The Concordance between Mumps and Rubella Sero-Positivity among the Israeli Population in 2015
Abstract
:1. Introduction
2. Methods
3. Results
4. Discussion
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- The Division of Epidemiology, Public Health Services, Israel Ministry of Health. Vaccines Instructions. 2022. Available online: https://www.health.gov.il/UnitsOffice/HD/PH/epidemiology/td/docs/tadrich_Chisunim.pdf (accessed on 18 June 2022).
- Israel Center for Diseases Control (ICDC) and Division of Epidemiology, Public Health Services. Notifiable Infectious Diseases in Israel: 65 Years of Surveillance 1951–2015; Israeli Ministry of Health: Jerusalem, Israel, 2020.
- Characterization and Classification of Local Authorities by the Socio-Economic Level of the Population in 2015. Local Councils and Municipalities—Rank, Cluster Membership, Population, Variable Values, Standardized Values and Ranking for the Variables Used in the Computation of the Index. Available online: https://www.cbs.gov.il/he/publications/Pages/2019/אפיון-יחידות-גאוגרפיות-וסיווגן-לפי-הרמה-החברתית-כלכלית-של-האוכלוסייה-בשנת-2015.aspx (accessed on 18 June 2022).
- Bassal, R.; Cohen, D.; Green, M.S.; Keinan-Boker, L. The Israel National Sera Bank: Methods, Representativeness, and Challenges. Int. J. Environ. Res. Public Health 2021, 18, 2280. [Google Scholar] [CrossRef] [PubMed]
- Devika, S.; Jeyaseelan, L.; Sebastian, G. Analysis of sparse data in logistic regression in medical research: A newer approach. J. Postgrad. Med. 2016, 62, 26–31. [Google Scholar] [PubMed]
- Santacruz-Sanmartín, E.; Hincapié-Palacio, D.; Ospina, M.C.; Perez-Toro, O.; Bernal-Restrepo, L.M.; Buitrago-Giraldo, S.; Lenis-Ballesteros, V.; Díaz, F.J. Seroprevalence of mumps in an epidemic period in Medellín, Colombia. Vaccine 2015, 33, 5606–5612. [Google Scholar] [CrossRef] [PubMed]
- Domínguez, A.; Plans, P.; Costa, J.; Torner, N.; Cardenosa, N.; Batalla, J.; Plasencia, A.; Salleras, L. Seroprevalence of measles, rubella, and mumps antibodies in Catalonia, Spain: Results of a cross-sectional study. Eur. J. Clin. Microbiol. Infect. Dis. 2006, 25, 310–317. [Google Scholar] [CrossRef] [PubMed]
- González-Escalada, A.; García-García, L.; Viguera-Ester, P.; Marín-García, P.; García, J.; Gil-de-Miguel, A.; Gil-Prieto, R. Seroprevalence of antibodies against measles, rubella, mumps, varicella-zoster, and B. Pertussis in young adults of Madrid, Spain. Hum. Vaccines Immunother. 2013, 9, 1918–1925. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lebo, E.J.; Kruszon-Moran, D.M.; Marin, M.; Bellini, W.J.; Schmid, S.; Bialek, S.R.; Wallace, G.S.; McLean, H.Q. Seroprevalence of measles, mumps, rubella and varicella antibodies in the United States population, 2009–2010. Open Forum. Infect. Dis. 2015, 2, ofv006. [Google Scholar] [CrossRef] [PubMed]
- Pang, H.; Zhou, Y.; Zhao, W.; Jiang, Q. Seroprevalence and Determinants Associated with Mumps Antibodies after 20 Years of MMR Vaccination in Urban Area of Shanghai, China. Int. J. Environ. Res. Public Health 2018, 15, 2089. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Tharmaphornpilas, P.; Yoocharean, P.; Rasdjarmrearnsook, A.O.; Theamboonlers, A.; Poovorawan, Y. Seroprevalence of antibodies to measles, mumps, and rubella among Thai population: Evaluation of measles/MMR immunization programme. J. Health Popul. Nutr. 2009, 27, 80–86. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ho, Y.H.; Tsai, C.C.; Tsai, Y.W.; Wang, Y.C.; Lin, T.Y.; Lee, D.J.; Chen, C.J. Humoral immunity to mumps in a highly vaccinated population in Taiwan. J. Microbiol. Immunol. Infect. 2019, 52, 379–385. [Google Scholar] [CrossRef]
