COVID-19 Vaccination Might Induce Postural Orthostatic Tachycardia Syndrome: A Case Report
Abstract
:1. Introduction
2. Case Presentation
3. Discussion
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Bryarly, M.; Phillips, L.T.; Fu, Q.; Vernino, S.; Levine, B.D. Postural Orthostatic Tachycardia Syndrome: JACC Focus Seminar. J. Am. Coll. Cardiol. 2019, 73, 1207–1228. [Google Scholar] [CrossRef]
- Raj, S.R. The Postural Tachycardia Syndrome (POTS): Pathophysiology, Diagnosis & Management. Indian Pacing Electrophysiol. J. 2006, 6, 84–99. [Google Scholar]
- Sheldon, R.S.; Grubb, B.P.; Olshansky, B.; Shen, W.-K.; Calkins, H.; Brignole, M.; Raj, S.R.; Krahn, A.D.; Morillo, C.A.; Stewart, J.M.; et al. 2015 heart rhythm society expert consensus statement on the diagnosis and treatment of postural tachycardia syndrome, inappropriate sinus tachycardia, and vasovagal syncope. Heart Rhythm. 2015, 12, e41–e63. [Google Scholar] [CrossRef] [Green Version]
- Johansson, M.; Ståhlberg, M.; Runold, M.; Nygren-Bonnier, M.; Nilsson, J.; Olshansky, B.; Bruchfeld, J.; Fedorowski, A. Long-Haul Post-COVID-19 Symptoms Presenting as a Variant of Postural Orthostatic Tachycardia Syndrome: The Swedish Experience. JACC Case Rep. 2021, 3, 573–580. [Google Scholar] [CrossRef]
- Venkatesan, P. NICE guideline on long COVID. Lancet Respir. Med. 2021, 9, 129. [Google Scholar] [CrossRef]
- Bisaccia, G.; Ricci, F.; Recce, V.; Serio, A.; Iannetti, G.; Chahal, A.A.; Ståhlberg, M.; Khanji, M.Y.; Fedorowski, A.; Gallina, S. Post-Acute Sequelae of COVID-19 and Cardiovascular Autonomic Dysfunction: What Do We Know? J. Cardiovasc. Dev. Dis. 2021, 8, 156. [Google Scholar] [CrossRef]
- Long COVID: Let Patients Help Define Long-Lasting COVID Symptoms. Available online: https://www.nature.com/articles/d41586-020-02796-2 (accessed on 2 June 2022).
- Larsen, N.W.; Stiles, L.E.; Miglis, M.G. Preparing for the long-haul: Autonomic complications of COVID-19. Auton. Neurosci. Basic Clin. 2021, 235, 102841. [Google Scholar] [CrossRef]
- Goodman, B.P.; Khoury, J.A.; Blair, J.E.; Grill, M.F. COVID-19 Dysautonomia. Front. Neurol. 2021, 12, 624968. [Google Scholar] [CrossRef]
- Carfì, A.; Bernabei, R.; Landi, F. Gemelli against COVID-19 Post-Acute Care Study Group Persistent Symptoms in Patients after Acute COVID-19. JAMA 2020, 324, 603–605. [Google Scholar] [CrossRef]
- Kamal, M.; Abo Omirah, M.; Hussein, A.; Saeed, H. Assessment and characterisation of post-COVID-19 manifestations. Int. J. Clin. Pract. 2021, 75, e13746. [Google Scholar] [CrossRef]
- Blitshteyn, S.; Whitelaw, S. Postural orthostatic tachycardia syndrome (POTS) and other autonomic disorders after COVID-19 infection: A case series of 20 patients. Immunol. Res. 2021, 69, 205–211. [Google Scholar] [CrossRef]
- Novak, P.; Mukerji, S.S.; Alabsi, H.S.; Systrom, D.; Marciano, S.P.; Felsenstein, D.; Mullally, W.J.; Pilgrim, D.M. Multisystem Involvement in Post-Acute Sequelae of Coronavirus Disease 19. Ann. Neurol. 2022, 91, 367–379. [Google Scholar] [CrossRef]
- Chadda, K.R.; Blakey, E.E.; Huang, C.L.-H.; Jeevaratnam, K. Long COVID-19 and Postural Orthostatic Tachycardia Syndrome- Is Dysautonomia to Be Blamed? Front. Cardiovasc. Med. 2022, 9, 860198. Available online: https://www.frontiersin.org/article/10.3389/fcvm.2022.860198 (accessed on 2 June 2022). [CrossRef]
