COVID-19 Vaccine Acceptance among Low- and Lower-Middle-Income Countries: A Rapid Systematic Review and Meta-Analysis
Abstract
:1. Introduction
2. Materials and Methods
2.1. Search Strategy
2.2. Study Selection
2.3. Data Extraction
2.4. Assessment of Study Quality
2.5. Data Analysis
3. Results
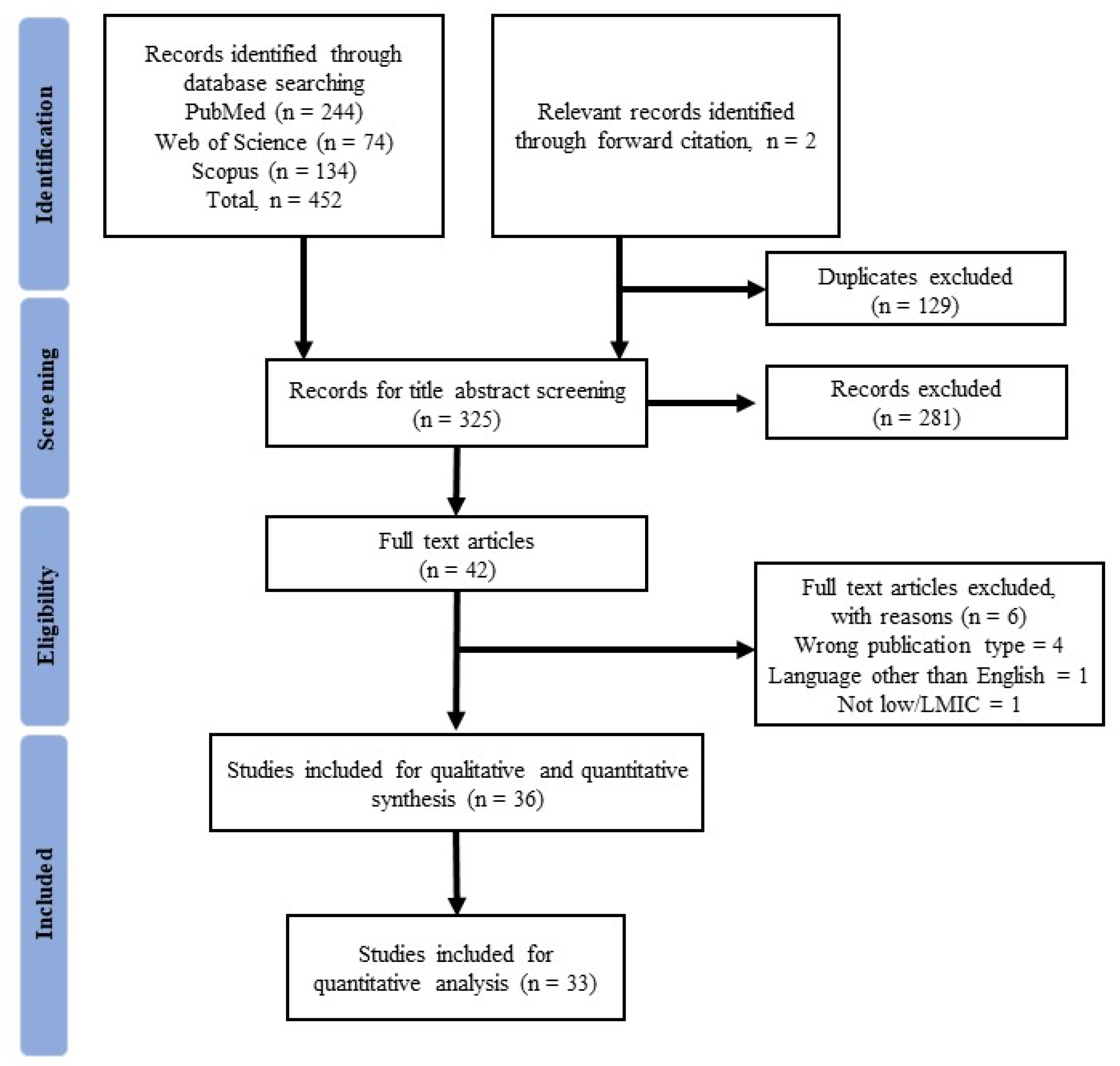
3.1. Search Results
3.2. Characteristics of the Included Studies
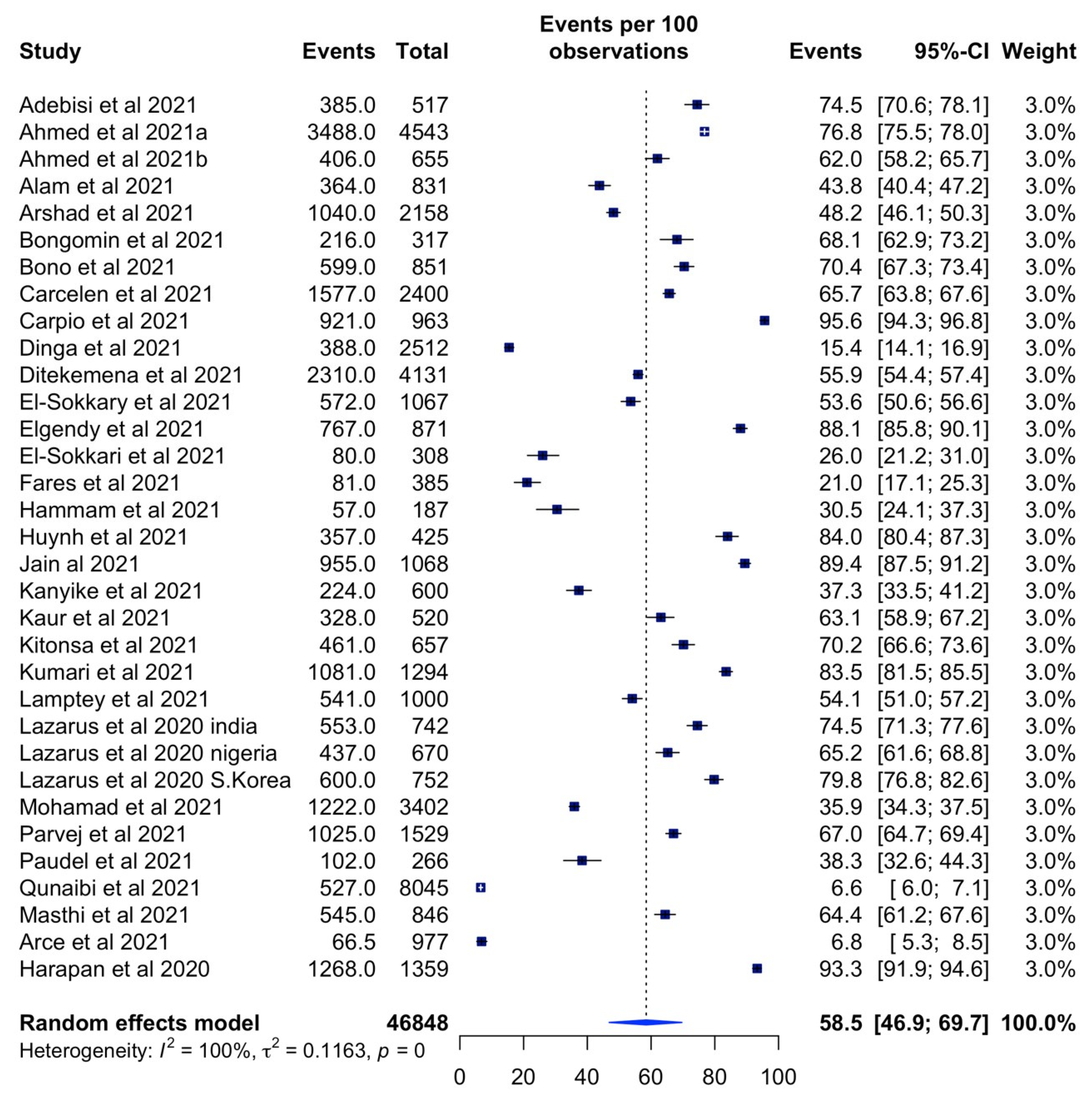
