COVID-19 Outbreak and BNT162b2 mRNA Vaccination Coverage in a Correctional Facility during Circulation of the SARS-CoV-2 Omicron BA.1 Variant in Italy
Abstract
:1. Introduction
2. Materials and Methods
2.1. Correctional Facilities
2.2. SARS-CoV-2 Vaccination Campaign
2.3. COVID-19 Outbreak and Screening Campaign
- (1)
- Unvaccinated: never received a vaccine dose.
- (2)
- Partially vaccinated: received the first dose of the two-dose series.
- (3)
- Fully vaccinated: received the second dose of the two-dose series.
- (4)
- Boostered: received a third dose after the two-dose series.
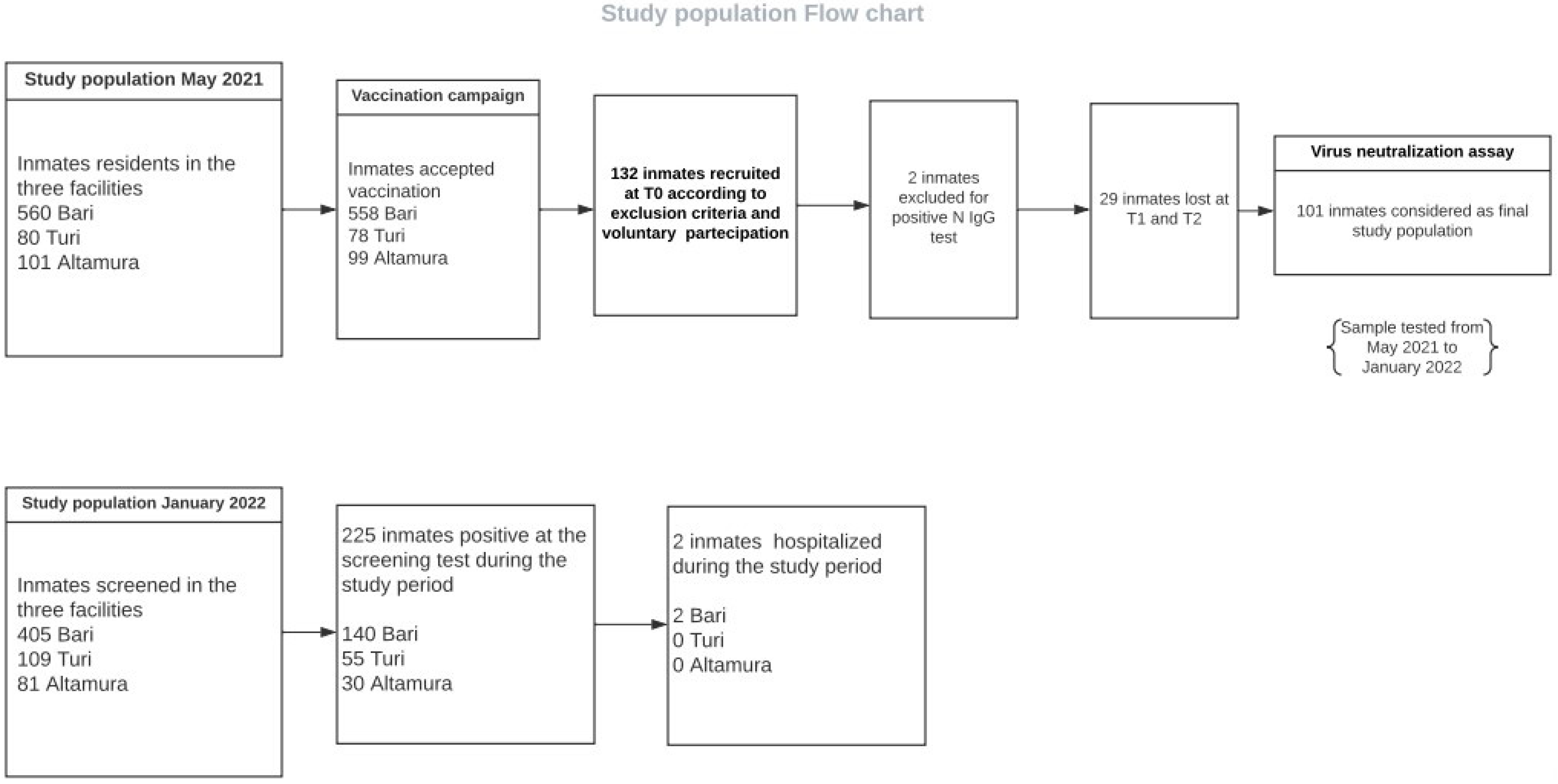
2.4. Humoral Immunity Survey: Study Cohort and Sample Collection
2.4.1. Enzyme-Linked Immunosorbent Assay (ELISA)
2.4.2. Virus Neutralization Assay
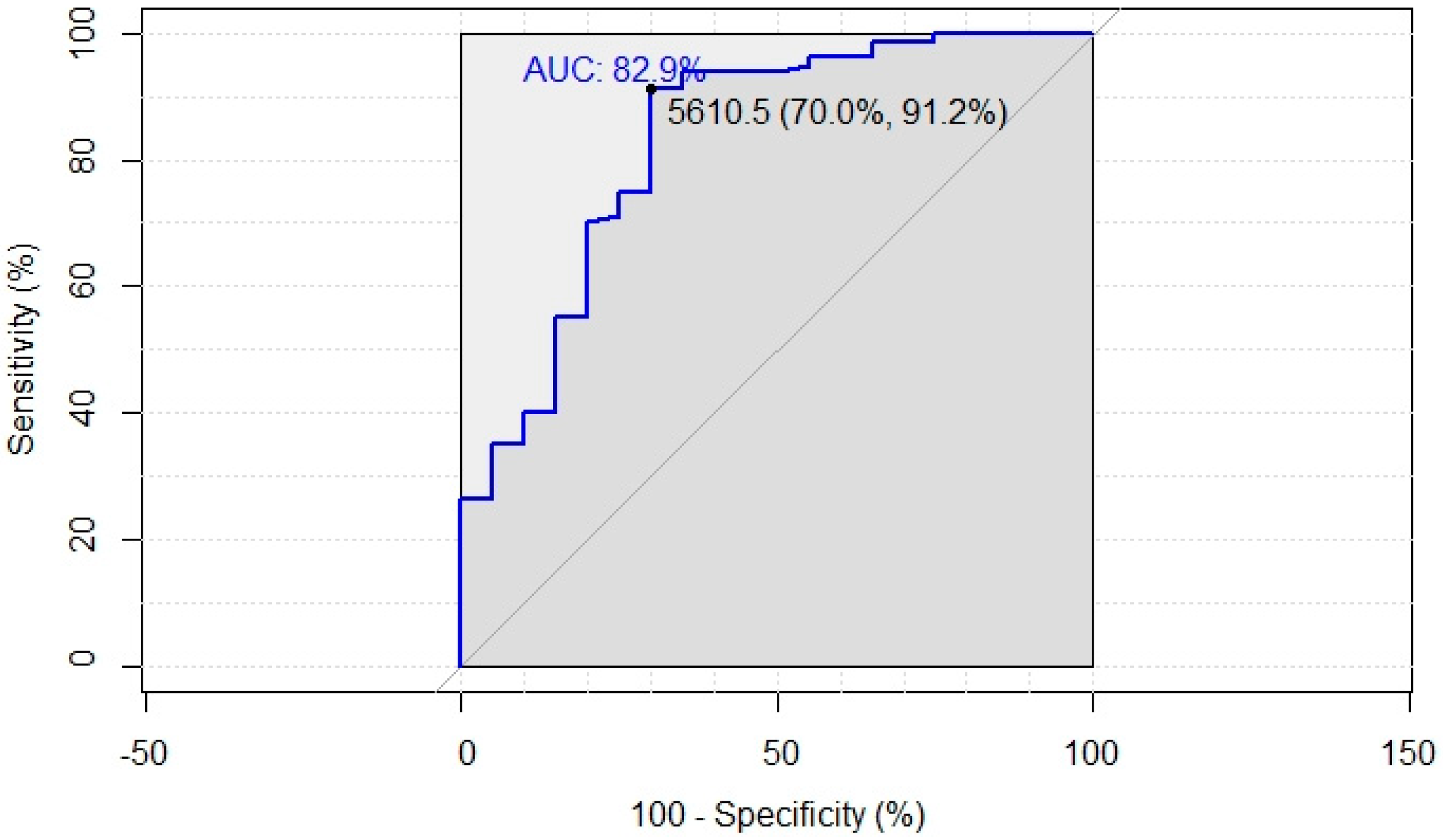
2.5. Statistical Analysis
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Mistry, P.; Barmania, F.; Mellet, J.; Peta, K.; Strydom, A.; Viljoen, I.M.; James, W.; Gordon, S.; Pepper, M.S. SARS-CoV-2 Variants, Vaccines, and Host Immunity. Front. Immunol. 2022, 12, 809244. [Google Scholar] [CrossRef] [PubMed]
