Curcumin Stimulates the Antioxidant Mechanisms in Mouse Skin Exposed to Fractionated γ-Irradiation
Abstract
:1. Introduction
2. Materials and Methods
2.1. Preparation of Drug and Mode of Administration
2.2. Experimental Protocol
2.3. Irradiation
2.4. Preparation of Animals
2.5. Biochemical Parameters
2.5.1. Glutathione (GSH)
2.5.2. Glutathione Peroxidase (GSHPx)
2.5.3. Superoxide dismutase (SOD)
2.5.4. Lipid Peroxidation (LOO)
2.5.5. Estimation of Protein
2.6. Analysis of Data
3. Results
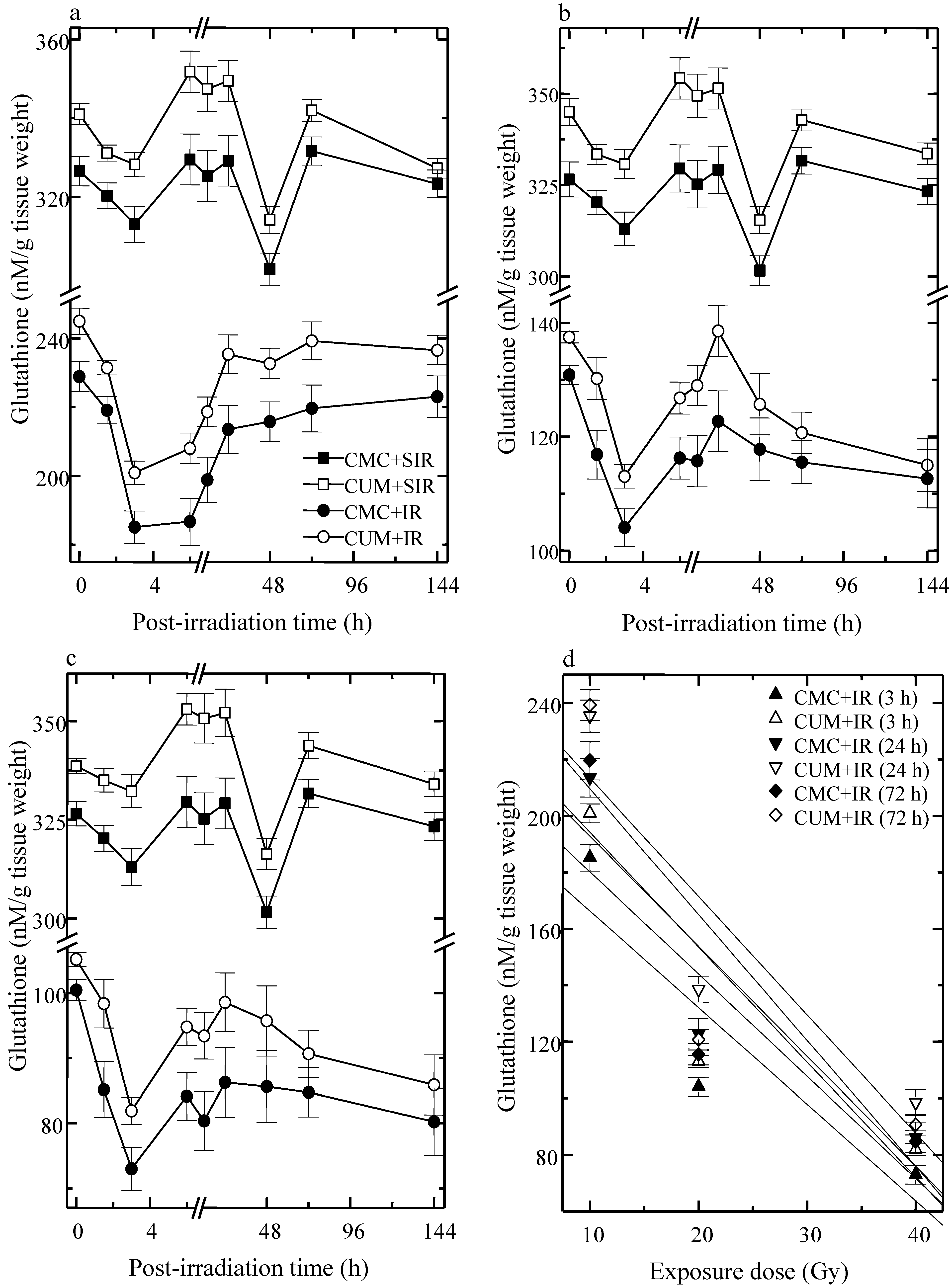
3.1. Glutathione (GSH)
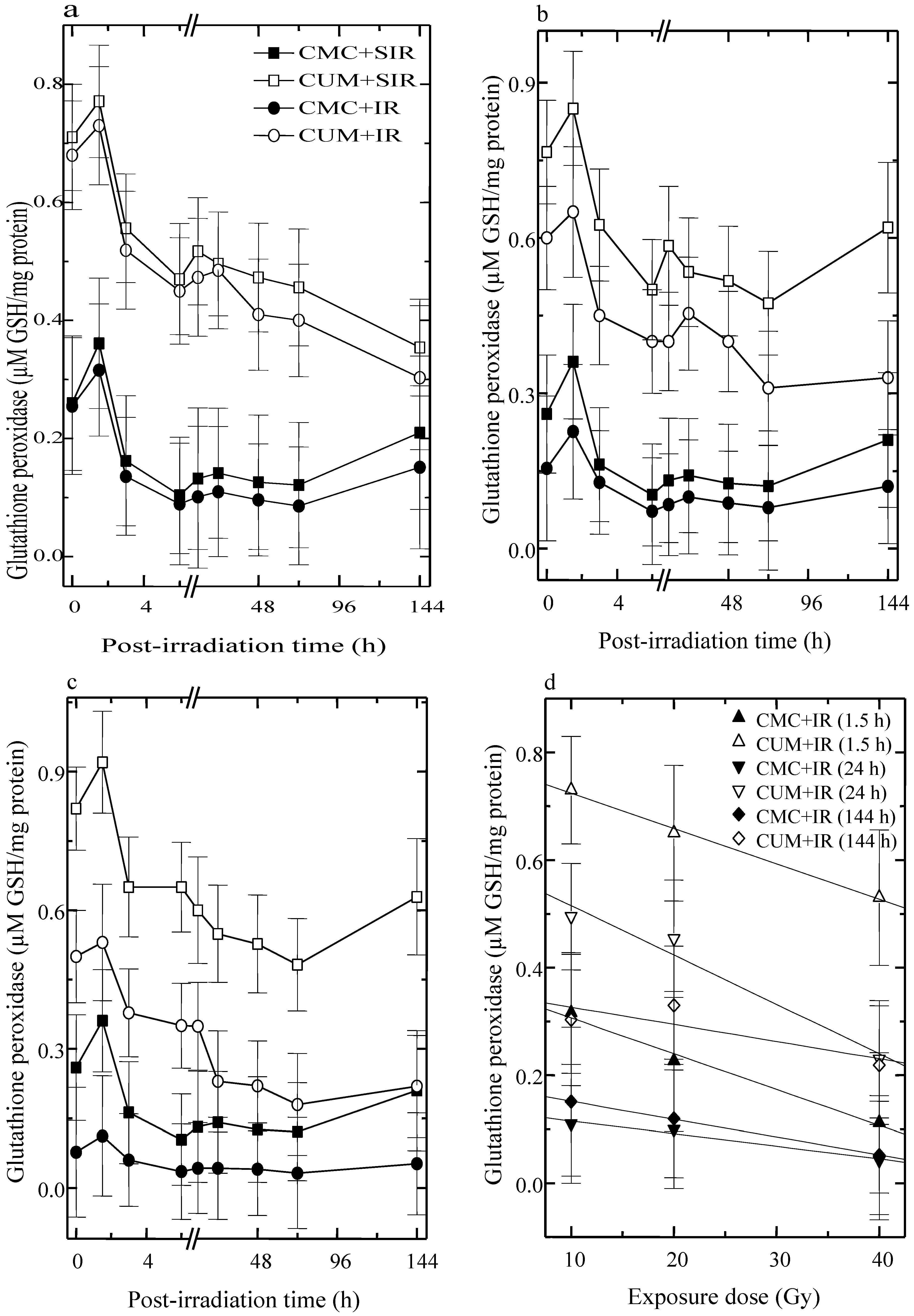
3.2. Glutathione Peroxidase (GSHPx)
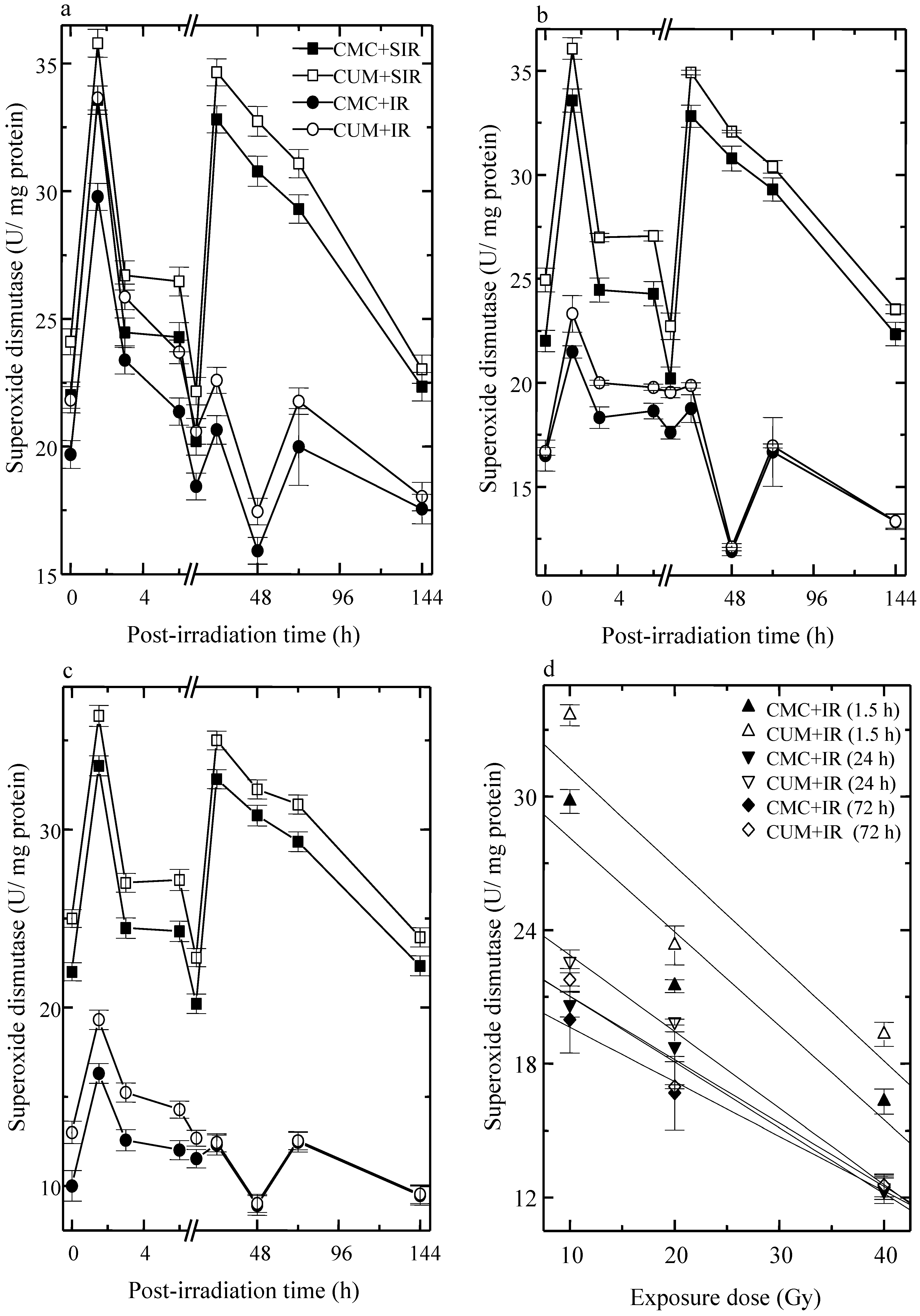
3.3. Superoxide Dismutase (SOD)
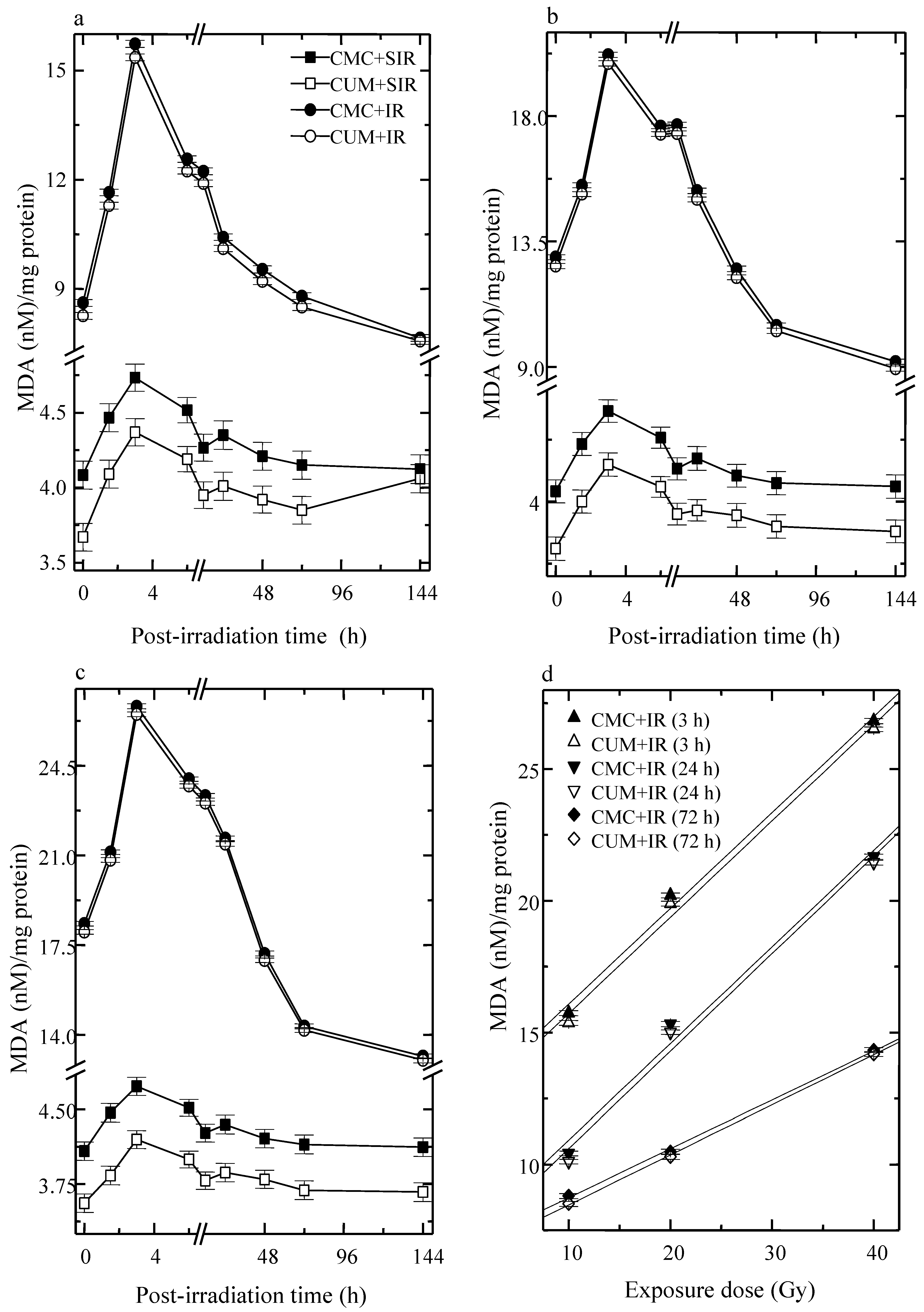
3.4. Lipid Peroxidation (LOO)
4. Discussion
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Zhou, Y.; Ni, H.; Balint, K.; Sanzari, J.K.; Dentchev, T.; Diffenderfer, E.S.; Wilson, J.M.; Cengel, K.A.; Weissman, D. Ionizing Radiation Selectively Reduces Skin Regulatory T Cells and Alters Immune Function. PLoS One 2014, 9, e100800. [Google Scholar] [CrossRef] [PubMed]
- Monson, R.R.; Cleaver, J.E.; Abrams, H.L.; Bingham, E.; Buffler, P.A.; Cardis, E.; Cox, R.; Davis, S.; Dewey, W.C.; Gilbert, E.S.; et al. Health Risks from Exposure to Low Levels of Ionizing Radiation: BEIR VII—Phase 2; National Academies Press: Washigton, DC, USA, 2006. [Google Scholar]
- Quintiliani, M. The oxygen effect in radiation interaction on DNA and enzymes. Int. J. Radiat. Biol. 1986, 50, 573–594. [Google Scholar] [CrossRef]
- Shindo, Y.; Witt, E.; Han, D.; Epstein, W.; Packer, L. Enzymic and non-enzymic antioxidants in epidermis and dermis of human skin. J. Investig. Dermatol. 1994, 102, 122–124. [Google Scholar] [CrossRef] [PubMed]
- Yu, B.P. Cellular defenses against damage from reactive oxygen species. Physiol. Rev. 1994, 74, 139–162. [Google Scholar] [PubMed]