- Smetana, J.; Chlibek, R.; Hanovcova, I.; Sosovickova, R.; Smetanova, L.; Polcarova, P.; Gal, P.; Dite, P. Serological survey of mumps antibodies in adults in the Czech Republic and the need for changes to the vaccination strategy. Hum. Vaccines Immunother. 2018, 14, 887–893. [Google Scholar] [CrossRef]
- Anderson, R.M.; May, R.M. Immunisation and herd immunity. Lancet 1990, 335, 641–645. [Google Scholar] [CrossRef]
- Eriksen, J.; Davidkin, I.; Kafatos, G.; Andrews, N.; Barbara, C.; Cohen, D.; Duks, A.; Griskevicius, A.; Johansen, K.; Bartha, K.; et al. Seroepidemiology of mumps in Europe (1996–2008): Why do outbreaks occur in highly vaccinated populations? Epidemiol. Infect. 2013, 141, 651–666. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- World Health Organization. WHO Vaccine-Preventable Diseases: Monitoring System. Available online: https://apps.who.int/immunization_monitoring/globalsummary/timeseries/tscoveragemcv2.html (accessed on 18 June 2022).
- Mossong, J.; Putz, L.; Schneider, F. Seroprevalence of measles, mumps and rubella antibodies in Luxembourg: Results from a national cross-sectional study. Epidemiol. Infect. 2004, 132, 11–18. [Google Scholar] [CrossRef] [PubMed]
- Muhsen, K.; Aboudy, Y.; Mendelson, E.; Green, M.S.; Cohen, D. Prevalence of mumps antibodies in the Israeli population in relation to mumps vaccination policy and incidence of disease. Epidemiol. Infect. 2008, 136, 688–693. [Google Scholar] [CrossRef]
- Seagle, E.E.; Bednarczyk, R.A.; Hill, T.; Fiebelkorn, A.P.; Hickman, C.J.; Icenogle, J.P.; Belongia, E.A.; McLean, H.Q. Measles, mumps, and rubella antibody patterns of persistence and rate of decline following the second dose of the MMR vaccine. Vaccine 2018, 36, 818–826. [Google Scholar] [CrossRef] [Green Version]
- Zamir, C.S.; Schroeder, H.; Shoob, H.; Abramson, N.; Zentner, G. Characteristics of a large mumps outbreak: Clinical severity, complications and association with vaccination status of mumps outbreak cases. Hum. Vaccines Immunother. 2015, 11, 1413–1417. [Google Scholar] [CrossRef] [Green Version]
- Lam, E.; Rosen, J.B.; Zucker, J.R. Mumps: An Update on Outbreaks, Vaccine Efficacy, and Genomic Diversity. Clin. Microbiol. Rev. 2020, 33, e00151-19. [Google Scholar] [CrossRef]
- Smits, G.; Mollema, L.; Hahné, S.; de Melker, H.; Tcherniaeva, I.; van der Klis, F.; Berbers, G. Seroprevalence of rubella antibodies in The Netherlands after 32 years of high vaccination coverage. Vaccine 2014, 32, 1890–1895. [Google Scholar] [CrossRef]
- Edirisuriya, C.; Beard, F.H.; Hendry, A.J.; Dey, A.; Gidding, H.F.; Hueston, L.; Dwyer, D.E.; Wood, J.G.; Macartney, K.K.; McIntyre, P.B. Australian rubella serosurvey 2012–2013: On track for elimination? Vaccine 2018, 36, 2794–2798. [Google Scholar] [CrossRef]
- World Health Organization. The Immunological Basis for Immunization Series—Module 11—Rubella. Available online: https://apps.who.int/iris/bitstream/handle/10665/43922/9789241596848_eng.pdf;jsessionid=52CEB6A406A2ED553A762ABD35AFB08E?sequence=1 (accessed on 18 June 2022).