- Maslo, C.; Friedland, R.; Toubkin, M.; Laubscher, A.; Akaloo, T.; Kama, B. Characteristics and Outcomes of Hospitalized Patients in South Africa during the COVID-19 Omicron Wave Compared with Previous Waves. JAMA 2022, 327, 583. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Rocklöv, J. The Effective Reproductive Number of the Omicron Variant of SARS-CoV-2 Is Several Times Relative to Delta. J. Travel Med. 2022, 29, taac037. [Google Scholar] [CrossRef]
- LeMasters, K.; Ranapurwala, S.; Maner, M.; Nowotny, K.M.; Peterson, M.; Brinkley-Rubinstein, L. COVID-19 Community Spread and Consequences for Prison Case Rates. PLoS ONE 2022, 17, e0266772. [Google Scholar] [CrossRef] [PubMed]
- Beaudry, G.; Zhong, S.; Whiting, D.; Javid, B.; Frater, J.; Fazel, S. Managing Outbreaks of Highly Contagious Diseases in Prisons: A Systematic Review. BMJ Glob. Health 2020, 5, e003201. [Google Scholar] [CrossRef] [PubMed]
- Hagan, L.M.; Dusseau, C.; Crockett, M.; Rodriguez, T.; Long, M.J. COVID-19 Vaccination in the Federal Bureau of Prisons, December 2020–April 2021. Vaccine 2021, 39, 5883–5890. [Google Scholar] [CrossRef]
- Kim, H.; Hughes, E.; Cavanagh, A.; Norris, E.; Gao, A.; Bondy, S.J.; McLeod, K.E.; Kanagalingam, T.; Kouyoumdjian, F.G. The Health Impacts of the COVID-19 Pandemic on Adults Who Experience Imprisonment Globally: A Mixed Methods Systematic Review. PLoS ONE 2022, 17, e0268866. [Google Scholar] [CrossRef]
- Stufano, A.; Buonvino, N.; Cagnazzo, F.; Armenise, N.; Pontrelli, D.; Curzio, G.; De Benedictis, L.; Lovreglio, P. Efficacy of the Measures Adopted to Prevent COVID-19 Outbreaks in an Italian Correctional Facility for Inmates Affected by Chronic Diseases. Front. Public Health 2021, 9, 694795. [Google Scholar] [CrossRef]
- EC. Safe COVID-19 Vaccines for Europeans. Available online: https://ec.europa.eu/info/live-work-travel-eu/coronavirus-response/safe-covid-19-vaccines-europeans_en (accessed on 26 May 2022).
