Vitamin E Bioavailability: Mechanisms of Intestinal Absorption in the Spotlight
Abstract
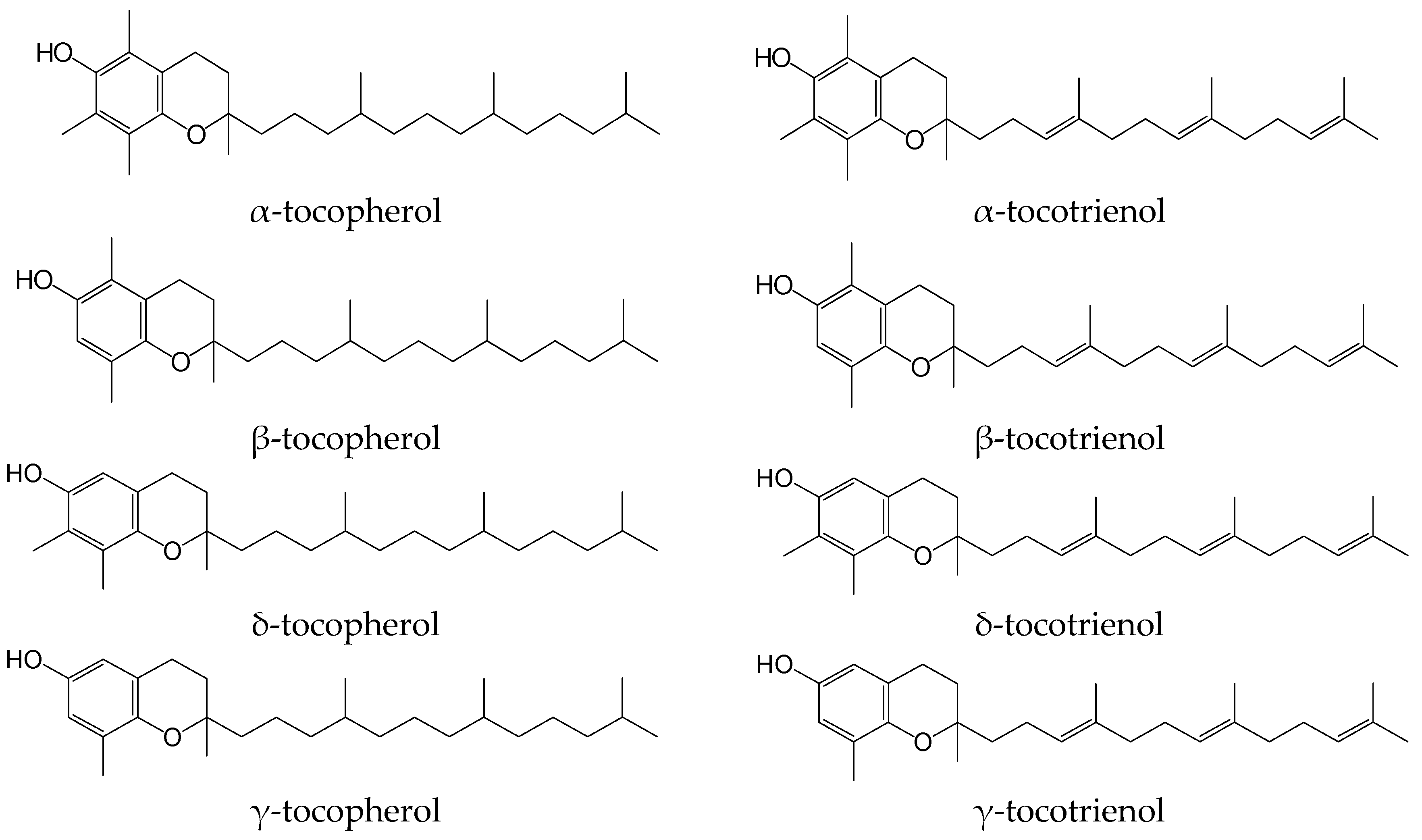
1. Introduction
2. Vitamin E Digestion Process
2.1. The Fate of Vitamin E in the Gastrointestinal Tract
2.2. Factors Affecting Vitamin E Transfer to Mixed Micelles
2.3. Vitamin E Ester Hydrolysis
3. Vitamin E Absorption Mechanisms by the Enterocyte
3.1. Apical Transport at the Brush Border Level
3.2. Vitamin E Trafficking across the Enterocyte
3.3. Basolateral Secretion to the Lymph or to the Blood Circulation
3.4. Vitamin E Absorption Site in the Intestine
3.5. Factors Modulating Vitamin E Absorption by the Intestinal Cell
4. Conclusions
Conflicts of Interest
References
- DellaPenna, D. A decade of progress in understanding vitamin E synthesis in plants. J. Plant Physiol. 2005, 162, 729–737. [Google Scholar] [CrossRef] [PubMed]
- Martin, A. Apports Nutritionnels Conseillés Pour la Population Française, 3rd ed.; Tec & Doc. Lavoisier: Paris, France, 2001; p. 605. [Google Scholar]
- Azzi, A.; Meydani, S.N.; Meydani, M.; Zingg, J.M. The rise, the fall and the renaissance of vitamin E. Arch. Biochem. Biophys. 2016, 595, 100–108. [Google Scholar] [CrossRef] [PubMed]
- Jiang, Q.; Christen, S.; Shigenaga, M.K.; Ames, B.N. gamma-Tocopherol, the major form of vitamin E in the US diet, deserves more attention. Am. J. Clin. Nutr. 2001, 74, 714–722. [Google Scholar] [PubMed]
- Table de Composition Nutritionnelle des Aliments Ciqual. 2016. Available online: https://www.anses.fr/fr/content/ciqual-la-table-de-composition-nutritionnelle-des-aliments (accessed on 31 October 2017).