- Poethko-Müller, C.; Mankertz, A. Seroprevalence of measles-, mumps- and rubella-specific IgG antibodies in German children and adolescents and predictors for seronegativity. PLoS ONE 2012, 7, e42867. [Google Scholar] [CrossRef]
- Béraud, G.; Abrams, S.; Beutels, P.; Dervaux, B.; Hens, N. Resurgence risk for measles, mumps and rubella in France in 2018 and 2020. Eurosurveillance 2018, 23, 1700796. [Google Scholar] [CrossRef] [PubMed]
| Tested | Sero-Positive | ||||||
|---|---|---|---|---|---|---|---|
| N | % | N | % | 95% CI £ | p-Value | ||
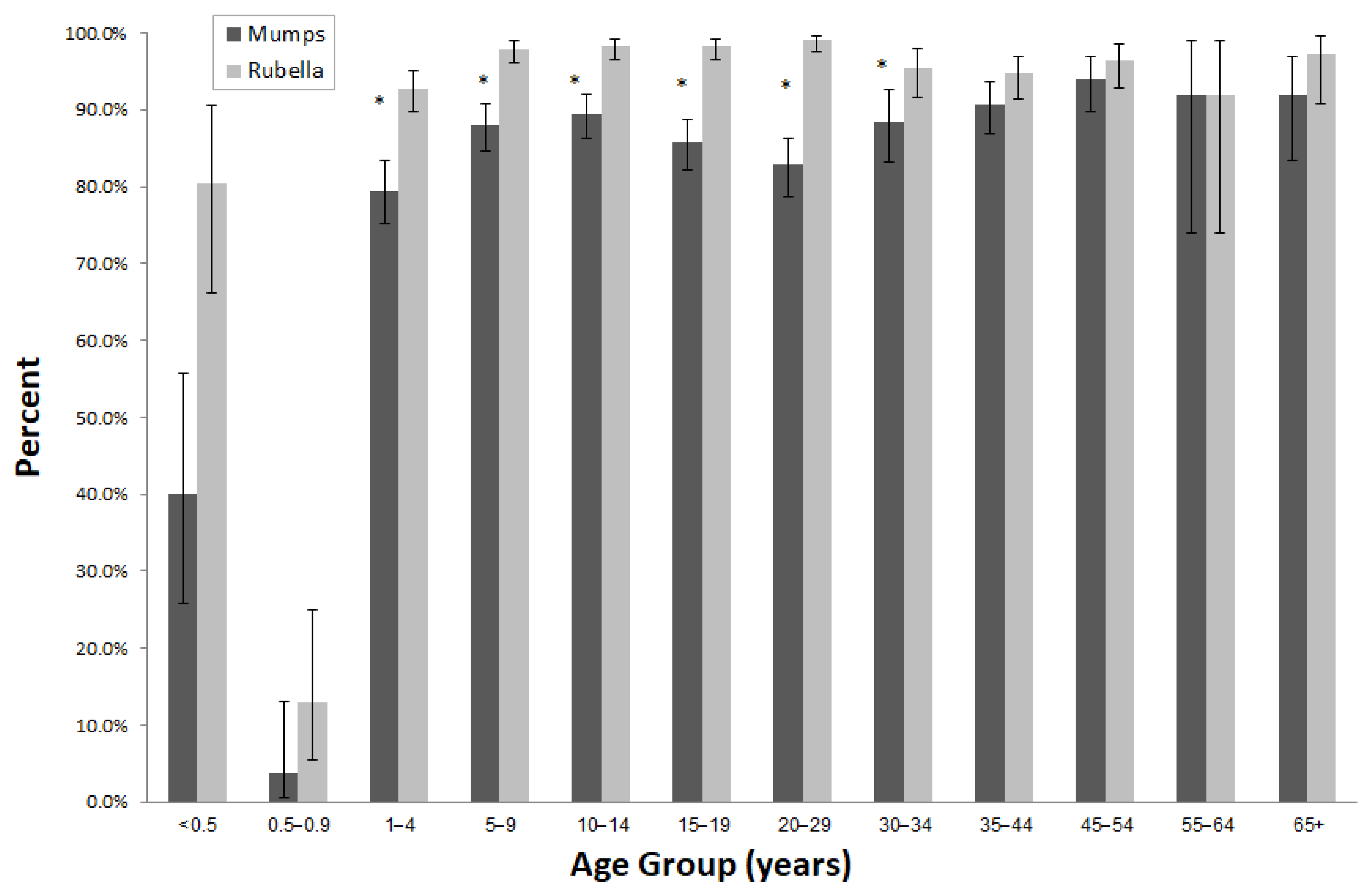
| Age group (years) | <0.5 | 45 | 1.4 | 18 | 40.0 | 25.7–55.7 | <0.0001 |
| 0.5–0.9 | 53 | 1.7 | 2 | 3.8 | 0.5–13.0 | ||
| 1–4 | 395 | 12.6 | 314 | 79.5 | 75.2–83.4 | ||
| 5–9 | 467 | 14.9 | 411 | 88.0 | 84.7–90.8 | ||
| 10–14 | 489 | 15.6 | 437 | 89.4 | 86.3–92.0 | ||
| 15–19 | 482 | 15.4 | 413 | 85.7 | 82.2–88.7 | ||
| 20–29 | 400 | 12.8 | 331 | 82.8 | 78.7–86.3 | ||
| 30–34 | 200 | 6.4 | 177 | 88.5 | 83.2–92.6 | ||
| 35–44 | 300 | 9.6 | 272 | 90.7 | 86.8–93.7 | ||
| 45–54 | 200 | 6.4 | 188 | 94.0 | 89.8–96.9 | ||
| 55–64 | 25 | 0.8 | 23 | 92.0 | 74.0–99.0 | ||
| 65+ | 75 | 2.4 | 69 | 92.0 | 83.4–97.0 | ||
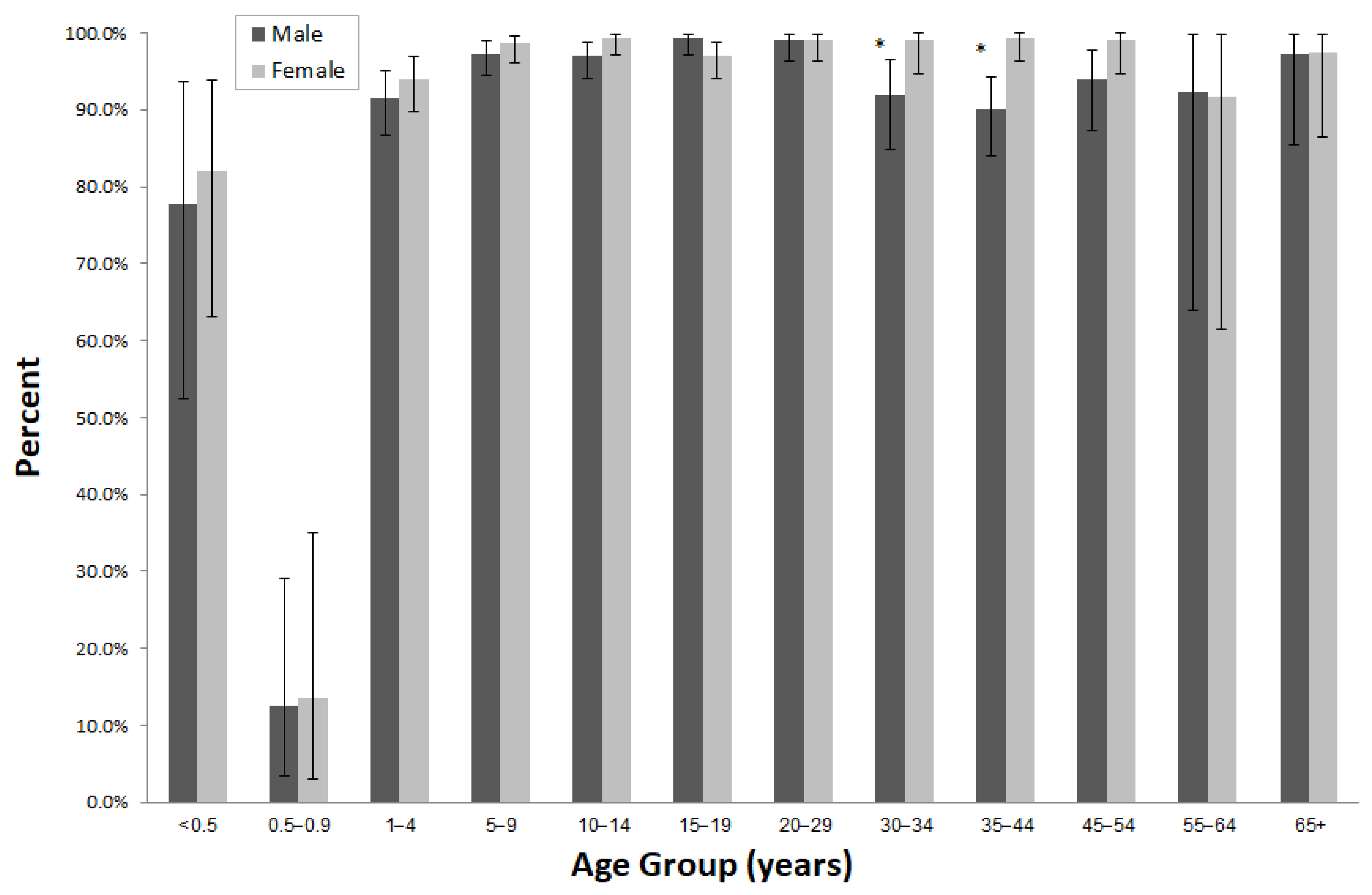
| Sex | Male | 1581 | 50.5 | 1322 | 83.6 | 81.7–85.4 | 0.0634 |
| Female | 1550 | 49.5 | 1333 | 86.0 | 84.2–87.7 | ||
| Birth country | Israel | 2957 | 94.5 | 2507 | 84.8 | 83.4–86.1 | 0.9463 |
| Others | 173 | 5.5 | 147 | 85.0 | 78.8–89.9 | ||