- Pegu, A.; O’Connell, S.E.; Schmidt, S.D.; O’Dell, S.; Talana, C.A.; Lai, L.; Albert, J.; Anderson, E.; Bennett, H.; Corbett, K.S.; et al. Durability of MRNA-1273 Vaccine–Induced Antibodies against SARS-CoV-2 Variants. Science 2021, 373, 1372–1377. [Google Scholar] [CrossRef]
- Mannar, D.; Saville, J.W.; Zhu, X.; Srivastava, S.S.; Berezuk, A.M.; Tuttle, K.S.; Marquez, A.C.; Sekirov, I.; Subramaniam, S. SARS-CoV-2 Omicron Variant: Antibody Evasion and Cryo-EM Structure of Spike Protein–ACE2 Complex. Science 2022, 375, 760–764. [Google Scholar] [CrossRef]
- Strassle, C.; Jardas, E.; Ochoa, J.; Berkman, B.E.; Danis, M.; Rid, A.; Taylor, H.A. COVID-19 Vaccine Trials and Incarcerated People—The Ethics of Inclusion. N. Engl. J. Med. 2020, 383, 1897–1899. [Google Scholar] [CrossRef]
- Strodel, R.; Dayton, L.; Garrison-Desany, H.M.; Eber, G.; Beyrer, C.; Arscott, J.; Rubenstein, L.; Sufrin, C. COVID-19 Vaccine Prioritization of Incarcerated People Relative to Other Vulnerable Groups: An Analysis of State Plans. PLoS ONE 2021, 16, e0253208. [Google Scholar] [CrossRef] [PubMed]
- Wang, E.A.; Zenilman, J.; Brinkley-Rubinstein, L. Ethical Considerations for COVID-19 Vaccine Trials in Correctional Facilities. JAMA 2020, 324, 1031–1032. [Google Scholar] [CrossRef] [PubMed]
- Khoury, D.S.; Cromer, D.; Reynaldi, A.; Schlub, T.E.; Wheatley, A.K.; Juno, J.A.; Subbarao, K.; Kent, S.J.; Triccas, J.A.; Davenport, M.P. Neutralizing Antibody Levels Are Highly Predictive of Immune Protection from Symptomatic SARS-CoV-2 Infection. Nat. Med. 2021, 27, 1205–1211. [Google Scholar] [CrossRef] [PubMed]
- CDC. Overview of Testing for SARS-CoV-2, the Virus That Causes COVID-19. Available online: https://www.cdc.gov/coronavirus/2019-ncov/hcp/testing-overview.html (accessed on 26 May 2022).
- WHO. Laboratory Testing for 2019 Novel Coronavirus (2019-NCoV) in Suspected Human Cases. Available online: https://www.who.int/publications-detail-redirect/10665-331501 (accessed on 26 May 2022).
- Manenti, A.; Maggetti, M.; Casa, E.; Martinuzzi, D.; Torelli, A.; Trombetta, C.M.; Marchi, S.; Montomoli, E. Evaluation of SARS-CoV-2 Neutralizing Antibodies Using a CPE-based Colorimetric Live Virus Micro-neutralization Assay in Human Serum Samples. J. Med. Virol. 2020, 92, 2096–2104. [Google Scholar] [CrossRef]
- Italian Health Ministry. Piano Vaccini Anti COVID-19. Available online: https://www.salute.gov.it/portale/nuovocoronavirus/dettaglioContenutiNuovoCoronavirus.jsp?lingua=italiano&id=5452&area=nuovoCoronavirus&menu=vuoto (accessed on 26 May 2022).
- Vella, R.; Giuga, G.; Piizzi, G.; AlunniFegatelli, D.; Petroni, G.; Tavone, A.M.; Potenza, S.; Cammarano, A.; Mandarelli, G.; Marella, G.L. Health Management in Italian Prisons during COVID-19 Outbreak: A Focus on the Second and Third Wave. Healthcare 2022, 10, 282. [Google Scholar] [CrossRef]
- Ritchie, H.; Mathieu, E.; Rodés-Guirao, L.; Appel, C.; Giattino, C.; Ortiz-Ospina, E.; Hasell, J.; Macdonald, B.; Beltekian, D.; Roser, M. Coronavirus Pandemic (COVID-19). Our World Data. 2020. Available online: https://ourworldindata.org/coronavirus (accessed on 26 May 2022).
- COVID-19 Vaccinations for Prison Populations and Staff: Report on Global Scan. Available online: https://www.penalreform.org/resource/covid-19-vaccinations-report-on-global-scan/ (accessed on 26 May 2022).
