Strategy for Designing a Synthetic Tumor Vaccine: Multi-Component, Multivalency and Antigen Modification
Abstract
:1. Introduction
2. The Challenges of Synthetic Tumor Vaccines
3. Multi-Component Vaccine
3.1. Carrier Proteins in Vaccine Design
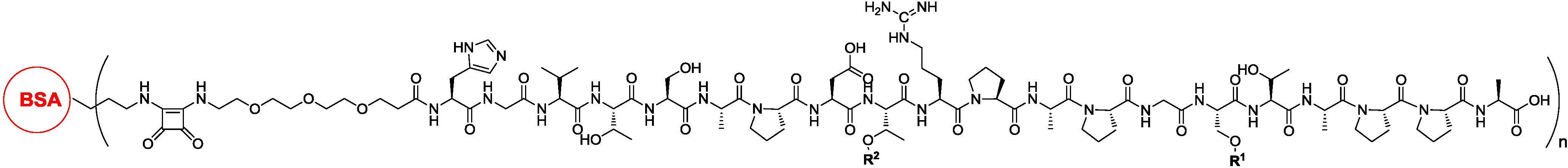
3.2. T-Cell Epitope in Vaccine Design

3.3. Toll-Like Receptor Agonist in Vaccine Design

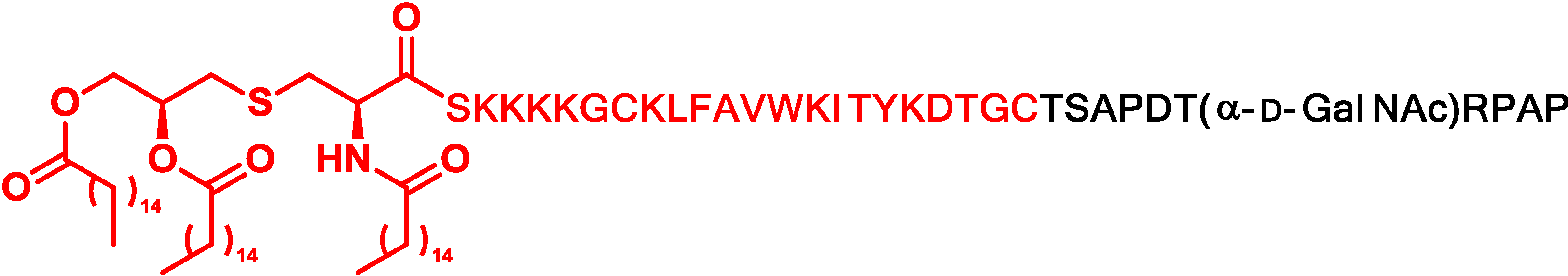
4. Multivalency in Vaccine Design


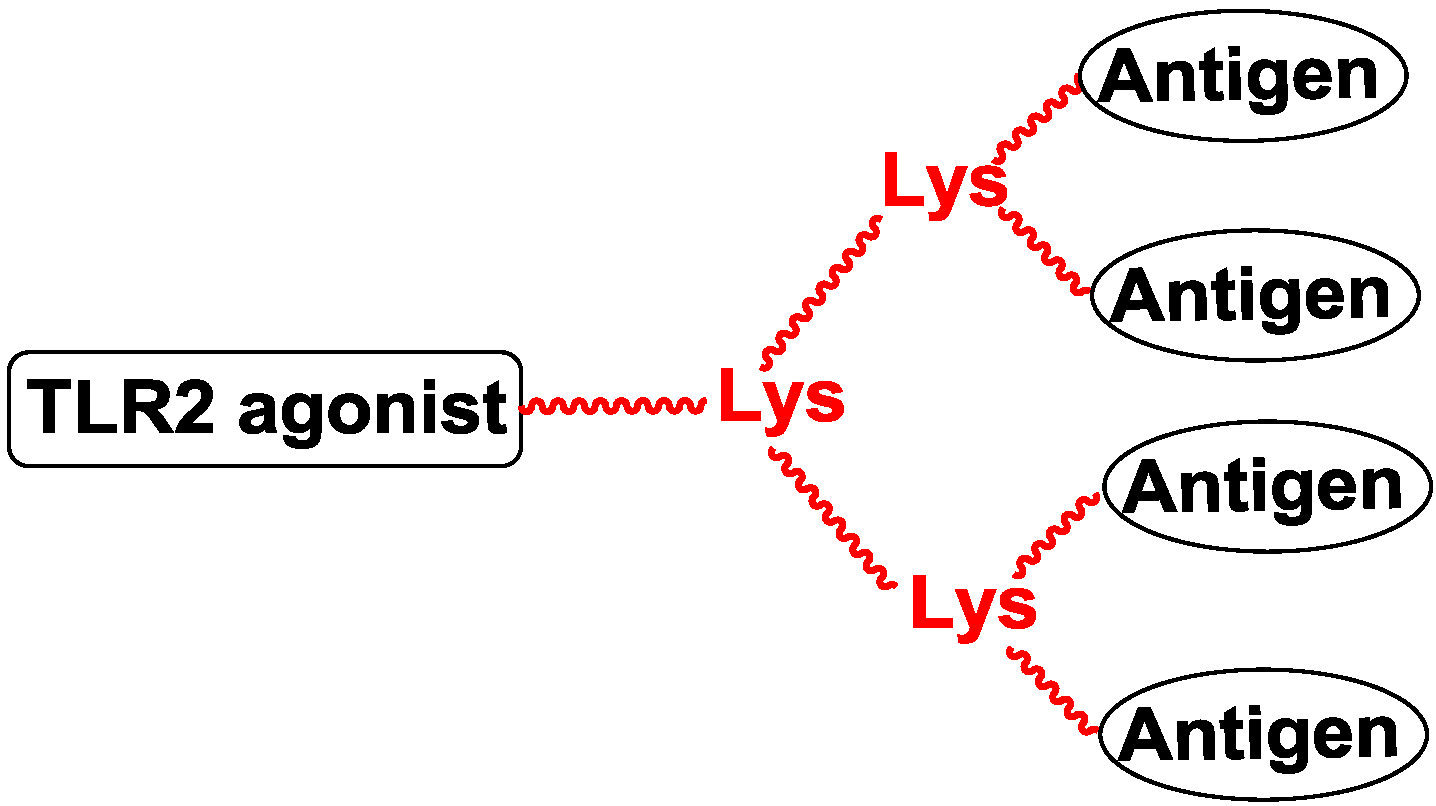
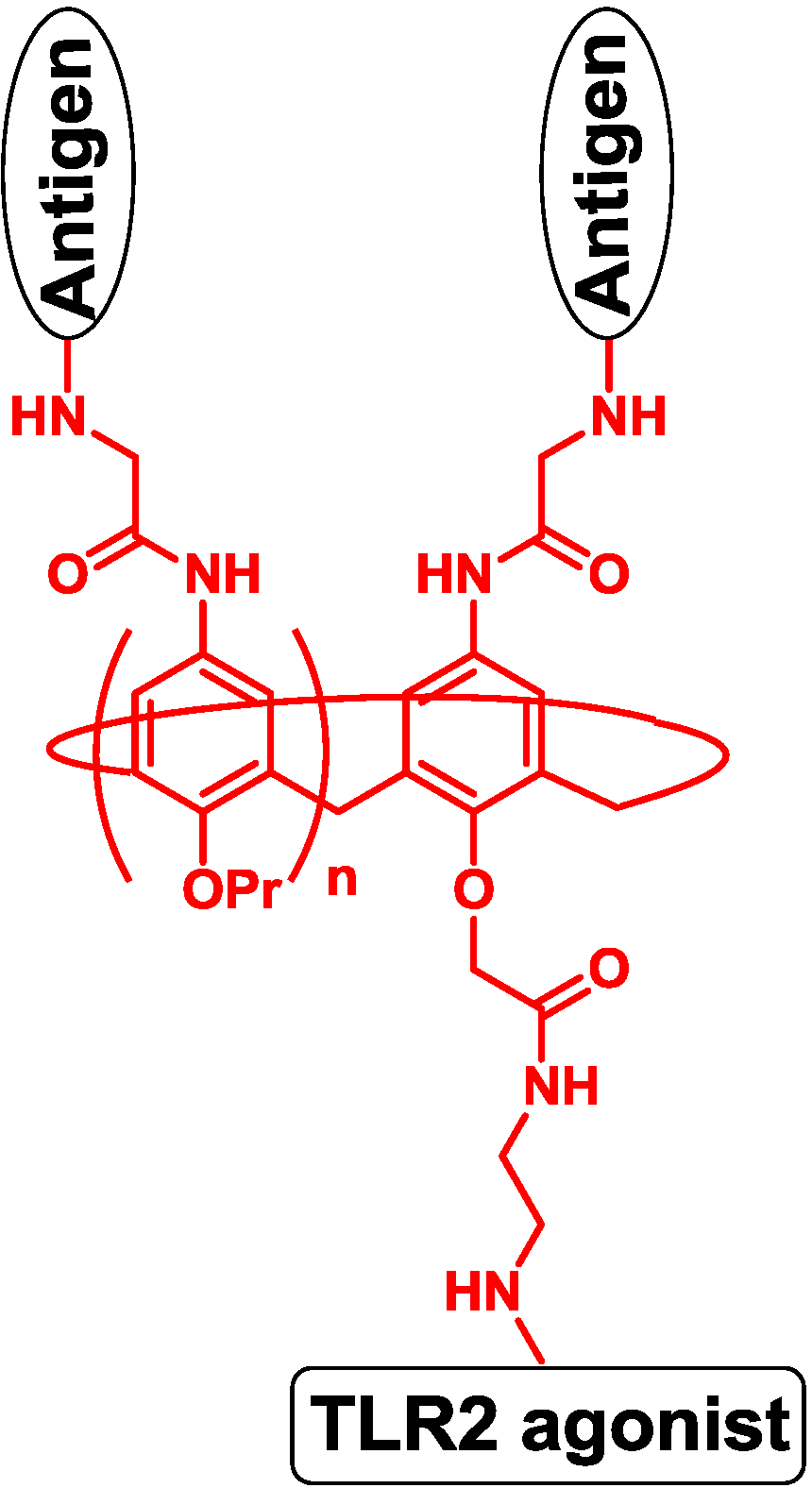
5. Antigen Modification
5.1. Modification in Vitro
5.2. Modification in Vivo
6. Conclusions and Perspective
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Mellman, I.; Coukos, G.; Dranoff, G. Cancer immunotherapy comes of age. Nature 2011, 480, 480–489. [Google Scholar] [CrossRef]
- Waldmann, T.A. The biology of interleukin-2 and interleukin-15: Implications for cancer therapy and vaccine design. Nat. Rev. Immunol. 2006, 6, 595–601. [Google Scholar] [CrossRef]
- Pejawar-Gaddy, S.; Finn, O.J. Cancer vaccines: Accomplishments and challenges. Crit. Rev. Oncol. Hematol. 2008, 67, 93–102. [Google Scholar] [CrossRef]
- Ward, S.; Casey, D.; Labarthe, M.C.; Whelan, M.; Dalgleish, A.; Pandha, H.; Todryk, S. Immunotherapeutic potential of whole tumour cells. Cancer Immunol. Immunother. 2002, 51, 351–357. [Google Scholar]
- Wang, R.F.; Rosenberg, S.A. Human tumor antigens for cancer vaccine development. Immunol. Rev. 1999, 170, 85–100. [Google Scholar] [CrossRef]
- Karsten, U.; von Mensdorff-Pouilly, S.; Goletz, S. What makes muc1 a tumor antigen? Tumor Biol. 2005, 26, 217–220. [Google Scholar] [CrossRef]
- Guo, Z.; Wang, Q. Recent development in carbohydrate-based cancer vaccines. Curr. Opin. Chem. Biol. 2009, 13, 608–617. [Google Scholar] [CrossRef]
- Galonic, D.P.; Gin, D.Y. Chemical glycosylation in the synthesis of glycoconjugate antitumour vaccines. Nature 2007, 446, 1000–1007. [Google Scholar]
- Beatson, R.E.; Taylor-Papadimitriou, J.; Burchell, J.M. MUC1 immunotherapy. Immunotherapy 2010, 2, 305–327. [Google Scholar] [CrossRef]
- Wesseling, J.; van der Valk, S.W.; Vos, H.L.; Sonnenberg, A.; Hilkens, J. Episialin (MUC1) overexpression inhibits integrin-mediated cell adhesion to extracellular matrix components. J. Cell Biol. 1995, 129, 255–265. [Google Scholar] [CrossRef]
- Takada, A.; Ohmori, K.; Yoneda, T.; Tsuyuoka, K.; Hasegawa, A.; Kiso, M.; Kannagi, R. Contribution of carbohydrate antigens sialyl Lewis a and sialyl lewis X to adhesion of human cancer cells to vascular endothelium. Cancer Res. 1993, 53, 354–361. [Google Scholar]
