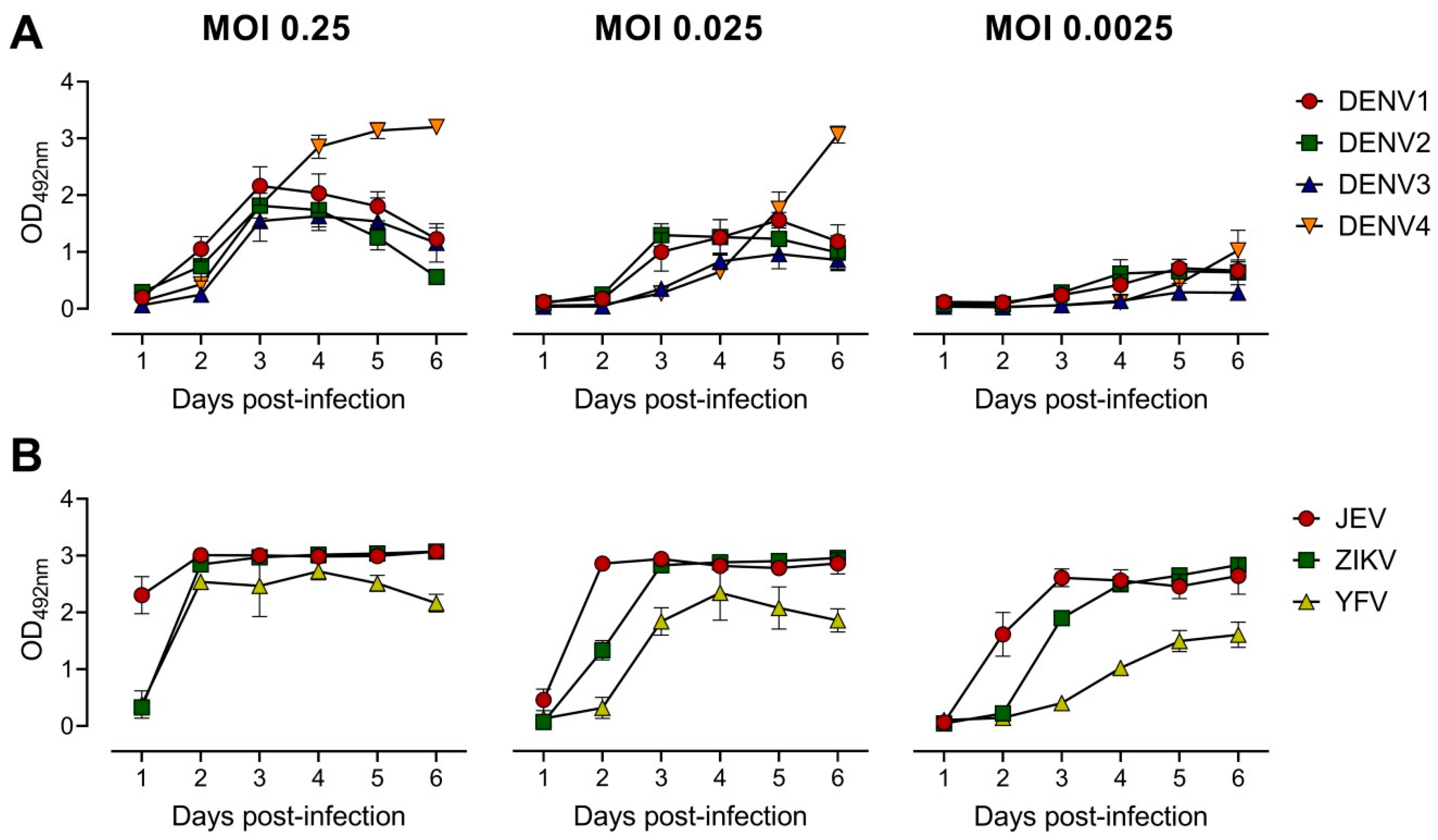
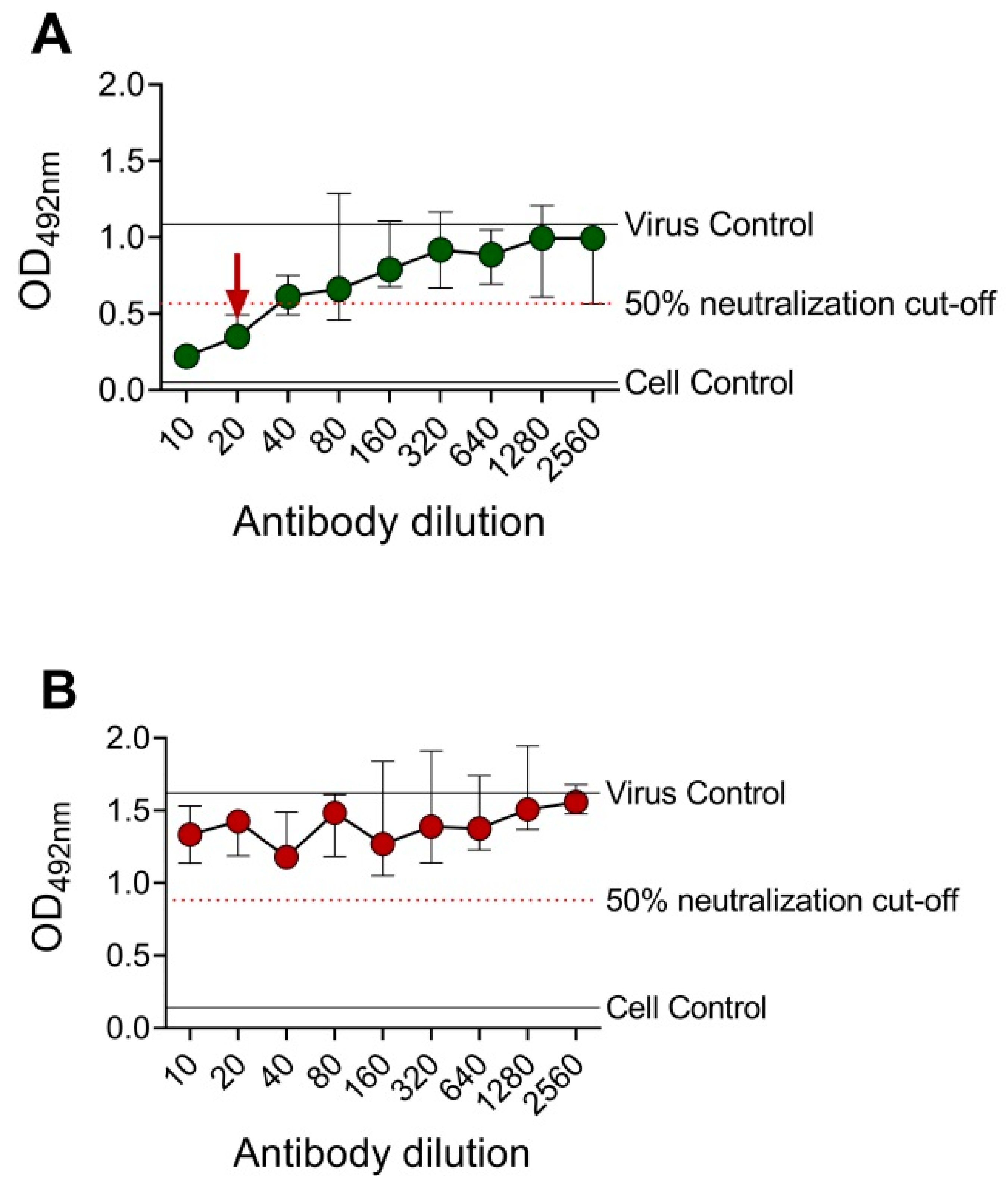
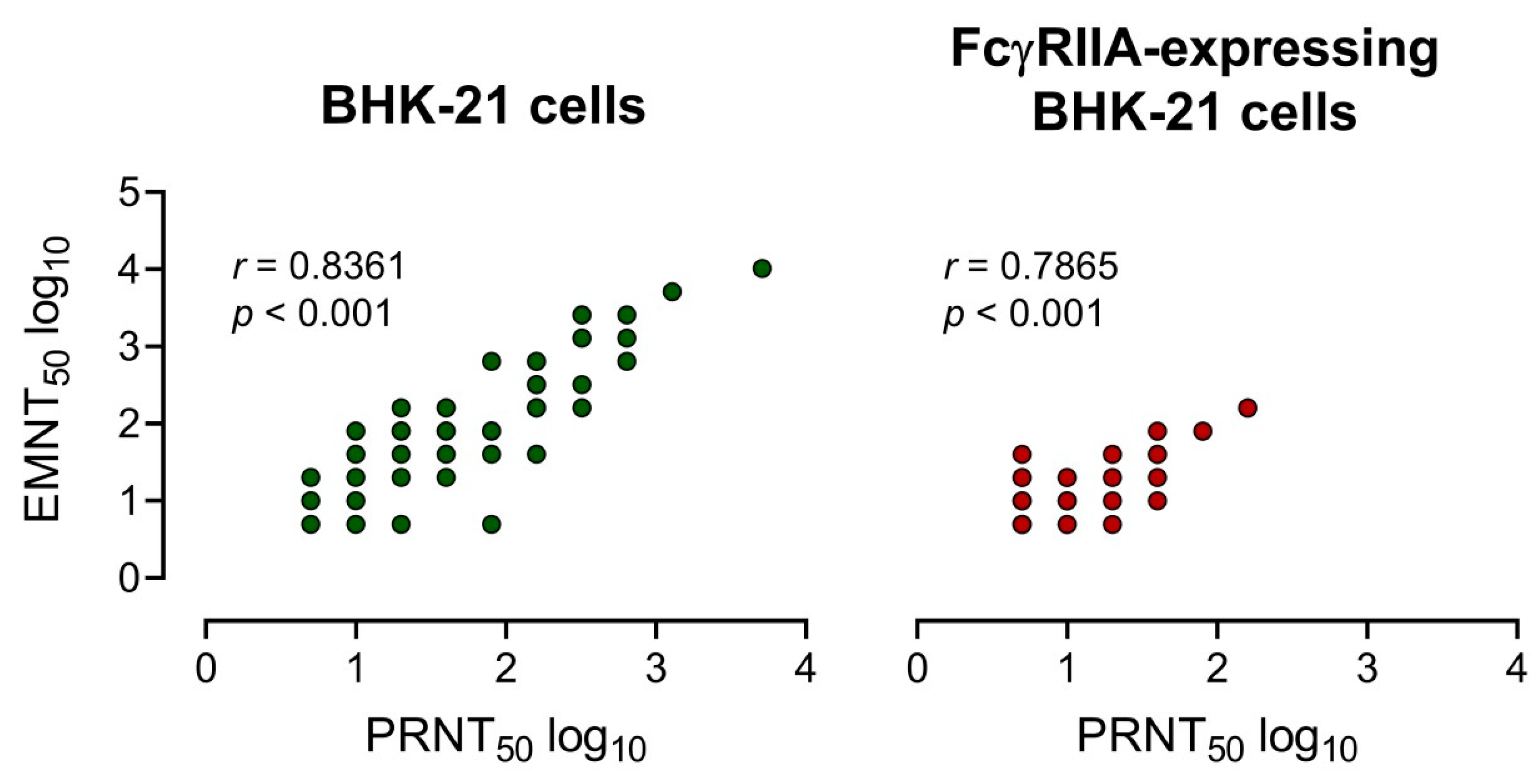
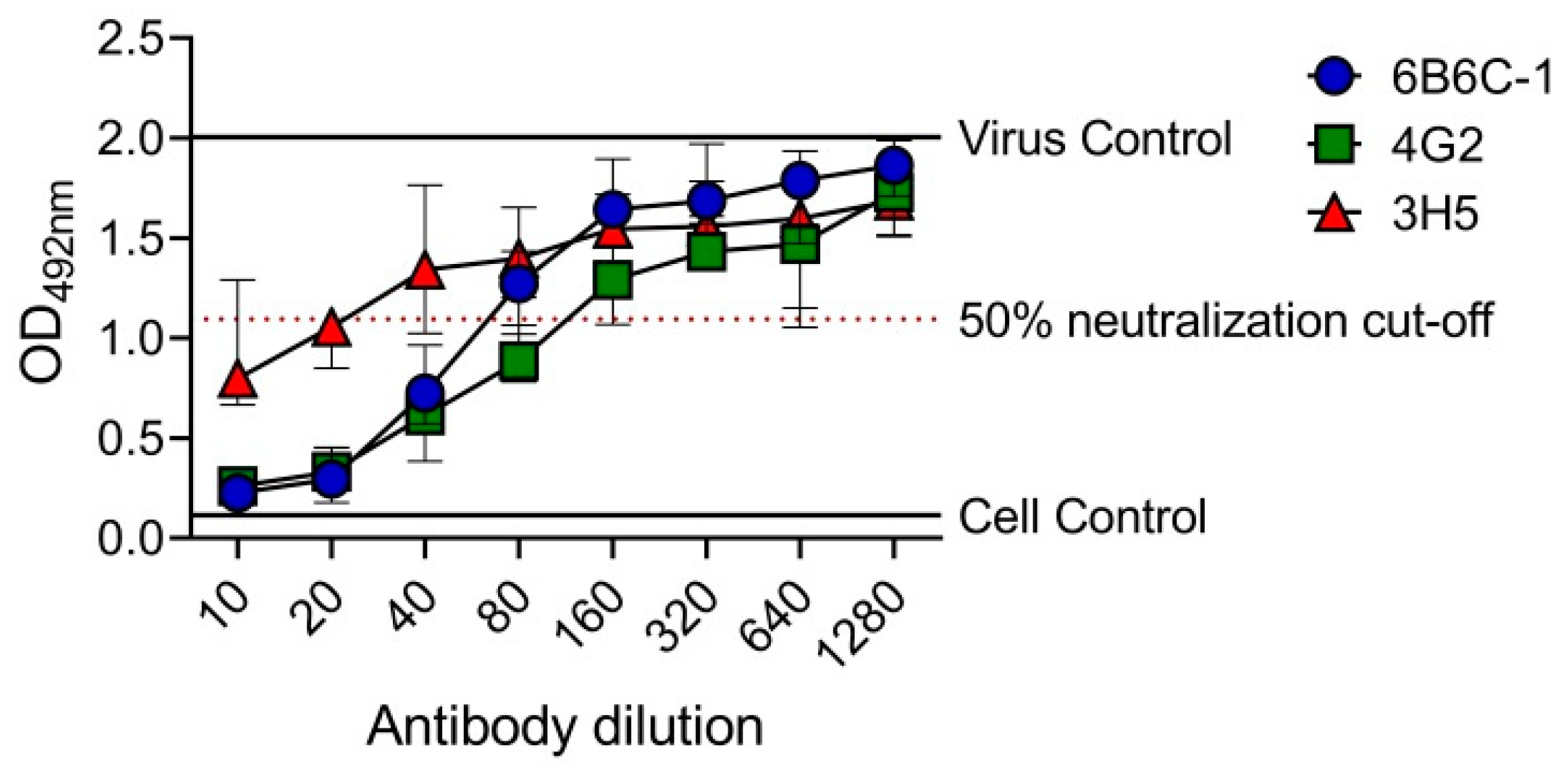
| Virus | Flavivirus Cross-Reactive Monoclonal Antibody | DENV-2 Type-Specific Monoclonal Antibody | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| mAb 6B6C-1 | mAb 4G2 | mAb 3H5 | ||||||||||
| BHK-21 | FcγR-BHK-21 | BHK-21 | FcγR-BHK-21 | BHK-21 | FcγR-BHK-21 | |||||||
| EMNT a | PRNT b | EMNT | PRNT | EMNT | PRNT | EMNT | PRNT | EMNT | PRNT | EMNT | PRNT | |
| DENV-1 | 40 | 40 | 10 | <10 c | 40 | 40 | 10 | <10 | <10 | <10 | <10 | <10 |
| DENV-2 | 80 | 40 | 20 | 10 | 80 | 80 | 10 | 10 | 20 | 40 | <10 | <10 |
| DENV-3 | 20 | 10 | <10 | <10 | 10 | 10 | <10 | <10 | <10 | <10 | <10 | <10 |
| DENV-4 | 40 | 20 | <10 | <10 | 40 | 80 | <10 | <10 | <10 | <10 | <10 | <10 |
| JEV | 20 | <10 | 20 | <10 | 20 | <10 | 10 | <10 | <10 | <10 | <10 | <10 |