- Di Giuseppe, G.; Pelullo, C.P.; Della Polla, G.; Montemurro, M.V.; Napolitano, F.; Pavia, M.; Angelillo, I.F. Surveying Willingness toward SARS-CoV-2 Vaccination of Healthcare Workers in Italy. Expert Rev. Vaccines 2021, 20, 881–889. [Google Scholar] [CrossRef]
- Berk, J.; Murphy, M.; Kane, K.; Chan, P.; Rich, J.; Brinkley-Rubinstein, L. Initial SARS-CoV-2 Vaccination Uptake in a Correctional Setting: Cross-Sectional Study. JMIRx Med. 2021, 2, e30176. [Google Scholar] [CrossRef]
- Stern, M.F.; Piasecki, A.M.; Strick, L.B.; Rajeshwar, P.; Tyagi, E.; Dolovich, S.; Patel, P.R.; Fukunaga, R.; Furukawa, N.W. Willingness to Receive a COVID-19 Vaccination Among Incarcerated or Detained Persons in Correctional and Detention Facilities—Four States, September–December 2020. MMWR Morb. Mortal. Wkly. Rep. 2021, 70, 473–477. [Google Scholar] [CrossRef]
- WHO. Regional Office for Europe. In Preparedness, Prevention and Control of COVID-19 in Prisons and Other Places of Detention: Interim Guidance, 8 February 2021; World Health Organization, Regional Office for Europe: Geneva, Switzerland, 2021. [Google Scholar]
- Wilburn, J.; Blakey, E.; Trindall, A.; Burr, H.; Tanti, V.; Doolan, S.; Palmer, I.; Jewell, T.; Balakrishnan, R. COVID-19 within a Large UK Prison with a High Number of Vulnerable Adults, March to June 2020: An Outbreak Investigation and Screening Event. Int. J. Infect. Dis. 2021, 104, 349–353. [Google Scholar] [CrossRef]
- Vusirikala, A.; Flannagan, J.; Czachorowski, M.; Zaidi, A.; Twohig, K.A.; Plugge, E.; Ellaby, N.; Rice, W.; Dabrera, G.; Chudasama, D.Y.; et al. Impact of SARS-CoV-2 Alpha Variant (B.1.1.7) on Prisons, England. Public Health 2022, 204, 21–24. [Google Scholar] [CrossRef] [PubMed]
- Mazzilli, S.; Tavoschi, L.; Soria, A.; Fornili, M.; Cocca, G.; Sebastiani, T.; Scardina, G.; Cairone, C.; Arzilli, G.; Lapadula, G.; et al. COVID-19 Infection Among Incarcerated Individuals and Prison Staff in Lombardy, Italy, March 2020 to February 2021. JAMA Netw. Open 2022, 5, e224862. [Google Scholar] [CrossRef]
- Sigal, A. Milder Disease with Omicron: Is It the Virus or the Pre-Existing Immunity? Nat. Rev. Immunol. 2022, 22, 69–71. [Google Scholar] [CrossRef] [PubMed]
- Chin, E.T.; Ryckman, T.; Prince, L.; Leidner, D.; Alarid-Escudero, F.; Andrews, J.R.; Salomon, J.A.; Studdert, D.M.; Goldhaber-Fiebert, J.D. COVID-19 in the California State Prison System: An Observational Study of Decarceration, Ongoing Risks, and Risk Factors. J. Gen. Intern. Med. 2021, 36, 3096–3102. [Google Scholar] [CrossRef] [PubMed]
- Brinkley-Rubinstein, L.; Peterson, M.; Martin, R.; Chan, P.; Berk, J. Breakthrough SARS-CoV-2 Infections in Prison after Vaccination. N. Engl. J. Med. 2021, 385, 1051–1052. [Google Scholar] [CrossRef]
