Retrospective Analysis of the Impact of Vaccination with an Inactivated Vaccine on Toxoplasmosis-Associated Mortality in Captive Wildlife
Abstract
1. Introduction
2. Materials and Methods
2.1. The Ethics Statement
2.2. VXN-Toxo Vaccine Preparation
2.3. Vaccinations
2.4. Safety of VXN-Toxo in Captive Wildlife
2.5. Immunization
2.6. Toxoplasma-Related Mortality in Zoos
- Did you record any toxoplasmosis-related deaths before the vaccination was introduced? (Yes/No)
- Is a specific diagnosis systematically made when a deceased animal is suspected of having toxoplasmosis? (Yes/No/Not applicable)
- If yes, at what moment was the diagnosis carried out? (When the symptoms were detected/Post-mortem/Not applicable)
- What was the diagnostic method used? (Open question)
- If no, is it possible that any of the undiagnosed animals died from toxoplasmosis, given the clinical signs? (Yes/No/Not applicable)
- Have you implemented any measures other than vaccination to prevent toxoplasmosis? (Yes/No/Not applicable)
- If yes, what measures? (Open question)
- What prompted you to set up a vaccination campaign with VXN-Toxo? (Open question)
- Would you recommend the vaccination to other zoos? (Yes/No/Not applicable)
2.7. Toxoplasmosis Diagnosis
3. Results
3.1. Vaccinated Animals
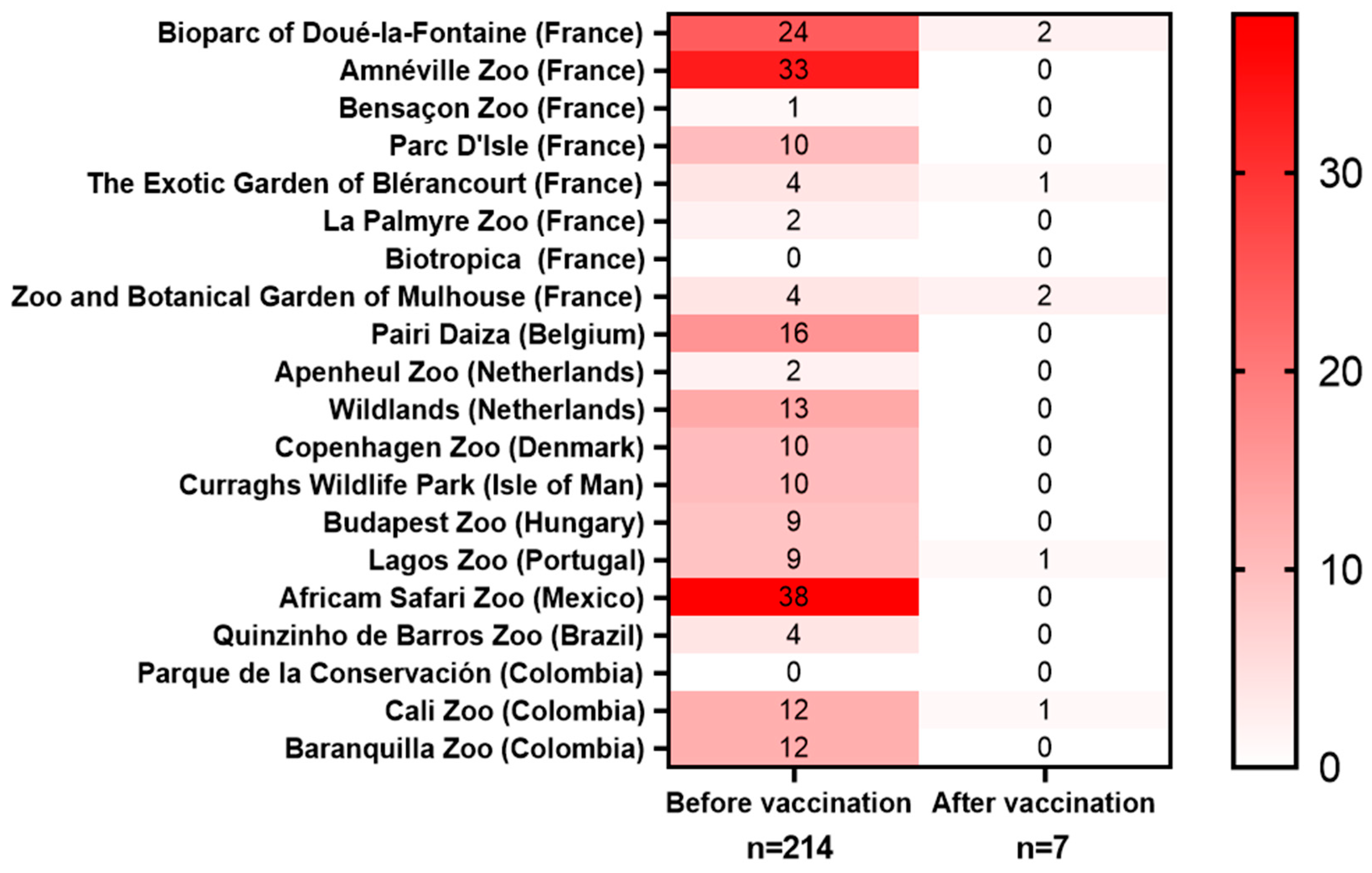
3.2. Toxoplasmosis-Related Mortality in Captive Animals
3.2.1. Bioparc of Doué-la-Fontaine (France, Doué-la-Fontaine)
3.2.2. Amnéville Zoo (France, Amnéville)
3.2.3. Besançon Zoo (France, Besançon)
3.2.4. Parc d’Isle (France, Saint-Quentin)
3.2.5. The Exotic Garden, Foster’s Center for Small Primates (France, Blérancourt)
3.2.6. La Palmyre Zoo (France, Les Mathes)
3.2.7. Biotropica Zoo (France, Val-de-Reuil)
3.2.8. Zoo and Botanical Garden of Mulhouse (France, Mulhouse)
3.2.9. Pairi Daiza Zoo (Belgium, Brugelette)
3.2.10. Apenheul Zoo (The Netherlands, Apeldoorn)
3.2.11. Wildlands (The Netherlands, Emmen)
3.2.12. Copenhagen Zoo (Denmark, Copenhagen)
3.2.13. Curraghs Wildlife Park (Isle of Man)
3.2.14. Budapest Zoo and Botanical Garden (Hungary, Budapest)
3.2.15. Lagos Zoo (Portugal, Barão de São João)
3.2.16. Africam Safari Zoo (Mexico, Puebla)
3.2.17. Quinzinho de Barros Zoo (Brazil, Sorocaba)
3.2.18. Parque de la Conservación (Medellín—Colombia)
3.2.19. Cali Zoo (Colombia, Santiago de Cali)
3.2.20. Baranquilla Zoo (Colombia, Baranquilla)
3.3. Summary of Toxoplasmosis-Associated Mortality and Vaccinations
3.4. Toxoplasma Prevention Survey
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Correction Statement
Abbreviations
| EU | European Union |
| FVE | Federation of Veterinarians of Europe |
| NPLs | Lipid core maltodextrin nanoparticles |
| GMP | Good Manufacturing Processes |
| IHC | Immunohistochemistry |
| IUCN | International Union for Conservation of Nature |
| EAZA | European Association of Zoos and Aquaria |
| PCR | Polymerase Chain Reaction |
References