- Fuchs, J. Potentials and limitations of the natural antioxidants RRR-α tocopherol, l-ascorbic acid, and β-carotene in cutaneous photoprotection. Free Radic. Biol. Med. 1998, 25, 848–873. [Google Scholar] [CrossRef] [PubMed]
- Ichihashi, M.; Ahmed, N.U.; Budiyanto, A.; Wu, A.; Bito, T.; Ueda, M.; Osawa, T. Preventive effect of antioxidant on ultraviolet-induced skin cancer in mice. J. Dermatol. Sci. 2000, 23, 45–50. [Google Scholar] [CrossRef]
- Darr, D.; Fridovich, I. Free radicals in cutaneous biology. J. Investig. Dermatol. 1994, 102, 671–675. [Google Scholar] [CrossRef] [PubMed]
- Brenner, D.J.; Doll, R.; Goodhead, D.T.; Hall, E.J.; Land, C.E.; Little, J.B.; Lubin, J.H.; Preston, D.L.; Preston, R.J.; Puskin, J.S.; et al. Cancer risks attributable to low doses of ionizing radiation: Assessing what we really know. Proc. Natl. Acad. Sci. USA 2003, 100, 13761–13766. [Google Scholar] [CrossRef] [PubMed]
- Gagliano, N.; Bedoni, M.; Mantovani, G.; Chiriva-Internati, M.; Castelli, D.; Torri, C.; Donetti, E. Effect of a topical treatment in organotypic culture of human breast skin after exposure to gamma-rays. Eur. J. Histochem. 2007, 51, 283–290. [Google Scholar] [PubMed]
- Chan, R.J.; Webster, J.; Chung, B.; Marquart, L.; Ahmed, M.; Garantziotis, S. Prevention and treatment of acute radiation-induced skin reactions: A systematic review and meta-analysis of randomized controlled trials. BMC Cancer 2014, 14, 53. [Google Scholar] [CrossRef] [PubMed]
- Hu, Z.P.; Kim, Y.M.; Sowa, M.B.; Robinson, R.J.; Gao, X.; Metz, T.O.; Morgan, W.F.; Zhang, Q. Metabolomic response of human skin tissue to low dose ionizing radiation. Mol. Biosyst. 2012, 8, 1979–1986. [Google Scholar] [CrossRef] [PubMed]
- Fuchs, J.; Hufljet, M.; Rothfuss, L.; Wilson, D.S.; Carcamo, G.; Packer, L. Impairment of enzymatic and non-enzymatic antioxidants in skin by UVB irradiation. J. Investig. Dermatol. 1989, 93, 769–773. [Google Scholar] [CrossRef] [PubMed]