- Muik, A.; Lui, B.G.; Wallisch, A.-K.; Bacher, M.; Mühl, J.; Reinholz, J.; Ozhelvaci, O.; Beckmann, N.; Garcia, R.d.l.C.G.; Poran, A.; et al. Neutralization of SARS-CoV-2 Omicron by BNT162b2 MRNA Vaccine–Elicited Human Sera. Science 2022, 375, 678–680. [Google Scholar] [CrossRef]
- Cele, S.; Jackson, L.; Khoury, D.S.; Khan, K.; Moyo-Gwete, T.; Tegally, H.; San, J.E.; Cromer, D.; Scheepers, C.; Amoako, D.G.; et al. Omicron Extensively but Incompletely Escapes Pfizer BNT162b2 Neutralization. Nature 2022, 602, 654–656. [Google Scholar] [CrossRef]
- Andrews, N.; Stowe, J.; Kirsebom, F.; Toffa, S.; Rickeard, T.; Gallagher, E.; Gower, C.; Kall, M.; Groves, N.; O’Connell, A.-M.; et al. COVID-19 Vaccine Effectiveness against the Omicron (B.1.1.529) Variant. N. Engl. J. Med. 2022, 386, 1532–1546. [Google Scholar] [CrossRef]
- Abu-Raddad, L.J.; Chemaitelly, H.; Ayoub, H.H.; AlMukdad, S.; Yassine, H.M.; Al-Khatib, H.A.; Smatti, M.K.; Tang, P.; Hasan, M.R.; Coyle, P.; et al. Effect of MRNA Vaccine Boosters against SARS-CoV-2 Omicron Infection in Qatar. N. Engl. J. Med. 2022, 386, 1804–1816. [Google Scholar] [CrossRef]
- Bergwerk, M.; Gonen, T.; Lustig, Y.; Amit, S.; Lipsitch, M.; Cohen, C.; Mandelboim, M.; Levin, E.G.; Rubin, C.; Indenbaum, V.; et al. COVID-19 Breakthrough Infections in Vaccinated Health Care Workers. N. Engl. J. Med. 2021, 385, 1474–1484. [Google Scholar] [CrossRef]
- Earle, K.A.; Ambrosino, D.M.; Fiore-Gartland, A.; Goldblatt, D.; Gilbert, P.B.; Siber, G.R.; Dull, P.; Plotkin, S.A. Evidence for Antibody as a Protective Correlate for COVID-19 Vaccines. Vaccine 2021, 39, 4423–4428. [Google Scholar] [CrossRef] [PubMed]
- Garcia-Beltran, W.F.; St Denis, K.J.; Hoelzemer, A.; Lam, E.C.; Nitido, A.D.; Sheehan, M.L.; Berrios, C.; Ofoman, O.; Chang, C.C.; Hauser, B.M.; et al. MRNA-Based COVID-19 Vaccine Boosters Induce Neutralizing Immunity against SARS-CoV-2 Omicron Variant. Cell 2022, 185, 457–466.e4. [Google Scholar] [CrossRef] [PubMed]
- Naranbhai, V.; Nathan, A.; Kaseke, C.; Berrios, C.; Khatri, A.; Choi, S.; Getz, M.A.; Tano-Menka, R.; Ofoman, O.; Gayton, A.; et al. T Cell Reactivity to the SARS-CoV-2 Omicron Variant Is Preserved in Most but Not All Individuals. Cell 2022, 185, 1041–1051.e6. [Google Scholar] [CrossRef] [PubMed]
- Lustig, Y.; Zuckerman, N.; Nemet, I.; Atari, N.; Kliker, L.; Regev-Yochay, G.; Sapir, E.; Mor, O.; Alroy-Preis, S.; Mendelson, E.; et al. Neutralising Capacity against Delta (B.1.617.2) and Other Variants of Concern Following Comirnaty (BNT162b2, BioNTech/Pfizer) Vaccination in Health Care Workers, Israel. Eurosurveillance 2021, 26, 2100557. [Google Scholar] [CrossRef]