| ZIKV | 80 | <10 | 80 | <10 | 80 | <10 | 40 | <10 | <10 | <10 | <10 | <10 |
| YFV | 320 | <10 | 40 | <10 | 160 | <10 | 80 | <10 | <10 | <10 | <10 | <10 |
| Sample Code | DENV-1 | DENV-2 | DENV-3 | DENV-4 | JEV | |||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| BHK-21 | FcγR-BHK-21 | BHK-21 | FcγR-BHK-21 | BHK-21 | FcγR-BHK-21 | BHK-21 | FcγR-BHK-21 | BHK-21 | FcγR-BHK-21 | |||||||||||
| EMNT a | PRNT b | EMNT | PRNT | EMNT | PRNT | EMNT | PRNT | EMNT | PRNT | EMNT | PRNT | EMNT | PRNT | EMNT | PRNT | EMNT | PRNT | EMNT | PRNT | |
| HN.15.001/1 | 20 | <10 c | 20 | <10 | <10 | <10 | <10 | <10 | <10 | 10 | <10 | <10 | 80 | 20 | <10 | <10 | 320 | 320 | 160 | 160 |
| HN.15.018/1 | <10 | <10 | 10 | 10 | 20 | 40 | <10 | <10 | <10 | <10 | <10 | <10 | 160 | 40 | <10 | <10 | 20 | 40 | <10 | 10 |
| HN.15.022/1 | 40 | 20 | 40 | <10 | 80 | 80 | 10 | 40 | 20 | 40 | 10 | <10 | 80 | 20 | <10 | <10 | 80 | 20 | <10 | <10 |