- Meffert, H.; Diezel, W.; Sönnichsen, N. Stable lipid peroxidation products in human skin: Detection, ultraviolet induced increase, and pathogenic importance. Experientia 1976, 32, 1397–1398. [Google Scholar] [CrossRef] [PubMed]
- Buttke, T.M.; Sandstrom, P.A. Oxidative stress as a mediator of apoptosis. Immunol. Today 1994, 15, 7–10. [Google Scholar] [CrossRef] [PubMed]
- Sato, Y.; Kamazawa, N.; Suzuki, M.; Wang, C.M.; Ohte, S.; Shinoda, M. Studies on chemical protectors against radiation XXXIII. Protective mechanisms of various compounds against skin injury induced by radiation. Yakugaku Zasshi 1991, 111, 51–58. [Google Scholar] [PubMed]
- Katiyar, S.K.; Afaq, F.; Perez, A.; Mukhtar, H. Green Tea polyphenol (−)- epigallocatechin-3-galate treatment of human skin inhibits ultraviolet radiation-induced oxidative stress. Carcinogenesis 2001, 22, 287–294. [Google Scholar] [CrossRef] [PubMed]
- Jagetia, G.C.; Rajanikant, G.K.; Rao, S.K.; Baliga, S.M. Alteration in the glutathione, glutathione peroxidase, superoxide dismutase and lipid peroxidation by ascorbic acid in the skin of mice exposed to fractionated gamma radiation. Clin. Chim. Acta 2003, 332, 111–121. [Google Scholar] [CrossRef] [PubMed]
- Jagetia, G.C.; Rajanikant, G.K. Acceleration of wound repair by curcumin in the excision wound of mice exposed to different doses of fractionated γ radiation. Int. Wound J. 2012, 9, 76–92. [Google Scholar] [CrossRef] [PubMed]
- Leung, A. Encyclopedia of Common Natural Ingredients Used in Food, Drugs, and Cosmetics; John Wiley: New York, NY, USA, 1980; pp. 313–314. [Google Scholar]
- Jagetia, G.C.; Aggarwal, B.B. “Spicing Up” of the Immune System by Curcumin. J. Clin. Immunol. 2007, 27, 19–35. [Google Scholar] [CrossRef] [PubMed]
- Chang, H.; But, P.P. Pharmacology and Applications of Chinese Materia Medica, 2nd ed.; World Scientific Publishing Company: Singapore, 1987. [Google Scholar]