- Hyseni, I.; Molesti, E.; Benincasa, L.; Piu, P.; Casa, E.; Temperton, N.J.; Manenti, A.; Montomoli, E. Characterisation of SARS-CoV-2 LentiviralPseudotypes and Correlation between Pseudotype-Based Neutralisation Assays and Live Virus-Based Micro Neutralisation Assays. Viruses 2020, 12, 1011. [Google Scholar] [CrossRef]
- Pannus, P.; Neven, K.Y.; De Craeye, S.; Heyndrickx, L.; VandeKerckhove, S.; Georges, D.; Michiels, J.; Francotte, A.; Van Den Bulcke, M.; Zrein, M.; et al. Poor Antibody Response to BioNTech/Pfizer Coronavirus Disease 2019 Vaccination in Severe Acute Respiratory Syndrome Coronavirus 2–Naive Residents of Nursing Homes. Clin. Infect. Dis. 2021, ciab998. [Google Scholar] [CrossRef]
- Makris, E.; Gourgoulianis, K.I.; Hatzoglou, C. Prisoners and Cigarettes or ‘Imprisoned in Cigarettes’? What Helps Prisoners Quit Smoking? BMC Public Health 2012, 12, 508. [Google Scholar] [CrossRef] [Green Version]
- Sahin, U.; Muik, A.; Vogler, I.; Derhovanessian, E.; Kranz, L.M.; Vormehr, M.; Quandt, J.; Bidmon, N.; Ulges, A.; Baum, A.; et al. BNT162b2 Vaccine Induces Neutralizing Antibodies and Poly-Specific T Cells in Humans. Nature 2021, 595, 572–577. [Google Scholar] [CrossRef]
- Servellita, V.; Syed, A.M.; Morris, M.K.; Brazer, N.; Saldhi, P.; Garcia-Knight, M.; Sreekumar, B.; Khalid, M.M.; Ciling, A.; Chen, P.-Y.; et al. Neutralizing Immunity in Vaccine Breakthrough Infections from the SARS-CoV-2 Omicron and Delta Variants. Cell 2022, 185, 1539–1548.e5. [Google Scholar] [CrossRef]
- Kustin, T.; Harel, N.; Finkel, U.; Perchik, S.; Harari, S.; Tahor, M.; Caspi, I.; Levy, R.; Leshchinsky, M.; Ken Dror, S.; et al. Evidence for Increased Breakthrough Rates of SARS-CoV-2 Variants of Concern in BNT162b2-MRNA-Vaccinated Individuals. Nat. Med. 2021, 27, 1379–1384. [Google Scholar] [CrossRef]
- CDC. COVID-19 and Your Health. Available online: https://www.cdc.gov/coronavirus/2019-ncov/need-extra-precautions/living-prisons-jails.html (accessed on 26 May 2022).
- Barsky, B.A.; Reinhart, E.; Farmer, P.; Keshavjee, S. Vaccination plus Decarceration—Stopping COVID-19 in Jails and Prisons. N. Engl. J. Med. 2021, 384, 1583–1585. [Google Scholar] [CrossRef] [PubMed]
- Parsons, T.L.; Worden, L. Assessing the Risk of Cascading COVID-19 Outbreaks from Prison-to-Prison Transfers. Epidemics 2021, 37, 100532. [Google Scholar] [CrossRef] [PubMed]
| Inmates (N. 595) | |||||
|---|---|---|---|---|---|
| Characteristics | Unvaccinated (15.8%) | Partially Vaccinated (7.9%) | Fully Vaccinated (27.4%) | Boostered (48.9%) | Total (100.0%) |
| Age groups (years) a | |||||
| 18–29 | 4.1 | 1.6 | 5.5 | 6.1 | 17.3 |
| 30–39 | 5.1 | 1.2 | 10.7 | 13.0 | 30.0 |
| 40–49 | 4.3 | 3.1 | 5.5 | 13.3 | 26.2 |
| 50–59 | 1.0 | 1.5 | 3.9 | 9.5 | 16.1 |
| ≥60 | 1.3 | 0.5 | 1.6 | 7.0 | 10.4 |
| Days since the last vaccine dose a | |||||
| ≤14 | - | 0.8 | 2.0 | 13.2 | 16.0 |