- Dubey, J.P. Toxoplasmosis of Animals and Humans, 3rd ed.; CRC Press: Boca Raton, FL, USA, 2021; ISBN 978-1-00-319937-3. [Google Scholar]
- Anderson, K.; Dennis, P.M. Mortality Trends for Five Species of Macropods from a Single Institution from 1995 to 2016. Zoo Biol. 2022, 41, 44–49. [Google Scholar] [CrossRef]
- Rocchigiani, G.; Fonti, N.; Nardoni, S.; Cavicchio, P.; Mancianti, F.; Poli, A. Toxoplasmosis in Captive Ring-Tailed Lemurs (Lemur catta). Pathogens 2022, 11, 1142. [Google Scholar] [CrossRef]
- Salant, H.; Weingram, T.; Spira, D.T.; Eizenberg, T. An Outbreak of Toxoplasmosis amongst Squirrel Monkeys in an Israeli Monkey Colony. Vet. Parasitol. 2009, 159, 24–29. [Google Scholar] [CrossRef] [PubMed]
- Nishimura, M.; Goyama, T.; Tomikawa, S.; Fereig, R.M.; El-Alfy, E.-S.N.; Nagamune, K.; Kobayashi, Y.; Nishikawa, Y. Outbreak of Toxoplasmosis in Four Squirrel Monkeys (Saimiri sciureus) in Japan. Parasitol. Int. 2019, 68, 79–86. [Google Scholar] [CrossRef] [PubMed]
- Burger, M.; Du Plessis, E.C.; Suleman, E.; Gardner, B.R. Fatal Disseminated Toxoplasmosis in a Zoological Collection of Meerkats (Suricata suricatta). J. S. Afr. Vet. Assoc. 2017, 88, 1–5. [Google Scholar] [CrossRef]
- Girling, S.J.; Pizzi, R.; Naylor, A.D.; Richardson, D.; Richardson, U.; Harley, J.; Cole, G.; Brown, D.; Fraser, M.; Tillman, E.; et al. Use of clindamycin in pallas’ cats [Otocolobus (felis) manul] to reduce juvenile toxoplasmosis-associated mortality rates. J. Zoo Wildl. Med. 2020, 51, 39. [Google Scholar] [CrossRef] [PubMed]
- Basso, W.; Edelhofer, R.; Zenker, W.; Möstl, K.; Kübber-Heiss, A.; Prosl, H. Toxoplasmosis in Pallas’ Cats (Otocolobus manul) Raised in Captivity. Parasitology 2005, 130, 293–299. [Google Scholar] [CrossRef]
- Denk, D.; De Neck, S.; Khaliq, S.; Stidworthy, M.F. Toxoplasmosis in Zoo Animals: A Retrospective Pathology Review of 126 Cases. Animals 2022, 12, 619. [Google Scholar] [CrossRef]
- Stock, M.L.; Elazab, S.T.; Hsu, W.H. Review of Triazine Antiprotozoal Drugs Used in Veterinary Medicine. Vet. Pharm. Ther. 2018, 41, 184–194. [Google Scholar] [CrossRef]
- De Camps, S.; Dubey, J.P.; Saville, W.J.A. Seroepidemiology of Toxoplasma gondii in Zoo Animals in Selected Zoos in the Midwestern United States. J. Parasitol. 2008, 94, 648–653. [Google Scholar] [CrossRef]
- Dubey, J.P. Clinical Toxoplasmosis in Zoo Animals and Its Management. Emerg. Anim. Species 2022, 2, 100002. [Google Scholar] [CrossRef]
- Angarita-Corzo, K.; Henao-Montoya, L.M.; Franco-G, M.; Riva, H.G.; Correa-Valencia, N.M. Occurrence of Toxoplasma gondii in Three Tamarin Species (Saguinus geoffroyi, Saguinus oedipus, and Leontocebus fuscicollis) Ex Situ in Colombia. J. Med. Primatol. 2025, 54, e70005. [Google Scholar] [CrossRef] [PubMed]
- Anderson, D.C.; McClure, H.Μ. Acute Disseminated Fatal Toxoplasmosis in a Squirrel Monkey. J. Am. Vet. Med. Assoc. 1982, 181, 1363–1366. [Google Scholar] [CrossRef] [PubMed]