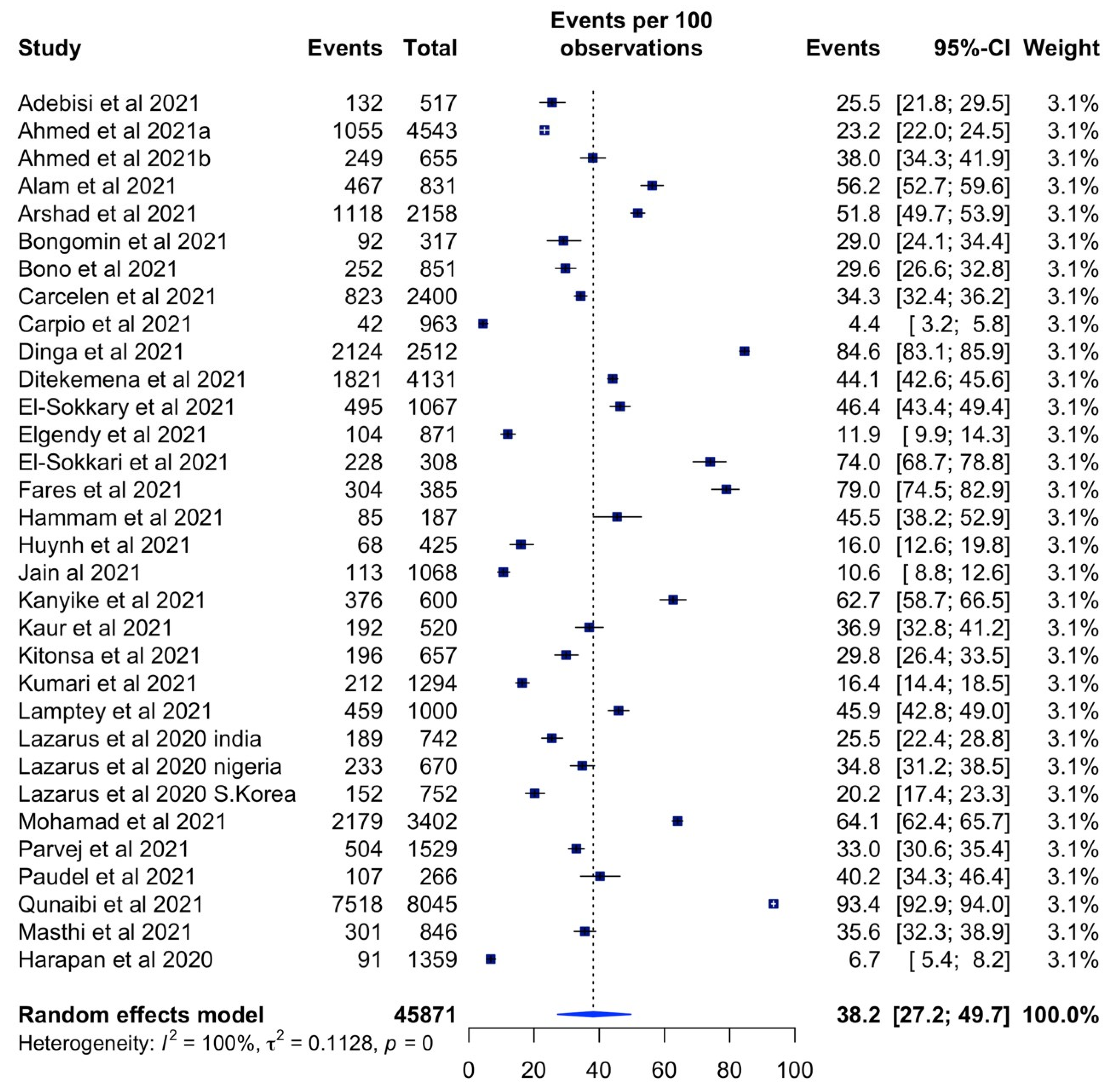
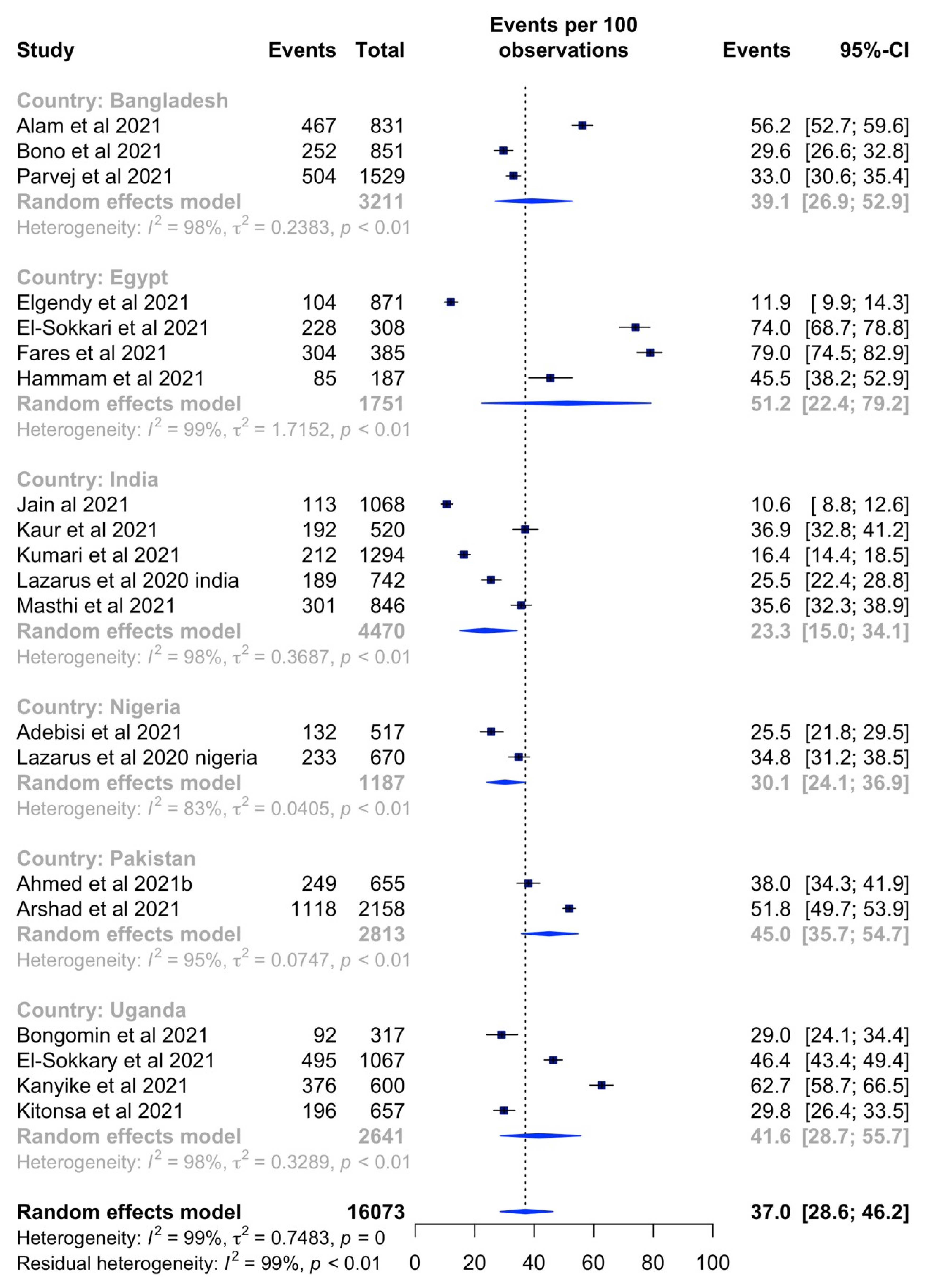
3.3. Prevalence of Vaccine Acceptance and Hesitancy
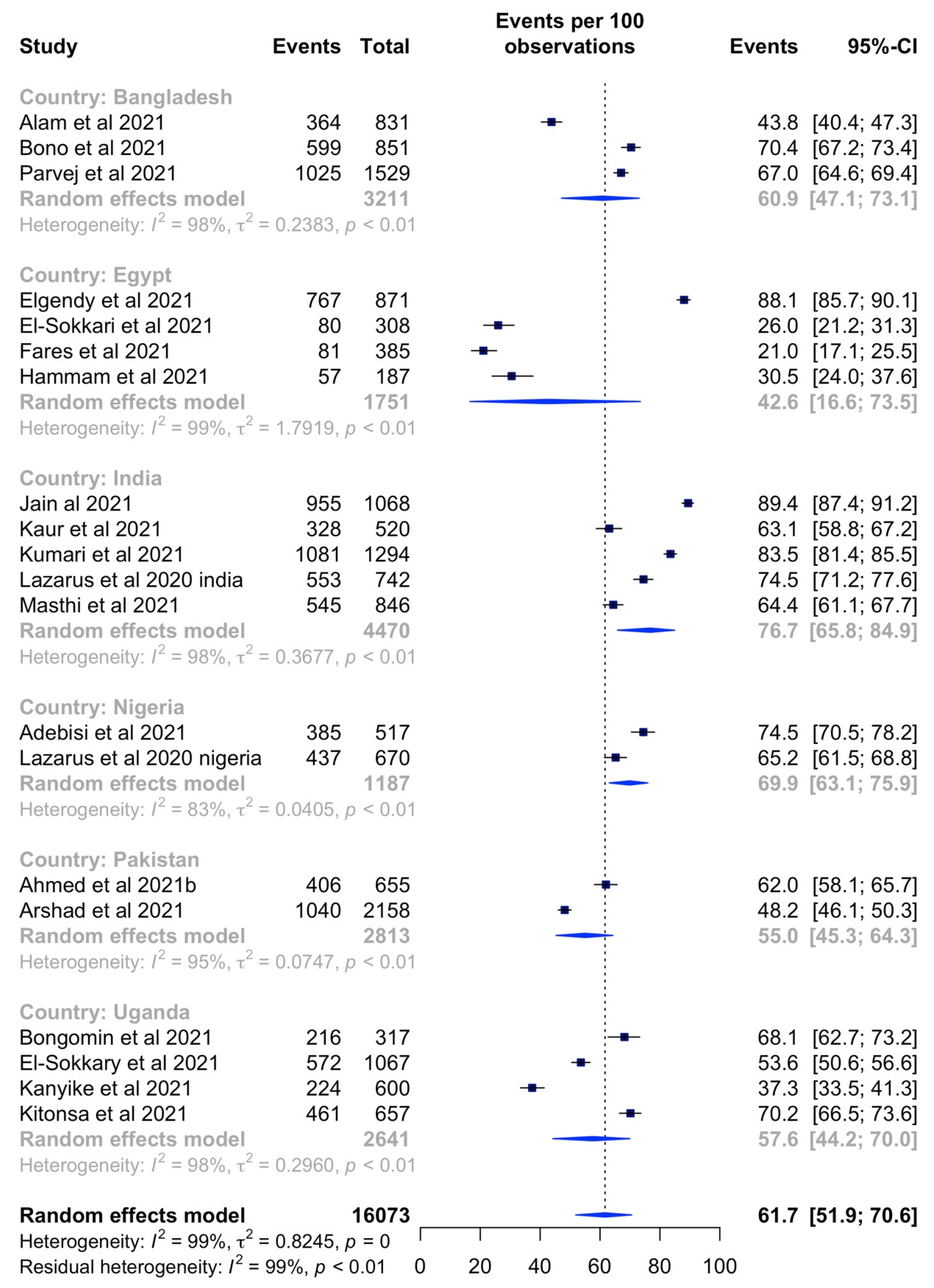
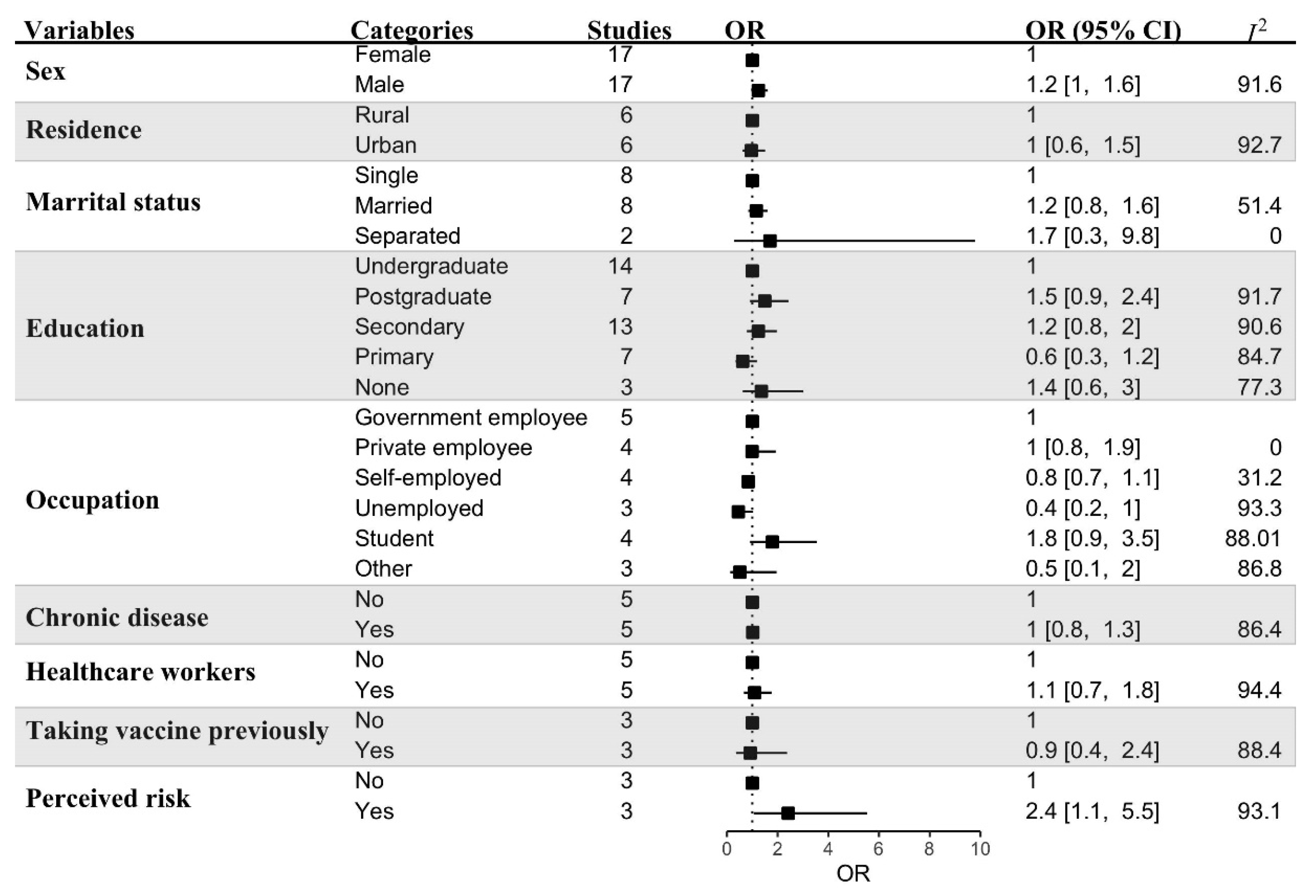
3.4. Sub-Group Analyses
3.5. Risk of Bias
4. Discussion
4.1. Implications and Future Research Needs
4.2. Strengths and Limitations
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Grochowska, M.; Ratajczak, A.; Zdunek, G.; Adamiec, A.; Waszkiewicz, P.; Feleszko, W. A comparison of the level of acceptance and hesitancy towards the influenza vaccine and the forthcoming COVID-19 vaccine in the medical community. Vaccines 2021, 9, 475. [Google Scholar] [CrossRef] [PubMed]
- Worldometer COVID Live—Coronavirus Statistics—Worldometer. Available online: https://www.worldometers.info/coronavirus/ (accessed on 5 February 2022).