- Shouman, K.; Vanichkachorn, G.; Cheshire, W.P.; Suarez, M.D.; Shelly, S.; Lamotte, G.J.; Sandroni, P.; Benarroch, E.E.; Berini, S.E.; Cutsforth-Gregory, J.K.; et al. Autonomic dysfunction following COVID-19 infection: An early experience. Clin. Auton. Res. Off. J. Clin. Auton. Res. Soc. 2021, 31, 385–394. [Google Scholar] [CrossRef]
- Reddy, S.; Reddy, S.; Arora, M. A Case of Postural Orthostatic Tachycardia Syndrome Secondary to the Messenger RNA COVID-19 Vaccine. Cureus 2021, 13, e14837. [Google Scholar] [CrossRef]
- Carroll, H.A.; Millar, E.; Deans, K.A. Vitamin B12 and D deficiency as cofactors of COVID-19 vaccine-induced chronic neurological adverse reactions: Two cases and a hypothesis. Res. Sq. 2022, 1–33. Available online: https://www.researchsquare.com/article/rs-1425014/v1 (accessed on 2 June 2022).
- Karimi Galougahi, K. Autonomic dysfunction post-inoculation with ChAdOx1 nCoV-19 vaccine. Eur. Heart J. Case Rep. 2021, 5, ytab472. [Google Scholar] [CrossRef]
- Hermel, M.; Sweeney, M.; Ni, Y.-M.; Bonakdar, R.; Triffon, D.; Suhar, C.; Mehta, S.; Dalhoumi, S.; Gray, J. Natural Supplements for COVID19-Background, Rationale, and Clinical Trials. J. Evid.-Based Integr. Med. 2021, 26, 2515690X211036875. [Google Scholar] [CrossRef]
- Schondorf, R.; Low, P.A. Idiopathic postural orthostatic tachycardia syndrome: An attenuated form of acute pandysautonomia? Neurology 1993, 43, 132–137. [Google Scholar] [CrossRef]
- Arana, J.; Mba-Jonas, A.; Jankosky, C.; Lewis, P.; Moro, P.L.; Shimabukuro, T.T.; Cano, M. Reports of Postural Orthostatic Tachycardia Syndrome After Human Papillomavirus Vaccination in the Vaccine Adverse Event Reporting System. J. Adolesc. Health Off. Publ. Soc. Adolesc. Med. 2017, 61, 577–582. [Google Scholar] [CrossRef]
- Blitshteyn, S. Postural tachycardia syndrome following human papillomavirus vaccination. Eur. J. Neurol. 2014, 21, 135–139. [Google Scholar] [CrossRef]
- Tomljenovic, L.; Colafrancesco, S.; Perricone, C.; Shoenfeld, Y. Postural Orthostatic Tachycardia with Chronic Fatigue after HPV Vaccination as Part of the “Autoimmune/Auto-inflammatory Syndrome Induced by Adjuvants”: Case Report and Literature Review. J. Investig. Med. High Impact Case Rep. 2014, 2, 2324709614527812. [Google Scholar] [CrossRef]
- Novak, P. Hypocapnic cerebral hypoperfusion: A biomarker of orthostatic intolerance. PLoS ONE 2018, 13, e0204419. [Google Scholar] [CrossRef] [Green Version]
- Raj, S.R.; Arnold, A.C.; Barboi, A.; Claydon, V.E.; Limberg, J.K.; Lucci, V.-E.M.; Numan, M.; Peltier, A.; Snapper, H.; Vernino, S.; et al. Long-COVID postural tachycardia syndrome: An American Autonomic Society statement. Clin. Auton. Res. Off. J. Clin. Auton. Res. Soc. 2021, 31, 365–368. [Google Scholar] [CrossRef]
- Huang, H.; Deb, A.; Culbertson, C.; Morgenshtern, K.; DePold Hohler, A. Dermatological Manifestations of Postural Tachycardia Syndrome Are Common and Diverse. J. Clin. Neurol. Seoul Korea 2016, 12, 75–78. [Google Scholar] [CrossRef] [Green Version]
- COVID-19 Infection Induced Raynaud’s Syndrome|Proceedings. Available online: https://proceedings.med.ucla.edu/index.php/2021/01/11/covid-19-infection-induced-raynauds-syndrome/ (accessed on 2 June 2022).