- Hensel, M.E.; Landsgaard, K.; Wang, W.; Neupane, S.; Su, C.; Blue-McClendon, A.; Porter, B.F.; Uzal, F.A.; Rech, R. Toxoplasmosis in Ring-Tailed Lemurs (Lemur catta) and a Peahen (Pavo cristatus) in a Zoological Collection Caused by the Common Toxoplasma Genotype in Wild Animals in the US. Vet. Parasitol. Reg. Stud. Rep. 2023, 41, 100876. [Google Scholar] [CrossRef]
- Dubey, J.P.; Murata, F.H.A.; Cerqueira-Cézar, C.K.; Kwok, O.C.H.; Su, C.; Grigg, M.E. Recent Aspects on Epidemiology, Clinical Disease, and Genetic Diversity of Toxoplasma Gondii Infections in Australasian Marsupials. Parasites Vectors 2021, 14, 301. [Google Scholar] [CrossRef] [PubMed]
- Sós, E.; Szigeti, A.; Fok, É.; Molnár, V.; Erdélyi, K.; Perge, E.; Biksi, I.; Gál, J. Toxoplasmosis in Tammar Wallabies (Macropus eugenii) in the Budapest Zoo and Botanical Garden (2006–2010). Acta Vet. Hung. 2012, 60, 361–370. [Google Scholar] [CrossRef]
- Gong, H.; Wang, Q.; Jin, Y.; Qiu, S.; Chen, Z.; Han, X.; Chen, Z.; Jiang, W. Fatal Toxoplasmosis in Red Kangaroos (Macropus rufus) in East China. Pathogens 2025, 14, 202. [Google Scholar] [CrossRef]
- Yang, L.; Xin, S.; Zhu, N.; Li, J.; Su, C.; Yang, Y. Two Viable Toxoplasma gondii Isolates from Red-Necked Wallaby (Macropus rufogriseus) and Red Kangaroo (M. rufus). Parasitol. Int. 2023, 92, 102687. [Google Scholar] [CrossRef]
- Basso, W.; Moré, G.; Quiroga, M.A.; Pardini, L.; Bacigalupe, D.; Venturini, L.; Valenzuela, M.C.; Balducchi, D.; Maksimov, P.; Schares, G.; et al. Isolation and Molecular Characterization of Toxoplasma gondii from Captive Slender-Tailed Meerkats (Suricata suricatta) with Fatal Toxoplasmosis in Argentina. Vet. Parasitol. 2009, 161, 201–206. [Google Scholar] [CrossRef]
- Paula, N.F.D.; Dutra, K.S.; Oliveira, A.R.D.; Santos, D.O.D.; Rocha, C.E.V.; Vitor, R.W.D.A.; Tinoco, H.P.; Costa, M.E.L.T.D.; Paixão, T.A.D.; Santos, R.L. Host Range and Susceptibility to Toxoplasma gondii Infection in Captive Neotropical and Old-world Primates. J. Med. Primatol. 2020, 49, 202–210. [Google Scholar] [CrossRef]
- Henrik Dietz, H.; Henriksen, P.; Bille-Hansen, V.; Aage Henriksen, S. Toxoplasmosis in a Colony of New World Monkeys. Vet. Parasitol. 1997, 68, 299–304. [Google Scholar] [CrossRef]
- Catão-Dias, J.L.; Epiphanio, S.; Kierulff, M.C.M. Neotropical Primates and Their Susceptibility to Toxoplasma gondii: New Insights for an Old Problem. In Primates, Pathogens, and Evolution; Brinkworth, J.F., Pechenkina, K., Eds.; Springer: New York, NY, USA, 2013; pp. 253–289. ISBN 978-1-4614-7180-6. [Google Scholar]
- Dubey, J.P. Toxoplasmosis in Sheep—The Last 20 Years. Vet. Parasitol. 2009, 163, 1–14. [Google Scholar] [CrossRef] [PubMed]
- Fasquelle, F.; Scuotto, A.; Howsam, M.; Betbeder, D. Maltodextrin-Nanoparticles as a Delivery System for Nasal Vaccines: A Review Article. Pharmaceutics 2024, 16, 247. [Google Scholar] [CrossRef]