- Machingaidze, S.; Wiysonge, C.S. Understanding COVID-19 vaccine hesitancy. Nat. Med. 2021, 27, 1338–1339. [Google Scholar] [CrossRef] [PubMed]
- Eroglu, B.; Nuwarda, R.F.; Ramzan, I.; Kayser, V. A Narrative Review of COVID-19 Vaccines. Vaccines 2022, 10, 62. [Google Scholar] [CrossRef] [PubMed]
- Wang, Q.; Yang, L.; Jin, H.; Lin, L. Vaccination against COVID-19: A systematic review and meta-analysis of acceptability and its predictors. Prev. Med. 2021, 150, 106694. [Google Scholar] [CrossRef] [PubMed]
- Dubé, E.; Bettinger, J.; Fisher, W.; Naus, M.; Mahmud, S.; Hilderman, T. Improving Vaccination Rates: Vaccine acceptance, hesitancy and refusal in Canada: Challenges and potential approaches. Canada Commun. Dis. Rep. 2016, 42, 246. [Google Scholar] [CrossRef] [PubMed]
- Li, M.; Luo, Y.; Watson, R.; Zheng, Y.; Ren, J.; Tang, J.; Chen, Y. Healthcare workers’ (HCWs) attitudes and related factors towards COVID-19 vaccination: A rapid systematic review. Postgrad. Med. J. 2021. [Google Scholar] [CrossRef] [PubMed]
- Harapan, H.; Wagner, A.L.; Yufika, A.; Winardi, W.; Anwar, S.; Gan, A.K.; Setiawan, A.M.; Rajamoorthy, Y.; Sofyan, H.; Mudatsir, M. Acceptance of a COVID-19 Vaccine in Southeast Asia: A Cross-Sectional Study in Indonesia. Front. Public Health 2020, 8, 381. [Google Scholar] [CrossRef] [PubMed]
- Ayhan, S.G.; Oluklu, D.; Atalay, A.; Beser, D.M.; Tanacan, A.; Tekin, O.M.; Sahin, D. COVID-19 vaccine acceptance in pregnant women. Int. J. Gynecol. Obstet. 2021, 154, 291–296. [Google Scholar] [CrossRef]
- Charron, J.; Gautier, A.; Jestin, C. Influence of information sources on vaccine hesitancy and practices. Médecine Mal. Infect. 2020, 50, 727–733. [Google Scholar] [CrossRef] [PubMed]
- Feemster, K.A. Building vaccine acceptance through communication and advocacy. Hum. Vaccin. Immunother. 2020, 16, 1004–1006. [Google Scholar] [CrossRef]
- MacDonald, N.E.; Butler, R.; Dubé, E. Addressing barriers to vaccine acceptance: An overview. Hum. Vaccin. Immunother. 2017, 14, 218–224. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sallam, M. COVID-19 vaccine hesitancy worldwide: A concise systematic review of vaccine acceptance rates. Vaccines 2021, 9, 160. [Google Scholar] [CrossRef] [PubMed]
- Dubé, È.; Farrands, A.; Lemaitre, T.; Boulianne, N.; Sauvageau, C.; Boucher, F.D.; Tapiero, B.; Quach, C.; Ouakki, M.; Gosselin, V.; et al. Overview of knowledge, attitudes, beliefs, vaccine hesitancy and vaccine acceptance among mothers of infants in Quebec, Canada. Hum. Vaccines Immunother. 2019, 15, 113–120. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Solís Arce, J.S.; Warren, S.S.; Meriggi, N.F.; Scacco, A.; McMurry, N.; Voors, M.; Syunyaev, G.; Malik, A.A.; Aboutajdine, S.; Adeojo, O.; et al. COVID-19 vaccine acceptance and hesitancy in low- and middle-income countries. Nat. Med. 2021, 27, 1385–1394. [Google Scholar] [CrossRef] [PubMed]
- Patwary, M.M.; Bardhan, M.; Disha, A.S.; Hasan, M.; Haque, M.Z.; Sultana, R.; Hossain, M.R.; Browning, M.H.E.M.; Alam, M.A.; Sallam, M. Determinants of COVID-19 Vaccine Acceptance among the Adult Population of Bangladesh Using the Health Belief Model and the Theory of Planned Behavior Model. Vaccines 2021, 9, 1393. [Google Scholar] [CrossRef] [PubMed]
- Akiful Haque, M.M.; Rahman, M.L.; Hossian, M.; Matin, K.F.; Nabi, M.H.; Saha, S.; Hasan, M.; Manna, R.M.; Barsha, S.Y.; Hasan, S.M.R.; et al. Acceptance of COVID-19 vaccine and its determinants: Evidence from a large sample study in Bangladesh. Heliyon 2021, 7, e07376. [Google Scholar] [CrossRef] [PubMed]
- Alam, A.B.M.M.; Majumder, M.A.A.; Haque, M.; Ashraf, F.; Khondoker, M.U.; Mashreky, S.R.; Wahab, A.; Siddiqui, T.H.; Uddin, A.; Joarder, T.; et al. Disproportionate COVID-19 vaccine acceptance rate among healthcare professionals on the eve of nationwide vaccine distribution in Bangladesh. Expert Rev. Vaccines 2021, 20, 1167–1175. [Google Scholar] [CrossRef] [PubMed]
- Arshad, M.S.; Hussain, I.; Mahmood, T.; Hayat, K.; Majeed, A.; Imran, I.; Saeed, H.; Iqbal, M.O.; Uzair, M.; Rehman, A.U.; et al. A National Survey to Assess the COVID-19 Vaccine-Related Conspiracy Beliefs, Acceptability, Preference, and Willingness to Pay among the General Population of Pakistan. Vaccines 2021, 9, 720. [Google Scholar] [CrossRef] [PubMed]
- Ahmed, T.F.; Ahmed, A.; Ahmed, S.; Ahmed, H.U. Understanding COVID-19 vaccine acceptance in Pakistan: An echo of previous immunizations or prospect of change? Expert Rev. Vaccines 2021, 20, 1185–1193. [Google Scholar] [CrossRef] [PubMed]
- Dinga, J.N.; Sinda, L.K.; Titanji, V.P.K. Assessment of vaccine hesitancy to a COVID-19 vaccine in Cameroonian adults and its global implication. Vaccines 2021, 9, 175. [Google Scholar] [CrossRef]
- Fares, S.; Elmnyer, M.M.; Mohamed, S.S.; Elsayed, R. COVID-19 Vaccination Perception and Attitude among Healthcare Workers in Egypt. J. Prim. Care Community Health 2021, 12, 21501327211013304. [Google Scholar] [CrossRef] [PubMed]
- Nehal, K.R.; Steendam, L.M.; Ponce, M.C.; van der Hoeven, M.; Smit, G.S.A. Worldwide vaccination willingness for COVID-19: A systematic review and meta-analysis. Vaccines 2021, 9, 1071. [Google Scholar] [CrossRef]
- Moola, S.; Gudi, N.; Nambiar, D.; Dumka, N.; Ahmed, T.; Sonawane, I.R.; Kotwal, A. A rapid review of evidence on the determinants of and strategies for COVID-19 vaccine acceptance in low- and middle-income countries. J. Glob. Health 2021, 11, 05027. [Google Scholar] [CrossRef] [PubMed]