- EFSA Panel on Dietetic Products, Nutrition, and Allergies (NDA). Dietary Reference Values for vitamin E as α-tocopherol. EFSA J. 2015, 13. [Google Scholar] [CrossRef]
- Medicine, I.O. Dietary Reference Intakes for Vitamin C, Vitamin E, Selenium, and Carotenoids; The National Academies Press: Washington, DC, USA, 2000; p. 529. [Google Scholar]
- De Carvalho, M.J.; Guilland, J.C.; Moreau, D.; Boggio, V.; Fuchs, F. Vitamin status of healthy subjects in Burgundy (France). Ann. Nutr. Metab. 1996, 40, 24–51. [Google Scholar] [CrossRef] [PubMed]
- Troesch, B.; Hoeft, B.; McBurney, M.; Eggersdorfer, M.; Weber, P. Dietary surveys indicate vitamin intakes below recommendations are common in representative western countries. Br. J. Nutr. 2012, 108, 692–698. [Google Scholar] [CrossRef] [PubMed]
- Traber, M.G. Vitamin E inadequacy in humans: Causes and consequences. Adv. Nutr. 2014, 5, 503–514. [Google Scholar] [CrossRef] [PubMed]
- Pignitter, M.; Stolze, K.; Gartner, S.; Dumhart, B.; Stoll, C.; Steiger, G.; Kraemer, K.; Somoza, V. Cold fluorescent light as major inducer of lipid oxidation in soybean oil stored at household conditions for eight weeks. J. Agric. Food Chem. 2014, 62, 2297–2305. [Google Scholar] [CrossRef] [PubMed]
- Borel, P.; Pasquier, B.; Armand, M.; Tyssandier, V.; Grolier, P.; Alexandre-Gouabau, M.C.; Andre, M.; Senft, M.; Peyrot, J.; Jaussan, V.; et al. Processing of vitamin A and E in the human gastrointestinal tract. Am. J. Physiol. Gastrointest. Liver Physiol. 2001, 280, G95–G103. [Google Scholar] [PubMed]
- Reboul, E.; Richelle, M.; Perrot, E.; Desmoulins-Malezet, C.; Pirisi, V.; Borel, P. Bioaccessibility of carotenoids and vitamin E from their main dietary sources. J. Agric. Food Chem. 2006, 54, 8749–8755. [Google Scholar] [CrossRef] [PubMed]
- Werner, S.; Bohm, V. Bioaccessibility of carotenoids and vitamin e from pasta: Evaluation of an in vitro digestion model. J. Agric. Food Chem. 2011, 59, 1163–1170. [Google Scholar] [CrossRef] [PubMed]
- Sriwichai, W.; Berger, J.; Picq, C.; Avallone, S. Determining Factors of Lipophilic Micronutrient Bioaccessibility in Several Leafy Vegetables. J. Agric. Food Chem. 2016, 64, 1695–1701. [Google Scholar] [CrossRef] [PubMed]
- Mandalari, G.; Faulks, R.M.; Rich, G.T.; Lo Turco, V.; Picout, D.R.; Lo Curto, R.B.; Bisignano, G.; Dugo, P.; Dugo, G.; Waldron, K.W.; et al. Release of protein, lipid, and vitamin E from almond seeds during digestion. J. Agric. Food Chem. 2008, 56, 3409–3416. [Google Scholar] [CrossRef] [PubMed]
- Cilla, A.; Alegria, A.; de Ancos, B.; Sanchez-Moreno, C.; Cano, M.P.; Plaza, L.; Clemente, G.; Lagarda, M.J.; Barbera, R. Bioaccessibility of tocopherols, carotenoids, and ascorbic acid from milk- and soy-based fruit beverages: Influence of food matrix and processing. J. Agric. Food Chem. 2012, 60, 7282–7290. [Google Scholar] [CrossRef] [PubMed]
- Nagao, A.; Kotake-Nara, E.; Hase, M. Effects of fats and oils on the bioaccessibility of carotenoids and vitamin E in vegetables. Biosci. Biotechnol. Biochem. 2013, 77, 1055–1060. [Google Scholar] [CrossRef] [PubMed]
- Yang, Y.; McClements, D.J. Vitamin E bioaccessibility: Influence of carrier oil type on digestion and release of emulsified alpha-tocopherol acetate. Food Chem. 2013, 141, 473–481. [Google Scholar] [CrossRef] [PubMed]
