Paraoxonase 1 Phenotype and Protein N-Homocysteinylation in Patients with Rheumatoid Arthritis: Implications for Cardiovascular Disease
Abstract
:1. Introduction
2. Materials and Methods
2.1. Patients and Control Subjects
2.2. Anthropometric, Clinical, and Laboratory Data
2.3. PON1 Activity Toward Synthetic Substrates
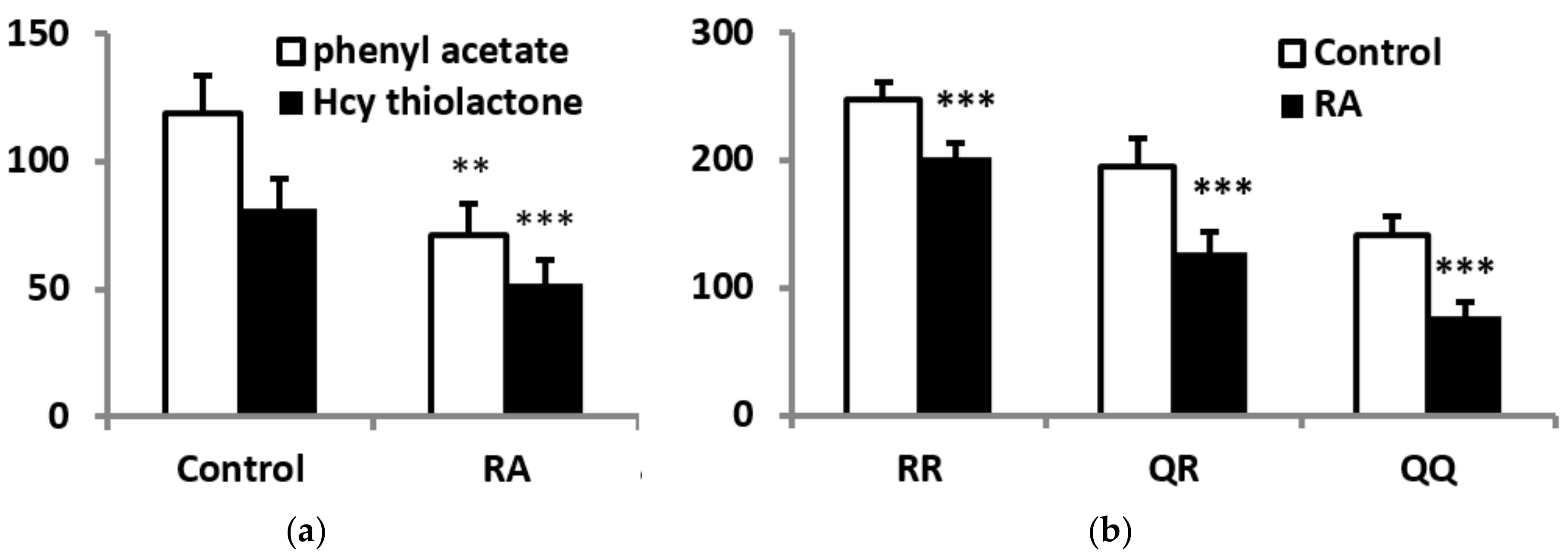
2.4. PON1 Polymorphism
2.5. PON1 Activity Toward Homocysteine Thiolactone
2.6. PON1 Protein Concentration
2.7. Serum Homocysteine Concentration and Protein N-Homocysteinylation
2.8. Reagents
2.9. Statistical Analysis
3. Results
3.1. Characteristics of RA Patients and Control Subjects
3.2. PON1 Activities, Protein Concentrations, and Phenotype
3.3. Plasma Homocysteine and N-Hcy-Protein
3.4. PON1 Status, Homocysteine, and N-Hcy Protein in RA Patients Stratified According to Disease Activity
3.5. PON1 Status, Homocysteine, and N-Hcy Protein in RA Patients and Control Subjects with Normal Renal Function
3.6. Lipid Profile, PON1 Status, Homocysteine, and N-Hcy Protein in Healthy and RA Females Startified According to Menopausal Status
3.7. Correlations
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Van Vollenhoven, R.F. Sex differences in rheumatoid arthritis: More than meets the eye. BMC Med. 2009, 7, 12. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Van den Hoek, J.; Boshuizen, H.C.; Roorda, L.D.; Tijhuis, G.J.; Nurmohamed, M.T.; van den Bos, G.A.; Dekker, J. Mortality in patients with rheumatoid arthritis: A 15-year prospective cohort study. Rheumatol. Int. 2017, 37, 487–493. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Urman, A.; Taklalsingh, N.; Sorrento, C.; McFarlane, I.M. Inflammation beyond the Joints: Rheumatoid Arthritis and cardiovascular disease. Scifed. J. Cardiol. 2018, 2, 1000019. [Google Scholar]
- Zavodovsky, B.V.; Sivordova, L.E. Cardiovascular safety of non-steroidal anti-inflammatory drugs in chronic inflammatory rheumatic diseases. Ter. Arkhiv 2018, 90, 101–106. [Google Scholar] [CrossRef] [PubMed]
- Peters, M.J.L.; Symmons, D.P.M.; McCarey, D.; Dijkmans, B.A.C.; Nicola, P.; Kvien, T.K.; McInnes, I.B.; Haentzschel, H.; Gonzalez-Gay, A.; Provan, S.; et al. EULAR evidence-based recommendations for cardiovascular risk management in patients with rheumatoid arthritis and other forms of inflammatory arthritis. Ann. Rheum Dis. 2010, 69, 325–331. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Conelly, P.W.; Picardo, C.M.; Potter, P.M.; Teiber, J.F.; Maguire, G.F.; Ng, D.S. Mouse serum paraoxonase-1 lactonase activity is specific for medium-chain length fatty acid lactones. Biochim. Biophys. Acta 2011, 1811, 39–45. [Google Scholar] [CrossRef]