- Aw, J.; Seng, J.J.B.; Seah, S.S.Y.; Low, L.L. COVID-19 vaccine hesitancy—A scoping review of literature in high-income countries. Vaccines 2021, 9, 900. [Google Scholar] [CrossRef] [PubMed]
- Biswas, M.R.; Alzubaidi, M.S.; Shah, U.; Abd-Alrazaq, A.A.; Shah, Z. A scoping review to find out worldwide COVID-19 vaccine hesitancy and its underlying determinants. Vaccines 2021, 9, 1243. [Google Scholar] [CrossRef] [PubMed]
- Haby, M.M.; Chapman, E.; Clark, R.; Barreto, J.; Reveiz, L.; Lavis, J.N. What are the best methodologies for rapid reviews of the research evidence for evidence-informed decision making in health policy and practice: A rapid review. Health Res. Policy Syst. 2016, 14, 83. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Page, M.J.; McKenzie, J.E.; Bossuyt, P.M.; Boutron, I.; Hoffmann, T.C.; Mulrow, C.D.; Shamseer, L.; Tetzlaff, J.M.; Akl, E.A.; Brennan, S.E.; et al. The PRISMA 2020 statement: An updated guideline for reporting systematic reviews. BMJ 2021, 372, 71. [Google Scholar] [CrossRef] [PubMed]
- Adebisi, Y.A.; Alaran, A.J.; Bolarinwa, O.A.; Akande-Sholabi, W.; Lucero-Prisno, D.E. When it is available, will we take it? Social media users’ perception of hypothetical COVID-19 vaccine in Nigeria. Pan Afr. Med. J. 2021, 38, 230. [Google Scholar] [CrossRef] [PubMed]
- Ahmed, M.A.M.; Colebunders, R.; Gele, A.A.; Farah, A.A.; Osman, S.; Guled, I.A.; Abdullahi, A.A.M.; Hussein, A.M.; Ali, A.M.; Siewe Fodjo, J.N. COVID-19 Vaccine Acceptability and Adherence to Preventive Measures in Somalia: Results of an Online Survey. Vaccines 2021, 9, 543. [Google Scholar] [CrossRef]
- Bongomin, F.; Olum, R.; Andia-Biraro, I.; Nakwagala, F.N.; Hassan, K.H.; Nassozi, D.R.; Kaddumukasa, M.; Byakika-Kibwika, P.; Kiguli, S.; Kirenga, B.J. COVID-19 vaccine acceptance among high-risk populations in Uganda. Ther. Adv. Infect. Dis. 2021, 8, 20499361211024376. [Google Scholar] [CrossRef]
- Bono, S.A.; Faria de Moura Villela, E.; Siau, C.S.; Chen, W.S.; Pengpid, S.; Hasan, M.T.; Sessou, P.; Ditekemena, J.D.; Amodan, B.O.; Hosseinipour, M.C.; et al. Factors Affecting COVID-19 Vaccine Acceptance: An International Survey among Low- and Middle-Income Countries. Vaccines 2021, 9, 515. [Google Scholar] [CrossRef] [PubMed]
- Carcelen, A.C.; Prosperi, C.; Mutembo, S.; Chongwe, G.; Mwansa, F.D.; Ndubani, P.; Simulundu, E.; Chilumba, I.; Musukwa, G.; Thuma, P.; et al. COVID-19 vaccine hesitancy in Zambia: A glimpse at the possible challenges ahead for COVID-19 vaccination rollout in sub-Saharan Africa. Hum. Vaccin. Immunother. 2022, 18, 1–6. [Google Scholar] [CrossRef] [PubMed]
- Carpio, C.E.; Sarasty, O.; Hudson, D.; Macharia, A.; Shibia, M. The demand for a COVID-19 vaccine in Kenya. Hum. Vaccin. Immunother. 2021, 17, 3463–3471. [Google Scholar] [CrossRef] [PubMed]
- Echoru, I.; Ajambo, P.D.; Keirania, E.; Bukenya, E.E.M. Sociodemographic factors associated with acceptance of COVID-19 vaccine and clinical trials in Uganda: A cross-sectional study in western Uganda. BMC Public Health 2021, 21, 1106. [Google Scholar] [CrossRef] [PubMed]
- Elgendy, M.O.; Abdelrahim, M.E.A. Public awareness about coronavirus vaccine, vaccine acceptance, and hesitancy. J. Med. Virol. 2021, 93, 6535–6543. [Google Scholar] [CrossRef] [PubMed]
- El-Sokkary, R.H.; El Seifi, O.S.; Hassan, H.M.; Mortada, E.M.; Hashem, M.K.; Gadelrab, M.R.M.A.; Tash, R.M.E. Predictors of COVID-19 vaccine hesitancy among Egyptian healthcare workers: A cross-sectional study. BMC Infect. Dis. 2021, 21, 762. [Google Scholar] [CrossRef] [PubMed]
- Hammam, N.; Tharwat, S.; Shereef, R.R.E.; Elsaman, A.M.; Khalil, N.M.; Fathi, H.M.; Salem, M.N.; El-Saadany, H.M.; Samy, N.; El-Bahnasawy, A.S.; et al. Rheumatology university faculty opinion on coronavirus disease-19 (COVID-19) vaccines: The vaXurvey study from Egypt. Rheumatol. Int. 2021, 41, 1607–1616. [Google Scholar] [CrossRef] [PubMed]
- Huynh, G.; Van Nguyen, T.; Nguyen, D.D.; Lam, Q.M.; Pham, T.N.; Nguyen, H.T.N. Knowledge About COVID-19, Beliefs and Vaccination Acceptance Against COVID-19 Among High-Risk People in Ho Chi Minh City, Vietnam. Infect. Drug Resist. 2021, 14, 1773–1780. [Google Scholar] [CrossRef]
- Jain, J.; Saurabh, S.; Kumar, P.; Verma, M.K.; Goel, A.D.; Gupta, M.K.; Bhardwaj, P.; Raghav, P.R. COVID-19 vaccine hesitancy among medical students in India. Epidemiol. Infect. 2021, 149, e132. [Google Scholar] [CrossRef]
- Kanyike, A.M.; Olum, R.; Kajjimu, J.; Ojilong, D.; Akech, G.M.; Nassozi, D.R.; Agira, D.; Wamala, N.K.; Asiimwe, A.; Matovu, D.; et al. Acceptance of the coronavirus disease-2019 vaccine among medical students in Uganda. Trop. Med. Health 2021, 49, 37. [Google Scholar] [CrossRef] [PubMed]
- Kaur, A.; Kaur, G.; Kashyap, A.; Singh, G.; Singh Sandhu, H.; Khilji, I.; Singh Gambhir, R. Attitude and acceptance of COVID-19 vaccine amongst medical and dental fraternity—A questionnaire survey. Rocz. Panstw. Zakl. Hig. 2021, 72, 185–191. [Google Scholar] [CrossRef] [PubMed]
- Kitonsa, J.; Kamacooko, O.; Bahemuka, U.M.; Kibengo, F.; Kakande, A.; Wajja, A.; Basajja, V.; Lumala, A.; Ssemwanga, E.; Asaba, R.; et al. Willingness to participate in COVID-19 vaccine trials; a survey among a population of healthcare workers in Uganda. PLoS ONE 2021, 16, e0251992. [Google Scholar] [CrossRef] [PubMed]