- Yang, Y.; Decker, E.A.; Xiao, H.; McClements, D.J. Enhancing vitamin E bioaccessibility: Factors impacting solubilization and hydrolysis of alpha-tocopherol acetate encapsulated in emulsion-based delivery systems. Food Funct. 2015, 6, 84–97. [Google Scholar] [CrossRef] [PubMed]
- Yang, Y.; Xiao, H.; McClements, D.J. Impact of Lipid Phase on the Bioavailability of Vitamin E in Emulsion-Based Delivery Systems: Relative Importance of Bioaccessibility, Absorption, and Transformation. J. Agric. Food Chem. 2017, 65, 3946–3955. [Google Scholar] [CrossRef] [PubMed]
- Yang, Y.; McClements, D.J. Vitamin E and vitamin E acetate solubilization in mixed micelles: Physicochemical basis of bioaccessibility. J. Colloid Interface Sci. 2013, 405, 312–321. [Google Scholar] [CrossRef] [PubMed]
- Lombardo, D.; Guy, O. Studies on the substrate specificity of a carboxyl ester hydrolase from human pancreatic juice. II. Action on cholesterol esters and lipid-soluble vitamin esters. Biochim. Biophys. Acta 1980, 611, 147–155. [Google Scholar] [CrossRef]
- Reboul, E.; Berton, A.; Moussa, M.; Kreuzer, C.; Crenon, I.; Borel, P. Pancreatic lipase and pancreatic lipase-related protein 2, but not pancreatic lipase-related protein 1, hydrolyze retinyl palmitate in physiological conditions. Biochim. Biophys. Acta 2006, 1761, 4–10. [Google Scholar] [CrossRef] [PubMed]
- Desmarchelier, C.; Tourniaire, F.; Preveraud, D.P.; Samson-Kremser, C.; Crenon, I.; Rosilio, V.; Borel, P. The distribution and relative hydrolysis of tocopheryl acetate in the different matrices coexisting in the lumen of the small intestine during digestion could explain its low bioavailability. Mol. Nutr. Food Res. 2013, 57, 1237–1245. [Google Scholar] [CrossRef] [PubMed]
- Nagy, K.; Ramos, L.; Courtet-Compondu, M.C.; Braga-Lagache, S.; Redeuil, K.; Lobo, B.; Azpiroz, F.; Malagelada, J.R.; Beaumont, M.; Moulin, J.; et al. Double-balloon jejunal perfusion to compare absorption of vitamin E and vitamin E acetate in healthy volunteers under maldigestion conditions. Eur. J. Clin. Nutr. 2013, 67, 202–206. [Google Scholar] [CrossRef] [PubMed]
- Mathias, P.M.; Harries, J.T.; Peters, T.J.; Muller, D.P. Studies on the in vivo absorption of micellar solutions of tocopherol and tocopheryl acetate in the rat: Demonstration and partial characterization of a mucosal esterase localized to the endoplasmic reticulum of the enterocyte. J. Lipid Res. 1981, 22, 829–837. [Google Scholar] [PubMed]
- Muralidhara, K.S.; Hollander, D. Intestinal absorption of alpha-tocopherol in the unanesthetized rat. The influence of luminal constituents on the absorptive process. J. Lab. Clin. Med. 1977, 90, 85–91. [Google Scholar] [PubMed]
- Hollander, D.; Rim, E.; Muralidhara, K.S. Mechanism and site of small intestinal absorption of alpha-tocopherol in the rat. Gastroenterology 1975, 68, 1492–1499. [Google Scholar] [PubMed]
- Reboul, E.; Klein, A.; Bietrix, F.; Gleize, B.; Malezet-Desmoulins, C.; Schneider, M.; Margotat, A.; Lagrost, L.; Collet, X.; Borel, P. Scavenger receptor class B type I (SR-BI) is involved in vitamin E transport across the enterocyte. J. Biol. Chem. 2006, 281, 4739–4745. [Google Scholar] [CrossRef] [PubMed]
- Narushima, K.; Takada, T.; Yamanashi, Y.; Suzuki, H. Niemann-pick C1-like 1 mediates alpha-tocopherol transport. Mol. Pharmacol. 2008, 74, 42–49. [Google Scholar] [CrossRef] [PubMed]