- Ammon, H.P.; Wahl, M.A. Pharmacology of Curcuma longa. Planta Med. 1991, 57, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Lin, J.K.; Pan, M.H.; Lin-Shiau, S.Y. Recent studies on the biofunctions and biotransformations of curcumin. Biofactors 2000, 13, 153–158. [Google Scholar] [CrossRef] [PubMed]
- Lao, C.D.; Ruffin, M.T.; Normolle, D.; Heath, D.D.; Murray, S.I.; Bailey, J.M.; Boggs, M.E.; Crowell, J.; Rock, C.L.; Brenner, D.E. Dose escalation of a curcuminoid formulation. BMC Complem. Altern. Med. 2006, 6, 10. [Google Scholar] [CrossRef]
- Susan, M.; Rao, M.N. Induction of glutathione-S-transferase activity by curcumin in mice. Arzneim Forsch. 1992, 42, 962–964. [Google Scholar]
- Ak, T.; Gulcin, I. Antioxidant and radical scavenging properties of curcumin. Chem. Biol. Interact 2008, 174, 27–37. [Google Scholar] [CrossRef] [PubMed]
- Ruby, A.J.; Kuttan, G.; Babu, K.D.; Rajasekharan, K.N.; Kuttan, R. Anti-tumour and antioxidant activity of natural curcuminoids. Cancer Lett. 1995, 94, 79–83. [Google Scholar] [CrossRef] [PubMed]
- Sreejayan, N.; Rao, M.N.A. Curcuminoids as potent inhibitors of lipid peroxidation. J. Pharm. Pharmacol. 1994, 46, 1013–1016. [Google Scholar] [CrossRef] [PubMed]
- Jelveh, S.; Kaspler, P.; Bhogal, N.; Mahmood, J.; Lindsay, P.E.; Okunieff, P.; Doctrow, S.R.; Bristow, R.G.; Hill, R.P. Investigations of antioxidant-mediated protection and mitigation of radiation-induced DNA damage and lipid peroxidation in murine skin. Int. J. Radiat. Biol. 2013, 89, 618–627. [Google Scholar] [CrossRef] [PubMed]
- Jagetia, G.C.; Rajanikant, G.K. Role of curcumin, a naturally occurring phenolic compound of turmeric in accelerating the repair of excision wound in mice whole-body exposed to various doses of γ-radiation. J. Surg. Res. 2004, 120, 127–138. [Google Scholar] [CrossRef] [PubMed]