| Population group | Jews and Others | 1750 | 55.9 | 1494 | 85.4 | 83.6–87.0 | 0.3137 |
| Arabs | 1381 | 44.1 | 1161 | 84.1 | 82.0–86.0 | ||
| District | Jerusalem | 71 | 2.3 | 63 | 88.7 | 79.0–95.0 | 0.0005 |
| North | 820 | 26.2 | 727 | 88.7 | 86.3–90.8 | ||
| Haifa | 354 | 11.3 | 313 | 88.4 | 84.6–91.6 | ||
| Central | 576 | 18.4 | 468 | 81.2 | 77.8–84.4 | ||
| Tel-Aviv | 292 | 9.3 | 238 | 81.5 | 76.6–85.8 | ||
| South | 906 | 28.9 | 753 | 83.1 | 80.5–85.5 | ||
| Judea and Samaria | 112 | 3.6 | 93 | 83.0 | 74.8–89.5 | ||
| Socioeconomic status (SES) | Low | 2014 | 64.3 | 1689 | 83.9 | 82.2–85.4 | 0.0506 |
| High | 1117 | 35.7 | 966 | 86.5 | 84.3–88.4 |
| Sero-negativity-Univariable | Sero-negativity-Multivariable | ||||||
|---|---|---|---|---|---|---|---|
| OR € | 95% CI £ | p-Value | OR € | 95% CI £ | p-Value | ||
| Age group (years) | <0.5 | 11.01 | 5.70–21.27 | <0.0001 | 9.95 | 5.12–19.34 | <0.0001 |
| 0.5–0.9 | 187.15 | 44.34–790.00 | <0.0001 | 173.46 | 41.02–733.45 | <0.0001 | |
| 1–4 | 1.89 | 1.31–2.74 | 0.0007 | 1.83 | 1.26–2.66 | 0.0015 | |
| 5–9 | Ref. | ||||||
| 10–14 | 0.87 | 0.58–1.30 | 0.5077 | 0.86 | 0.58–1.29 | 0.4752 | |
| 15–19 | 1.23 | 0.84–1.79 | 0.2905 | 1.21 | 0.83–1.77 | 0.3220 | |
| 20–29 | 1.53 | 1.04–2.24 | 0.0287 | 1.51 | 1.03–2.22 | 0.0351 | |
| 30–34 | 0.95 | 0.57–1.60 | 0.8572 | 0.94 | 0.56–1.58 | 0.8137 | |
| 35–44 | 0.76 | 0.47–1.22 | 0.2511 | 0.73 | 0.45–1.18 | 0.1965 | |
| 45–54 | 0.47 | 0.24–0.90 | 0.0216 | 0.46 | 0.24–0.88 | 0.0186 | |