- Reboul, E.; Soayfane, Z.; Goncalves, A.; Cantiello, M.; Bott, R.; Nauze, M.; Terce, F.; Collet, X.; Comera, C. Respective contributions of intestinal Niemann-Pick C1-like 1 and scavenger receptor class B type I to cholesterol and tocopherol uptake: In vivo v. in vitro studies. Br. J. Nutr. 2012, 107, 1296–1304. [Google Scholar] [CrossRef] [PubMed]
- Abuasal, B.; Sylvester, P.W.; Kaddoumi, A. Intestinal absorption of gamma-tocotrienol is mediated by Niemann-Pick C1-like 1: In situ rat intestinal perfusion studies. Drug Metab. Dispos. 2010, 38, 939–945. [Google Scholar] [CrossRef] [PubMed]
- Goncalves, A.; Roi, S.; Nowicki, M.; Niot, I.; Reboul, E. Cluster-determinant 36 (CD36) impacts on vitamin E postprandial response. Mol. Nutr. Food Res. 2014, 58, 2297–2306. [Google Scholar] [CrossRef] [PubMed]
- Werder, M.; Han, C.H.; Wehrli, E.; Bimmler, D.; Schulthess, G.; Hauser, H. Role of scavenger receptors SR-BI and CD36 in selective sterol uptake in the small intestine. Biochemistry 2001, 40, 11643–11650. [Google Scholar] [CrossRef] [PubMed]
- Altmann, S.W.; Davis, H.R., Jr.; Zhu, L.J.; Yao, X.; Hoos, L.M.; Tetzloff, G.; Iyer, S.P.; Maguire, M.; Golovko, A.; Zeng, M.; et al. Niemann-Pick C1 Like 1 protein is critical for intestinal cholesterol absorption. Science 2004, 303, 1201–1204. [Google Scholar] [CrossRef] [PubMed]
- Borel, P.; Lietz, G.; Goncalves, A.; Szabo de Edelenyi, F.; Lecompte, S.; Curtis, P.; Goumidi, L.; Caslake, M.J.; Miles, E.A.; Packard, C.; et al. CD36 and SR-BI Are Involved in Cellular Uptake of Provitamin A Carotenoids by Caco-2 and HEK Cells, and Some of Their Genetic Variants Are Associated with Plasma Concentrations of These Micronutrients in Humans. J. Nutr. 2013, 143, 448–456. [Google Scholar] [CrossRef] [PubMed]
- Reboul, E.; Abou, L.; Mikail, C.; Ghiringhelli, O.; Andre, M.; Portugal, H.; Jourdheuil-Rahmani, D.; Amiot, M.J.; Lairon, D.; Borel, P. Lutein transport by Caco-2 TC-7 cells occurs partly by a facilitated process involving the scavenger receptor class B type I (SR-BI). Biochem. J. 2005, 387, 455–461. [Google Scholar] [CrossRef] [PubMed]
- Reboul, E.; Goncalves, A.; Comera, C.; Bott, R.; Nowicki, M.; Landrier, J.F.; Jourdheuil-Rahmani, D.; Dufour, C.; Collet, X.; Borel, P. Vitamin D intestinal absorption is not a simple passive diffusion: Evidences for involvement of cholesterol transporters. Mol. Nutr. Food Res. 2011, 55, 691–702. [Google Scholar] [CrossRef] [PubMed]
- Goncalves, A.; Margier, M.; Roi, S.; Collet, X.; Niot, I.; Goupy, P.; Caris-Veyrat, C.; Reboul, E. Intestinal scavenger receptors are involved in vitamin K1 absorption. J. Biol. Chem. 2014, 289, 30743–30752. [Google Scholar] [CrossRef] [PubMed]
- Davis, H.R., Jr.; Zhu, L.J.; Hoos, L.M.; Tetzloff, G.; Maguire, M.; Liu, J.; Yao, X.; Iyer, S.P.; Lam, M.H.; Lund, E.G.; et al. Niemann-Pick C1 Like 1 (NPC1L1) is the intestinal phytosterol and cholesterol transporter and a key modulator of whole-body cholesterol homeostasis. J. Biol. Chem. 2004, 279, 33586–33592. [Google Scholar] [CrossRef] [PubMed]
- Takada, T.; Yamanashi, Y.; Konishi, K.; Yamamoto, T.; Toyoda, Y.; Masuo, Y.; Yamamoto, H.; Suzuki, H. NPC1L1 is a key regulator of intestinal vitamin K absorption and a modulator of warfarin therapy. Sci. Transl. Med. 2015, 7. [Google Scholar] [CrossRef] [PubMed]