- Jagetia, G.C.; Rajanikant, G.K. Effect of curcumin on radiation-impaired healing of excisional wounds in mice. J. Wound Care 2004, 13, 107–109. [Google Scholar] [CrossRef] [PubMed]
- Jagetia, G.C.; Rajanikant, G.K. Curcumin treatment enhances the repair and regeneration of wounds in mice exposed to hemibody γ-irradiation. Plast Reconstr. Surg. 2005, 115, 515–528. [Google Scholar] [CrossRef] [PubMed]
- López-Jornet, P.; Camacho-Alonso, F.; Jiménez-Torres, M.J.; Orduña-Domingo, A.; Gómez-García, F. Topical curcumin for the healing of carbon dioxide laser skin wounds in mice. Photomed. Laser Surg. 2011, 29, 809–814. [Google Scholar] [CrossRef] [PubMed]
- Moron, M.S.; Depierre, J.W.; Mannervik, B. Levels of glutathione, glutathione reductase and glutathione-s-transferase activities in rat lung and liver. Biochim. Biophys. Acta 1979, 582, 67–78. [Google Scholar] [CrossRef] [PubMed]
- Sazuka, Y.; Tanizawa, H.; Takino, Y. Effect of adriamycin on the activities of superoxide dismutase, glutathione peroxidase and catalase in tissues of mice. Jpn. J. Cancer Res. 1989, 80, 89–94. [Google Scholar] [CrossRef] [PubMed]
- Fried, R. Enzymatic and non-enzymatic assay of superoxide dismutase. Biochimie 1975, 57, 657–60. [Google Scholar] [CrossRef] [PubMed]
- Beuege, J.A.; Aust, S.D. Microsomal lipid peroxidation. Method Enzymol. 1978, 30, 302–310. [Google Scholar]
- Siems, W.; Gartner, C.; Kranz, D.; Schneider, W.; Grune, T.; Schimke, J.; Gau, S.; Wege, U.; Gerber, G. Long term effects of monthly low dose whole body irradiation on the glutathione status and thiobarbituric acid-reactive substance in different organs of male Wister rats. Radiobiol. Radiother. 1990, 31, 257–263. [Google Scholar]
- Kaya, H.; Delibas, N.; Serteser, M.; Ulukaya, E.; Ozkaya, O. The effect of melatonin on lipid peroxidation during radiotherapy in female rats. Strahlenther. Onkol. 1999, 175, 285–258. [Google Scholar] [CrossRef] [PubMed]