- Senapati, S.; Das, S.; Batra, S.K. Mucin-interacting proteins: From function to therapeutics. Trends Biochem. Sci. 2010, 35, 236–245. [Google Scholar] [CrossRef]
- Li, Y.Q.; Liu, D.; Chen, D.S.; Kharbanda, S.; Kufe, D. Human DF3/MUC1 carcinoma-associated protein functions as an oncogene. Oncogene 2003, 22, 6107–6110. [Google Scholar] [CrossRef]
- Singh, R.; Bandyopadhyay, D. Muc1—A target molecule for cancer therapy. Cancer Biol. Ther. 2007, 6, 481–486. [Google Scholar] [CrossRef]
- Dunn, G.P.; Bruce, A.T.; Ikeda, H.; Old, L.J.; Schreiber, R.D. Cancer immunoediting: From immunosurveillance to tumor escape. Nat. Immunol. 2002, 3, 991–998. [Google Scholar] [CrossRef]
- Guy, B. The perfect mix: Recent progress in adjuvant research. Nat. Rev. Microbiol. 2007, 5, 505–517. [Google Scholar] [CrossRef]
- Kaiser, A.; Gaidzik, N.; Westerlind, U.; Kowalczyk, D.; Hobel, A.; Schmitt, E.; Kunz, H. A synthetic vaccine consisting of a tumor-associated sialyl-T-N-MUC1 tandem-repeat glycopeptide and tetanus toxoid: Induction of a strong and highly selective immune response. Angew. Chem. Int. Ed. 2009, 48, 7551–7555. [Google Scholar] [CrossRef]
- Hoffmann-Roder, A.; Kaiser, A.; Wagner, S.; Gaidzik, N.; Kowalczyk, D.; Westerlind, U.; Gerlitzki, B.; Schmitt, E.; Kunz, H. Synthetic antitumor vaccines from tetanus toxoid conjugates of Muc1 glycopeptides with the Thomsen-Friedenreich antigen and a Fluorine-Substituted analogue. Angew. Chem. Int. Ed. 2010, 49, 8498–8503. [Google Scholar] [CrossRef]
- Gaidzik, N.; Kaiser, A.; Kowalczyk, D.; Westerlind, U.; Gerlitzki, B.; Sinn, H.P.; Schmitt, E.; Kunz, H. Synthetic antitumor vaccines containing MUC1 glycopeptides with two immunodominant domains-induction of a strong immune response against breast tumor tissues. Angew. Chem. Int. Ed. 2011, 50, 9977–9981. [Google Scholar] [CrossRef]
- Cai, H.; Huang, Z.H.; Shi, L.; Sun, Z.Y.; Zhao, Y.F.; Kunz, H.; Li, Y.M. Variation of the glycosylation pattern in MUC1 glycopeptide Bsa vaccines and its influence on the immune response. Angew. Chem. Int. Ed. 2012, 51, 1719–1723. [Google Scholar] [CrossRef]
- Tam, J.P.; Lu, Y.A. Vaccine engineering—Enhancement of immunogenicity of synthetic peptide vaccines related to hepatitis in chemically defined models consisting of T-cell and B-cell epitopes. Proc. Natl. Acad. Sci. USA 1989, 86, 9084–9088. [Google Scholar] [CrossRef]
- Keil, S.; Claus, C.; Dippold, W.; Kunz, H. Towards the development of antitumor vaccines: A synthetic conjugate of a tumor-associated MUC1 glycopeptide antigen and a tetanus toxin epitope. Angew. Chem. Int. Ed. 2001, 40, 366–369. [Google Scholar] [CrossRef]
