Micelle-Based Adjuvants for Subunit Vaccine Delivery
Abstract
:1. Introduction
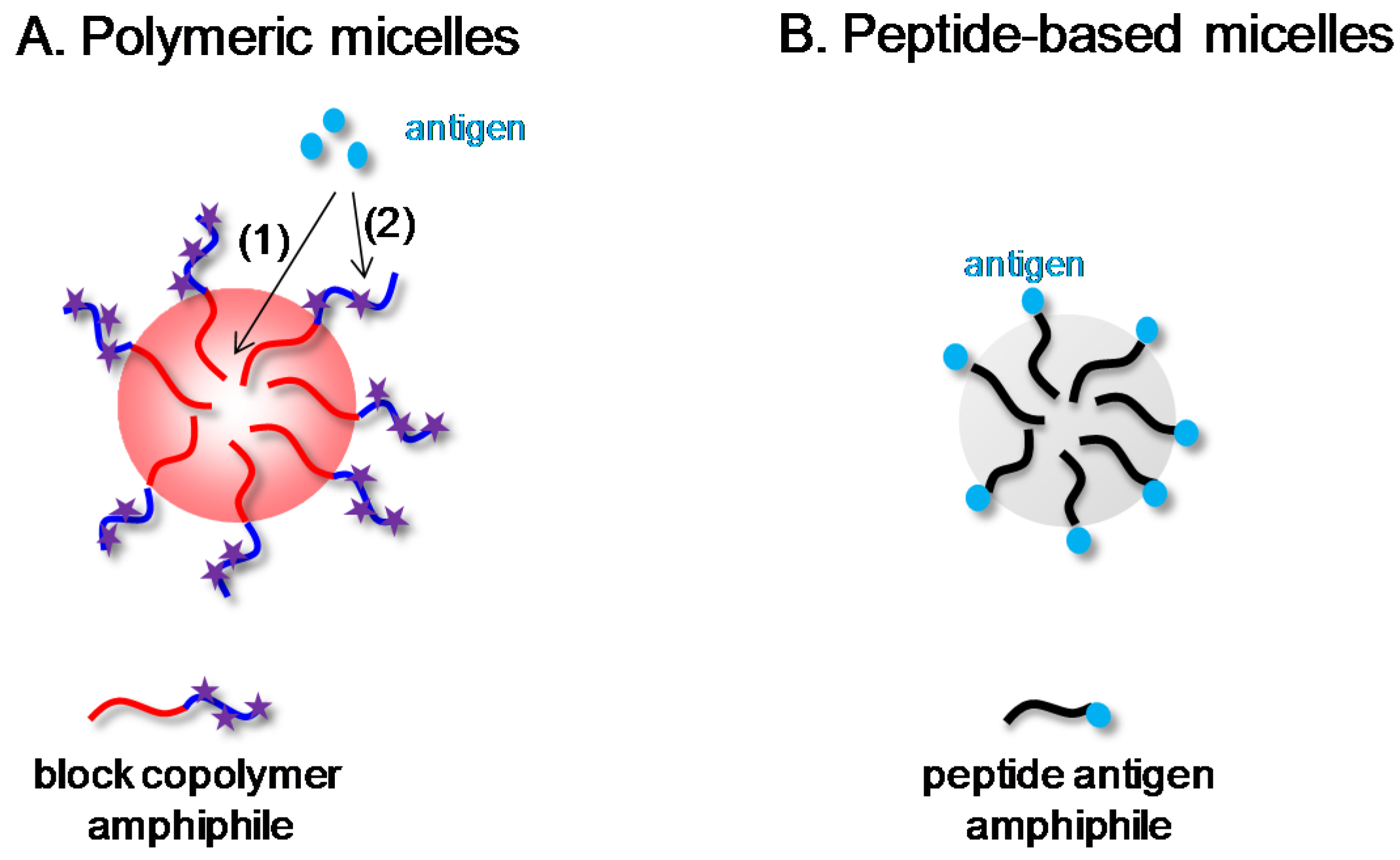
2. Polymeric Micelles
2.1. Polylactide (PLA) Based Micelles
2.2. Polypeptide Based Micelles
2.3. pH Responsive Micelles
2.4. Other Polymeric Micelles
2.5. DNA Vaccination with Polymeric Micelles
3. Peptide Amphiphiles
4. Conclusions and Outlooks
Acknowledgments
Author Contribution
Conflicts of Interest
References
- De Gregorio, E.; Rappuoli, R. From empiricism to rational design: A personal perspective of the evolution of vaccine development. Nat. Rev. Immunol. 2014, 14, 505–514. [Google Scholar] [CrossRef] [PubMed]
- Alving, C.R.; Peachman, K.K.; Rao, M.; Reed, S.G. Adjuvants for human vaccines. Curr. Opin. Immunol. 2012, 24, 310–315. [Google Scholar] [CrossRef] [PubMed]
- Fox, C.B.; Haensler, J. An update on safety and immunogenicity of vaccines containing emulsion-based adjuvants. Expert Rev. Vaccines 2013, 12, 747–758. [Google Scholar] [CrossRef] [PubMed]
- Gregoriadis, G.; McCormack, B.; Obrenovic, M.; Saffie, R.; Zadi, B.; Perrie, Y. Liposomes as immunological adjuvants and vaccine carriers. Methods 1999, 19, 156–162. [Google Scholar] [CrossRef] [PubMed]
- Rice-Ficht, A.C.; Arenas-Gamboa, A.M.; Kahl-McDonagh, M.M.; Ficht, T.A. Polymeric particles in vaccine delivery. Curr. Opin. Microbiol. 2010, 13, 106–112. [Google Scholar] [CrossRef] [PubMed]
- Croy, S.R.; Kwon, G.S. Polymeric micelles for drug delivery. Curr. Pharm. Des. 2006, 12, 4669–4684. [Google Scholar] [CrossRef] [PubMed]
- Luo, Z.; Shi, S.; Jin, L.; Xu, L.; Yu, J.; Chen, H.; Li, X. Cationic micelle based vaccine induced potent humoral immuneresponse through enhancing antigen uptake and formation of germinal center. Colloids Surf. B 2015, 135, 556–564. [Google Scholar] [CrossRef] [PubMed]
- Moyano, D.F.; Goldsmith, M.; Solfiell, D.J.; Landesman-Milo, D.; Miranda, O.R.; Peer, D.; Rotello, V.M. Nanoparticle hydrophobicity dictates immune response. J. Am. Chem. Soc. 2012, 134, 3965–3967. [Google Scholar] [CrossRef] [PubMed]
- Hara, E.; Ueda, M.; Makino, A.; Hara, I.; Ozeki, E.; Kimura, S. Factors influencing in vivo disposition of polymeric micelles on multiple administrations. ACS Med. Chem. Lett. 2014, 5, 873–877. [Google Scholar] [CrossRef] [PubMed]