- Jakubowski, H. Calcium-dependent human serum homocysteine thiolactone hydrolase. A protective mechanism against protein N-homocysteinylation. J. Biol. Chem. 2000, 275, 3957–3962. [Google Scholar] [CrossRef] [Green Version]
- Perla-Kajan, J.; Jakubowski, H. Paraoxonase 1 protects against protein N-homocysteinylation in humans. FASEB J. 2010, 24, 931–936. [Google Scholar] [CrossRef]
- Borowczyk, K.; Shih, D.M.; Jakubowski, H. Metabolism and neurotoxicity of homocysteine thiolactone in mice: Evidence for a protective role of paraoxonase 1. J. Alzheimers Dis. 2012, 30, 225–231. [Google Scholar] [CrossRef]
- Perła-Kaján, J.; Jakubowski, H. Paraoxonase 1 and homocysteine metabolism. Amino Acids 2012, 43, 1405–1417. [Google Scholar] [CrossRef]
- Shih, D.M.; Gu, L.; Xia, Y.-R.; Navab, M.; Li, W.-F.; Hama, S.; Castellani, L.W.; Furlong, C.E.; Costa, L.G.; Fogelman, A.M.; et al. Mice lacking serum paraoxonase are susceptible to organophosphate toxicity and atherosclerosis. Nature 1998, 394, 284–287. [Google Scholar] [CrossRef] [PubMed]
- Moya, C.; Máñez, S. Paraoxonases: Metabolic role and pharmacological projection. Naunyn Schmiedebergs Arch. Pharmacol. 2018, 391, 349–359. [Google Scholar] [CrossRef] [PubMed]
- Tanimoto, N.; Kumon, Y.; Suehiro, T.; Ohkubo, S.; Ikeda, Y.; Nishiya, K.; Hashimoto, K. Serum paraoxonase activity decreases in rheumatoid arthritis. Life Sci. 2003, 72, 2877–2885. [Google Scholar] [CrossRef]
- Baskol, G.; Demir, H.; Baskol, M.; Kilic, E.; Ates, F.; Kocer, D.; Muhtaroglu, S. Assessment of paraoxonase 1 activity and malondialdehyde levels in patients with rheumatoid arthritis. Clin. Biochem. 2005, 38, 951–955. [Google Scholar] [CrossRef]
- Isik, A.; Koca, S.S.; Ustundag, B.; Celik, H.; Yildirim, A. Paraoxonase and arylesterase levels in rheumatoid arthritis. Clin. Rheumatol. 2007, 26, 342–348. [Google Scholar] [CrossRef] [PubMed]
- Hashemi, M.; Moazeni-Roodi, A.K.; Fazaeli, A.; Sandoughi, M.; Bardestani, G.R.; Kordi-Tamandani, D.M.; Ghavami, S. Lack of association between paraoxonase-1 Q192R polymorphism and rheumatoid arthritis in southeast Iran. Genet Mol. Res. 2010, 9, 333–339. [Google Scholar] [CrossRef]
- Arnett, F.C.; Edworthy, S.M.; Bloch, D.A.; McShane, D.J.; Fries, J.F.; Cooper, N.S.; Healey, L.A.; Kaplan, S.R.; Liang, M.H.; Luthra, H.S.; et al. The American Rheumatism Association 1987 revised criteria for the classification of rheumatoid arthritis. Arthritis Rheum. 1988, 31, 315–324. [Google Scholar] [CrossRef]
- Anderson, J.; Caplan, L.; Yazdany, J.; Robbins, M.L.; Neogi, T.; Michaud, K.; Saag, K.G.; O’Dell, J.R.; Kazi, S. Rheumatoid arthritis disease activity measures: American college of rheumatology recommendations for use in clinical practice. Arthritis Care Res Hob. 2012, 64, 640–647. [Google Scholar] [CrossRef]