- Kumari, A.; Ranjan, P.; Chopra, S.; Kaur, D.; Kaur, T.; Upadhyay, A.D.; Isaac, J.A.; Kasiraj, R.; Prakash, B.; Kumar, P.; et al. Knowledge, barriers and facilitators regarding COVID-19 vaccine and vaccination programme among the general population: A cross-sectional survey from one thousand two hundred and forty-nine participants. Diabetes Metab. Syndr. 2021, 15, 987–992. [Google Scholar] [CrossRef] [PubMed]
- Lamptey, E.; Serwaa, D.; Appiah, A.B. A nationwide survey of the potential acceptance and determinants of COVID-19 vaccines in Ghana. Clin. Exp. Vaccine Res. 2021, 10, 183–190. [Google Scholar] [CrossRef] [PubMed]
- Lazarus, J.V.; Ratzan, S.C.; Palayew, A.; Gostin, L.O.; Larson, H.J.; Rabin, K.; Kimball, S.; El-Mohandes, A. A global survey of potential acceptance of a COVID-19 vaccine. Nat. Med. 2021, 27, 225–228. [Google Scholar] [CrossRef] [PubMed]
- Lazarus, J.V.; Wyka, K.; Rauh, L.; Rabin, K.; Ratzan, S.; Gostin, L.O.; Larson, H.J.; El-Mohandes, A. Hesitant or Not? The Association of Age, Gender, and Education with Potential Acceptance of a COVID-19 Vaccine: A Country-level Analysis. J. Health Commun. 2020, 25, 799–807. [Google Scholar] [CrossRef] [PubMed]
- Mohamad, O.; Zamlout, A.; AlKhoury, N.; Mazloum, A.A.; Alsalkini, M.; Shaaban, R. Factors associated with the intention of Syrian adult population to accept COVID19 vaccination: A cross-sectional study. BMC Public Health 2021, 21, 1310. [Google Scholar] [CrossRef] [PubMed]
- Panda, D.S.; Giri, R.K.; Nagarajappa, A.K.; Basha, S. COVID-19 vaccine, acceptance, and concern of safety from public perspective in the state of Odisha, India. Hum. Vaccin. Immunother. 2021, 17, 3333–3337. [Google Scholar] [CrossRef]
- Parvej, M.I.; Sultana, S.; Tabassum, M.; Mannan, S.E.; Ahmed, F. Determinants of COVID-19 vaccine acceptance and encountered side-effects among the vaccinated in Bangladesh. Asian Pac. J. Trop. Med. 2021, 14, 341. [Google Scholar] [CrossRef]
- Paudel, S.; Palaian, S.; Shankar, P.R.; Subedi, N. Risk Perception and Hesitancy Toward COVID-19 Vaccination Among Healthcare Workers and Staff at a Medical College in Nepal. Risk Manag. Healthc. Policy 2021, 14, 2253–2261. [Google Scholar] [CrossRef]
- Qunaibi, E.A.; Helmy, M.; Basheti, I.; Sultan, I. A high rate of COVID-19 vaccine hesitancy in a large-scale survey on Arabs. Elife 2021, 10, e68038. [Google Scholar] [CrossRef] [PubMed]
- Ramesh Masthi, N.R.; Sowmyashree, U. Awareness of COVID 19 vaccine in a rural area near Bangalore, Karnataka. Natl. J. Community Med. 2021, 12, 72–75. [Google Scholar] [CrossRef]
- Saied, S.M.; Saied, E.M.; Kabbash, I.A.; Abdo, S.A.E.-F. Vaccine hesitancy: Beliefs and barriers associated with COVID-19 vaccination among Egyptian medical students. J. Med. Virol. 2021, 93, 4280–4291. [Google Scholar] [CrossRef] [PubMed]
- Skjefte, M.; Ngirbabul, M.; Akeju, O.; Escudero, D.; Hernandez-Diaz, S.; Wyszynski, D.F.; Wu, J.W. COVID-19 vaccine acceptance among pregnant women and mothers of young children: Results of a survey in 16 countries. Eur. J. Epidemiol. 2021, 36, 197–211. [Google Scholar] [CrossRef] [PubMed]
- Joanna Briggs Institute. Checklist for Analytical Cross Sectional Studies, Critical Appraisal Tools. Joanna Briggs Inst. 2017. Available online: https://jbi.global/critical-appraisal-tools (accessed on 15 January 2022).
- Al-Amer, R.; Maneze, D.; Everett, B.; Montayre, J.; Villarosa, A.R.; Dwekat, E.; Salamonson, Y. COVID-19 vaccination intention in the first year of the pandemic: A systematic review. J. Clin. Nurs. 2021, 31, 62–86. [Google Scholar] [CrossRef] [PubMed]
- Villarosa, A.R.; Maneze, D.; Ramjan, L.M.; Srinivas, R.; Camilleri, M.; George, A. The effectiveness of guideline implementation strategies in the dental setting: A systematic review. Implement. Sci. 2019, 14, 106. [Google Scholar] [CrossRef] [PubMed]
- Higgins, J.P.T.; Thompson, S.G. Quantifying heterogeneity in a meta-analysis. Stat. Med. 2002, 21, 1539–1558. [Google Scholar] [CrossRef]
- Patwary, M.M.; Hossain, M.R.; Shuvo, F.K.; Ashraf, S.; Sultana, R.; Alam, M.A. Protecting Sanitation Workers in Low-Middle Income Countries Amid COVID-19. Ann. Work Expo. Health 2021, 65, 492–493. [Google Scholar] [CrossRef]
- Anwar, S.; Nasrullah, M.; Hosen, M.J. COVID-19 and Bangladesh: Challenges and How to Address Them. Front. Public Health 2020, 8, 154. [Google Scholar] [CrossRef] [PubMed]
- Wouters, O.J.; Shadlen, K.C.; Salcher-Konrad, M.; Pollard, A.J.; Larson, H.J.; Teerawattananon, Y.; Jit, M. Challenges in ensuring global access to COVID-19 vaccines: Production, affordability, allocation, and deployment. Lancet 2021, 397, 1023–1034. [Google Scholar] [CrossRef]
- Maeda, J.M.; Nkengasong, J.N. The puzzle of the COVID-19 pandemic in Africa. Science 2021, 371, 27–28. [Google Scholar] [CrossRef] [PubMed]
- Christensen, D.; Dube, O.; Haushofer, J.; Siddiqi, B.; Voors, M. Building resilient health systems: Experimental evidence from sierra leone and the 2014 ebola outbreak. Q. J. Econ. 2021, 136, 1145–1198. [Google Scholar] [CrossRef]
- Barello, S.; Nania, T.; Dellafiore, F.; Graffigna, G.; Caruso, R. “Vaccine hesitancy” among university students in Italy during the COVID-19 pandemic. Eur. J. Epidemiol. 2020, 35, 781–783. [Google Scholar] [CrossRef] [PubMed]
- The Business Standard India’s Omicron Surge Explained: Reproduction Number up, Doubling Time down | Business Standard News. Available online: https://www.business-standard.com/article/current-affairs/india-s-omicron-surge-explained-reproduction-number-up-doubling-time-down-122010900082_1.html (accessed on 15 January 2022).