- Dziadek, S.; Hobel, A.; Schmitt, E.; Kunz, H. A fully synthetic vaccine consisting of a tumor-associated glycopeptide antigen and a T-cell epitope for the induction of a highly specific humoral immune response. Angew. Chem. Int. Ed. 2005, 44, 7630–7635. [Google Scholar] [CrossRef]
- Westerlind, U.; Hobel, A.; Gaidzik, N.; Schmitt, E.; Kunz, H. Synthetic vaccines consisting of tumor-associated MUC1 glycopeptide antigens and a T-cell epitope for the induction of a highly specific humoral immune response. Angew. Chem. Int. Ed. 2008, 47, 7551–7556. [Google Scholar] [CrossRef]
- Cai, H.; Chen, M.S.; Sun, Z.Y.; Zhao, Y.F.; Kunz, H.; Li, Y.M. Self-adjuvanting synthetic antitumor vaccines from MUC1 glycopeptides conjugated to T-cell epitopes from tetanus toxoid. Angew. Chem. Int. Ed. 2013, 52, 6106–6110. [Google Scholar]
- Trinchieri, G.; Sher, A. Cooperation of toll-like receptor signals in innate immune defence. Nat. Rev. Immunol. 2007, 7, 179–190. [Google Scholar] [CrossRef]
- Lemaitre, B. Landmark—The Road to Toll. Nat. Rev. Immunol. 2004, 4, 521–527. [Google Scholar] [CrossRef]
- Akira, S.; Takeda, K. Toll-like receptor signalling. Nat. Rev. Immunol. 2004, 4, 499–511. [Google Scholar] [CrossRef]
- Kaisho, T.; Akira, S. Toll-like receptors as adjuvant receptors. Biochim. Biophys. Acta 2002, 1589, 1–13. [Google Scholar] [CrossRef]
- Akira, S.; Takeda, K.; Kaisho, T. Toll-like receptors: Critical proteins linking innate and acquired immunity. Nat. Immunol. 2001, 2, 675–680. [Google Scholar] [CrossRef]
- Wang, Q.; Zhou, Z.; Tang, S.; Guo, Z. Carbohydrate-monophosphoryl lipid a conjugates are fully synthetic self-adjuvanting cancer vaccines eliciting robust immune responses in the mouse. ACS Chem. Biol. 2012, 7, 235–240. [Google Scholar] [CrossRef]
- Erridge, C.; Bennett-Guerrero, E.; Poxton, I.R. Structure and function of lipopolysaccharides. Microbes Infect. 2002, 4, 837–851. [Google Scholar]
- Reichel, F.; Roelofsen, A.M.; Geurts, H.P.M.; Hamalainen, T.I.; Feiters, M.C.; Boons, G.J. Stereochemical dependence of the self-assembly of the immunoadjuvants Pam(3)Cys and Pam(3)Cys-Ser. J. Am. Chem. Soc. 1999, 121, 7989–7997. [Google Scholar] [CrossRef]
- Ingale, S.; Awolfert, M.; Gaekwad, J.; Buskas, T.; Boons, G.J. Robust immune responses elicited by a fully synthetic three-component vaccine. Nat. Chem. Biol. 2007, 3, 663–667. [Google Scholar] [CrossRef]
- Ingale, S.; Wolfert, M.A.; Buskas, T.; Boons, G.J. Increasing the antigenicity of synthetic tumor-associated carbohydrate antigens by targeting toll-like receptors. Chembiochem 2009, 10, 455–463. [Google Scholar] [CrossRef]