- Vanaparthy, R.; Malayala, S.V.; Balla, M. COVID-19-Induced Vestibular Neuritis, Hemi-Facial Spasms and Raynaud’s Phenomenon: A Case Report. Cureus 2020, 12, e11752. [Google Scholar] [CrossRef]
- Sachdeva, M.; Gianotti, R.; Shah, M.; Bradanini, L.; Tosi, D.; Veraldi, S.; Ziv, M.; Leshem, E.; Dodiuk-Gad, R.P. Cutaneous manifestations of COVID-19: Report of three cases and a review of literature. J. Dermatol. Sci. 2020, 98, 75–81. [Google Scholar] [CrossRef]
- Urban, N.; Weber, B.; Deinsberger, J.; Gschwandtner, M.; Bauer, W.; Handisurya, A. Raynaud’s Phenomenon after COVID-19 Vaccination: Causative Association, Temporal Connection, or Mere Bystander? Case Rep. Dermatol. 2021, 13, 450–456. [Google Scholar] [CrossRef]
- Lee, W.S.; Wheatley, A.K.; Kent, S.J.; DeKosky, B.J. Antibody-dependent enhancement and SARS-CoV-2 vaccines and therapies. Nat. Microbiol. 2020, 5, 1185–1191. [Google Scholar] [CrossRef]
- Dani, M.; Dirksen, A.; Taraborrelli, P.; Torocastro, M.; Panagopoulos, D.; Sutton, R.; Lim, P.B. Autonomic dysfunction in “long COVID”: Rationale, physiology and management strategies. Clin. Med. Lond. Engl. 2021, 21, e63–e67. [Google Scholar] [CrossRef]
- Goldstein, D.S. The extended autonomic system, dyshomeostasis, and COVID-19. Clin. Auton. Res. Off. J. Clin. Auton. Res. Soc. 2020, 30, 299–315. [Google Scholar] [CrossRef]
- Davido, B.; Seang, S.; Tubiana, R.; de Truchis, P. Post-COVID-19 chronic symptoms: A postinfectious entity? Clin. Microbiol. Infect. Off. Publ. Eur. Soc. Clin. Microbiol. Infect. Dis. 2020, 26, 1448–1449. [Google Scholar] [CrossRef]