- Kabamba Nzaji, M.; Kabamba Ngombe, L.; Ngoie Mwamba, G.; Banza Ndala, D.B.; Mbidi Miema, J.; Luhata Lungoyo, C.; Lora Mwimba, B.; Cikomola Mwana Bene, A.; Mukamba Musenga, E. Acceptability of Vaccination Against COVID-19 Among Healthcare Workers in the Democratic Republic of the Congo. Pragmatic Obs. Res. 2020, 11, 103–109. [Google Scholar] [CrossRef] [PubMed]
- Jegede, A.S. What led to the Nigerian boycott of the polio vaccination campaign? PLoS Med. 2007, 4, e73. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Dzinamarira, T.; Nachipo, B.; Phiri, B.; Musuka, G. COVID-19 vaccine roll-out in South Africa and Zimbabwe: Urgent need to address community preparedness, fears and hesitancy. Vaccines 2021, 9, 250. [Google Scholar] [CrossRef] [PubMed]
- Goldman, R.D.; Yan, T.D.; Seiler, M.; Parra Cotanda, C.; Brown, J.C.; Klein, E.J.; Hoeffe, J.; Gelernter, R.; Hall, J.E.; Davis, A.L.; et al. Caregiver willingness to vaccinate their children against COVID-19: Cross sectional survey. Vaccine 2020, 38, 7668–7673. [Google Scholar] [CrossRef] [PubMed]
- Jiménez-García, R.; Hernández-Barrera, V.; de Andres, A.L.; Jimenez-Trujillo, I.; Esteban-Hernández, J.; Carrasco-Garrido, P. Gender influence in influenza vaccine uptake in Spain: Time trends analysis (1995–2006). Vaccine 2010, 28, 6169–6175. [Google Scholar] [CrossRef] [PubMed]
- Sallam, M.; Dababseh, D.; Yaseen, A.; Al-Haidar, A.; Taim, D.; Eid, H.; Ababneh, N.A.; Bakri, F.G.; Mahafzah, A. COVID-19 misinformation: Mere harmless delusions or much more? A knowledge and attitude cross-sectional study among the general public residing in Jordan. PLoS ONE 2020, 15, e0243264. [Google Scholar] [CrossRef] [PubMed]
- Nindrea, R.D.; Usman, E.; Katar, Y.; Sari, N.P. Acceptance of COVID-19 vaccination and correlated variables among global populations: A systematic review and meta-analysis. Clin. Epidemiol. Glob. Health 2021, 12, 100899. [Google Scholar] [CrossRef]
- Rajamoorthy, Y.; Radam, A.; Taib, N.M.; Rahim, K.A.; Wagner, A.L.; Mudatsir, M.; Munusamy, S.; Harapan, H. The relationship between perceptions and self-paid hepatitis B vaccination: A structural equation modeling approach. PLoS ONE 2018, 13, e0208402. [Google Scholar] [CrossRef]
- Sundaram, N.; Purohit, V.; Schaetti, C.; Kudale, A.; Joseph, S.; Weiss, M.G. Community awareness, use and preference for pandemic influenza vaccines in pune, India. Hum. Vaccines Immunother. 2015, 11, 2376–2388. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wong, L.P.; Alias, H.; Wong, P.-F.; Lee, H.Y.; AbuBakar, S. The use of the health belief model to assess predictors of intent to receive the COVID-19 vaccine and willingness to pay. Hum. Vaccin. Immunother. 2020, 16, 2204–2214. [Google Scholar] [CrossRef] [PubMed]
- Wu, D.C.; Jha, P.; Lam, T.; Brown, P.; Gelband, H.; Nagelkerke, N.; Birnboim, H.C.; Reid, A. Predictors of self-reported symptoms and testing for COVID-19 in Canada using a nationally representative survey. PLoS ONE 2020, 15, e0240778. [Google Scholar] [CrossRef] [PubMed]
- Johnson, N.F.; Velásquez, N.; Restrepo, N.J.; Leahy, R.; Gabriel, N.; El Oud, S.; Zheng, M.; Manrique, P.; Wuchty, S.; Lupu, Y. The online competition between pro- and anti-vaccination views. Nature 2020, 582, 230–233. [Google Scholar] [CrossRef] [PubMed]
- Daly, M.; Robinson, E. Willingness to Vaccinate against COVID-19 in the U.S.: Representative Longitudinal Evidence from April to October 2020. Am. J. Prev. Med. 2021, 60, 766–773. [Google Scholar] [CrossRef] [PubMed]
- Wright, K.B. Researching internet-based populations: Advantages and disadvantages of online survey research, online questionnaire authoring software packages, and web survey services. J. Comput. Commun. 2005, 10, JCMC1034. [Google Scholar] [CrossRef]
| SL | Author | Country | Study Design | Survey Period | Target Population | Sample Size, n | Vaccine Acceptance (%) | Factors Associated with Vaccine Acceptance |
|---|---|---|---|---|---|---|---|---|
| 1 | Adebisi et al. [29] | Nigeria | Cross-sectional | August 2020 | General population | 517 | 74.47 | Age, geopolitical location, education level. |
| 2 | Ahmed et al. [30] | Somalia | Cross-sectional | 26 December 2020–28 January 2021 | General population | 4543 | 76.78 | Female, living in Galmudug, Hirshabelle and Southwest, student, worker in the healthcare sector, adherence score, presence of flu symptoms. |
| 3 | Ahmed et al. [20] | Pakistan | Cross-sectional | April 2021 | General population | 655 | 61.98 | Older age, sometimes/not following Anti-COVID-19 SOPs, high chance of being infected, vaccination having the potential of preventing COVID-19 spread, observing the effects of the vaccine on others, knowing more about the vaccine, belief that a Muslim’s trust in God was sufficient to protect one from infection, the vaccine was prepared in a hurry without sufficient testing and could harm those with low immunity, seeing everyone else getting vaccinated, pressure from friends and family. |
| 4 | Akiful Haque et al. [17] | Bangladesh | Cross-sectional | 17 January–2 February 2021 | General population | 7357 | 65.05 | Graduates or above, age ≥ 50 years, students, monthly income ≥ 41,000 BDT, rural resident, respondents from Khulna division, family members diagnosed with COVID-19, presence of chronic disease, vaccinated in the last few years. |
| 5 | Alam et al. [18] | Bangladesh | Cross-sectional | 3–25 January 2021 | Healthcare professionals | 831 | 43.80 | Female, 18–34 age group, work in public/government institutes, nurses, not having received the flu vaccine in the previous year. |
| 6 | Arshad et al. [19] | Pakistan | Cross-sectional | January 2021 | General population | 2158 | 48.19 | Gender, age, marital status, education level, occupation, profession, monthly income, residential area, myths, conspiracy beliefs. |
| 7 | Bongomin et al. [31] | Uganda | Cross-sectional | 29 March–14 April 2021 | General population | 317 | 68.14 | Female, patients who agreed or strongly agreed that they had some immunity against COVID-19, patients who had a history of vaccine hesitancy for their children. |
| 8 | Bono et al. [32] | Bangladesh | Cross-sectional | 10 December 2020–9 February 2021 | General population | 230 | 89.57 | COVID-19 knowledge, worry/fear regarding COVID-19, higher income, younger age, testing negative for COVID-19. |
| DR Congo | 219 | 59.36 | ||||||
| Benin | 159 | 48.43 | ||||||
| Uganda | 107 | 88.79 | ||||||
| Malawi | 81 | 61.73 | ||||||
| Mali | 55 | 74.55 | ||||||
| 9 | Bono et al. [32] | The Democratic Republic of Congo | Cross-sectional | 24 August–8 September 2020 | General population | 4131 | 55.92 | Middle or high-income, being tested for COVID-19, COVID-19 community vaccine acceptance, acknowledging the existence of COVID-19, healthcare worker. |
| 10 | Carcelen et al. [33] | Zambia | Cross-sectional | 23–29 November 2020 | Caregivers | 2400 | 65.71 | Belief in the COVID-19 vaccine safety and efficacy. |
| 11 | Carpio et al. [34] | Kenya | Cross-sectional | 7–15 April 2020 | General population | 963 | 95.64 | Vaccine duration of protection and efficacy, perceived probability of being hospitalized, age, gender, education, location, region of residence, household income. |
| 12 | Dinga et al. [21] | Cameroon | Cross-sectional | May–August 2020 | General population | 2512 | 15.45 | NR * |
| 13 | Echoru et al. [35] | Western Uganda | Cross-sectional | July–September 2020 | General population | 1067 | 53.61 | Younger, male, tertiary level of students, Muslims, married, on-salary earners, rural dwellers. |
| 14 | Elgendy and Abdelrahim [36] | Egypt | Cross-sectional | April–May 2021 | General population | 871 | 88.06 | NR |