- Sato, Y.; Suzuki, R.; Kobayashi, M.; Itagaki, S.; Hirano, T.; Noda, T.; Mizuno, S.; Sugawara, M.; Iseki, K. Involvement of cholesterol membrane transporter Niemann-Pick C1-like 1 in the intestinal absorption of lutein. J. Pharm. Pharm. Sci. 2012, 15, 256–264. [Google Scholar] [CrossRef] [PubMed]
- Drover, V.A.; Nguyen, D.V.; Bastie, C.C.; Darlington, Y.F.; Abumrad, N.A.; Pessin, J.E.; London, E.; Sahoo, D.; Phillips, M.C. CD36 mediates both cellular uptake of very long chain fatty acids and their intestinal absorption in mice. J. Biol. Chem. 2008, 283, 13108–13115. [Google Scholar] [CrossRef] [PubMed]
- Hansen, G.H.; Niels-Christiansen, L.L.; Immerdal, L.; Danielsen, E.M. Scavenger receptor class B type I (SR-BI) in pig enterocytes: Trafficking from the brush border to lipid droplets during fat absorption. Gut 2003, 52, 1424–1431. [Google Scholar] [CrossRef] [PubMed]
- Ge, L.; Wang, J.; Qi, W.; Miao, H.H.; Cao, J.; Qu, Y.X.; Li, B.L.; Song, B.L. The cholesterol absorption inhibitor ezetimibe acts by blocking the sterol-induced internalization of NPC1L1. Cell Metab. 2008, 7, 508–519. [Google Scholar] [CrossRef] [PubMed]
- Kamishikiryo, J.; Haraguchi, M.; Nakashima, S.; Tasaka, Y.; Narahara, H.; Sugihara, N.; Nakamura, T.; Morita, T. N-terminal domain of the cholesterol transporter Niemann-Pick C1-like 1 (NPC1L1) is essential for alpha-tocopherol transport. Biochem. Biophys. Res. Commun. 2017, 486, 476–480. [Google Scholar] [CrossRef] [PubMed]
- Tran, T.T.; Poirier, H.; Clement, L.; Nassir, F.; Pelsers, M.M.; Petit, V.; Degrace, P.; Monnot, M.C.; Glatz, J.F.; Abumrad, N.A.; et al. Luminal lipid regulates CD36 levels and downstream signaling to stimulate chylomicron synthesis. J. Biol. Chem. 2011, 286, 25201–25210. [Google Scholar] [CrossRef] [PubMed]
- Beaslas, O.; Cueille, C.; Delers, F.; Chateau, D.; Chambaz, J.; Rousset, M.; Carriere, V. Sensing of dietary lipids by enterocytes: A new role for SR-BI/CLA-1. PLoS ONE 2009, 4, e4278. [Google Scholar] [CrossRef] [PubMed]
- Buttet, M.; Traynard, V.; Tran, T.T.; Besnard, P.; Poirier, H.; Niot, I. From fatty-acid sensing to chylomicron synthesis: Role of intestinal lipid-binding proteins. Biochimie 2014, 96, 37–47. [Google Scholar] [CrossRef] [PubMed]
- Briand, O.; Touche, V.; Colin, S.; Brufau, G.; Davalos, A.; Schonewille, M.; Bovenga, F.; Carriere, V.; de Boer, J.F.; Dugardin, C.; et al. Liver X Receptor Regulates Triglyceride Absorption Through Intestinal Down-regulation of Scavenger Receptor Class B, Type 1. Gastroenterology 2016, 150, 650–658. [Google Scholar] [CrossRef] [PubMed]
- Goncalves, A.; Gontero, B.; Nowicki, M.; Margier, M.; Masset, G.; Amiot, M.J.; Reboul, E. Micellar lipid composition affects micelle interaction with class B scavenger receptor extracellular loops. J. Lipid Res. 2015, 56, 1123–1133. [Google Scholar] [CrossRef] [PubMed]
- Anwar, K.; Kayden, H.J.; Hussain, M.M. Transport of vitamin E by differentiated Caco-2 cells. J. Lipid Res. 2006, 47, 1261–1273. [Google Scholar] [CrossRef] [PubMed]
- Zimmer, S.; Stocker, A.; Sarbolouki, M.N.; Spycher, S.E.; Sassoon, J.; Azzi, A. A novel human tocopherol-associated protein—Cloning, in vitro expression, and characterization. J. Biol. Chem. 2000, 275, 25672–25680. [Google Scholar] [CrossRef] [PubMed]