- Jagetia, G.C.; Reddy, T.K. Modulation of radiation-induced alteration in the antioxidant status of mice by naringin. Life Sci. 2005, 77, 780–794. [Google Scholar] [CrossRef] [PubMed]
- Jagetia, G.C.; Baliga, M.S.; Venkatesh, P.; Ulloor, J.N. Influence of ginger rhizome (Zingiber officinale Rosc) on survival, glutathione and lipid peroxidation in mice after whole-body exposure to γ-radiation. Radiat. Res. 2003, 160, 584–592. [Google Scholar] [CrossRef] [PubMed]
- Jagetia, G.C.; Venkatesh, P.; Baliga, M.S. Evaluation of the radioprotective effect of bael leaf (Aegle marmelos) extract in mice. Int. J. Radiat. Biol. 2004, 80, 281–290. [Google Scholar] [CrossRef] [PubMed]
- Jagetia, G.C.; Venkatesha, V.A. Effect of mangiferin on radiation-induced micronucleus formation in cultured human peripheral blood lymphocytes. Environ. Mol. Mutagen. 2005, 46, 12–21. [Google Scholar] [CrossRef] [PubMed]
- Tawfik, S.S.; Abouelella, A.M.; Shahein, Y.E. Curcumin protection activities against γ-rays-induced molecular and biochemical lesions. BMC Res. Notes 2013, 6, 375. [Google Scholar] [CrossRef] [PubMed]
- Davis, W., Jr.; Ronai, Z.; Tew, K.D. Cellular thiols and reactive oxygen species in drug-induced apoptosis. J. Pharmacol. Exp. Therapeut. 2001, 296, 1–6. [Google Scholar]
- Canales-Aguirre, A.A.; Gomez-Pinedo, U.A.; Luquin, S.; Ramırez-Herrera, M.A.; Mendoza-Magana, M.L.; Feria-Velasco, A. Curcumin protects against the oxidative damage induced by the pesticide parathion in the hippocampus of the rat brain. Nutr. Neurosci. 2012, 15, 62–69. [Google Scholar] [CrossRef] [PubMed]
- Xie, Y.; Zhao, Q.Y.; Li, H.Y.; Zhou, X.; Liu, Y.; Zhang, H. Curcumin ameliorates cognitive deficits heavy ion irradiation-induced learning and memory deficits through enhancing of Nrf2 antioxidant signaling pathways. Pharmacol. Biochem. Behav. 2014, 126, 181–186. [Google Scholar] [CrossRef] [PubMed]