- Lakshminarayanan, V.; Thompson, P.; Wolfert, M.A.; Buskas, T.; Bradley, J.M.; Pathangey, L.B.; Madsen, C.S.; Cohen, P.A.; Gendler, S.J.; Boons, G.-J. Immune recognition of tumor-associated mucin MUC1 is achieved by a fully synthetic aberrantly glycosylated MUC1 tripartite vaccine. Proc. Natl. Acad. Sci. USA 2012, 109, 261–266. [Google Scholar] [CrossRef]
- Wilkinson, B.L.; Day, S.; Malins, L.R.; Apostolopoulos, V.; Payne, R.J. Self-adjuvanting multicomponent cancer vaccine candidates combining per-glycosylated MUC1 glycopeptides and the toll-like receptor 2 agonist Pam(3)Cysser. Angew. Chem. Int. Ed. 2011, 50, 1635–1639. [Google Scholar] [CrossRef]
- Kaiser, A.; Gaidzik, N.; Becker, T.; Menge, C.; Groh, K.; Cai, H.; Li, Y.M.; Gerlitzki, B.; Schmitt, E.; Kunz, H. Fully synthetic vaccines consisting of tumor-associated MUC1 glycopeptides and a lipopeptide ligand of the toll-like receptor 2. Angew. Chem. Int. Ed. 2010, 49, 3688–3692. [Google Scholar] [CrossRef]
- Cai, H.; Sun, Z.Y.; Huang, Z.H.; Shi, L.; Zhao, Y.F.; Kunz, H.; Li, Y.M. Fully synthetic self-adjuvanting thioether-conjugated glycopeptidelipopeptide antitumor vaccines for the induction of complement-dependent cytotoxicity against tumor cells. Chem. Eur. J. 2013, 19, 1962–1970. [Google Scholar] [CrossRef]
- Abdel-Aal, A.B.; El-Naggar, D.; Zaman, M.; Batzloff, M.; Toth, I. Design of fully synthetic, self-adjuvanting vaccine incorporating the tumor-associated carbohydrate Tn antigen and lipoamino acid-based toll-like receptor 2 ligand. J. Med. Chem. 2012, 55, 6968–6974. [Google Scholar] [CrossRef]
- Sarkar, S.; Lombardo, S.A.; Herner, D.N.; Talan, R.S.; Wall, K.A.; Sucheck, S.J. Synthesis of a single-molecule l-rhamnose-containing three-component vaccine and evaluation of antigenicity in the presence of anti-l-rhamnose antibodies. J. Am. Chem. Soc. 2010, 132, 17236–17246. [Google Scholar] [CrossRef]
- Sarkar, S.; Salyer, A.C.; Wall, K.A.; Sucheck, S.J. Synthesis and immunological evaluation of a MUC1 glycopeptide incorporated into l-rhamnose displaying liposomes. Bioconjug. Chem. 2013, 24, 363–375. [Google Scholar] [CrossRef]
- Kudryashov, V.; Glunz, P.W.; Williams, L.J.; Hintermann, S.; Danishefsky, S.J.; Lloyd, K.O. Toward optimized carbohydrate-based anticancer vaccines: Epitope clustering, carrier structure, and adjuvant all influence antibody responses to Lewis(Y) conjugates in mice. Proc. Natl. Acad. Sci. USA 2001, 98, 3264–3269. [Google Scholar] [CrossRef]
- Zhang, S.; Walberg, L.A.; Ogata, S.; Itzkowitz, S.H.; Koganty, R.R.; Reddish, M.; Gandhi, S.S.; Longenecker, B.M.; Lloyd, K.O.; Livingston, P.O. Immune sera and monoclonal antibodies define two configurations for the Sialyl Tn tumor antigen. Cancer Res. 1995, 55, 3364–3368. [Google Scholar]