| 55–64 | 0.64 | 0.15–2.78 | 0.5498 | 0.66 | 0.15–2.86 | 0.5746 | |
| 65+ | 0.64 | 0.26–1.54 | 0.3170 | 0.62 | 0.26–1.50 | 0.2904 | |
| Sex | Male | 1.20 | 0.99–1.46 | 0.0637 | |||
| Female | Ref. | ||||||
| Birth country | Israel | 1.02 | 0.66–1.56 | 0.9465 | |||
| Others | Ref. | ||||||
| Population group | Jews and Others | 0.90 | 0.74–1.10 | 0.3139 | |||
| Arabs | Ref. | ||||||
| District | Jerusalem | 0.55 | 0.26–1.18 | 0.1258 | 0.64 | 0.30–1.39 | 0.2584 |
| North | 0.55 | 0.41–0.75 | 0.0001 | 0.64 | 0.46–0.87 | 0.0053 | |
| Haifa | 0.57 | 0.38–0.84 | 0.0041 | 0.62 | 0.41–0.93 | 0.0209 | |
| Central | Ref. | ||||||
| Tel-Aviv | 0.98 | 0.68–1.41 | 0.9269 | 0.90 | 0.60–1.35 | 0.6119 | |
| South | 0.88 | 0.67–1.16 | 0.3590 | 0.89 | 0.66–1.20 | 0.4463 | |
| Judea and Samaria | 0.88 | 0.52–1.51 | 0.6560 | 0.92 | 0.52–1.62 | 0.7729 | |
| Socioeconomic status (SES) | Low | 1.23 | 1.00–1.52 | 0.0509 | |||
| High | Ref. | ||||||
| Tested | Sero-Positive | ||||||
|---|---|---|---|---|---|---|---|
| N | % | N | % | 95% CI £ | p-Value | ||
| Age group (years) | <0.5 | 46 | 1.4 | 37 | 80.4 | 66.1–90.6 | <0.0001 |
| 0.5–0.9 | 54 | 1.7 | 7 | 13.0 | 5.4–24.9 | ||
| 1–4 | 400 | 12.6 | 371 | 92.8 | 89.8–95.1 | ||
| 5–9 | 480 | 15.2 | 470 | 97.9 | 96.2–99.0 | ||
| 10–14 | 500 | 15.8 | 491 | 98.2 | 96.6–99.2 | ||
| 15–19 | 489 | 15.4 | 480 | 98.2 | 96.5–99.2 | ||
| 20–29 | 400 | 12.6 | 396 | 99.0 | 97.5–99.7 | ||
| 30–34 | 200 | 6.3 | 191 | 95.5 | 91.6–97.9 | ||
| 35–44 | 300 | 9.5 | 284 | 94.7 | 91.5–96.9 | ||