- Bełtowski, J.; Wójcicka, G.; Jakubowski, H. Modulation of paraoxonase 1 and protein N-homocysteinylation by leptin and the synthetic liver X receptor agonist T0901317 in the rat. J. Endocrinol. 2010, 204, 191–198. [Google Scholar] [CrossRef] [Green Version]
- Ayub, A.; Mackness, M.I.; Arrol, S.; Mackness, B.; Patel, J.; Durrington, P.M. Serum paraoxonase after myocardial infarction. Arterioscler. Thromb. Vasc. Biol. 1999, 19, 330–335. [Google Scholar] [CrossRef] [Green Version]
- Eckerson, H.W.; Romson, J.; Wyte, C. The human serum paraoxonase polymorphism: Identification of phenotypes by their response to salts. Am. J. Hum. Genet. 1983, 35, 214–227. [Google Scholar] [PubMed]
- Samouilidou, E.; Kostopoulos, V.; Liaouri, A.; Kioussi, E.; Vassiliou, K.; Bountou, E.; Grapsa, E. Association of lipid profile with serum PON1 concentration in patients with chronic kidney disease. Ren. Fail. 2016, 38, 1601–1606. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Choy, E.; Sattar, N. Interpreting lipid levels in the context of high-grade inflammatory states with a focus on rheumatoid arthritis: A challenge to conventional cardiovascular risk actions. Ann. Rheum. Dis. 2009, 68, 460–469. [Google Scholar] [CrossRef] [PubMed]
- Robertson, J.; Peters, M.J.; McInnes, I.B.; Sattar, N. Changes in lipid levels with inflammation and therapy in RA: A maturing paradigm. Nat. Rev. Rheumatol. 2013, 9, 513–523. [Google Scholar] [CrossRef] [PubMed]
- Gómez Rosso, L.; Lhomme, M.; Meroño, T.; Sorroche, P.; Catoggio, L.; Soriano, E.; Saucedo, C.; Malah, V.; Dauteuille, C.; Boero, L.; et al. Altered lipidome and antioxidative activity of small, dense HDL in normolipidemic rheumatoid arthritis: Relevance of inflammation. Atherosclerosis 2014, 237, 652–660. [Google Scholar] [CrossRef]
- Charles-Schoeman, C.; Lee, Y.Y.; Shahbazian, A.; Gorn, A.H.; FitzGerald, J.; Ranganath, V.K.; Taylor, M.; Ragavendra, N.; McMahon, M.; Elashoff, D.; et al. Association of paraoxonase 1 gene polymorphism and enzyme activity with carotid plaque in rheumatoid arthritis. Arthritis Rheum. 2013, 65, 2765–2772. [Google Scholar] [CrossRef] [Green Version]
- Shahmohamadnejad, S.; Vaisi-Raygani, A.; Shakiba, Y.; Kiani, A.; Rahimi, Z.; Bahrehmand, F.; Shakiba, E.; Pourmotabbed, T. Association between butyrylcholinesterase activity and phenotypes, paraoxonase192 rs662 gene polymorphism and their enzymatic activity with severity of rheumatoid arthritis: Correlation with systemic inflammatory markers and oxidative stress, preliminary report. Clin. Biochem. 2015, 48, 63–69. [Google Scholar] [CrossRef]
- Bae, S.C.; Lee, Y.H. Associations between paraoxonase 1 (PON1) polymorphisms and susceptibility and PON1 activity in rheumatoid arthritis patients, and comparison of PON1 activity in patients and controls: A meta-analysis. Clin. Rheumatol. 2019, 38, 2141–2149. [Google Scholar] [CrossRef]