- Nakada, H.; Inoue, M.; Numata, Y.; Tanaka, N.; Funakoshi, I.; Fukui, S.; Mellors, A.; Yamashina, I. Epitopic structure of Tn glycophorin a for an anti-Tn antibody (Mls 128). Proc. Natl. Acad. Sci. USA 1993, 90, 2495–2499. [Google Scholar] [CrossRef]
- Yin, Z.; Comellas-Aragones, M.; Chowdhury, S.; Bentley, P.; Kaczanowska, K.; Benmohamed, L.; Gildersleeve, J.C.; Finn, M.G.; Huang, X. Boosting immunity to small tumor-associated carbohydrates with bacteriophage qbeta capsids. ACS Chem. Biol. 2013, 8, 1253–1262. [Google Scholar] [CrossRef]
- Ragupathi, G.; Koide, F.; Livingston, P.O.; Cho, Y.S.; Endo, A.; Wan, Q.; Spassova, M.K.; Keding, S.J.; Allen, J.; Ouerfelli, O.; et al. Preparation and evaluation of unimolecular pentavalent and hexavalent antigenic constructs targeting prostate and breast cancer: A synthetic route to anticancer vaccine candidates. J. Am. Chem. Soc. 2006, 128, 2715–2725. [Google Scholar] [CrossRef]
- Keding, S.J.; Danishefsky, S.J. Prospects for total synthesis: A vision for a totally synthetic vaccine targeting epithelial tumors. Proc. Natl. Acad. Sci. USA 2004, 101, 11937–11942. [Google Scholar] [CrossRef]
- Lee, D.; Danishefsky, S.J. “Biologic” level structures through chemistry: A total synthesis of a unimolecular pentavalent MUCI glycopeptide construct. Tetrahedron Lett. 2009, 50, 2167–2170. [Google Scholar] [CrossRef]
- Zhu, J.L.; Wan, Q.; Lee, D.; Yang, G.B.; Spassova, M.K.; Ouerfelli, O.; Ragupathi, G.; Damani, P.; Livingston, P.O.; Danishefsky, S.J. From synthesis to biologics: Preclinical data on a chemistry derived anticancer vaccine. J. Am. Chem. Soc. 2009, 131, 9298–9303. [Google Scholar] [CrossRef]
- Zhu, J.L.; Wan, Q.; Ragupathi, G.; George, C.M.; Livingston, P.O.; Danishefsky, S.J. Biologics through chemistry: Total synthesis of a proposed dual-acting vaccine targeting ovarian cancer orchestration of oligosaccharide and polypeptide domains. J. Am. Chem. Soc. 2009, 131, 4151–4158. [Google Scholar]
- Wang, J.; Li, H.; Zou, G.; Wang, L.-X. Novel template-assembled oligosaccharide clusters as epitope mimics for HIV-neutralizing antibody 2G12. Design, synthesis, and antibody binding study. Org. Biomol. Chem. 2007, 5, 1529–1540. [Google Scholar] [CrossRef]
- Ten Cate, M.G.J.; Severin, N.; Boerner, H.G. Self-assembling peptide-polymer conjugates comprising d-Alt-l)-cyclopeptides as aggregator domains. Macromolecules 2006, 39, 7831–7838. [Google Scholar] [CrossRef]
- Jeon, I.; Lee, D.; Krauss, I.J.; Danishefsky, S.J. A new model for the presentation of tumor-associated antigens and the quest for an anticancer vaccine: A solution to the synthesis challenge via ring-closing metathesis. J. Am. Chem. Soc. 2009, 131, 14337–14344. [Google Scholar] [CrossRef]