| 45–54 | 200 | 6.3 | 193 | 96.5 | 92.9–98.6 | ||
| 55–64 | 25 | 0.8 | 23 | 92.0 | 74.0–99.0 | ||
| 65+ | 75 | 2.4 | 73 | 97.3 | 90.7–99.7 | ||
| Sex | Male | 1600 | 50.5 | 1502 | 93.9 | 92.6–95.0 | 0.0006 |
| Female | 1569 | 49.5 | 1514 | 96.5 | 95.5–97.4 | ||
| Birth country | Israel | 2995 | 94.5 | 2852 | 95.2 | 94.4–96.0 | 0.5486 |
| Others | 173 | 5.5 | 163 | 94.2 | 89.6–97.2 | ||
| Population group | Jews and Others | 1781 | 56.2 | 1683 | 94.5 | 93.3–95.5 | 0.0448 |
| Arabs | 1388 | 43.8 | 1333 | 96.0 | 94.9–97.0 | ||
| District | Jerusalem | 72 | 2.3 | 71 | 98.6 | 92.5–100.00 | 0.0001 |
| North | 823 | 26.0 | 800 | 97.2 | 95.8–98.2 | ||
| Haifa | 357 | 11.3 | 340 | 95.2 | 92.5–97.2 | ||
| Central | 593 | 18.7 | 548 | 92.4 | 90.0–94.4 | ||
| Tel-Aviv | 302 | 9.5 | 278 | 92.0 | 88.4–94.8 | ||
| South | 908 | 28.6 | 868 | 95.6 | 94.0–96.8 | ||
| Judea and Samaria | 114 | 3.6 | 111 | 97.4 | 92.5–99.4 | ||
| Socioeconomic status (SES) | Low | 2028 | 64.0 | 1942 | 95.8 | 94.8–96.6 | 0.0397 |
| High | 1141 | 36.0 | 1074 | 94.1 | 92.6–95.4 |
| Sero-Negativity—Univariable | Sero-Negativity—Multivariable | ||||||
|---|---|---|---|---|---|---|---|
| OR € | 95% CI £ | p-Value | OR € | 95% CI £ | p-Value | ||
| Age group (years) | <0.5 | 47.84 | 47.16–48.52 | <0.0001 | 44.56 | 43.92–45.21 | <0.0001 |
| 0.5–0.9 | 18.05 | 8.85–27.25 | 0.0001 | 17.57 | 10.06–25.08 | <0.0001 | |
| 1–4 | 2.98 | 2.95–3.01 | <0.0001 | 3.01 | 2.98–3.04 | <0.0001 | |
| 5–9 | Ref. | ||||||