- Kim, C.J.; Hara, E.; Shimizu, A.; Sugai, M.; Kimura, S. Activation of B1a cells in peritoneal cavity by T cell-independent antigen expressed on polymeric micelle. J. Pharm. Sci. 2015, 104, 1839–1847. [Google Scholar] [CrossRef] [PubMed]
- Hara, E.; Ueda, M.; Kim, C.J.; Makino, A.; Hara, I.; Ozeki, E.; Kimura, S. Suppressive immune response of poly(sarcosine) chains in peptide-nanosheets in contrast to polymeric micelles. J. Pept. Sci. 2014, 20, 570–577. [Google Scholar] [CrossRef] [PubMed]
- Aguado, M.T.; Lambert, P.H. Controlled-release vaccines-biodegradable polylactide/polyglycolide (PL/PG) microspheres as antigen vehicles. Immunobiology 1992, 184, 113–125. [Google Scholar] [CrossRef]
- Pavot, V.; Rochereau, N.; Primard, C.; Genin, C.; Perouzel, E.; Lioux, T.; Paul, S.; Verrier, B. Encapsulation of Nod1 and Nod2 receptor ligands into poly(lactic acid) nanoparticles potentiates their immune properties. J. Control. Release 2013, 167, 60–67. [Google Scholar] [CrossRef] [PubMed]
- Jiménez-Sánchez, G.; Pavot, V.; Chane-Haong, C.; Handké, N.; Terrat, C.; Gigmes, D.; Trimaille, T.; Verrier, B. Preparation and In Vitro Evaluation of Imiquimod loaded polylactide-based micelles as potential vaccine adjuvants. Pharm. Res. 2015, 32, 311–320. [Google Scholar] [CrossRef] [PubMed]
- Jain, A.K.; Goyal, A.K.; Gupta, P.N.; Khatri, K.; Mishra, N.; Mehta, A.; Mangal, S.; Vyas, S.P. Synthesis, characterization and evaluation of novel triblock copolymer based nanoparticles for vaccine delivery against hepatitis B. J. Control. Release 2009, 136, 161–169. [Google Scholar] [CrossRef] [PubMed]
- Jain, A.K.; Goyal, A.K.; Mishra, N.; Vaidya, B.; Mangal, S.; Vyas, S.P. PEG-PLA-PEG block copolymeric nanoparticles for oral immunization against hepatitis B. Int. J. Pharm. 2010, 387, 253–262. [Google Scholar] [CrossRef] [PubMed]
- Luo, Z.; Li, P.; Deng, J.; Gao, N.; Zhang, Y.; Pan, H.; Liu, L.; Wang, C.; Cai, L.; Ma, Y. Cationic polypeptide micelle-based antigen delivery system: A simple and robust adjuvant to improve vaccine efficacy. J. Control. Release 2013, 170, 259–267. [Google Scholar] [CrossRef] [PubMed]
- Luo, Z.; Wang, C.; Yi, H.; Li, P.; Pan, H.; Liu, L.; Cai, L.; Ma, Y. Nanovaccine loaded with poly I:C and STAT3 siRNA robustly elicits anti-tumor immune responses through modulating tumor-associated dendritic cells in vivo. Biomaterials 2015, 38, 50–60. [Google Scholar] [CrossRef] [PubMed]