- Shokri, Y.; Variji, A.; Nosrati, M.; Khonakdar-Tarsi, A.; Kianmehr, A.; Kashi, Z.; Bahar, A.; Bagheri, A.; Mahrooz, A. Importance of paraoxonase 1 (PON1) as an antioxidant and antiatherogenic enzyme in the cardiovascular complications of type 2 diabetes: Genotypic and phenotypic evaluation. Diabetes Res. Clin. Pract. 2020, 161, 08067. [Google Scholar] [CrossRef]
- Liu, P.; Wang, Q.; Cui, Y.; Wang, J. A meta-analysis of the relationship between paraoxonase 1 polymorphisms and cancer. Free Radic Res. 2019, 53, 1045–1050. [Google Scholar] [CrossRef]
- Jarvik, G.P.; Rozek, L.S.; Brophy, V.H.; Hatsukami, T.S.; Richter, R.; Schellenberg, G.D.; Furlong, C.E. Paraoxonase (PON1) phenotype is a better predictor of vascular disease than is PON1192 or PON155 genotype. Arterioscler. Thromb. Vasc. Biol. 2000, 20, 2441–2447. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Mackness, M.; Mackness, B. Human paraoxonase-1 (PON1): Gene structure and expression, promiscuous activities and multiple physiological roles. Gene 2015, 567, 12–21. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gaidukov, L.; Tawfik, D.S. High affinity, stability, and lactonase activity of serum paraoxonase PON1 anchored on HDL with ApoA-I. Biochemistry 2005, 44, 11843–11854. [Google Scholar] [CrossRef] [PubMed]
- Gaidukov, L.; Rosenblat, M.; Aviram, M.; Tawfik, D.S. The 192R/Q polymorphs of serum paraoxonase PON1 differ in HDL binding, lipolactonase stimulation, and cholesterol efflux. J. Lipid Res. 2006, 47, 2492–2502. [Google Scholar] [CrossRef] [Green Version]
- Armitage, J.; Holmes, M.V.; Preiss, D. Cholesteryl ester transfer protein inhibition for preventing cardiovascular events: JACC Review Topic of the Week. J. Am. Coll. Cardiol. 2019, 73, 477–487. [Google Scholar] [CrossRef]
- Ormseth, M.J.; Stein, C.M. High-density lipoprotein function in rheumatoid arthritis. Curr. Opin. Lipidol. 2016, 27, 67–75. [Google Scholar] [CrossRef] [Green Version]
- Kim, J.Y.; Lee, E.Y.; Park, J.K.; Song, Y.W.; Kim, J.R.; Cho, K.H. Patients with rheumatoid arthritis show altered lipoprotein profiles with dysfunctional high-density lipoproteins that can exacerbate inflammatory and atherogenic process. PLoS ONE 2016, 11, e0164564. [Google Scholar] [CrossRef]
- Feingold, K.R.; Memon, R.A.; Moser, A.H.; Grunfeld, C. Paraoxonase activity in the serum and hepatic mRNA levels decrease during the acute phase response. Atherosclerosis 1998, 139, 307–315. [Google Scholar] [CrossRef]
- Kumon, Y.; Suehiro, T.; Ikeda, Y.; Hashimoto, K. Human paraoxonase-1 gene expression by HepG2 cells is downregulated by interleukin-1β and tumor necrosis factor-α, but is upregulated by interleukin-6. Life Sci. 2003, 73, 2807–2815. [Google Scholar] [CrossRef]