- Bettahi, I.; Dasgupta, G.; Renaudet, O.; Chentoufi, A.A.; Zhang, X.L.; Carpenter, D.; Yoon, S.; Dumy, P.; Benmohamed, L. Antitumor activity of a self-adjuvanting glyco-lipopeptide vaccine bearing B cell, CD4+ Nd CD8+ T Cell Epitopes. Cancer Immunol. Immunother. 2009, 58, 187–200. [Google Scholar] [CrossRef]
- Nardelli, B.; Lu, Y.A.; Shiu, D.R.; Delpierre-Defoort, C.; Profy, A.T.; Tam, J.P. A chemically defined synthetic vaccine model for HIV-1. J. Immunol. 1992, 148, 914–920. [Google Scholar]
- Cai, H.; Sun, Z.Y.; Chen, M.S.; Zhao, Y.F.; Kunz, H.; Li, Y.M. Synthetic multivalent glycopeptide-lipopeptide antitumor vaccines: Impact of the cluster effect on the killing of tumor cells. Angew. Chem. Int. Ed. 2014, 53, 1699–1703. [Google Scholar] [CrossRef]
- Cai, H.; Huang, Z.H.; Shi, L.; Zhao, Y.F.; Kunz, H.; Li, Y.M. Towards a fully synthetic Muc1-based anticancer vaccine: Efficient conjugation of glycopeptides with Mono-, Di-, and Tetravalent lipopeptides using click chemistry. Chem. Eur. J. 2011, 17, 6396–6406. [Google Scholar] [CrossRef]
- Geraci, C.; Consoli, G.M.; Granata, G.; Galante, E.; Palmigiano, A.; Pappalardo, M.; di Puma, S.D.; Spadaro, A. First self-adjuvant multicomponent potential vaccine candidates by tethering of four or eight MUC1 antigenic immunodominant PDTRP units on a calixarene platform: Synthesis and biological evaluation. Bioconjug. Chem. 2013, 24, 1710–1720. [Google Scholar] [CrossRef]
- Nuhn, L.; Hartmann, S.; Palitzsch, B.; Gerlitzki, B.; Schmitt, E.; Zentel, R.; Kunz, H. Water-soluble polymers coupled with glycopeptide antigens and T-cell epitopes as potential antitumor vaccines. Angew. Chem. Int. Ed. 2013, 52, 10652–10656. [Google Scholar] [CrossRef]
- Yang, F.; Zheng, X.J.; Huo, C.X.; Wang, Y.; Zhang, Y.; Ye, X.S. Enhancement of the immunogenicity of synthetic carbohydrate vaccines by chemical modifications of Stn antigen. ACS Chem. Biol. 2011, 6, 252–259. [Google Scholar] [CrossRef]
- Dubrovska, A.; Kim, C.; Elliott, J.; Shen, W.; Kuo, T.H.; Koo, D.I.; Li, C.; Tuntland, T.; Chang, J.; Groessl, T.; et al. A chemically induced vaccine strategy for prostate cancer. ACS Chem. Biol. 2011, 6, 1223–1231. [Google Scholar] [CrossRef]
- Qiu, L.; Gong, X.; Wang, Q.; Li, J.; Hu, H.; Wu, Q.; Zhang, J.; Guo, Z. Combining synthetic carbohydrate vaccines with cancer cell glycoengineering for effective cancer immunotherapy. Cancer Immunol. Immunother. 2012, 61, 2045–2054. [Google Scholar] [CrossRef]