- Noh, Y.W.; Hong, J.H.; Shim, S.M.; Park, H.S.; Bae, H.H.; Ryu, E.K.; Hwang, J.H.; Lee, C.H.; Cho, S.H.; Sung, M.H.; et al. Polymer nanomicelles for efficient mucus delivery and antigen-specific high mucosal immunity. Angew. Chem. Int. Ed. 2013, 52, 7684–7689. [Google Scholar] [CrossRef] [PubMed]
- Hyungjin, K.; Tomofumi, U.; Takami, A.; Masanori, B.; Mitsuru, A. Amphiphilic Poly(amino acid) nanoparticles induce size-dependent dendritic cell maturation. Adv. Funct. Mater. 2010, 20, 3925–3931. [Google Scholar]
- Matsuo, K.; Koizumi, H.; Akashi, M.; Nakagawa, S.; Fujita, T.; Yamamoto, A.; Okada, N. Intranasal immunization with poly(γ-glutamic acid) nanoparticles entrapping antigenic proteins can induce potent tumor immunity. J. Control. Release 2011, 152, 310–316. [Google Scholar] [CrossRef] [PubMed]
- Okamoto, S.; Yoshii, H.; Matsuura, M.; Kojima, A.; Ishikawa, T.; Akagi, T.; Akashi, M.; Takahashi, M.; Yamanishi, K.; Mori, Y. Poly-γ-glutamic acid nanoparticles and aluminium adjuvant used as an adjuvant in a single dose of Japanese Encephalitis virus-like particles provide effective protection from Japanese Encephalitis virus. Clin. Vaccine Immunol. 2012, 19, 17–22. [Google Scholar] [CrossRef] [PubMed]
- Hao, J.; Kwissa, M.; Pulendran, B.; Murthy, N. Peptide crosslinked micelles: A new strategy for the design and synthesis of peptide vaccines. Int. J. Nanomed. 2006, 1, 97–103. [Google Scholar] [CrossRef]
- Keller, S.; Wilson, J.T.; Patilea, G.I.; Kern, H.B.; Convertine, A.J.; Stayton, P.S. Neutral polymer micelle carriers with pH-responsive, endosome-releasing activity modulate antigen trafficking to enhance CD8+ T cell responses. J. Control. Release 2014, 191, 24–33. [Google Scholar] [CrossRef] [PubMed]
- Wilson, J.T.; Keller, S.; Manganiello, M.J.; Cheng, C.; Lee, C.C.; Opara, C.; Convertine, A.; Stayton, P.S. pH-Responsive nanoparticle vaccines for dual-delivery of antigens and immunostimulatory oligonucleotides. ACS Nano 2013, 7, 3912–3925. [Google Scholar] [CrossRef] [PubMed]
- Boudier, A.; Aubert-Pouessel, A.; Louis-Plence, P.; Gerardin, C.; Jorgensen, C.; Devoisselle, J.M.; Begu, S. The control of dendritic cell maturation by pH-sensitive polyion complex micelles. Biomaterials 2009, 30, 233–241. [Google Scholar] [CrossRef] [PubMed]