- Manor, D.; Atkinson, J. Is tocopherol associated protein a misnomer? J. Nutr. Biochem 2003, 14, 421–422. [Google Scholar] [CrossRef]
- Zingg, J.M.; Kempna, P.; Paris, M.; Reiter, E.; Villacorta, L.; Cipollone, R.; Munteanu, A.; De Pascale, C.; Menini, S.; Cueff, A.; et al. Characterization of three human sec14p-like proteins: Alpha-tocopherol transport activity and expression pattern in tissues. Biochimie 2008, 90, 1703–1715. [Google Scholar] [CrossRef] [PubMed]
- Anwar, K.; Iqbal, J.; Hussain, M.M. Mechanisms involved in vitamin E transport by primary enterocytes and in vivo absorption. J. Lipid Res. 2007, 48, 2028–2038. [Google Scholar] [CrossRef] [PubMed]
- Reboul, E.; Trompier, D.; Moussa, M.; Klein, A.; Landrier, J.F.; Chimini, G.; Borel, P. ATP-binding cassette transporter A1 is significantly involved in the intestinal absorption of alpha- and gamma-tocopherol but not in that of retinyl palmitate in mice. Am. J. Clin. Nutr. 2009, 89, 177–184. [Google Scholar] [CrossRef] [PubMed]
- Olivier, M.; Bott, G.R.; Frisdal, E.; Nowick, M.; Plengpanich, W.; Desmarchelier, C.; Roi, S.; Quinn, C.M.; Gelissen, I.; Jessup, W.; et al. ABCG1 is involved in vitamin E efflux. Biochim. Biophys. Acta 2014, 1841, 1741–1751. [Google Scholar] [CrossRef] [PubMed]
- Nicod, N.; Parker, R.S. Vitamin E secretion by Caco-2 monolayers to APOA1, but not to HDL, is vitamer selective. J. Nutr. 2013, 143, 1565–1572. [Google Scholar] [CrossRef] [PubMed]
- Ramasamy, I. Update on the molecular biology of dyslipidemias. Clin. Chim. Acta 2016, 454, 143–185. [Google Scholar] [CrossRef] [PubMed]
- Cuerq, C.; Restier, L.; Drai, J.; Blond, E.; Roux, A.; Charriere, S.; Michalski, M.C.; Di Filippo, M.; Levy, E.; Lachaux, A.; et al. Establishment of reference values of alpha-tocopherol in plasma, red blood cells and adipose tissue in healthy children to improve the management of chylomicron retention disease, a rare genetic hypocholesterolemia. Orphanet J. Rare Dis. 2016, 11, 114. [Google Scholar] [CrossRef] [PubMed]
- Burnett, J.R.; Hooper, A.J. Vitamin E and oxidative stress in abetalipoproteinemia and familial hypobetalipoproteinemia. Free Radic. Biol. Med. 2015, 88, 59–62. [Google Scholar] [CrossRef] [PubMed]
- Reboul, E.; Borel, P. Proteins involved in uptake, intracellular transport and basolateral secretion of fat-soluble vitamins and carotenoids by mammalian enterocytes. Prog. Lipid Res. 2011, 50, 388–402. [Google Scholar] [CrossRef] [PubMed]
- Goncalves, A.; Roi, S.; Nowicki, M.; Dhaussy, A.; Huertas, A.; Amiot, M.J.; Reboul, E. Fat-soluble vitamin intestinal absorption: Absorption sites in the intestine and interactions for absorption. Food Chem. 2015, 172, 155–160. [Google Scholar] [CrossRef] [PubMed]
- Nassir, F.; Wilson, B.; Han, X.; Gross, R.W.; Abumrad, N.A. CD36 is important for fatty acid and cholesterol uptake by the proximal but not distal intestine. J. Biol. Chem. 2007, 282, 19493–19501. [Google Scholar] [CrossRef] [PubMed]
- Bietrix, F.; Yan, D.; Nauze, M.; Rolland, C.; Bertrand-Michel, J.; Comera, C.; Schaak, S.; Barbaras, R.; Groen, A.K.; Perret, B.; et al. Accelerated lipid absorption in mice overexpressing intestinal SR-BI. J. Biol. Chem. 2006, 281, 7214–7219. [Google Scholar] [CrossRef] [PubMed]