| HN.15.056/1 | 20 | 40 | 40 | 20 | 10 | <10 | <10 | <10 | <10 | <10 | <10 | <10 | 20 | <10 | <10 | <10 | 160 | 160 | 80 | 80 |
| HN.15.068/1 | <10 | 20 | 10 | 10 | 80 | 80 | <10 | 10 | <10 | <10 | <10 | <10 | 80 | 20 | <10 | <10 | 40 | 40 | <10 | <10 |
| HN.15.071/1 | <10 | 10 | <10 | <10 | 80 | 80 | <10 | 10 | <10 | <10 | <10 | <10 | 40 | 10 | <10 | <10 | <10 | <10 | <10 | <10 |
| HN.15.082/1 | 320 | 160 | 160 | 160 | 20 | 40 | 10 | 20 | 160 | 160 | 40 | 40 | 160 | 20 | 10 | <10 | 160 | 160 | 80 | 40 |
| HN.15.084/1 | 640 | 160 | 160 | 160 | <10 | <10 | <10 | <10 | 160 | 160 | <10 | <10 | 80 | 10 | <10 | <10 | <10 | <10 | <10 | <10 |
| HN.15.086/1 | 160 | 160 | 160 | 160 | 40 | 160 | 20 | 40 | 80 | 80 | 20 | 10 | 320 | 160 | 20 | 20 | 40 | 20 | <10 | <10 |
| HN.15.097/1 | 640 | 320 | 160 | 160 | 40 | 40 | 40 | 40 | 160 | 160 | 80 | 80 | 320 | 160 | 20 | 10 | 40 | 20 | 10 | 10 |
| HN.15.011/1 | 640 | 640 | 160 | 160 | 80 | 40 | 40 | 20 | NT | NT | NT | NT | NT | NT | NT | NT | 640 | 160 | 40 | 40 |