- Van Lenten, B.J.; Hama, S.Y.; de Beer, F.C.; Stafforini, D.M.; McIntyre, T.M.; Prescott, S.M.; La Du, B.N.; Fogelman, A.M.; Navab, M. Anti-inflammatory HDL becomes pro-inflammatory during the acute phase response. Loss of protective effect of HDL against LDL oxidation in aortic wall cell cocultures. J. Clin. Investig. 1995, 96, 2758–2767. [Google Scholar] [CrossRef]
- Nys, G.; Cobraiville, G.; Servais, A.C.; Malaise, M.G.; de Seny, D.; Fillet, M. Targeted proteomics reveals serum amyloid A variants and alarmins S100A8-S100A9 as key plasma biomarkers of rheumatoid arthritis. Talanta 2019, 204, 507–517. [Google Scholar] [CrossRef] [PubMed]
- Jaouad, L.; Milochevitch, C.; Khalil, A. PON1 paraoxonase activity is reduced during HDL oxidation and is an indicator of HDL antioxidant capacity. Free Radic. Res. 2003, 37, 77–83. [Google Scholar] [CrossRef]
- Da Fonseca, L.J.S.; Nunes-Souza, V.; Goulart, M.O.F.; Rabelo, L.A. Oxidative stress in rheumatoid arthritis: What the future might hold regarding novel biomarkers and add-on therapies. Oxid. Med. Cell Longev. 2019, 2019, 7536805. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Nicholls, S.J.; Hazen, S.L. Myeloperoxidase, modified lipoproteins, and atherogenesis. J. Lipid Res. 2009, 50, S346–S351. [Google Scholar] [CrossRef] [Green Version]
- Charles-Schoeman, C.; Watanabe, J.; Lee, Y.Y.; Furst, D.E.; Amjadi, S.; Elashoff, D.; Park, G.; McMahon, M.; Paulus, H.E.; Fogelman, A.M.; et al. Abnormal function of high-density lipoprotein is associated with poor disease control and an altered protein cargo in rheumatoid arthritis. Arthritis Rheum. 2009, 60, 2870–2879. [Google Scholar] [CrossRef] [Green Version]
- Teng, N.; Maghzal, G.J.; Talib, J.; Rashid, I.; Lau, A.K.; Stocker, R. The roles of myeloperoxidase in coronary artery disease and its potential implication in plaque rupture. Redox Rep. 2017, 22, 51–73. [Google Scholar] [CrossRef] [Green Version]
- Shao, B.; Pennathur, S.; Heinecke, J.W. Myeloperoxidase targets apolipoprotein A-I, the major high density lipoprotein protein, for site-specific oxidation in human atherosclerotic lesions. J. Biol. Chem. 2012, 287, 6375–6386. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Huang, Y.; Wu, Z.; Riwanto, M.; Gao, S.; Levison, B.S.; Gu, X.; Fu, X.; Wagner, M.A.; Besler, C.; Gerstenecker, G.; et al. Myeloperoxidase, paraoxonase-1, and HDL form a functional ternary complex. J. Clin. Investig. 2013, 123, 3815–3828. [Google Scholar] [CrossRef] [Green Version]
- Deakin, S.; Moren, X.; James, R.W. HDL oxidation compromises its influence on paraoxonase-1 secretion and its capacity to modulate enzyme activity. Arterioscler. Thromb. Vasc. Biol. 2007, 27, 1146–1152. [Google Scholar] [CrossRef] [Green Version]