- Cai, S.F.; Kirby, R.J.; Howles, P.N.; Hui, D.Y. Differentiation-dependent expression and localization of the class B type I scavenger receptor in intestine. J. Lipid Res. 2001, 42, 902–909. [Google Scholar] [PubMed]
- Masson, C.J.; Plat, J.; Mensink, R.P.; Namiot, A.; Kisielewski, W.; Namiot, Z.; Fullekrug, J.; Ehehalt, R.; Glatz, J.F.; Pelsers, M.M. Fatty acid- and cholesterol transporter protein expression along the human intestinal tract. PLoS ONE 2010, 5, e10380. [Google Scholar] [CrossRef] [PubMed]
- Drevon, C.A. Absorption, transport and metabolism of vitamin E. Free Radic. Res. Commun. 1991, 14, 229–246. [Google Scholar] [CrossRef] [PubMed]
- Traber, M.G.; Sies, H. Vitamin E in humans: Demand and delivery. Annu. Rev. Nutr. 1996, 16, 321–347. [Google Scholar] [CrossRef] [PubMed]
- Cohn, W. Bioavailability of vitamin E. Eur. J. Clin. Nutr. 1997, 51, S80–S85. [Google Scholar] [PubMed]
- Bruno, R.S.; Leonard, S.W.; Park, S.I.; Zhao, Y.; Traber, M.G. Human vitamin E requirements assessed with the use of apples fortified with deuterium-labeled alpha-tocopheryl acetate. Am. J. Clin. Nutr. 2006, 83, 299–304. [Google Scholar] [PubMed]
- Traber, M.G.; Burton, G.W.; Ingold, K.U.; Kayden, H.J. RRR- and SRR-alpha-tocopherols are secreted without discrimination in human chylomicrons, but RRR-alpha-tocopherol is preferentially secreted in very low density lipoproteins. J. Lipid Res. 1990, 31, 675–685. [Google Scholar] [PubMed]
- Traber, M.G.; Kayden, H.J. Preferential incorporation of alpha-tocopherol vs. gamma-tocopherol in human lipoproteins. Am. J. Clin. Nutr. 1989, 49, 517–526. [Google Scholar] [PubMed]
- Porsgaard, T.; Hoy, C.E. Absorption by rats of tocopherols present in edible vegetable oils. Lipids 2000, 35, 1073–1078. [Google Scholar] [CrossRef] [PubMed]
- Bardowell, S.A.; Ding, X.; Parker, R.S. Disruption of P450-mediated vitamin E hydroxylase activities alters vitamin E status in tocopherol supplemented mice and reveals extra-hepatic vitamin E metabolism. J. Lipid Res. 2012, 53, 2667–2676. [Google Scholar] [CrossRef] [PubMed]
- Riedl, J.; Linseisen, J.; Hoffmann, J.; Wolfram, G. Some dietary fibers reduce the absorption of carotenoids in women. J. Nutr. 1999, 129, 2170–2176. [Google Scholar] [PubMed]
- Greenwood, D.C.; Cade, J.E.; White, K.; Burley, V.J.; Schorah, C.J. The impact of high non-starch polysaccharide intake on serum micronutrient concentrations in a cohort of women. Public Health Nutr. 2004, 7, 543–548. [Google Scholar] [CrossRef] [PubMed]
- Roodenburg, A.J.; Leenen, R.; van het Hof, K.H.; Weststrate, J.A.; Tijburg, L.B. Amount of fat in the diet affects bioavailability of lutein esters but not of alpha-carotene, beta-carotene, and vitamin E in humans. Am. J. Clin. Nutr. 2000, 71, 1187–1193. [Google Scholar] [PubMed]
- Jeanes, Y.M.; Hall, W.L.; Ellard, S.; Lee, E.; Lodge, J.K. The absorption of vitamin E is influenced by the amount of fat in a meal and the food matrix. Br. J. Nutr. 2004, 92, 575–579. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.E.; Ferruzzi, M.G.; Campbell, W.W. Egg Consumption Increases Vitamin E Absorption from Co-Consumed Raw Mixed Vegetables in Healthy Young Men. J. Nutr. 2016, 146, 2199–2205. [Google Scholar] [CrossRef] [PubMed]