- Kanjwal, K.; Jamal, S.; Kichloo, A.; Grubb, B.P. New-onset Postural Orthostatic Tachycardia Syndrome Following Coronavirus Disease 2019 Infection. J. Innov. Card. Rhythm Manag. 2020, 11, 4302–4304. [Google Scholar] [CrossRef]
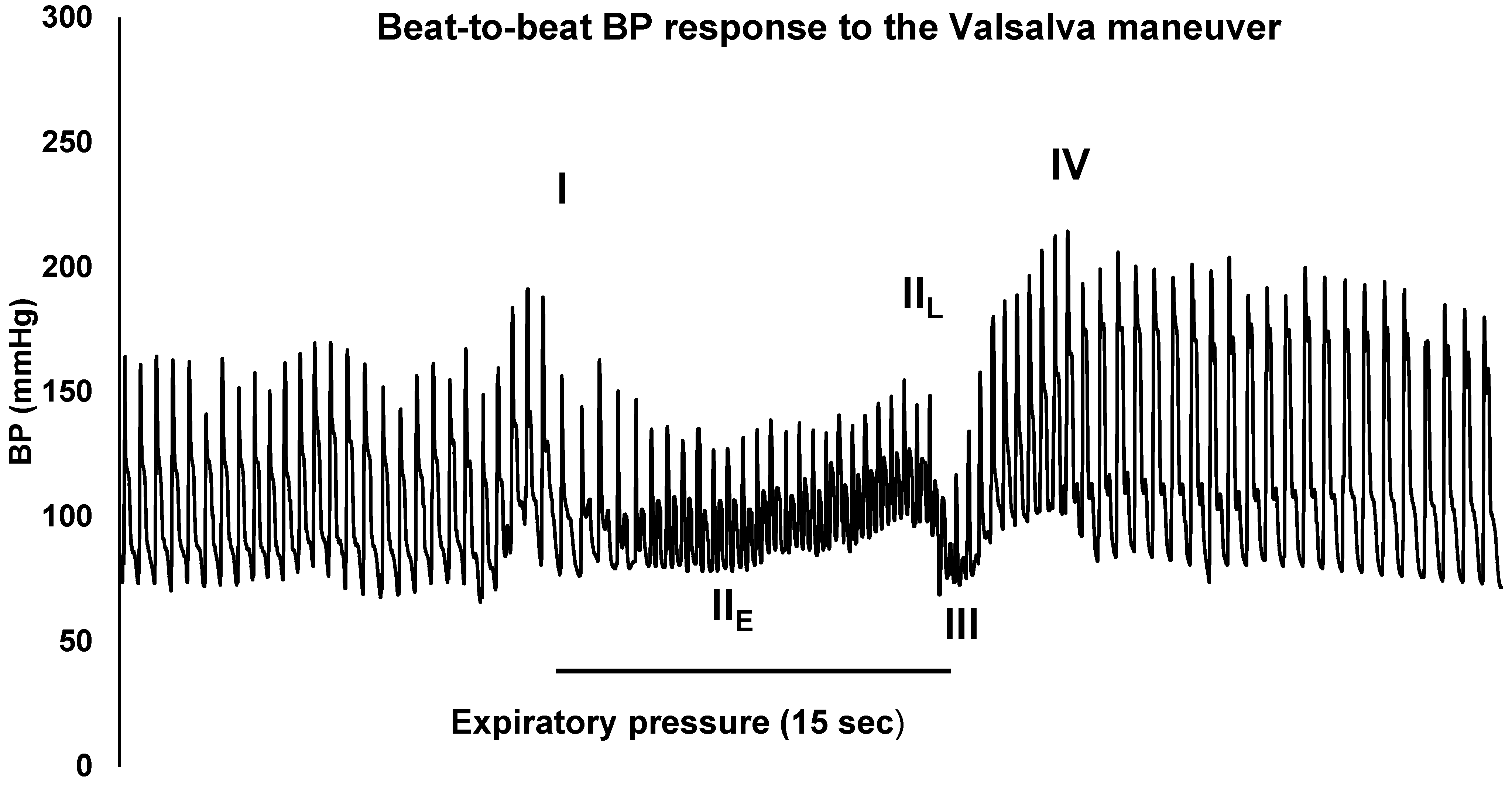
| Heart Rate Responses | ||||||||||||||||
| Test | Parameter | Result | Normal Values | Percentile % | ||||||||||||
| Deep Breathing | HR Range (bpm) | 20.4 | [>10.0] | 46 | ||||||||||||
| Valsalva Maneuver | Valsalva Ratio | 1.69 | [>1.51] | 19 | ||||||||||||
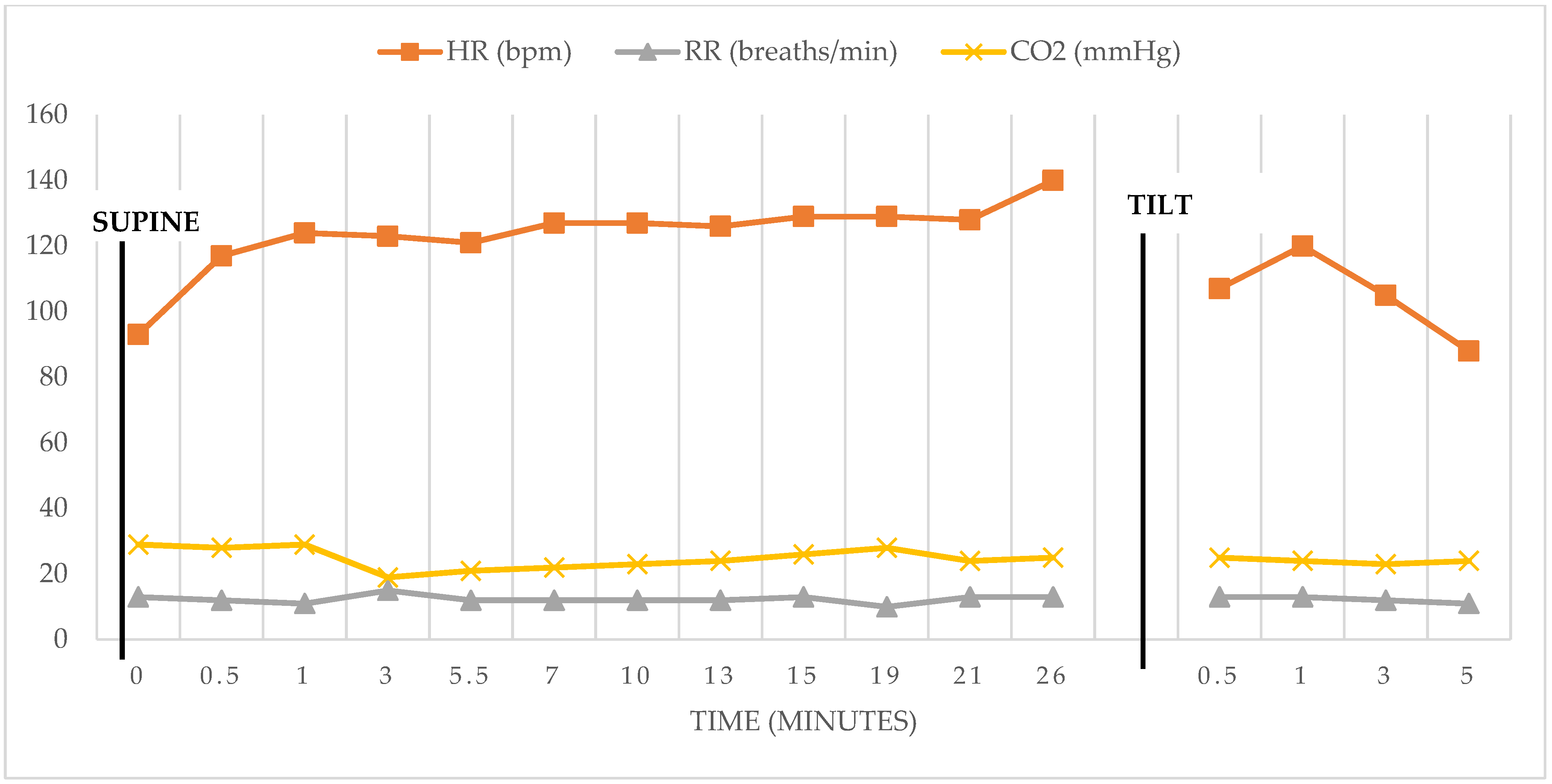
| Blood Pressure and Heart Rate Response to Head-up Tilt. | ||||||||||||||||
| Values with patient to 70 degrees head-up tilt | ||||||||||||||||
| Supine | 30 s | 1 min | 3 min | 5.5 min | 7 min | 10 min | 13 min | 15 min | 19 min | 21 min | 26 min | 30 s post tilt | 1 min post tilt | 3 min post tilt | 5 min post tilt | |
| BP (mmHg) | 102/69 | 115/75 | 122/85 | 106/77 | 101/76 | 114/80 | 113/83 | 113/77 | 111/82 | 114/83 | 117/84 | 113/78 | 102/68 | 99/65 | 109/72 | 108/74 |
| HR (bpm) | 93 | 117 | 124 | 123 | 121 | 127 | 127 | 126 | 129 | 129 | 128 | 140 | 107 | 120 | 105 | 88 |
| Respiration Rate (breaths/min) | 13 | 12 | 11 | 15 | 12 | 12 | 12 | 12 | 13 | 10 | 13 | 13 | 13 | 13 | 12 | 11 |
| EtCO2 (mmHg) | 29 | 28 | 29 | 19 | 21 | 22 | 23 | 24 | 26 | 28 | 24 | 25 | 25 | 24 | 23 | 24 |
| Author | Patient’s History | Vaccine | Presenting Symptoms/Timing | Diagnostic Test Results | Management |
|---|---|---|---|---|---|