- Jakubowski, H. Homocysteine Modification in Protein Structure/Function and Human Disease. Physiol. Rev. 2019, 99, 555–604. [Google Scholar] [CrossRef]
- Undas, A.; Brozek, J.; Jankowski, M.; Siudak, Z.; Szczeklik, A.; Jakubowski, H. Plasma homocysteine affects fibrin clot permeability and resistance to lysis in human subjects. Arterioscler. Thromb. Vasc. Biol. 2006, 26, 1397–1404. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Borowczyk, K.; Piechocka, J.; Głowacki, R.; Dhar, I.; Midtun, Ø.; Tell, G.S.; Ueland, P.M.; Nygård, O.; Jakubowski, H. Urinary excretion of homocysteine thiolactone and the risk of acute myocardial infarction in coronary artery disease patients: The WENBIT trial. J. Intern. Med. 2019, 285, 232–244. [Google Scholar] [CrossRef] [PubMed]
- Gurda, D.; Handschuh, L.; Kotkowiak, W.; Jakubowski, H. Homocysteine thiolactone and N-homocysteinylated protein induce pro-atherogenic changes in gene expression in human vascular endothelial cells. Amino Acids 2015, 47, 1319–1339. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Undas, A.; Perła, J.; Lacinski, M.; Trzeciak, W.; Kaźmierski, R.; Jakubowski, H. Autoantibodies against N-homocysteinylated proteins in humans: Implications for atherosclerosis. Stroke 2004, 35, 1299–1304. [Google Scholar] [CrossRef] [Green Version]
- Mendes, R.H.; Sirvente, R.A.; Candido, G.O.; Mostarda, C.; Salemi, V.M.; D’Almeida, V.; Jacob, M.H.; Ribeiro, M.F.; Belló-Klein, A.; Rigatto, K.; et al. Homocysteine thiolactone induces cardiac dysfunction: Role of oxidative stress. J. Cardiovasc. Pharmacol. 2010, 55, 198–202. [Google Scholar] [CrossRef]
- Wu, S.; Gao, X.; Yang, S.; Meng, M.; Yang, X.; Ge, B. The role of endoplasmic reticulum stress in endothelial dysfunction induced by homocysteine thiolactone. Fundam. Clin. Pharmacol. 2015, 29, 252–259. [Google Scholar] [CrossRef]
- Smith, R.M.; Kruzliak, P.; Adamcikova, Z.; Zulli, A. Role of Nox inhibitors plumbagin, ML090 and gp91ds-tat peptide on homocysteine thiolactone induced blood vessel dysfunction. Clin. Exp. Pharmacol. Physiol. 2015, 42, 860–864. [Google Scholar] [CrossRef]
- AnandBabu, K.; Sen, P.; Angayarkanni, N. Oxidized LDL, homocysteine, homocysteine thiolactone and advanced glycation end products act as pro-oxidant metabolites inducing cytokine release, macrophage infiltration and pro-angiogenic effect in ARPE-19 cells. PLoS ONE 2019, 14, e0216899. [Google Scholar] [CrossRef] [Green Version]

| Control (n = 70) | RA (n = 74) | |
|---|---|---|
| Age (years) | 52.3 ± 8.8 | 55.4 ± 12.2 |
| Males/females | 12/58 | 14/60 |
| Body weight (kg) | 62.1 ± 11.3 | 65.6 ± 14.4 |
| Body mass index (kg/m2) | 24.9 ± 12.0 | 26.4 ± 13.5 |
| Waist circumference (cm) | 82.3 ± 10.6 | 84.7 ± 13.8 |