- Ragupathi, G.; Liu, N.X.; Musselli, C.; Powell, S.; Lloyd, K.; Livingston, P.O. Antibodies against tumor cell glycolipids and proteins, but not mucins, mediate complement-dependent cytotoxicity. J. Immunol. 2005, 174, 5706–5712. [Google Scholar] [CrossRef]
- Pardoll, D.M. The blockade of immune checkpoints in cancer immunotherapy. Nat. Rev. Cancer 2012, 12, 252–264. [Google Scholar] [CrossRef]
- Li, X.; Fujio, M.; Imamura, M.; Wu, D.; Vasan, S.; Wong, C.-H.; Ho, D.D.; Tsuji, M. Design of a potent CD1d-binding NKT cell ligand as a vaccine adjuvant. Proc. Natl. Acad. Sci. USA 2010, 107, 13010–13015. [Google Scholar]
- Putta, M.R.; Zhu, F.G.; Wang, D.Q.; Bhagat, L.; Dai, M.R.; Kandimalla, E.R.; Agrawal, S. Peptide conjugation at the 5'-end of oligodeoxynucleotides abrogates toll-like receptor 9-mediated immune stimulatory activity. Bioconjug. Chem. 2010, 21, 39–45. [Google Scholar] [CrossRef]
- Zou, W. Immunosuppressive networks in the tumour environment and their therapeutic relevance. Nat. Rev. Cancer 2005, 5, 263–274. [Google Scholar] [CrossRef]
- Robert, C.; Thomas, L.; Bondarenko, I.; O’Day, S.; Weber, J.; Garbe, C.; Lebbe, C.; Baurain, J.F.; Testori, A.; Grob, J.J.; et al. Ipilimumab plus dacarbazine for previously untreated metastatic melanoma. N. Engl. J. Med. 2011, 364, 2517–2526. [Google Scholar] [CrossRef]
- Topalian, S.L.; Hodi, F.S.; Brahmer, J.R.; Gettinger, S.N.; Smith, D.C.; mcdermott, D.F.; Powderly, J.D.; Carvajal, R.D.; Sosman, J.A.; Atkins, M.B.; et al. Safety, activity, and immune correlates of anti-PD-1 antibody in cancer. N. Engl. J. Med. 2012, 366, 2443–2454. [Google Scholar] [CrossRef]
- Ni, J.; Powell, R.; Baskakov, I.V.; Devico, A.; Lewis, G.K.; Wang, L.X. Synthesis, conformation, and immunogenicity of monosaccharide-centered multivalent HIV-1 gp41 peptides containing the sequence of DP178. Bioorg. Med. Chem. 2004, 12, 3141–3148. [Google Scholar] [CrossRef]
- Zhong, W.; Skwarczynski, M.; Fujita, Y.; Simerska, P.; Good, M.F.; Toth, I. Design and synthesis of lipopeptide-carbohydrate assembled multivalent vaccine candidates using native chemical ligation. Aust. J. Chem. 2009, 62, 993–999. [Google Scholar] [CrossRef]
- Oyewumi, M.O.; Kumar, A.; Cui, Z. Nano-microparticles as immune adjuvants: Correlating particle sizes and the resultant immune responses. Expert Rev. Vaccines 2010, 9, 1095–1107. [Google Scholar] [CrossRef]
- Bachmann, M.F.; Jennings, G.T. Vaccine delivery: A matter of size, geometry, kinetics and molecular patterns. Nat. Rev. Immunol. 2010, 10, 787–796. [Google Scholar] [CrossRef]
- Ghasparian, A.; Riedel, T.; Koomullil, J.; Moehle, K.; Gorba, C.; Svergun, D.I.; Perriman, A.W.; Mann, S.; Tamborrini, M.; Pluschke, G.; et al. Engineered synthetic virus-like particles and their use in vaccine delivery. Chembiochem 2011, 12, 100–109. [Google Scholar] [CrossRef]