- Meister, A.; Anderson, M.E. Glutathione. Annu. Rev. Biochem. 1983, 52, 711–760. [Google Scholar] [CrossRef] [PubMed]
- Aquilano, K.; Baldelli, S.; Ciriolo, M.R. Glutathione: New roles in redox signaling for an old antioxidant. Front. Pharmacol. 2014, 5, 196. [Google Scholar] [CrossRef] [PubMed]
- Navarro, J.; Obrador, E.; Pellicer, J.A.; Aseni, M.; Vina, J.; Estrela, M. Blood glutathione as an index of radiation-induced oxidative stress in mice and humans. Free Radic. Biol. Med. 1997, 22, 1203–1207. [Google Scholar] [CrossRef] [PubMed]
- Younes, M.; Seigers, C.P. Mechanistic aspects of enhanced lipid peroxidation following glutathione depletion in vivo. Chem. Biol. Interact. 1981, 34, 257–266. [Google Scholar] [CrossRef] [PubMed]
- Miyachi, Y.; Imamura, S.; Niwa, Y. Decreased skin superoxide dismutase activity by a single exposure of ultraviolet radiation is reduced by liposomal superoxide dismutase pretreatment. J. Investig. Dermatol. 1987, 89, 111–112. [Google Scholar] [CrossRef] [PubMed]
- Record, I.R.; Dreosti, I.E.; Konstantinopoulos, M.; Buckley, R.A. The influence of topical and systemic vitamin E on ultraviolet light-induced skin damage in hairless mice. Nutr. Cancer 1991, 16, 219–225. [Google Scholar] [CrossRef] [PubMed]
- Iizawa, O.; Kato, T.; Tagami, H.; Akamatsu, H.; Niwa, Y. Long term follow-up study of changes in lipid peroxide levels and the activity of superoxide dismutase, catalase, and glutathione peroxidase in mouse skin after acute and chronic UV irradiation. Arch. Dermatol. Res. 1994, 286, 47–52. [Google Scholar] [CrossRef] [PubMed]
- Dickinson, D.A.; Forman, H.J. Cellular glutathione and thiols metabolism. Biochem. Pharmacol. 2002, 64, 1019–1026. [Google Scholar] [CrossRef] [PubMed]
- Altavilla, D.; Saitta, A.; Guarini, S.; Galeano, M.; Squadrito, G.; Cucinotta, D. Oxidative stress causes nuclear factor-κB activation in acute hypovolemic hemorrhagic shock. Free Radic. Biol. Med. 2001, 30, 1055–1066. [Google Scholar] [CrossRef] [PubMed]