| Our Case | 46-year-old woman, history of allergic rhinitis | First Pfizer-BioNTech COVID-19 Vaccine | Hives, urticaria, fatigue onset 1.5 h after vaccination; Dizziness, tremors, numbness, brain fog, headache onset 7-days after vaccination | CBC, Inflammatory Markers, Endocrine markers, electrolytes within normal limits. LFTs normal aside from elevated AST. Autonomic Testing including cardiovagal response to deep breathing and Valsalva maneuver assessment during tilt-table testing | Fluids, salt, compression stockings 20 mmHg, ivabradine 5 mg BID, natural supplements |
| Carroll, 2022 [17] | 30-year-old healthy female | First Oxford-AstraZeneca ChAdOx1nCoV-19 vaccination | Flu-like symptoms, dizziness, nausea, weakness, bradykinesia, brain fog, fatigue, appetite changes, loss of taste, polydipsia, numbness, tinnitus, micrographia and headaches onset 6-h post-vaccination | CBC, LFTs, Inflammatory Markers, Endocrine markers, electrolytes within normal limits. Deficiency of Vitamins B12, D2, D3, and total D. Chronically elevated D-dimer. Abnormal tilt-table test consistent with POTS, significant systolic blood pressure elevation | Replacement therapy of Vitamins B12 (1 mg intramuscular hydroxocobalamin, 3×/week) and Vitamin D (50,000 IU Stexerol-D3 weekly) |
| Galougahi, 2021 [18] | 29-year-old healthy male | First Oxford-AstraZeneca ChAdOx1nCoV-19 vaccination | Extremeity parasthesias 4-days post-vaccination; neuralgias, tachycardia, skin-color changes with mottling and blood pooling/dependent acrocyanosis, palpitations, dizziness 2-months post-vaccination | CBC, CMP, inflammatory markers, thyroid function, and folate were normal. B12 was mildly low. Positive low-titer ANA. Electrocardiogram and echocardiography were normal. Advanced neurologic serologies and imaging were negative. 5-min orthostatic tolerance testing | Vitamin B12 injections and Amitriptyline for neuralgia, 5-weeks of steroids, and lifestyle modifications |