| 10–14 | 0.94 | 0.93–0.95 | <0.0001 | 0.97 | 0.96–0.98 | <0.0001 | |
| 15–19 | 0.95 | 0.94–0.96 | <0.0001 | 0.99 | 0.98–1.00 | 0.0557 | |
| 20–29 | 0.80 | 0.79–0.81 | <0.0001 | 0.85 | 0.84–0.86 | <0.0001 | |
| 30–34 | 1.71 | 1.68–1.74 | <0.0001 | 1.77 | 1.74–1.80 | <0.0001 | |
| 35–44 | 2.01 | 1.98–2.03 | <0.0001 | 2.06 | 2.04–2.09 | <0.0001 | |
| 45–54 | 1.38 | 1.36–1.41 | <0.0001 | 1.44 | 1.41–1.46 | <0.0001 | |
| 55–64 | 4.98 | 4.84–5.12 | <0.0001 | 5.06 | 4.92–5.20 | <0.0001 | |
| 65+ | 1.26 | 1.22–1.30 | <0.0001 | 1.32 | 1.28–1.36 | <0.0001 | |
| Sex | Male | 1.80 | 1.28–2.52 | 0.0007 | 1.56 | 1.55–1.57 | <0.0001 |
| Female | Ref. | ||||||
| Birth country | Israel | Ref. | |||||
| Others | 1.22 | 0.63–2.37 | 0.5489 | ||||
| Population group | Jews and Others | 1.41 | 1.01–1.98 | 0.0458 | 1.10 | 1.10–1.11 | <0.0001 |
| Arabs | Ref. | ||||||
| District | Jerusalem | 0.17 | 0.02–1.26 | 0.0836 | 0.57 | 0.55–0.59 | <0.0001 |
| North | 0.35 | 0.21–0.58 | <0.0001 | 0.58 | 0.58–0.59 | <0.0001 | |
| Haifa | 0.61 | 0.34–1.08 | 0.0903 | 0.77 | 0.76–0.78 | <0.0001 | |
| Central | Ref. | ||||||
| Tel-Aviv | 1.05 | 0.63–1.76 | 0.8492 | 0.84 | 0.83–0.85 | <0.0001 | |
| South | 0.56 | 0.36–0.87 | 0.0099 | 0.60 | 0.59–0.60 | <0.0001 | |
| Judea and Samaria | 0.33 | 0.10–1.08 | 0.0664 | 0.55 | 0.54–0.56 | <0.0001 | |
| Socioeconomic status (SES) | Low | Ref. | |||||
| High | 1.41 | 1.02–1.96 | 0.0405 | ||||
| Rubella Sero-prevalence | ||||
|---|---|---|---|---|
| Mumps sero-prevalence | Negative N (%) | Positive N (%) | Total N (%) | |