- Mah, E.; Sapper, T.N.; Chitchumroonchokchai, C.; Failla, M.L.; Schill, K.E.; Clinton, S.K.; Bobe, G.; Traber, M.G.; Bruno, R.S. alpha-Tocopherol bioavailability is lower in adults with metabolic syndrome regardless of dairy fat co-ingestion: A randomized, double-blind, crossover trial. Am. J. Clin. Nutr. 2015, 102, 1070–1080. [Google Scholar] [CrossRef] [PubMed]
- Preveraud, D.P.; Devillard, E.; Borel, P. Dietary fat modulates dl-alpha-tocopheryl acetate (vitamin E) bioavailability in adult cockerels. Br. Poult. Sci. 2015, 56, 94–102. [Google Scholar] [CrossRef] [PubMed]
- Failla, M.L.; Chitchumronchokchai, C.; Ferruzzi, M.G.; Goltz, S.R.; Campbell, W.W. Unsaturated fatty acids promote bioaccessibility and basolateral secretion of carotenoids and alpha-tocopherol by Caco-2 cells. Food Funct. 2014, 5, 1101–1112. [Google Scholar] [CrossRef] [PubMed]
- Koo, S.I.; Noh, S.K. Phosphatidylcholine inhibits and lysophosphatidylcholine enhances the lymphatic absorption of alpha-tocopherol in adult rats. J. Nutr. 2001, 131, 717–722. [Google Scholar] [PubMed]
- Nishimukai, M.; Hara, H. Enteral administration of soybean phosphatidylcholine enhances the lymphatic absorption of lycopene, but reduces that of alpha-tocopherol in rats. J. Nutr. 2004, 134, 1862–1866. [Google Scholar] [PubMed]
- Reboul, E.; Thap, S.; Perrot, E.; Amiot, M.J.; Lairon, D.; Borel, P. Effect of the main dietary antioxidants (carotenoids, gamma-tocopherol, polyphenols, and vitamin C) on alpha-tocopherol absorption. Eur. J. Clin. Nutr. 2007, 61, 1167–1173. [Google Scholar] [CrossRef] [PubMed]
- Sklan, D.; Donoghue, S. Vitamin E response to high dietary vitamin A in the chick. J. Nutr. 1982, 112, 759–765. [Google Scholar] [PubMed]
- Griffiths, D.W. The inhibition of digestive enzymes by polyphenolic compounds. Adv. Exp. Med. Biol. 1986, 199, 509–516. [Google Scholar] [PubMed]
- Fardet, A.; Morise, A.; Kalonji, E.; Margaritis, I.; Mariotti, F. Influence of phytosterol and phytostanol food supplementation on plasma liposoluble vitamins and provitamin A carotenoid levels in humans: An updated review of the evidence. Crit. Rev. Food Sci. Nutr. 2017, 57, 1906–1921. [Google Scholar] [CrossRef] [PubMed]
- Richelle, M.; Enslen, M.; Hager, C.; Groux, M.; Tavazzi, I.; Godin, J.P.; Berger, A.; Metairon, S.; Quaile, S.; Piguet-Welsch, C.; et al. Both free and esterified plant sterols reduce cholesterol absorption and the bioavailability of beta-carotene and alpha-tocopherol in normocholesterolemic humans. Am. J. Clin. Nutr. 2004, 80, 171–177. [Google Scholar] [PubMed]
- Borel, P.; Desmarchelier, C. Genetic Variations Involved in Vitamin E Status. Int. J. Mol. Sci. 2016, 17. [Google Scholar] [CrossRef] [PubMed]
© 2017 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Reboul, E. Vitamin E Bioavailability: Mechanisms of Intestinal Absorption in the Spotlight. Antioxidants 2017, 6, 95. https://doi.org/10.3390/antiox6040095
Reboul E. Vitamin E Bioavailability: Mechanisms of Intestinal Absorption in the Spotlight. Antioxidants. 2017; 6(4):95. https://doi.org/10.3390/antiox6040095
Chicago/Turabian StyleReboul, Emmanuelle. 2017. "Vitamin E Bioavailability: Mechanisms of Intestinal Absorption in the Spotlight" Antioxidants 6, no. 4: 95. https://doi.org/10.3390/antiox6040095
APA StyleReboul, E. (2017). Vitamin E Bioavailability: Mechanisms of Intestinal Absorption in the Spotlight. Antioxidants, 6(4), 95. https://doi.org/10.3390/antiox6040095