- Dimier-Poisson, I.; Carpentier, R.; N’Guyen, T.T.L.; Dahmani, F.; Ducournau, C.; Betbeder, D. Porous Nanoparticles as Delivery System of Complex Antigens for an Effective Vaccine against Acute and Chronic Toxoplasma gondii Infection. Biomaterials 2015, 50, 164–175. [Google Scholar] [CrossRef]
- Ducournau, C.; Moiré, N.; Carpentier, R.; Cantin, P.; Herkt, C.; Lantier, I.; Betbeder, D.; Dimier-Poisson, I. Effective Nanoparticle-Based Nasal Vaccine Against Latent and Congenital Toxoplasmosis in Sheep. Front. Immunol. 2020, 11, 2183. [Google Scholar] [CrossRef]
- Fasquelle, F.; Scuotto, A.; Vreulx, A.-C.; Petit, T.; Charpentier, T.; Betbeder, D. Nasal Vaccination of Six Squirrel Monkeys (Saimiri sciureus): Improved Immunization Protocol Against Toxoplasma gondii with a Nanoparticle-Born Vaccine. Int. J. Parasitol. Parasites Wildl. 2023, 22, 69–74. [Google Scholar] [CrossRef]
- European Union. Regulation (EU) 2019/6 of the European Parliament and of the Council of 11 December 2018 on veterinary medicinal products and repealing Directive 2001/82/EC. Off. J. Eur. Union 2019, L4, 43–167. Available online: https://eur-lex.europa.eu/legal-content/EN/TXT/?uri=CELEX:32019R0006 (accessed on 24 August 2025).
- European Union. Directive 2010/63/EU of the European Parliament and of the Council of 22 September 2010 on the protection of animals used for scientific purposes. Off. J. Eur. Union 2010, L276, 33–79. Available online: https://eur-lex.europa.eu/legal-content/EN/TXT/?uri=CELEX:32010L0063 (accessed on 24 August 2025).
- Ogonczyk-Makowska, D.; Fasquelle, F.; Vreulx, A.-C.; Scuotto, A.; Degraeve, A.; Thiroux, B.; Martin, L.F.; Tahas, S.A.; Jakobsen, J.T.; Jungersen, G.; et al. Inactivated Toxoplasma gondii Nanovaccine Boosts T-Cell Memory Response in a Seropositive Yellow-Footed Rock Wallaby (Petrogale xanthopus)—A Case Report from Copenhagen Zoo. Int. J. Parasitol. Parasites Wildl. 2024, 25, 101017. [Google Scholar] [CrossRef]
- Fernández-Escobar, M.; Schares, G.; Maksimov, P.; Joeres, M.; Ortega-Mora, L.M.; Calero-Bernal, R. Toxoplasma gondii Genotyping: A Closer Look into Europe. Front. Cell Infect. Microbiol. 2022, 12, 842595. [Google Scholar] [CrossRef] [PubMed]
- Galal, L.; Hamidović, A.; Dardé, M.L.; Mercier, M. Diversity of Toxoplasma gondii Strains at the Global Level and Its Determinants. Food Waterborne Parasitol. 2019, 15, e00052. [Google Scholar] [CrossRef]
- Hosseini, S.A.; Amouei, A.; Sharif, M.; Sarvi, S.; Galal, L.; Javidnia, J.; Pagheh, A.S.; Gholami, S.; Mizani, A.; Daryani, A. Human Toxoplasmosis: A Systematic Review for Genetic Diversity of Toxoplasma gondii in Clinical Samples. Epidemiol. Infect. 2019, 147, e36. [Google Scholar] [CrossRef] [PubMed]

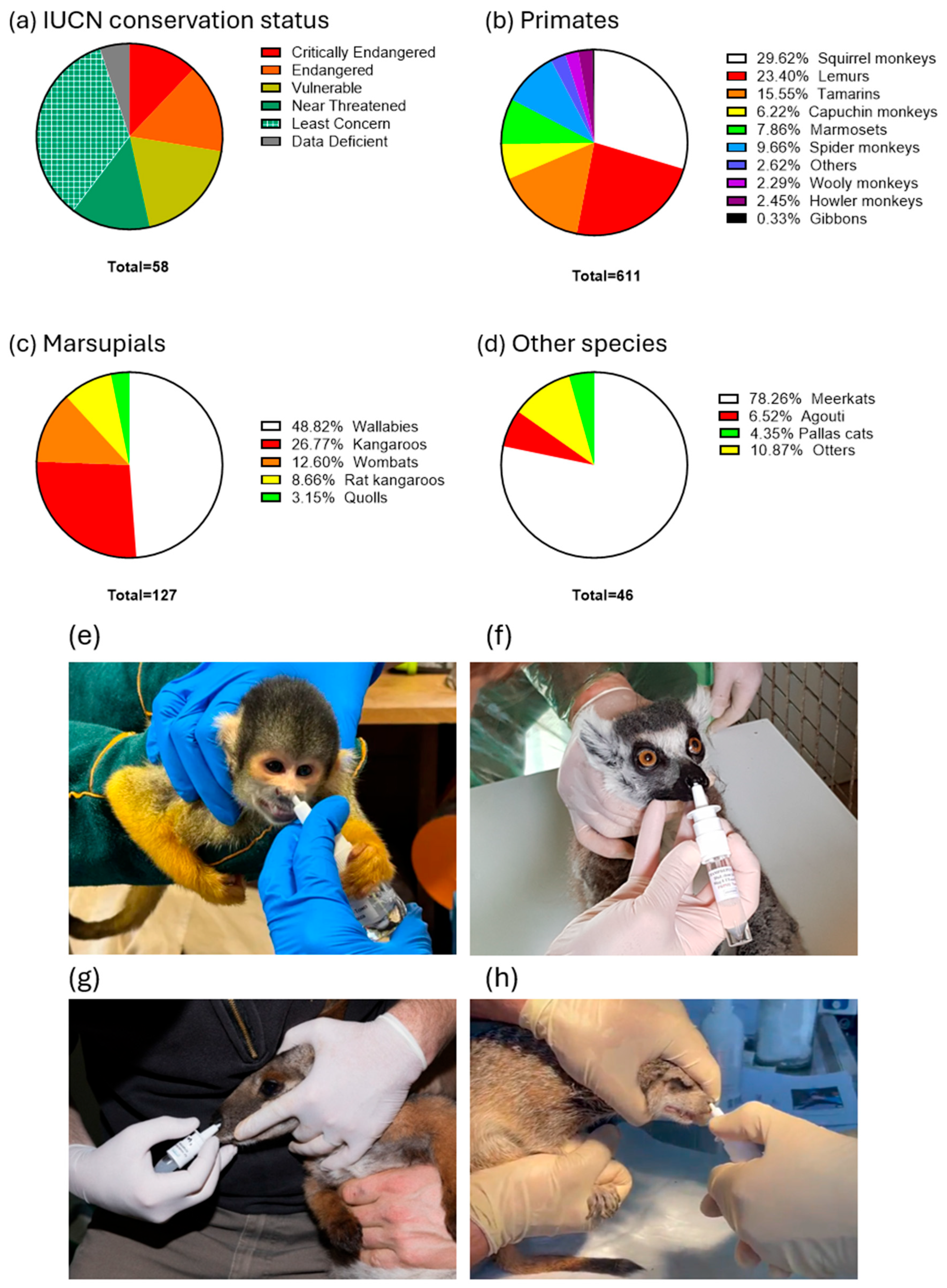
| Family | Common Name | Scientific Name | IUCN | Number | Adverse Effects Observed |
|---|---|---|---|---|---|
| Primates | Common squirrel monkey | Saimiri sciureus | LC | 24 | No |