| Negative N (%) | 121 (3.9) | 355 (11.3) | 476 (15.2) | |
| Positive N (%) | 29 (0.9) | 2626 (83.9) | 2655 (84.8) | |
| Total N (%) | 150 (4.8) | 2981 (95.2) | 3131 (100.0) | |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bassal, R.; Shohat, T.; Levin, T.; Pando, R.; Shinar, E.; Amichay, D.; Barak, M.; Ben-Dor, A.; Bar-Haim, A.; Mendelson, E.; et al. The Concordance between Mumps and Rubella Sero-Positivity among the Israeli Population in 2015. Vaccines 2022, 10, 996. https://doi.org/10.3390/vaccines10070996
Bassal R, Shohat T, Levin T, Pando R, Shinar E, Amichay D, Barak M, Ben-Dor A, Bar-Haim A, Mendelson E, et al. The Concordance between Mumps and Rubella Sero-Positivity among the Israeli Population in 2015. Vaccines. 2022; 10(7):996. https://doi.org/10.3390/vaccines10070996
Chicago/Turabian StyleBassal, Ravit, Tamy Shohat, Tal Levin, Rakefet Pando, Eilat Shinar, Doron Amichay, Mira Barak, Anat Ben-Dor, Adina Bar-Haim, Ella Mendelson, and et al. 2022. "The Concordance between Mumps and Rubella Sero-Positivity among the Israeli Population in 2015" Vaccines 10, no. 7: 996. https://doi.org/10.3390/vaccines10070996
APA StyleBassal, R., Shohat, T., Levin, T., Pando, R., Shinar, E., Amichay, D., Barak, M., Ben-Dor, A., Bar-Haim, A., Mendelson, E., Cohen, D., Keinan-Boker, L., & Indenbaum, V. (2022). The Concordance between Mumps and Rubella Sero-Positivity among the Israeli Population in 2015. Vaccines, 10(7), 996. https://doi.org/10.3390/vaccines10070996