| Hip circumference (cm) | 97.9 ± 8.2 | 99.6 ± 8.6 |
| Waist-to-hip ratio | 0.81 ± 0.10 | 0.84 ± 0.11 |
| Disease duration (months) | - | 151 ± 107 |
| Control (n = 70) | RA (n = 74) | |
|---|---|---|
| Hemoglobin (g/dL) | 13.2 ± 1.8 | 12.3 ± 1.7 |
| Red blood cell count (mln/μL) | 4.51 ± 0.49 | 4.42 ± 0.53 |
| MCV | 88.7 ± 7.6 | 84.8 ± 8.3 |
| Platelet count (103/ μL) | 336 ± 115 | 324 ± 120 |
| White blood cells (103/ μL) | 7.23 ± 2.04 | 7.84 ± 2.47 |
| Total plasma protein (g/dL) | 7.23 ± 0.68 | 6.98 ± 0,59 |
| AST (U/L) | 22.1 ± 11.1 | 24.5 ± 13.3 |
| ALT (U/L) | 24.7 ± 11.3 | 26.1 ± 28.6 |
| Total cholesterol (mg/dL) | 211 ± 32 | 185 ± 46 |
| LDL-cholesterol (mg/dL) | 123 ± 21 | 106 ± 36 |
| HDL-cholesterol (mg/dL) | 54 ± 15 | 58 ± 18 |
| Non-HDL cholesterol (mg/dL) | 157 ± 28 | 128.3 ± 41.6 |
| Triglycerides (mg/dL) | 132 ± 25 | 108 ± 48 |
| Apolipoprotein A-I (μg/mL) | 1.12 ± 0.02 | 1.05 ± 0.03 |
| Apolipoprotein A-II (μg/mL) | 0.03 ± 0.01 | 0.04 ± 0.02 |
| ESR (mm/1 h) | 5.6 ± 3.8 | 35.4 ± 25.8 *** |
| CRP (mg/L) | 3.34 ± 0.76 | 27.9 ± 31.9 *** |
| MPO (ng/mL) | 1.5 ± 1.2 | 54.5 ± 13.2 *** |
| Remission-to-Moderate Activity (n = 32) | High Activity (n = 42) | |
|---|---|---|
| PON1 polymorphism | ||
| 17 | 22 | |
| QR | 13 | 16 |
| RR | 2 | 4 |
| PON1 concentration (μg/mL) | 27.4 ± 3.2 | 18.6 ± 2.1 ** |
| PON1 activity toward Hcy thiolactone (nmol × min−1 × mL−1) | 57.2 ± 12.1 | 42.1 ± 10.2 ** |
| Specific PON1 activity toward Hcy thiolactone (nmol × min−1 × μg−1) | 2.11 ± 0.53 | 2.44 ± 0.61 |
| Total Hcy (μM) | 9.12 ± 0.97 | 10.26 ± 1.24 |
| N-Hcy-protein (μM) | 2.53 ± 1.04 | 3.04 ± 0.88 ** |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Parada-Turska, J.; Wójcicka, G.; Beltowski, J. Paraoxonase 1 Phenotype and Protein N-Homocysteinylation in Patients with Rheumatoid Arthritis: Implications for Cardiovascular Disease. Antioxidants 2020, 9, 899. https://doi.org/10.3390/antiox9090899
Parada-Turska J, Wójcicka G, Beltowski J. Paraoxonase 1 Phenotype and Protein N-Homocysteinylation in Patients with Rheumatoid Arthritis: Implications for Cardiovascular Disease. Antioxidants. 2020; 9(9):899. https://doi.org/10.3390/antiox9090899
Chicago/Turabian StyleParada-Turska, Jolanta, Grażyna Wójcicka, and Jerzy Beltowski. 2020. "Paraoxonase 1 Phenotype and Protein N-Homocysteinylation in Patients with Rheumatoid Arthritis: Implications for Cardiovascular Disease" Antioxidants 9, no. 9: 899. https://doi.org/10.3390/antiox9090899
APA StyleParada-Turska, J., Wójcicka, G., & Beltowski, J. (2020). Paraoxonase 1 Phenotype and Protein N-Homocysteinylation in Patients with Rheumatoid Arthritis: Implications for Cardiovascular Disease. Antioxidants, 9(9), 899. https://doi.org/10.3390/antiox9090899