| Black-capped squirrel monkey | Saimiri boliviensis | LC | 149 | No | |
| Squirrel monkey | Saimiri spp. | LC | 8 | No | |
| Ring-tailed lemur | Lemur catta | EN | 104 | No | |
| Crowned lemur | Eulemur coronatus | EN | 2 | No | |
| Brown lemur | Eulemur fulvus | NT | 12 | No | |
| Black-and-white ruffed lemur | Varecia variegata variegata | CR | 16 | No | |
| Black lemur | Eulemur macaco | VU | 2 | No | |
| Red ruffed lemur | Varecia variegata rubra | CR | 5 | No | |
| Lac Alaotra Gentle Lemur | Hapalemur alaotrensis | CR | 2 | No | |
| White-faced saki | Pithecia pithecia | LC | 8 | No | |
| Cotton-top tamarin | Saguinus oedipus | CR | 20 | No | |
| Emperor tamarin | Saguinus imperator | LC | 6 | No | |
| Golden-handed tamarin | Saguinus midas | LC | 8 | No | |
| Geoffroy’s tamarin | Saguinus geoffroyi | NT | 11 | No | |
| White-footed tamarin | Saguinus leucopus | VU | 10 | No | |
| Golden-headed lion tamarin | Leontopithecus chrysomelas | EN | 6 | No | |
| Pied tamarin | Saguinus bicolor | CR | 21 | No | |
| Black tamarin | Saguinus niger | DD | 1 | No | |
| Brown-mantled tamarin | Leontocebus fuscicollis | LC | 10 | No | |
| Golden lion tamarin | Leontopithecus rosalia | EN | 2 | No | |
| Pygmy marmoset | Cebuella pygmaea | LC | 31 | No | |
| Geoffroy’s marmoset | Callithrix geoffroyi | LC | 8 | No | |
| Common marmoset | Callithrix jacchus | LC | 6 | No | |
| Goeldi’s marmoset | Callimico goeldii | VU | 3 | No | |
| White-cheeked spider monkey | Ateles marginatus | EN | 4 | No | |
| White-bellied spider monkey | Ateles belzebuth | EN | 1 | No | |
| Brown spider monkey | Ateles hybridus | CR | 29 | No | |
| Black-headed spider monkey | Ateles fusciceps | CR | 23 | No | |
| Black-faced black spider monkey | Ateles chamek | VU | 2 | No | |
| Southern muriqui/wooly spider monkey | Brachyteles arachnoides | EN | 3 | No | |
| Gray woolly monkey | Lagothrix cana | VU | 3 | No | |
| Colombian woolly monkey | Lagothrix lagothricha | VU | 8 | No | |
| White-handed gibbon | Hylobates lar | EN | 2 | No | |
| Howler monkey | Alouatta caraya | LC | 5 | No | |
| Venezuelan red howler monkey | Alouatta seniculus | LC | 10 | No | |
| White-fronted capuchin | Cebus albifrons | VU | 11 | No | |
| White-faced capuchin | Cebus capucinus | LC | 8 | No | |
| Tufted capuchin | Sapajus apella | LC | 19 | No | |
| Vieira’s titi monkey | Plecturocebus vieirai | VU | 2 | No | |
| Grey-necked night monkey | Aotus lemurinus | VU | 3 | No | |
| Northern night monkey | Aotus trivirgatus | DD | 3 | No | |
| Marsupials | Red-necked/Bennett’s wallaby | Notamacropus rufogriseus | LC | 33 | No |
| Swamp wallaby | Wallabia bicolor | LC | 9 | No | |
| Yellow-footed rock wallaby | Petrogale xanthopus | NT | 18 | No | |
| Parma wallaby | Notamacropus parma | VU | 2 | No | |
| Red kangaroo | Osphranter rufus | LC | 14 | No | |
| Western Grey kangaroo | Macropus fuliginosus | DD | 4 | No | |
| Bennett’s tree-kangaroo | Dendrolagus bennettianus | NT | 16 | No | |