- Boudier, A.; Aubert-Pouessel, A.; Mebarek, N.; Chavanieu, A.; Quentin, J.; Martire, D.; Boukhaddaoui, H.; Gerardin, C.; Jorgensen, C.; Devoisselle, J.M.; et al. Development of tripartite polyion micelles for efficient peptide delivery into dendritic cells without altering their plasticity. J. Control. Release 2011, 154, 156–163. [Google Scholar] [CrossRef] [PubMed]
- Eby, J.K.; Dane, K.Y.; O’Neil, C.P.; Hirosue, S.; Swartz, M.A.; Hubbell, J.A. Polymer micelles with pyridyl disulfide-coupled antigen travel through lymphatics and show enhanced cellular responses following immunization. Acta Biomater. 2012, 8, 3210–3217. [Google Scholar] [CrossRef] [PubMed]
- Zeng, Q.; Jiang, H.; Wang, T.; Zhang, Z.; Gong, T.; Sun, X. Cationic micelle delivery of Trp2 peptide for efficient lymphatic draining and enhanced cytotoxic T-lymphocyte responses. J. Control. Release 2015, 200, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Layek, B.; Lipp, L.; Singh, J. APC targeted micelle for enhanced intradermal delivery of hepatitis B DNA vaccine. J. Control. Release 2015, 207, 143–153. [Google Scholar] [CrossRef] [PubMed]
- Yang, Y.; Kuang, Y.; Liu, Y.; Li, W.; Jiang, Z.; Xiao, L.; Li, M. Immunogenicity of multiple-epitope antigen gene of HCV carried by novel biodegradable polymers. Comp. Immunol. Microbiol. Infect. Dis. 2011, 34, 65–72. [Google Scholar] [CrossRef] [PubMed]
- Furugaki, K.; Cui, L.; Kunisawa, Y.; Osada, K.; Shinkai, K.; Tanaka, M.; Kataoka, K.; Nakano, K. Intraperitoneal administration of a tumor-associated antigen SART3, CD40L, and GM-CSF gene-loaded polyplex micelle elicits a vaccine effect in mouse tumor models. PLoS ONE 2014, 9, e101854. [Google Scholar] [CrossRef] [PubMed]
- Cui, L.; Osada, K.; Imaizumi, A.; Kataoka, K.; Nakano, K. Feasibility of a subcutaneously administered block/homo-mixed polyplex micelle as a carrier for DNA vaccination in a mouse tumor model. J. Control. Release 2015, 206, 220–231. [Google Scholar] [CrossRef] [PubMed]
- Miyata, K.; Oba, M.; Nakanishi, M.; Fukushima, S.; Yamasaki, Y.; Koyama, H.; Nishiyama, N.; Kataoka, K. Polyplexes from poly(aspartamide) bearing 1,2-diaminoethane side chains induce pH-selective, endosomal membrane destabilization with amplified transfection and negligible cytotoxicity. J. Am. Chem. Soc. 2008, 130, 16287–16294. [Google Scholar] [CrossRef] [PubMed]
- Zhao, M.; Li, M.; Zhang, Z.; Gong, T.; Sun, X. Induction of HIV-1 gag specific immune responses by cationic micelles mediated delivery of gag mRNA. Drug Deliv. 2015. [Google Scholar] [CrossRef] [PubMed]