| HN.15.026/1 | 20 | 40 | <10 | <10 | 160 | 160 | <10 | 20 | NT | NT | NT | NT | NT | NT | NT | NT | <10 | <10 | <10 | <10 |
| Neutralizing Titers to ZIKV | ||
|---|---|---|
| Sample Code | EMNT a | PRNT b |
| Z67 SRII | 2560 | 640 |
| Z68 SRII | 2560 | 320 |
| Z77 | 1280 | 320 |
| Z78 | 10240 | 5120 |
| Z79 | 1280 | 640 |
| Z84 | 2560 | 320 |
| Z78H2 | 5120 | 1280 |
| Z79H2 | 1280 | 320 |
| Z120 | <20 c | <20 |
| Z123 | 640 | 80 |
| Z125 | 40 | 20 |
| Z126 | 20 | 20 |
| Z129 | 40 | 40 |
| Monoclonal Antibody | 384-well | 96-well |
|---|---|---|
| 6B6C-1 | 40 | 80 |
| 4G2 | 80 | 80 |
| 3H5 | 20 | 20 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Balingit, J.C.; Phu Ly, M.H.; Matsuda, M.; Suzuki, R.; Hasebe, F.; Morita, K.; Moi, M.L. A Simple and High-Throughput ELISA-Based Neutralization Assay for the Determination of Anti-Flavivirus Neutralizing Antibodies. Vaccines 2020, 8, 297. https://doi.org/10.3390/vaccines8020297
Balingit JC, Phu Ly MH, Matsuda M, Suzuki R, Hasebe F, Morita K, Moi ML. A Simple and High-Throughput ELISA-Based Neutralization Assay for the Determination of Anti-Flavivirus Neutralizing Antibodies. Vaccines. 2020; 8(2):297. https://doi.org/10.3390/vaccines8020297
Chicago/Turabian StyleBalingit, Jean Claude, Minh Huong Phu Ly, Mami Matsuda, Ryosuke Suzuki, Futoshi Hasebe, Kouichi Morita, and Meng Ling Moi. 2020. "A Simple and High-Throughput ELISA-Based Neutralization Assay for the Determination of Anti-Flavivirus Neutralizing Antibodies" Vaccines 8, no. 2: 297. https://doi.org/10.3390/vaccines8020297
APA StyleBalingit, J. C., Phu Ly, M. H., Matsuda, M., Suzuki, R., Hasebe, F., Morita, K., & Moi, M. L. (2020). A Simple and High-Throughput ELISA-Based Neutralization Assay for the Determination of Anti-Flavivirus Neutralizing Antibodies. Vaccines, 8(2), 297. https://doi.org/10.3390/vaccines8020297