- Boato, F.; Thomas, R.M.; Ghasparian, A.; Freund-Renard, A.; Moehle, K.; Robinson, J.A. Synthetic virus-like particles from self-assembling coiled-coil lipopeptides and their use in antigen display to the immune system. Angew. Chem. Int. Ed. 2007, 46, 9015–9018. [Google Scholar] [CrossRef]
- Skwarczynski, M.; Zaman, M.; Urbani, C.N.; Lin, I.C.; Jia, Z.F.; Batzloff, M.R.; Good, M.F.; Monteiro, M.F.; Toth, I. Polyacrylate dendrimer nanoparticles: A self-adjuvanting vaccine delivery system. Angew. Chem. Int. Ed. 2010, 49, 5742–5745. [Google Scholar] [CrossRef]
- Kakwere, H.; Chun, C.K.Y.; Jolliffe, K.A.; Payne, R.J.; Perrier, S. Polymer-peptide chimeras for the multivalent display of immunogenic peptides. Chem. Commun. 2010, 46, 2188–2190. [Google Scholar] [CrossRef]
- Huang, Z.H.; Shi, L.; Ma, J.W.; Sun, Z.Y.; Cai, H.; Chen, Y.X.; Zhao, Y.F.; Li, Y.M. A totally synthetic, self-assembling, adjuvant-free muc1 glycopeptide vaccine for cancer therapy. J. Am. Chem. Soc. 2012, 134, 8730–8733. [Google Scholar]
- Skwarczynski, M.; Toth, I. Peptide-based subunit nanovaccines. Curr. Drug Deliv. 2011, 8, 282–289. [Google Scholar] [CrossRef]
- Parry, A.L.; Clemson, N.A.; Ellis, J.; Bernhard, S.S.; Davis, B.G.; Cameron, N.R. “Multicopy Multivalent” glycopolymer-stabilized gold nanoparticles as potential synthetic cancer vaccines. J. Am. Chem. Soc. 2013, 135, 9362–9365. [Google Scholar] [CrossRef]
© 2014 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Huang, Z.-H.; Sun, Z.-Y.; Gao, Y.; Chen, P.-G.; Liu, Y.-F.; Chen, Y.-X.; Li, Y.-M. Strategy for Designing a Synthetic Tumor Vaccine: Multi-Component, Multivalency and Antigen Modification. Vaccines 2014, 2, 549-562. https://doi.org/10.3390/vaccines2030549
Huang Z-H, Sun Z-Y, Gao Y, Chen P-G, Liu Y-F, Chen Y-X, Li Y-M. Strategy for Designing a Synthetic Tumor Vaccine: Multi-Component, Multivalency and Antigen Modification. Vaccines. 2014; 2(3):549-562. https://doi.org/10.3390/vaccines2030549
Chicago/Turabian StyleHuang, Zhi-Hua, Zhan-Yi Sun, Yue Gao, Pu-Guang Chen, Yan-Fang Liu, Yong-Xiang Chen, and Yan-Mei Li. 2014. "Strategy for Designing a Synthetic Tumor Vaccine: Multi-Component, Multivalency and Antigen Modification" Vaccines 2, no. 3: 549-562. https://doi.org/10.3390/vaccines2030549
APA StyleHuang, Z.-H., Sun, Z.-Y., Gao, Y., Chen, P.-G., Liu, Y.-F., Chen, Y.-X., & Li, Y.-M. (2014). Strategy for Designing a Synthetic Tumor Vaccine: Multi-Component, Multivalency and Antigen Modification. Vaccines, 2(3), 549-562. https://doi.org/10.3390/vaccines2030549