| 15–40 | - | 3.6 | 4.5 | 25.1 | 33.4 |
| 41–89 | - | 0.8 | 8.7 | 8.7 | 18.3 |
| ≥90 | - | 2.6 | 12.2 | 1.6 | 16 |
| Characteristics | No. of Cases (Population) | Attack Rate % | RR (CI 95%) |
|---|---|---|---|
| Vaccination status * | |||
| -Unvaccinated a | 59 (94) | 62.7 | 1.8 (1.5–2.3) |
| Partially vaccinated | 23 (42) | 52.3 | 1.5 (1.0–1.9) |
| Fully vaccinated | 68 (151) | 45.0 | 1.3 (1.0–1.6) |
| Boostered a | 68 (213) | 31.4 | 0.7 (0.6–0.9) |
| Total | 218 (500) | 43.6 | 0.8 (0.2–3.0) |
| Days since the last vaccine dose | |||
| 15–40 a | 57 (199) | 28.6 | 0.6 (0.5–0.8) |
| 41–90 | 50 (109) | 45.8 | 1.2 (1.0–1.6) |
| >90 | 52 (99) | 52.5 | 1.5 (1.2–1.8) |
| Age groups (years) | |||
| 18–29 | 40 (103) | 38.9 | 1.0 (0.7–1.3) |
| 30–39 | 74 (178) | 41.5 | 1.1 (0.9–1.4) |
| 40–49 | 62 (156) | 39.7 | 1.1 (0.8–1.3) |
| 50–59 | 27 (96) | 28.0 | 0.7 (0.5–0.9) |
| ≥60 | 22 (62) | 35.4 | 0.9 (0.6–1.3) |
| Neutralizing Antibodies | T1 (N.101) GMT | T2 (N.101) GMT a |
|---|---|---|
| Anti-wild type | 6.1 | 53.1 |
| Anti-Omicron | 5.0 | 5.5 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Stufano, A.; Buonvino, N.; Trombetta, C.M.; Pontrelli, D.; Marchi, S.; Lobefaro, G.; De Benedictis, L.; Lorusso, E.; Carofiglio, M.T.; Vasinioti, V.I.; et al. COVID-19 Outbreak and BNT162b2 mRNA Vaccination Coverage in a Correctional Facility during Circulation of the SARS-CoV-2 Omicron BA.1 Variant in Italy. Vaccines 2022, 10, 1137. https://doi.org/10.3390/vaccines10071137
Stufano A, Buonvino N, Trombetta CM, Pontrelli D, Marchi S, Lobefaro G, De Benedictis L, Lorusso E, Carofiglio MT, Vasinioti VI, et al. COVID-19 Outbreak and BNT162b2 mRNA Vaccination Coverage in a Correctional Facility during Circulation of the SARS-CoV-2 Omicron BA.1 Variant in Italy. Vaccines. 2022; 10(7):1137. https://doi.org/10.3390/vaccines10071137
Chicago/Turabian StyleStufano, Angela, Nicola Buonvino, Claudia Maria Trombetta, Daniela Pontrelli, Serena Marchi, Giuseppe Lobefaro, Leonarda De Benedictis, Eleonora Lorusso, Maria Teresa Carofiglio, Violetta Iris Vasinioti, and et al. 2022. "COVID-19 Outbreak and BNT162b2 mRNA Vaccination Coverage in a Correctional Facility during Circulation of the SARS-CoV-2 Omicron BA.1 Variant in Italy" Vaccines 10, no. 7: 1137. https://doi.org/10.3390/vaccines10071137
APA StyleStufano, A., Buonvino, N., Trombetta, C. M., Pontrelli, D., Marchi, S., Lobefaro, G., De Benedictis, L., Lorusso, E., Carofiglio, M. T., Vasinioti, V. I., Montomoli, E., Decaro, N., & Lovreglio, P. (2022). COVID-19 Outbreak and BNT162b2 mRNA Vaccination Coverage in a Correctional Facility during Circulation of the SARS-CoV-2 Omicron BA.1 Variant in Italy. Vaccines, 10(7), 1137. https://doi.org/10.3390/vaccines10071137