| Eastern Bettong | Bettongia gaimardi | NT | 6 | No | |
| Brush-tailed rat-kangaroo | Bettongia penicillata | NT | 3 | No | |
| Long-nosed potoroo | Potorous tridactylus | NT | 2 | No | |
| Tasmanian wombat | Vombatus ursinus | LC | 16 | No | |
| Eastern quoll | Dasyurus viverrinus | EN | 4 | No | |
| Felids | Pallas’ cat | Otocolobus manul | NT | 2 | No |
| Rodents | Azara’s agouti | Dasyprocta azarae | DD | 3 | No |
| Mustelidae | Small-clawed Asian otter | Aonyx cinereus | VU | 5 | No |
| Herpestidae | Meerkat | Suricata suricatta | LC | 36 | No |
| Total number | 784 |
| Institution/ Zoo (Country) | Toxoplasmosis-Related Mortality Before the Vaccination | Vaccinations | Toxoplasmosis-Related Mortality After the Vaccination | ||
|---|---|---|---|---|---|
| Period | Dead Animals | Vaccinated Animals | Period | Details | |
| Bioparc of Doué-la- Fontaine Zoo (France, Doué-la- Fontaine) | 2005–2017 | S. boliviensis n = 23 L. catta n = 1 (total n = 24) | S. boliviensis n = 17 | 2017–2025 | S. boliviensis n = 4 (2 vaccinated twice, and 2 others were vaccinated while being seropositive; vaccine administration before the optimization of nasal spray delivery) Procavia capensis n = 1 (unvaccinated) |
| Amnéville Zoo (France, Amnéville) | 2010–2017 | S. boliviensis n = 33 | S. boliviensis n = 9, N. rufogriseus n = 18 (total n = 27) | 2017–2025 | N. rufogriseus n = 1 (vaccinated while being seropositive, 3 doses) |
| Besançon Zoo (France, Besançon) | 2010 | Petrogale spp. n = 1 | S. boliviensis n = 11 | 2018–2025 | 0 |
| Parc d’Isle (France, Saint-Quentin) | 2023 | N. rufogriseus n = 10 | Saimiri spp. n = 6, L. catta n = 13, N. rufogriseus n = 9, S. suricatta n = 4, (total n = 32) | 2023–2025 | L. catta n = 2, (vaccinated while showing signs of clinical toxoplasmosis) |
| The Exotic Garden, Foster’s Center For Small Primates (France, Blérancourt) | 2023–2024 | L. catta n = 4 | Saimiri spp. n = 2 L. catta n = 5, E. fulvus n = 12, C. jacchus n = 6, S. imperator n = 4, C. pygmaea n = 4, C. goeldii n = 3, S. midas n = 3, E. macaco n = 2, S. Oedipus n = 2, V. variegata variegata n = 1 (total n = 44) | 2024–2025 | L. catta, n = 1 (2 doses) |
| La Palmyre Zoo (France, Les Mathes) | 2013–2016 | S. suricata n = 2 | S. sciureus n = 6 | 2022–2025 | 0 |
| Biotropica Zoo (France, La Coudrette) | unavailable | S. boliviensis n = 21 | 2023–2025 | N. parma n = 2 (unvaccinated), and n = 1 suspected (unvaccinated) M. fuliginosus n = 1 (unvaccinated) | |
| Zoo and Botanical Garden of Mulhouse (France, Mulhouse) | 2017 | S. boliviensis n = 4 | S. boliviensis n = 5, P. xanthopus n = 14, O. manul n = 2 (total n = 21) | 2017–2025 | S. boliviensis n = 2, (5 doses of the vaccine and died 3–4 years after the last booster) |
| Pairi Daiza (Belgium, Brugelette) | 2016 | S. boliviensis peruviensis n = 16 | S. boliviensis peruviensis n = 32, L. catta n = 25, S. oedipus n = 7, P. pithecia n = 6, V. variegata variegata n = 5, E. coronatus n = 2 (total n = 77) | 2021–2025 | L. catta, n = 2, unvaccinated |