| 15 | El-Sokkary et al. [37] | Egypt | Cross-sectional | 25–31 January 2021 | Healthcare professionals | 308 | 25.97 | Income, years of experience. |
| 16 | Fares et al. [22] | Egypt | Observational | December 2020–January 2021 | Healthcare professionals | 385 | 21.04 | Male, interacting directly with COVID-19 patients, taking non-compulsory vaccines, recommending COVID-19 vaccination to others, receiving advice from hospitals to get the vaccine, trust in vaccine producers, pharmaceutical companies, and authorities. |
| 17 | Hammam et al. [38] | Egypt | Cross-sectional | April 2021 | Healthcare professionals | 187 | 30.48 | NR |
| 18 | Harapan et al. [8] | Indonesia | Cross-sectional | 25 March–6 April 2020 | General population | 1359 | 93.30 | Female, middle-aged, retired, married, healthcare worker, moderate perceived risk of COVID-19 infection. |
| 19 | Huynh et al. [39] | Vietnam | Cross-sectional | December 2020–January 2021 | General population | 425 | 84.00 | Knowledge of COVID-19, cues to action toward the vaccine. |
| 20 | Jain et al. [40] | India | Cross-sectional | 2 February–7 March 2021 | Healthcare students | 1068 | 89.42 | NR |
| 21 | Kanyike et al. [41] | Uganda | Cross-sectional | 15–21 March 2021 | Healthcare students | 600 | 37.33 | Male, being single, very high or moderate perceived risk of contracting COVID-19, receiving any vaccine in the past five years, COVID-19 vaccine hesitancy, |
| 22 | Kaur et al. [42] | India | Cross-sectional | January 2021 | Healthcare professionals | 520 | 63.08 | Dental professional, involved in COVID-19 duties, preference for natural immunity over the vaccine, belief in COVID-19 vaccine safety, interest in vaccine information, belief that vaccine should be compulsory. |
| 23 | Kitonsa et al. [43] | Uganda | Cross-sectional | September–November 2020 | Healthcare professionals | 657 | 70.17 | NR |
| 24 | Kumari et al. [44] | India | Cross-sectional | 13–25 March 2021 | General population | 1294 | 83.54 | Older, belief that the vaccine is harmless, belief that vaccine benefits outweigh the risks, belief that getting vaccinated is a societal responsibility, belief that sufficient data about the vaccine is available, belief that the vaccine will eradicate COVID-19, role model getting vaccinated, many other people getting vaccinated, higher socioeconomic status, developed place of residence. |
| 25 | Lamptey et al. [45] | Ghana | Cross-sectional | 14 October–12 December 2020 | General population | 1000 | 54.10 | Being married, government worker, high-risk perceptions. |
| 26 | Lazarus et al. [46] | India | Cross-sectional | 16–20 June 2020 | General population | 742 | 74.53 | Male, older, higher education. |
| Nigeria | 670 | 65.22 | ||||||
| South Korea | 752 | 79.79 | ||||||
| 27 | Lazarus et al. [47] | South Korea | Cross-sectional | 16–20 June 2020 | General population | 619 to 773 | 79.79 | NR |
| India | 74.53 | |||||||
| Nigeria | 65.22 | |||||||
| 28 | Mohamad et al. [48] | Syria | Cross-sectional | 23 December 2020–5 January 2021 | General population | 3402 | 35.82 | Female, younger, urban resident, not married, no kids, not a healthcare worker, not a smoker, no fear of COVID-19, perceived severity of COVID-19, belief in the natural origin of the virus, knowledge on vaccine hesitancy. |
| 29 | Panda et al. [49] | India | Cross-sectional | February 2021 | General population | 359 | 8.08 | NR |
| 30 | Parvej et al. [50] | Bangladesh | Cross-sectional | 17–26 April 2021 | General population | 1529 | 67.04 | Muslim, highly educated, living in urban areas, believing vaccines protect against infectious diseases and vaccines, having no health-related risks. |
| 31 | Paudel et al. [51] | Nepal | Cross-sectional | 27 January–3 February 2021 | Healthcare professionals | 266 | 38.35 | NR |
| 32 | Qunaibi et al. [52] | Algeria | Cross-sectional | 14–29 January 2021 | General population | 2706 | 3.62 | Receiving the influenza vaccine regularly, health care worker, resident in country with higher rates of COVID-19 infections. |
| Egypt | 5339 | 8.04 | ||||||
| Mauritania | 99 | 8.08 | ||||||
| Morocco | 3775 | 7.89 | ||||||
| Sudan | 313 | 15.34 | ||||||
| Syria | 1232 | 10.71 | ||||||
| Tunisia | 665 | 6.47 | ||||||
| Yemen | 226 | 9.29 | ||||||
| 33 | Ramesh Masthi and Sowmyashree [53] | India | Cross-sectional | January 2021 | General population | 846 | 64.42 | NR |
| 34 | Saied et al. [54] | Egypt | Cross-sectional | 8–15 January 2021 | Healthcare students | 2133 | 34.79 | Pharmacy student, higher academic year or graduate, average to very good self-perception of health status, good self-rated COVID-19 knowledge level, presence of confirmed COVID-19 infection in a close social network. |
| 35 | Skjefte et al. [55] | India | Cross-sectional | 28 October–18 November 2020 | Pregnant women, mothers of young children | 1639 | Pregnant women (52) Non-pregnant woman (73.4) | NR |
| Philippines | 1034 | NR | ||||||
| 36 | Solis Arce et al. [15] | Burkina Faso | Cross-sectional | 15 October–4 December 2020 | General population | 977 | 66.53 | Protection for self, family, and community, recommendation from health workers and government. |
| India | 17 June 2020–18 January 2021 | General population | 1680 | 84.29 | ||||
| Mozambique | 30 October–30 November 2020 | General population | 862 | 89.10 | ||||
| Nepal | 1–11 December 2020 | General population | 1389 | 96.62 | ||||
| Nigeria | 18 November–18 December 2020 | General population | 1868 | 76.18 | ||||
| Pakistan 1 | 24 July–9 September 2020 | General population | 1633 | 76.12 | ||||
| Pakistan 2 | 2 September–13 October 2020 | General population | 1492 | 66.49 | ||||
| Rwanda | 22 October–15 November 2020 | General population | 1355 | 84.87 | ||||
| Sierra Leone 1 | 2–19 October 2020 | General population | 1070 | 78.04 | ||||
| Sierra Leone 2 | 7 October 2020–20 January 2021 | General population | 2110 | 87.91 | ||||
| Uganda 1 | 21 September–12 December 2020 | General population | 3362 | 85.81 | ||||
| Uganda 2 | 23 November–12 December 2020 | General population | 1366 | 76.50 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Patwary, M.M.; Alam, M.A.; Bardhan, M.; Disha, A.S.; Haque, M.Z.; Billah, S.M.; Kabir, M.P.; Browning, M.H.E.M.; Rahman, M.M.; Parsa, A.D.; et al. COVID-19 Vaccine Acceptance among Low- and Lower-Middle-Income Countries: A Rapid Systematic Review and Meta-Analysis. Vaccines 2022, 10, 427. https://doi.org/10.3390/vaccines10030427
Patwary MM, Alam MA, Bardhan M, Disha AS, Haque MZ, Billah SM, Kabir MP, Browning MHEM, Rahman MM, Parsa AD, et al. COVID-19 Vaccine Acceptance among Low- and Lower-Middle-Income Countries: A Rapid Systematic Review and Meta-Analysis. Vaccines. 2022; 10(3):427. https://doi.org/10.3390/vaccines10030427
Chicago/Turabian StylePatwary, Muhammad Mainuddin, Md Ashraful Alam, Mondira Bardhan, Asma Safia Disha, Md. Zahidul Haque, Sharif Mutasim Billah, Md Pervez Kabir, Matthew H. E. M. Browning, Md. Mizanur Rahman, Ali Davod Parsa, and et al. 2022. "COVID-19 Vaccine Acceptance among Low- and Lower-Middle-Income Countries: A Rapid Systematic Review and Meta-Analysis" Vaccines 10, no. 3: 427. https://doi.org/10.3390/vaccines10030427
APA StylePatwary, M. M., Alam, M. A., Bardhan, M., Disha, A. S., Haque, M. Z., Billah, S. M., Kabir, M. P., Browning, M. H. E. M., Rahman, M. M., Parsa, A. D., & Kabir, R. (2022). COVID-19 Vaccine Acceptance among Low- and Lower-Middle-Income Countries: A Rapid Systematic Review and Meta-Analysis. Vaccines, 10(3), 427. https://doi.org/10.3390/vaccines10030427