| Reddy, 2021 [16] | 42-year-old male with history of hypothyroidism and B12 deficiency | First Pfizer-BioNTech COVID-19 Vaccine | Sinus tachycardia, dizziness, headaches, and fatigue onset 6-days after vaccination | CBC, LFTs, Inflammatory Markers, Endocrine markers, electrolytes within normal limits. Echocardiogram normal however noted sinus tachycardia (158 bpm) | Lifestyle modifications, compression socks, and sodium |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hermel, M.; Sweeney, M.; Abud, E.; Luskin, K.; Criado, J.P.; Bonakdar, R.; Gray, J.; Ahern, T. COVID-19 Vaccination Might Induce Postural Orthostatic Tachycardia Syndrome: A Case Report. Vaccines 2022, 10, 991. https://doi.org/10.3390/vaccines10070991
Hermel M, Sweeney M, Abud E, Luskin K, Criado JP, Bonakdar R, Gray J, Ahern T. COVID-19 Vaccination Might Induce Postural Orthostatic Tachycardia Syndrome: A Case Report. Vaccines. 2022; 10(7):991. https://doi.org/10.3390/vaccines10070991
Chicago/Turabian StyleHermel, Melody, Megan Sweeney, Edsel Abud, Kathleen Luskin, Jose P. Criado, Robert Bonakdar, James Gray, and Thomas Ahern. 2022. "COVID-19 Vaccination Might Induce Postural Orthostatic Tachycardia Syndrome: A Case Report" Vaccines 10, no. 7: 991. https://doi.org/10.3390/vaccines10070991
APA StyleHermel, M., Sweeney, M., Abud, E., Luskin, K., Criado, J. P., Bonakdar, R., Gray, J., & Ahern, T. (2022). COVID-19 Vaccination Might Induce Postural Orthostatic Tachycardia Syndrome: A Case Report. Vaccines, 10(7), 991. https://doi.org/10.3390/vaccines10070991