| Apenheul Zoo (The Netherlands, Apeldoorn) | 2021–2022 | A. seniculus n = 2 | A. seniculus n = 4, P. pithecia n = 2, L. chrysomelas n = 2, S. bicolor n = 1 (total n = 9) | 2023–2025 | 0 |
| Wildlands (The Netherlands, Emmen) | 2012–2019 | S. boliviensis n = 1, L. catta n = 12 (total n = 13) | S. boliviensis n = 21 L. catta n = 15, W. bicolor n = 5 C. pygmaea n = 4 V. variegata n = 2 (total n = 47) | 2022–2025 | 0 |
| Copenhagen Zoo (Denmark, Frederiksberg) | 2014–2020 | L. catta n = 4, S. suricatta n = 1, M. giganteus n = 2, V. ursinus n = 1 Vulpes lagopus n = 1 Ovibos moschatus n = 1 (total n = 10) | L. catta n = 11, V. ursinus n = 13, P. xanthopus n = 4, D. viverrinus n = 4, B. penicillata n = 3, P. tridactylus n = 2 (total n = 37) | 2021–2025 | S. suricatta n = 1, unvaccinated |
| Curraghs Wildlife Park (Isle de Man, Ballaugh) | 2017–2020 | S. boliviensis peruviensis n = 1 S. suricatta n = 1 A. cinereus n = 8 (total n = 10) | S. boliviensis boliviensis n = 7, L. catta n = 9, S. suricatta n = 11, A. cinereus n = 5 D. azarae n = 3, A. fusciceps robustus n = 2, H. alaotrensis n = 2, V. variegata rubra n = 2, V. variegata variegata n = 2 (total n = 43) | 2023–2025 | 0 |
| Budapest Zoo and Botanical Garden (Hungary, Budapest) | 2013–2018 | Notamacropus eugenii n = 5 D. bennettianus n = 3 Capra aegagrus hircus n = 1 (total n = 9) | D. bennettianus n = 16, B. gaimardi n = 6, M. fuliginosus n = 4 V. ursinus n = 3 N. parma n = 2 (total n = 31) | 2023–2025 | 0 |
| Lagos Zoo (Portugal, Lagos) | 2018–2020 | S. boliviensis n = 6 L. catta n = 3 (total n = 9) | S. boliviensis n = 26, L. catta n = 16, S. suricatta n = 5, C. geoffroyi n = 8, V. variegata rubra n = 3, C. pygmaea n = 6, S. imperator n = 2, S. midas n = 2, S. oedipus n = 2 V. variegata variegata n = 2, W. bicolor n = 4 (total n = 76) | 2021–2025 | S. boliviensis n = 2 (1 vaccinated, 1 of unknown status) |
| Africam Safari (Mexico, Puebla) | 2012–2024 | S. sciureus n = 4 L. catta n = 5 N. rufogriseus n = 20 S. suricatta n = 2 O. rufus n = 7 (total n = 38) | O. rufus n = 14 S. suricatta n = 5 (total n = 19) | 2024–2025 | 0 |
| Quinzinho de Barros Zoo (Brazil, Sorocaba) | 2021–2024 | L. catta n = 1 B. arachnoides n = 3 (total n = 4) | L. catta n = 3, S. bicolor n = 20, A. caraya n = 5, L. chrysomelas n = 4, A. marginatus n = 4, S. midas n = 3, L. fuscicollis n = 3, L. cana n = 3, B. arachnoides n = 3, P. vieirai n = 2, A. chamek n = 2, A. trivirgatus n = 2, L. rosalia n = 2, A. trivirgatus n = 1, S. niger n = 1 (total n = 58) | 2024–2025 | 0 |
| Conservacion Park of Medellín (Colombia, Medellín) | 2015–2022 | - | S. sciureus n = 9, A. hybridus n = 21, C. albifrons n = 11, A. fusciceps n = 8, A. seniculus n = 6, S. geoffroyi, n = 4 C. capucinus n = 4, S. apella n = 3, S. oedipus n = 5, C. pygmaea n = 1, L. lagothricha n = 1 (total n = 73) | 2022–2025 | 0 |