- Brigelius-Fhole, R. Tissue-specific functions of individual glutathione peroxidases. Free Radic. Biol. Med. 1999, 27, 951–965. [Google Scholar] [CrossRef] [PubMed]
- Kunchandy, E.; Rao, M.N.A. Oxygen radical scavenging activity of curcumin. Int. J. Pharm. 1990, 58, 237–240. [Google Scholar] [CrossRef]
- Inano, H.; Onoda, M. Radioprotective action of curcumin extracted from Curcuma longa Linn: Inhibitory effect on formation of urinary 8-hydroxy-2′-deoxyguanosine, tumorigenesis, but not mortality, induced by γ-ray irradiation. Int. J. Radiat. Oncol. Biol. Phys. 2002, 53, 735–743. [Google Scholar] [CrossRef] [PubMed]
- Iqbal, M.; Sharma, S.D.; Okazaki, Y.; Fujisawa, M.; Okada, S. Dietary supplementation of curcumin enhances antioxidant and phase II metabolizing enzymes in ddY male mice: Possible role in protection against chemical carcinogenesis and toxicity. Pharmacol. Toxicol. 2003, 92, 33–38. [Google Scholar] [CrossRef] [PubMed]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Jagetia, G.C.; Rajanikant, G.K. Curcumin Stimulates the Antioxidant Mechanisms in Mouse Skin Exposed to Fractionated γ-Irradiation. Antioxidants 2015, 4, 25-41. https://doi.org/10.3390/antiox4010025
Jagetia GC, Rajanikant GK. Curcumin Stimulates the Antioxidant Mechanisms in Mouse Skin Exposed to Fractionated γ-Irradiation. Antioxidants. 2015; 4(1):25-41. https://doi.org/10.3390/antiox4010025
Chicago/Turabian StyleJagetia, Ganesh Chandra, and Golgod Krishnamurthy Rajanikant. 2015. "Curcumin Stimulates the Antioxidant Mechanisms in Mouse Skin Exposed to Fractionated γ-Irradiation" Antioxidants 4, no. 1: 25-41. https://doi.org/10.3390/antiox4010025
APA StyleJagetia, G. C., & Rajanikant, G. K. (2015). Curcumin Stimulates the Antioxidant Mechanisms in Mouse Skin Exposed to Fractionated γ-Irradiation. Antioxidants, 4(1), 25-41. https://doi.org/10.3390/antiox4010025