- Black, M.; Trent, A.; Kostenko, Y.; Lee, J.S.; Olive, C.; Tirrell, M. Self-assembled peptide amphiphile micelles containing a cytotoxic T-cell epitope promote a protective immune response in vivo. Adv. Mater. 2012, 24, 3845–3849. [Google Scholar] [CrossRef] [PubMed]
- Trent, A.; Ulery, B.D.; Black, M.J.; Barrett, J.C.; Liang, S.; Kostenko, Y.; David, N.A.; Tirrell, M.V. Peptide amphiphile micelles self-adjuvant group A streptococcal vaccination. AAPS J. 2015, 17, 380–388. [Google Scholar] [CrossRef] [PubMed]
- Accardo, A.; Vitiello, M.; Tesauro, D.; Galdiero, M.; Finamore, E.; Martora, F.; Mansi, R.; Ringhieri, P.; Morelli, G. Self-assembled or mixed peptide amphiphile micelles from Herpes simplex virus glycoproteins as potential immunomodulatory treatment. Int. J. Nanomed. 2014, 9, 2137–2148. [Google Scholar]
- Liu, H.; Moynihan, K.D.; Zheng, Y.; Szeto, G.L.; Li, A.V.; Huang, B.; van Egeren, D.S.; Park, C.; Irvine, D.J. Structure-based programming of lymph-node targeting in molecular vaccines. Nature 2014, 507, 519–522. [Google Scholar] [CrossRef] [PubMed]
- Skwarczynski, M.; Zaman, M.; Urbani, C.N.; Lin, I.C.; Jia, Z.; Batzloff, M.R.; Good, M.F.; Monteiro, M.J.; Toth, I. Polyacrylate dendrimer nanoparticles: A self-adjuvanting vaccine delivery system. Angew. Chem. Int. Ed. 2010, 49, 5742–5745. [Google Scholar] [CrossRef] [PubMed]
- Zope, H.; Quer, C.B.; Bomans, P.H.; Sommerdijk, N.A.; Kros, A.; Jiskoot, W. Peptide amphiphile nanoparticles enhance the immune response against a CpG-adjuvanted influenza antigen. Adv. Healthc. Mater. 2014, 3, 343–348. [Google Scholar] [CrossRef] [PubMed]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Trimaille, T.; Verrier, B. Micelle-Based Adjuvants for Subunit Vaccine Delivery. Vaccines 2015, 3, 803-813. https://doi.org/10.3390/vaccines3040803
Trimaille T, Verrier B. Micelle-Based Adjuvants for Subunit Vaccine Delivery. Vaccines. 2015; 3(4):803-813. https://doi.org/10.3390/vaccines3040803
Chicago/Turabian StyleTrimaille, Thomas, and Bernard Verrier. 2015. "Micelle-Based Adjuvants for Subunit Vaccine Delivery" Vaccines 3, no. 4: 803-813. https://doi.org/10.3390/vaccines3040803
APA StyleTrimaille, T., & Verrier, B. (2015). Micelle-Based Adjuvants for Subunit Vaccine Delivery. Vaccines, 3(4), 803-813. https://doi.org/10.3390/vaccines3040803