| Cali Zoo (Colombia, Cali) | 2020–2023 | S. sciureus n = 1 L. catta n = 1 N. rufogriseus n = 2 C. pygmaea n = 1 S. oedipus n = 1 A. lemurinus n = 2 O. leucopus n = 2 L. lagothricha n = 1 V. variegata variegata n = 1 (total n = 12) | S. sciureus n = 9, L. catta n = 7, N. rufogriseus n = 6, S. suricatta n = 11, C. pygmaea n = 7, S. leucopus n = 8, L. lagothricha n = 7, S. apella n = 7, L. fuscicollis n = 5, C. capucinus n = 4, V. variegata variegata n = 4, A. fusciceps n = 2, H. lar n = 2 (total n = 79) | 2023–2025 | N. rufogriseus n = 3. (2 unvaccinated and 1 vaccinated animal in which vaccine administration failure was suspected) |
| Baranquilla Zoo (Colombia, Barranquilla) | 2007–2022 | N. rufogriseus n = 2, L. catta n = 2, V. variegata variegata n = 1, S. geoffroyi n = 3, S. oedipus n = 1, A. lemurinus n = 1, S. suricatta n = 2 (total n = 12) [13] | A. fusciceps n = 11, S. oedipus n = 4, L. fuscicollis n = 2, S. geoffroyi n = 7, S. apella n = 9, C. pygmaea n = 9, A. hybridus n = 8, A. lemurinus n = 3, A. belzebuth n = 1, S. leucopus n = 2 (total n = 56) | 2022–2025 | |
| Total | 214 | 784 | 23 A, 7 B | ||
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Scuotto, A.; Ogonczyk-Makowska, D.; Quiévy, A.; Berthet, M.; Schlax, K.; Boussarie, D.; Maillot, A.; Popelin-Wedlarski, F.; Charpentier, T.; Perot, M.; et al. Retrospective Analysis of the Impact of Vaccination with an Inactivated Vaccine on Toxoplasmosis-Associated Mortality in Captive Wildlife. Vaccines 2025, 13, 910. https://doi.org/10.3390/vaccines13090910
Scuotto A, Ogonczyk-Makowska D, Quiévy A, Berthet M, Schlax K, Boussarie D, Maillot A, Popelin-Wedlarski F, Charpentier T, Perot M, et al. Retrospective Analysis of the Impact of Vaccination with an Inactivated Vaccine on Toxoplasmosis-Associated Mortality in Captive Wildlife. Vaccines. 2025; 13(9):910. https://doi.org/10.3390/vaccines13090910
Chicago/Turabian StyleScuotto, Angelo, Daniela Ogonczyk-Makowska, Alicia Quiévy, Mélanie Berthet, Kévin Schlax, Didier Boussarie, Alexis Maillot, Florine Popelin-Wedlarski, Thomas Charpentier, Maïalen Perot, and et al. 2025. "Retrospective Analysis of the Impact of Vaccination with an Inactivated Vaccine on Toxoplasmosis-Associated Mortality in Captive Wildlife" Vaccines 13, no. 9: 910. https://doi.org/10.3390/vaccines13090910
APA StyleScuotto, A., Ogonczyk-Makowska, D., Quiévy, A., Berthet, M., Schlax, K., Boussarie, D., Maillot, A., Popelin-Wedlarski, F., Charpentier, T., Perot, M., Quintard, B., van Elderen, M., Stumpel, J. B. G., Tahas, S. A., Modlinska, A., Sós-Koroknai, V., Azevedo, A., Muciño, M. d. C. C., Martins, M. C., ... Betbeder, D. (2025). Retrospective Analysis of the Impact of Vaccination with an Inactivated Vaccine on Toxoplasmosis-Associated Mortality in Captive Wildlife. Vaccines, 13(9), 910. https://doi.org/10.3390/vaccines13090910







